Introduction
Contamination with radionuclides can occur in the context of large-scale incidents or accidents, at workplaces, or purposefully if used as a weapon. Internal decorporation treatment aims to support removal of incorporated radionuclides from the human body,1 i.e. the aim is to reduce absorption or to enhance excretion of radioactive contaminants.1, Reference Guilmette, Bertelli, Miller and Little2 Diethylenetriamine pentaacetate (DTPA) treatment is commonly used in cases of internal contamination with transuranium radionuclides.
DTPA is available in 2 forms: calcium (Ca-DTPA) and zinc (Zn-DTPA). Both forms work by tightly chelating Plutonium (Pu), Americium (Am), and Curium (Cm), with Ca-DTPA considered to act somewhat faster but with more risks for pregnant women.3 These radioactive materials are then passed from the body and excreted in the urine or feces.4 DTPA has been approved by the United States Food and Drug Administration (FDA) since 2004.5 Furthermore, the World Health Organization (WHO) has included DTPA in their policy advice for national stockpiles in radiological and nuclear emergencies.6
Although the drug is used and approved, there is little systematic overview of evidence on the topic so far. In particular, there are questions related to timing of treatment, effectiveness, and side effects of DTPA. For evidence-based guideline development, there is a need to search, map, and analyze existing research using principles of evidence-based medicine. The work supports the development of a new international guideline and recommendation from the WHO, similar to a previous review.Reference Pfinder, Dreger, Christianson, Lhachimi and Zeeb7, 8 The Guideline Development Group (GDG), consisting of clinical experts and researchers in the field of treatment and research in internal radionuclide contamination, is supporting the systematic review process by providing guidance on the theoretical and clinical framework.
The systematic review aims to assess:
-
a. the optimal ways to assess (monitor, measure) internal contamination in individuals and populations exposed to transuranium nuclides due to large-scale nuclear incidents, workplace accidents, or other incidents with contamination potential,
-
b. the effectiveness of treatment of exposed persons receiving chelating agents (DTPAs), and
-
c. the efficacy of treatment of experimental animals receiving chelating agents (DTPAs).
Methods
The systematic review protocol, registered on the Open Science Framework (OSF),Reference Gill, Dreger, Zeeb and Christianson9 covered 2 topics, namely measurement methods and effects of decorporation treatment. Search was developed based on relevant population, intervention, comparator, and outcome, so-called PICO questions. Two PICO questions were developed for each topic through iterative discussions by the GDG and the authors. Details are described in the protocol. PICO 1 and PICO 2 focus on assessment methods, while PICO 3 and PICO 4 consider (comparative) effects of decorporation treatment.
Eligibility Criteria
For the first objective of the review, no limit on study design or publication type was set. As the research questions for this review do not lend themselves to assessment in randomized clinical trials (RCTs), primarily non-randomized studies were included. Controlled animal experimental studies were relevant for the second objective which focused on treatment with chelating agents.Reference Gill, Dreger, Zeeb and Christianson9
Search and Data Extraction
Databases Medical Literature Analysis and Retrieval System Online (MEDLINE), Excerpta Medica Database (EMBASE), and Cochrane Library were searched on December 06, 2023. Search terms for the measurement methods were, among others, “decontamination, internal contamination, radiation monitoring, bioassay, manage, assess.” For treatment, the search terms were, among others, “internal contamination, chelation, DTPA, diethylenetriamine penta.” The complete search terms are described in the appendix (Supplementary Files S1-S6). Search of reports published outside of traditional commercial publishing (e.g. technical reports, recommendations, guidelines, etc.), i.e. gray literature, was performed on the December 24, 2024. Reviewers (HZ, SG) screened titles and abstracts independently using Covidence© software; conflicts were resolved by consensus. Extracted outcomes are described in the protocol.Reference Gill, Dreger, Zeeb and Christianson9 Extracted data included publication details, study characteristics (radionuclide and exposure pathway), details of measurement methods and treatment, and other relevant information. Two reviewers independently performed data extraction. Discrepancies were resolved by discussion.
Study Quality
Risk of bias (RoB) was assessed with the National Toxicology Program—Office of Health Assessment and Translation (OHAT) tool for RoB assessment10 in animal studies and the Joanna Briggs Institute (JBI) critical appraisal tool for human studies.Reference Moola, Munn and Tufanaru11 The OHAT tool assesses the overall experimental conditions and allocation to the experimental groups (performance, selection bias), details on exposure characterization and outcome assessment (detection bias), completeness of outcome data (attrition/exclusion bias, selective reporting bias), and other sources of bias. The JBI tool is used to assess whether a case report provides a clear and detailed description that makes it useful for application in similar clinical conditions. It checks if sufficient detailed information is provided in a timeframe, ranging from patient’s history to reporting of post-interventions clinical conditions and potential adverse effects. Certainty of evidence was assessed with the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach for a narrative synthesis.Reference Murad, Mustafa, Schunemann, Sultan and Santesso12 GRADE rates the overall body of evidence for each outcome by considering methodological limitations of the study design, indirectness, imprecision, inconsistency, and publication bias. It concludes with Summary of Finding (SoF) tables for each outcome. SoF (Supplementary Files S7-S23) were generated according to the GDG’s prioritization rating of outcomes.
Synthesis
Heterogeneity was assessed by systematically going through the reported outcomes based on population and treatment or measurement methods according to the PICO questions. No meta-analysis was performed but a narrative synthesis based on the Cochrane handbook chapter 12.Reference McKenzie, Brennan, Higgins, Chandler, Cumpston, Li, Page and Welch13 The presentation of the results was based on the Synthesis Without Meta-analysis (SWiM) reporting guidelinesReference Campbell, McKenzie and Sowden14 and Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA).Reference Page, McKenzie and Bossuyt15
The measurement of treatment effect as described in the protocol was not possible, because numerous different outcomes and highly variable exposure levels were reported. Therefore, grouping was done according to PICO components: population (human, animal species), type of measurement method, or type of decorporation treatment (Ca-DTPA, Zn-DTPA, Ca-DTPA and Zn-DTPA, other). As far as feasible, outcomes were grouped according to conceptual similarities (Supplementary Files S24 and S25). Because of heterogeneity in outcomes and methods used, no standardized metric or transformation method was feasible. Instead, a harvest plotReference Ogilvie, Fayter and Petticrew16 was generated. Any reduction in the reported outcome in the treatment group (i.e. an effect associated with treatment) compared to the control group was defined as beneficial (any numerical reduction of treatment group compared to control group or reported in-text), no reduction was defined as no effect, and increase or reported side effects as negative.
Deviations from Protocol
During the systematic review, deviations from the protocol were necessary after data extraction. These were made with careful consideration, while maintaining the systematic approach and the review’s objectives.
For the RoB assessment the OHAT10 tool was initially planned to be applied for human and experimental animal studies. After 2 reviewers tested the tool independently, it was found to have no sufficient fit for the assessment in human studies, since these were case reports. Thus, the JBI critical appraisal toolReference Moola, Munn and Tufanaru11 for case reports and case series was tested and applied by the two reviewers independently.
A SoF could not be created with the GRADEpro software as the included studies were too heterogeneous and had multiple estimates of effect that could not be transferred into a single metric. Therefore, the certainty of evidence assessment and the SoF tables were based on the overall GRADE approach and data was summarized narratively.Reference Schünemann, Higgins, Vist, Higgins, Thomas and Chandler17, Reference Murad, Mustafa, Schünemann, Sultan and Santesso18
The assessment of reporting bias and sensitivity analyses could not be performed with funnel plots as outcomes were predominantly descriptive.
