Introduction
A double-inlet, double-outlet right ventricle is a rare congenital malformation of the heart.Reference Jacobs, Franklin and Béland1–Reference Keeton, Macartney and Hunter3 Because of its relative rarity, large series describing double-inlet, double-outlet right ventricles are not readily available. The primary aim of this report was to pool morphologic data from reports of hearts with double-inlet, double-outlet right ventricles. The secondary aim of this report was to determine if natural groupings exist among these hearts.
Methods
Study design
This was a systematic review of the literature with statistical analyses conducted on the pooled data. As the data were already deidentified published data, there was no institutional review board approval necessary for this. This was a new systematic review protocol without a previously published protocol. This was done in concordance with the PRISMA checklist.
The primary objective of these analyses was to characterise the morphologic features of hearts with double-inlet, double-outlet right ventricles. The secondary objective was to determine if such hearts segregate into specific groups based on morphologic features.
Study identification
A systematic review of the literature was conducted by querying the following databases: PubMed, OVID, and EMBASE. The following keywords were used in various combinations across the electronic databases: “double outlet,” “double inlet,” “right ventricle,” “ventricle.” References of resulting studies from the electronic search were manually reviewed as well to identify additional studies that may be pertinent.
The title and abstract for identified studies were reviewed to determine the relevance of the studies to the objectives of the systematic review. Studies deemed to be relevant then had their full text retrieved. The full text was then reviewed to confirm relevance to the objectives of the systematic review.
Studies that described hearts with double-inlet, double-outlet right ventricles and included heart-level data were included in the final analyses. Heart-level data implies that the reported data were not summative findings of a group of hearts but rather that specific data for individual hearts was included.
Data extraction
Once relevant studies were identified, a data extraction tool was used to extract data from the studies. This tool was made for the systematic review, specifically. The following endpoints were included in the tool for extraction: gender, age at diagnosis, systemic venous connections, atria, atrial septal defect, atrioventricular connections, atrioventricular valve abnormalities, ventricular topology, apex direction, right ventricular abnormality, left ventricular abnormality, ventriculo-arterial connections, aortic stenosis, aortic atresia, pulmonary stenosis, pulmonary atresia, relative position of the aorta to the pulmonary trunk, coarctation or interruption of the aorta, aortic arch sidedness, coronary arteries, and isomerism.
Statistical analyses
Absolute counts and percentages were tabulated for descriptive variables, while mean and standard deviation were tabulated for continuous variables. Cluster analyses were performed using a K-means method. The number of optimal clusters was determined using the elbow method and the silhouette score. Two separate cluster analyses were performed: the first was done using ventricular morphology as assigned by the original reports, while the second was done using the ventricular morphology as assigned by reassessment. This repeat analysis was done, as it is difficult to assign ventricular morphology for a truly solitary ventricle. Thus, for hearts without a left ventricle, the authors do not find it logical or possible to assign a solitary right or left morphology.
All statistical analyses were conducted using the R statistical package. A p-value of less than 0.05 was considered statistically significant. Any use of the word “significant” or “significantly” refers to statistical significance and not clinical significance.
Results
Initial analyses based on ventricular morphology by report
A total of 42 hearts were included in analyses based on ventricular morphology by report.Reference Keeton, Macartney and Hunter3–Reference Thies, Soto, Diethelm, Bargeron and Pacifico12 The majority of bias, subjectively, came from a lack of reporting specific features in some reports. The elbow method demonstrated that 2 or 3 clusters would be optimal, while the silhouette score demonstrated that 3 or 4 clusters would be optimal. Thus, 3 clusters were utilised for the K-means clusters. The resulting clusters had good discrimination. The silhouette score was 0.56, demonstrating strong cluster assignment. The Davies-Bouldin index was 0.84, demonstrating clear separation of clusters with some overlap.
