Introduction
Cardiovascular MRI with magnetic resonance angiography is commonly used to support clinical indications for pulmonary valve replacement in patients with significant pulmonary insufficiency. Reference Stout, Daniels and Aboulhosn1 While surgical pulmonary valve replacement has traditionally been undertaken for patients with native or surgically palliated right ventricular outflow tracts, new transcatheter valve technologies allow for primary catheter-based valve implantation in select patients.
Currently, cardiac CT is required to evaluate candidacy for transcatheter pulmonary valve replacement using new right ventricular outflow tract reducer valve technologies in patients with native or surgically repaired right ventricular outflow tracts. Cardiac CTs are required by the transcatheter valve manufacturers despite the high prevalence of robust magnetic resonance angiography datasets in this population. These cardiac CT datasets are sent to industry, and a fit analysis is performed in order to assess patient anatomic candidacy for right ventricular outflow tract reducer platforms. This requirement for cardiac CT creates an extra burden for patients. Furthermore, while higher spatial resolution can be achieved on cardiac CT relative to magnetic resonance angiography, cardiac CT can be subject to limitations such as blooming and beam hardening artefacts and motion artefacts, which may impact dataset quality. We have previously suggested that high-quality magnetic resonance angiography datasets can provide information comparable to that obtained by cardiac CT. Reference Wilson, Hashemi, Smith, D Sallee, Kim and Slesnick2 Furthermore, there is growing experience with successful implantation of these transcatheter pulmonary valve replacement platforms in patients whose cardiac CT datasets resulted in an industry-based “screen failure” but are felt by the operator to be favourable candidates. Therefore, strict adherence to industry cardiac CT-derived fit analyses as an absolute requirement for candidacy may unnecessarily exclude patients from transcatheter pulmonary valve replacement. Reference McElhinney, Gillespie and Aboulhosn3
Virtual reality is increasingly used in the congenital cardiology community to evaluate complex anatomic questions. Reference Zablah, Than, Browne, Rodriguez and Morgan4,Reference Wilson, Hashemi and Ligon5 However, data on virtual reality modelling using magnetic resonance angiography data are less robust. In this study, we aimed to (1) assess the feasibility of generating 3-dimensional (3D) virtual right ventricular outflow tracts models from clinical magnetic resonance angiography datasets in patients undergoing consideration for transcatheter pulmonary valve replacement and (2) compare transcatheter pulmonary valve replacement candidacy and outcomes with visualisation of right ventricular outflow tracts and valve models in virtual reality to the disposition suggested by industry fit analysis.
Materials and methods
This was a retrospective study. We identified the patients at our centre with native or surgically palliated right ventricular outflow tracts who underwent cardiac CT to evaluate for transcatheter pulmonary valve replacement using right ventricular outflow tract reducer valve technologies. We included patients who had a cardiac MRI with magnetic resonance angiography completed within one year of cardiac CT imaging. We excluded patients with unknown transcatheter pulmonary valve replacement outcomes or those with palliative interventions performed elsewhere.
Cardiac magnetic resonance angiography was performed on a 1.5 tesla magnet (Avanto Fit, Siemens Healthcare, Forchheim, Germany). 0.2 mmol/kg gadolinium contrast was administered to acquire a 1.2–1.4 mm isotropic 3D inversion recovery prepared sequence with gradient echo readout (3D IR-GRE) contrast-enhanced magnetic resonance angiography using vectorcardiogram gating to systole and respiratory navigation. We preferentially gate to systole at our centre per institutional protocol, as we have found that this technique consistently results in high-quality datasets, although gating to diastole would also be feasible. There was one patient who had a non-contrast 3D steady state free precession magnetic resonance angiography completed. Cardiac CTs were completed using retrospective cardiac gating on a dual source scanner (SOMATOM Force, Siemens Healthcare, Forchheim, Germany) according to industry specifications to generate 0.5 mm isotropic datasets covering the right ventricular outflow tracts and branch pulmonary arteries.