Results
Study Characteristics
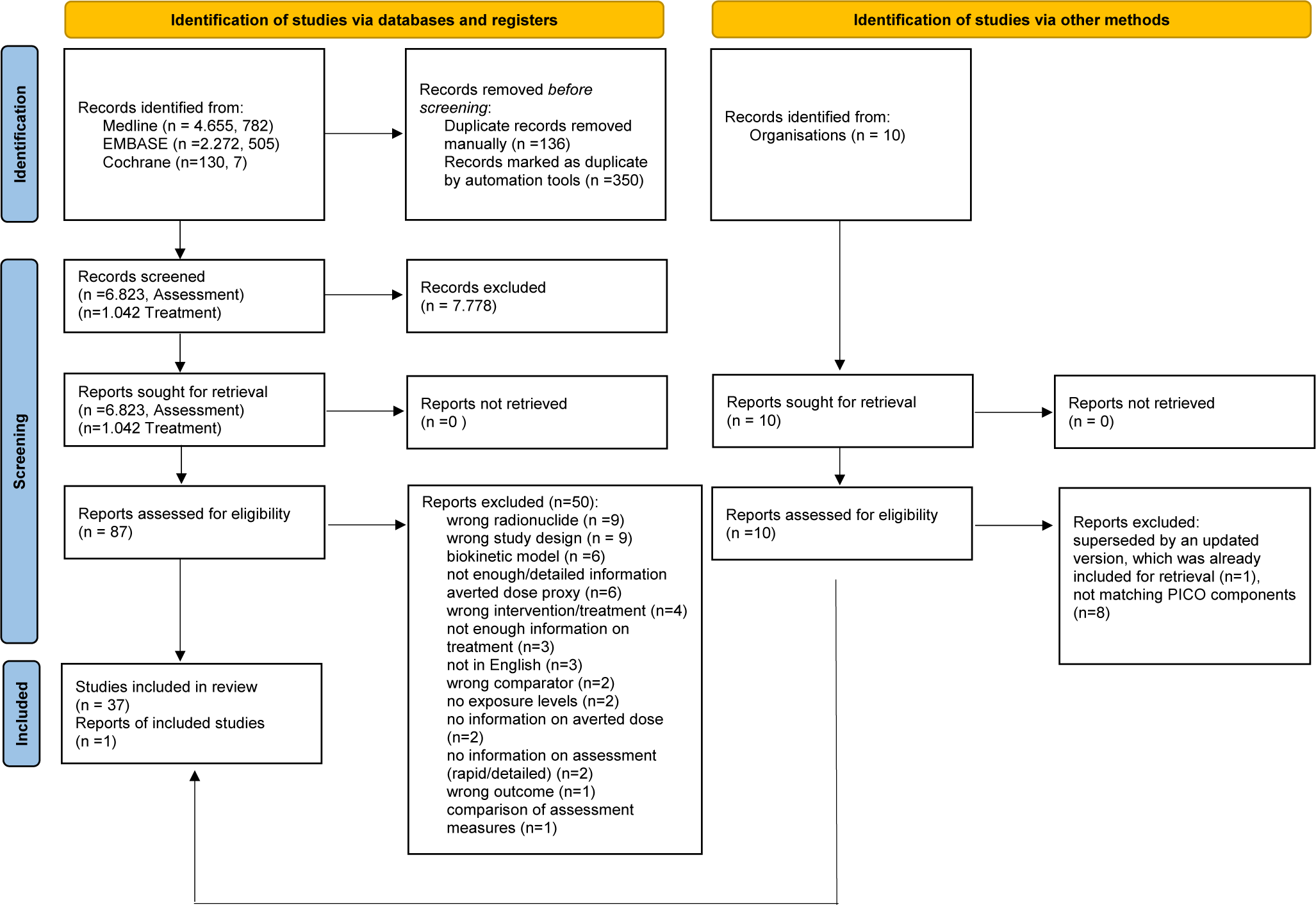
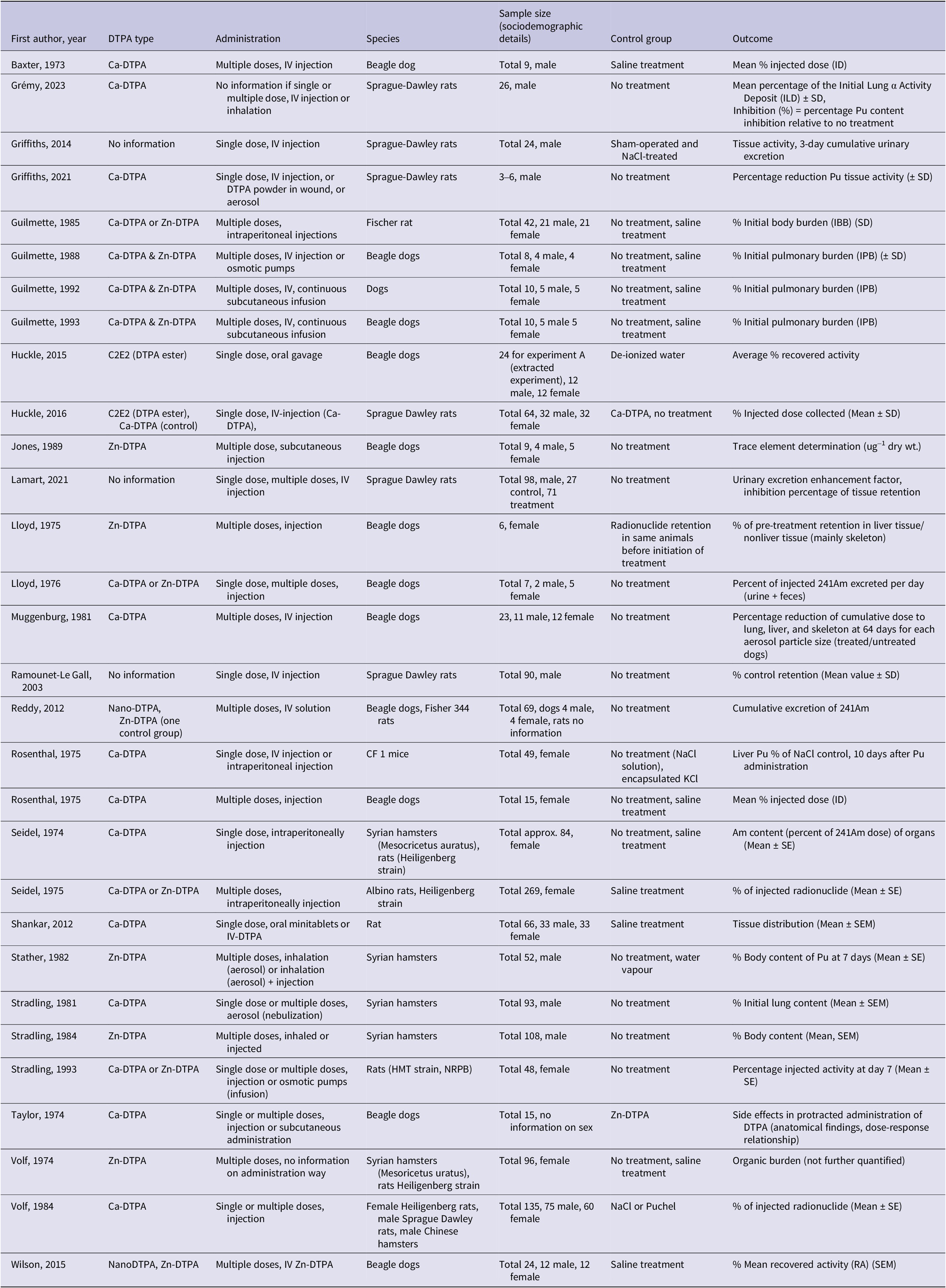
In total, 37 studies were included (Figure 1), of which 7 were human studies (case reports or case series) and 30 controlled experimental animal studies. All human studies reported workplace accidents (n = 12). The number of animals ranged from 3 to 269 in their designs. The species studied were dog (n = 13), rat (n = 11), mouse (n = 1), or hamster (n = 5).
