Introduction
The Mediterranean fruit fly, medfly (Ceratitis capitata [Wiedemann]) (Diptera: Tephritidae), is a highly invasive pest of many agricultural commodities that has expanded, in less than two centuries, its geographic range from the ancestral habitats of sub-Saharan Africa to nearly all temperate and tropical regions worldwide (Gomulski et al., Reference Gomulski, Dimopoulos, Xi, Soares, Bonaldo, Malacrida and Gasperi2008). Its extreme polyphagy, high invasive potential, and development of insecticide resistance in certain populations continue to challenge sustainable management efforts (Szyniszewska and Tatem, Reference Szyniszewska and Tatem2014; Giunti et al., Reference Giunti2023). Medfly has become a model organism in pest management research, contributing to advances in invasion biology, integrated pest management (IPM) approaches, and the development of innovative pest suppression and eradication strategies.
The sterile insect technique (SIT) is considered one of the most environmentally friendly and species-specific methods for IPM applications among those available. SIT involves mass-rearing, sterilisation – typically by irradiation – and release of sterile males to field, where mating with wild females results in no viable offspring, leading to population suppression or even eradication over time (Pereira et al., Reference Pereira, Yuval, Liedo, Teal, Shelly, McInnis and Hendrichs2013; Enkerlin, Reference Enkerlin2021).
Releasing ‘female-free’ sterile males is important for the successful application of SIT (Hendrichs et al., Reference Hendrichs, Franz and Rendon1995). Medfly is a model for male-only releases, due to extended research for many decades that led to the development of the Vienna 7 and Vienna 8 genetic sexing strains (GSSs) that are currently used in all major medfly mass-rearing facilities (Franz et al., Reference Franz, Bourtzis, Cáceres, Dyck, Hendrichs and Robinson2021), with Vienna 8 GSS representing the most widely applied and current strain in industrial mass-rearing facilities worldwide, while Vienna 7, although historically important, is largely replaced by Vienna 8 due to improved performance and stability. For the present study, the Vienna 8 GSS lacking the D53 inversion was selected due to its higher productivity, while the potentially reduced genetic stability associated with the absence of the inversion was not expected to affect the outcomes under the controlled experimental conditions applied here. Industrial-scale mass-rearing facilities produce billions of sterile males weekly, which are irradiated and shipped worldwide for release (Pereira et al., Reference Pereira, Yuval, Liedo, Teal, Shelly, McInnis and Hendrichs2013; Franz et al., Reference Franz, Bourtzis, Cáceres, Dyck, Hendrichs and Robinson2021). A major success example is the SIT-based area-wide IPM programme in Valencia, Spain, which reduced insecticide use by over 90% and enhanced citrus exports (Plá et al., Reference Plá, de Oteyza, Tur, Martínez, Laurín, Alonso, Martínez, Martín, Sanchis and Navarro2021). Such sustained suppression depends on large-scale production capacity, exemplified by the El Pino facility in Guatemala – the world’s largest – producing more than 2 billion sterile C. capitata males per week (Parker et al., Reference Parker, Mamai and Maiga2021).
The efficacy of SIT depends on the biological quality and field performance of released sterile males. Traits such as survival, mating competitiveness, sexual signalling, and flight ability are essential, but can be negatively affected by mass-rearing, sterilisation, and handling (USDA, Reference USDA2003; Pereira et al., Reference Pereira, Yuval, Liedo, Teal, Shelly, McInnis and Hendrichs2013; Dyck et al., Reference Dyck, Hendrichs and Robinson2021). Strategies to mitigate these effects include dietary enrichment (Yuval et al., Reference Yuval, Kaspi, Field, Blay and Taylor2002), hormonal treatment (Teal et al., Reference Teal, Gomez-Simuta and Proveaux2000), exposure to species-specific semiochemicals (Shelly, Reference Shelly2001), and improvements in rearing technology, such as cellulose III pupation substrates for the Vienna 8 GSS and irradiation under hypoxic conditions (Bourtzis and Vreysen, Reference Bourtzis and Vreysen2021; Giustina et al., Reference Giustina, Mastrangelo, Ahmad, Mascarin and Caceres2021; Pascacio-Villafan et al., Reference Pascacio-Villafan, Quintero-Fong, Guillen, Rivera-Ciprian, Aguilar and Aluja2021). Optimising male performance remains a cornerstone of SIT programmes, as emphasised by ongoing FAO/IAEA (Food and Agriculture Organization/ International Atomic Energy Agency) research.
Environmental stress, particularly thermal stress, is a major determinant of sterile male performance, affecting survival, flight, mating competitiveness, and longevity (Nyamukondiwa et al., Reference Nyamukondiwa, Kleynhans and Terblanche2010). The widely used Vienna 8 temperature-sensitive lethal (tsl) strain (Caceres, Reference Caceres2002) shows increased heat sensitivity, particularly in females, while heterozygous sterile males may also be affected to a lesser extent. This sensitivity has been attributed to the tsl mutation or to heat treatments applied at the egg stage for female elimination (Nyamukondiwa et al., Reference Nyamukondiwa, Weldon, Chown, le Roux and Terblanche2013; Franz et al., Reference Franz, Bourtzis, Cáceres, Dyck, Hendrichs and Robinson2021; Cáceres et al., Reference Cáceres, Bourtzis, Gouvi, Vreysen, Bimbilé Somda, Hejníčková, Marec and Meza2023). Additional stress from irradiation and handling can further reduce thermal performance under fluctuating field temperatures (Collins et al., Reference Collins, Weldon, Banos and Taylor2008). Rapid acclimation and physiological plasticity, reflected in critical thermal minimum (CTmin), critical thermal maximum (CTmax), or chill-coma recovery times (CCRT), are key for predicting performance under climatic extremes (Nyamukondiwa and Terblanche, Reference Nyamukondiwa and Terblanche2010; Chidawanyika and Terblanche, Reference Chidawanyika and Terblanche2011).
