Bipolar disorder is a chronic psychiatric condition that requires long-term pharmacotherapy to prevent mood episodes and hospitalisations. Reference Goes1,Reference Yatham, Kennedy, Parikh, Schaffer, Bond and Frey2 Clinical practice guidelines strongly recommend lithium, second-generation antipsychotics and combinations of a second-generation antipsychotic with either lithium or divalproex. Reference Goes1,Reference Yatham, Kennedy, Parikh, Schaffer, Bond and Frey2 However, these recommendations are primarily based on randomised clinical trials (RCTs), which often lack generalisability to real-world clinical settings. Reference Wong, Jones, Timko and Humphreys3,Reference Storosum, Fouwels, Gispen-de Wied, Wohlfarth, van Zwieten and van den Brink4 Patients enrolled in RCTs typically differ from routine clinical populations due to highly restrictive exclusion criteria, such as comorbid substance use disorders, elevated suicide risk and coexisting medical conditions. Reference Wong, Jones, Timko and Humphreys3
By contrast, population-based observational studies offer complementary insights into treatment effectiveness in broader and more representative populations. Although meta-analyses of RCTs have shown that both lithium and quetiapine are superior to placebo in the treatment of acute mania and demonstrate comparable efficacy, Reference Huang, He, Liu and Xu5,Reference Cipriani, Barbui, Salanti, Rendell, Brown and Stockton6 findings from observational studies suggest differential effectiveness. One study found that lithium was the most effective mood stabiliser, being associated with a 33% reduction in the risk of psychiatric hospitalisation, whereas quetiapine was associated with a reduction in risk of only 8%. Reference Lahteenvuo, Tanskanen, Taipale, Hoti, Vattulainen and Vieta7 A more recent study reported that lithium was the only treatment associated with reduced risks of both psychiatric and non-psychiatric hospitalisations, whereas quetiapine was associated with an increased risk of non-psychiatric hospitalisation. Reference Lahteenvuo, Paljarvi, Tanskanen, Taipale and Tiihonen8
Although observational studies are inherently vulnerable to confounding by indication, Reference Booth and Tannock9 they remain essential for evaluating pharmacological effectiveness in routine clinical practice. However, existing population-based studies on treatment effectiveness in bipolar disorder are limited, Reference Lahteenvuo, Tanskanen, Taipale, Hoti, Vattulainen and Vieta7,Reference Lahteenvuo, Paljarvi, Tanskanen, Taipale and Tiihonen8,Reference Joas, Karanti, Song, Goodwin, Lichtenstein and Landen10–Reference Ermis, Taipale, Tanskanen, Vieta, Correll and Mittendorfer-Rutz12 have been conducted primarily in northern Europe and have rarely assessed the effectiveness of combination therapy involving specific drug regimens. Reference Wingard, Boden, Brandt, Tiihonen, Tanskanen and Kieler11,Reference Ermis, Taipale, Tanskanen, Vieta, Correll and Mittendorfer-Rutz12
To address these gaps, we conducted a population-based cohort study to assess the real-world effectiveness of both mono- and combination therapy with mood stabilisers and antipsychotics in preventing psychiatric hospitalisations among patients with bipolar disorder. We hypothesised that treatment effectiveness would vary by specific drug or drug combination.
Method
Study design
The overall study design is illustrated in Fig. 1 and the Supplementary Methods available at https://doi.org/10.1192/bjp.2026.10636. This was a population-based cohort study using data from the National Database of Health Insurance Claims and Specific Health Check-ups of Japan (NDB). The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation, and with the Helsinki Declaration of 1975 as revised in 2013. The study was approved by Juntendo University Ethics Committee (no. E22-0372-M01). The requirement for informed consent was waived under Japanese regulatory guidelines because the data were anonymised secondary use. The use of NDB was reviewed by a specialist committee and approved by the Ministry of Health, Labour and Welfare of Japan. The study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology reporting guidelines. People with lived experience of bipolar disorder were not involved in every phase of this study.
Design diagram. aFollow-up censored at the last recorded visit, death or May 2023, whichever occurred first.

