Introduction
Although antipsychotics can alleviate positive symptoms, many schizophrenia (SCZ) patients suffer from negative symptoms and functional impairments (Couture, Penn, & Roberts, Reference Couture, Penn and Roberts2006; Halverson et al., Reference Halverson, Orleans-Pobee, Merritt, Sheeran, Fett and Penn2019). SCZ patients also have markedly elevated risk of suicide, compared to the general populations (Bertuccio et al., Reference Bertuccio, Amerio, Grande, La Vecchia, Costanza, Aguglia and Odone2024). Empathy refers to the ability to understand and feel others’ emotional states (Preston & de Waal, Reference Preston and de Waal2002). Cognitive and affective empathy are impaired in patients with SCZ (Bonfils, Lysaker, Minor, & Salyers, Reference Bonfils, Lysaker, Minor and Salyers2016, Reference Bonfils, Lysaker, Minor and Salyers2017), undermining patients’ everyday social interactions (Green, Horan, & Lee, Reference Green, Horan and Lee2015; Vaskinn et al., Reference Vaskinn, Andersson, Østefjells, Andreassen and Sundet2018). Moreover, impaired empathy is associated with suicidal behavior and risk of bullying by others (Chu, Lu, & Huang, Reference Chu, Lu and Huang2025; Serafini et al., Reference Serafini, Aguglia, Amerio, Canepa, Adavastro, Conigliaro and Amore2023; van Noorden, Haselager, Cillessen, & Bukowski, Reference van Noorden, Haselager, Cillessen and Bukowski2015; Wang et al., Reference Wang, Zhou, Wang, Xu, Wei, Wang and Zhang2020a). Multisensory processing (mostly visual and auditory modalities) is the primary channel for social perception and empathic accuracy (Gesn & Ickes, Reference Gesn and Ickes1999; Hall & Schmid Mast, Reference Hall and Schmid Mast2007; Jospe et al., Reference Jospe, Genzer, Selle, Ong, Zaki and Perry2020; Kraus, Reference Kraus2017; Ong et al., Reference Ong, Jospe, Reddan, Wu, Kahhale, Chen and Perry2023). Emotion recognition is a core process of empathy (Derntl et al., Reference Derntl, Finkelmeyer, Toygar, Hülsmann, Schneider, Falkenberg and Habel2009) and is impaired in SCZ patients who are presented with audio-only and audiovisual information (Giannitelli et al., Reference Giannitelli, Xavier, François, Bodeau, Laurent, Cohen and Chaby2015; Thaler et al., Reference Thaler, Strauss, Sutton, Vertinski, Ringdahl, Snyder and Allen2013). Vogel et al. (Reference Vogel, Brück, Jacob, Eberle and Wildgruber2016) found SCZ patients having relatively preserved emotion recognition in audio-only condition, suggesting that the impaired emotion recognition might emerge primarily from altered processing in audiovisual condition. However, Simpson, Pinkham, Kelsven and Sasson (Reference Simpson, Pinkham, Kelsven and Sasson2013) found that SCZ patients showed poorer emotion recognition in audio-only and visual-only conditions, rather than audiovisual condition. Indeed, visual and auditory information exert differential effects on the recognition of positive versus negative emotions (Paulmann & Pell, Reference Paulmann and Pell2011; Zhang et al., Reference Zhang, Chen, Chen, Li, Chen, Long and Yuan2018). For instance, positive emotions tend to be more accurately recognized under visual (e.g. facial expressions) instead of auditory (e.g. vocal tones) conditions, whereas negative emotions are recognized comparably well in the two modalities (Zhang et al., Reference Zhang, Chen, Chen, Li, Chen, Long and Yuan2018).