PRISMA flowchart of study selection.Reference Page, McKenzie and Bossuyt15

The distribution of DTPA types was the following: Ca-DTPA (n = 14), Zn-DTPA (n = 7), Ca-DTPA or Zn-DTPA (n = 6), combined Ca-DTPA and Zn-DTPA (n = 5), no information (n = 4), and C2E2 (DTPA ester drug) (n = 1). Two of the studies relevant for the PICOs 3 and 4 were also applicable for the PICOs 1 and 2. Therefore, these 2 studiesReference Carbaugh, Lynch, Cannon and Lewis19, Reference Sugarman, Findley, Toohey and Dainiak20 appear twice in the data extraction. Results are presented according to the DTPA types, as the efficacy of treatment could differ among the groups. Results from the group “combined” are presented in text for the group Zn-DTPA, as the treatment was continued for a longer period with Zn-DTPA compared to Ca-DTPA in the respective studies. Studies in the group “Ca or Zn” are presented either for Ca-DTPA or Zn-DTPA, depending on the treatment schedules. Four studiesReference Carbaugh, Lynch, Cannon and Lewis19, Reference Bertelli, Waters, Miller, Gadd, Eaton and Guilmette21–Reference Guilmette and Muggenberg23 administered either Ca or Zn-DTPA to different participants within the study. The results for each DTPA type are presented separately in the text but in the tables and figures, the data is still grouped under their broader category (Supplementary File S26). The group “no information” and “C2E2” are presented combined under “other grouping” in the text.
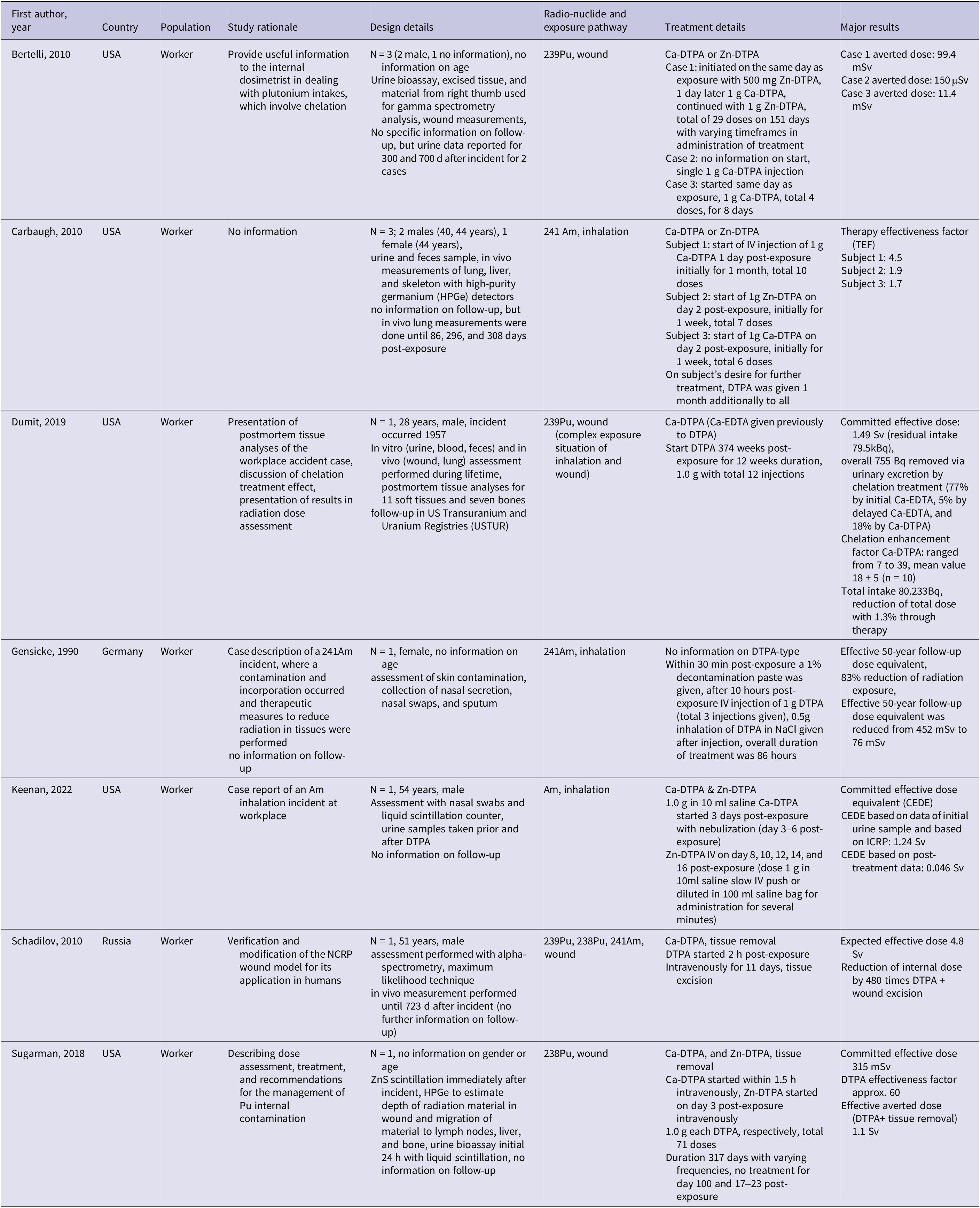
Two studies reported the treatment effect with the priority outcome averted doseReference Sugarman, Findley, Toohey and Dainiak20, Reference Bertelli, Waters, Miller, Gadd, Eaton and Guilmette21 (Table 1). Side effects were reported in 1 studyReference Taylor, Williams, Roberts, Atherton and Shabestari24 and 1 report25. In discussion with the GDG the authors decided to use exposure level as a surrogate outcome for averted dose. The majority of included studies presented their results with exposure levels as an outcome (n = 28 animal studies). Most human studies reported outcomes related to effective dose (n = 6). No clinical outcomes such as cancer incidence or mortality were reported in human studies.
