Nitrogen is one of the fundamental elements for crop growth and is therefore essential to meet the increasing food demand linked to population growth (FAO, 2009; Pan et al., Reference Pan, Zhang, Zou, Zhao, Deng, Chen and Huang2019). It is supplied to the soil through the application of chemical fertilizers, such as urea, or organic fertilizers. However, the industrial processes required for the production of synthetic fertilizers are no longer sustainable, as they require high energy consumption and release large amounts of greenhouse gases to the atmosphere (Pesendorfer & Ellersdorfer, Reference Pesendorfer and Ellersdorfer2021). Moreover, once applied to agricultural soils, synthetic fertilizers release nutrients rapidly, often resulting in low use efficiency by crops and the consequent release of large amounts of nitrogen to the environment (Hooda et al., Reference Hooda, Edwards, Anderson and Miller2000; Ferretti et al., Reference Ferretti, Galamini, Medoro, Coltorti, Di Giuseppe and Faccini2020). As an alternative to synthetic fertilizers, liquid digestates derived from the anaerobic digestion of livestock or the organic fraction of municipal solid waste (MD; i.e. municipal digestate) are often applied to sustain crop growth (Ferretti et al., Reference Ferretti, Galamini, Medoro, Coltorti, Di Giuseppe and Faccini2020; Kovačić et al., Reference Kovačić, Lončarić, Jović, Samac, Popović and Tišma2022; Rossi et al., Reference Rossi, Beni, Benedetti, Felici and Neri2023; Galamini et al., Reference Galamini, Ferretti, Rosinger, Huber, Mentler and Pines2025). The nutrient content of these matrices varies depending on the type of livestock and animal diet or the composition of waste (in the case of municipal solid waste wastewaters), as well as the operating conditions of anaerobic digestion plants (Makádi et al., Reference Makádi, Tomócsik, Orosz and Kumar2012). This variability makes it difficult to determine the correct fertilizer dosage to use (Pesendorfer & Ellersdorfer, Reference Pesendorfer and Ellersdorfer2021), increasing the risk of nutrient losses and associated negative environmental impacts, such as water eutrophication, CH4, CO2, N2O and ammonia (NH3) emissions and the release of pathogens into water bodies (Huang et al., Reference Huang, Kankanamge, Chow, Welsh, Li and Teasdale2017; Ferretti et al., Reference Ferretti, Galamini, Medoro, Coltorti, Di Giuseppe and Faccini2020; Fu et al., Reference Fu, Li, Yu, Shen, Li and Zhang2020; Nhubu et al., Reference Nhubu, Mbohwa and Muzenda2020; Nguyen et al., Reference Nguyen, Nguyen, Van, Nguyen, Nguyen and Nguyen2022; Policastro & Cesaro, Reference Policastro and Cesaro2023; Galamini et al., Reference Galamini, Ferretti, Rosinger, Huber, Mentler and Pines2025).
To address these issues, several regulations have been introduced by the European Union (EU), including the Nitrates Directive (91/676/EEC) in 1991 (Huygens et al., Reference Huygens, Orveillon, Lugato and Tavazzi2020). Additionally, the European Green Deal objectives include: (1) reducing nitrogen concentrations in effluents and digestates; (2) limiting greenhouse gas emissions during storage and field application; and (3) preventing soil and water contamination (European Union, 1998; Martinez et al., Reference Martinez, Dabert, Barrington and Burton2009; United Nations, 2015; European Commission, 2020).
