Introduction
Haskap, Lonicera caerulea Linnaeus (Caprifoliceae), also known as blue honeysuckle or honeyberry, is a fruit crop of growing importance in Canada (Agriculture and Agri-Food Canada 2024). The plant is native to northern regions of North America, Europe, and Asia and is primarily suited for high latitudes and temperate cold climates (Hummer Reference Hummer2006; Plekhanova Reference Plekhanova2000; Hummer et al. Reference Hummer, Pomper, Postman, Graham, Stover, Mercure, Badenes and Byrne2012; Vahenurm et al. Reference Vahenurm, Rätsep and Arus2020). Haskap berries are dark blue or purple, oval, and elongated and have a tart–sweet flavour. Having high levels of flavonoids and phenolic acids, the berries are increasingly recognised for their antioxidant and anti-inflammatory properties and are promoted for their health benefits (Jurikova et al. Reference Jurikova, Rop, Mlcek, Sochor, Balla and Szekeres2011; Rupasinghe et al. Reference Rupasinghe, Boehm, Sekhon-Loodu, Parmar, Bors and Jamieson2015, Reference Rupasinghe, Arumuggam, Amararathna and De Silva2018; Dharmawansa et al. Reference Dharmawansa, Stadnyk and Rupasinghe2024).
Lonicera caerulea has a two-flower inflorescence typical of other species in the genus Lonicera. The ovaries of each flower are enclosed by bracteoles, and the haskap berry forms from the combined structures, creating a single compound fruit from the pollination of two flowers (Zhu et al. Reference Zhu, Zhang, Gao, Qin and Huo2022). If only one flower of the inflorescence is pollinated, fruit set is lower, and the resulting fruit is typically smaller with fewer seeds (Frier et al. Reference Frier, Somers and Sheffield2016a). Plants are self-incompatible, and fruit set requires cross-pollination of two compatible cultivars, which is largely dependent on insect pollination (Bozek Reference Bozek2012; Boyarskikh Reference Boyarskikh2017). Haskap flowers have high nectar production and long floral longevity and anther dehiscence (4–5 days). These floral traits maximise opportunities for pollination by increasing the flowers’ attractiveness to a range of insect pollinators (Frier et al. Reference Frier, Somers and Sheffield2016b).
Bloom of haskap typically begins in late April to early May in southwest Nova Scotia, Canada. This period usually has uncertain and inclement weather in the province, while also having high pollen demand for honey bee, Apis mellifera Linnaeus (Hymenoptera: Apidae), colonies, corresponding with onset of brood production and spring build up. Honey bees are effective pollinators of haskap (Frier et al. Reference Frier, Somers and Sheffield2016a), but they will not forage in weather that is too cool, wet, or windy (Abrol Reference Abrol2012), such as that commonly experienced in Nova Scotia in spring. Because of this, the Nova Scotia haskap industry is interested in optimising contributions of the wild pollinator community for pollination of haskap flowers. In other regions, bumble bees, Bombus spp. (Hymenoptera: Apidae), are particularly effective pollinators of haskap, but other taxa, such as cavity-nesting Osmia spp., (Hymenoptera: Megachilidae) may also pollinate the crop (Frier et al. Reference Frier, Somers and Sheffield2016a; Leung and Forrest Reference Leung and Forrest2019).
Currently in Nova Scotia, the majority of haskap growers rent commercially managed honey bee colonies to help pollinate their crop. It is unclear how effective honey bees are at pollinating haskap compared to other bees and what other pollinator taxa actively work haskap flowers during crop bloom. We therefore assessed the activity of honey bees in haskap orchards and compared this with native bumble bees, in particular, while also documenting the early season diversity and abundance of other wild bees in haskap orchards. Because Megachilidae pollinate haskap in other regions (Frier et al. Reference Frier, Somers and Sheffield2016a), we also wanted to evaluate the potential of nesting boxes to attract cavity-nesting bees in haskap orchards in Nova Scotia, particularly during bloom. Many different nest box designs have been developed (MacIvor Reference MacIvor2017), but the success of nesting boxes or “bee hotels” in attracting cavity-nesting bees has been mixed (Rahimi et al. Reference Rahimi, Barghjelveh and Dong2021). We incorporated tubes of different diameters and spacings into our nest box designs to determine if these factors affected bee nesting success in Nova Scotia.
Materials and methods
Study sites
Field studies were conducted in 2016 and 2017 in three haskap orchards belonging to Lahave Natural Farms (Lunenburg County, Nova Scotia, Canada): Honeyberry Hurst (6.36 ha; 44° 28' 02.9'' N, 64° 32' 15.8'' W); Silver Hurst (5.32 ha; 44° 28' 45.3'' N, 64° 35' 08.3'' W); and Lohnnes (1.97 ha; 44° 29' 24.4'' N, 64° 35' 55.0'' W). Honey bee colonies were placed by the producer in a single location along an edge of each haskap orchard at a stocking density of approximately 0.4 hives per hectare. In 2017, the producer increased hive density at Honeyberry Hurst to approximately 0.8 hives per hectare. At Honeyberry Hurst, many haskap plants were 4–5 years old, whereas those at Silver Hurst and Lohnnes were 2–3 years old. For the 2017 nesting box experiment, we used an additional four orchards: Barrs Corner (1.77 ha; 44° 33' 43.3'' N, 64° 40' 32.7'' W); Lonetree (18.08 ha; 44° 28' 10.4'' N, 64° 39' 24.9'' W); Lahave River Berry (0.21 ha; 44° 19' 08.5'' N, 64° 23' 41.3'' W); and Mahone Bay (1.48 ha; 44° 27' 12.6'' N, 64° 22' 21.7'' W). All orchards contained Borealis, Tundra, and Indigo Gem cultivars, as well as the Honeybee cultivar, which was used as a polliniser. All orchards were in rural areas surrounded by forest and landscapes that consisted mostly of tolerant mixed-wood drumlins and, to a lesser extent, spruce–hemlock–pine hummocks, hills, and ridges (Nova Scotia Natural Resources and Renewables 2025), interspersed with small areas of cropland, fallow, pasture, lakes, ponds, rivers, and creeks.