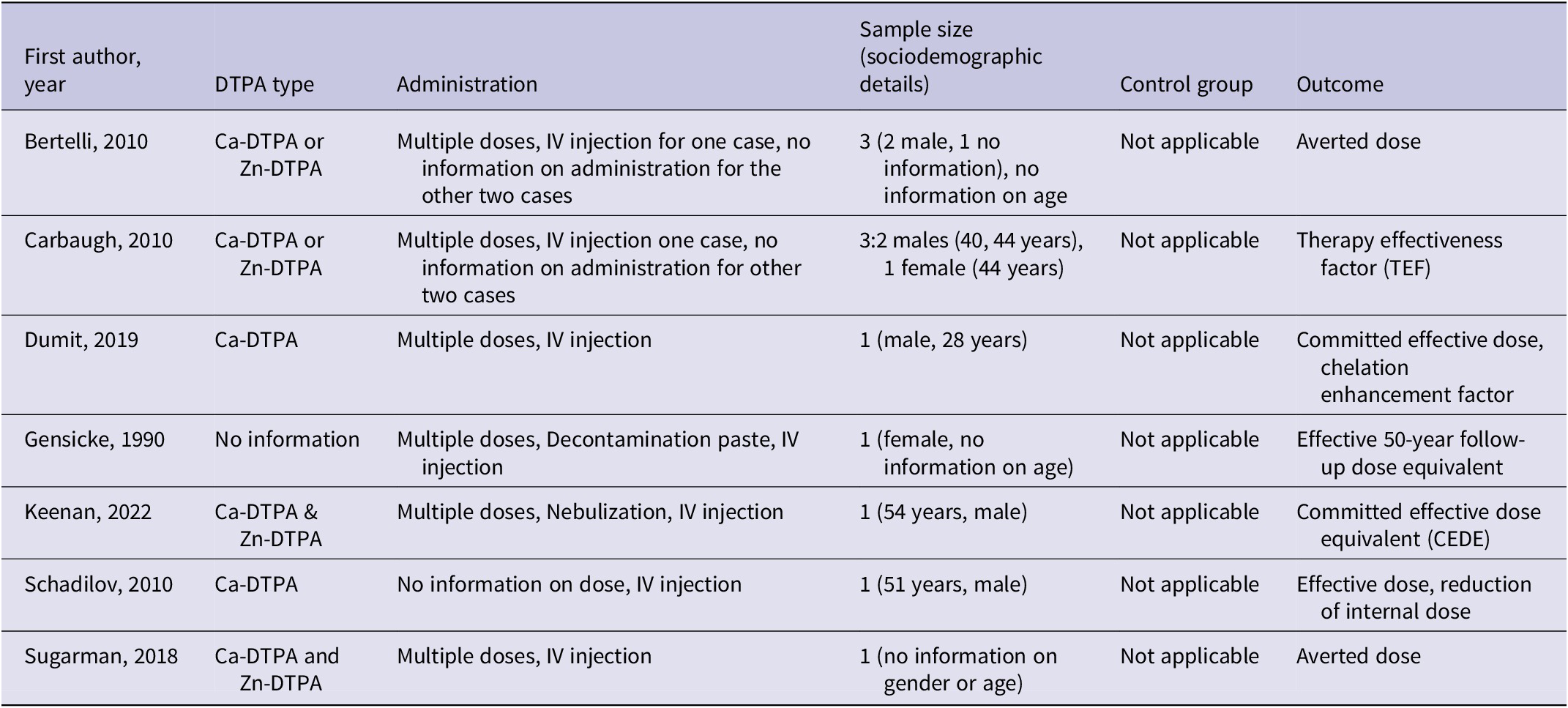
Characteristics of included human studies in DTPA treatment (PICO 3)

Measurement Methods
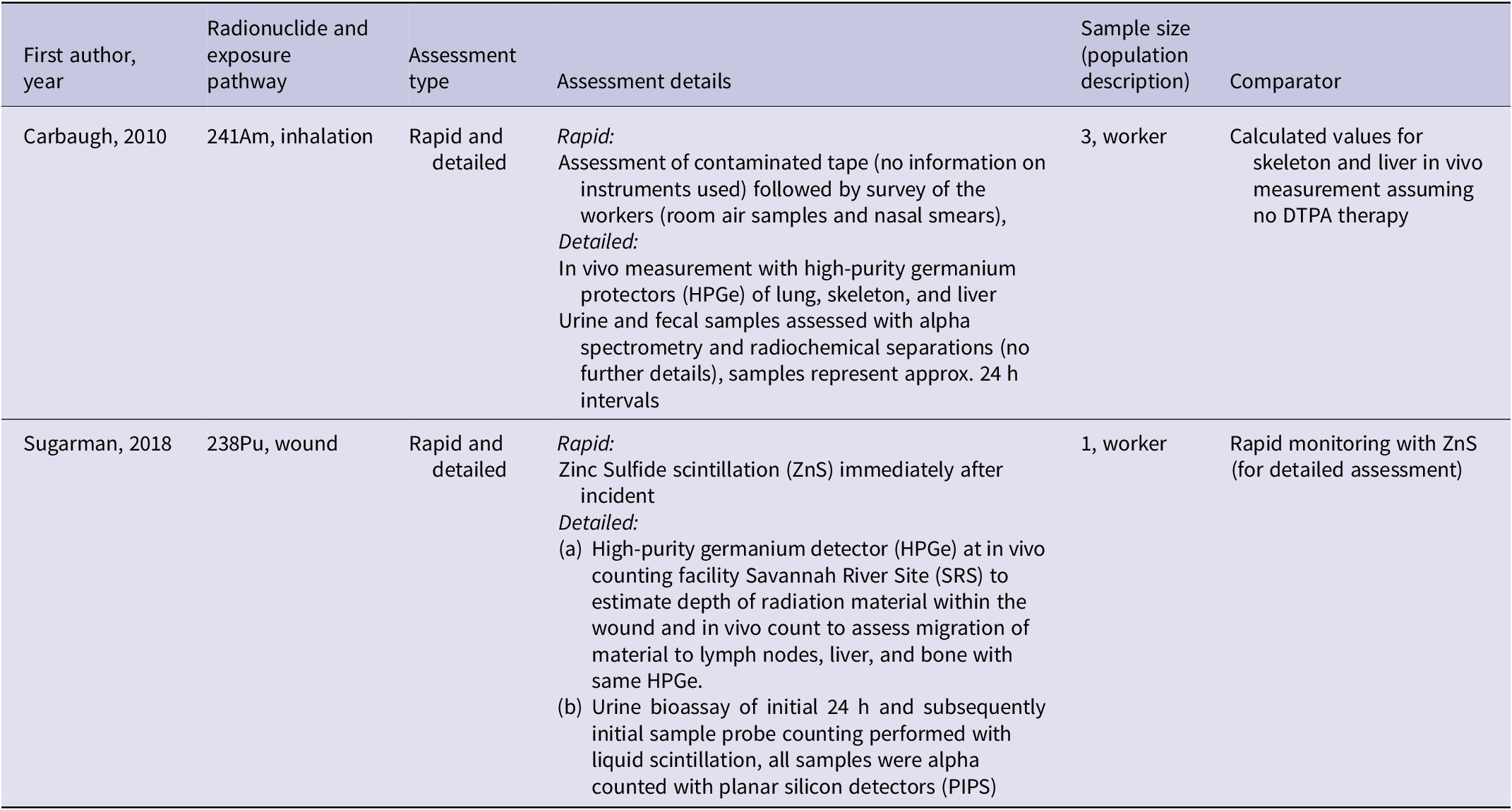
Of the 37 included studies, 2Reference Carbaugh, Lynch, Cannon and Lewis19, Reference Sugarman, Findley, Toohey and Dainiak20 described rapid and detailed assessment methods to allow an estimation of contamination magnitude (Table 2).
