Introduction
Iron is a key trace element in biological systems, integral to numerous biochemical processes, which support the structural integrity of proteins and enzymes (Zhang et al. Reference Zhang, Ghosh and Rouault2014). In mammals, iron metabolism is critical for numerous physiological processes, including oxygen transport via hemoglobin (Hb), energy production, and cell proliferation (Le and Richardson Reference Le and Richardson2002; Pantopoulos et al. Reference Pantopoulos, Porwal and Tartakoff2012). The regulation of iron absorption, transport, storage, and utilization directly affects energy production, growth, and overall physiological health (Chua et al. Reference Chua, Graham and Trinder2007). Abnormal iron metabolism can cause various health problems, with anemia being the most prevalent. Anemia is characterized by a reduction in red blood cell count or Hb concentration (Bolliger et al. Reference Bolliger, Jahn and Schüpbach-Regula2025), which impairs oxygen transport, thereby diminishing energy supply and compromising physiological function (Van Swelm et al. Reference Van Swelm, Wetzels and Swinkels2019).
As the cornerstone of pork production, sows require optimal health to ensure high productivity. However, anemia in pregnant sows impacts reproductive performance, leading to increased stillbirth rate, an increase in the number of weak piglets and increased farrowing duration (Bhattarai et al. Reference Bhattarai, Framstad and Nielsen2019a; Hollema et al. Reference Hollema, Zwiers and Hermesch2020; McClellan et al. Reference McClellan, Sheffield and Levesque2024). These weak piglets often exhibit impaired growth and development (Perri et al. Reference Perri, Friendship and Harding2016), thereby posing substantial challenges to the sustainability of the swine industry. Moreover, maternal iron deficiency can induce various reproductive disorders, including delayed uterine recovery (Zilliacus and Putkinen Reference Zilliacus and Putkinen1952), impaired follicular development (Mehta et al. Reference Mehta, Goyal and Meena2018), and disrupted estrous cycle in anemic sows following parturition. Collectively, these factors prolong non-production days, significantly increase breeding costs (Peng et al. Reference Peng, Shi and Jin2015), and exert a sustained negative impact on the long-term production efficiency and economic profitability of swine operations.
This review summarizes recent advances in this field, explores the relationship between iron metabolism, anemia, and sustainable production in pregnant sows, and evaluates the potential implications for the swine industry.
Methods
We searched PubMed, Web of Science, Google Scholar, and Science Direct using keywords related to anemia, sows, iron deficiency anemia (IDA), iron, iron supplementation, iron metabolism, and reproductive performance. No restrictions were placed on publication date or language. Following duplicate removal, the retrieved records underwent a two-stage screening process: an initial assessment based on titles and abstracts, followed by a full-text review of potentially eligible studies to determine final inclusion. During literature synthesis, priority was given to studies characterized by rigorous experimental designs, adequate sample sizes, and appropriate statistical analyses. Given the relative scarcity of literature specifically addressing sow anemia, studies on piglet anemia and iron supplementation strategies were also consulted to provide supplementary insights and contextual understanding.
Iron metabolism
Iron metabolism is a complex physiological process involving mechanisms for the absorption, transport, storage, utilization, and homeostatic regulation of this essential micronutrient (Vogt et al. Reference Vogt, Arsiwala and Mohsen2021). This section describes iron metabolism with a specific focus on maternal-fetal iron transport in sows.
Iron absorption
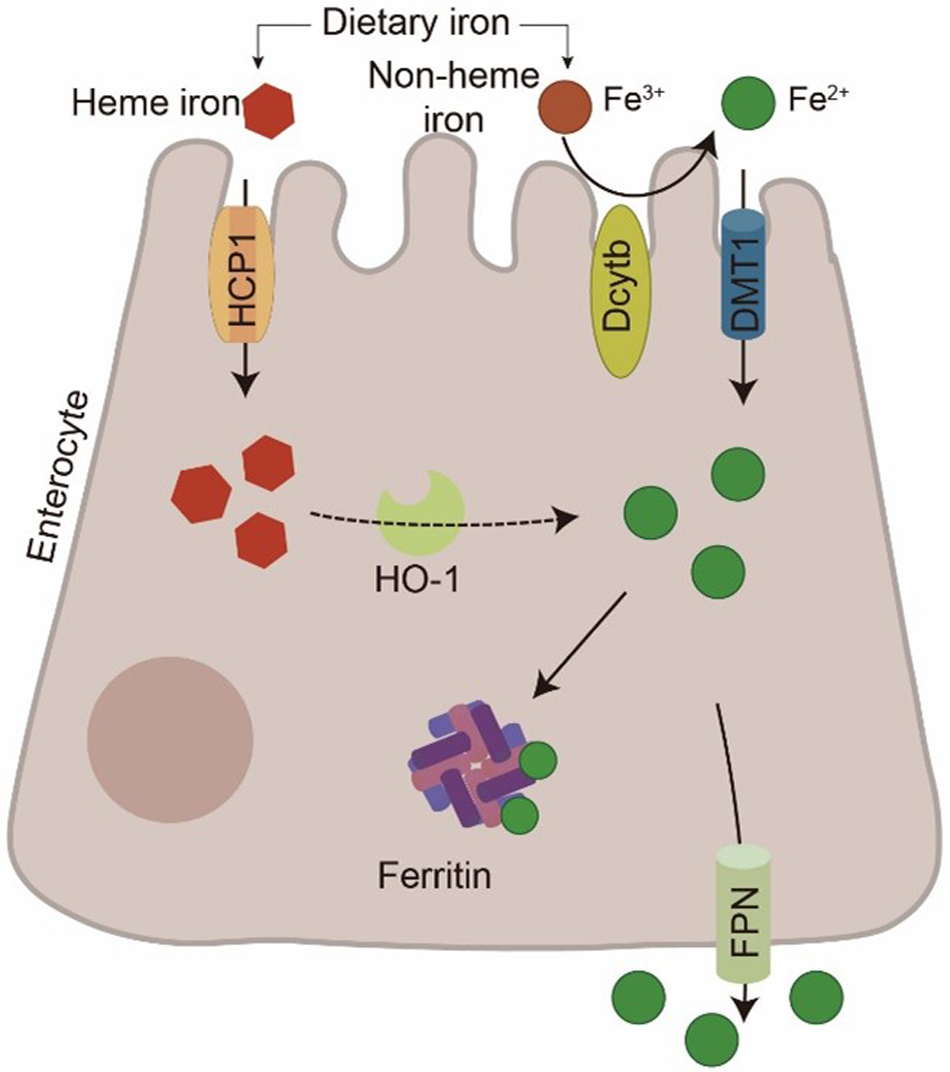
Iron absorption primarily occurs in the duodenum and proximal jejunum (Correnti et al. Reference Correnti, Gammella and Cairo2024). Dietary iron exists in two forms: heme iron, derived from animal sources such as meat and fish, and non-heme iron, found in plant-based foods and fortified products (Zhou et al. Reference Zhou, Schilling and Makrides2005). After heme iron enters the enterocyte, heme oxygenase-1 (HO-1) decomposes it, releasing ferrous iron (Fe2+), which then enters the labile iron pool for further metabolism (Yanatori et al. Reference Yanatori, Tabuchi and Kawai2010; Zhuo et al. Reference Zhuo, Yu and Li2019). Heme iron can be absorbed intact by enterocytes; its absorption efficiency is higher than that of non-heme iron (Abboud and Haile Reference Abboud and Haile2000; Quintero-Gutiérrez et al. Reference Quintero-Gutiérrez, González-Rosendo and Sánchez-Muñoz2008).