Data source
NDB is one of the largest health-related databases worldwide. Reference Suto, Iba, Sugiyama, Kodama, Takegami and Taguchi13,Reference Suto, Sugiyama, Imai, Furuno, Hosozawa and Ichinose14 Established in the fiscal year 2009 by the Ministry of Health, Labour and Welfare, it was developed under Japan’s universal health insurance system and covers the entire Japanese population (approximately 126 million individuals). The database contains all electronic claims data submitted from insurance medical care facilities to health insurance associations and publicly funded healthcare programmes (e.g. the public assistance system). These claims include information on diagnoses, medical procedures and prescriptions, enabling population-based observational studies.
The source data for this study were extracted by the data vendor on 17 May 2024. The vendor identified patients aged 20 years or older who had a recorded diagnosis of schizophrenia, bipolar disorder or mitochondrial disease in any diagnosis status between April 2009 and May 2023 in NDB. Subsequently, data on diagnoses, procedures and prescriptions were extracted for these patients during the specified period. Notably, dispensed prescription data were available for in-house pharmacies, where nearly all patients were expected to receive their medications, and filled prescription data were available for outside pharmacies, where only prescriptions that were actually dispensed were captured, because some patients might not have filled in their prescriptions.
Patient selection
The study included patients who met the following eligibility criteria. First, they were required to have claims data for psychiatric in-patient care fees or psychiatric visit fees between April 2013 and March 2022, with a primary diagnosis of bipolar disorder (ICD-10 codes F30–1). The earliest date of psychiatric in-patient care or psychiatric visit during this period was defined as the index date. Second, patients must have received at least 2 prescriptions for lithium at a dosage of 200 mg/day or higher during the study period between April 2009 and May 2023. This criterion was established for the following reasons: (a) lithium is approved solely for the treatment of bipolar disorder, whereas other mood stabilisers and antipsychotics are approved for additional indications such as schizophrenia and epilepsy; (b) lithium is the only first-line agent recommended for the maintenance treatment of bipolar disorder by the treatment guidelines of the Japanese Society of Mood Disorders during the study period; Reference Kanba, Kato, Terao and Yamada15 and (c) lithium was the most frequently prescribed medication for the treatment of bipolar disorder during this period. Reference Kawamata, Yasui-Furukori, Adachi, Ueda, Hongo and Azekawa16 This criterion is unlikely to compromise the representativeness of our study population, because the proportion of patients receiving lithium at a given time point is much higher in Japan Reference Kawamata, Yasui-Furukori, Adachi, Ueda, Hongo and Azekawa16 (47%) than in either the UK (16%) Reference Ng, Man, Gao, Chan, Lee and Hayes17 or USA (17%), Reference Rhee, Olfson, Nierenberg and Wilkinson18 and our assessment window for lithium prescriptions covered the entire study period (approximately 14 years) rather than a single time point. Third, patients had to be aged 20 years or older on the index date. Fourth, patients were required to have at least 366 days of data available prior to the index date. Fifth, patients with a history of dementia diagnosis (ICD-10 codes F00–3, 05.1, G30–1) or dementia-related treatment prior to the index date were excluded. Finally, all included patients were required to have at least one day of follow-up data.
Outcomes
The primary outcome was time to psychiatric hospitalisation. Under Japan’s reimbursement system and the Medical Care Act, patients with mental illness are typically admitted to designated psychiatric beds, except when hospitalisation is required for the treatment of physical comorbidities. The secondary outcome was time to all-cause hospitalisation, including psychiatric and non-psychiatric hospitalisations. This outcome was used as a proxy measure for the overall effectiveness of treatments reflecting tolerability, as in previous studies. Reference Lahteenvuo, Tanskanen, Taipale, Hoti, Vattulainen and Vieta7,Reference Tiihonen, Tanskanen, Hoti, Vattulainen, Taipale and Mehtala19 For out-patients at the index date, follow-up began the day following index date; for in-patients, follow-up began the day following discharge. Follow-up continued until either the last recorded visit, death or May 2023, whichever occurred first.