Pollinator surveys
Pan trapping
In 2016, pan traps were placed along three 36-m transects spaced 25 m apart (five crop rows) in each of the three orchards. Pan traps consisted of blue, yellow, and white plastic bowls (355 mL; Solo Cup Company, Urbana, Illinois, United States of America) half-filled with water. Two drops of unscented dish soap were added to each bowl to break water-surface tension and to reduce escape of captured insects. Transects were set between haskap rows but near haskap bushes. Each transect ran parallel to the row of haskap bushes and had traps spaced 3 m apart with alternating colours of blue, yellow, and white. Pan traps were placed at ground level near the centre of each orchard. Pan traps were set out between 08:00 and 16:00 hours, local time, on sampling days. Sampling was done on sunny days that had temperatures above 12 °C and low wind. Thirty pan traps per transect were set in pre-bloom sampling, but during this time, we observed a high number of bumble bee queens being trapped. We therefore reduced the number of pan traps per transect to 12 for subsequent bloom and post-bloom sampling periods. Two collections were made pre-bloom, five collections were made during bloom, and three were made post-bloom. Sampling during bloom in 2016 occurred on five separate days during 4–17 May, with the entire sampling period spanning 21 April–21 June. Bees removed from pan traps were stored in ethanol and returned to the laboratory for counting and identification. Bees captured were identified to genus using Packer et al.’s (Reference Packer, Genaro and Sheffield2007) key and the Discover Life online key (DiscoverLife.org 2017). Flies were identified to family (data not shown).
Pan trap sampling was modified in 2017 to examine field edge effects. Two grids of pan traps were placed in each orchard. A grid consisted of a randomised 3 × 3 arrangement of blue, yellow, and white pans (nine pans total) set 3 m apart from each other. One grid of pans was established on the westernmost field edge, and the other was established 60 m within the field. Two collections were made during the pre-bloom period, five collections were made during bloom, and three were made post-bloom. Sampling during bloom in 2017 occurred on five separate days during 3–18 May, with the entire sampling period spanning 21 April–8 June. Insects were handled and identified as described above.
Transect walks
Transect walks were completed during bloom in the afternoon of days when pan traps were sampled. One 30-m transect along a row of haskap in each orchard was observed for 30 minutes. Transects were set in areas of low human disturbance and were done as quietly and as inconspicuously as possible. To maintain consistency, only one person counted pollinators along transects, and transects were set in the same locations in both 2016 and 2017. Pollinators were classified as bumble bees, honey bees, solitary bees, or hover flies (Diptera: Syrphidae) and were counted only if they were actively foraging on haskap flowers.
Statistical analysis – pollinator surveys
Some taxa were collected infrequently or not at all at certain periods, and we therefore conducted statistical analyses on only the four most common genera. For 2016, all pan trap collections were standardised to a per-trap-per-day measure for each orchard for each sampling period (pre-bloom, bloom, or post-bloom). In 2017, we standardised all collections to a per-trap-per-day measure for each orchard, trap location (0 m or 60 m from the field edge), and sampling period. For both years, the number of pollinators was the response variable. Assumptions of normality-of-error terms and constant variance of residuals were verified, and analysis of variance using the Proc GLM procedure in SAS (SAS Institute Inc. 2014) was used to examine effects of sampling period, trap location and their interaction, or both, with orchard as a blocking factor, on pollinator counts. Multiple means comparison where appropriate was done using Fisher’s least-significant difference test at α = 0.05.
Transect walk pollinator count data were analysed as a mixed model using the Proc MIXED procedure (SAS Institute Inc. 2014). Data were square-root transformed to achieve normality of residuals and constant variance. Transect walks were completed along the same transects and fields in 2016 and 2017, permitting interyear comparisons. Pollinator category, year, and their interaction were treated as fixed factors, and orchard was treated as a random blocking factor. Multiple means comparison was done using Fisher’s least-significant difference test at α = 0.05.
Pollinator behaviour
Floral visit pollen deposition
Counts of single-visit pollen deposition from visits to virgin haskap flowers were used to measure pollinator effectiveness. This study was done at Honeyberry Hurst in 2016 and 2017. In 2016, pollinator exclusion bags were placed over pairs of haskap flower buds and fastened to the branches using twist ties, such that pollination could not occur unless bags were removed. Pollinator exclusion bags were 150 mm × 100 mm and made of 1-mm white nylon mesh. After flowers opened, we waited at a bagged flower until a bee was nearby, at which time we removed the exclusion bag to allow the bee to visit the flower for pollination. Honey bees and bumble bees were used to measure single-visit pollen deposition because they were abundant, readily identifiable, and amenable to flower offerings. We randomly selected bees for flower offerings but tried to alternate between honey bees and bumble bees when doing so to avoid clustering of data collection by taxon on a given day. After a flower was visited by a bee, the stigma was carefully removed (taking care to minimise pollen dislodgement) and mounted on a microscope slide in basic fuchsin gel (Kearns and Inouye Reference Kearns and Inouye1993). To do so, an approximately 5-mm3 block of fuchsin gel was placed on a microscope slide, and the stigma was placed on top of the fuchsin gel cube. Using a pocket lighter, the microscope slide was slowly heated to melt the gel, and a coverslip was placed atop the melted gel and haskap stigma. The microscope slide was labelled and stored in a horizontal position until the fuchsin gel hardened. After returning to the laboratory, slides were observed under a dissection microscope at 5.6× magnification. Pollen grains were counted and used to infer single-visit pollen deposition rates for honey bees and bumble bees. A control “no insect pollination” treatment, which examined pollen on stigmas from bagged flowers not subject to insect visit, was also included in the experiment. Control flowers were randomly distributed throughout the orchard and were kept within pollinator exclusion bags until stigma collection.
Pollinator flower visitation rates
In 2016 and 2017, honey bees and bumble bees were tracked during haskap bloom at Honeyberry Hurst orchard to determine the number of flowers visited in 60 seconds. If a pollinator went out of sight during the tracking period, the pollinator was no longer followed and data for that insect were not used. Data from 2016 and 2017 were combined for statistical analysis.