Unlike the heme iron absorption pathway, non-heme iron is primarily absorbed in the small intestine. First, ferric iron (Fe3+) is reduced to Fe2+ at the brush border of enterocytes by reductase, notably duodenal cytochrome b (Dcytb) (Balusikova et al. Reference Balusikova, Dostalikova‐Cimburova and Tacheci2022; Lane et al. Reference Lane, Bae and Merlot2015; McKie Reference McKie2008). It is then transported across the apical membrane into the enterocyte by the divalent metal transporter 1 (DMT1) (Okazaki Reference Okazaki2024; Yanatori and Kishi Reference Yanatori and Kishi2019). Following its absorption, iron is either stored as ferritin or exported into the bloodstream via ferroportin (FPN) located on the basolateral membrane (Fig. 1).
Intestinal iron absorption pathways. At the intestinal enterocyte membrane, Fe3+ is reduced to Fe2+ by Dcytb and subsequently transported into the cell via DMT1. Heme iron is taken up by cells through hemolysin-coregulated protein 1 (HCP1) and catabolized by HO-1 to release Fe2+. Subsequently, Fe2+ is exported to the blood via FPN.

Iron transport, storage, and utilization
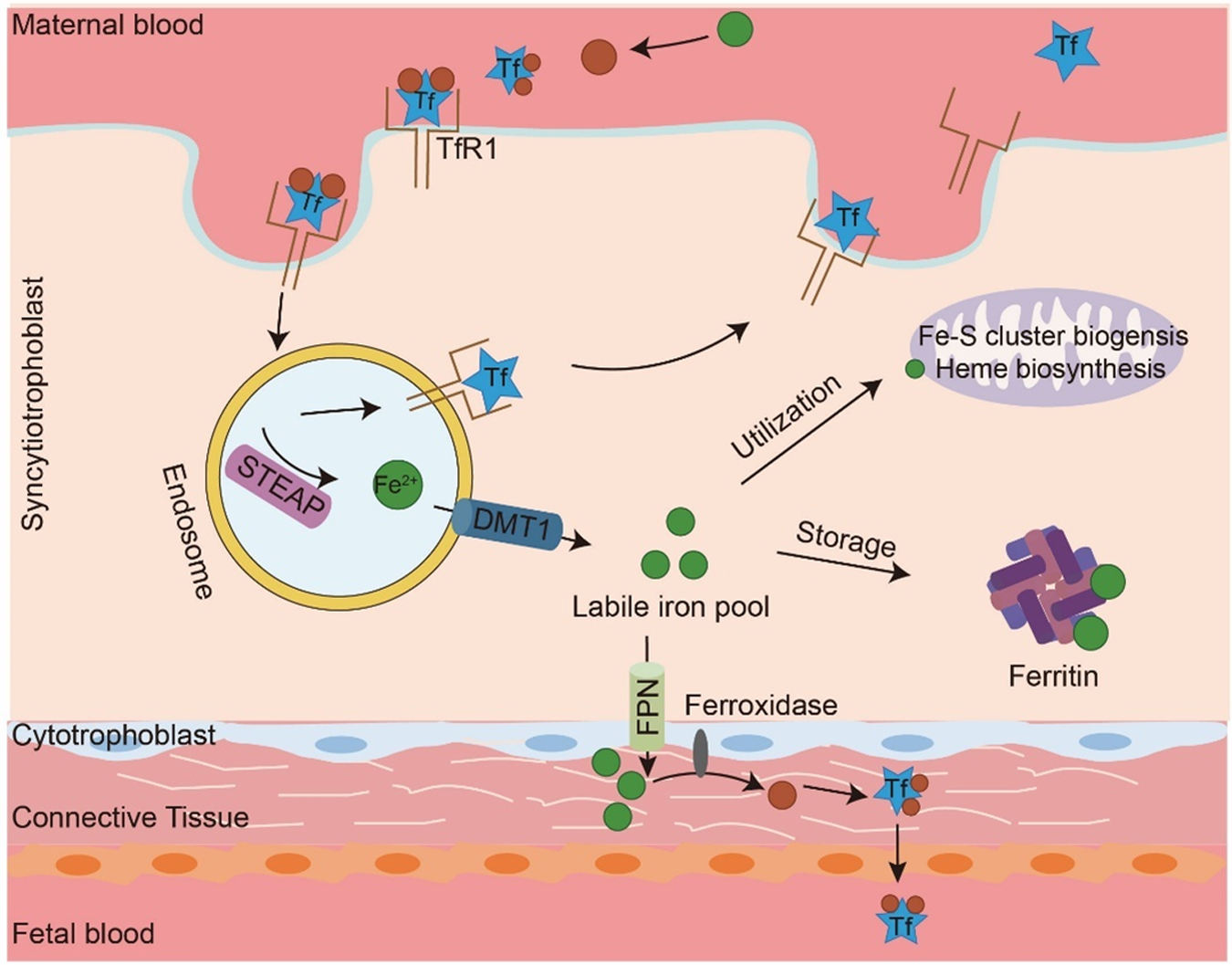
In maternal blood, Fe2+ is oxidized to Fe3+ and subsequently binds to transferrin (Tf) (Eid et al. Reference Eid, Hémadi and Ha-Duong2014; White et al. Reference White, Conesa and Sánchez2012). Tf is a glycoprotein with a high affinity for iron. Iron-bound holo-transferrin then binds to transferrin receptor 1 (TfR1) on the syncytiotrophoblast membrane, triggering receptor-mediated endocytosis (Chen Reference Chen2007; Fuqua et al. Reference Fuqua, Vulpe and Anderson2012). Within the acidified endosome, Fe3+ is released from Tf, reduced to Fe2+ by six-transmembrane epithelial antigen of prostate (STEAP), and transported into the cytoplasm via DMT1 to enter the labile iron pool (Conway and Henderson Reference Conway and Henderson2022; Correnti et al. Reference Correnti, Gammella and Cairo2024). Following iron release, the TfR1–Tf complex is recirculated to the cell surface and Tf is released (Fig. 2).
Iron transport, storage and utilization. In plasma, Fe2+ is oxidized to Fe3+ and binds to Tf to form the Tf–iron complex. This complex binds to TfR1 to trigger endocytosis. Within the endosome, Fe3+ is reduced to Fe2+ by the STEAP and transported into the cytoplasm via DMT1. Tf and TfR1 are recycled back to the cell surface and released into the bloodstream to continue participating in iron transport. Some of the Fe2+ in the cytoplasm are stored in ferritin, some are used for biosynthesis of heme and Fe–S clusters in mitochondria, and others are exported to the placental connective tissue via FPN. In the placenta, Fe2+ is oxidized to Fe3+ and transferred to the fetal circulation.

Fe2+ released into syncytiotrophoblasts is stored within these cells as ferritin (O’Brien Reference O’Brien2022). Ferritin is a ubiquitous protein complex composed of 24 heavy and light chain subunits that stores iron in a nontoxic and soluble form (Dunn et al. Reference Dunn, Rahmanto and Richardson2007; Dutt et al. Reference Dutt, Hamza and Bartnikas2022; Piskin et al. Reference Piskin, Cianciosi and Gulec2022). Each ferritin molecule can bind up to 4500 iron atoms, though it typically contains approximately 2000. This makes ferritin an efficient, dynamic iron reserve capable of rapidly releasing iron in response to increased physiological demand (Theil Reference Theil2003). Consequently, insufficient iron storage can lead to disorders such as anemia, underscoring the critical role of ferritin in maintaining systemic iron homeostasis. Fe2+ is also available to the placenta to support mitochondrial function.