In this context, nutrient recovery and recycling from wastewater are crucial, as they could help save energy in production processes while reducing environmental contamination risks (Galamini et al., Reference Galamini, Ferretti, Rosinger, Huber, Medoro and Mentler2023, Reference Galamini, Ferretti, Rosinger, Huber, Mentler and Pines2025). Several technologies have been proposed for nitrogen removal from wastewater, including chemical precipitation, ammonia stripping, electrolytic methods, biological degradation and physical adsorption (Kovačić et al., Reference Kovačić, Lončarić, Jović, Samac, Popović and Tišma2022). Among these, adsorption stands out for its versatility, low operating costs and process simplicity. Materials such as biochar (Hou et al., Reference Hou, Huang, Yang, Zhao and Deng2016; Banik et al., Reference Banik, Bakshi, Andersen, Laird, Smith and Brown2023), nanoparticles (Zare et al., Reference Zare, Sadegh, Shahryari-Ghoshekandi and Asif2016) and natural zeolites (Faccini et al., Reference Faccini, Di Giuseppe, Malferrari, Coltorti, Abbondanzi and Campisi2015; Tasić et al., Reference Tasić, Bogdanović and Antonijević2019; Galamini et al., Reference Galamini, Ferretti, Medoro, Tescaro and Faccini2020) have been widely studied for their ability to adsorb ammonium (NH4+). Zeolites in particular are hydrated aluminosilicates with a porous three-dimensional structure composed of [SiO4]4– and [AlO4]5– tetrahedra, forming channels and cavities capable of hosting exchangeable cations (Coombs et al., Reference Coombs, Alberti, Armbruster, Artioli, Colella and Galli1998; Dyer & Zubair, Reference Dyer and Zubair1998). Their affinity for NH4+ depends on their crystal structure, chemical composition and any chemical treatments they have undergone (Nguyen & Tanner, Reference Nguyen and Tanner1998). Several studies have demonstrated their applicability to the removal of NH4+ from a large variety of wastewaters, and their application to soil also improves soil health in the long term and promotes crop growth while reducing nitrogen losses (Di Giuseppe et al., Reference Di Giuseppe, Ferretti, Faccini, Blasi, Passeri, Bianchini and Coltorti2016; Ali et al., Reference Ali, Nakhli and Delkash2017; Eroglu et al., Reference Eroglu, Emekci and Athanassiou2017; Ghasemi et al., Reference Ghasemi, Sourinejad, Kazemian and Rohani2018; Mpanga et al., Reference Mpanga, Braun and Walworth2020; Cataldo et al., Reference Cataldo, Salvi, Paoli, Fucile, Masciandaro and Manzi2021; Jarosz et al., Reference Jarosz, Szerement, Gondek and Mierzwa-Hersztek2022; Galamini et al., Reference Galamini, Ferretti, Rosinger, Huber, Medoro and Mentler2023; Ferretti et al., Reference Ferretti, Rosinger, Diaz-Pines, Faccini, Coltorti and Keiblinger2024).
Most studies in this area focus on NH4+ adsorption from synthetic solutions (Pan et al., Reference Pan, Zhang, Zou, Zhao, Deng, Chen and Huang2019; Fu et al., Reference Fu, Li, Yu, Shen, Li and Zhang2020; Pesendorfer & Ellersdorfer, Reference Pesendorfer and Ellersdorfer2021; Muscarella et al., Reference Muscarella, Laudicina, Cano, Badalucco, Conte and Mannina2023), while few evaluate its removal efficiency using real wastewaters (Huang et al., Reference Huang, Yang, Xue, Liu, Hou and Ding2015; Galamini et al., Reference Galamini, Ferretti, Medoro, Tescaro and Faccini2020). Moreover, existing research tends to consider a single type of wastewater, whereas a comparative analysis of different types of wastewater is still lacking.
Recently, Alberghini et al. (Reference Alberghini, Ferretti, Galamini, Botezatu and Faccini2025) evaluated the NH4+ adsorption performance of a zeolitic tuff composed of two distinct zeolite phases: phillipsite and chabazite. The study involved testing five anaerobic digestates originating from both livestock effluents and municipal solid waste, which had undergone various pre-treatment processes, including screw separation, microfiltration and centrifugation. The findings demonstrated the high potential of that specific tuff for efficient NH4+ recovery under realistic operational conditions. Moreover, the study offered a preliminary farm-scale assessment, estimating annual nitrogen recovery at between ∼440 and ∼6000 kg N year–1, depending on the digestate and solid-to-liquid (S/L) ratio.
Given the wide diversity of natural zeolitic tuffs, their varying availability across different regions and the distinct properties associated with each zeolite type, further investigation is essential. To enhance both the understanding and scalability of this recovery process, detailed studies on the behaviour and performance of other commercially available zeolitic tuffs are thus necessary.
The aim of this study was therefore to broaden the characterization of commercially available materials for potential use in nutrient recycling technologies by characterizing the behaviour of another diffused zeolitic tuff across a wide spectrum of treatable matrices.
To achieve this aim, the experimental design of Alberghini et al. (Reference Alberghini, Ferretti, Galamini, Botezatu and Faccini2025) was adopted to assess the performance of a zeolitic tuff composed predominantly of chabazite (∼70%) in NH4+ removal from three types of digestates derived from municipal solid waste, swine and cattle livestock. The study focused on the practical applicability of the chabazite-rich tuff for ammonium recovery under both laboratory and preliminary farm-scale conditions, including the effects of digestate composition, solids content, pre-treatment processes, competing ions and NH4+ concentrations. Adsorption isotherms and kinetics were constructed to describe the process, and the potential amounts of nitrogen recoverable on a daily and annual basis by a model farm implementing this approach were estimated, providing actionable insights to support the development of scalable nutrient recovery applications. The NH4+-saturated tuff produced through this process is designed for direct use as a slow-release soil amendment, being fully consistent with the objectives of the European Green Deal.