Pollination of two-flower inflorescence
Although haskap fruit set can occur if only one flower in the two-flower inflorescence is pollinated, fruit set is higher if both flowers are pollinated (Frier et al. Reference Frier, Somers and Sheffield2016a). Determining if pollinators of haskap tend to visit one or both flowers of an inflorescence is therefore of interest. In the spring of 2017, honey bee and bumble bee floral visits were observed during haskap bloom at the Honeyberry Hurst orchard. Bees were followed during their consecutive visits to 10 different inflorescences, and the number of instances in which both flowers in the inflorescence were pollinated was recorded. If a bee went out of sight of the observer before 10 inflorescences were visited, data for that bee were disregarded.
Honey bee pollen collections
On 17 May 2017, plastic front-mounted pollen traps (Country Fields Beekeeping Supplies, Fall River, Nova Scotia) were placed on three honey bee hives at Honeyberry Hurst and Lohnnes orchards during haskap bloom. Pollen traps were mounted to hives deemed to be strong by previous visual inspection and were activated for 24 hours. Collected pollen was stored on ice, returned to the laboratory, and placed in a freezer at –20 °C until it was sent away for morphometric analysis. Floral source analysis for pollen was conducted by Johanne Parent (Laboratoire BSL, Rimouski, Quebec, Canada), using previously described methods (Cutler and Scott-Dupree Reference Cutler and Scott-Dupree2014).
Statistical analysis – pollinator behaviour
Analysis of variance using the Proc GLM procedure (SAS Institute Inc. 2014) was used to determine if pollinators (honey bee versus bumble bee) differed in their deposition of pollen grains, number of flower visits per minute, and tendency to visit both flowers of an inflorescence. Assumptions of normality of error terms and constant variance of residuals were met. Multiple means comparison was done using Fisher’s least-significant difference test at α = 0.05.
Nesting box studies
Nest boxes consisted of a 23-cm-long, 10-cm-diameter cylindrical piece of white PVC pipe with a cap to seal one end. A 10-cm-diameter piece of 2.5-cm-thick purple polystyrene insulation containing 12 cardboard nest tubes (Crown Bees, Woodinville, Washington, United States of America) was inserted into the front of each cylinder. Nest tubes were 15 cm long and bluntly rounded with a 4-mm orifice at one end. Spray foam insulation was sprayed around the nest tubes to provide stability. Nest boxes contained either 6-mm-diameter or 8-mm-diameter tube openings, which were arranged either snugly together or spaced 2.5 cm apart, giving four different nest designs: (1) 6 mm snug; (2) 6 mm spaced; (3) 8 mm snug; and (4) 8 mm spaced. In 2017, in each of the seven orchards, we established three clusters of the four nest box designs, totalling 12 nest boxes per orchard. Within a cluster, the four nest boxes were placed 1 m apart, and clusters were spaced approximately 50 m apart. Nest boxes were placed in the field on 13 April and checked once every 2–3 weeks for capped nest tubes, both at the front opening and within tubes. Grass was cut around the nest boxes once a month. On 13 September, the nesting boxes were collected and returned to the lab, and the total number of capped tubes was recorded.
Nest boxes with one or more capped tubes were stored in 4 °C cold storage (MacIvor and Packer Reference MacIvor and Packer2015) until 5 March of the following year. Capped cardboard nest tubes were removed from the nest boxes, labelled, and placed in separate inflated plastic bags (McCallum et al. Reference McCallum, McLean and Cutler2018). Bags with tubes were placed in an environmental chamber at 8 °C and warmed with daily increments of 4 °C, up to 24 °C by 9 March. After 10 days at 24 °C, the temperature was increased to 30 °C until 2 April. Relative humidity was maintained at 60% throughout the hatching period (MacIvor and Packer Reference MacIvor and Packer2015; McCallum et al. Reference McCallum, McLean and Cutler2018). Bees and wasps that emerged were frozen. Wasps were identified to family; and bees were identified to genus or species.
Statistical analysis – nest box studies
We had intended to use a 2 × 2 factorial design, with two levels of nest tube diameter (6 mm versus 8 mm) and two levels of nest tube arrangement (snug versus spaced). However, occupancy in 8-mm-sized tubes was limited, and therefore only the data collected for 6-mm-sized tubes were statistically analysed. For each orchard, we determined the total number of capped nest tubes for each nest tube style (snug or spaced) on different dates. Data were analysed for variance with the Proc GLM procedure (SAS Institute Inc. 2014). Assumptions of normality of error terms and constant variance of residuals were verified before analyses.
Results
Pollinator surveys
Pan trapping
In 2016, 362 bees from nine genera were captured in pan traps. In 2017, 268 bees from 10 genera were captured. Andrena was the most common genus captured, comprising 42% and 34% of all bees captured in 2016 and 2017, respectively. Approximately 70% of Andrena were captured during bloom (Table 1). Andrena, Apis, Bombus, and Lasioglossum spp. comprised 87% and 76% of all pollinators captured during haskap bloom in 2016 and 2017, respectively. Two families of pollinating flies, Syrphidae and Bombyliidae (Diptera), were captured in relatively low numbers. Pollinator captures were generally greatest in 2016 and during haskap bloom in both years, although Bombus queens were captured mostly pre-bloom in 2016, likely because of greater pre-bloom sampling effort in that year. Except for Lasioglossum and Sphecodes, most Halictidae, including the genera Agapostemon, Augochlorella, and Halictus, were captured post-bloom. All captured taxa were collected in both years, except for one individual Sphecodes sp. that was captured only during bloom in 2017. Osmia were not captured in high numbers in any sampling period (Table 1).
Table 1. Wild and managed bees captured in pan traps during three sampling periods in three haskap orchards in southern Nova Scotia, Canada, 2016 and 2017

The four most common taxa – Andrena, Apis, Bombus, and Lasioglossum – were collected during all time periods and were subject to statistical analysis. In both 2016 and 2017, the number of Apis, Bombus, and Lasioglossum collected pre-bloom, bloom, or post-bloom did not differ significantly (P > 0.10 in all cases). However, the number of Andrena collected during bloom was significantly greater than that collected pre-bloom and post-bloom in both 2016 (F 2,6 = 8.18, P = 0.019) and 2017 (F 2,12 = 8.01, P = 0.006). In 2017, when we incorporated the factor of “trap location” into the study, neither trap location nor interaction of sampling period with trap location significantly affected any taxa (P > 0.05 in all cases).
