Introduction
Enterovirus A71 (EV-A71) is a single-stranded, positive-sense RNA virus belonging to the enterovirus genus in the family Picornaviridae [Reference Wong1]. As one of the main pathogens causing hand, foot, and mouth disease (HFMD) in children, EV-A71 infections can cause mucocutaneous and respiratory manifestations, such as fever, sore throat, and multiple rashes on palms, soles, buttocks, and oral cavity. In rare instances, EV-A71 infection can result in more severe neurological and systemic manifestations, like aseptic meningitis, acute flaccid paralysis, and encephalitis [Reference Ooi2].
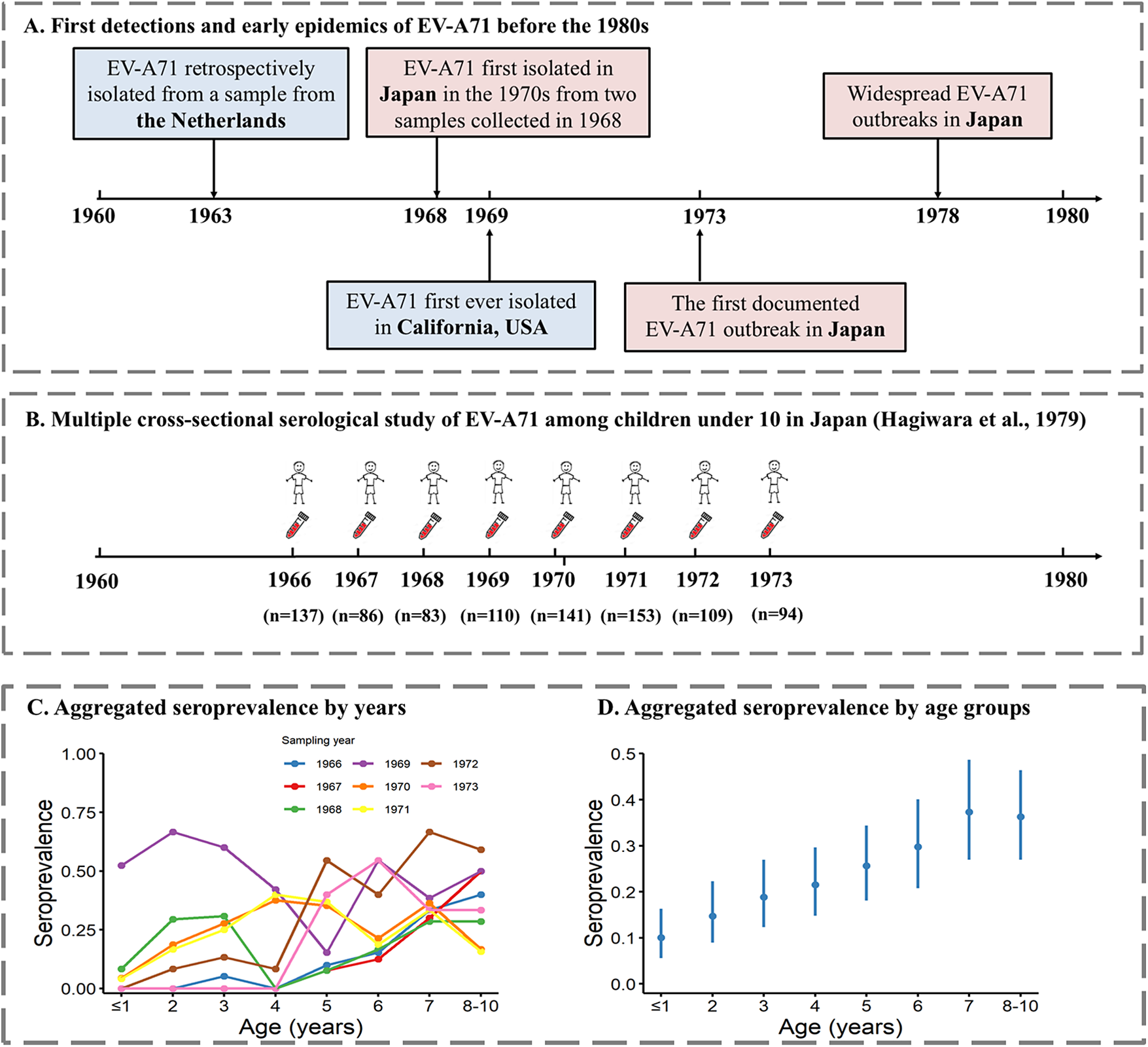
EV-A71 was first recognized as a new enterovirus serotype when isolated from more than 20 patients with disease of the central nervous system (CNS), mostly encephalitis and meningitis, in California, between 1969 and 1972 (Figure 1a) [Reference Schmidt4]. However, an extensive study of the epidemiology of EV-A71 in the Netherlands, which retrospectively tested isolates from 1963 onwards, identified EV-A71 in one isolate from 1963 (Figure 1a) and in a few others from 1965, 1966, and 1967 [Reference Van Der Sanden5]. In the 1970s, outbreaks of diseases associated with EV-A71 of different sizes and severity occurred around the globe, including in Australia [Reference Kennett6], the USA [Reference Deibel7], Sweden [Reference Blomberg8], Bulgaria [Reference Chumakov9, Reference Shindarov10], and Hungary [Reference Nagy11]. Most cases of these outbreaks reported neurological symptoms, often similar to poliomyelitis, although a few cases of skin rashes were also described [Reference Kennett6, Reference Blomberg8]. In Japan, two large-scale outbreaks of HFMD associated with EV-A71 occurred in 1973 and 1978, with most patients experiencing typical symptoms of HFMD (including skin manifestations) and a low incidence of disorders of the CNS (even lower in 1978 than in 1973), compared to previous and concurrent EV-A71 outbreaks in Europe and elsewhere [Reference Ishimaru12, Reference Tagaya13]. These were the first large-scale outbreaks of HFMD associated with EV-A71 ever reported [Reference Ishimaru12, Reference Tagaya13]. In the 1980s and early 1990s, sporadic epidemics of EV-A71 continued to occur (e.g. in Hong Kong, China, in 1985, and Australia in 1986) [Reference Gilbert14, Reference Samuda15]. In 1997, over 2500 HFMD cases and 34 deaths were reported in Sarawak, Malaysia [Reference Cardosa16], which marked the beginning of a series of large-scale outbreaks of HFMD in Southeast Asia [Reference Solomon17]. According to data from China’s national enhanced surveillance system, between 2008 and 2012, there were approximately two million HFMD cases each year, resulting in around 500–900 deaths annually [Reference Xing18].
Timeline of early EV-A71 detections worldwide and serology study in Japan. (a) Early detections and epidemics of EV-A71 worldwide and in Japan. (b) Sampling of EV-A71 serology study in Japan by Hagiwara et al. [Reference Hagiwara3] (c) Seroprevalence by age for different sampling years from Hagiwara et al. [Reference Hagiwara3]. (d) Age-specific seroprevalence aggregated across all sampling years.