Exposures
The primary exposures of interest were periods of monotherapy with individual mood stabilisers or antipsychotics. Mood stabilisers included carbamazepine, lamotrigine, lithium and valproate, all of which are approved in Japan for the treatment of bipolar disorder. Antipsychotics included 33 oral formulations and 5 long-acting injectables (LAIs), regardless of their regulatory approval status for bipolar disorder (Supplementary Table 1). Short-acting injectable antipsychotics and reserpine were excluded. A transdermal patch formulation of blonanserin was categorised as an oral antipsychotic.
Periods of mood stabiliser use were categorised into three groups: (a) monotherapy with a specific mood stabiliser (exposure period); (b) non-use of any mood stabilisers (control period); and (c) other regimens, including combination therapy with mood stabilisers (e.g. lithium plus valproate) or monotherapy with a different mood stabiliser. For antipsychotics, periods of use were similarly categorised into three groups: (a) monotherapy with a specific antipsychotic (exposure period); (b) non-use of any antipsychotics (control period); and (c) other regimens, including combination therapy with antipsychotics (e.g. quetiapine plus sulpiride) or monotherapy with a different antipsychotic.
For oral medications, the period of drug use was determined based on the drug code, prescription date and days supplied recorded in the claims data. For long-acting formulations, the period of drug use was identified using the drug code and prescription date from claims data, along with the dosing interval specified in the package insert.
The exploratory exposures of interest were periods of quetiapine monotherapy stratified by daily dosage. Periods were categorised into four groups: (a) quetiapine monotherapy at a dosage of ≥300 mg/day (first exposure period); (b) quetiapine monotherapy at a dosage of <300 mg/day (second exposure period); (c) non-use of any antipsychotics (control period); and (d) other regimens, including combination therapy with antipsychotics or monotherapy with a different antipsychotic. The cut-off of 300 mg/day was based on prior literature regarding the treatment of bipolar disorder. Reference Vieta, Calabrese, Goikolea, Raines, Macfadden and Group20,Reference Muneer21
The secondary exposures of interest were periods of combination therapy. Although numerous drug combinations are used in clinical practice for the treatment of bipolar disorder, this study focused on the following specific combinations based on clinical relevance: (a) lithium plus a specific mood stabiliser; (b) lithium plus a specific antipsychotic; (c) valproate plus a specific antipsychotic; and (d) lamotrigine plus a specific antipsychotic. For this analysis, the ten most frequently prescribed antipsychotics – aripiprazole, quetiapine, olanzapine, risperidone, levomepromazine, sulpiride, chlorpromazine, lurasidone, brexpiprazole and zotepine – were included regardless of formulation (i.e. oral or LAI). Periods of drug use were categorised into (a) a specific combination therapy (e.g. lithium plus valproate, exposure period); (b) monotherapy (e.g. lithium only, control period); (c) other regimens (e.g. lithium plus valproate plus quetiapine); and (d) non-use of any mood stabilisers or antipsychotics.
Covariates and additional variables
Time-varying covariates included antipsychotic use (in models assessing mood stabiliser effectiveness), mood stabiliser use (in models assessing antipsychotic effectiveness), benzodiazepine use, antidepressant use, use of drugs with adverse effects on mood and time since cohort entry. Additional variables describing patient characteristics included year of cohort entry, age, gender, care setting at the index date and comorbidities, including cardiovascular disease, diabetes, thyroid disorders, asthma or chronic obstructive pulmonary disease, cancer, substance use disorder, poisoning, injury and the Sundararajan version of the Charlson Comorbidity Index. Reference Sundararajan, Quan, Halfon, Fushimi, Luthi and Burnand22 Details are provided in Supplementary Methods.
Statistical analyses
Patient characteristics are summarised as counts and percentages. Follow-up duration and incidence rates of outcome events were reported as events per 100 person-years. Stratified Cox proportional hazards models, in which each patient served as their own control to minimise selection bias, were used to evaluate the association between each exposure and the outcome event. Reference Lichtenstein, Halldner, Zetterqvist, Sjolander, Serlachius and Fazel23 To enable within-individual comparisons of treatment duration, follow-up time was reset to zero after each outcome event and restarted on the day following discharge, consistent with previous studies. Reference Lahteenvuo, Paljarvi, Tanskanen, Taipale and Tiihonen8,Reference Taipale, Lieslehto, Lahteenvuo, Hamina, Tanskanen and Mittendorfer-Rutz24 Adjusted hazard ratios (aHRs) and their 95% confidence intervals were estimated from models adjusted for time-varying covariates.