Transect walks
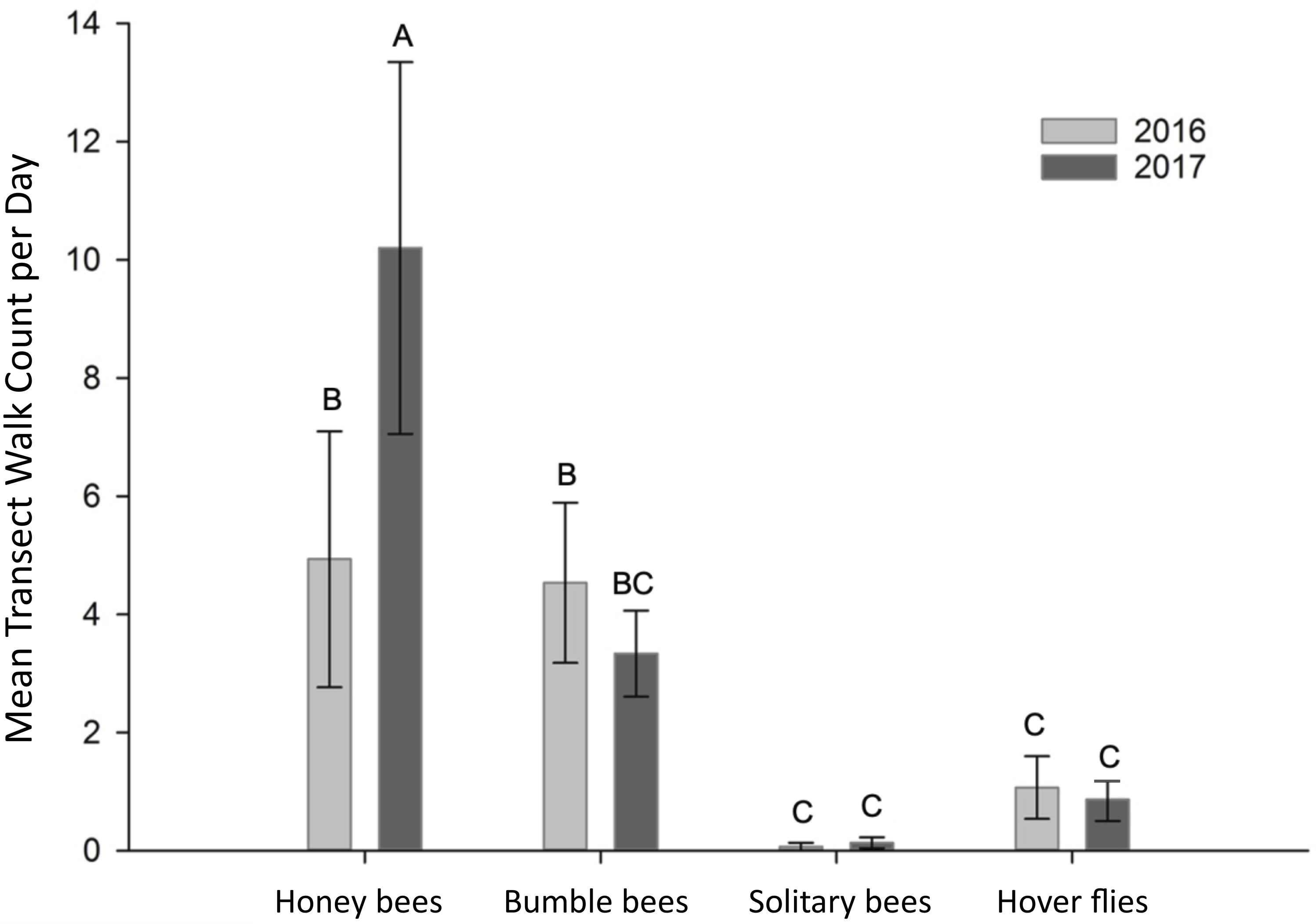
Overall, transect walk counts did not vary by year (F 1,32 = 1.36, P = 0.25), but counts of each type of pollinators differed significantly (F 3,32 = 15.97, P < 0.0001; Fig. 1). Honey bees and bumble bee queens were the pollinators most commonly observed on haskap flowers. Only half as many honey bees were observed in 2016 as in 2017, with noticeably more honey bees counted per day in 2017 at the Honeyberry Hurst site than at other orchards (23.6 ± 12.9 versus 3.5 ± 2.7 (mean ± standard deviation)). Counts of other taxa were greatest in 2016 or equivalent in 2016 and 2017 (taxa–year interaction; F 3,32 = 2.96, P = 0.047). No significant difference in the mean number of bumble bee queens observed on transect walks during haskap bloom was detected between years (Fig. 1), but the number of Bombus queens on haskap flowers noticeably increased during mid–late bloom of the crop, compared to early bloom (data not shown). Few solitary bees and syrphid flies were observed on transect walks.

Figure 1. Mean (+/– standard error of the mean) number of pollinators counted on haskap flowers during 30-minute transect walks through haskap orchards in southern Nova Scotia, Canada, in 2016 and 2017. In each year, data were recorded along a single transect, on five different dates at three different orchards. Bars with different letter groupings differ significantly (Fisher’s least-significant difference test, α = 0.05).
Pollinator behaviour
The average number of pollen grains deposited by honey bees or bumble bees did not differ significantly, but pollen deposition was greater in both treatments than in the control (F 2,62 = 8.31, P = 0.0006; Fig. 2A). Honey bees deposited a mean of 111.4 pollen grains per stigma (range = 0–335, n = 30), and bumble bees deposited an average of 95.3 pollen grains per stigma (range = 0–240, n = 27), compared to only 7.5 pollen grains deposited per stigma in control flowers (range = 0–29, n = 8; Fig. 2A). Bumble bees visited approximately three times more haskap flowers per minute than honey bees did (F 1,54 = 127.2, P < 0.001), visiting approximately 27 flowers per minute (n = 30) compared to the nine flowers per minute visited by honey bees (n = 26; Fig. 2B). The number of flowers that bumble bees visited per minute was more variable than that visited by honey bees. Bumble bees tended to visit both flowers on an inflorescence more often than honey bees did (F 1,38 = 86.83, P = < 0.0001; Fig. 2C), visiting both flowers of an inflorescence approximately 80% of the time (n = 20), whereas honey bees visited both flowers only approximately 35% of the time (n = 20).