Although a few species A enteroviruses can cause HFMD (including EV-A71, Coxsackievirus (CV)A6, CVA10, and CVA16), EV-A71 infections are responsible for the majority of severe and fatal cases [Reference Xing18], and EV-A71 is now considered the most neurotropic enterovirus after the three poliovirus serotypes. In 2015 and 2016, China approved the use of three inactivated EV-A71 vaccines, which had shown high efficacy against EV-A71–associated HFMD during clinical trials [Reference Adminstration19, Reference Li20]. However, vaccine coverage is low, and questions about the target population, optimal immunization schedule, and long-term effectiveness continue to exist. Today, EV-A71 infections remain a public health concern, posing a significant disease burden, especially in the paediatric population in Southeast Asia.
In order to clarify whether EV-A71 circulated in Japan before the first large epidemic of 1973, a retrospective serosurvey of neutralizing antibodies against EV-A71 among healthy children was conducted in the Takatsu area, Kawasaki City (near Tokyo) [Reference Hagiwara3]. Data from this serosurvey, with detailed age stratifications among children under 10 years old (yo) and annual sampling between 1966 and 1973, provide us with an excellent opportunity to reconstruct the historical circulation of EV-A71 in this area. Here, we use serocatalytic models and the seroprevalence data reported in Hagiwara et al. [Reference Hagiwara3] to reconstruct the historical circulation of EV-A71 in Kawasaki City, Japan, and quantitatively assess the likelihood of historical EV-A71 outbreaks during the 1960s.
Methods
Data
Age-stratified seroprevalence data were extracted from a retrospective cross-sectional serology study in Japan that tested archived serum samples collected each March between 1966 and 1973 [Reference Hagiwara3]. This study recruited 913 healthy children aged 0–10 years in the Takatsu area of Kawasaki City, near Tokyo, with an average of 114 (range: 83–141) participants recruited each year ( Figure 1b ). A live virus neutralization assay was used to detect EV-A71–specific antibody titres [Reference Moritsugu21]. Briefly, serial two-fold dilutions of serum were prepared in duplicate, starting from 1:4 to 1:512. Serum was mixed with titred virus (BrCr strain) in equal volume and then incubated at 36°C for 3 h and placed overnight at 4°C. After incubation, a continuous cynomolgus monkey kidney cell line (CMK1-S1) was added, and the resulting mixture was incubated at 36°C. The results were read on the fifth and seventh day after inoculation, and 50% endpoints were calculated. The seropositivity cut-off was set as 1:4.
The number of samples tested per age class and year is available in Hagiwara et al. [Reference Hagiwara3], and the aggregated age-specific seroprevalence per age class and year is displayed in figures [Reference Hagiwara3]. We used the online data extractor WebPlotDigitizer, version 4.8, to extract the crude seroprevalence for each age group and year, and then derived the number of seropositive samples by multiplying the extracted seroprevalence by the number of samples tested [Reference Rohatgi22]. Age-specific seroprevalence was provided aggregated for 2 consecutive years, 1966–1967 and 1970–1971, because age-specific seroprevalence patterns were very similar, according to the authors [Reference Hagiwara3]. For those years, we proportionally estimated the sample size for each age group based on the total number of samples tested each year.