Additionally, a portion of this Fe2+ is utilized within the placenta itself to support essential mitochondrial functions, such as energy production. Mitochondria are the central hubs of cellular iron metabolism (Paul et al. Reference Paul, Manz and Torti2017) and are responsible for two critical processes: heme synthesis and iron–sulfur (Fe–S) cluster (ISC) biogenesis (Drysdale et al. Reference Drysdale, Arosio and Invernizzi2002). In heme synthesis, the enzyme 5-aminolevulinate synthase 2 (ALAS2) catalyzes the initial reaction to produce 5-aminolevulinic acid (ALA) (La et al. Reference La, Oved and Ghiaccio2020). Finally, Fe2+ is inserted into protoporphyrin IX by ferrochelatase to form heme. Notably, ferrochelatase activity itself depends on an intrinsic [2Fe–2S] cluster (Wu et al. Reference Wu, Wang and Ta2021; Ye and Rouault Reference Ye and Rouault2010b). Mitochondrial ISC biogenesis is executed by the ISC assembly machinery. Initially, a cysteine desulfurase complex provides sulfur, while iron is likely delivered by frataxin. These elements combine on the ISC scaffold protein to form an initial [2Fe–2S] cluster (Ye and Rouault Reference Ye and Rouault2010a). This cluster is subsequently transferred and reshaped by chaperone proteins like Glutaredoxin 5, ultimately yielding mature [4Fe–4S] clusters for insertion into apoproteins (Lill and Freibert Reference Lill and Freibert2020). Increased ISC biogenesis regulates cellular iron homeostasis. It inhibits the iron-regulatory proteins (IRPs), stimulates ALAS2 expression, and thereby promotes heme synthesis and erythropoiesis. Thus, heme synthesis is functionally coupled to and dependent on efficient ISC biogenesis (Braymer and Lill Reference Braymer and Lill2017; Lill et al. Reference Lill, Hoffmann and Molik2012).
When placental iron demand is low, cytosolic Fe2+ is exported via FPN into the connective tissue space on the fetal side of the placenta (Roberts et al. Reference Roberts, Bourque and Renaud2020; Sangkhae and Nemeth Reference Sangkhae and Nemeth2019). Within this space, ferroxidase oxidizes Fe2+ to Fe3+, enabling its binding to Tf in fetal circulation. The resulting Tf-bound iron then enters the fetal bloodstream and is transported to fetal tissues for utilization and storage (McArdle et al. Reference McArdle, Andersen and Jones2008).
Iron recycling
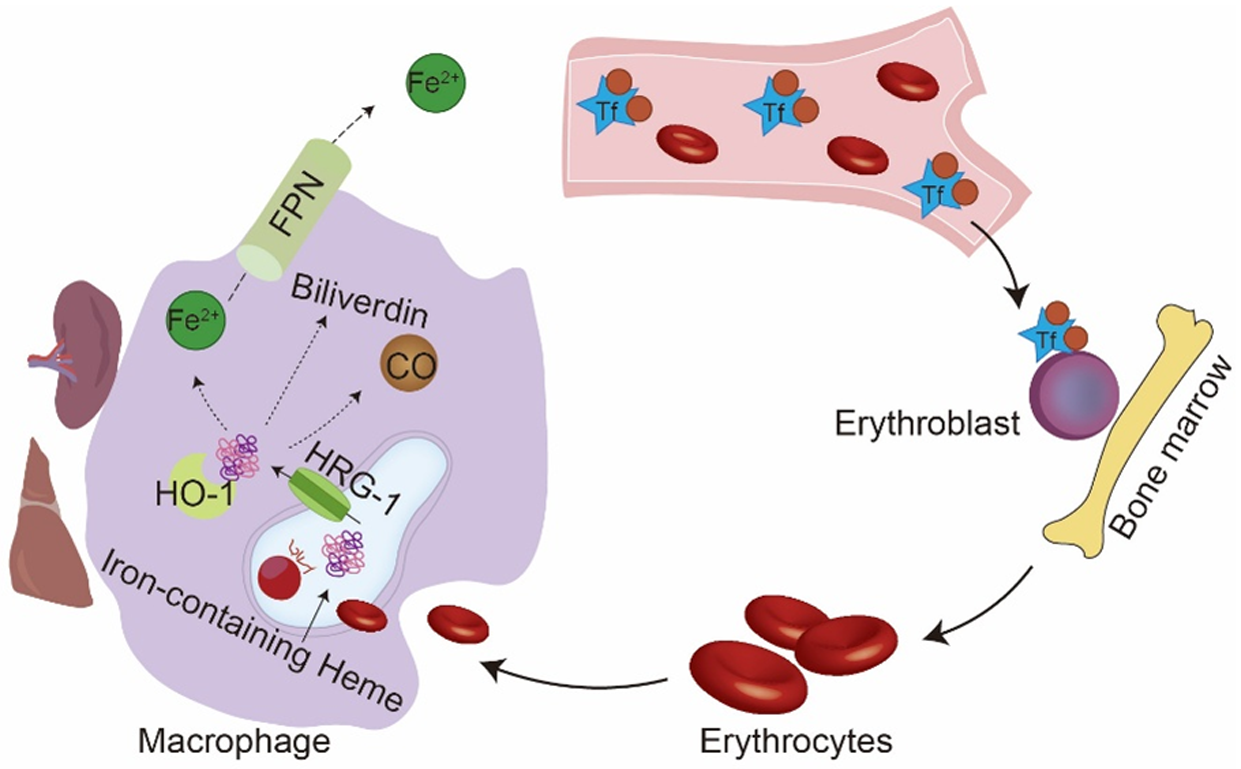
The body efficiently recycles iron, and senescent erythrocytes are phagocytosed by macrophages primarily in the spleen and liver. Within the phagolysosome, Hb is catabolized. Heme is then transported into the cytoplasm via heme-responsive gene-1 (HRG-1), where it is cleaved by HO-1 to release Fe2+, biliverdin, and carbon monoxide (CO). The Fe2+ is subsequently exported into the bloodstream via FPN to complete the iron cycle (Ni et al. Reference Ni, Yuan and Kuang2022) (Fig. 3).
Iron recycling. The Tf-bound iron complex enters the bone marrow and combines with erythroblasts, precursors of red blood cells (erythrocytes). Senescent or damaged erythrocytes are phagocytosed by macrophages. Within phagolysosomes, erythrocytes are digested and Hb is degraded. Heme is then transported to the cytoplasm via HRG-1, where it is catabolized by HO-1 to release Fe2+, biliverdin, and CO. Fe2+ is exported to the bloodstream through FPN.

Regulation of iron metabolism
Iron metabolism is tightly regulated to maintain systemic homeostasis, ensuring sufficient iron availability for essential physiological processes while preventing the toxicity associated with its excess accumulation (Himmelfarb Reference Himmelfarb2007). When systemic iron levels exceed physiological demand, these labile Fe2+ catalyze the generation of a highly reactive radical via the Fenton reaction, leading to severe oxidative stress that causes extensive damage to cellular structures and impairs function (Fraga and Oteiza Reference Fraga and Oteiza2002; Papanikolaou and Pantopoulos Reference Papanikolaou and Pantopoulos2005). This regulation occurs primarily through two key mechanisms: the systemic hepcidin–ferroprotein axis and the intracellular IRP/iron-responsive element (IRE) system (Bonadonna et al. Reference Bonadonna, Altamura and Tybl2022; Nemeth and Ganz Reference Nemeth and Ganz2021).