Characteristics of included studies for assessment methods (PICO 1 and PICO 2)

Note: Present studies also included in data extraction for PICO3 on DTPA treatment in human studies
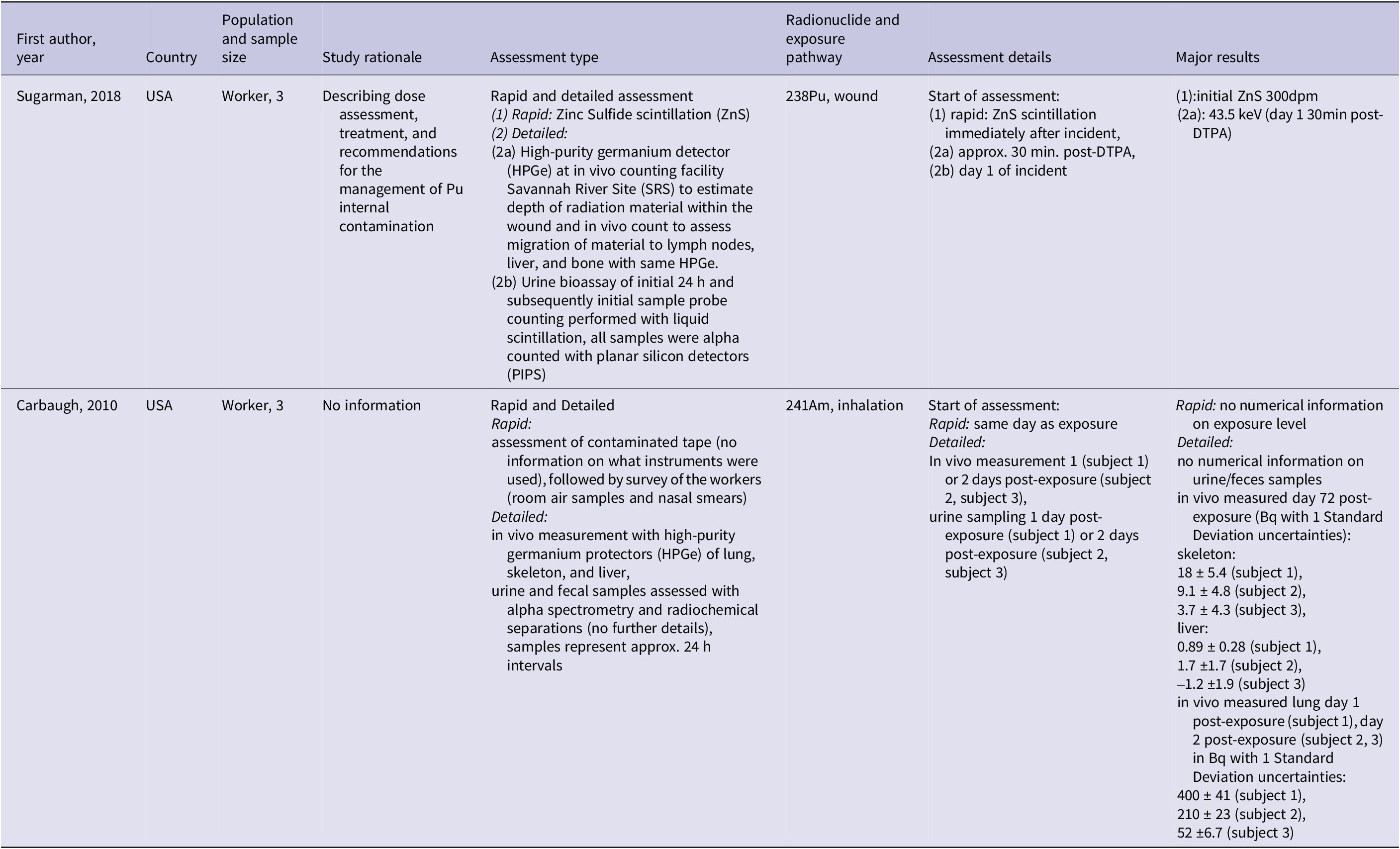
In a case reportReference Sugarman, Findley, Toohey and Dainiak20 a worker was rapidly assessed after a 238Pu wound contamination using Zinc Sulfide (ZnS) scintillation. A detailed assessment with a high-purity germanium detector (HPGe) and urine bioassay was performed afterward. HPGe detected 43.5 keV at 30 min post-DTPA on day 1 post-exposure. The initial ZnS-based count underestimated the contamination and the HPGe results guided further treatment, including tissue removal. No information on the quantity of the exposure level was available, except for information on the activity reduced for day 1 (24,500 dpm), day 3 (1,398 dpm), and day 12 (434 dpm) post-exposure based on urine samples. Although estimated exposure levels were reported, bioassay range data were unavailable.
A case seriesReference Carbaugh, Lynch, Cannon and Lewis19 examined 241Am inhalation contamination in three workers. Rapid assessment included contaminated tape analysis, room air samples, and nasal smears but lacked numerical exposure data. Detailed assessment involved in vivo HPGe measurements of the lung, skeleton, and liver, along with urine and fecal alpha spectrometry. The in vivo measurement results were described for day 72 post-exposure and after DTPA treatment, ranging from 3.7 to 18 Bq for the skeleton (Table 3). The rapid assessment identified the case with the highest contamination, warranting in vivo measurement the next day. Chelation therapy was initiated based on the in vivo results, starting on day 1 for subject 1 and day 2 for subjects 2 and 3.
Key findings and characteristics of included studies for assessment methods (PICO 1 and PICO 2)

Note: Present studies also included in treatment data extraction (PICO 3).
CA-DTPA
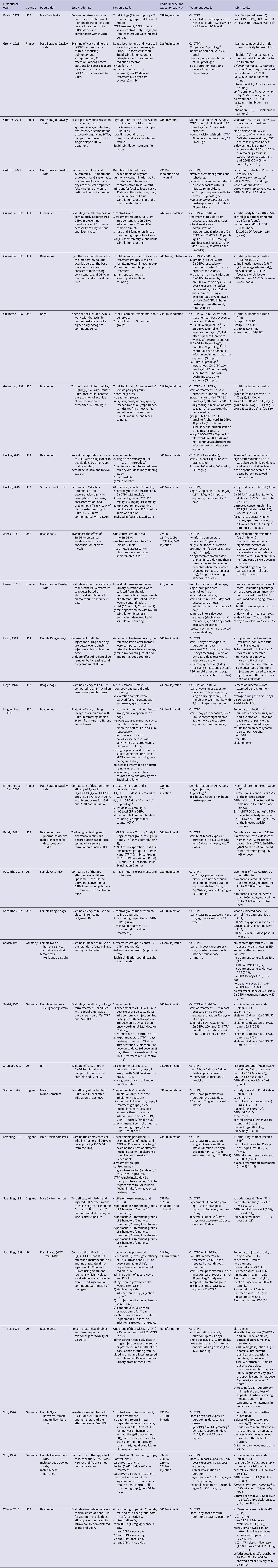
Four human studiesReference Carbaugh, Lynch, Cannon and Lewis19, Reference Bertelli, Waters, Miller, Gadd, Eaton and Guilmette21, Reference Dumit, Avtandilashvili and Tolmachev26, Reference Schadilov, Belosokhov and Levina27and 15 animal studiesReference Seidel22–Reference Taylor, Williams, Roberts, Atherton and Shabestari24, Reference Baxter, Rosenthal and Lindenbaum28–Reference Lloyd, Mays, McFarland, Taylor and Atherton39 administered Ca-DTPA, and 4 of these studies also described results for Zn-DTPA.Reference Carbaugh, Lynch, Cannon and Lewis19, Reference Bertelli, Waters, Miller, Gadd, Eaton and Guilmette21–Reference Guilmette and Muggenberg23 Outcomes were averted dose and generally some measure of dose in relation to the initially injectedReference Baxter, Rosenthal and Lindenbaum28–Reference Volf and Peter31 or inhaled dose.Reference Gremy, Devilliers and Miccoli32, Reference Stradling, Stather, Ham and Sumner33 Further detailed characteristics (treatment start, duration, number of treatments, and isotope) and results of the treatment are described in Tables 1, 4, 5, and 6. Grouping categories of the outcomes are described in the appendix (Supplementary Files S24 and 25).
Characteristics of included animal studies in DTPA treatment (PICO 4)

Key findings and characteristics of included human studies for DTPA treatment (PICO 3)

In humans, Ca-DTPA was administered with multiple intravenous (IV) injections and a small positive treatment effect was observed, though different outcomes were used and magnitude of treatment effect varied, among others, depending on the complexity and severity of the internal contamination and its management. The first studyReference Dumit, Avtandilashvili and Tolmachev26 reported a committed effective dose of 1.49 Sv (residual intake 79.5 kBq), revealing that only a very small proportion (755 Bq) was excreted due to chelation treatment. In the second studyReference Schadilov, Belosokhov and Levina27 an expected effective dose (over 50 y) of 4.8 Sv was reported. Within 10 days after the exposure approximately 200 Bq of 239Pu, 4 Bq of 238Pu, and 40 Bq of 241Am were excreted in urine. The third studyReference Bertelli, Waters, Miller, Gadd, Eaton and Guilmette21 reported an averted dose varying between 99.4 mSv (case 1) and 11.4 mSv (case 3) or 150 μSv (case 2). A further study on three wound contamination casesReference Carbaugh, Lynch, Cannon and Lewis19 calculated the Therapy Effectiveness Factor (TEF) to range between 1.7 and 4.5, indicating effective removal of dose varying with frequency and number of DTPA treatments. The authors described the concept of TEF as based on the reference organ or radiation content without therapy relative to the observed organ or tissue content with therapy. The authors used the skeleton as the reference for the TEF calculation.