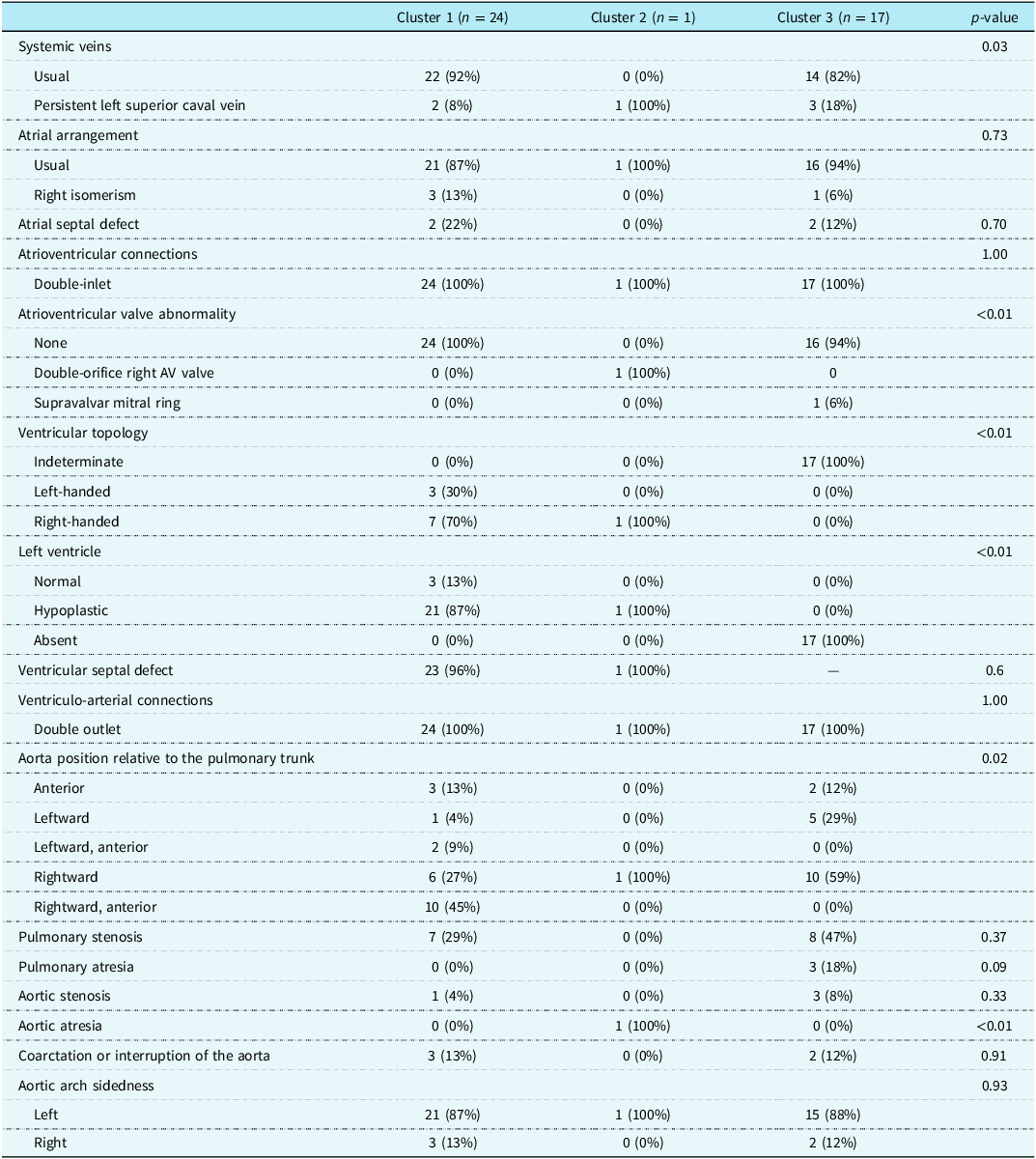
The characteristics of each cluster are specifically outlined in Table 1. Cluster 1 consisted of 24 hearts, cluster 2 consisted of 1 heart, and cluster 3 consisted of 17 hearts.