Materials and methods
Materials
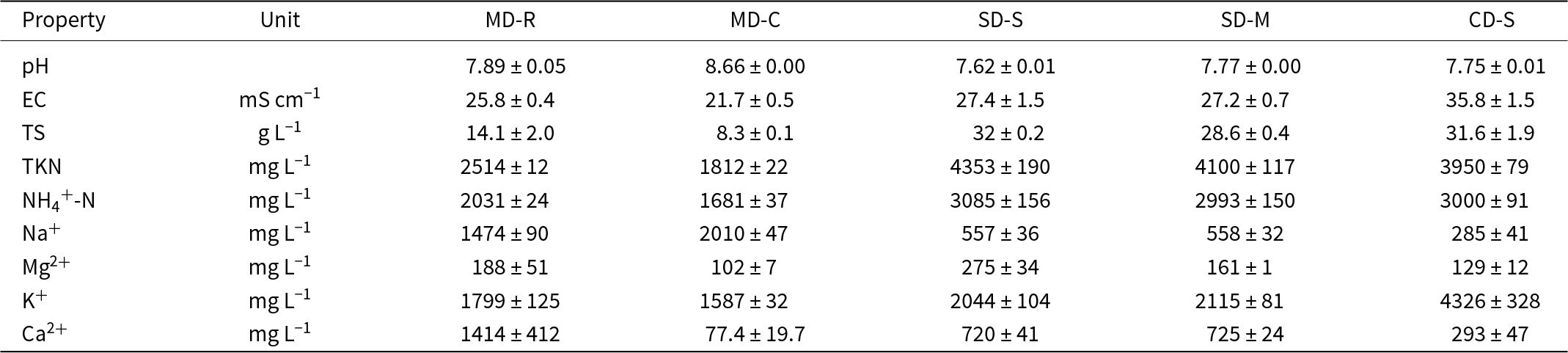
The chemical characteristics of the digestates used are reported in Table 1. The cattle-derived digestate, collected from a biogas plant near San Biagio (Ferrara province, Italy), was available only after separation by screw compression process (CD-S). The swine-derived digestate was sampled from a swine livestock near Formigine (Modena province, Italy) and was analysed in two different forms: after separation by screw compression (SD-S) and after microfiltration (SD-M). This latter process consisted of a microfiltration step after the screw compression using a 40 MFT SAVECO filter from WAM Group S.p.A. (Modena province, Italy). The MD-derived digestates were collected from an anaerobic digestion plant near Spilamberto (Modena province, Italy). These were available in two fractions: the raw digestate (MD-R), corresponding to the liquid fraction leaving the digester, and the clarified digestate (MD-C), obtained through a centrifugation process aimed at reducing suspended solids.
Table 1. Physicochemical properties of the digestates. Values are expressed as mean ± standard deviation, based on three replicates.

EC = electrical conductivity; TKN = total Kjeldahl nitrogen.
All digestates exhibited a high concentration of NH4+-N and total solids (TS), with particularly elevated values observed in those derived from livestock manure. The cattle digestate (CD-S) also showed a notably high potassium (K+) content.
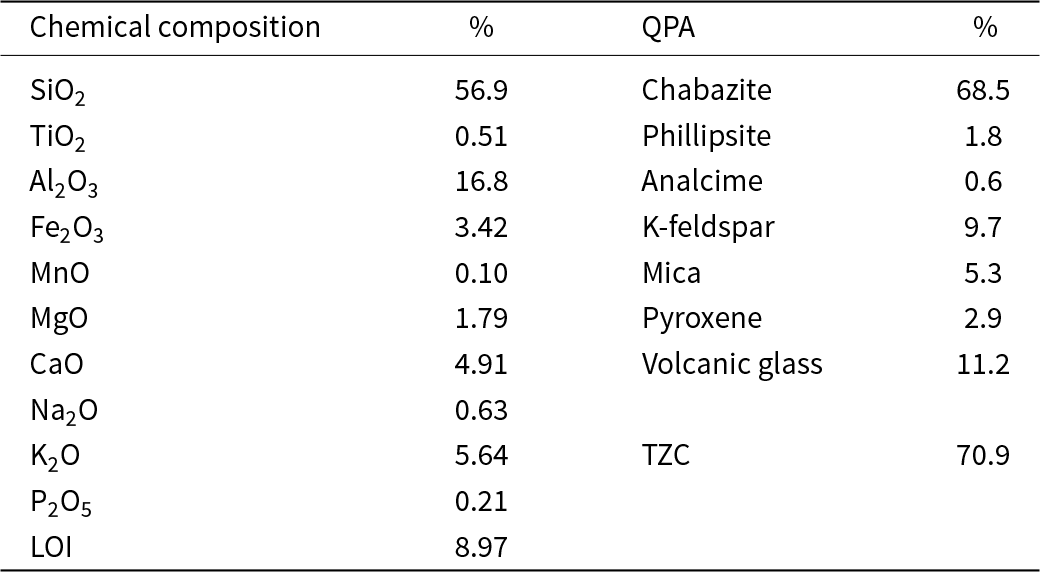
The zeolitic tuff used is a chabazite-rich tuff extracted from central Italy. The material belongs to the Lithic Yellow Tuff (Sorano Formation) and is part of the Latera Volcanic Complex, a stratigraphic sequence primarily composed of pyroclastic deposits (Vezzoli et al., Reference Vezzoli, Conticelli, Innocenti, Landi, Manetti, Palladino and Trigila1987). This material is the same as that employed in many previous studies (Di Giuseppe et al., Reference Di Giuseppe, Ferretti, Faccini, Blasi, Passeri, Bianchini and Coltorti2016; Galamini et al., Reference Galamini, Ferretti, Rosinger, Huber, Medoro and Mentler2023, Reference Galamini, Ferretti, Medoro, Eftekhari, Favero, Faccini and Coltorti2024, Reference Galamini, Ferretti, Rosinger, Huber, Mentler and Pines2025; Ferretti et al., Reference Ferretti, Rosinger, Diaz-Pines, Faccini, Coltorti and Keiblinger2024). The tuff was quarried from the ‘Piandirena’ site, an area of ∼60,000 m2 near Sorano (Grosseto