Optimising larval diet is a central priority in SIT programmes to ensure high-quality sterile males while maintaining economical large-scale production. Larval diet formulation is one of the most essential and costly components of mass rearing, and its design must balance nutrient adequacy, ingredient availability, storage requirements, labour demands, and overall cost (Nikolouli et al., Reference Nikolouli, Sassù, Ntougias, Stauffer, Cáceres and Bourtzis2021; Parker et al., Reference Parker, Mamai and Maiga2021). An effective diet must provide all the necessary nutrients to support proper larval development, successful adult emergence, and high adult performance, while remaining economically feasible and free from negative impacts on insect fitness or colony productivity (Cohen, Reference Cohen2018; Nikolouli et al., Reference Nikolouli, Sassù, Ntougias, Stauffer, Cáceres and Bourtzis2021; Parker et al., Reference Parker, Mamai and Maiga2021). While Enterobacter supplementation has been shown to improve developmental parameters and reproductive traits in mass-reared tephritids, its potential effects on thermal stress physiology – a critical factor determining field performance – remain unexplored.
Gut symbiotic bacteria play critical roles in insect nutrition, reproduction, immunity, and fitness (Douglas, Reference Douglas1998, Reference Douglas2009; Bourtzis and Miller, Reference Bourtzis and Miller2003; Behar et al., Reference Behar, Yuval and Jurkevitch2008). In C. capitata, Enterobacteriaceae such as Klebsiella, Enterobacter, and Pantoea aid digestion and nutrient assimilation, and may influence pathogen resistance (Behar et al., Reference Behar, Yuval and Jurkevitch2005; Ami et al., Reference Ami, Yuval and Jurkevitch2010; Augustinos et al., Reference Augustinos, Kyritsis, Papadopoulos, Abd-Alla, Cáceres and Bourtzis2015), mating behaviour, and competitiveness – traits crucial for SIT success (Ami et al., Reference Ami, Yuval and Jurkevitch2010). Symbionts in other insects enhance longevity, resistance to natural enemies, and social interactions (Oliver et al., Reference Oliver, Russell, Moran and Hunter2003; Dillon et al., Reference Dillon, Vennard, Buckling and Charnley2005; Behar et al., Reference Behar, Yuval and Jurkevitch2008; Augustinos et al., Reference Augustinos, Kyritsis, CáCeres and Bourtzis2021).
Artificial rearing and irradiation often alter gut microbiota, reducing Enterobacteriaceae and favouring opportunistic taxa such as Pseudomonas and Serratia, which can impair physiological and behavioural performance (Marchini et al., Reference Marchini, Rosetto, Dallai and Marri2002; Ami et al., Reference Ami, Yuval and Jurkevitch2010). This has driven interest in gut bacteria as probiotics or functional dietary additives to enhance SIT efficiency (Niyazi et al., Reference Niyazi, Lauzon and Shelly2004; Meats et al., Reference Meats, Streamer and Gilchrist2009; Ami et al., Reference Ami, Yuval and Jurkevitch2010; Augustinos et al., Reference Augustinos, Kyritsis, Papadopoulos, Abd-Alla, Cáceres and Bourtzis2015; Kyritsis et al., Reference Kyritsis, Augustinos, Cáceres and Bourtzis2017, Reference Kyritsis, Augustinos, Ntougias, Papadopoulos, Bourtzis and Cáceres2019; Nikolouli et al., Reference Nikolouli, Sassù, Ntougias, Stauffer, Cáceres and Bourtzis2021). In C. capitata, larval diet supplementation with live Enterobacteriaceae accelerates development, improves survival, and increases male mating success (Niyazi et al., Reference Niyazi, Lauzon and Shelly2004; Meats et al., Reference Meats, Streamer and Gilchrist2009; Ami et al., Reference Ami, Yuval and Jurkevitch2010; Augustinos et al., Reference Augustinos, Kyritsis, Papadopoulos, Abd-Alla, Cáceres and Bourtzis2015; Kyritsis et al., Reference Kyritsis, Augustinos, Cáceres and Bourtzis2017, Reference Kyritsis, Augustinos, Ntougias, Papadopoulos, Bourtzis and Cáceres2019; Nikolouli et al., Reference Nikolouli, Sassù, Ntougias, Stauffer, Cáceres and Bourtzis2021). Similar findings have been reported for Bactrocera oleae, with Enterobacteriaceae supplementation improving fecundity (Sacchetti et al., Reference Sacchetti, Ghiardi, Granchietti, Stefanini and Belcari2014; Ben-Yosef et al., Reference Ben-Yosef, Pasternak, Jurkevitch and Yuval2015) and other symbionts, such as Candidatus Erwinia dacicola and Pseudomonas putida, enhancing larval development and female fecundity (Sacchetti et al., Reference Sacchetti, Ghiardi, Granchietti, Stefanini and Belcari2014; Ben-Yosef et al., Reference Ben-Yosef, Pasternak, Jurkevitch and Yuval2015).
Although most studies focus on live bacteria, inactive bacterial biomass has received limited attention. Kyritsis et al. (Reference Kyritsis, Augustinos, Ntougias, Papadopoulos, Bourtzis and Cáceres2019) showed that supplementation with inactive Enterobacter sp. AA26 biomass, partially or fully replacing yeast, improved biological quality and productivity. Enterobacter sp. AA26 can be efficiently cultured under diverse conditions, including commercial yeast media or agro-industrial wastewater. As a probiotic, it provides essential and nonessential amino acids and vitamins sufficient to support mass-rearing and SIT applications (Azis et al., Reference Azis, Zerva, Melidis, Caceres, Bourtzis and Ntougias2019). Its use offers a cost-effective, practical, and potentially locally producible alternative, reducing dependence on external suppliers and enhancing the sustainability of SIT programmes.