SCZ patients have impaired cognitive empathy, regardless of the different types of tasks, including paradigms involving visual stimuli (i.e. Berger et al., Reference Berger, Bitsch, Jakobi, Nagels, Straube and Falkenberg2019; Kamp et al., Reference Kamp, Hartmann, Frommann, Lowe, Pintgen, Weide and Wölwer2025; Karpouzian-Rogers et al., Reference Karpouzian-Rogers, Cobia, Petersen, Wang, Mittal, Csernansky and Smith2021; Smith et al., Reference Smith, Horan, Cobia, Karpouzian, Fox, Reilly and Breiter2014), auditory stimuli (i.e. Atoui et al., Reference Atoui, El Jamil, El Khoury, Doumit, Syriani, Khani and Nahas2018), and audiovisual stimuli (i.e. Baez et al., Reference Baez, Herrera, Villarin, Theil, Gonzalez-Gadea, Gomez and Ibañez2013; de Jong et al., Reference de Jong, van Donkersgoed, Renard, Carter, Bokern, Lysaker and Pijnenborg2018; Harenski et al., Reference Harenski, Brook, Kosson, Bustillo, Harenski, Caldwell and Kiehl2017; van Donkersgoed et al., Reference van Donkersgoed, de Jong, Het Rot, Wunderink, Lysaker, Hasson-Ohayon and Pijnenborg2019). Using the Empathic Accuracy Task (EAT), some studies found that intensity of emotional expression in the stimuli could differentially affect empathy in SCZ patients, that is, they performed well in low-expressive stimuli, but poorly in high-emotional expressions (Harvey et al., Reference Harvey, Zaki, Lee, Ochsner and Green2013; Lee et al., Reference Lee, Zaki, Harvey, Ochsner and Green2011). Studies using visual information tasks showed that SCZ patients had poorer affective empathy than controls (i.e. Benedetti et al., Reference Benedetti, Bernasconi, Bosia, Cavallaro, Dallaspezia, Falini and Scotti2009; Derntl et al., Reference Derntl, Finkelmeyer, Toygar, Hülsmann, Schneider, Falkenberg and Habel2009; Lee et al., Reference Lee, Kang, Kim, Gu, Park, Choi and Kwon2010, Smith et al., Reference Smith, Horan, Cobia, Karpouzian, Fox, Reilly and Breiter2014); but the two groups showed similar affective empathy in tasks using silent or short audiovisual video clips (Knobloch et al., Reference Knobloch, Leiding, Wagels, Regenbogen, Kellermann, Mathiak and Habel2024; Ramos-Loyo, Mora-Reynoso, Sánchez-Loyo, & Medina-Hernández, Reference Ramos-Loyo, Mora-Reynoso, Sánchez-Loyo and Medina-Hernández2012).
Schizotypy is a latent personality organization reflecting vulnerability to schizophrenia (Meehl, Reference Meehl1962). Physical and social anhedonia are negative dimension of schizotypy (Wang, Lui, & Chan, Reference Wang, Lui and Chan2024). In particular, high social anhedonia (HSoA) constitutes a high-risk condition for developing SCZ (Kwapil, Reference Kwapil1998) and is associated with attenuated empathy deficits relative to SCZ (Dominguez et al., Reference Dominguez, Wichers, Lieb, Wittchen and van Os2011). Previous studies found a significant negative correlation between social anhedonia and empathy (Wang et al., Reference Wang, Shi, Liu, Zheng, Wong, Cheung and Chan2020b). HSoA individuals exhibited poorer empathy than people with low social anhedonia (Gu et al., Reference Gu, Ding, Yang, Meng, Hu, Li and Chan2025; Guo et al., Reference Guo, Zheng, Ruan, Wang, Wang and Chan2023; Pflum & Gooding, Reference Pflum and Gooding2018). Research using samples with HSoA can circumvent the confounding variables (e.g. antipsychotics) inherent in clinical patients, and provide early detection frameworks (Barrantes-Vidal, Grant, & Kwapil, Reference Barrantes-Vidal, Grant and Kwapil2015).
Several unresolved issues concerning empathy in SCZ spectrum disorders are notable. First, previous studies emphasized the influence of audiovisual information on empathy, but the traditional video materials were mostly real-human videos, lacking standardization and manipulation of visual information. Avatar technology (computer-generated agents) could replicate human morphology and kinematics (Boulic, Bécheiraz, Emering, & Thalmann, Reference Boulic, Bécheiraz, Emering, Thalmann, Thalmann, Feiner and Singh1997; Sung, Han, Bae, & Kwon, Reference Sung, Han, Bae and Kwon2022). These programmable virtual humans enable precise control over facial muscle dynamics and emotional expression gradients, better than real-human video materials. However, few empirical studies have investigated the difference between Avatar and real-human videos in influencing empathy. Moreover, very few studies have explored whether empathy deficits in SCZ would be related to the mode/channels (visual and auditory) for presenting materials. Furthermore, previous research on empathy in HSoA samples was limited to unimodal stimulation materials, and seldom used multimodal audiovisual materials. Finally, no study has explored the differential impacts of visual and auditory information on the empathy performance of SCZ patients and individuals with HSoA.
This study aimed to examine the impacts of visual modalities on empathy using the modified Chinese version of the EAT (Wang et al., Reference Wang, Zhang, Fu, Wang, Jiang, Cao and Raymond2025) in SCZ patients and people with HSoA. We hypothesized that SCZ patients would have significantly reduced empathic accuracy, cognitive and affective empathy relative to controls, across different conditions. We also hypothesized that empathy deficits would be inversely correlated with clinical symptoms (in particular negative symptoms) in SCZ patients. Finally, we hypothesized that people with HSoA would have attenuated deficit of empathy relative to SCZ patients, involving affective empathy.