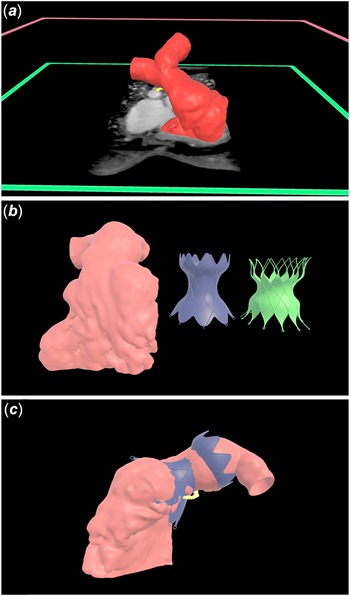
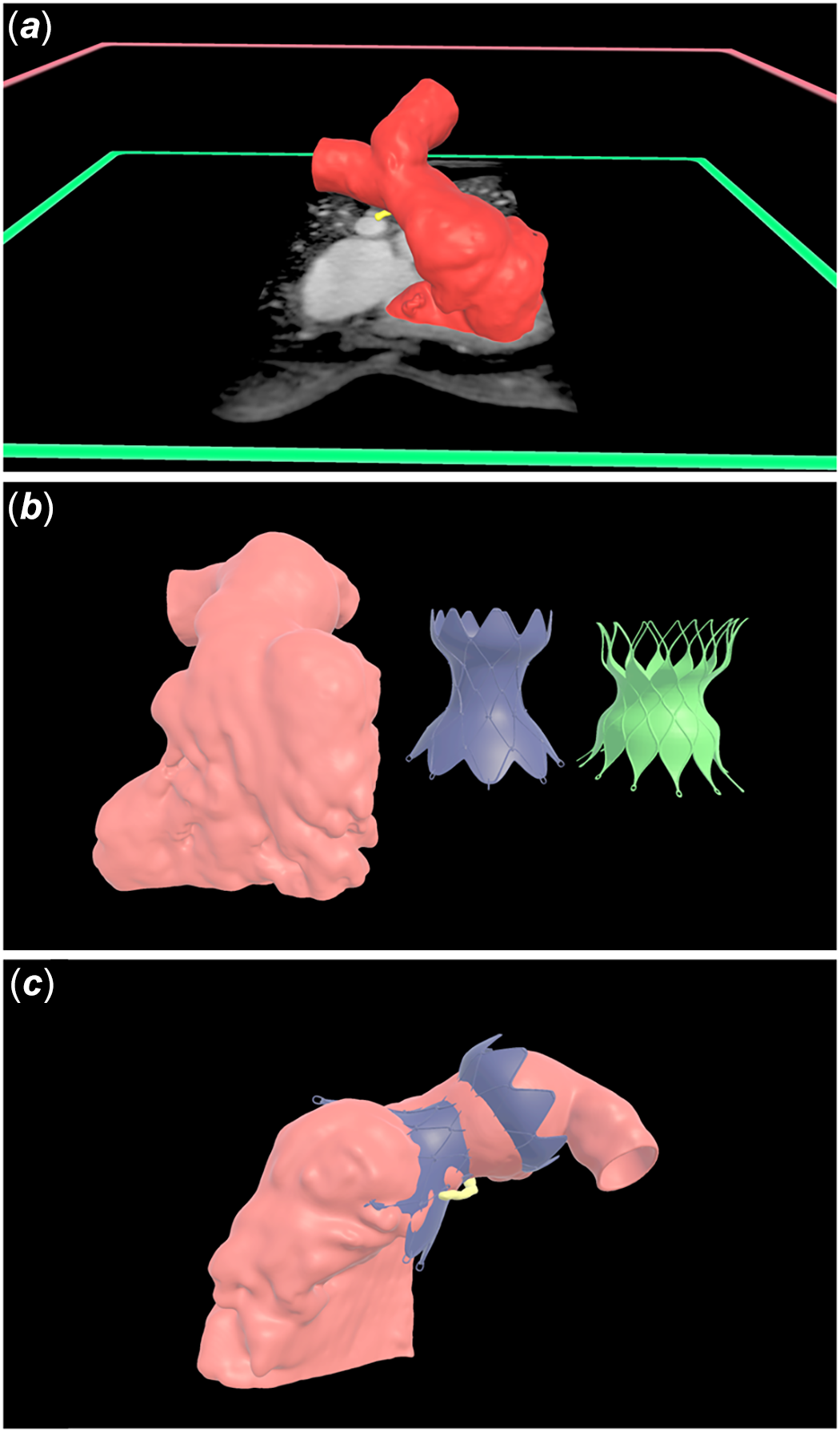
Magnetic resonance angiography datasets were segmented using Elucis™ (Realize Medical, Ottawa, Canada) in order to generate 3D models of right ventricular outflow tracts, branch pulmonary arteries, and the left coronary artery system. Anatomic scale and structure relationships were maintained during the segmentation process. Virtual models of the Medtronic Harmony TPV 25 mm™ valve and Edwards Alterra Adaptive Prestent™ were created locally using industry-reported dimensions in a 1:1 anatomic scale and visualised with right ventricular outflow tract models within the virtual reality environment. The valve could be manipulated in virtual reality and superimposed upon the anatomic right ventricular outflow tract models. While a Harmony TPV 22 mm™ valve model is also available for clinical use, a virtual model of this valve was not generated, as in our experience, patients rarely satisfy the criteria for usage of this valve.
Clinical data were extracted from the electronic medical record. Industry screening reports from cardiac CT datasets for the Harmony TPV 25 mm™ valve were tabulated. An experienced interventional cardiologist, blinded to the industry-driven cardiac CT fit analysis results and clinical outcome, visualised models and the virtual valve in virtual reality and made a determination of transcatheter pulmonary valve replacement candidacy. The interventional cardiologist received a training session in the use of the virtual reality platform before the datasets were evaluated. The criterion for transcatheter pulmonary valve replacement candidacy was perception by the interventional cardiologist of adequate overlap of the proximal and/or distal aspect of the valve with the entirety of the anatomy on virtual