The aim of the current paper was to evaluate whether partial replacement of brewer’s yeast with autoclaved Enterobacter biomass in the larval diet (5% yeast replacement, maintaining equivalent protein content), and complete replacement of yeast in the adult diet, can enhance the thermal stress responses of sterile C. capitata males. We hypothesised that Enterobacter supplementation would enhance thermal tolerance through improved nutritional status and metabolic reserves, thereby mitigating the negative effects of sterilisation and mass rearing on male performance and mating competitiveness. To test this hypothesis, we measured the CTmin, CTmax, and CCRT as indicators of thermal tolerance and recovery capacity. In addition, sterile males were compared with wild males to evaluate whether Enterobacter supplementation could reduce the possible performance gap between them. Given the crucial roles of food quality and nutrient provision in sterile fly nutrition, stress resistance, and behaviour, we expected that microbial supplementation in their diet would improve their resilience and field effectiveness under thermally challenging conditions, thereby enhancing the success of biological control programmes in the context of ongoing environmental change.
Materials and methods
Enterobacter production
Bacterial strain and maintenance and production medium and culture conditions
Enterobacter sp. AA26 was used for all experiments and production runs; the strain was maintained on standard laboratory medium and transferred to fresh plates at regular intervals to ensure viability and phenotypic stability. To minimise genetic drift and batch-to-batch variability, a cryopreserved stock was used to initiate each production batch. For every production cycle, a pre-culture was prepared by inoculating a single colony of strain AA26 into liquid medium and incubating it under aerobic conditions until it reached the mid- to late-exponential phase. This pre-culture served as the inoculum for the main culture, using a defined volume ratio to ensure reproducible starting cell densities.
Enterobacter cultures were grown in half-strength Luria–Bertani (½ LB) broth, which had been prepared in bulk and sterilised prior to inoculation. Production volumes of 50 l per batch were used. Cultures were incubated under aerated conditions for 48 h. At the end of the 48-h period, growth was assessed spectrophotometrically using a Helios instrument by measuring optical density (OD) as a proxy for total biomass.
Biomass quantification and yield estimation
OD measurements at 48 h were used to estimate wet and dry biomass, and process calculations indicated that approximately 50 l of liquid culture were required to obtain 90–100 g of Enterobacter dry weight. OD–biomass correlations established in preliminary runs were used to monitor production performance and to determine the harvesting time point. At the end of the incubation period, the cultures were subjected to a heat treatment at 100°C to inactivate bacterial cells and stabilise the biomass prior to drying. Following heat inactivation, bacterial biomass was collected by centrifugation at 6,000 rpm for 20 min and washed three times with phosphate-buffered saline. The final pellet was transferred into 50 ml polypropylene tubes (Falcon) and dehydrated in a laboratory oven at 65°C for 24–48 h until they were completely dry. Recovered Enterobacter biomass was dried at 65°C until mass stabilisation, as determined by consecutive weightings showing no further weight loss, to obtain a dry, stable bacterial product. The resulting dried biomass constituted the final Enterobacter preparation used for downstream applications in enhanced larval diet formulations.
Insect strains and colony maintenance
Vienna 8 GSS origin and maintenance
The Vienna 8 GSS of C. capitata, provided by the Insect Pest Control Laboratory (FAO/IAEA), was used in all sterile male experiments. This strain is widely applied in SIT mass-rearing programmes due to its favourable balance between productivity and genetic traits.
The bacterial strain has been continuously maintained for over 31 generations in insectaries at the Department of Plant Protection, University of Patras (Greece), under controlled laboratory conditions. Colonies were maintained both under filtering, to ensure genetic integrity, and without filtering, to allow colony upscaling for experimental purposes (Franz et al., Reference Franz, Bourtzis, Cáceres, Dyck, Hendrichs and Robinson2021). Rearing conditions kept constant at 25 ± 1°C, 55 ± 5% relative humidity (RH), and a 14:10 h light:dark photoperiod.
Larvae were reared on a standard larval diet (SLD) commonly used in medfly mass-rearing, while adults were maintained on standard adult diet (SAD) consisting of hydrolysed yeast and crystalline sugar (4:1) with water supplied via soaked sponges placed in 150-ml containers. Colony maintenance was conducted using a filter rearing system, in which a low-density mother colony was maintained under reduced numbers, used exclusively to supply individuals to initiate an amplification cycle. In contrast, non-filtered material was maintained under a continuous mass-rearing system for a maximum of four amplification cycles for experimental purposes. No material originating from non-filtered amplification cycles was returned to the filtered colonies. The use of filtering is relevant, as it minimises genetic and behavioural deterioration associated with long-term mass rearing (Franz et al., Reference Franz, Bourtzis, Cáceres, Dyck, Hendrichs and Robinson2021).
For tsl-based female elimination, eggs were collected daily within a 6 h oviposition window. Eggs were maintained under high-humidity conditions to allow normal embryonic development. In accordance with standard tsl protocols used in GSSs, 24–30-h-old embryos were subjected to a heat treatment at 34°C for 24 h, a temperature known to selectively affect female embryos homozygous for the tsl mutation without compromising male viability (Augustinos et al., Reference Augustinos, Targovska, Cancio-Martinez, Schorn, Franz, Cáceres, Zacharopoulou and Bourtzis2017; Franz et al., Reference Franz, Bourtzis, Cáceres, Dyck, Hendrichs and Robinson2021; Plá et al., Reference Plá, de Oteyza, Tur, Martínez, Laurín, Alonso, Martínez, Martín, Sanchis and Navarro2021). After completion of the thermal treatment, eggs were transferred to the larval diet.
Wild C. capitata population collection and establishment
Wild C. capitata colonies were established from pupae obtained from infested persimmon fruits collected in the plain of Central Macedonia, northern Greece. Fruits were transported to the laboratory and maintained at 25 ± 1°C and 60–70% RH. Infested fruits were placed in plastic trays containing a 2 cm layer of sterilised dry sand to allow larval exit and pupation.
Pupae were collected by sieving the sand and maintained under the same laboratory conditions until adult emergence. Adults were housed in wooden cages (30 × 30 × 30 cm) covered with wire mesh on all sides except for one glass window, with each cage containing 100–150 flies of mixed sex. Flies were provided ad libitum with water and a SAD consisting of hydrolysed yeast, crystalline sugar, and water at a rate of 1:4:5, respectively.