Serocatalytic models
Serocatalytic models are designed to estimate the force of infection (FOI), which represents the per-capita rate at which susceptible individuals become infected, based on age-stratified seroprevalence data [Reference Rees23]. Here, we used the ‘RSero’ package within R (version 4.4.1) to test different models that make different assumptions about how the FOI changes over time and/or age [Reference Hozé24]. We considered three types of models: time-independent model, one-outbreak model, and two-outbreak model. Specifically, the time-independent model assumes that the FOI changes every year (hereinafter referred to as ‘independent model’), and a separate posterior distribution is estimated for the FOI in each year, resulting in a relatively large number of parameters in our setting. Because the FOI is reconstructed retrospectively from the sampling period, earlier years are informed by progressively fewer observations, reducing parameter identifiability and increasing posterior uncertainty. The outbreak model assumes that there have been a fixed number
![]() $ K $
of epidemics in the past, where
$ K $
of epidemics in the past, where
![]() $ K $
is a positive integer. When
$ K $
is a positive integer. When
![]() $ K $
= 1, it is referred to as the ‘one-outbreak model’, and when
$ K $
= 1, it is referred to as the ‘one-outbreak model’, and when
![]() $ K $
= 2, it is referred to as the ‘two-outbreak model’. Given a fixed value of
$ K $
= 2, it is referred to as the ‘two-outbreak model’. Given a fixed value of
![]() $ K $
, posterior inference is performed on a set of two-outbreak-specific parameters, including outbreak intensity (
$ K $
, posterior inference is performed on a set of two-outbreak-specific parameters, including outbreak intensity (
![]() $ {\alpha}_k $
) and timing (
$ {\alpha}_k $
) and timing (
![]() $ {T}_k $
). This parsimonious parameterization reduces model complexity relative to the independent model, leading to lower parameter uncertainty and a reduced risk of overfitting. For each of the three time-varying FOI models, we further tested four versions of the model: 1) the basic model, 2) a model with age-dependent FOI, 3) a model with seroreversion, and 4) a model with both age-dependent FOI and seroreversion. Therefore, a total of 12 models were considered. A summary of all models and their assumptions is available in Supplementary Tables S1 and S2.
$ {T}_k $
). This parsimonious parameterization reduces model complexity relative to the independent model, leading to lower parameter uncertainty and a reduced risk of overfitting. For each of the three time-varying FOI models, we further tested four versions of the model: 1) the basic model, 2) a model with age-dependent FOI, 3) a model with seroreversion, and 4) a model with both age-dependent FOI and seroreversion. Therefore, a total of 12 models were considered. A summary of all models and their assumptions is available in Supplementary Tables S1 and S2.
Model fitting, selection and comparison
All models were fitted to the seroprevalence data using Markov Chain Monte Carlo (MCMC), with the No-U-Turn sampler (NUTS) sampling algorithm to update the parameters. Four independent chains with 5000 iterations were used, with the first 2500 corresponding to the warm-up period. Convergence was evaluated both visually by inspecting the MCMC chains, and through the Rhat values and effective sample size (ESS) of the parameters. Specifically, the Rhat values should be below 1.01, while the bulk and tail ESS should exceed 400 for each parameter. Leave-One-Out Cross-Validation (LOO) and Deviance Information Criterion (DIC) were used for model selection [Reference Allen25]. To estimate the credible intervals (CrIs) for the parameters, we performed posterior sampling by drawing 1000 samples from the posterior distribution and extracted the 2.5th and 97.5th percentiles of these samples.
Results
Aggregated seroprevalence
Age-specific seroprevalence for each sampling year showed generally higher estimates in older children than in younger children. However, in contrast to other years, in 1969, seroprevalence in children under 3 yo exceeded 50% (Figure 1c). Across all years, the overall seroprevalence increased with age, from 10.1% (95% confidence interval (CI): 5.6%–16.3%) for children aged 0–1 years to 36.3% (95% CI: 27.0%–46.4%) for children aged 8–10 years (Figure 1d).
Model selection
We fitted a total of 12 serocatalytic models to the data, and based on the LOO and DIC criterion, we found that the independent model with age-dependent FOI was the best model, closely followed by the independent model with seroreversion and the independent model with both age-dependent FOI and seroreversion (Supplementary Table S3). The small differences in LOO and DIC values among these top three models suggest that they are statistically indistinguishable in terms of predictive performance. The two-outbreak model with age-dependent FOI and the two-outbreak model with seroreversion ranked fourth and fifth, respectively. The 4 one-outbreak models ranked lowest in both LOO and DIC comparisons, clearly indicating poor performance relative to the other models. Given these results, we decided to present and discuss the results of the independent and two-outbreak models with either age-dependent FOI (Models 2 and 10) or seroreversion (Models 3 and 11).