To enhance diagnostic specificity and address potential diagnostic transitions from bipolar disorder to schizophrenia or schizoaffective disorder, Reference Kendler, Abrahamsson, Sundquist and Sundquist25,Reference Hung, Yang, Kuo and Lin26 we conducted sensitivity analyses for the primary outcome, excluding patients with a primary diagnosis of schizophrenia or schizoaffective disorder and without any diagnosis of bipolar disorder during psychiatric hospitalisation following the index date. We also performed sensitivity analyses that omitted the first 30 days after each new treatment pattern, to control for potential protopathic bias.
All statistical analyses were conducted using R version 4.3.1 for Windows (R Foundation for Statistical Computing, Vienna, Austria; https://cran.r-project.org/).
Role of funding source
The funder of the study had no role in study design, data collection, data analysis, data interpretation or writing of the report.
Results
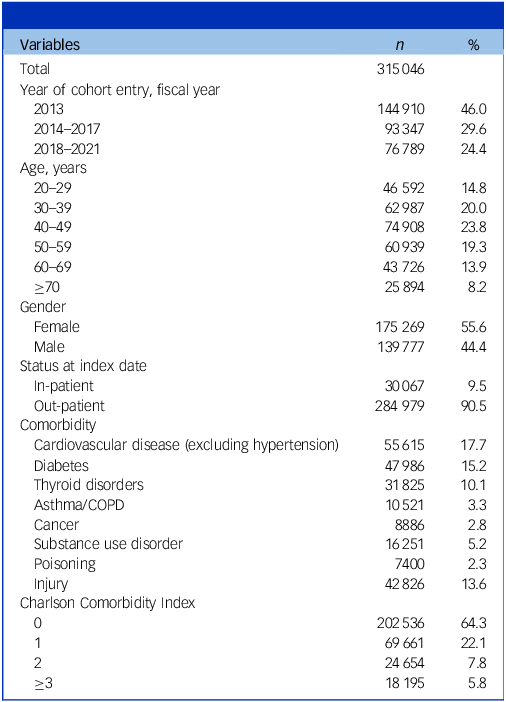
The study population comprised 315 046 patients with bipolar disorder (Table 1 and Supplementary Fig. 1). The most prevalent age group was 40–49 years (23.8%). Of the total sample, 175 269 (55.6%) were females and 284 979 (90.5%) had received treatment in out-patient settings on the index date. Within the past year, 42 826 patients (13.6%) had a history of injury, 7400 (2.3%) had a history of poisoning and 16 251 (5.2%) had received treatment for substance use disorders. A Charlson Comorbidity Index score of ≥1 was observed in 112 510 patients (35.7%). The median follow-up period was 7.1 years (interquartile range 3.7–9.7 years). Lithium was the most commonly used mood stabiliser, and aripiprazole the most frequently prescribed antipsychotic (Supplementary Table 2). During follow-up, 208 896 psychiatric hospitalisations were reported, corresponding to an incidence rate of 10.2 per 100 person-years.
Demographic characteristics

COPD, chronic obstructive pulmonary disease.
Figure 2 presents the aHRs for psychiatric hospitalisations associated with monotherapy using individual mood stabilisers or major antipsychotics, compared with non-use of medications within the same pharmacologic class. To conserve space, Fig. 1 includes only drugs used by ≥1000 patients; results for all antipsychotics are available in Supplementary Table 2.
Risk of psychiatric hospitalisation associated with mood stabiliser monotherapy or major antipsychotic monotherapy, compared with non-use. Adjusted hazard ratios (aHRs) and corresponding 95% confidence intervals are plotted on a logarithmic scale. Exposures are defined as periods of monotherapy with a specific mood stabiliser or antipsychotic, each compared with periods of non-use of drugs within the same pharmacologic class. Results are shown only for drugs with 1000 or more users. LAI, long-acting injectable.