Eggs were collected using artificial oviposition devices consisting of perforated red plastic hemispheres (5 cm diameter) fitted into Petri dish lids. Eggs collected over 24 h were transferred with a fine brush to a carrot-based larval diet following the FAO/IAEA/USDA (2019) formulation. Approximately 100 eggs were placed per dish, and larval development proceeded for 7–8 days at 25°C before pupation in sand. The wild colony was maintained for three consecutive generations under these laboratory conditions prior to its use in experiments, ensuring sufficient population size while minimising changes in gut microbiota. All manipulations, including egg collection and larval transfers, were conducted under sterile conditions to reduce the risk of microbial contamination.
The wild (Macedonia) strain was not included in the factorial dietary manipulation applied to sterile males. Instead, it was maintained under standard larval and adult rearing conditions and used as an external biological reference to benchmark sterile male performance relative to a non-genetically modified population. Comparisons involving the wild strain were conducted separately from the sterile male factorial analyses.
Experimental diets
Larval diets
Larvae destined for sterile male production were reared on one of two larval diets:
1. SLD: Cellulose-based diet commonly used in medfly mass-rearing.
2. Enterobacter-enhanced larval diet (ELD): Identical to SLD, except that 5% of brewer’s yeast was replaced with dried biomass of Enterobacter sp. AA26, while maintaining equal total protein content (Augustinos et al., Reference Augustinos, Kyritsis, CáCeres and Bourtzis2021).
Detailed diet compositions are provided in table 1.
Ingredients of the two larval diets. A ‘standard diet’ is one in which the main ingredients are more common in different mass-rearing facilities

Note: The ‘Enterobacter-enhanced diet’ was based on the standard diet but was supplemented with 5% Enterobacter-dried biomass, keeping the total amount of the protein the same in the two diets.
Adult diets
Upon emergence, adult males received one of two liquid dietary treatments:
1. SAD: Hydrolysed yeast, sugar, and water (1:4:7).
2. Enterobacter adult diet (EAD): Hydrolysed yeast fully replaced with Enterobacter sp. AA26 biomass mixed with sugar and water at the same ratios.
Thus, while larval diets involved partial yeast replacement, adult diets involved complete yeast replacement. The reasoning behind the different approaches is that larval development is highly sensitive to nutrient composition, so only a partial replacement of yeast with Enterobacter sp. AA26 was used to maintain optimal protein levels while testing the effect of the bacteria. In contrast, adult males are less sensitive to complete yeast replacement, which allowed us to fully substitute yeast with the bacterial biomass in the adult diet.
Experimental groups
Combining larval and adult dietary regimes resulted in four experimental treatments in a 2 × 2 factorial design for sterile males:
1. Vienna 8 GSS–SLD_SAD: SLD; SAD
2. Vienna 8 GSS–SLD_EAD: SLD; EAD
3. Vienna 8 GSS–ELD_SAD: Enterobacter-ELD; SAD
4. Vienna 8 GSS–ELD_EAD: Enterobacter-ELD; EAD
To clarify the experimental design, table 2 summarises the sterile male dietary treatments in the 2 × 2 factorial design along with the wild (Macedonia) strain, which served as an external biological reference. Sterile male comparisons were conducted within the factorial groups, while the wild strain was analysed separately to benchmark performance.
Experimental groups, larval and adult diets, and their role in the study

Irradiation protocol
Vienna 8 GSS pupae were sterilised using a Caesium-137 irradiator located at the General University Hospital of Patras, originally designed for blood irradiation. The irradiator delivered a dose of 100 Gy over approximately 36 min, with sufficient dose uniformity to process up to 2.2 L of pupae per session (approximately 100,000 individuals). The proximity of the irradiation facility to the rearing site allowed efficient transport and timely processing.
Thermal stress assays
Critical thermal limits (CTmin and CTmax)
To assess CTmin and CTmax, we used a standard CTLs protocol previously described for C. capitata (Papadogiorgou et al., Reference Papadogiorgou, Verykouki and Papadopoulos2025), based on the methodological framework of Nyamukondiwa and Terblanche (Reference Nyamukondiwa and Terblanche2010). In brief 5-day-old males were placed into jacketed chambers containing organ pipes (10 ml). These chambers were connected to a programmable circulating refrigerated bath (Lauda Eco RE 2025 S) filled with Kryo 30 (a mixture of water, ethylene glycol, and corrosion inhibitors), enabling precise temperature control. Multiple chambers were connected simultaneously to assess the CTmin and CTmax.
Temperature monitoring within each chamber was conducted using type T thermocouples (36 SWG) inserted into one of the organ pipes. Thermocouples were linked to a Picotech TC-08 interface (Pico Technology, Cambridge, UK) and used to record the temperature at a frequency of 1 Hz via PicoLog software. CTmin and CTmax assessments commenced at 25°C, with an initial 10-min equilibration period before the temperature was either reduced (for CTmin) or increased (for CTmax) at a controlled rate of 0.25°C/min. The CTLs were defined as the temperature at which individuals ceased to exhibit coordinated movement in response to gentle prodding with a thermally inert object (e.g. a fine paintbrush) (Nyamukondiwa and Terblanche, Reference Nyamukondiwa and Terblanche2010).
For each combination of larval and adult diet (2 × 2), 20 males were tested for CTmin and 20 males for CTmax, resulting in a total of 80 males (n = 80).
Chill-coma recovery time
On the fifth day post-emergence, groups of 10 males were transferred into empty 35-ml glass vials sealed with a cotton wool stopper. Vials were submerged in an ice-water slurry at 0°C for 4 h in complete darkness within a Styrofoam cooler. The internal temperature of the vials was monitored using either an analog thermometer or a HOBO data logger placed in an empty reference vial immersed in the slurry.