FOI estimates
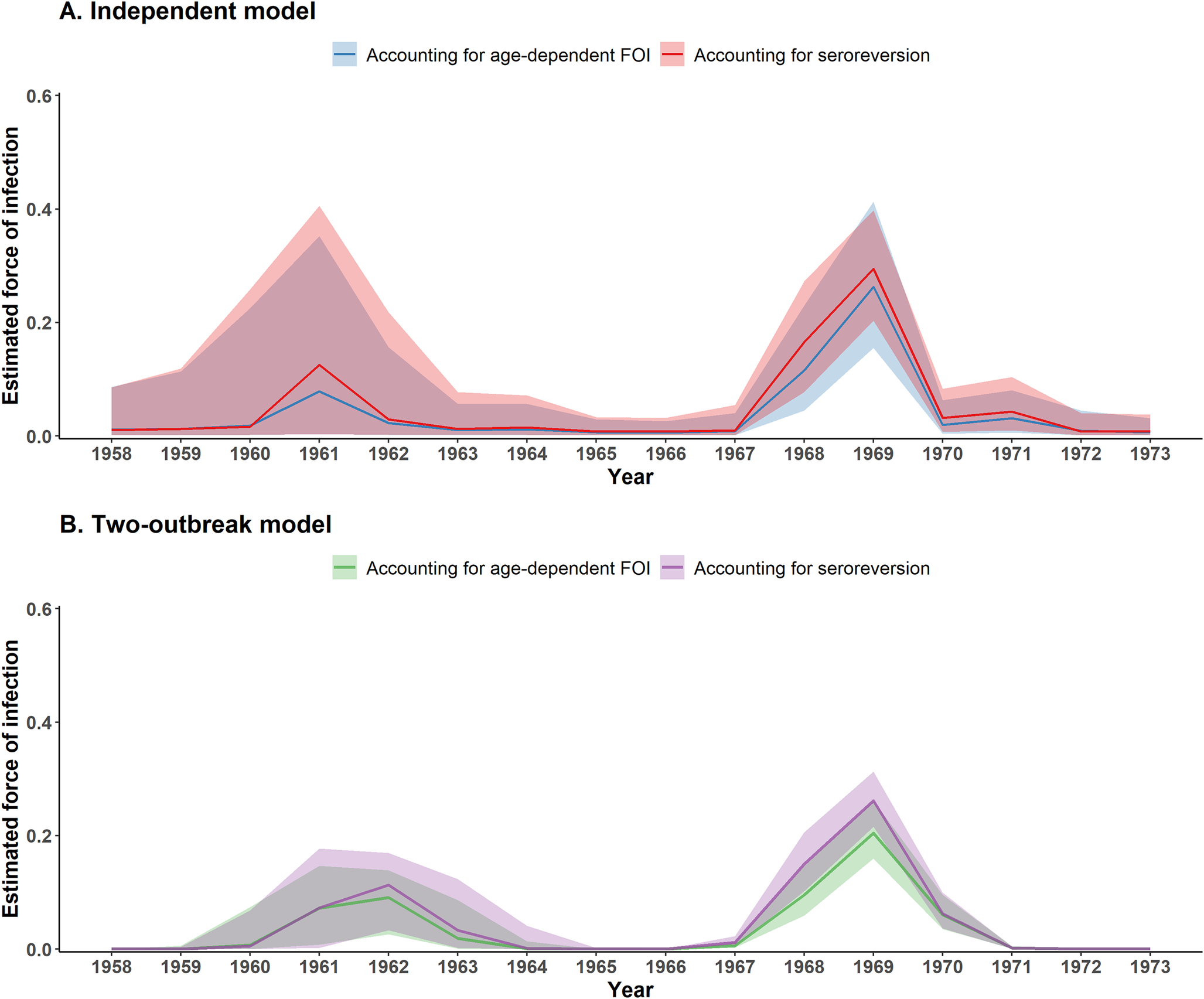
Similar temporal trends in FOI estimates were observed across the independent and two-outbreak models (Figure 2). All four models identified two main periods of EV-A71 circulation, one around 1968–1969 and an earlier one around 1961–1962. However, the annual FOI estimates from the two-outbreak model showed less uncertainty than those from the independent model, likely due to its simpler parameterization and the imposition of a strong temporal structure. As a result, the independent model resulted in larger uncertainty in the FOI estimates of the earliest outbreak. This uncertainty is evident in the wide variability of individual FOI trajectories (Supplementary Figure S1). Across the two-outbreak models, individual FOI trajectories showed a highly consistent timing for the 1968–1969 outbreak and a bit more variability in the timing of the earliest outbreak. In contrast, the individual FOI trajectories estimated with the two independent models showed greater variability in the timing of the 1968–1969 outbreak, which continued to increase towards the earliest outbreak, with the timing and magnitude of the FOI estimates varying considerably across trajectories.