reality inspection. This judgement was based on operator characterisation of the intended landing zone in terms of device stability, longitudinal fit within the outflow tract, risk of embolisation, possibility for paravalvular leak, risk of affecting adjacent structures (pulmonary or coronary arteries), and other criteria operators account for in determining right ventricular outflow tract reducer clinical candidacy. Ultimately, candidacy for transcatheter valve replacement was derived using a subjective assessment of the fit of the valve with the anatomy in virtual reality, informed by the above considerations. Data are presented using descriptive statistics. This study was approved by the local Institutional Review Board; the requirement for informed consent was waived.
Results
A total of 16 Patients (N = 7 males, 44%) with a median age of 15.5 years (interquartile range [IQR] 13.9, 17.8) were identified. The underlying congenital heart defect was tetralogy of Fallot in 15 patients and double outlet right ventricle in one patient. All cardiac magnetic resonance angiography datasets were adequate for virtual reality model creation. The median time for model creation was 20.6 minutes (IQR 18.5, 22.0) (Figure 1). The mean time the interventional cardiologist spent reviewing datasets was 3.0 ± 2.4 minutes.
Right ventricular outflow tracts were segmented from magnetic resonance angiography datasets and virtual models were created (Panel a). Scale models of the Harmony TPV 25 mm™ valve and Edwards Alterra Adaptive Prestent™ were built using industry specified dimensions (Panel b). The virtual Harmony TPV 25 mm™ valve is superimposed on a patient-specific three-dimensional right ventricular outflow tract model to evaluate valve fit and candidacy for transcatheter pulmonary valve replacement (Panel c).