Following the cold exposure, flies were individually placed in petri dishes (5 cm Ø) in a supine position using a fine paintbrush. Recovery was monitored for 1 h under controlled laboratory conditions (25 ± 1°C, 65 ± 5% RH). Petri dishes were sealed with a transparent plastic lid to prevent escape. Recovery was defined as the ability of a fly to regain an upright position or initiate flight without any external stimulation. The time required for each individual to assume an upright posture at 25°C was recorded as the CCRT, following the methodology described previously (Overgaard and MacMillan, Reference Overgaard and MacMillan2017). Mortality during the CCRT assay (i.e. flies that did not survive the 0°C exposure) and individuals (flies that remained alive but failed to recover within the 1-h observation period) were documented.
For each combination of larval and adult diet (2 × 2), 20 males were tested for CCRT, resulting in a total of 80 males per replicate. The experiment was replicated six times, yielding a total of 480 males tested.
Data analysis
Statistical analyses were performed using IBM SPSS Statistics version 29. Data were examined for normality and homogeneity of variance prior to analysis. In all cases, the significance level was set at α = 0.05.
For sterile (Vienna 8 GSS) males, the effects of larval diet (standard vs Enterobacter-enhanced), adult diet (standard vs Enterobacter), and their interaction on CTmin, CTmax, and CCRT were assessed using General Linear Model (GLM) univariate analyses (2 × 2 factorial design).
To directly compare all experimental groups, including the wild (Macedonia) strain, separate one-way GLM was conducted with treatment as a fixed factor comprising five levels (four sterile dietary combinations and wild males).
When significant effects were detected, pairwise comparisons were performed using Bonferroni-adjusted post hoc tests.
Results
Critical thermal limits (CTmin and CTmax)
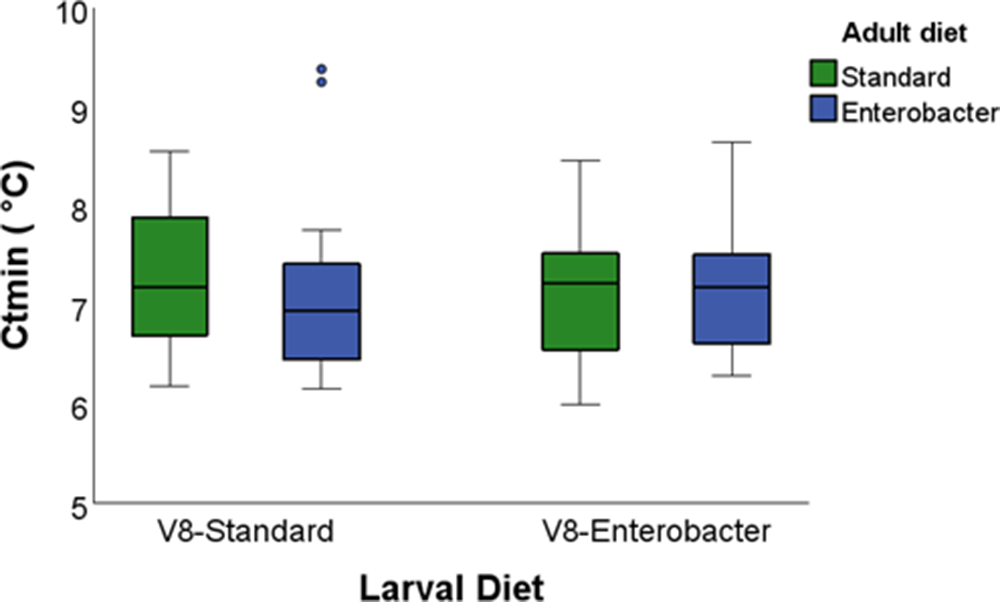
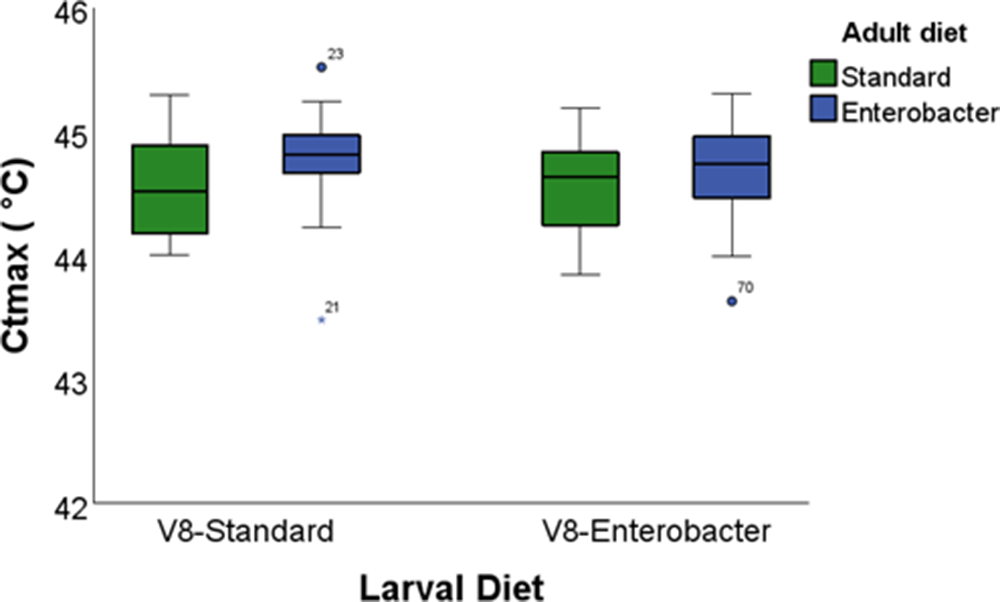
We first examined the effects of larval and adult diet supplementation on the critical thermal limits of Vienna 8 GSS sterile males using a 2 × 2 factorial GLM (table3). When only sterile males were considered, neither larval diet, adult diet, nor their interaction significantly affected CTmin or CTmax (P > 0.05; figs. 1 and 2; table 3).
Box plots of the critical thermal minimum (CTmin) of the Vienna 8 GSS males reared on two larval diets (standard and Enterobacter-enhanced) and fed on two adult diets (standard and Enterobacter-enhanced). Boxes represent the interquartile range (IQR), horizontal lines indicate medians, whiskers denote minimum and maximum values, and circles denote outliers.