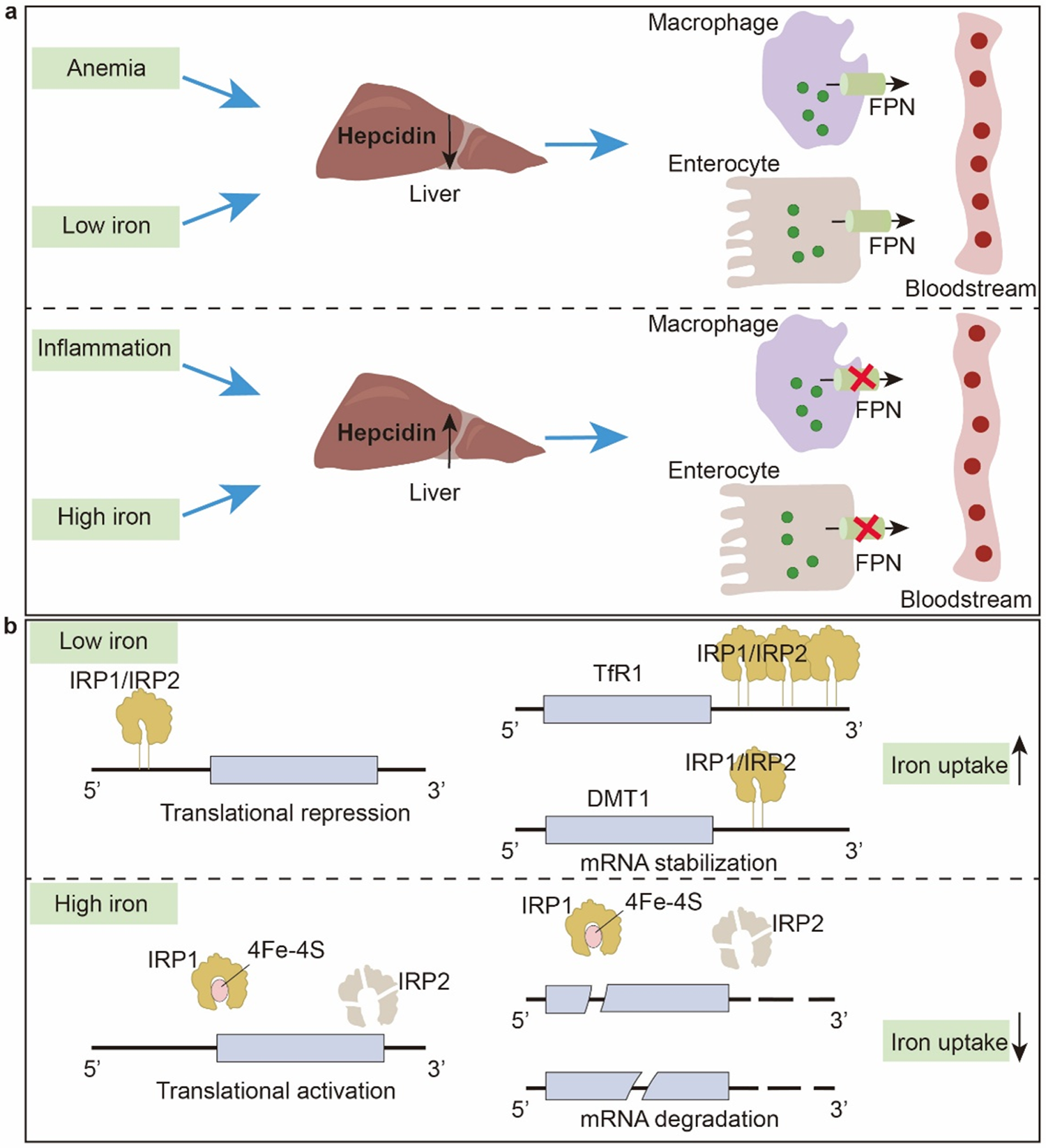
The liver is a central organ in systemic iron metabolism, serving as the body’s primary iron storage site (Fig. 4a). The iron-regulatory hormone hepcidin, synthesized and secreted by the liver, is a central regulator of systemic iron homeostasis (Anderson et al. Reference Anderson, Shen and Eisenstein2012). Hepcidin regulates systemic iron levels by binding to FPN, inducing its internalization and degradation. This process reduces dietary iron absorption from the intestine and inhibits iron recycling from macrophages. Under conditions of iron overload or inflammation, hepcidin expression is upregulated, thereby suppressing iron absorption and release (Cassat James and Skaar Eric Reference Cassat James and Skaar Eric2013). Conversely, during iron deficiency or anemia, hepcidin expression is downregulated to enhance iron availability (Yiannikourides and Latunde-Dada Reference Yiannikourides and Latunde-Dada2019).
Iron homeostasis regulation. (a) Hepcidin, secreted by the liver, regulates systemic iron circulation by controlling FPN-mediated iron efflux from absorptive enterocytes and macrophages. (b) IRPs bind to IREs located in either the 5′ or 3′ untranslated regions of specific mRNAs. Under low iron conditions, IRPs bind to IREs to stabilize target mRNAs and promote iron uptake. Under high iron conditions, IREs’ interactions are inhibited, thereby reducing iron uptake.

At the cellular level, iron metabolism is primarily regulated by the IRP/IRE system (Wang and Pantopoulos Reference Wang and Pantopoulos2011) (Fig. 4b). IRPs are RNA-binding proteins, comprising IRP1 and IRP2. An IRE is a highly conserved stem-loop structure located in the 5′ or 3′ untranslated region of mRNAs encoding proteins involved in iron metabolism (Hentze et al. Reference Hentze, Muckenthaler and Galy2010). When intracellular iron levels are low, IRPs bind to IREs. This binding inhibits the translation of FPN mRNA while stabilizing the TfR1 and DMT1 mRNAs, thereby increasing cellular iron uptake (Mleczko‐Sanecka and Silvestri Reference Mleczko‐Sanecka and Silvestri2021). When intracellular iron levels are high, IRP1 incorporates a [4Fe–4S] cluster, converting it into a cytosolic aconitase, while IRP2 undergoes degradation. Both processes prevent IRP binding to IREs. Consequently, TfR1 and DMT1 expression decreases, reducing cellular iron import. Simultaneously, the translation of ferritin mRNA is enhanced, promoting the storage of excess iron (Muckenthaler et al. Reference Muckenthaler, Galy and Hentze2008; Pantopoulos Reference Pantopoulos2004).
The absorption, transport, and storage of iron collectively constitute a homeostatic system that ensures the efficient utilization of this essential micronutrient. This homeostatic system is a complex and tightly regulated process, which is crucial for maintaining normal physiological functions and overall health.
Anemia in pregnant sows
Iron is a key structural component of Hb and myoglobin, playing an indispensable role in oxygen metabolism. Within erythrocytes, each Hb molecule binds to four oxygen molecules, enabling precise oxygen delivery to tissues via the circulatory system (Abbas et al. Reference Abbas, Hayirli and Drakesmith2022; Mairbäurl and Weber Reference Mairbäurl and Weber2012). In muscle tissue, myoglobin acts as an oxygen reservoir, facilitating oxygen availability for muscle contraction and energy production. The operation of this precision system is highly dependent on the dynamic balance of iron metabolism. A disruption in any aspect of iron metabolism can impair Hb synthesis, ultimately leading to reduced erythropoiesis. During the mid- to late-gestation period, rapid fetal growth increases maternal iron demand, which can disrupt iron homeostasis and precipitate gestational anemia.
Anemia factors in pregnant sows
Anemia is defined as a reduction in red blood cell count or Hb concentration below the physiological normal range (Bhattarai et al. Reference Bhattarai, Framstad and Nielsen2019c). Multiple factors contribute to the development of anemia in pregnant sows.