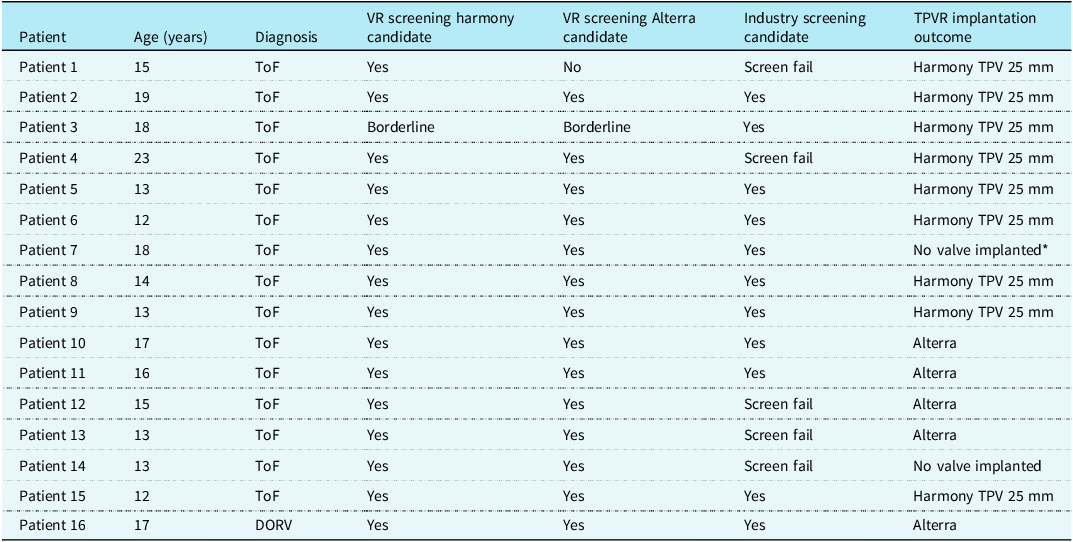
Of the cohort, there were 11/16 (69%) patients with industry fit analyses which passed industry screening. Industry screening failures were predominantly secondary to inadequate overlap of the proximal/inflow portion of the valve within the anatomy to meet the specific industry-set criteria, as demonstrated in the centreline perimeter plot. Of the entire cohort, 14/16 (88%) ultimately underwent transcatheter pulmonary valve replacement. Of patients undergoing transcatheter pulmonary valve replacement, 9/14 (64%) underwent implantation of the Harmony TPV 25 mm™ valve, and 5/14 (36%) underwent implantation of the Edwards Alterra Adaptive Prestent.™ When viewed in virtual reality, there were 15/16 (94%) patients whose anatomy was felt to be amenable to transcatheter pulmonary valve replacement and 1/16 (6%) patient whose anatomy was felt to be borderline for the virtual Medtronic Harmony TPV 25 mm™ valve (Table 1). There were 14/16 (88%) patients whose anatomy was felt to be amenable to transcatheter pulmonary valve replacement with Edwards Alterra Adaptive Prestent.™ 1/16 (6%) was felt not to be a candidate for Alterra Adaptive Prestent™, and another 1/16 (6%) felt to be borderline in candidacy.