Methods
Participants
This study utilized two independent samples. The SCZ–Control sample comprised 50 SCZ patients recruited from the psychiatric department of Weifang Mental Health Center, and 50 healthy controls from the neighboring community. The inclusion criteria for clinical patients were: (1) DSM-5 (American Psychiatric Association, 2013) diagnosis of SCZ, (2) aged 18–60, (3) IQ ≥70, (4) normal (or corrected-to-normal) hearing and visual acuity, and (5) clinical stabilization. The exclusion criteria for clinical patients included (1) history of brain injury, major physical illness, or organic brain lesions, (2) lifetime history of psychoactive substance dependence or abuse, (3) neurological organic diseases, and (4) history of transcranial magnetic stimulation therapy or electroconvulsive therapy in the past 14 days. The inclusion criteria for controls included (1) absence of personal and family history of psychiatric disorder as confirmed by the MINI International Neuropsychiatric Interview (M.I.N.I.) (Lecrubier et al., Reference Lecrubier, Sheehan, Weiller, Amorim, Bonora, Sheehan and Dunbar1997), (2) normal (or corrected) hearing and visual acuity, and (3) IQ ≥70. The exclusion criteria for controls included (1) history of traumatic brain injury, (2) organic brain lesions, (3) psychoactive substance abuse, and (4) neurological diseases.
The HSoA–LSoA sample comprised 59 participants with HSoA and 60 participants with low social anhedonia (LSoA). The inclusion criteria for HSoA included (1) CSAS ≥17 (Zhang et al., Reference Zhang, Yang, Wang, Wang, Yang, Cheung and Chan2020), (2) aged 18–25, and (3) normal (or corrected-to-normal) hearing and visual acuity. The inclusion criteria for LSoA included (1) CSAS <11 (Zhang et al., Reference Zhang, Yang, Wang, Wang, Yang, Cheung and Chan2020); (2) aged 18–25, (3) absence of traumatic brain injury and neurological diseases, and (4) normal (or corrected-to-normal) hearing and visual acuity. The exclusion criteria for both HSoA and LSoA included (1) M.I.N.I.-identified psychiatric illness, and (2) lifetime history of drug dependence, substance dependence, and alcohol dependence.
This study was approved by the Ethics Committee of Shandong Second Medical University (2023YX080). All participants gave written informed consent.
Measures
The modified Chinese version of the EAT (Wang et al., Reference Wang, Zhang, Fu, Wang, Jiang, Cao and Raymond2025) was used in the current study, which comprised three Modality Conditions: audio-only, audiovisual, and audioavatar visual condition. Six videos were randomly selected from a standardized video library (Hu et al., Reference Hu, Guo, Zheng, Yan, Lui, Wang and Chan2024) and assigned to each condition (one positive and one negative clips). The audiovisual condition presented the raw, unprocessed footage; the audio-only condition extracted the audio from the original video; the audioavatar visual condition were created using Reallusion® Character Creator©, Reallusion® iClone®, and Adobe® Premiere Pro® software (Wang et al., Reference Wang, Zhang, Fu, Wang, Jiang, Cao and Raymond2025). As in the original version of EAT, participants were asked to watch each video of emotional stories, and continuously rated the narrator’s emotions using a 9-point scale (1 = very negative, 9 = very positive). The ratings of Perspective Taking, the Target’s Emotional Valence and Arousal reflected the participants’ cognitive empathy; while the Emotional Contagion, self-emotional valence and arousal reflected the participants’ affective empathy. The ratings of empathic concern, help, and effort level were filled in only after watching the negative videos, and were used to measure the participants’ empathic motivation. Spearman’s correlation coefficient between the participant and the narrator’s ratings was calculated and Fisher’s z transformed to measure the Empathic Accuracy (EA).