Figure 2. Boxplots of A, single-visit pollen deposition on haskap stigmas (n = 30 honey bees, n = 27 bumble bees, n = 8 control), B, rates of haskap flower visitation (n = 20 honey bees, n = 20 bumble bees), and C, percentage of instances visiting both haskap flowers in the two-flower inflorescence (n = 20 honey bees, n = 20 bumble bees), at haskap orchards in southern Nova Scotia, Canada, in 2016, 2017, or both. Boxes with different letters are significantly different (Fisher’s least-significant difference test, α = 0.05).
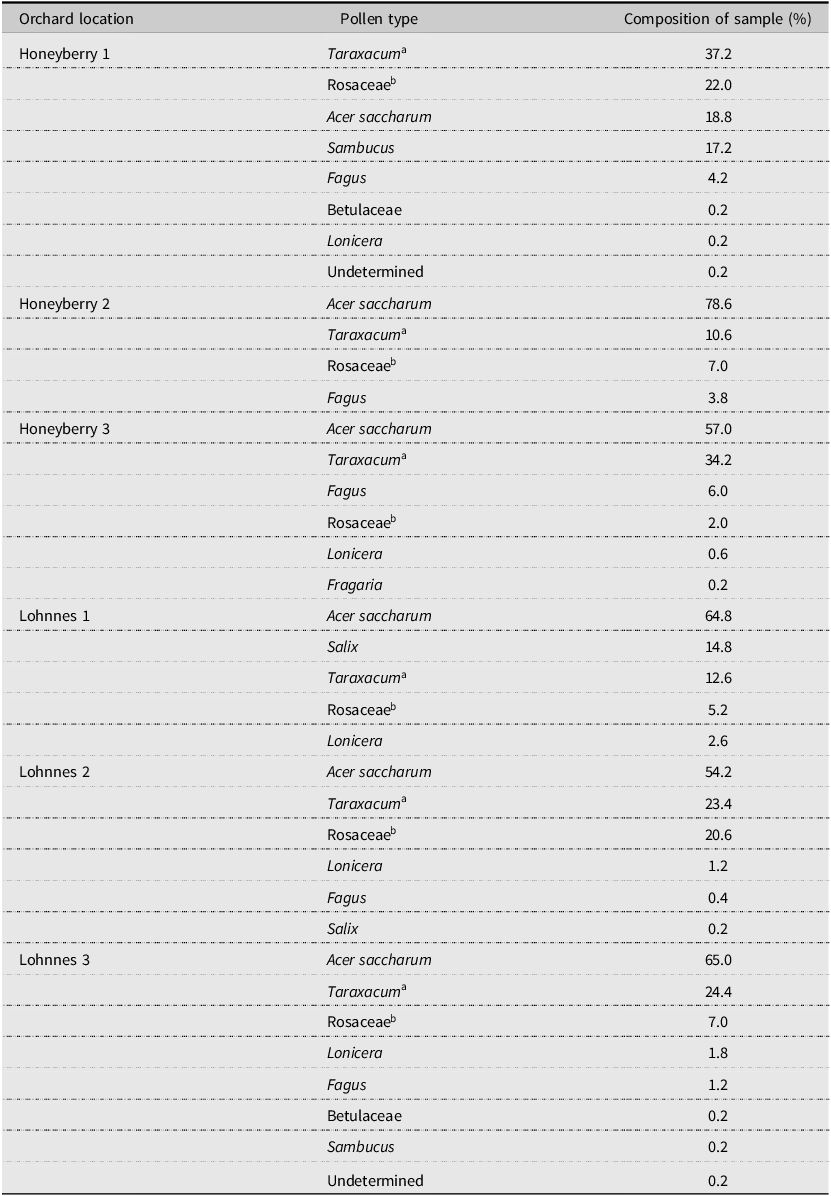
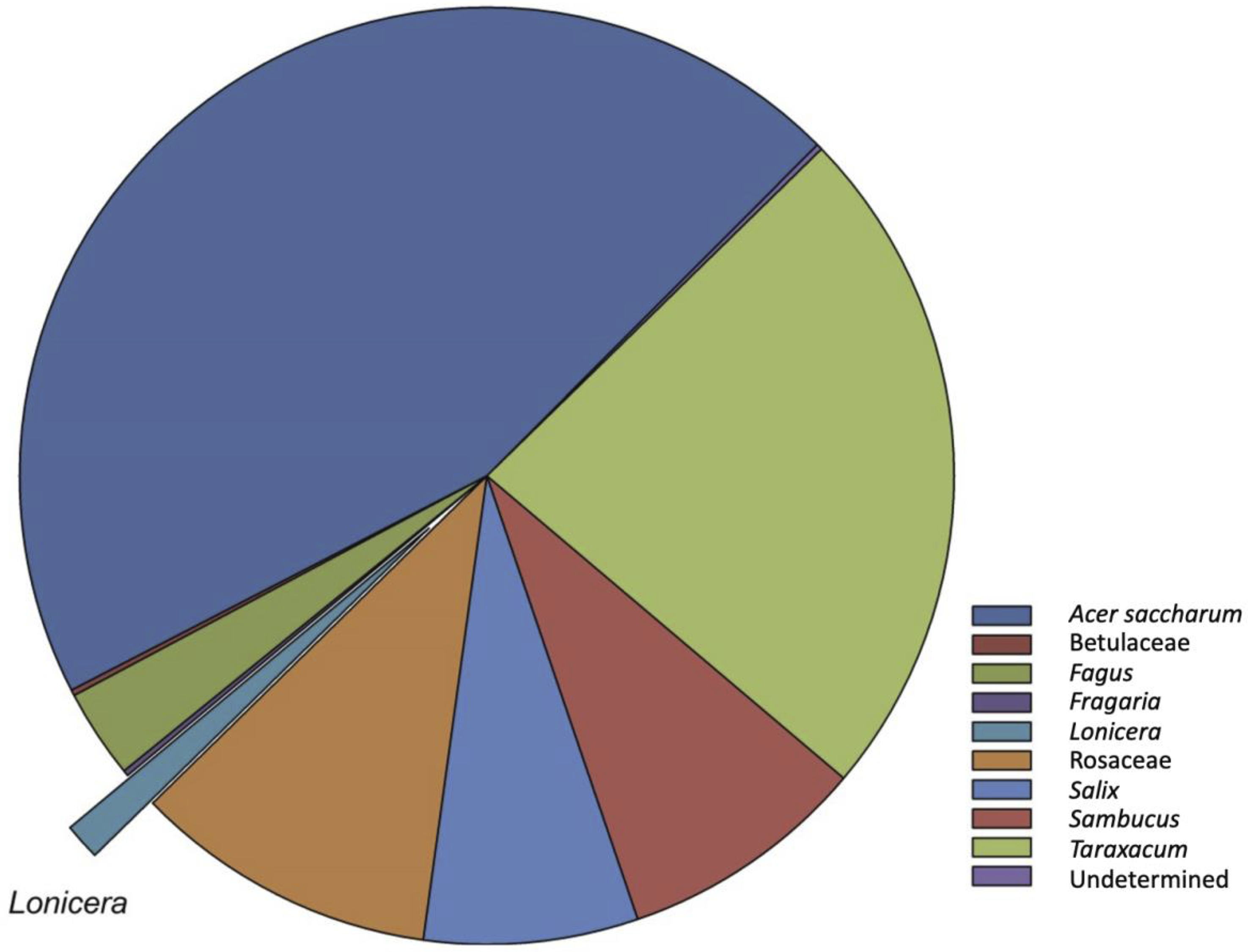
Pollen samples collected from pollen traps placed on honey bee hives were generally composed of 4–7 types of pollen, and only a few pollen types were unidentifiable (Table 2). Haskap pollen was present in five of six (83.3%) samples but comprised a relatively small percentage (1.3%) of the total amount of pollen collected (Fig. 3). Sugar maple, Acer saccharum (Sapindaceae), comprised 45% of the pollen collected across all sites. Dandelion, Taraxacum (Asteraceae), was the second most abundant pollen collected, at 22%. Pollen from Rosaceae and willow, Salix (Salicaceae), were also collected in high proportions at some sites (Table 2).
Table 2. Pollen collected by honey bees at six haskap orchards in southern Nova Scotia, Canada, 2017

a Taraxacum is a large genus of flowers in the aster family, but the pollen collected in the present study is most likely dandelion.
b Rosaceae in these samples are fruit tree types.

Figure 3. Mean pollen composition (%) of samples collected in pollen traps placed on the entrance of honey bee colonies at n = 6 haskap orchards in southern Nova Scotia, Canada, in 2017. Haskap pollen (Lonicera) comprised approximately 1.3% of the samples. The group “Rosaceae” includes all members of this family except Fragaria.
Nesting box studies
Cavity-nesting bees occupied nest tubes in six of the seven fields in 2017, with a total occupancy of 13.5% of all tubes available. Occupancy was 25.7% in 6-mm-diameter tubes and 1.4% in 8-mm-diameter tubes. Excluding the field with no nesting, 26 (72%) nest boxes containing 6-mm-diameter tubes had at least one capped nest tube, with up to 