Outcomes of blinded virtual reality screening for transcatheter pulmonary valve candidacy, industry-based screening for candidacy using cardiac computed tomography datasets, and clinical outcome of whether valve implantation was successfully completed
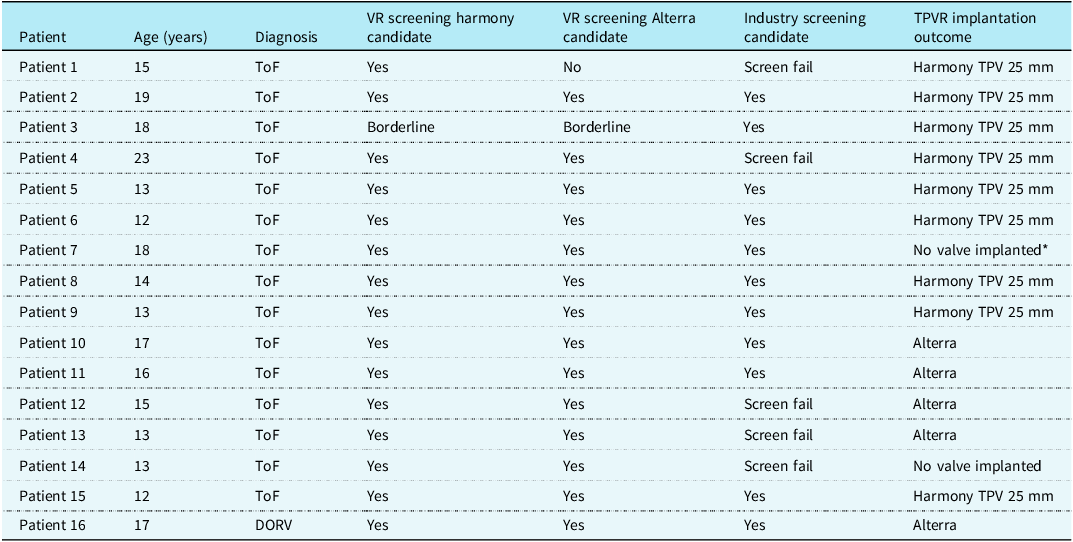
ToF = tetralogy of Fallot; DORV = double outlet right ventricle; VR = virtual reality; TPVR = transcatheter pulmonary valve replacement.
*A left pulmonary artery stent was present.
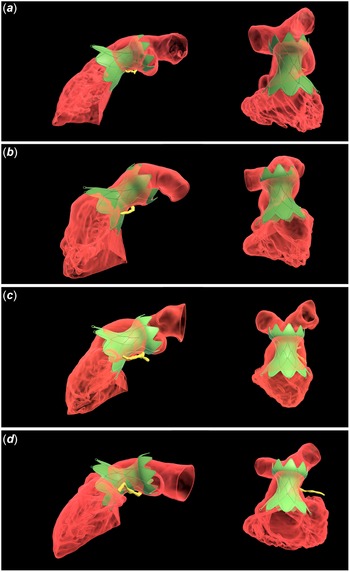
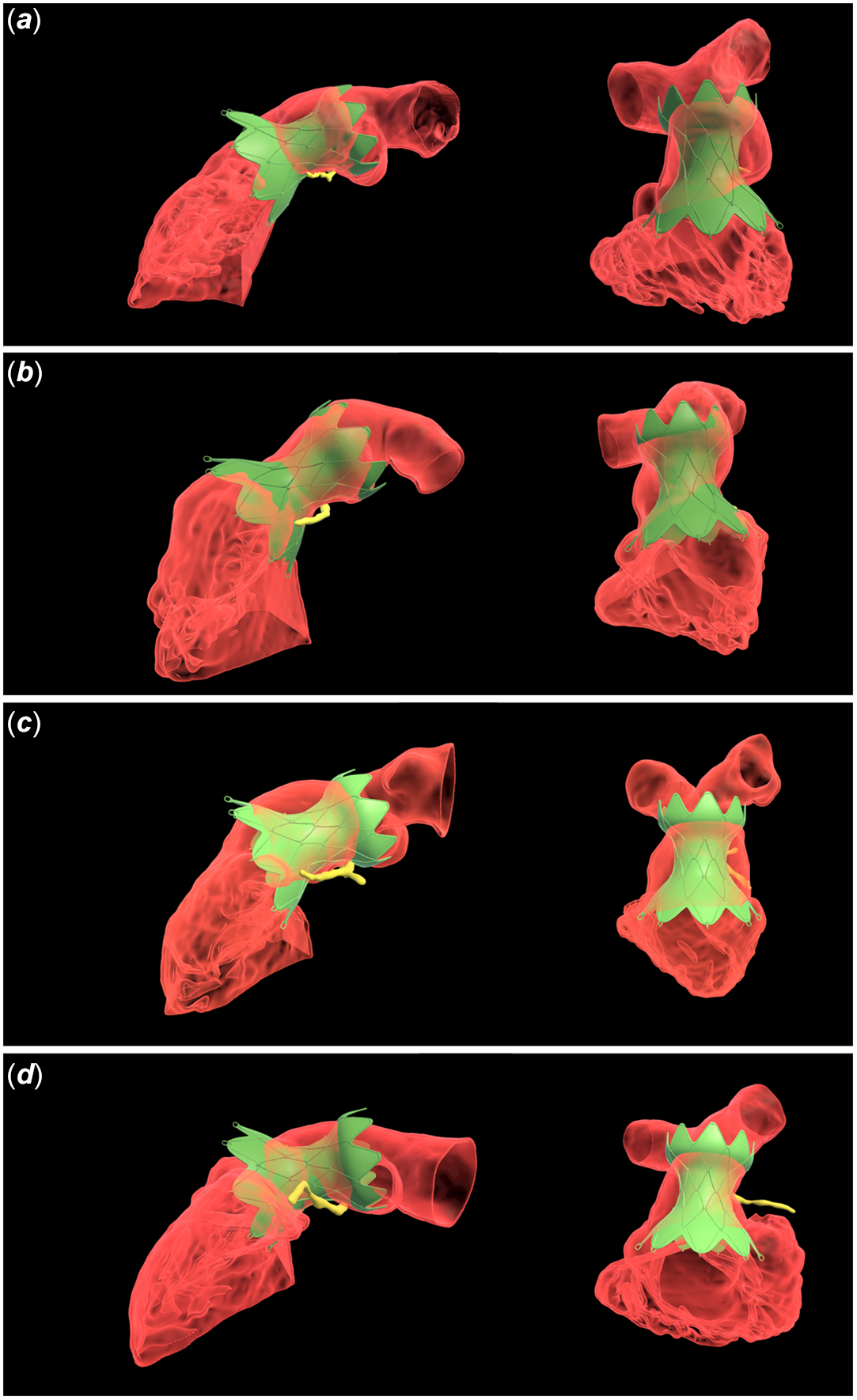
When specific patients were reviewed, there were four patients who passed virtual reality screening for the Harmony TPV 25 mm™ valve but failed industry screening who ultimately underwent successful transcatheter pulmonary valve replacement implantation (patients 1, 4, 12, and 13). Each of these patients failed industry screening due to inadequate overlap of the proximal aspect of the Harmony TPV 25 mm valve™ with the right ventricular outflow tract anatomy (Figure 2). There was one patient who passed both virtual reality and industry screening but did not undergo transcatheter pulmonary valve replacement secondary to the presence of a left pulmonary artery stent (patient 7). One patient passed virtual reality screening but failed industry screening and was not offered transcatheter pulmonary valve replacement (patient 14). There was one patient who passed virtual reality screening for the Harmony TPV 25 mm™ valve but failed virtual reality screening using the virtual Edwards Alterra Adaptive Prestent.™ In this case, the distal “tines” of the valve extended beyond the anatomy to the extent that there was felt to be potential risk for extension beyond the anatomy in vivo, as has been described in prior works. Reference Gillespie, Maschietto, Aboulhosn, Balzer, Qureshi and McElhinney6
Right ventricular outflow tract models visualised in virtual reality from sagittal oblique (left) and cranially oriented views (right) for patients 1 (Panel a), 4 (Panel b), 12 (Panel c), and 14 (Panel d) who failed industry fit analysis but were deemed candidates for the Harmony TPV 25 mm™ valve based on review of datasets in virtual reality. Right ventricular outflow tract models are visualised in red with opacification to allow for visualisation of the orientation of the Harmony TPV 25 mm valve™ (green). The proximal left coronary artery system is shown in yellow.
Discussion
We report the feasibility of generating virtual 3D models of right ventricular outflow tracts anatomy utilising clinical magnetic resonance angiography datasets. Magnetic resonance angiography datasets were robust for the generation of high-quality models in our cohort, and patient-specific anatomy was able to be successfully viewed in the virtual reality environment along with virtual valves. Virtual reality-aided models may serve as an important adjunct means of evaluating transcatheter pulmonary valve replacement candidacy using new valve technologies.