In addition, the Positive and Negative Syndrome Scale (PANSS) (Kay, Fiszbein, & Opler, Reference Kay, Fiszbein and Opler1987; Si et al., Reference Si, Yang, Shu-Liang, Wang, Kong, Zhou-Mo and Liu-cui2004) and the Clinical Assessment Interview for Negative Symptoms (CAINS) (Chan et al., Reference Chan, Shi, Lui, Ho, Hung, Lam and Yu2015; Kring et al., Reference Kring, Gur, Blanchard, Horan and Reise2013) were administered to assess the clinical symptoms in SCZ patients. The Chinese version of the Wechsler Adult Intelligence Scale–Revised (WAIS-R) (Gong & Dai, Reference Gong and Dai1984) was used to assess estimated IQ through four subtests, that is, General Knowledge, Arithmetic, Similarities and Digit Span. The Chapman Social Anhedonia Scale (CSAS) and the Chapman Physical Anhedonia Scale (CPAS) (Chapman, Chapman, & Raulin, Reference Chapman, Chapman and Raulin1976) had been found to have excellent psychometric properties (with Cronbach’s alpha coefficients of 0.84 and 0.86, respectively) and were used to measure the participants’ levels of anhedonia. Likewise, the Cognitive and Affective Empathy Scale (QCAE) (Liang et al., Reference Liang, Yang, Ma, Lui, Cheung, Wang and Chan2019; Reniers et al., Reference Reniers, Corcoran, Drake, Shryane and Völlm2011) demonstrated a Cronbach’s alpha coefficient of 0.88, and was used to assess the participants’ trait cognitive and affective empathy. Finally, the First Episode Social Functioning Scale (FESFS) (Lecomte et al., Reference Lecomte, Corbière, Ehmann, Addington, Abdel-Baki and Macewan2014; Wang et al., Reference Wang, Yeh, Tsang, Liu, Shi, Li and Lui2013) demonstrated a Cronbach’s alpha coefficient of 0.94, and was used to evaluate the participants’social functioning.
Data analysis
We conducted independent samples t tests to compare the differences in demographic variables and scores on the self-report scales between the groups. The 3 (Modality: audio-only, audiovisual, audioavatar visual condition) × 2 (Group: SCZ vs. HC; or HSoA vs. LSoA) mixed models of analysis of variance (ANOVA) were performed to examine the Modality, and Group main effects, and the Modality-by-Group interaction on the EAT performance, during the positive and negative video conditions separately for each sample. Bonferroni corrections were applied for post hoc pairwise comparisons. Pearson’s correlation analysis was used to explore the relationship between clinical symptoms and empathy deficits in the patient group. False discovery rate (FDR)-adjusted p values were reported for correlation analyses. Data analyses were performed using the SPSS, with a significance level set at p or adjusted p < 0.05.
Results
SCZ–control sample
Table 1 shows the demographics, self-report scales, and clinical symptoms. The two groups did not differ in gender and age. However, SCZ patients had lower years of education and estimated IQ, higher levels of physical and social anhedonia, and lower scores of cognitive empathy than healthy controls.
Demographics and clinical characteristics of schizophrenia–control sample

Note: AE, affective empathy; CAINS, clinical assessment interview for negative symptoms; CE, cognitive empathy; CSAS, Chapman Social Anhedonia Scale; CPAS, Chapman Physical Anhedonia Scale; EXP, expression; MAP, motivation and pleasure; QCAE, Cognitive and Affective Empathy Scale; PANSS, Positive and Negative Syndrome Scale; SFS, First Episode Social Functioning Scale.
EAT performance in positive-valenced videos
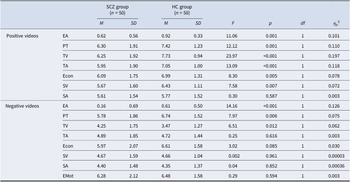
As shown in Table 2 and Figure 1A, significant Group main effect was found, suggesting that SCZ patients scored significantly lower than controls across different cognitive empathy variables (i.e. Empathic Accuracy, Perspective Taking, Target’s Emotional Valence, and Target’s Emotional Arousal) and affective empathy variables (Emotional Contagion and Self-Emotional Valence). Moreover, we found a significant Modality main effect on the Target’s Emotional Valence ratings of cognitive empathy, F(2, 196) = 3.43, p = 0.034, η p 2 = 0.034. Specifically, ratings in the audiovisual condition (M = 7.21, SD = 2.01) were significantly higher than those in the audioavatar visual condition (M = 6.72, SD = 2.10, Bonferroni-adjusted p = 0.032, Cohen’s d = − 0.26). We also found a significant Modality-by-Group interaction effect for the Empathic Accuracy, F(2, 196) = 5.61, p = 0.004, η p 2 = 0.054 (see Supplementary Table S1). As shown in Figure 1B, SCZ patients showed significantly lower empathic accuracy than controls under both audiovisual (Bonferroni-adjusted p = 0.039, Cohen’s d = − 0.42) and audioavatar visual conditions (Bonferroni-adjusted p < 0.001, Cohen’s d = −0.93), whereas no significant group difference was found under audio-only condition. Within-group comparisons further indicated that, among SCZ patients, the Empathic Accuracy in audio-only condition was significantly higher than audioavatar visual condition (Bonferroni-adjusted p = 0.017), but no significant differences could be found among three conditions in the control participants.