10 capped nest tubes (83.3%) occupied in a nest box. Only three (8.3%) of the nest boxes with 8-mm-diameter tubes contained one or more capped nest tubes. Nest tube capping was first observed on 22 June, approximately one month after haskap bloom, and increased steadily up to 13 September. Most nesting occurred during July and August (Fig. 4).

Figure 4. Mean (+/– standard error of the mean) nest tube occupancy over time by solitary bees in nest boxes in haskap orchards in southern Nova Scotia, Canada, 2017. There were seven orchards, each with 12 nest boxes, with each box containing 12 6-mm-diameter or 8-mm-diameter nesting tubes.
With the exception of three Megachile spp. individuals that emerged from a single 8-mm-diameter tube, all cavity-nesting bees in our study emerged from 6-mm-diameter tubes. Because little nesting occurred in the 8-mm-diameter nest tubes, only data from 6-mm-diameter nest tubes were analysed statistically. For the 6-mm-diameter tubes, no significant difference was observed in uptake by bees in the snug versus spaced nest tube arrangement (F 1,10 = 0.03, P = 0.86; Fig. 5A), tube nesting rate over time did not differ (F 1,60 = 1.47, P = 0.23), and no interaction between date and tube arrangement was observed (F 5,60 = 0.05, P = 0.99). A total of 156 bees and wasps emerged from 41 of 50 (82%) nest tubes placed in the chamber: 51 Osmia tersula, 27 Megachile spp., 75 Sapygidae wasps, two Chrysididae wasps, and one Ichneumonidae wasp (Tripplehorn and Johnson Reference Tripplehorn and Johnson2005; VanDyk et al. Reference VanDyk, Sellers and Bartlett2018). Wasp emergence began on 22 March 2018, 17 days after being placed in the environmental chamber, and was completed on 25 March (Fig. 5B). The first bees to emerge were O. tersula, on 26 March 2018. By 29 March, all bees had emerged (Fig. 5B).

Figure 5. Cavity-nesting bee nesting box occupancy in hapskap orchards (n = 7) in southern Nova Scotia, Canada, 2017: A, boxplots of nest tube uptake (capping) per orchard according to nest tube size (6 mm diameter versus 8 mm diameter) and arrangement (snug versus spaced), and B, emergence of bees and parasitic wasps from capped nest tubes (n = 50) held in an environmental chamber.