Box plots of the critical thermal maximum (CTmax) of the Vienna 8 GSS males reared on two larval diets (standard and Enterobacter-enhanced) and fed on two adult diets (standard and Enterobacter-enhanced). Boxes represent the interquartile range (IQR), horizontal lines indicate medians, whiskers denote minimum and maximum values, and circles denote outliers.

Results of General Linear Model (GLM) analyses testing the effects of larval diet, adult diet, and their interaction on CTmin and CTmax of sterile Vienna 8 GSS males

Specifically, CTmin values ranged from 7.10°C in males reared on the Enterobacter-ELD and maintained on the SAD to 7.25 °C in males reared on the SLD and maintained on the EAD. CTmax values ranged from 44.55°C to 44.77°C, with slightly higher – but non-significant – thermal tolerance observed in males provided with the EAD.
To directly compare all experimental groups, including the wild (Macedonia) strain, a separate one-way GLM was conducted including five treatment levels (four sterile dietary combinations and wild males; table 4). In this analysis, CTmin did not differ significantly among treatments (P > 0.05; Supplementary Figure S1; table 4), with values ranging from 6.9°C in wild males to 7.25°C in sterile males.
Results of one-way General Linear Model (GLM) analyses testing the effect of treatment on the CTmin and CTmax of sterile Vienna 8 GSS males and wild (Macedonia) males of Ceratitis capitata

Note: Five experimental groups were included: (1) Vienna 8 GSS–SLD_SAD (standard larval diet; standard adult diet), (2) Vienna 8 GSS–SLD_EAD (standard larval diet; Enterobacter adult diet), (3) Vienna 8 GSS–ELD_SAD (Enterobacter-enhanced larval diet; standard adult diet), (4) Vienna 8 GSS–ELD_EAD (Enterobacter-enhanced larval diet; Enterobacter adult diet), and (5) wild (Macedonia) males reared on standard larval and adult diets.
In contrast, CTmax differed significantly among treatments (P < 0.001; Supplementary Figure S2; table 4). Wild males exhibited the lowest CTmax (44.0°C), whereas sterile males showed consistently higher upper thermal limits across dietary treatments.
Chill-coma recovery time
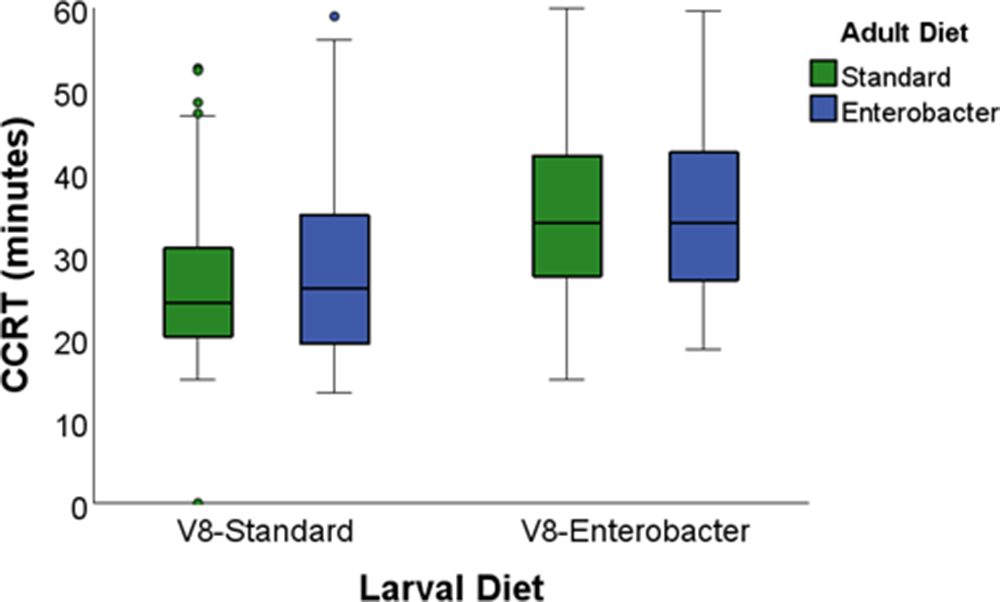
CCRT of Vienna 8 GSS sterile males was first analysed using a 2 × 2 factorial GLM including larval diet, adult diet, and their interaction (table 5). Larval diet significantly affected CCRT (P < 0.001), whereas adult diet was not significant (P > 0.05; fig. 3; table 5).
Results of General Linear Model (GLM) analyses testing the effects of larval diet, adult diet, and their interaction on CCRT of sterile Vienna 8 GSS males

Specifically, sterile males reared on the SLD recovered significantly faster (28.10 min) than those reared on the Enterobacter-ELD (35.12 min), irrespective of adult dietary treatment.
Box plots of the chill-coma recovery time (CCRT) of the Vienna 8 GSS males reared on two larval diets (standard and Enterobacter-enhanced) and fed on two adult diets (srandard and Enterobacter-enhanced). Boxes represent the interquartile range (IQR), horizontal lines indicate medians, whiskers denote minimum and maximum values, and circles denote outliers.

To compare all experimental groups directly, including the wild (Macedonia) strain, a separate one-way GLM was conducted including five treatment levels (four sterile dietary combinations and wild males; table 6). This analysis revealed significant variation in CCRT among treatments (P < 0.001; table 6). Wild males exhibited markedly faster recovery (14.58 min) than sterile males, which required between 27.27 and 35.20 min to recover (P < 0.001; Supplementary Figure S3).
Results of one-way General Linear Model (GLM) analyses testing the effect of treatment on CCRT of sterile Vienna 8 GSS males and wild (Macedonia) males of Ceratitis capitata

Note: Five experimental groups were included: (1) Vienna 8 GSS–SLD_SAD (standard larval diet; standard adult diet), (2) Vienna 8 GSS–SLD_EAD (standard larval diet; Enterobacter adult diet), (3) Vienna 8 GSS–ELD_SAD (Enterobacter-enhanced larval diet; standard adult diet), (4) Vienna 8 GSS–ELD_EAD (Enterobacter-enhanced larval diet; Enterobacter adult diet), and (5) wild (Macedonia) males reared on standard larval and adult diets.