Monotherapy with lithium (aHR 0.67, 95% CI 0.66–0.68), valproate (aHR 0.71, 95% CI 0.70–0.73), lamotrigine (aHR 0.72, 95% CI 0.69–0.75) and carbamazepine (aHR 0.74, 95% CI 0.70–0.78) was associated with a lower risk of psychiatric hospitalisation compared with non-use of any mood stabilisers.
The associations between antipsychotic monotherapy and psychiatric hospitalisation were heterogeneous. A lower risk was observed during monotherapy with sultopride (aHR 0.53, 95% CI 0.43–0.66), paliperidone LAI (aHR 0.61, 95% CI 0.53–0.71), aripiprazole LAI (aHR 0.62, 95% CI 0.56–0.69), propericiazine (aHR 0.65, 95% CI 0.51–0.83), oral aripiprazole (aHR 0.73, 95% CI 0.70–0.75), oral paliperidone (aHR 0.73, 95% CI 0.65–0.82), zotepine (aHR 0.74, 95% CI 0.69–0.79), brexpiprazole (aHR 0.74, 95% CI 0.68–0.81), olanzapine (aHR 0.82, 95% CI 0.79–0.84), haloperidol (aHR 0.82, 95% CI 0.75–0.90), quetiapine (aHR 0.82, 95% CI 0.80–0.84), tiapride (aHR 0.82, 95% CI 0.69–0.99), levomepromazine (aHR 0.86, 95% CI 0.83–0.90), risperidone (aHR 0.87, 95% CI 0.83–0.90), lurasidone (aHR 0.88, 95% CI 0.82–0.95), blonanserin (aHR 0.91, 95% CI 0.83–1.00) and chlorpromazine (aHR 0.95, 95% CI 0.90–1.00). For other antipsychotics – including asenapine, bromperidol, fluphenazine, haloperidol LAI, perospirone, perphenazine, prochlorperazine, risperidone LAI and sulpiride – the associations with psychiatric hospitalisation were inconclusive. A dose-dependent effect was observed for quetiapine, with higher doses associated with a greater reduction in the risk of psychiatric hospitalisation (aHR for ≥300 mg/day, 0.75; aHR for<300 mg/day, 0.85; Supplementary Table 2).
Similar patterns were observed for all-cause hospitalisations across most medications. Notably, prochlorperazine was associated with a significantly higher risk of all-cause hospitalisation, although its association with psychiatric hospitalisation remained uncertain (Supplementary Table 3).
Figure 3 illustrates the risk of psychiatric hospitalisation associated with combination therapy regimens compared with monotherapy (see Supplementary Table 4). Compared with lithium monotherapy, a decreased risk was observed for the following combinations: lithium plus carbamazepine (aHR 0.73, 95% CI 0.64–0.83), lithium plus zotepine (aHR 0.82, 95% CI 0.72–0.93), lithium plus aripiprazole (aHR 0.87, 95% CI 0.82–0.92) and lithium plus valproate (aHR 0.92, 95% CI 0.87–0.97). By contrast, an increased risk was observed for lithium plus sulpiride (aHR 1.16, 95% CI 1.03–1.30).
Risk of psychiatric hospitalisation associated with combination therapy involving lithium plus another mood stabiliser, or a mood stabiliser plus a major antipsychotic, compared with monotherapy. Adjusted hazard ratios (aHRs) and corresponding 95% confidence intervals are plotted on a logarithmic scale. Exposures are defined as periods of combination therapy involving (a) lithium plus another mood stabiliser compared with periods of lithium monotherapy; and (b) lithium, valproate or lamotrigine plus a commonly prescribed antipsychotic compared with periods of monotherapy with the respective mood stabiliser.

Compared with valproate monotherapy, combination therapy with valproate plus brexpiprazole (aHR 0.79, 95% CI 0.64–0.98), aripiprazole (aHR 0.84, 95% CI 0.78–0.90), olanzapine (aHR 0.89, 95% CI 0.83–0.96), risperidone (aHR 0.90, 95% CI 0.82–0.99) and quetiapine (aHR 0.92, 95% CI 0.86–0.98) was associated with a lower risk of psychiatric hospitalisation.