Group comparisons between schizophrenia patients and healthy controls on the EAT performance

Note: EA, empathic accuracy; Econ, emotional contagion; EMot, empathic motivation; HC, healthy control; PT, perspective taking; SA, self-emotional arousal; SCZ, schizophrenia; TV, target’s emotional valence; TA, target’s emotional arousal; SV, self-emotional valence.
Performance on the empathic accuracy task. Note: Panel A illustrates the main effects of group (SCZ vs. Control); Panel B illustrates the Modality-by-Group interaction effect. Panel C illustrates the main effects of group (HSoA vs. LSoA). EA, empathic accuracy; Econ, emotional contagion; EMot, empathic motivation; HSoA, high social anhedonia; LSoA, low social anhedonia; PT, perspective taking; SA, self-emotional arousal; SCZ, schizophrenia; SV, self-emotional valence; TA, target’s emotional arousal; TV, target’s emotional valence.

EAT performance in negative-valenced videos
We found significant main effects of Group on the Empathic Accuracy, Perspective Taking, and Target’s Emotional Valence of cognitive empathy, with poorer performance in SCZ patients relative to healthy controls (see Table 2). Moreover, we found a significant main effect of Modality on the Emotional Contagion ratings of affective empathy, F(2, 196) = 8.54, p < 0.001, η p 2 = 0.080. Post hoc comparisons revealed that Emotional Contagion ratings in audio-only (M = 6.66, SD = 2.09, Bonferroni-adjusted p < 0.001, Cohen’s d = 0.40) and audiovisual (M = 6.35, SD = 2.21, Bonferroni-adjusted p = 0.056, Cohen’s d = 0.24) conditions were significantly higher than in audioavatar visual condition (M = 5.87, SD = 2.19). We found a significant Modality-by-Group interaction effect for the Empathic Accuracy, F(2, 196) = 3.08, p = 0.048, η p 2 = 0.031. As shown in Figure 1B, SCZ patients had lower EA scores than healthy controls in audio-only (Bonferroni-adjusted p = 0.002, Cohen’s d = −0.63) and audiovisual conditions (Bonferroni-adjusted p < 0.001, Cohen’s d = −0.83), but not in audioavatar visual condition (see Supplementary Table S1). No significant difference was found among three conditions within each group.
HSoA–LSoA sample
Table 3 shows the demographics and self-reported measures of participants with HSoA and LSoA. The two groups were matched in demographics and estimated IQ. As expected, HSoA participants had higher physical and social anhedonia, but lower empathy and social functioning than LSoA participants.
The characteristics of the HSoA–LSoA sample

Note: AE, affective empathy; CE, cognitive empathy; CPAS, Chapman Physical Anhedonia Scale; CSAS, Chapman Social Anhedonia Scale; HSoA, high social anhedonia; LSoA, low social anhedonia; QCAE, Cognitive and Affective Empathy Scale; SFS, First Episode Social Functioning Scale.
EAT performance in positive-valenced videos
We found significant main effects of Group for Empathic Accuracy and Target’s Emotional Valence within the domain of cognitive empathy, with the HSoA participants scoring significantly lower than the LSoA participants. We did not find any significant group difference in the remaining indices (see Table 4 and Figure 1C). The main effect of Modality on the Target’s Emotional Valence was significant, F(1.89, 221.02) = 4.78, p = 0.011, η p 2 = 0.039. Post hoc comparison showed that the audiovisual condition (M = 7.79, SD = 1.04) was significantly higher than audio-only (M = 7.39, SD = 1.16; Bonferroni-adjusted p = 0.009, Cohen’s d = −0.28) and audioavatar visual (M = 7.45, SD = 1.27; Bonferroni-adjusted p = 0.027, Cohen’s d = −0.24) conditions. The significant main effect of Modality for the Target’s Emotional Arousal was observed, F(2, 234) = 3.84, p = 0.023, η p 2 = 0.032. Audiovisual condition (M = 7.08, SD = 1.34) yielded significantly higher ratings than audio-only condition (M = 6.62, SD = 1.67; Bonferroni-adjusted p = 0.027, Cohen’s d = −0.25). Regarding affective empathy, we found significant main effect of Modality on the Self-emotional Valence, F(2, 234) = 8.94, p <0.001, η p 2 = 0.071. The audiovisual condition (M = 6.83, SD = 1.17) was significantly higher than audio-only (M = 6.36, SD = 1.41; Bonferroni-adjusted p = 0.002, Cohen’s d = 0.32) and audioavatar visual (M = 6.34, SD = 1.32; Bonferroni-adjusted p <0.001, Cohen’s d = −0.37) conditions. Similarly, the main effect of Modality for the Self-emotional Arousal was significant, F(2, 234) = 8.56, p <0.001, η p 2 = 0.068. Participants rated audiovisual condition (M = 6.08, SD = 1.76) higher than audio-only condition (M = 5.41, SD = 2.00; Bonferroni-adjusted p = 0.001, Cohen’s d = 0.35). However, we did not find any significant Modality-by-Group interaction in the HSoA–LSoA sample (see Supplementary Table S2).