Estimated FOI between 1958 and 1973 obtained with serocatalytic models. (a) Independent model. (b) Two-outbreak model. Coloured lines and envelopes indicate the annual FOI estimated median and corresponding 95% CrIs.

The models with an age-dependent risk of infection estimated that the FOI increased with age, with a parameter for the exponential increase of 0.01 (95% CI: −0.05–0.08) for the independent model and 0.04 (95% CI: 0–0.09) for the two-outbreak model. These correspond to a 1.15-fold (95% CI: 0.66–1.86) increase in FOI at age 10 compared to age 1 for the independent model and a 1.43-fold (95% CI: 1.02–1.96) increase for the two-outbreak model (Supplementary Figures S2 and S3). The CrI for the independent model included 1, indicating little to no evidence for age-related differences in risk. In contrast, the two-outbreak model excluded 1 from its CrI, suggesting a modest but statistically supported increase in infection risk with age. The models with seroreversion estimated similar seroreversion rates of 0.03 (95% CrI: 0.01–0.08) for the independent model and 0.02 (95% CrI: 0.01–0.06) for the two-outbreak model (Supplementary Table S4). These correspond to 13.9% (95% CrI: 4.8%–33.0%) and 9.5% (95% CrI: 4.8%–25.9%) of children losing detectable antibodies within 5 years, respectively, for the independent and two-outbreak models. Trace plots and posterior density plots for all estimated parameters are shown in Supplementary Figures S4–S11.
The outbreak models are parameterised to return the overall probability of infection over the course of an outbreak, also referred to as the attack rate. In the two-outbreak model with seroreversion, the estimated attack rates were 21.7% (95% CrI: 15.9%–29.2%) for the earlier outbreak and 38.7% (95% CrI: 34.1%–43.9%) for the later outbreak (Figure 2b). For the two-outbreak model with age-dependent FOI, the corresponding attack rates were lower, 19.8% (95% CrI: 0.1%–27.1%) and 34.9% (95% CrI: 27.5%–44.0%) for the earlier (1960–1964) and later outbreaks (1967–1971), respectively (Figure 2b ). To compare these estimates with those from the independent model, we computed the overall probability of infection over the period 1967–1971 (i.e. the years with a non-zero FOI) using the annual FOI estimates from the independent model (Figure 2a). The overall probability of infection was higher with the independent model compared to the two-outbreak model: 41.9% (95% CrI: 36.4%–47.9%) for the seroreversion model and 36.9% (95% CrI: 24.8%–50.3%) for the age-dependent model. We did not compute the attack rate for the earlier outbreak using the independent model, due to substantial uncertainty in FOI estimates prior to 1965.