Among the study cohort, there were five patients who passed virtual reality screening using the virtual Harmony TPV 25 mm™ model but failed to meet the industry-based screening requirements. There is growing evidence that transcatheter pulmonary valve replacement can be successfully undertaken in patients who do not pass traditional industry screening requirements such as oversizing criteria. This suggests imperfect sensitivity for the use of industry screening reports alone in determining transcatheter pulmonary valve replacement candidacy. Reference McElhinney, Gillespie and Aboulhosn3 Industry reports based on cardiac CT data are derived using cross-sectional right ventricular outflow tracts data at different points along the right ventricular outflow tracts length. For example, the Harmony™ screening report uses the perimeter of the right ventricular outflow tracts at different points along the length of the right ventricular outflow tracts to calculate a perimeter-derived radius at each level in systole and diastole. A centreline plot is generated, and 2-dimensional renditions of valves are superimposed. An industry-driven screen pass or fail is determined by whether there is adequate overlap of the proximal and distal ends of the valve with the right ventricular outflow tract anatomy. While these reports provide a useful distillation of complex geometry, they may not fully encapsulate all aspects of anatomy, which may be better visualised with 3D renderings. There is a growing sense within the right ventricular outflow tract reducer implanter community to focus on details within the report outside of the oversizing criteria, which we feel is greatly enhanced by virtual reality visualisation.
The five patients who failed industry screening were all felt to be suitable candidates for transcatheter pulmonary valve replacement for the Harmony TPV 25 mm™ based on virtual reality review, and four out of five patients ultimately underwent transcatheter pulmonary valve replacement, underscoring the notion of clinical suitability for transcatheter pulmonary valve replacement in certain patients who do not pass industry criteria. When viewed in virtual reality, these anatomic datasets demonstrated adequate overlap of virtual valves with right ventricular outflow tract anatomy in a systolic phase. We speculate that the third patient, who underwent evaluation early in our institutional experience, who passed virtual reality screening but failed industry screening (patient 14) and did not undergo transcatheter pulmonary valve replacement, may have been more strongly considered for transcatheter pulmonary valve replacement in the contemporary era. Finally, it is worth noting that the one patient who was felt by virtual reality to be a suitable candidate for the Harmony TPV 25 mm™ valve but not the Edwards Alterra Adaptive Prestent™ failed virtual reality screening for the latter secondary to perception of potential for protrusion of “tines” beyond the anatomy. This phenomenon has been described, and virtual reality modelling of anatomy may allow for a more comprehensive qualitative assessment of the potential impact of this complication on surrounding structures. Reference Gillespie, Maschietto, Aboulhosn, Balzer, Qureshi and McElhinney6
While virtual reality offers a powerful means of evaluating 3D anatomy, it is essential that the parameters of the datasets are understood. Our magnetic resonance angiography datasets are generated in systole and therefore, proximal right ventricular outflow tract dimensions are likely smaller and main pulmonary artery dimensions larger than in diastole. This was understood by the blinded interventional cardiologist when the determination of transcatheter pulmonary valve replacement candidacy was assigned. While systolic magnetic resonance angiography datasets were generated at our centre, generation of a second contrast-enhanced magnetic resonance angiography diastolic dataset could also be feasible during the same magnetic resonance angiography acquisition, or a second dataset could be generated using a non-contrast technique, insofar as these data are helpful for device planning. Furthermore, advancement of newer cardiac magnetic resonance angiography techniques may allow for more widespread acquisition of whole heart multiphase dataset which would allow for generation of 3D datasets and measurements in systole and diastole from a single imaging sequence. Reference Roy, Di Sopra and Whitehead7 Notably, screening reports for the Medtronic Harmony Transcatheter Pulmonary Valve™ and Edwards Alterra Adaptive Prestent™ have focused on different phases of the cardiac cycle for determination of anatomical candidacy for transcatheter pulmonary valve replacement. Reference Patel, Levi, Cheatham, Qureshi, Shahanavaz and Zahn8 Visualisation of the right ventricular outflow tract anatomy in 3D allowed for improved visualisation of other important aspects of the right ventricular outflow tract anatomy, such as “choke points” near the level of the annulus which may allow for stable implant positioning. It is also worth noting that one patient (patient 7) passed both virtual reality and industry screening, but transcatheter pulmonary valve replacement was not implanted, given the presence of a left pulmonary artery stent and concern for malposition of the right ventricular outflow tract reducer system within the anatomy. This example emphasises the need to consider all imaging and clinical data in totality when evaluating candidacy for transcatheter pulmonary valve replacement.