In experimental animal studies with an outcome related to injected radionuclide dose, treatment was administered via injection with multiple DTPA doses in 5 studiesReference Seidel22, Reference Baxter, Rosenthal and Lindenbaum28, Reference Rosenthal, Lindenbaum, Baxter, Kalesperis, Moretti and Russell29, Reference Volf and Peter31, Reference Lloyd, Mays, McFarland, Taylor and Atherton39 and in 1 study with a single doseReference Huckle, Sadgrove and Leed30. The first studyReference Baxter, Rosenthal and Lindenbaum28 reported that in liver the mean % injected dose in μCi (ID) was 1.19 after treatment against 30.9 in controls. The second studyReference Rosenthal, Lindenbaum, Baxter, Kalesperis, Moretti and Russell29 reported a mean % ID after 90 days of Pu exposure with 77.8% in liver for treated and 92.2% in liver for untreated animals. The third studyReference Volf and Peter31 showed lower values of the injected radionuclide (% mean ± standard error (SE)) in rats with 48.3% (3.6) in skeleton and 3.7% (0.8) in liver compared to the control group with 62.8% (4.5) in skeleton and 10.1% (1.2) in liver. Treated hamsters showed 22.3% (0.9) in skeleton and 8.6% (0.4) injected dose in liver compared to the control group with 33.3% (2.4) in skeleton and 41.6% (1.1) in liver. A study in dogsReference Lloyd, Mays, McFarland, Taylor and Atherton39 reported the average excreted percent of injected 241Am during the first 3 days for Ca-DTPA with 6.5% and for Zn-DTPA with 14.8%. In a study comparing early (1.5 min post-exposure) and delayed (4 days post-exposure) treatment, differences between Ca-DTPA and Zn-DTPA were observed in % of injected radionuclide dose (mean ± SE) for skeleton.Reference Seidel22 Ca-DTPA treatment resulted in a lower % remaining dose compared to Zn-DTPA (2.79 vs 5.09) and both treatments showed a similar value in late treatment (11.7 vs. 11.3% of injected dose). For the study with the single dose administration, treated animals showed lower values of injected % dose in liver (6.1 ± 0.7), skeleton (12 ± 2.0), and at wound site (22.4±7.4) compared to the untreated animals with 17.2 (2.8) in liver, 26 (2.0) in skeleton, and 35.1 (5.7) at wound site.Reference Huckle, Sadgrove and Leed30 The last studyReference Stradling, Gray and Moody40 described a reduced percentage of injected activity (mean ± standard error (SE)) with 9.1 (0.9) for Pu and 8.3 (0.7) for Am in treated animals compared to the control with 23.9 (1.5). This study compared the efficacies of the linear hydroxypyridinone-based ligand 3,4,3-Li(1,2-HOPO) and DTPA after different routes of administration and both were effective after a local administration (Table 6).
Key findings and characteristics of included animal studies for DTPA treatment (PICO 4)

Of 2 animal studies on the outcome lung content/deposition,Reference Gremy, Devilliers and Miccoli32, Reference Stradling, Stather, Ham and Sumner33 the first described the mean percentage of the Initial Lung α Activity Deposit (ILD) ± standard deviation (SD) for early treatment (1 hour post-exposure) or delayed treatment (14 days post-exposure).Reference Gremy, Devilliers and Miccoli32 ILD ± SD in kBq in delayed treatment was 8.6% (2.2) for IV injection, 8.1% (2.0) for inhalation against 17.0% (2.8) in the control group. Results for the early treatment were 3.0% (0.7) for IV injection, 1.9% (0.3) for inhalation, and 11.4% (2.0) for the control group. The second studyReference Stradling, Stather, Ham and Sumner33 nebulized DTPA in a single dose or multiple doses. Hamsters receiving multiple doses showed 7.9% (0.9) initial lung content compared to control animals with 30.5% (0.7).
Five animal studies reported distinct outcomes beyond those of other included studies. One study compared liposome-encapsulated vs. conventional Ca-DTPA in mice, showing liver Pu dose reduction to 60.2% (injected dose 500 mg/kg) and 56.8% (injected dose 1000 mg/kg) of control levels.Reference Rosenthal, Rahman, Moretti and Cerny35 A study comparing oral mini Ca-DTPA tablets to IV-Ca-DTPA and untreated controls reported a kidney retention in mean and SE of 1.67 ± 0.05 nCi (IV), 1.94 ± 0.08 nCi (oral), and 2.30 ± 0.14 nCi (control).Reference Shankar, Weber, Doyle-Eisele and Guilmette36 A study in hamsters and rats showed lower Am content in Ca-DTPA-treated groups across organs.Reference Seidel37 Finally, a combination of lung lavage and Ca-DTPA in dogs resulted in reduced reduction of cumulative dose for the mean aerodynamic aerosol particle size (50% in lungs, 90% in liver, 85% in skeleton).Reference Muggenburg and Mewhinney38 One study reported the percentage reduction of Pu tissue activity (± SD).Reference Griffiths, Van der Meeren and Gremy34 A single dose was administered but in different administration ways (IV injection, DTPA powder in wound, or aerosol) and treatment (Table 6). In pulmonary contaminated rats, treated animals had a reduction of 21% (SD 7) in the lung. In wound-contaminated rats a reduction of 56% (SD 10) in skeleton and 80% (SD 3) in the liver was reported. Another study in rats used treatment either through discrete administration with intraperitoneal injection (Ca-DTPA or Zn-DTPA) or through continuous administration (Zn-DTPA).Reference Guilmette and Muggenberg23 Results were presented in % initial body burden (IBB) (SD). In bone the IBB for the continuous Zn-DTPA was 0.082% (0.036) and for the injected Ca-DTPA 0.16% (1.4). Another study in dogs examined side effects and reported that the highest toxicity, given the specific condition, was reached at dose 6 μmols/kg given every 5 hours.Reference Taylor, Williams, Roberts, Atherton and Shabestari24 In the report identified from the hand search25 information on the occurrence of adverse incidents was documented for 310 out of a total of 646 human individuals treated with at least 1 dose of either Ca- or Zn-DTPA. No information on administered dose was available. Of the 310 individuals, 19 reported at least 1 adverse incident. In total, 20 adverse incidents were reported. One individual treated with Zn-DTPA reported headache, light-headiness, and pelvic pain. Two individuals treated with nebulized Ca-DTPA reported cough and/or wheezing.
ZN-DTPA
A total of 12 animal studies and 2 human studies considered Zn-DTPA treatment in multiple doses. Outcomes were grouped as body content,Reference Stradling, Stather, Sumner, Strong, Lennox and Ham41, Reference Stather, Stradling and Smith42 excretion,Reference Reddy, Cobb and Dungan43 activity,Reference Wilson, Cobb and Dungan44 retention,Reference Lloyd, Mc Farland, Taylor, Williams and Mays45 and other.Reference Jones, Mays and Taylor46, Reference Volf and Seidel47 Zn-DTPA was either given solelyReference Stradling, Stather, Sumner, Strong, Lennox and Ham41–Reference Volf and Seidel47 or for continued treatment after initiation with Ca-DTPA.Reference Seidel22, Reference Guilmette and Muggenberg23, Reference Guilmette and Muggenburg48–Reference Guilmette and Muggenburg50 All outcomes reported a reduction in exposure level compared to the control group but with varying magnitudes depending on the statistical method used and treatment administration (e.g. IV, inhaled, infusionReference Seidel22, doseReference Guilmette and Muggenburg49, start of treatmentReference Seidel22).
The first human case reportReference Sugarman, Findley, Toohey and Dainiak20 described a workplace incident with initial Ca-DTPA and continued Zn-DTPA and tissue removal. A committed effective dose of 315 mSv was reported and an effective averted dose for the combined treatment (DTPA + tissue removal) as 1.1 Sv. The second case reportReference Keenan, Behrens and Bravenec51 administered a combined treatment of initiation with Ca-DTPA and continuation with Zn-DTPA. Committed effective dose equivalent (CEDE) was calculated once based on data of initial urine sample and calculated based on ICRP Publication (66 and 67) resulting in 1.24 Sv. CEDE based on post-treatment data was 0.046 Sv.