Group comparisons between participants with high and low social anhedonia on the EAT performance

Note: EA, empathic accuracy; Econ, emotional contagion; EMot, empathic motivation; HSoA, high social anhedonia; LSoA, low social anhedonia; PT, perspective taking; SA, self-emotional arousal; SV, self-emotional valence; TV, target’s emotional valence; TA, target’s emotional arousal.
EAT performance in negative-valenced videos
We found significant main effects of Group in the ratings of Perspective Taking of cognitive empathy, Self-emotional Arousal of affective empathy, and Empathic Motivation, with HSoA participants scoring lower than LSoA participants (see Table 4). HSoA and LSoA participants did not differ in the remaining indices. However, the main effects of Modality and Modality-by-Group interaction effects were nonsignificant.
Association between empathy deficit and clinical symptoms in SCZ patients
In positive-valenced videos, Perspective Taking was negatively correlated with the PANSS negative factor (FDR-adjusted p = 0.032), the CSAS (FDR-adjusted p = 0.006) and the CPAS scores (FDR-adjusted p = 0.003). Emotional Contagion was also negatively correlated with the CSAS (FDR-adjusted p = 0.008) and CPAS (FDR-adjusted p = 0.012). Self-emotional Valence showed significant negative correlations with PANSS general symptoms (FDR-adjusted p = 0.045), the CSAS (FDR-adjusted p = 0.012) and CPAS scores (FDR-adjusted p = 0.008). Target’s Emotional Arousal was negatively correlated with the CSAS (FDR-adjusted p = 0.045), and CPAS scores (FDR-adjusted p = 0.012). Target’s Emotional Valence negatively correlated with the CPAS scores (FDR-adjusted p = 0.012). In negative-valenced videos, Perspective Taking was negatively correlated with the CAINS total score (FDR-adjusted p = 0.045) and the CSAS (FDR-adjusted p = 0.012), and CPAS scores (FDR-adjusted p = 0.012) (see Figure 2 and Supplementary Table S3). No significant correlations were found between dosage of antipsychotic medication and EAT performance.
Association between empathy deficit and clinical symptoms in patients with schizophrenia. Note: CAINS, clinical assessment interview for negative symptoms; CPAS, Chapman Physical Anhedonia Scale; CSAS, Chapman Social Anhedonia Scale; EA, empathic accuracy; Econ, emotional contagion; EXP, expression; MAP, motivation and pleasure; PANSS, Positive and Negative Syndrome Scale; PT, perspective taking; QAE, affective empathy; QCAE, Cognitive and Affective Empathy Scale; QCE, cognitive empathy; SV, self-emotional valence; TA, target’s emotional arousal; TV, target’s emotional valence. *: FDR-adjusted p < 0.05.

Discussion
Regardless of the emotional valence, we found significant Group main effect across the samples. Both SCZ patients and HSoA individuals showed lower cognitive empathy (especially Empathic Accuracy, Perspective Taking, Target’s Emotional Valence) relative to the respective comparison groups, which is consistent with previous research (Guo et al., Reference Guo, Zheng, Ruan, Wang, Wang and Chan2023; Kamp et al., Reference Kamp, Hartmann, Frommann, Lowe, Pintgen, Weide and Wölwer2025; van Donkersgoed et al., Reference van Donkersgoed, de Jong, Het Rot, Wunderink, Lysaker, Hasson-Ohayon and Pijnenborg2019). Specifically, SCZ patients showed marked difficulties in understanding and inferring others’ mental states, regardless of positive or negative emotional valences. Such deficits appeared to be pervasive and trait-like features of SCZ patients, closely associated with social functioning (Green, Horan, & Lee, Reference Green, Horan and Lee2015). Similarly, HSoA individuals also exhibited cognitive empathy impairments, which may serve as an early indicator of SCZ spectrum disorders. Regarding affective empathy, SCZ patients exhibited deficits only in positive-valenced context (such as Emotional Contagion and Self-emotional Valence), while people with HSoA retained relatively intact affective empathy. Under the negative-valenced context, both SCZ patients and people with HSoA showed relatively intact affective empathy. This may indicate that SCZ patients have difficulty in processing of positive emotions, though they are able to experience and perceive others’ emotions in negative emotional stories. Such difficulty may stem from anhedonia and the weaker neural responses to social rewards (such as smiles and praise) (Guo et al., Reference Guo, Zheng, Ruan, Wang, Wang and Chan2023; Lee et al., Reference Lee, Jimenez, Reavis, Horan, Wynn and Green2019), which adversely affect patients’ mental simulation abilities of others’ positive emotional states. In other words, SCZ patients may be unable to accurately map others’ happiness based on their own experiences, leading to a failure in affective empathy. Notably, our findings