Iron is an essential micronutrient that supports normal maternal growth and embryonic development during gestation, and is indispensable for Hb synthesis (Sperling et al. Reference Sperling, Guerra and Dimitrov2021). Iron deficiency arises when bodily iron stores are depleted, dietary intake is inadequate, or iron loss is excessive, preventing sufficient Hb synthesis. Without timely intervention, iron deficiency progresses to IDA (Mazgaj et al. Reference Mazgaj, Lipiński and Starzyński2024). Hematological monitoring indicates that the normal Hb concentration for sows ranges from 100 to 160 g/L. A concentration below 100 g/L is diagnostic for anemia (Bhattarai et al. Reference Bhattarai, Framstad and Nielsen2019b). It is noteworthy that Hb concentrations fluctuate throughout the physiological stages of sows. Hb concentration typically peaks during mid-gestation, gradually declines in late gestation, and reaches its nadir during early lactation (Castevens et al. Reference Castevens, Ferreira and Gillespie2020). A longitudinal study of 247 pregnant sows revealed that Hb levels decreased by 2.7 g/L at 4 weeks before farrowing, compared to 7 weeks before farrowing, by 8.1 g/L at farrowing, and by 9.1 g/L at 3 weeks after farrowing (Normand et al. Reference Normand, Perrin and Auvigne2012). Furthermore, sow iron reserves decline gradually with advancing parity. From the first to the third parity, the average Hb concentration decreased linearly from 117 to 108 g/L, and remained at this low level in subsequent parities (Sawhney et al. Reference Sawhney, Sarkar and Begum2023). This cumulative effect is reflected in the higher anemia prevalence observed in parity ≥ 4 sows (58.4%, 321/550) compared to gilts (38.8%, 206/531) (Castevens et al. Reference Castevens, Ferreira and Gillespie2020). This irreversible depletion of iron reserves may be associated with two key factors: first, blood volume expansion during gestation induces a relative iron deficiency; second, high-producing sows cannot fully replenish their iron reserves between successive parities (Anderson et al. Reference Anderson, Elsley and McDonald1970).
Blood loss represents another major etiology of anemia in pregnant sows, particularly hemorrhage associated with parturition. Excessive hemorrhage during farrowing can result from uterine dystocia, retained placenta, or lacerations of the reproductive tract (Feyera et al. Reference Feyera, Pedersen and Krogh2018). Severe, untreated blood loss can precipitate a rapid decline in erythrocyte count, consequently inducing acute anemia. Gastrointestinal bleeding is also a potential cause. Gastric or small intestinal ulcers in sows can lead to chronic or acute blood loss. The pathogenesis of this type of peptic ulcer is complex, commonly involving stressors, high-grain/low-fiber diets, and certain medications.
Infectious diseases can cause anemia in pregnant sows. The Porcine Reproductive and Respiratory Syndrome Virus (PRRSV) infects hematopoietic cells in the bone marrow, disrupting erythropoiesis (Zhang et al. Reference Zhang, Wang and Zhang2024) and leading to a reduced circulating red blood cell count, thereby causing anemia (Wang et al. Reference Wang, Yu and He2019). The clinical symptoms of PRRSV-associated anemia may include pale mucous membranes, lethargy, anorexia, and, in severe cases, mortality (Hu et al. Reference Hu, Zhang and Wang2020).
Beyond iron, deficiencies in other nutrients can also induce anemia in pregnant sows (Lillie and Frobish Reference Lillie and Frobish1978). Vitamin B12 is essential for the normal maturation and division of erythrocytes in bone marrow. A deficiency of vitamin B12 can lead to megaloblastic anemia, characterized by the production of numerous immature erythrocytes that are inefficient in oxygen transport (Cartwright et al. Reference Cartwright, Tatting and Robinson1951; Obeid et al. Reference Obeid, Heil and Verhoeven2019). Vitamin B12 deficiency anemia in sows can result from either inadequate dietary intake or impaired absorption in the gastrointestinal tract (Bruun et al. Reference Bruun, Lyderik and Dall2024). Survey data reveal that 49.7% of prepartum sows exhibit Hb concentrations below 100 g/L. This study encompassed 2683 sows from 11 commercial farms across North Carolina and Indiana, revealing significant regional variations in sow Hb concentrations. Notably, the average Hb concentration was significantly higher in Indiana herds compared to those in North Carolina. This disparity may be attributed to differing dietary phytase supplementation levels (Indiana: 1250 FTU/kg, North Carolina: 750 FTU/kg). Phytase enzymatically hydrolyzes phytic acid, liberating chelated minerals such as iron and thereby enhancing their bioavailability (Castevens et al. Reference Castevens, Ferreira and Gillespie2020). This finding suggests that anemia in sows is not solely a consequence of direct iron deficiency but can also be influenced by dietary imbalances that affect iron absorption efficiency, such as the level of phytase supplementation.
Effects of anemia on sow reproduction
Effects of anemia on reproductive performance
Anemia in pregnant sows significantly impacts piglet Hb levels and survival rate. Related researches indicate that the levels of Hb in piglets show a positive correlation with the Hb content of sows before and after delivery (Bhattarai et al. Reference Bhattarai, Framstad and Nielsen2019a; Mahan et al. Reference Mahan, Watts and St-Pierre2009). The stillbirth rate is a key factor of piglets weaned per sow per year (PSY). A high stillbirth rate directly reduces the number of live piglets. Consequently, even with a constant preweaning survival rate, the number of piglets weaned is diminished due to this smaller initial cohort. Furthermore, stillbirths increase the cost per weaned piglet. Inputs such as gestation feed, housing, and labor represent largely fixed costs per sow. Therefore, an elevated stillbirth rate effectively reduces the PSY and undermines overall farm profitability. A further study involving 160 sows with an average parity of 2.8 reported that a higher number of stillbirths occurred in anemic sows (1.7 ± 2.6) compared to non-anemic sows (1.1 ± 2.1). Analysis using a linear mixed model revealed a negative correlation between the probability of stillbirth during delivery and maternal Hb concentration, particularly in higher-parity sows (Bhattarai et al. Reference Bhattarai, Framstad and Nielsen2018; Noblett et al. Reference Noblett, Bonin Ferriera and Bhattarai2021). Therefore, interventions that increase maternal Hb concentration are likely to correspondingly elevate Hb levels in newborn piglets, thereby reducing the stillbirth rate.
McClellan et al. recorded the farrowing duration of 101 sows and found that it was significantly longer in anemic sows (7.2 ± 0.92 h) than in non-anemic sows (3.5 ± 0.88 h) (McClellan et al. Reference McClellan, Sheffield and Levesque2024). Prolonged farrowing duration, indicative of sow fatigue, fetal macrosomia, or malpresentation, frequently necessitates interventions such as oxytocin administration, vaginal examinations, positional adjustments, and manual extraction. These interventions not only increase labor demands but also elevate the risk of birth canal damage and infection (Nam et al. Reference Nam, Anh Dao and Sukon2024). Manual extraction, in particular, compromises the sow’s natural reproductive tract barrier, thereby substantially increasing the risk of endometritis (Björkman et al. Reference Björkman, Oliviero and Kauffold2018). Consequently, postpartum antibiotic therapy often becomes necessary, directly increasing medication expenditures. Studies in humans have demonstrated that maternal anemia is associated with adverse outcomes, including fetal death (Shi et al. Reference Shi, Chen and Wang2022), spontaneous abortion, preterm delivery (Scholl et al. Reference Scholl, Hediger and Fischer1992), and low birth weight. Collectively, this evidence suggests that anemia in pregnant sows may impair oxygen supply and uterine contractility (Rydal et al. Reference Rydal, Bhattarai and Nielsen2021; Seip et al. Reference Seip, Friendship and Amezcua2020), resulting in intrapartum hypoxia in piglets and a consequent increase in stillbirth rate (Lipiński et al. Reference Lipiński, Starzyński and Canonne-Hergaux2010).