In animal studies reduced exposure levels compared to controls were reported for outcomes related to % body contentReference Guilmette and Muggenberg23, Reference Stradling, Stather, Sumner, Strong, Lennox and Ham41, Reference Stather, Stradling and Smith42 with injected DTPA treatment showing a mean of 2.2% body content in liver and a mean of 4.4% after DTPA administration by inhalation.Reference Stradling, Stather, Sumner, Strong, Lennox and Ham41
A new formulation of DTPA (NanoDTPA) was tested in 2 animal studies.Reference Reddy, Cobb and Dungan43, Reference Wilson, Cobb and Dungan44 Am excretion in urine and feces was higher in DTPA-treated groups (NanoDTPA or Zn-DTPA) (70-90% of injected dose) compared to untreated group (30-40% of injected dose)Reference Reddy, Cobb and Dungan43.
For the outcome trace element depletion rate one long-term study in beagle dogs reported the trace element determination in μg g−1 dry weight.Reference Jones, Mays and Taylor46 Treatment was given for 10 years and in liver and bone tissue no significant increases or decreases (P >.05) of trace metal concentration in treated (30 μmol Zn-DTPA kg−1) and control dogs were seen. For treatment and other details see Table 6.
Three animal studies reported a reduced % initial pulmonary burden compared to the untreated control.Reference Guilmette and Muggenberg23, Reference Guilmette and Muggenburg48, Reference Guilmette and Muggenburg49 Infused DTPA led to an average whole body initial pulmonary burden (IPB) of 8.2% vs. 76.7% in control.Reference Guilmette and Muggenburg48 IPB described the quantity of pulmonary deposited radionuclide. And the average whole body IPB described the average amount of total radionuclide retained in the body shortly after exposure. The second study showed varying % IPB (3.0-11%) depending on the administered dose compared to 86% in the control.Reference Guilmette and Muggenburg49 The third study reported 72% of IPB in control compared to 12-19% in treated animals again based on different administration pathways and dosesReference Guilmette and Muggenburg50 (Table 6).
Other Grouping
One study examined the decorporation efficacy of C2E2 (DTPA ester drug) and 4 studies (1 human, 3 animal) had no information on DTPA type. The first studyReference Huckle, Sadgrove and Pacyniak52 observed a significant reduction in recovered activity in tissue for liver, kidney, and lung. The human case studyReference Gensicke, Stopp and Scheler53 reported a reduction in the effective 50-year follow-up dose equivalent from 452 mSv to 76 mSv. One animal studyReference Griffiths, Coudert, Wilk, Renault, Angulo and Van der Meeren54 reported that a single delayed DTPA administration led to a 73% decrease in activity in liver, 30% in kidney, and 35% in lymph nodes. The second animal study evaluated and compared the efficacy of different DTPA treatment schedules based on a statistical simulation of animal wound experimental data.Reference Lamart, Van der Meeren, Coudert, Baglan and Griffiths55 Authors reported that the inhibition percentage of tissue retention at day 7 post-exposure ranged from −40% to 80% in kidneys, liver, and in skeleton. The last animal study aimed at comparing the decorporation efficacy of hydroxypyridinone (HOPO) with DTPA.Reference Ramounet-Le Gall, Grillon, Rateau, Burgada, Bailly and Fritsch56 In DTPA-treated animals 34.6% of the injected activity remained in liver, bone, and kidneys. In HOPO-treated animals 6.0% or less of the injected activity remained.
Study Heterogeneity
The heterogeneity of the studies relates to different domains: clinical and methodological diversity and wide variations in the outcomes, with some outcomes reported in a small number of studies. Only two studies reported the priority outcome defined for this review (averted dose). Although we grouped dose level of internal contamination after treatment as surrogate outcome for averted dose following GDG consultation, these surrogate outcomes do not directly address the PICO questions. Additionally, study characteristics like species and type of experimental design were very diverse (Table 4).
Certainty of Evidence and Potential Bias
Overall, the certainty of evidence is very low (Supplementary Files S8, S20, and S23), primarily because of serious inconsistency resulting from different magnitudes of the outcome, serious indirectness resulting from the high heterogeneity of the PICO characteristics across the studies and various measures being used as a surrogate outcome for averted dose, and serious risk of bias in many studies. Most human studies (6/7) clearly reported assessment methods and results, treatment details (5/7), and takeaway lessons (5/7). However, all lacked clear description of the patient’s condition at presentation (7/7), with unclear patient history (5/7) and post-intervention outcome (6/7). Overall, the 30 animal studies were judged as “probably high risk of bias.” In particular, the majority of studies showed selection and performance bias. Two studies were assessed as having low risk of bias.Reference Huckle, Sadgrove and Leed30, Reference Wilson, Cobb and Dungan44
Treatment Effect
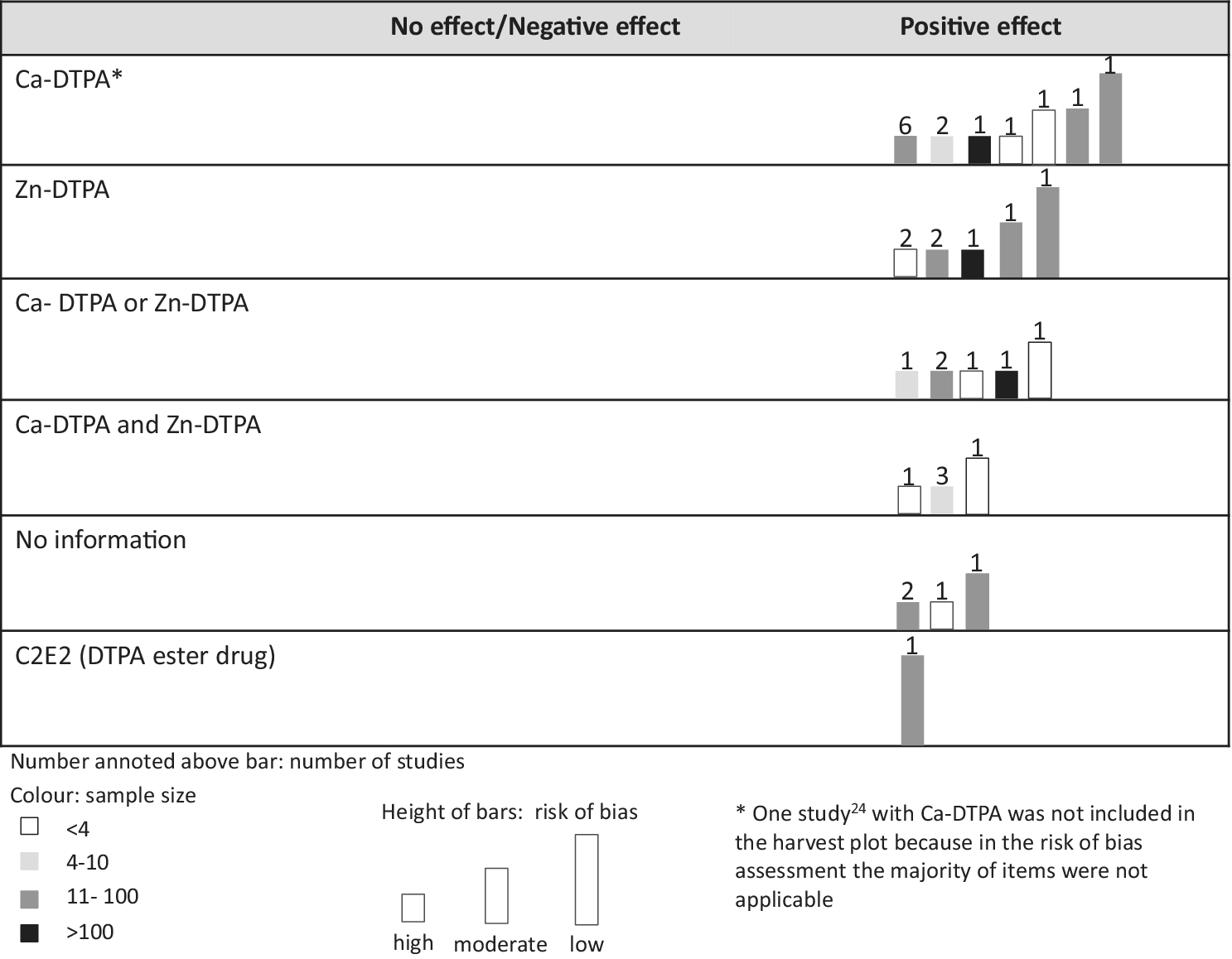
The harvest plot illustrates that all DTPA types showed a positive treatment effect compared to the control group (Figure 2, Supplementary File S27). One studyReference Taylor, Williams, Roberts, Atherton and Shabestari24 was not included in the harvest plot because, in the RoB assessment, the majority of items were not applicable.