Characteristics by cluster

Cluster 1 consisted of hearts that had mostly usual systemic venous connections, a usual atrial arrangement, an intact atrial septum, normal atrioventricular valves, right-handed ventricular topology, a hypoplastic left ventricle, a ventricular septal defect, a rightward and anterior aorta, normal semilunar valves, a normal ascending and transverse aorta, and a left aortic arch. Figure 1 demonstrates the most common findings in cluster 1.
An illustration of a heart characteristic of hearts in cluster 1.

Cluster 2 consisted of hearts that had a persistent left superior caval vein, an intact atrial septum, a double-orifice right atrioventricular valve, right-handed ventricular topology, a hypoplastic left ventricle, a ventricular septal defect, a rightward aorta, aortic atresia, a normal pulmonary valve, and a left aortic arch. Figure 2 demonstrates the most common findings in cluster 2.
Cluster 3 consisted of hearts that had usual systemic venous connections, a usual atrial arrangement, an intact atrial septum, normal atrioventricular valves, indeterminate ventricular topology with an absent left ventricle, a rightward aorta, pulmonary stenosis or atresia, a normal aortic valve, a normal ascending and transverse aorta, and a left aortic arch.
An illustration of a heart characteristic of hearts in cluster 2.

The following morphologic characteristics were most important in cluster segregation: systemic venous connections, atrioventricular valve abnormalities, ventricular topology, left ventricular size, aortic atresia, and position of the aorta. Keeping these characteristics in mind, cluster 1 mostly usually had usual systemic venous connections, normal atrioventricular valves, right-handed ventricular topology, a hypoplastic left ventricle, a normal aortic valve, and a rightward and anterior aorta. Cluster 2 most usually had a persistent left superior caval vein, a double-orifice right atrioventricular valve, right-handed ventricular topology, a hypoplastic left ventricle, aortic atresia, and a rightward aorta. Cluster 3 most usually had normal systemic venous connections, normal atrioventricular valves, indeterminate ventricular topology with an absent left ventricle, pulmonary atresia, and a rightward aorta.
Repeat analyses based on a reassessment of ventricular morphology
A total of 25 hearts were included in repeat analyses based on reassessment of ventricular morphology. This repeat analysis was done, as it is difficult to assign ventricular morphology for a truly solitary ventricle. Thus, for hearts without a left ventricle, the authors do not find it logical or possible to assign a solitary right or left morphology.
The elbow method and the silhouette score both demonstrated that 2 clusters would be optimal. Thus, 2 clusters were used, or K-means clusters. The resulting clusters had good discrimination.
The clusters were identical to cluster 1 and cluster 2 reported earlier.
Cluster 1 consisted of hearts that had mostly usual systemic venous connections, a usual atrial arrangement, an intact atrial septum, normal atrioventricular valves, right-handed ventricular topology, a hypoplastic left ventricle, a ventricular septal defect, a rightward and anterior aorta, normal semilunar valves, a normal ascending and transverse aorta, and a left aortic arch.
Cluster 2 consisted of hearts that had a persistent left superior caval vein, an intact atrial septum, a double-orifice right atrioventricular valve, right-handed ventricular topology, a hypoplastic left ventricle, a ventricular septal defect, a rightward aorta, aortic atresia, a normal pulmonary valve, and a left aortic arch.
The following morphologic characteristics were most important in cluster segregation: systemic venous connections and aortic atresia.
Discussion
These analyses demonstrate that a double-outlet double-inlet right ventricle is quite a rare entity and that natural groupings of these hearts are largely based on systemic venous connections, atrioventricular valve abnormalities, ventricular topology, left ventricular size, aortic atresia, and position of the aorta.
Three distinct clusters were identified when ventricular morphology was captured as initially reported. All three clusters were similar in that they had an intact atrial septum, double-inlet atrioventricular connections, double-outlet ventriculo-arterial connections, and a left-sided aortic arch. Cluster 1 consisted of hearts that additionally had normal atrioventricular valves, a hypoplastic left ventricle, an interventricular communication, and a rightward and anterior aorta. Cluster 2 consisted of hearts that additionally had a double-orifice right atrioventricular valve, a hypoplastic left ventricle, an interventricular communication, and aortic atresia. Cluster 3 consisted of hearts that had an absent left ventricle, indeterminate ventricular topology, and pulmonary stenosis or atresia.