Compared with lamotrigine monotherapy, a lower risk was observed for lamotrigine plus aripiprazole (aHR 0.79, 95% CI 0.71–0.88) and lamotrigine plus quetiapine (aHR 0.83, 95% CI 0.75–0.92), whereas a higher risk was found for lamotrigine plus chlorpromazine (aHR 1.47, 95% CI 1.19–1.81).
Similar patterns were observed for all-cause hospitalisations across all combination regimens (Supplementary Table 5).
To evaluate the robustness of findings for the primary outcome, we conducted two sensitivity analyses. In the first of these, 5888 patients whose diagnosis transitioned from bipolar disorder to schizophrenia or schizoaffective disorder during follow-up were excluded, and the results were consistent with those of the main analyses (Supplementary Tables 6 and 7). In the second, which omitted the first 30 days following each treatment change, the results were largely consistent although confidence intervals were wider owing to frequent changes in treatment patterns (Supplementary Tables 8 and 9).
Discussion
To our knowledge, this is the largest population-based cohort study to evaluate the real-world effectiveness of mono- and combination therapy with mood stabilisers and antipsychotics in reducing psychiatric hospitalisations among patients with bipolar disorder. Analyses of mood stabiliser and antipsychotic monotherapy revealed substantial heterogeneity in preventive effectiveness. The risk of psychiatric hospitalisation was lower during periods of lithium monotherapy compared with periods of non-use of any mood stabilisers. The order of risk reduction among mood stabilisers – lithium, followed by valproate, lamotrigine and carbamazepine – suggests that lithium had the greatest preventive effectiveness, consistent with prior studies; Reference Lahteenvuo, Tanskanen, Taipale, Hoti, Vattulainen and Vieta7,Reference Lahteenvuo, Paljarvi, Tanskanen, Taipale and Tiihonen8 however, the difference compared with other mood stabilisers was not large (aHR 0.67 v. 0.71, 0.72 and 0.74).
Among antipsychotics the greatest risk reduction was observed with sultopride, followed by paliperidone LAI, aripiprazole LAI, propericiazine and oral aripiprazole. In particular, better outcomes for sultopride, paliperidone LAI and aripiprazole LAI (aHRs 0.53–0.62) than other medications would be a novel finding. Although quetiapine was associated with only modest effectiveness, doses of ≥300 mg/day were more strongly associated with reduced risk. We consider that this dose–response pattern, along with the frequent use of low-dose quetiapine as a hypnotic agent, may have contributed to its overall limited effectiveness in the treatment of bipolar disorder reported in prior studies. Reference Lahteenvuo, Tanskanen, Taipale, Hoti, Vattulainen and Vieta7,Reference Lahteenvuo, Paljarvi, Tanskanen, Taipale and Tiihonen8 Nevertheless, a more recent study reported that quetiapine was not associated with a decreased risk of psychiatric hospitalisation at any dose, but rather increased the risk of relapse at ≥440 mg/day. Reference Lintunen, Hamina, Lahteenvuo, Paljarvi, Tanskanen and Tiihonen27 These inconsistent findings might be explained by a bell-shaped dose–response curve, and underscore the need for further studies.
The preventive effectiveness of monotherapy with certain antipsychotics was either limited (e.g. chlorpromazine) or remained uncertain, with this uncertainty possibly due to insufficient effectiveness (e.g. asenapine, sulpiride) or a small number of users (e.g. fluphenazine, risperidone LAI). These findings raise concerns about the use of specific antipsychotics in the treatment of bipolar disorder, given their limited or unclear effectiveness.
To the best of our knowledge, this is the first observational study to systematically evaluate the additional benefits of combination therapy over monotherapy, despite the widespread use of the former in clinical practice. In our analyses, the risk of psychiatric hospitalisation was lower during periods of lithium combined with carbamazepine, valproate, aripiprazole or zotepine compared with periods of lithium monotherapy. These findings differ from those of a previous study, Reference Wingard, Boden, Brandt, Tiihonen, Tanskanen and Kieler11 which reported that lithium plus olanzapine was the only lithium-based combination more effective than lithium monotherapy. The reason for this discrepancy is unclear; however, our results may be more robust to confounding bias due to the within-individual design used in this study. Additionally, among combinations of mood stabilisers and antipsychotics, aripiprazole consistently provided additional benefits over mood stabiliser monotherapy (i.e. lithium, valproate or lamotrigine). This is partly consistent with a network meta-analysis of RCTs, Reference Kishi, Sakuma, Okuya, Matsuda, Esumi and Hashimoto28 which found that a combination of a mood stabiliser (lithium or valproate) with aripiprazole was more effective than a mood stabiliser alone.