Harvest plot of evidence for DTPA treatment effect by DTPA type.

Discussion
Overall, 37 studies (30 animal studies, 7 human studies) and 1 report were included. Only 2 studies reported the priority outcome averted dose. For the 2 PICOs regarding assessment methods, 2 studies described that detailed assessment influenced the overall treatment plan. The rapid assessment underestimated the initial contamination in 1 study and in the other rapid assessment was used to prioritize the timing of detailed assessment. In human studies on DTPA treatment effects 2 studies reported averted dose for 4 persons (99.4 mSv, 150 μSv, 11.4 mSv, and 1.1 Sv). The averted dose varied substantially, depending on the specific exposure circumstances and time periods considered. No other outcomes, in particular no evidence on the occurrence of cancer or other non-communicable diseases, could be judged based on the available primary scientific literature on use of DTPA in humans.
Most scientific evidence was available for the DTPA treatment effect in animal studies, but many papers were outdated. Diverse outcomes and heterogeneity in treatment precluded a meta-analysis. Studies generally included a non-treated control group. As seen in the harvest plot all studies indicated efficacy in reducing the exposure level of internal contamination after treatment. However, there were large differences with respect to sample size and other conditions like administration pathway or dose. Side effects in experimental animal studies regarding DTPA toxicity for Ca-DTPA and Zn-DTPA were anorexia, emesis, diarrhea, melena, and hematuria, with very limited anecdotal mentioning of potential side effects in the few human studies. A dose-response relationship for Ca-DTPA was observed, with highest toxicity given the specific condition reached at dose 6 μmols/kg given every 5 hours. Overall, most studies showed a probably high risk of bias and the certainty of the evidence was judged as very low.
As this is the first systematic review on assessment methods and DTPA treatment after internal contamination with transuranium radionuclides, no other systematic reviews are available for result comparison. However, narrative and non-systematic reviews on radiation emergency management and preparedness are available and generally recommend the use of DTPA.Reference Kazzi, Heyl and Ruprecht57–Reference Breitenstein, Fry, Lushbaugh, Ricks and Fry59 One paper reviewed different chelating agents in actinides and reported a time-dependent effect based on the level of incorporated plutonium in the body.Reference Durbin, Kullgren, Xu and Raymond60 As long as plutonium is available in blood and extracellular fluids or loosely bound to skeletal surfaces of soft tissues, treatment can be effective.Reference Durbin, Kullgren, Xu and Raymond60 A simulation study based on biokinetic models for 239Pu, 241Am, and 137Cesium calculated the efficacy of DTPA treatment.Reference Rump, Stricklin, Lamkowski, Eder, Abend and Port61 Initiation and duration of treatment were considered as well as invasion pathways and physicochemical properties of the inhaled radionuclides. It was concluded that efficacy is less for 241Am or 137Cesium than for 239Pu, even when treatment started early. Treatment duration of up to 90 days showed an increase in efficacy for plutonium and cesium. In americium, continuation after that time might enhance the efficacy of treatment. Another review on delivery systems to increase the local concentration of chelating agents described DTPA and HOPOs as the best candidates for the practice.Reference Fattal, Tsapis and Phan62 Another study reviewed workplace accident cases that were treated with DTPA and described a positive treatment effect after Pu or Am contamination.Reference Grappin, Berard and Menetrier63
To strengthen the evidence for DTPA or other chelating treatments, systematically collected evidence from all cases undergoing treatment, with a set of harmonized indicators, including suitable endpoints, is relevant. This seems to be the best possible option given the rare occurrence of emergency situations and the impossibility to obtain RCT or cohort study evidence.
Highest quality and reporting standards for animal studies should be applied to reduce the risk of bias and strengthen the evidence. Details on the randomization procedure as well as sample size calculations were frequently missing, impeding research and/or reporting quality. This should be improved in future studies.
Although standardized protocols for rapid and delayed assessment of radiation dose are already developed1, 25, 64, 65 none of the included human studies reported using these protocols. Future research could investigate the question of how the current protocols are being used and whether they are guiding the clinical practice. Some studies investigated new agents including HOPO,Reference Gremy, Devilliers and Miccoli32, Reference Fattal, Tsapis and Phan62, Reference An, Kullgren, Jarvis and Abergel66 which may show better efficacy. More high-quality research on HOPO preparations seems warranted.
Limitations
The synthesis was limited by the availability of human studies, which were mostly case reports or small case series, leading to very low certainty evidence. We decided against specifying outcomes to maximize inclusivity, given the limited evidence.
Animal studies provided more extensive data, but their heterogeneity—variations in species, exposure routes, dosing regimens, and outcome measures—posed challenges for synthesis. Synthesizing based on each study’s specific aim and corresponding PICO elements may have resulted in the omission of important contextual details relevant for interpretation. Incomplete reporting, lack of standardized methodologies, and high risk of bias reduced the certainty of evidence. While these studies support DTPA’s effectiveness in reducing radionuclide burden, their generalizability to human exposures, especially regarding long-term outcomes, remains complex and uncertain.
Publication bias could not be formally assessed, as many studies lacked standardized reporting, but the inclusion of diverse study conditions suggests no systematic exclusion of findings. Much of the literature was published decades ago, and not always in the scientific literature which increases the likelihood that such evidence was not included in the current review. In addition, the limited number of included databases and incomplete gray literature screening may have led to unidentified studies, though independent study selection by 2 reviewers aimed to mitigate this risk.
Conclusions
Whether rapid dose assessment contributes to timely treatment decisions could not be assessed with the limited amount of informative studies available for this topic, even though there are anecdotal indications that rapid assessment can influence treatment decisions.
DTPA treatment following contamination with transuranium nuclides seems to be effective in terms of reducing radiation burden and other proximal outcomes in animal experimental studies, but the certainty of the evidence is very low. Evidence for humans is based on a small number of case studies in this review where reductions in body burden have been reported, but no control conditions were assessed. Given these finding, as well as evidence from case reports and gray literature, treatment with DTPA after incorporation of transuranium radionuclides appears to be reasonable from a practical perspective.
Supplementary material
The supplementary material for this article can be http://doi.org/10.1017/dmp.2025.10224.
Funding statement
The work was funded by the World Health Organization (WHO) and was part of a guideline development process.
Author contribution
Sehajpreet Gill (SG) wrote the first draft of the manuscript and performed the screening, data extraction, and analyses of results with Hajo Zeeb (HZ). Lara Christianson (LC) developed the search strategy with HZ and SG. LC performed the search. Steffen Dreger, SG, and HZ wrote the protocol for the systematic review. All authors have contributed to the final draft of the manuscript.
Competing interests
None.