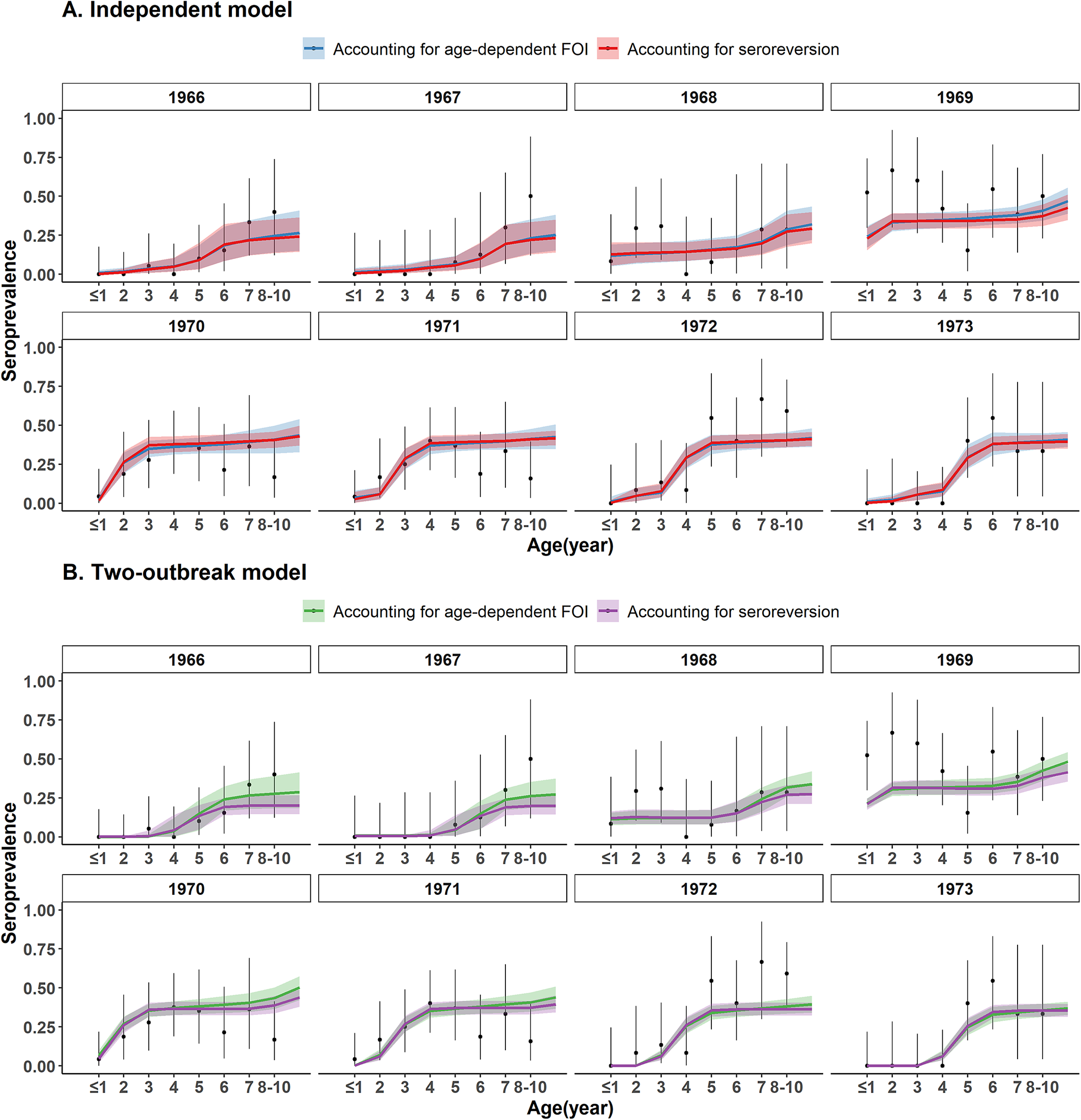
Modelled seroprevalence
All four models explained the age-patterns of seroprevalence well and provided very similar fits to the data (Figure 3). Seroprevalence was estimated close to zero in the 1–3 yo in 1966 and in the 1–4 yo in 1967, and then increased with age, reaching 26.5% (95% CrI: 15.1%–40.9%) in the 8–10 yo with the independent model with age-dependent FOI and 24.3% (95% CrI: 14.5%–36.5%) with the independent model with seroreversion. With the two-outbreak model, seroprevalence increased from less than 1% in children aged 1–3 years to 27.3% (95% CrI: 18.7%–37.5%) and 19.8% (95% CrI: 14.4%–26.4%) in those aged 8–10 years in 1967, respectively, when accounting for age-dependent FOI and seroreversion. Seroprevalence then increased in the younger age classes when it almost plateaued at around 12% in 1968 and 31% in 1969 for 2 yo children. In 1970 and 1971, seroprevalence increased sharply with age for children under 4 yo, and then plateaued between the ages of 4 and 10 years at around 40%, in agreement with the outbreak around 1968–1969. A similar age pattern, shifted in age, was estimated for 1972 and 1973, when seroprevalence in the 1 yo and in the 1–2 yo, respectively, was estimated null or close to zero.
Model fit to EV-A71 seroprevalence data. (a) Independent model. (b) Two-outbreak model. Black points indicate the original seroprevalence data from Hagiwara et al. and intervals are the 95% binomial CIs. Coloured lines and envelopes are the mean values and 95% CrIs estimated with the serocatalytic models.

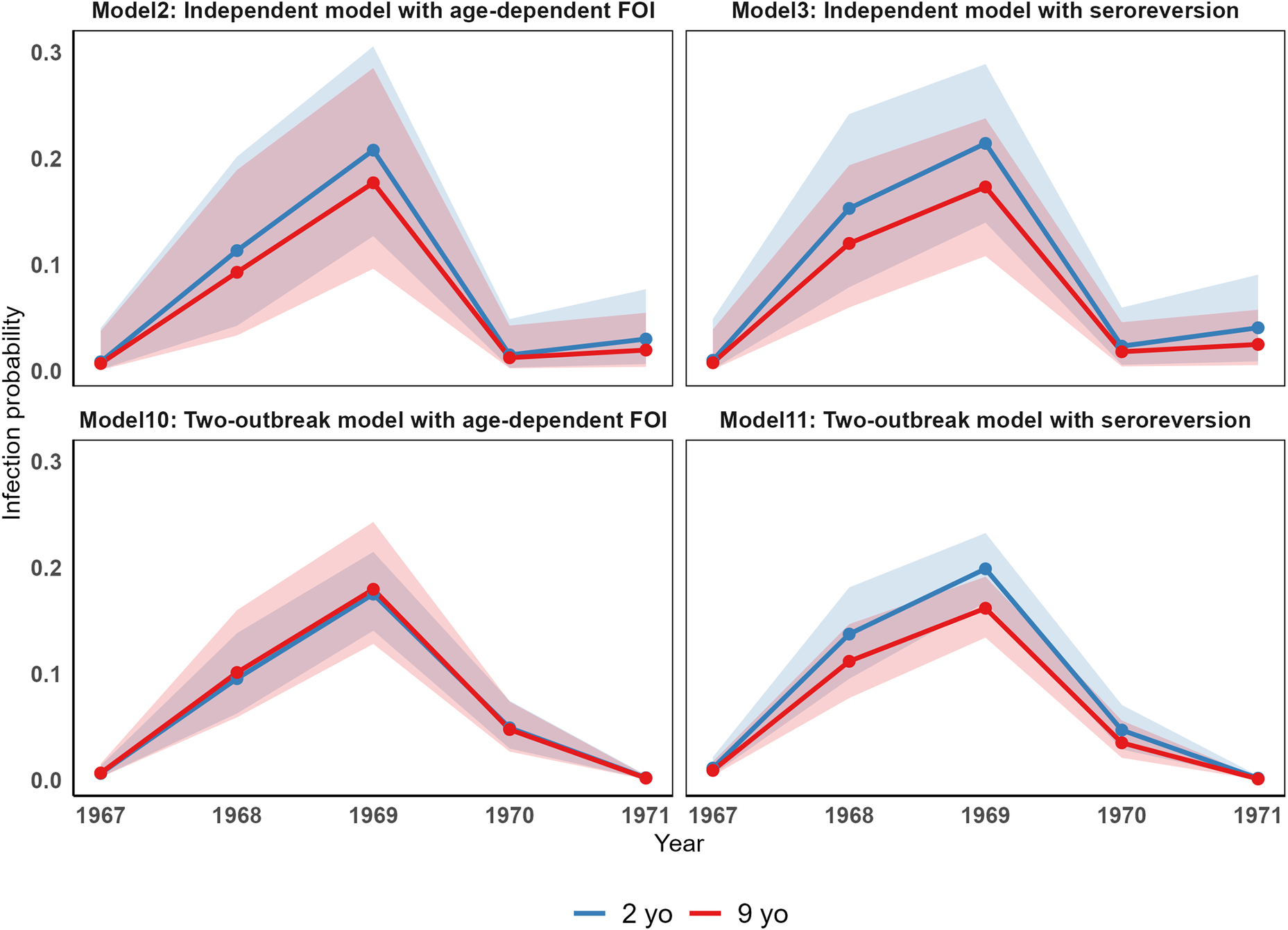
Seroconversion across age
Although the two versions of the two models (independent and two-outbreak) provided similar results in terms of the annual FOI estimates and model fits, the two underlying assumptions (age dependency in the risk of seroconversion vs. seroreversion) may result in different dynamics of seroconversion across different age groups. To investigate these differences through age under the four models, we computed the probability of infection for a typical 2-yo individual and a typical 9-yo individual (Figure 4). The two models with seroreversion estimated a higher probability of seroconversion for the 2 yo than the 9 yo. This pattern aligns with a smaller proportion of 9 yo being susceptible, a constant risk of infection across age, and a low seroreversion rate. In contrast, the two models with age-dependent FOI showed smaller differences