Discussion
Our study examined how larval and adult diets affect thermal tolerance and CCRT in Vienna 8 GSS C. capitata males, with comparisons to wild males. Larval and adult diets had little effect on CTmin or CTmax in sterile males, though Enterobacter-supplemented adult diet slightly increased heat tolerance. CCRT was strongly influenced by larval diet: males reared on the SLD recovered faster than those on the Enterobacter diet, regardless of adult diet. Wild males showed similar CTmin but lower CTmax and much faster CCRT than sterile males.
Within this framework, Enterobacter sp. AA26 represents a nutritionally rich dietary supplement with the potential to influence thermal tolerance. The strain can ferment multiple sugars, including adonitol, glucose, lactose, and sorbitol, hydrolyse urea, decarboxylate ornithine, utilise citrate, and produce acetoin, while lacking potentially deleterious reactions such as lysine decarboxylation, H₂S production, tryptophan bioconversion to indole, phenylalanine deamination, or pectinase activity. Moreover, it is oxidase-negative and strongly catalase-positive, suggesting its capacity to withstand oxidative stress (Azis et al., Reference Azis, Zerva, Melidis, Caceres, Bourtzis and Ntougias2019). By providing a spectrum of amino acids and metabolites without producing harmful by-products, Enterobacter sp. AA26 could enhance larval nutritional status, metabolic reserves, and stress resilience, thereby potentially modulating CTmin-CTmax and CCRT in C. capitata.
Despite these nutritional advantages, in our study, Enterobacter supplementation of larval or adult diets did not alter the CTmin or CTmax in Vienna 8 GSS males. This aligns with previous studies showing that the effects of microbiota on tephritids are highly context-dependent. Enterobacter-enriched larval diets often improve developmental parameters, including immature survival and egg-to-adult recovery, especially in males (Augustinos et al., Reference Augustinos, Kyritsis, Papadopoulos, Abd-Alla, Cáceres and Bourtzis2015; Kyritsis et al., Reference Kyritsis, Augustinos, Ntougias, Papadopoulos, Bourtzis and Cáceres2019), and may enhance traits such as flight ability, pupal weight, and mating competitiveness (Hamden et al., Reference Hamden, Guerfali, Fadhl, Saidi and Chevrier2013). However, these benefits do not necessarily extend to shifting the absolute thermal limits. Thus, while Enterobacter supplements can support physiological performance and stress resilience, its influence on the upper and lower thermal limits of Vienna 8 GSS males appears to be limited. It should be further noted that only a single heat-inactivated Enterobacter strain was tested. Therefore, the absence of strong effects on CTLs cannot be generalised to other bacterial strains or to live microbial supplementation. Live bacteria may exert additional effects through gut colonisation and metabolic interactions, and responses may be strain-specific. Thus, the impact of microbial enrichment on thermal limits may depend on both bacterial identity and viability.
While Enterobacter did not affect CTLs, the delayed CCRT in males reared on Enterobacter-supplemented larval diets suggests that the effects of microbiota can manifest through more indirect physiological pathways. Rapid CCRT requires efficient mobilisation of metabolic reserves and re-establishment of ion homeostasis (MacMillan and Sinclair, Reference MacMillan and Sinclair2011). Diets, which are known to modify nutrient profiles and developmental rates, may influence these physiological processes (Yuval et al., Reference Yuval, Kaspi, Shloush and Warburg1998). Bacteria and yeast differ in their biological value as protein sources, and accelerated development triggered by Enterobacter can lead to variations in adult nutritional reserves or stress-related physiology (Augustinos et al., Reference Augustinos, Kyritsis, Papadopoulos, Abd-Alla, Cáceres and Bourtzis2015). Additionally, both live and inactive Enterobacter cells can interact with immune and metabolic pathways (paraprobiotic effects), imposing energetic costs under acute cold exposure (Adams, Reference Adams2010; Taverniti and Guglielmetti, Reference Taverniti and Guglielmetti2011; Van Loveren et al., Reference Van Loveren, Sanz and Salminen2012; Kyritsis et al., Reference Kyritsis, Augustinos, Ntougias, Papadopoulos, Bourtzis and Cáceres2019). These mechanisms may explain why males reared on a typical larval diet recovered more quickly from a chill coma than those reared on Enterobacter-supplemented diets, despite the generally beneficial developmental effects of Enterobacter.
Turning to the comparison with wild males, the higher CTmax observed in Vienna 8 GSS males relative to wild males is unlikely to reflect selection or enrichment processes associated with tsl tests. In Vienna strains, tsl-treated individuals do not return to colony maintenance but are designed exclusively for sterilisation and release or experimental use. Moreover, the Vienna 8 GSS used in this study is not subject to periodic genetic enrichment; instead, it is reconstructed at regular intervals from two highly inbred parental lines, with females originating from the white pupae (wp)–tsl strain and males from the original translocation line (Franz et al., Reference Franz, Bourtzis, Cáceres, Dyck, Hendrichs and Robinson2021). Consequently, repeated heat exposure during tsl–tsl‐based female elimination is unlikely to have imposed directional selection on males contributing to colony maintenance.