Effects of anemia on milk production
Anemia impairs sow performance, leading to reduced milk yield and a consequent decrease in the iron concentration of sow’s milk (El‐Farrash et al. Reference El‐Farrash, Ismail and Nada2011). Sow’s milk is low in iron, providing piglets with only approximately 1 mg of iron per day (Li et al. Reference Li, Yin and Dang2011; Perri et al. Reference Perri, Friendship and Harding2016). Lactoferrin (LF) is a key regulator of iron absorption and oxidative stress. It facilitates iron transfer to piglets and exerts anti-inflammatory effects by inhibiting interleukin-6 secretion (Laskow et al. Reference Laskow, Langdon and Abadir2021; Presti et al. Reference Presti, Manti and Parisi2021).
A study supplementing LF to the sow’s diet demonstrated that milk yield was higher in the experimental group than in the control group throughout lactation. This difference was particularly pronounced on day 7, when the yield in the experimental group (629.76 g) was more than double that of the control group (207 g) (Jahan et al. Reference Jahan, Kracht and Ho2017). The combined supplementation of LF and iron effectively elevates iron status in piglets by upregulating LF receptor gene expression, enhancing antioxidant capacity, and modulating cytokine activity (Hu et al. Reference Hu, Zhao and Zhao2019). Consequently, a decline in milk production corresponds to reduced LF intake by piglets, leading to inadequate iron acquisition. Furthermore, maternal anemia reduces the concentration of various nutrients and antibodies in milk, which can impair piglet growth and development (Svoboda and Drabek Reference Svoboda and Drabek2005).
Iron metabolism and anemia in sustainable sow production
IDA in sustainable sow production
Iron metabolism in pregnant sows shows dynamic changes throughout gestation. Sow iron requirements are relatively low during early gestation, increase progressively with fetal development, and peak during late gestation. During early and mid-gestation, nutrients are prioritized for maternal body growth and maintenance, with a limited proportion allocated to reproductive demands (Solà-Oriol and Gasa Reference Solà-Oriol and Gasa2017). However, this nutrient partitioning shifts markedly during late gestation to support accelerated fetal growth (Theil et al. Reference Theil, Farmer and Feyera2022). At this stage, the daily dietary iron absorption of sows (80 mg/kg of dietary iron) becomes insufficient to meet escalating fetal demands. The maternal system mobilizes iron reserves, prioritizing allocation to the fetus. This physiological adaptation is a primary reason for the high susceptibility of pregnant sows to IDA (Bush et al. Reference Bush, Jensen and Ashenbrucker1956). Optimal sow nutrition is crucial for supporting mammary development, litter performance, and colostrum quality. McPherson et al. reported that 33% of fetal growth occurs after day 102 of gestation (McPherson et al. Reference McPherson, Ji and Wu2004). The rapid fetal growth, mammary development, and colostrum formation create a state of nutrient imbalance in late gestation and early lactation (Feyera and Theil Reference Feyera and Theil2017). Sows have an increased demand for iron during late gestation and lactation. This demand is driven by the high iron requirements of the rapidly growing fetus in late gestation and the need to secrete iron-rich milk during lactation. The iron in sow’s milk is vital for newborn piglets, as it is their source of this essential nutrient (El‐Farrash et al. Reference El‐Farrash, Ismail and Nada2011). Although routine iron supplementation is administered to piglets after birth, this does not mean that the iron provided in the sow’s milk is not important. From a production management perspective, ameliorating anemia in piglets or sows through physical injections may increase labor input and management costs. If every newborn piglet requires an iron injection, it will lead to a large amount of repetitive labor input in large-scale farms, which not only increases labor hours and personnel costs but also causes stress to the piglets. Furthermore, iron preparations for injection are veterinary drugs, and their use must strictly adhere to statutory withdrawal periods, dosage standards, and operational protocols to avoid drug residues and food safety risks. Simultaneously, improper injection practices also carry the risk of causing local inflammation, allergic reactions, or even accidental death. Therefore, anemia is a key issue that sows need to address, and currently, iron additives are mainly used to improve anemia.
Iron source selection and supplementation
Comparison of organic and inorganic iron
Feed additives emerged in the 1940s, and the advent of intensive feeding practices has promoted research and application in this field (Summons Reference Summons1968). Despite constituting less than 1% of the feed, additives play a crucial role in supplementing and balancing nutrition, enhancing animal product quality, and preventing feed spoilage (Lin et al. Reference Lin, Ye and Zw2020; Pluske Reference Pluske2013). To date, the development of iron additives has progressed through three distinct stages.
The first generation of iron additives primarily consisted of inorganic salts, such as sulfates, carbonates, and phosphates. Despite their high iron content and cost-effectiveness, which have led to widespread use in livestock production, these additives exhibit several limitations (Additives and Feed Posui Reference Additives and Feed Posui2016). Studies have indicated that the bioavailability of inorganic iron is generally low. First, during storage, Fe2+ is easily oxidized to Fe3+, which is poorly absorbed (Dietzfelbinger Reference Dietzfelbinger1987; Schmitz and Müller Reference Schmitz and Müller1971). Second, inorganic iron is sensitive to environmental factors like humidity and pH, leading to deliquescence and agglomeration, thereby compromising storage stability (Burkett et al. Reference Burkett, Stalder and Powers2009). Furthermore, Sulfate ions can disrupt the intestinal acid–base balance and osmotic pressure homeostasis of piglets, inducing diarrhea (Lee et al. Reference Lee, Shinde and Choi2008). Additionally, inorganic iron sources may react with other feed components, reducing the bioavailability of nutrients (Al-Jaf and Del Reference Al-Jaf and Del2019).
The second generation of additives comprises organic acid salts, such as ferrous citrate and ferrous fumarate, which exhibit significantly higher bioavailability and safety compared to inorganic iron sources (Hemery et al. Reference Hemery, Laillou and Fontan2018; O’sullivan et al. Reference O’sullivan, Byrne and Stagsted2002). This type of additive, by forming coordination compounds or chelates with organic acids or amino acids, possesses characteristics such as high biological potency, low addition amount, and excellent absorption and utilization rate (Freitag and Lückstädt Reference Freitag and Lückstädt2007; Veum et al. Reference Veum, Gallo and Pond1965). Studies have demonstrated that supplementing sow diets with ferrous fumarate, compared to ferrous sulfate, effectively improves the iron status of piglets and enhances their growth performance (Fu et al. Reference Fu, Zhou and Liu2025; Luiggi et al. Reference Luiggi, Berto and Mello2014; Wang et al. Reference Wang, Li and Che2014).