offer a new explanatory framework for previous inconsistent findings. The use of mixed emotional stimuli without distinguishing positive from negative emotional valence in previous research might have confounded prior results. After better delineating positive and negative emotion valences, affective empathy in SCZ patients was only found to be impaired in positive-valenced context. The Research Domain Criteria (RDoC) of the National Institute of Mental Health (NIMH), posits that dysfunction in the Positive Valence Systems is a core feature of SCZ (Cuthbert & Kozak, Reference Cuthbert and Kozak2013; Insel et al., Reference Insel, Cuthbert, Garvey, Heinssen, Pine, Quinn and Wang2010). Our findings of SCZ patients having difficulty in processing rewards and positive stimuli concurred with the NIMH RDoC framework. Moreover, this study offers a new clinical direction for social cognitive interventions. The differential impairments in affective empathy implicate that targeted interventions for positive affective empathy can be more effective. For instance, positive emotion facial recognition training, and social reward scenario simulations can be applied in future research. Moreover, virtual reality technology can be used in the future to create positive social scenarios, enhancing patients’ experiences of others’ positive emotions, thereby improving their empathy abilities. In addition, HSoA individuals showed lower empathic motivation than people without social anhedonia, while no significant difference was observed between SCZ patients and controls. This may be due to the fact that we applied the extreme-group design to identify the HSoA and LSoA groups based on extremely-high scores on the CSAS, such as the CSAS ratings in HSoA participants were higher than SCZ participants (SCZ: HSoA = 13.7: 21.02) in this study. The HSoA group therefore expected less pleasure in social situations, resulting in social withdrawal (Mishlove & Chapman, Reference Mishlove and Chapman1985; Zhang et al., Reference Zhang, Yang, Wang, Wang, Yang, Cheung and Chan2020).
Additionally, we found differential impacts of stimuli modalities by examining the Modality main effect and Group-by-Modality interaction. In the SCZ–Control sample, our findings indicated significant Modality main effect on both cognitive empathy (indexed by Target’s Emotional Valence) and affective empathy (indexed by Emotional Contagion). Specifically, participants exhibited poorer performance under audioavatar visual condition than audio-only and/or audiovisual conditions. Inaccurate facial expression information of Avatars may interfere with emotional recognition and empathic processing. In addition, we found significant Modality-by-Group interaction on Empathic Accuracy for both positive and negative valenced EAT. Under the positive-valenced context, visual information affected SCZ patients’ empathic processing, while under the negative-valenced context, SCZ patients had poorer empathic accuracy for audiovisual condition, but performed similarly to controls in interacting with the Avatar audiovideo condition. In the HSoA–LSoA sample, we found significant Modality main effect on both cognitive and affective empathy, with audiovisual condition outperformed than the other two conditions. However, the Modality types on empathy performance exhibited similar impacts to both groups with different levels of social anhedonia, which differed from the findings gathered in the SCZ–Control sample. In positive emotional events, individuals may exhibit more intense visual cues (e.g. clear facial expressions, dynamic gestures) (Zaki, Bolger, & Ochsner, Reference Zaki, Bolger and Ochsner2009), while SCZ patients are impaired in the early stage of visual perception (Adámek, Langová, & Horáček, Reference Adámek, Langová and Horáček2022; Matsumoto, Takahashi, Murai, & Takahashi, Reference Matsumoto, Takahashi, Murai and Takahashi2015), and there are significant deficits in audiovisual integration (Hirano, Nakamura, & Tamura, Reference Hirano, Nakamura and Tamura2024; Lin, Ding, & Zhang, Reference Lin, Ding and Zhang2020). Furthermore, in these contexts, the visual information in audiovideo stimuli may increase the cognitive load for SCZ patients, acting as a distraction that interferes one’s ability to process auditory information. Consequently, both visual and auditory information are inadequately processed, impairing empathy accuracy in SCZ patients. However, differing from their counterparts with positive schizotypy, the visual perception of individuals with negative schizotypy such as social anhedonia may be preserved. This might be the reason why we did not find any significant interaction in HSoA–LSoA sample. Future research should include individuals with positive schizotypy.