Discussion
Although haskap requires insect pollination and is an emerging fruit crop in Canada, few studies on pollinators of haskap have been completed to date. Bozek (Reference Bozek2012) made qualitative observations in Poland on visitation to haskap flowers by honey bees, bumble bees, and solitary bees, and Frier et al. (Reference Frier, Somers and Sheffield2016a) examined the pollination performance of honey bees, bumble bees, and Osmia lignaria on haskap but did not specifically study the crop’s wild pollinator community. The only other study we are aware of that has looked broadly at the haskap pollinator community was by Leung and Forrest (Reference Leung and Forrest2019), who studied diversity and abundance of haskap pollinators in the Yukon Territory, Canada. They observed four genera of cavity-nesting bees, as well as bumble bees, honey bees, syrphid flies, and butterflies, of which bumble bees were most common. In the present study, we found bumble bees and honey bees to be common, comprising approximately 10–20% of captures over two years, but our pan trap collections were dominated by Andrena and Lasioglossum, which made up more than 60% of bees captured in both years. The prominence of these genera has been observed in other crops before, during, and after bloom in Nova Scotia, including blueberry (Ericaceae) – where Lasioglossum and Andrena comprise more than 90% of all specimens captured (Cutler et al. Reference Cutler, Nams, Craig, Sproule and Sheffield2015) – and apple (Rosaceae) (Sheffield et al. Reference Sheffield, Kevan and Smith2003, Reference Sheffield, Kevan, Pindar and Packer2013; McCallum et al. Reference McCallum, McLean and Cutler2021). In the present study, we captured only 75% of the number of specimens in 2017 as we did in 2016, although numbers of some taxa increased in 2017 (e.g., Nomada (Apidae), Augochlorella (Halictidae)), and other taxa remained relatively constant (e.g., Lasioglossum). Annual fluctuations – mild or extreme – in pollinator population diversity and abundance are expected due to climate, ecological, and anthropogenic factors (Roubik Reference Roubik2001; Herrera Reference Herrera2008; Turley et al. Reference Turley, Biddinger, Joshi and López-Uribe2022).
We did not see a difference in pollinator abundance when pan traps were placed 0 m versus 60 m from the field edge. Habitat surrounding agricultural fields can be an important driver of pollinator communities within agricultural fields, such that abundance and diversity of pollinators and pollination services can decrease with increased distance from natural habitat (Carvalheiro et al. Reference Carvalheiro, Seymour, Veldtman and Nicolson2010; Bailey et al. Reference Bailey, Requier, Nusillard, Roberts, Potts and Bouget2014; Cutler et al. Reference Cutler, Nams, Craig, Sproule and Sheffield2015). However, the relatively short distance of 60 m from the field edge used in our study, owing to the small size of fields, probably was below the threshold distance where edge effects become evident. For example, in Nova Scotia blueberry fields, Cutler et al. (Reference Cutler, Nams, Craig, Sproule and Sheffield2015) found that the overall abundance of many pollinator taxa remained relatively constant up to 100 m from the field edge. Likewise, Chacoff and Aizen (Reference Chacoff and Aizen2006) found that abundance of pollinators within grapefruit plantations decreased only when distance exceeded 100 m from the forest edge.
Despite many solitary bees being collected in pan traps placed in haskap fields, we observed few solitary bees foraging on haskap flowers. This result highlights the limitations of relying on pan trap data as a proxy for pollination and the importance of floral visitation observations to confirm crop pollination (Broussard et al. Reference Broussard, Rao, Stephen and White2011; Bushmann and Drummond Reference Bushmann and Drummond2015). Our transect walks were conducted over 30 minutes on warm sunny days, with care taken to move slowly and carefully in order to not disturb or overlook pollinators along the transect. We did qualitatively notice during our fieldwork many Andrena and Lasioglossum visiting dandelion (Taraxacum spp.), willow, apple, and cherry (Rosaceae) blossoms around certain orchards during haskap bloom. This suggests that the role of solitary bees in pollination of haskap may be limited and that their presence in and around haskap fields may be more a function of noncrop floral resources than an affinity for haskap pollen and nectar. Frier et al. (Reference Frier, Somers and Sheffield2016a) noted that, when released in haskap orchards, Osmia lignaria were more interested in other floral sources than in haskap. Due to the relatively large size of haskap flowers and the exserted position of the stigma relative to the petals, small solitary bees probably are not effective pollinators of haskap. The small size and inconspicuous nature of many solitary bees also mean that, despite our best efforts, some small solitary bees may not have been detected during our transect walks.
Unlike solitary bees and hover flies, we frequently observed honey bees and bumble bees on haskap. Our transect walk observations and pollinator behaviour studies suggest that honey bees and bumble bee queens are important visitors of haskap flowers and effective pollinators of this crop (Bozek Reference Bozek2012; Frier et al. Reference Frier, Somers and Sheffield2016a; Leung and Forrest Reference Leung and Forrest2019). Similar numbers of honey bees and bumble bees were observed on haskap flowers in 2016, but twice as many honey bees were observed on transect walks in 2017. This may be due to the doubling of the honey bee stocking rate to 0.8 hives/ha at the Honeyberry Hurst orchard that year, which was nonetheless well below the stocking rate recommended for pollination of other crops in the region, such as 4–10 hives/ha for lowbush blueberry (Drummond Reference Drummond2012; Eaton and Nams Reference Eaton and Nams2012).
As Frier et al. (Reference Frier, Somers and Sheffield2016a) did, we found honey bees and bumble bee queens deposit roughly the same amount of pollen per single visit, although the amount of pollen deposited by these bees varied significantly. In other fruit crops, pollen deposition differences between bees are more pronounced: in lowbush blueberry, Bombus queens (Javorek et al. Reference Javorek, Mackenzie and Vander Kloet2002) deposit more than four times as much pollen per single visit than honey bees do, a difference that may be attributable to many honey bees being nectar foragers and the “buzz pollination” behaviour of bumble bees that is more suitable for blueberry and other plants that have poricidal anthers.
Although honey bees and bumble bee queens deposited equal amounts of pollen per visit, we found that bumble bees visited threefold more flowers per minute than honey bees did. This result aligns with Frier et al. (Reference Frier, Somers and Sheffield2016a), who found haskap flower visit duration for honey bees to be 15 seconds, compared to six seconds for bumble bees. In the present study, bumble bee queens also visited both flowers of the haskap flower inflorescence twice as often as honey bees did, at a frequency of around 80% versus 35%. These results are important not only in that bumble bees are faster pollinators of haskap but also because visitation to both flowers of the haskap inflorescence results in a higher percent fruit set and typically larger fruit with more seeds (Frier et al. Reference Frier, Somers and Sheffield2016a). On the other hand, the pollinator force of a honey bee hive is orders of magnitude greater than that of a Bombus queen or colony, which means the increased efficiency of bumble bees may not necessarily translate into better pollination service compared to that provided by honey bees.