in the probability of infection between the two age groups. Notably, in the two-outbreak model, the probability was almost identical for both age classes. This pattern may be explained by the estimated higher risk of seroconversion with age (which is higher for the two-outbreak model than that for the independent model) that counterbalances the decreasing proportion of susceptible with age and the absence of seroreversion.
Differences in the risk of seroconversion through age across the independent and two-outbreak models.

Discussion
Using serocatalytic models and publicly available serological data, this study estimates the timing and magnitude of the earliest EV-A71 outbreaks ever reported in Japan. We estimated that during the decade of the 1960s, there were two main circulation periods in Kawasaki City (near Tokyo), one around 1961–1962 and the other around 1968–1969. The timing of these outbreaks coincides with the first detections of EV-A71 reported from clinical samples around the globe: in 1963 from one sample in the Netherlands [Reference Van Der Sanden5], followed by a few other detections between 1965 and 1967; in 1967 from six cases of aseptic meningitis in Sweden [Reference Blomberg8]; and in 1969 from a patient with encephalitis in California, USA [Reference Schmidt4], followed by other isolations from patients with CNS disease during 1970–1972.
Bayesian evolutionary analysis of the VP1 capsid sequence estimated that EV-A71 diverged from its closely related ancestor CVA16 around 1940, with relatively large uncertainty around that estimate (95% CI: 1928.8–1952.2) [Reference Tee26]. EV-A71 is therefore a relatively recent human pathogen. However, the exact location of emergence and initial routes of spread remain uncertain. Our results, jointly with the currently available literature, suggest that by the second half of the 1960s, EV-A71 had already widespread through Europe, the USA, and Japan.
Our results are robust to the model structure. Both the time-independent and two-outbreak models (either with age-dependent FOI or seroreversion) provided highly consistent estimates of the FOI between 1964 and 1973. There was a bit more uncertainty around the FOI estimates between 1959 and 1963, when less data informed the parameters. Nevertheless, both models clearly supported the circulation of EV-A71 during two different periods between 1959 and 1973. In addition, with seroprevalence >10% in those aged 5 years and older in the first time-point of Hagiwara et al. in 1966, it is likely that EV-A71 already circulated before 1966 in Japan.