The degree of expressiveness of video characters may influence empathy accuracy. For videos with less expressive targets, no significant difference in empathy accuracy was observed between SCZ patients and controls, while for more expressive targets, SCZ patients’ empathy accuracy was significantly lower (van Donkersgoed et al., Reference van Donkersgoed, de Jong, Het Rot, Wunderink, Lysaker, Hasson-Ohayon and Pijnenborg2019). Our findings confirmed these previous results in the context of negative emotional videos. Real-life emotion expressions can be sophisticated and complex, but Avatar technology can maximize characteristic features of emotion expression, thus reducing the complexity of emotional recognition. In negative emotional contexts, Avatar format may be simpler and easier for SCZ patients, and tax less on patients’ limited cognitive resources, thus improving empathy performance. Furthermore, interacting with Avatar virtual humans in a friendly environment may reduce social pressure for SCZ patients. Previous research has shown that children with autism perform better when interacting with virtual humans than with real humans (Pino, Vagnetti, Valenti, & Mazza, Reference Pino, Vagnetti, Valenti and Mazza2021). The nonjudgmental nature of virtual humans may also facilitate SCZ patients to focus more on perceiving the core emotional content, rather than inferring on others’ intention. Recent studies have combined virtual reality (VR) technology with social cognitive interaction training, and found this approach effective in improving SCZ patients’ social cognition (Cella et al., Reference Cella, Tomlin, Robotham, Green, Griffiths, Stahl and Valmaggia2022; Shen et al., Reference Shen, Liu, Wu, Lin and Wang2022).
Finally, the correlations analysis showed that performance of cognitive and affective empathy as measured by the EAT task in SCZ patients was inversely correlated with negative symptoms, consistent with previous research (Wang et al., Reference Wang, Zhou, Liu, Wei, Xu, Wang and Wang2021). SCZ patients having severe negative symptoms are believed to have greater difficulties in perceiving and inferring others’ emotional states. Given that diminished emotion expression is a feature of negative symptoms, it is plausible that SCZ patients having severe negative symptoms are unable to resonate with others’ emotions (Ma et al., Reference Ma, Tian, Li, Liu, Wang and Zhang2024). Moreover, the CSAS and CPAS ratings in SCZ patients were inversely correlated with cognitive and affective empathy performance as measured using the EAT. The same patterns of correlations were also observed in the subclinical group (Wang et al., Reference Wang, Shi, Liu, Zheng, Wong, Cheung and Chan2020b), which reported that cognitive empathy and affective empathy were negatively correlated with schizotypal traits, particularly social anhedonia. By integrating the findings from both samples, empathy deficits in SCZ spectrum disorder may be linked to negative symptoms.
Several limitations of our study should be borne in mind. First, we did not measure participants’ attention to the presented information, but variations in attention allocation could confound EAT task performance. Eye-tracking technology to explore gaze patterns while watching the videos can be used in future research, such that it is possible to analyze how visual information processing would affect empathy. Second, although we measured affective empathy and empathic motivation using postvideo ratings, these subjective ratings were prone to social desirability bias. Future research should incorporate more objective indicators (e.g. heart rate, skin conductance response, or other physiological data) to assess participants’ empathic responses. Third, we did not differentiate the clinical group into different subtypes of SCZ. The heterogeneity of our sample may affect the clinical validity of our findings. Fourth, our subclinical sample did not include people with high levels of positive schizotypal traits. Future studies should recruit larger samples, differentiate SCZ subtypes, and explore the empathic characteristics of clinical and subclinical groups.
In conclusion, both SCZ patients and HSoA individuals have impaired cognitive empathy, but cognitive empathy deficits in SCZ patients is influenced by the presentation modality and emotional valence. SCZ patients are impaired in affective empathy under positive-valenced context only, but HSoA individuals show relatively normal affective component of empathy.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S003329172610364X.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgements
We thank all of the participants for participating in this project.
Author contributions
Miao Wang: Data curation, Investigation, Formal analysis, Writing – original draft. Guo-hui Zhu: Data curation, Investigation. Juan Yang: Data curation, Investigation. Xin-wei Fu: Data curation, Investigation. Li-Ying Zhang: Data curation, Investigation. Ding-ding Hu: Data curation, Formal analysis, Methodology, Writing – review & editing. Simon S.Y. Lui: Writing – review & editing. Yan-Yu Wang: Resources, Supervision, Writing – review & editing. Yi Wang: Conceptualization, Formal analysis, Funding acquisition, Methodology, Supervision, Writing – review & editing. Raymond C.K. Chan: Conceptualization, Funding acquisition, Project administration, Supervision, Writing – review & editing.
Funding statement
Raymond Chan was supported by the National Natural Science Foundation of China (32061160468) and the Philip K. H. Wong Foundation. Yi Wang was supported by the National Natural Science Foundation of China (31871114).
Competing interests
None.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. CRediT authorship contribution statement.