The third generation of iron additives is represented by chelated iron compounds, primarily ferric glycinate and ferric lysine complexes (Hertrampf and Olivares Reference Hertrampf and Olivares2004). The five-or six-membered cyclic chelate structure confers several superior properties (Jacob et al. Reference Jacob, Afify and Shanab2024). First, chelated iron exhibits excellent stability, capable of resisting interference from antinutritional factors and maintaining high iron solubility within the digestive tract (Heli et al. Reference Heli, Mirtorabi and Karimian2011; Wan et al. Reference Wan, Zhang and Wu2018; Wu et al. Reference Wu, Yang and Sun2020). Second, chelated iron has a high absorbability. The iron ions are bonded to amino acid molecules via ionic or covalent bonds, resulting in stable chemical properties (Hertrampf and Olivares Reference Hertrampf and Olivares2004; Nuñez et al. Reference Nuñez, Mazariegos and Pizarro2004), preventing metal ions from reacting with stomach acid to form insoluble precipitates (Cheng et al. Reference Cheng, Chen and Cao2023; Cocato et al. Reference Cocato, Trindade Neto and Berto2008; Ettle et al. Reference Ettle, Schlegel and Roth2007). Finally, chelated iron has good palatability. The neutral amino acid ligand can reduce gastrointestinal irritation, promote appetite (Xiong et al. Reference Xiong, Zhao and Li2025), and enhance the bioavailability of other nutrients (Ashmead Reference Ashmead2001; Ma et al. Reference Ma, Sun and Zhou2012).
Evaluation of the dosage and effect of iron in feeds
According to the NRC (2012) guidelines, the recommended dietary iron supplementation for gestating and lactating sows is 80 mg/kg. In modern swine production, numerous other imbalances in iron nutrient supply are prevalent. Research indicates that conventional feed raw materials have a relatively high natural mineral iron content, enabling the iron level in basal diets to often meet or exceed the actual nutritional requirements of pigs (Buffler et al. Reference Buffler, Becker and Windisch2017). However, industry survey data reveal that the amount of iron added to commercial swine feed is generally significantly higher than the levels recommended by the NRC (2012). A large-scale survey in the U.S. found that approximately 4.24 million commercial swine were fed diets with iron levels 1.3 times to 3.7 times higher than the NRC (2012) standard (Faccin et al. Reference Faccin, Tokach and Goodband2023), highlighting a significant over-addition. Notably, despite the overall excessive iron supply, the incidence of IDA in pig herds on large-scale farms has not diminished. A survey conducted across 10 EU countries on randomly selected farms that agreed to participate found that 47.1% (300/637) of sows were anemic, as determined by the assessment of Hb levels at weaning for 637 sows of different parities (Sperling et al. Reference Sperling, Guerra and Dimitrov2021). However, it should be pointed out that there is still a lack of comprehensive quantitative data on the prevalence of anemia in sows worldwide, mainly from surveys in Europe. The EU study lacks critical background information on feeding practices, precluding definitive conclusions regarding the actual dietary iron levels, the bioavailability of the iron sources used, or the presence of other dietary factors influencing iron absorption and metabolism in the anemic pig herd. In summary, direct comparisons across disparate regions and management systems can readily lead to misinterpretations of the underlying causes of anemia. Future research should concurrently monitor dietary iron levels and anemia biomarkers within standardized feeding management systems. This approach is essential for accurately evaluating the efficacy and potential risks of iron supplementation strategies. Furthermore, large-scale, cross-regional epidemiological studies are warranted to more accurately determine the global prevalence and impact of sow anemia.
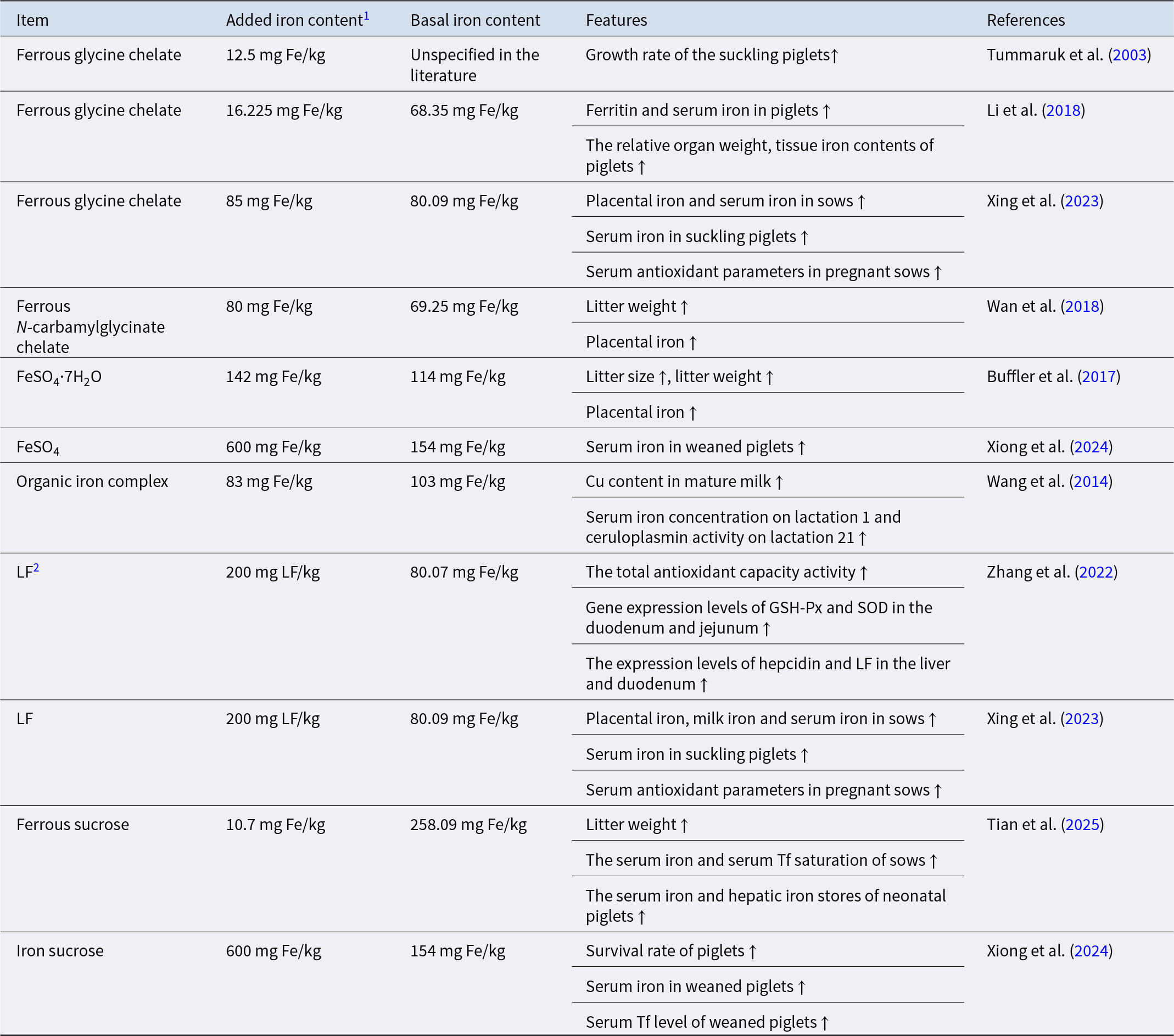
The iron imbalance may be attributed to several factors. First, iron supplementation protocols vary considerably among farms. These variations encompass the mode of iron supplementation, whether by injection or oral administration, the choice between organic and inorganic iron sources, and the dosage standards employed (Table 1). Second, the expansion of farming scale, increased density, and complex management have reduced the utilization efficiency of iron. These factors contribute to a mismatch between supply and demand in swine iron nutrition management, highlighting the need for establishing more scientific and precise iron supplementation systems.
The forms and effects of iron supplementation in gestating sow diets

1 Added iron content: actual iron content calculated based on the amount of iron additive added.
2 LF: the corresponding literature did not mention the iron dosage contained in LF; it was directly referred to as LF.
Iron balance
While maintaining optimal iron status in sows and their offspring is critical, the principle of “more is better” does not apply. Therefore, iron supplementation strategies must strike a balance between correcting deficiency and avoiding the adverse effects of overload.