A countrywide HFMD epidemic associated with CVA16 occurred in Japan between mid-1969 and the end of 1970 [Reference Tagaya27]. However, it was reported that a substantial proportion of all tested specimens was not positive for CVA16 [Reference Tagaya27]. In addition, isolates from those years were not neutralized by antiserum against the CVA16 prototype. It may be that EV-A71 (which was not recognized as a new enterovirus serotype by that time) co-circulated with CVA16 during the time of that outbreak years. In addition, EV-A71 circulation is also corroborated by the first isolation of this virus from two samples collected in 1968 [Reference Hagiwara3]. Japan also experienced a nationwide EV-A71 outbreak in 1973 [Reference Tagaya28]. However, our study failed to capture this outbreak, probably because our extracted data for 1973 were based on samples from March and the outbreak occurred later in the year [Reference Tagaya28]. There were no seropositive children younger than 4 years in 1973 in our dataset.
Interestingly, our findings are consistent with EV-A71 case-based surveillance data from Japan from 2000 onwards, as well as estimates from two modelling studies that fitted transmission models to those data [Reference Pons-Salort29, Reference Yan30]. These studies reported recurrent EV-A71 outbreak cycles occurring approximately every 4–5 years, with major outbreaks infecting around 20%–30% of the population. Similarly, our two-outbreak model estimated a mean attack rate of 21.8%–37.8%, alongside an approximately 5-year interval between the two reconstructed outbreaks. While the previous studies considered the entire population, our analysis focused on children under 10 years of age; however, given that EV-A71 infections predominantly occur in young children, the results remain broadly comparable. Together, these comparisons suggest that the reconstructed epidemiological patterns for EV-A71 infection in Kawasaki City in the 1960s are consistent with those observed in Japan over the last two decades.
In our study, the independent and the two-outbreak models with seroreversion performed better than their counterparts without it. Specifically, we estimated a seroreversion rate of 0.03 (95% CrI: 0.01–0.08) and 0.02 (95% CrI: 0.01–0.06) for the independent and the two-outbreak models, respectively. This corresponds to a mean duration of antibody detection of 32.4 (95% CrI: 13.1–95.4) and 42.8 (95% CrI: 18.6–123.4) years, respectively. However, the decay of seropositivity may not be very well estimated, given that we only have data for individuals up to 10 years of age. Furthermore, the long duration of seropositivity could also partly be explained by the low seropositivity cut-off (1:4) used in the serological study. Additionally, the long EV-A71 duration of seropositivity is supported by a study that reported that up to 97% of young children maintained neutralizing antibody titres above 1:8 even 5 years after receiving a single dose of the inactivated EV-A71 vaccine [Reference Hu31]. Moreover, findings from a seroepidemiology study conducted in Southern China between 2013 and 2018 reported that following natural infection, geometric mean titers (GMTs) among seropositive individuals slowly declined with age [Reference Yang32]. However, GMTs remained above 64 in children aged 12 years, indicating long-lasting EV-A71 antibody titres and possibly low seroreversion rates. Given the wide CrIs around our estimated seroreversion rates, as well as data limitations noted above, it is difficult to draw definitive conclusions about the true underlying seroreversion rate.
Our study has several limitations, mainly inherent to the data used to inform the models. Firstly, data were extracted from a published study that reported data twice for 2 consecutive years together. However, the authors explained in their paper that the age profile was very similar for those 2 years, and that was the reason why they plotted it together. This may slightly affect our estimates of the timing of inferred outbreaks. Second, we do not have detailed information on the children who were sampled; only their age and that they were healthy were available, but we do not know to what extent they were representative of the children who lived in Kawasaki City at the time of data collection (1966–1973).
In conclusion, our study provides evidence of the first EV-A71 outbreaks in Japan, by reconstructing historical circulation from detailed retrospective seroprevalence data from consecutive cross-sectional surveys that sampled children over 8 consecutive years. This captured well the first infections and allowed reconstructing EV-A71 circulation on a fine timescale. This work highlights the value of testing archived serum samples to clarify the emergence and spread of human pathogens.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0950268826101435.
Data availability statement
The data and code are available at https://github.com/cxhhhh24/ev71_foi.git
Author contribution
X.C.: formal analysis, investigation, visualization, writing – original draft; N.H.: methodology, writing – review and editing; M.P.-S.: conceptualization, resources, methodology, writing – review and editing.
Funding statement
X.C. and M.P.-S. acknowledge funding from the Medical Research Council (MRC) Centre for Global Infectious Disease Analysis (reference MR/X020258/1), funded by the UK MRC. This UK-funded award is carried out in the frame of the Global Health EDCTP3 Joint Undertaking. M.P.-S. is a Sir Henry Dale fellow, a programme jointly funded by the Wellcome Trust and the Royal Society (grant number 216427/Z/19/Z).
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Patient consent statement
This study is based on secondary aggregated data and does not involve any individual participants or patient information.
Disclaimer
The views expressed are those of the authors and do not necessarily represent the institutions with which the authors are affiliated.