Factors such as weather are also key determinants of pollination success. Honey bees will generally forage at a minimum ambient temperature of 13 °C (Kevan and Baker Reference Kevan and Baker1983), whereas temperate bumble bee species can forage at temperatures as low as 5 °C (Heinrich Reference Heinrich1972). Some temperate bumble bee species forage best at temperatures above 10–12 °C (Kenna et al. Reference Kenna, Pawar and Gill2021; Koppert 2025), but pollination performance of bumble bees usually exceeds that of honey bees in the cool, wet conditions often experienced in Nova Scotia and other parts of Canada in spring.
Fidelity of a pollinator to a crop can also affect pollination success. The results of our pollen analysis suggest that, despite frequently foraging on haskap flowers, honey bees placed in haskap orchards also forage a variety of floral resources in and around the agricultural landscape. This is not surprising because bees regularly collect a diversity of pollen types to satisfy their nutritional requirements (Somerville Reference Somerville2001). In the present study, haskap pollen comprised only 1.2% (range = 0–2.6%) of pollen in samples from our honey bee hives, suggesting honey bees cannot easily access haskap pollen or that haskap pollen is of low value to honey bees. Similarly, honey bees are effective pollinators of lowbush blueberry in Nova Scotia (Aras et al. Reference Aras, De Oliveira and Savoie1996; Eaton and Murray Reference Eaton and Murray1996; Eaton and Nams Reference Eaton and Nams2012), but the amount of blueberry pollen returned to the hive can be very low when hives are placed in blueberry fields (Colwell Reference Colwell2014). Therefore, the proportion of crop pollen in honey bee hive samples is not always a useful proxy of pollinator effectiveness for the crop. Because we did not collect pollen samples from bumble bees or their colonies, we do not know if bumble bee colonies use proportionally more haskap pollen.
We investigated the potential of placing cavity-nesting bee nesting boxes around haskap fields to support populations of solitary bees that may be useful haskap pollinators. We found occupancy was much greater in 6-mm-diameter tubes than in 8-mm-diameter tubes. Uptake into cavities or tubes of different sizes can vary greatly and depends on the bee community composition, because bees select cavities matching their own body width. In Nova Scotia, nest tubes that are 5–7 mm in diameter are effective sites for cavity-nesting bees (Sheffield et al. Reference Sheffield, Kevan, Westby and Smith2008; McCallum et al. Reference McCallum, McLean and Cutler2018), with tubes as large as 9 mm in diameter being preferred by some Megachile spp. (Sheffield et al. Reference Sheffield, Kevan, Westby and Smith2008). Patterning of the area around the entrance of nest box tubes can be important for nest recognition, and tube spacing may affect nesting activity (Bosch and Kemp Reference Bosch and Kemp2001; Sheffield et al. Reference Sheffield, Kevan, Westby and Smith2008), but we found that arranging nesting tubes in snug versus spaced arrangements had no bearing on nesting uptake.
Many nesting box designs and materials have been developed to study cavity-nesting bees (MacIvor Reference MacIvor2017), and testing other nest box designs would be worthwhile. However, our results suggest nesting boxes may have limited viability as a tactic to improve haskap pollination in Nova Scotia. Less than 15% of 1000 nesting tubes deployed in the present study were occupied, and only 78 total bees emerged. An equal number of parasitic wasps emerged from our nests (all of which emerged before the bees), corroborating previous reports of high levels of parasitism in trap nests (Sheffield et al. Reference Sheffield, Kevan, Westby and Smith2008; McCallum et al. Reference McCallum, McLean and Cutler2018). The low numbers of Megachilidae recorded from our pan traps and transect walks also suggest that cavity-nesting bees make a limited contribution to the natural haskap pollination force at our test sites. Furthermore, the activity of cavity-nesting bees does not seem to be well synchronised with haskap bloom: nest tube capping began almost a month after haskap bloom ended. Indeed, although some Osmia spp. – including O. tersula recovered from our nest boxes – are active in early spring, most other Megachilidae are active later in summer and into autumn (Sheffield et al. Reference Sheffield, Kevan and Smith2003). McCallum et al. (Reference McCallum, McLean and Cutler2018) reported that most capping of nesting tubes in blueberry fields in Nova Scotia also occurs well after crop bloom. To be effective for crop pollination, a bee would have to be actively foraging on the crop for the purpose of provisioning its nest during bloom of that crop. The viability of commercially managed Megachile rotundata (Fabricius) for haskap pollination should be tested; however, cool spring weather during haskap bloom, coupled with the need for temperatures warmer than 16 °C for M. rotundata flight (Argall et al. Reference Argall, Mackenzie, Javorek and Chiasson1996), would likely limit the use of this cavity-nesting bee for haskap pollination in Nova Scotia.
In conclusion, haskap orchards in southern Nova Scotia support a diverse pollinator community, but wild solitary bees were observed infrequently on haskap blossoms and probably do not contribute strongly to pollination of the crop. Although honey bees prefer warmer weather and move between haskap flowers at a lower rate than Bombus queens do, they visit haskap flowers often and deposit as much pollen as bumble bees do. The large numbers of foragers in honey bee hives will help provide an adequate pollination force for haskap growers when native bumble bees numbers are low. Although testing other nest box designs would be useful, our results suggest establishment of nesting boxes around fields probably would not boost the local community of cavity-nesting bees substantially and, in fact, may increase parasitism of pollinators occupying nesting blocks, thereby inadvertently harming the native bee population (MacIvor and Packer Reference MacIvor and Packer2015).
Data availability
Raw data are available to others upon request.
Acknowledgements
This research was supported through Research Acceleration Program funding to G.C.C. (Project RA16-010) from the Nova Scotia Department of Agriculture, in partnership with LaHave Forests/Haskapa. The authors thank the LaHave staff for their assistance and cooperation during our fieldwork. They also thank A. Loureiro, D.A. Joseph, C. Andrews, J. Baak, E. Vance, J. Hamlin, R. Slater, A. McOnie, and J. MacKeil for assistance with laboratory and fieldwork.
Author contributions
G.C.C. and S.O. conceptualised and designed the experiments. S.O. collected the data. S.O., R.R., and G.C.C. analysed the data and wrote the manuscript.
Competing interests
The authors declare there are no competing interests.