Newer virtual reality platforms offer a smooth user interface and a relatively short learning curve for users to become facile with basic manipulations in the virtual reality environment. In this effort, the blinded interventionalist felt comfortable exploring anatomy in the virtual reality environment after an introductory training session. A current challenge with the use of virtual reality technology is identifying objective measures for evaluating complex data in the virtual reality environment. However, in the current era, virtual reality platforms are more accessible and user-friendly. More widespread use of virtual reality may ultimately allow our community to develop more robust paradigms for the use of virtual reality in challenging structural heart disease.
While still a new technology, virtual reality is increasingly used in cardiology for the evaluation of complex anatomic questions. Kanshik et al demonstrated use of virtual reality to generate precise measurements for guidance of transcatheter aortic valve implantation. Reference Kanschik, Haschemi and Heidari9 Virtual reality anatomic datasets have also been used for ventricular assist device planning using virtual models of ventricular assist devices. Reference Davies, Hussain and Tandon10 In the congenital cardiology space, Zablah et al have shown good capability of virtual reality to predict transcatheter pulmonary valve replacement outcome using cardiac CT datasets. Reference Zablah, Than, Browne, Rodriguez and Morgan4 We extend this work by showing that segmentation of right ventricular outflow tracts anatomy and clinical evaluation for valve candidacy is feasible using magnetic resonance angiography datasets. Segmentation of the totality of cardiac CT datasets offers the ability to view dynamic anatomy throughout the cardiac cycle, but comes with a time cost given the need to segment multiple phases. Therefore, segmentation of static magnetic resonance angiography datasets may ultimately be more clinically feasible. Ultimately, it would be ideal if clinical magnetic resonance angiography data could be used for the primary determination of transcatheter pulmonary valve replacement candidacy, removing the time, cost, and ionising radiation associated with cardiac CT.
This study is subject to several limitations, including its retrospective nature, small sample size, and the ultimately subjective nature of the assignment of transcatheter pulmonary valve replacement candidacy based on visualisation in virtual reality. Future efforts include completion of additional comparisons of cardiac magnetic resonance angiography and cardiac CT datasets and derivation of more objective means of evaluating the overlap of devices within patient anatomy using a virtual reality platform.
In conclusion, creating 3D models using clinical magnetic resonance angiography datasets is feasible and allows for the conceptualisation of valve overlap with patient-specific anatomy within the virtual reality environment. Virtual reality-aided models may serve as an adjunct means of evaluating transcatheter pulmonary valve replacement candidacy using new transcatheter valve technologies but should be judiciously used in conjunction with other clinical and imaging data.
Acknowledgements
None.
Financial support
This work was supported by a grant from the Children’s Healthcare of Atlanta 1998 Society helping fund the virtual reality software utilised. This organisation had no role in study design; in the collection, analysis and interpretation of data; in the writing of the report; or in the decision to submit the article for publication.
Competing interests
R Allen Ligon reports consulting and advisory relationships with Abbott Vascular Inc., Medtronic, and Penumbra Design Inc. Dennis Kim reports consulting and advisory relationships with Edwards Lifesciences. All other authors disclose no relevant relationships.