From a practical standpoint, one could describe cluster 1 as having normal aortic and pulmonary roots, cluster 2 as having an abnormal aortic root but normal pulmonary root, and cluster 3 as having an abnormal pulmonary root but normal aortic root. These clusters are of interest, as these are not human-defined but rather are statistically defined, making it more likely they represent natural groupings.
It should be noted that cluster 3 should not, technically, be considered to represent a double-inlet double-outlet right ventricle, as if there is a solitary ventricle, it is not feasible to assign a specific ventricular morphology. None of these hearts were placed in other clusters, and thus, when cluster analysis was repeated without hearts with a solitary ventricle, the natural groupings remained the same with cluster 1 and cluster 2. The only difference was that cluster 3 no longer existed. These hearts were included in the analyses, as they were included in the source papers as double-inlet double-outlet right ventricles. This highlights some of the shortcomings in labelling such hearts.
Conventionally, a majority of these hearts would undergo functionally univentricular palliation with an eventual Fontan procedure.Reference Anderson, Sleeper and Mahony13 Some centres are now, however, attempting to septate functionally univentricular hearts with double-inlet left ventricles.Reference Nguyen, Quaegebeur and Corda14–Reference Prasanna, Beroukhim, Ghelani, Feins, Del Nido and Emani16 This is still not the norm. A large issue in attempting to septate hearts with a double-inlet left ventricle is that the plane of septation is arbitrary. How the plane of septation impacts ventricular function long-term remains unknown. Additionally, ensuring the conduction system is not damaged during such an arbitrary septation is difficult since the conduction system in the setting of a double-inlet left ventricle can have variable locations.Reference Gikandi, Davee and O’Leary17,Reference Anderson18 Septating a double-outlet right ventricle may present additional difficulty. The conduction system most likely consists of an atrioventricular node that is displaced postero-inferiorly and lies close to the roof of the coronary sinus. The bundle of His generally courses inferiorly and leftward, passing along the postero-inferior rim of the remnant of the ventricular septum, eventually giving rise to a dominant left bundle. The right bundle is often diminutive.
This review collates morphologic characteristics of hearts with double-inlet and double-outlet right ventricles and identifies natural groupings using cluster analysis. Such an understanding allows for a guide to characteristics that clinical imaging must highlight for such hearts. It should be highlighted that the collation of such findings from a variety of reports (spanning a large time period and across different providers with different sets of descriptors) can introduce some room for error. However, the authors of the manuscript are trained cardiac morphologists with years of combined experience. The authors are all members of the International Society of Nomenclature for Paediatric and Congenital Heart Disease, which is a multinational effort to cross-map descriptors across different cardiac anatomy schools of thought and has produced the ICD-11 codes for congenital heart disease. We believe this has allowed for minimising any such errors. Another limitation of the analyses was that the details regarding the geography and borders of the ventricular septal defect were often not described with the precision needed to define what kind of ventricular septal defect it truly was. Alignment between the atrial and ventricular septa, as well as the infundibular presence, was similarly not described in an overwhelming majority of the source reports. It is important to highlight that these clusters are based on a relatively small sample size. Despite the strong model quality, there are still limitations. Nonetheless, these are rare hearts, and these data provide some early guidance on what hearts with double-inlet double-outlet right ventricles are like. Future studies, as more cases are described, will certainly be warranted.
Conclusion
Hearts with a double-inlet double-outlet right ventricle are infrequent. They tend to fall into two clusters that are largely based on aortic atresia and systemic venous connections.
Acknowledgements
We thank Yaeji Kim of CHD Doodles for her wonderful illustrations for this manuscript. Her wonderful work illustrating congenitally malformed hearts is an exceptional tool for patients, families, nurses, physicians, and the many others who care for those with congenitally malformed hearts.