These findings may help guide clinicians in selecting adjunctive agents to use alongside a mood stabiliser – for example, when rigorous control of manic relapse is needed in patients with bipolar I disorder experiencing severe manic episodes, Reference Goes1 or when monotherapy yields an insufficient response. Reference Fornaro, De Berardis, Koshy, Perna, Valchera and Vancampfort29 The results partly support treatment guideline recommendations that favour combination therapy (i.e. a mood stabiliser plus an antipsychotic or two mood stabilisers) over monotherapy for bipolar disorder. Reference Kato, Ogasawara, Motomura, Kato, Tanaka and Takaesu30 This may reflect the distinct mechanisms of action of lithium, other mood stabilisers (valproate, lamotrigine and carbamazepine) and antipsychotics. Reference Kato, Ogasawara, Motomura, Kato, Tanaka and Takaesu30 However, caution is warranted regarding the risk of additional adverse events – such as extrapyramidal symptoms, akathisia, weight gain and prolactin elevation – that may not require hospitalisation but could impair treatment adherence and quality of life.
Moreover, our findings highlight the potential utility of zotepine in the treatment of bipolar disorder – a drug that remains under-recognised internationally – because it was associated with a reduced risk of psychiatric hospitalisation both as monotherapy and in combination with lithium. Although zotepine has been available in Japan since 1982 for the treatment of schizophrenia, and is also approved in a limited number of other countries including South Korea, India and the UK, it has not been approved by major regulatory agencies such as the U.S. Food and Drug Administration or the European Medicines Agency. Despite being classified as a first-generation antipsychotic based on its year of development, zotepine exhibits a pharmacological profile similar to that of second-generation antipsychotics. Reference Harada, Ebara and Otsuki31 However, evidence regarding its comparative efficacy remains limited and inconclusive, even for the treatment of schizophrenia. Reference DeSilva, Fenton and Rathbone32,Reference Subramanian, Rummel-Kluge, Hunger, Schmid, Schwarz and Kissling33 In bipolar disorder, RCTs evaluating zotepine are scarce and therefore our findings provide novel real-world evidence supporting its potential effectiveness in the treatment of bipolar disorder, consistent with an earlier trial that showed its superiority over lithium. Reference Harada, Otsuki, Yamashita, Okumra, Kazamatsuri and Yamaguchi34
One notable discrepancy with prior RCTs is the absence of an additive benefit from combining lithium with lamotrigine, in contrast to trials demonstrating the superiority of this combination for treating bipolar depression. Reference van der Loos, Mulder, Hartong, Blom, Vergouwen and de Keyzer35,Reference van der Loos, Mulder, Hartong, Blom, Vergouwen and van Noorden36 One possible explanation is the difference in outcome measures – psychiatric hospitalisation in our study versus clinician-reported outcomes in RCTs. Manic episodes are more likely to result in hospitalisation whereas depressive episodes may not. Additional explanations include generally lower treatment adherence, longer follow-up periods and greater heterogeneity in observational study populations compared with those in RCTs.
Our study design has several strengths, including a large sample size, high representativeness, a long follow-up period and the use of a within-individual design that minimises confounding by indication. However, several limitations should be acknowledged. First, the design does not adequately account for time-varying covariates such as symptom severity, psychiatric comorbidities or changes in concomitant medications other than psychotropic medications. Second, residual confounding by indication may remain because medications are often initiated during periods of clinical deterioration, potentially inflating the observed risk of outcome events during exposure periods. Third, exposure misclassification may have occurred because prescription records may not accurately reflect actual medication use during the exposure and control periods, whereas therapeutic drug monitoring would be required to objectively ascertain medication adherence. Fourth, transportability is limited because the within-individual design includes only patients who experienced both exposed and control periods and who had outcome events. Fifth, the inclusion criterion requiring at least two lithium prescriptions may further limit the transportability of the findings to broader populations (see Supplementary Discussion), although this restriction may help to increase the specificity of bipolar disorder identification. Sixth, people with lived experience of bipolar disorder were not involved in all phases of this study. Seventh, because the reason for psychiatric hospitalisation cannot be identified in the reimbursement system, we were unable to determine whether any preventive effect was attributable to a manic episode, a depressive episode or other psychiatric causes. Eighth, we did not account for the chronological sequence of exposure and control periods (e.g. first lithium monotherapy, second non-use of any mood stabilisers, third lithium plus valproate) because of the substantial complexity and heterogeneity of treatment patterns. Finally, ethnicity data were not available.