Alternative explanations therefore appear more plausible. First, the wild males used for comparison were derived from recently established laboratory colonies and may not yet be fully adapted to these conditions. Early generation laboratory colonies often show increased sensitivity to handling and experimental stressors, including thermal challenges, which could result in lower CTmax values compared to long established laboratory chains (Gilchrist et al., Reference Gilchrist, Cameron, Sved and Meats2012; Raphael et al., Reference Raphael, Shearman, Gilchrist, Sved, Morrow, Sherwin, Riegler and Frommer2014; Zygouridis et al., Reference Zygouridis, Argov, Nemny‐Lavy, Augustinos, Nestel and Mathiopoulos2014). Second focusing on the genetic background of the Vienna 8 GSS, sterile males may retain an ancestral genomic region derived from the original translocation line, which itself traces back to the Egypt II genetic background, historically considered relatively stress resistant in comparison to the wp–tsl lineage (Augustinos et al., Reference Augustinos, Targovska, Cancio-Martinez, Schorn, Franz, Cáceres, Zacharopoulou and Bourtzis2017). The long‐term isolation of this translocation line since its creation, together with suppressed male recombination in C. capitata, may contribute to the maintenance of these traits when Vienna 8 is periodically reconstructed (Porras et al., Reference Porras, Meza, Rajotte, Bourtzis and Cáceres2020; Ward et al., Reference Ward, Aumann, Whitehead, Nikolouli, Leveque, Gouvi, Fung, Reiling, Djambazian and Hughes2021; Sollazzo et al., Reference Sollazzo, Gouvi, Nikolouli, Martinez, Schetelig and Bourtzis2022).
Although male embryos carrying the tsl⁺ allele survive tsl tests, repeated heat exposure at the egg stage can influence thermal responses. Importantly, these males do not return to the colony after TSLTs, so no enrichment occurs in the maintained colony. Nevertheless, previous work has shown that even within Vienna strains, thermal sensitivity at the egg stage can vary depending on the specific genetic background (Augustinos et al., Reference Augustinos, Targovska, Cancio-Martinez, Schorn, Franz, Cáceres, Zacharopoulou and Bourtzis2017; Sollazzo et al., Reference Sollazzo, Gouvi, Nikolouli, Martinez, Schetelig and Bourtzis2022). Together these factors suggest that inherent differences in the ancestral translocation line and the wp–tsl background may contribute to higher CTmax observed in Vienna 8 males compared to wild males.
The same mechanisms may also help explain why wild males recovered from chill coma faster than sterile males. Reduced genetic diversity, pleiotropic effects linked to the wp–tsl inversion, and long-term maintenance under constant laboratory conditions can impair cold-stress resilience in GSS colonies, whereas wild males exposed to natural thermal fluctuations and stronger selection may maintain physiological traits that promote rapid recovery. Thus, although CTmax and CCRT represent different thermal endpoints, both patterns are consistent with the contrasting evolutionary histories of mass-reared and wild flies.
Overall, our results demonstrate that the thermal responses of Vienna 8 GSS males are driven mainly by the evolutionary and operational history of mass-rearing rather than by dietary supplementation. Enterobacter enrichment did not affect CTmin or CTmax, suggesting that these thermal limits are robust traits, under the dietary conditions tested, although the larval diet did influence cold-stress physiology through its effects on CCRT. However, CTmin and CTmax are not immutable traits; they exhibit phenotypic plasticity and can be influenced by developmental temperature, acclimation, age, and physiological state (Nyamukondiwa and Terblanche, Reference Nyamukondiwa and Terblanche2010; Papadogiorgou et al., Reference Papadogiorgou, Verykouki and Papadopoulos2025). In the present study, these factors were standardised across treatments, minimising potential confounding effects. Wild males showed lower heat tolerance but faster chill-coma recovery, reflecting the contrasting selective pressures in natural and laboratory environments. Furthermore, the bacterial supplementation was not evaluated in the wild colony. Therefore, we cannot exclude that wild males might respond differently to microbial enrichment compared to laboratory strains. Host genetic background and laboratory adaptation history may modulate dietary or microbiota effects, and extrapolation of these results to wild populations should be made cautiously.
These patterns have practical relevance for SIT programmes, as released males inevitably encounter more variable temperatures in the field than in controlled mass-rearing facilities. While cold stress is minimal in tropical and subtropical areas, more temperate regions, such as Valencia, Trentino (north Italy), Central Macedonia (north Greece), and other Mediterranean locations, experience episodic cold spells and heat waves. Establishing the CTLs of sterile males is therefore important for identifying vulnerability periods and optimising release timing, strain management, and rearing conditions. Incorporating strain-specific thermal tolerance traits into IPM and SIT population models may contribute to improving predictions of field performance and induced sterility. However, given that the present study was conducted under controlled laboratory conditions, further validation under semi-field and field environments is necessary before these parameters can be fully integrated into operational models. Nevertheless, our results highlight the value of characterising thermal physiology in GSS lines as part of quality assessment frameworks.
Future studies should investigate whether alternative probiotic strains, different supplementation regimes, or combinations of dietary additives could more effectively enhance thermal tolerance in GSS males. In addition, field-based evaluations of sterile male performance under naturally fluctuating thermal conditions would provide essential validation of these laboratory findings and help determine their operational relevance.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0007485326101035.
Acknowledgements
We thank Eleutheria Maria Bali (UTH) and Kostas Zarpas (UTH) for support with data collection, experimental measurements and laboratory maintenance, and Paraschos Prekas (UTH) for assistance with the rearing of wild Ceratitis capitata populations.
Author contributions
G. Papadogiorgou: Data curation, Formal analysis, Methodology [equal], Writing – original draft, Writing-review & editing; P. Koskinioti: Methodology, Writing-review & editing; V. Rodovitis: Methodology, Writing-review & editing; G. Giannatos: Methodology, Writing-review & editing; A. Augustinos: Methodology, Resources, Writing-review & editing; G. Tsiamis: Methodology, Resources, Writing-review & editing; N. Papadopoulos: Conceptualisation, Investigation, Methodology, Resources, Supervision, Visualisation, Writing-review & editing.
Financial support
Funding for this research was provided by the European Union’s Horizon Europe Research and Innovation Programme REACT (Grant agreement 101059523).
Competing interests
Authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Authorship statement
All authors listed on this manuscript meet the criteria for authorship according to disciplinary norms. All authors have made substantial contributions to the conception and design of the study, data acquisition and/or analysis, and the drafting or critical revision of the manuscript. All authors have approved the final version of the manuscript and agree to be accountable for all aspects of the work. No individuals who meet the criteria for authorship have been omitted.
Data availability
The data supporting the findings of this study are available on the Open Science Framework (OSF) https://doi.org/10.17605/OSF.IO/FYW78.