Sows possess a degree of iron tolerance; prolonged dietary iron supplementation exceeding NRC (2012) recommendations may result in its accumulation in vital organs, including the liver, kidneys, and pancreas. This accumulated iron promotes free radical generation, exacerbating oxidative cellular damage, and potentially inducing cell death, thereby compromising organ function (Hider and Kong Reference Hider and Kong2013). Notably, feeding iron-supplemented diets increases dry matter intake, which in turn leads to a linear rise in the dietary intake of iron, zinc, copper, and manganese. Consequently, fecal excretion of these minerals, particularly iron, zinc, and manganese, is significantly elevated (Lee et al. Reference Lee, Shinde and Choi2008; Rincker et al. Reference Rincker, Hill and Link2005). The long-term application of such iron-rich manure to farmland poses potential environmental risks, including alterations to soil physicochemical properties, disruption of trace element bioavailability, and contribution to water eutrophication (Creech et al. Reference Creech, Spears and Flowers2004).
Physiologically, excess iron catalyzes reactive oxygen species generation via the Fenton reaction, leading to oxidative stress, protein carbonylation, lipid peroxidation, and associated cellular damage (Altamura et al. Reference Altamura, Kessler and Gröne2014). Furthermore, dietary iron is a crucial nutrient for gut microorganisms, including potential pathogens. Consequently, elevated dietary iron can disrupt microbial homeostasis, promote the proliferation of detrimental bacteria, and compromise intestinal health (Rusu et al. Reference Rusu, Suharoschi and Vodnar2020). Lee et al. demonstrated that as the iron level in the feed increases, the number of coliform bacteria in the feces of piglets increases, and the diarrhea incidence also increases, indicating that iron supplementation may stimulate coliform proliferation (Lee et al. Reference Lee, Shinde and Choi2008). In conclusion, iron supplementation must be guided by the principle of precision nutrition, moving beyond the notion of “more is better.”
Perspectives
As livestock intensification and scale-up accelerate, the imbalance of iron metabolism in sows and the resulting anemia have emerged as key constraints on the sustainable development of the pig industry. IDA is one of the most widespread nutritional disorders in the sow population. This iron metabolic disorder not only reduces antioxidant capacity and immunoglobulin synthesis in sows but also significantly increases the risk of disease (Stoffel and Drakesmith Reference Stoffel and Drakesmith2024). Iron is essential for normal immune system development, participating in cytokine production, immune cell proliferation, and the differentiation of monocytes and macrophages (Ganz and Nemeth Reference Ganz and Nemeth2015). Consequently, iron deficiency can impair the immune response to intestinal infections and reduce the bactericidal activity of macrophages (Cronin et al. Reference Cronin, Woolf and Weiss2019). Research indicates that iron deficiency can also disrupt intestinal morphology and compromise nutrient digestion and absorption (Kovac et al. Reference Kovac, Anderson and Baldwin2011). Low-iron feeding can reduce gastric acid secretion in piglets. While this reduction may increase the bioavailability of non-heme iron by competing for iron-binding sites in the diet and maintaining iron solubility, the overall decrease in secretion further compromises gastrointestinal digestive and absorptive functions and may induce diarrhea (Ding et al. Reference Ding, Yu and Feng2020).
Sustainable sow production hinges on key performance metrics, including fertility, feed efficiency, and piglet birth/weaning weights (Ek-Mex et al. Reference Ek-Mex, Segura-Correa and Batista-Garcia2014), which directly determine herd productivity and farm profitability. PSY is a pivotal metric for evaluating both the production efficiency of pig farms and the reproductive performance of sows, with significant variations across countries (Bell et al. Reference Bell, Urioste and Barlocco2015). For instance, the average PSY reaches 30.9 in Denmark (Lopes et al. Reference Lopes, Kruse and Nielsen2019) and 25.3 in North America (Kraeling and Webel Reference Kraeling and Webel2015). In stark contrast, China, as the world’s largest pork producer and consumer, has an average PSY of only approximately 20 (Luo et al. Reference Luo, Li and Sun2014). A higher PSY enables the fixed costs associated with sow purchase and maintenance to be amortized across a greater number of weaned piglets, thereby enhancing the profitability of commercial operations. Litter size and stillbirth rate are two key determinants strongly correlated with PSY. Iron-deficient sows typically exhibit prominent issues, including reduced litter size, decreased piglet birth weight, and a lower survival rate, which directly impair the PSY and undermine the sustainability of swine production system.
Driven by the sustainable development goals of the pig industry, future research on sow anemia should focus on three main directions: multi-omics integrated and mechanism analysis, development of novel iron supplementation technologies, and the formulation of precise nutritional control strategies. A primary reason for the long-standing neglect of sow anemia is the lack of systematic, large-scale data on the global prevalence and economic impact of IDA in sow populations. Current evidence is largely derived from fragmented clinical observations and regional studies, which likely obscure the potential universality and severity of the problem. Consequently, this review addresses not only the established fact of anemia but, more critically, its potential pervasiveness and the attendant uncertainties. A foremost priority for future research is to implement large-scale, cross-sectional surveys across major pig farms worldwide. These surveys should employ standardized biomarkers, including Hb and ferritin, to establish reliable baseline measurements. Equipped with such foundational data, targeted iron supplementation strategies developed for specific populations and physiological stages, whether by revealing individual variations in iron metabolism through multi-omics technologies or employing novel iron sources to enhance utilization efficiency, will have a solid foundation and clear objectives. A study on rats revealed that IDA alters gut microbiota composition. The colons of anemic rats showed increased abundance of certain short-chain fatty acids (SCFAs)-producing genera and SCFAs-related metabolic pathways. This suggests that SCFAs or SCFAs-producing bacteria during IDA may play important roles in mitigating local and systemic diseases and enhancing disease management (Soriano-Lerma et al. Reference Soriano-Lerma, Garcia-Burgos and Alferez2022). Human studies demonstrate that transplanting washed microbiota from healthy donors to anemic patients can reshape gut microbiota composition and function, indicating its potential role in ameliorating anemia (Zhong et al. Reference Zhong, Chen and Lu2023). In recent years, the advent of novel embedding and formulation technologies has enhanced the stability and bioavailability of iron. Edible toys, developed via capsule technology using whey, sodium alginate, ferrous sulfate, and nebulized bovine red blood cells as raw materials, can generate new iron carriers to improve the iron nutrition status of weaned piglets (Anticoi et al. Reference Anticoi, Duran and Avendano2021). Additionally, studies indicate that peptide-iron encapsulated in nanoliposomes exhibits minimal side effects, significant iron supplementation effects, and protective effects on internal organs (Chen et al. Reference Chen, Chen and Zhang2022).
The iron requirement of sows is not constant throughout the reproductive cycle, but changes dynamically according to fetal growth and development, maternal metabolism, and postpartum lactation demands. During early gestation, sows’ iron requirements are relatively low and primarily support maternal metabolism along with embryo implantation and development. Mid-to-late gestation and lactation represent two critical peaks in iron demand. During mid-to-late gestation, both fetal growth rate and maternal weight increase substantially. Therefore, developing precise iron supplementation programs based on dynamic nutritional demand models will be crucial for optimizing sow reproductive performance.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (32230099 and 31925037), the Foundation of Hubei Hongshan Laboratory (No. 2021hszd018), and the Fundamental Research Funds for the Central Universities (Project 2662023DKPY002).
Author Contributions
LLS: Investigation, Writing – original draft. XHY: Supervision, Writing – review & editing.
Conflicts of interest
The authors declare none.