In conclusion, this large, population-based observational study found that both mono- and combination therapy with mood stabilisers and antipsychotics exhibited substantial heterogeneity in their effectiveness for preventing psychiatric hospitalisation among patients with bipolar disorder.
Supplementary material
The supplementary material is available online at https://doi.org/10.1192/bjp.2026.10636
Data availability
The data used in this study are proprietary to the Ministry of Health, Labour and Welfare of Japan, which granted the authors permission to access and analyse them. Due to legal and regulatory restrictions, the data-set used by the authors cannot be shared with third parties. Researchers interested in accessing the data must submit a separate application to the Ministry; access cannot be granted by the authors. The analytic code used to generate the findings of this study is available at https://doi.org/10.17605/OSF.IO/PMBCT. Reproducible code for drugs, procedures and diagnoses is also available at https://doi.org/10.17605/OSF.IO/PMBCT.
Acknowledgements
The authors used ChatGPT-4o (OpenAI) for language editing in July 2025. Y.O. takes responsibility for the integrity of the content.
Author contributions
Y.O. and T.K. had full access to all data in the study, and take responsibility for their integrity and the accuracy of data analysis. Concept and design: Y.O., T.K. Acquisition, analysis or interpretation of data: Y.O., H.T., H.H., T.K. Drafting of manuscript: Y.O., T.K. Revision of manuscript: all authors. Statistical analysis: Y.O. Data access and verification of data: Y.O., T.K. Funding acquisition: T.K. Administrative, technical and material support: all authors. Supervision: T.K.
Funding
This study was supported by the Japan Agency for Medical Research and Development under grant no. JP23dk0307124.
Declaration of interest
For the past 3 years Y.O. has been President of the Initiative for Clinical Epidemiological Research, and is a former employee of Real World Data Co., Ltd. H.T. reports personal fees from Asahi Kasei Co., Ltd, Daiichi Sankyo Co., Ltd, Janssen Pharmaceutical K.K., Meiji Seika Pharma Co., Ltd, Otsuka Pharmaceutical Co., Ltd and Shionogi Co., Ltd, outside the submitted work. H.H. declares no conflicts of interest. T.K. reports grants from Sumitomo Pharma Co., Ltd, Otsuka Pharmaceutical Co., Ltd, Takeda Pharmaceutical Co., Ltd, Eli Lilly Japan K.K., Teijin Pharma, Daiichi Sankyo Co., Ltd, EA Pharma Co., Ltd and Eisai Co., Ltd; and personal fees from Sumitomo Pharma Co., Ltd, Otsuka Pharmaceutical Co., Ltd, Takeda Pharmaceutical Co., Ltd, Eisai Co., Ltd, Meiji Seika Pharma Co., Ltd, Shionogi & Co., Ltd, Mochida Pharmaceutical Co., Janssen Pharmaceutical K.K., Janssen Asia Pacific, Vista Health, Yoshitomiyakuhin, MSD K.K., Japan Boehringer Ingelheim, Kyowa Pharmaceutical Industry Co., Ltd, Viatris, Mylan EPD, H.U. Frontier, Lundbeck Japan K.K., Nihon Medi-physics Co., Ltd, Glaxo-SmithKline, Novartis Pharma, EA Pharma Co., Ono Pharmaceutical Co., Ltd, Ground UP Solutions PTE Ltd and Sumitomo Pharma Asia, outside the submitted work. These companies played no role in study design, data collection and analysis, decision to publish and preparation of the manuscript.






eLetters
No eLetters have been published for this article.