province, Italy), located at ∼450 m above sea level (42°41′26.52″N, 11°44′35.54″E), and it was provided by Verdi S.p.A. company (Reggio Emilia, Italy). The mineralogical composition of the zeolitic tuff is listed in Table 2. The cation-exchange capacity (CEC) previously determined by Malferrari et al. (Reference Malferrari, Laurora, Brigatti, Coltorti, Di Giuseppe and Faccini2013) is 217 cmol kg⁻1, with Ca2+ being the prevalent exchangeable cation (146 cmol kg⁻1), followed by K+ (60 cmol kg⁻1), Na+ (7 cmol kg⁻1) and Mg2+ (4 cmol kg⁻1). The particle-size distribution of the tuff was determined using the sequential sieving method as follows: 12.5% between 5 and 2 mm, 60.4% between 2 and 0.8 mm, 24.1% between 0.8 and 0.425 mm and 3.0% below 0.425 mm. Prior to the experiments, the zeolitic tuff was repeatedly rinsed with Type I ultrapure water (18.2 MΩ cm–1 at 25°C) and dried at 105°C for 48 h to remove gravimetric water.
Table 2. The zeolitic tuff characteristics. Chemical composition data were obtained through X-ray fluorescence analysis.

LOI = loss on ignition; QPA = quantitative phase analysis; TZC = total zeolitic content.
Methods
Batch adsorption experiment
From an industrial perspective, it is important to evaluate the NH4+ sorption efficiency of the adsorbent by studying both adsorption isotherm and kinetics. Isotherms describe the equilibrium between adsorption and desorption at thermodynamic equilibrium and are useful for optimizing a process, particularly for determining the amount of adsorbent required to achieve a specific removal efficiency. Kinetics, on the other hand, are useful to define the ideal contact time between the digestate and the zeolitic tuff needed to reach the desired removal efficiency.
The NH4+ removal efficiency (R E%) of the zeolitic tuff was determined using Equation 1 (Detho et al., Reference Detho, Memon, Memon, Almohana, Daud and Rosli2023), whereas the amount of NH4+ adsorbed at equilibrium (q e; mg g⁻1) was determined using Equation 2 (Fu et al., Reference Fu, Li, Yu, Shen, Li and Zhang2020):
 \begin{equation}{R_{\text{E}}}\left( {\text{% }} \right) = {\text{ }}\frac{{\left( {{C_0} - {C_{\text{e}}}} \right)}}{{{C_0}}} \times 100\end{equation}
\begin{equation}{R_{\text{E}}}\left( {\text{% }} \right) = {\text{ }}\frac{{\left( {{C_0} - {C_{\text{e}}}} \right)}}{{{C_0}}} \times 100\end{equation}where C 0 (mg L–1) is the initial NH4+ concentration and C e (mg L–1) is the equilibrium concentration; and
 \begin{equation}{q_{\text{e}}} = {\text{ }}\frac{{\left( {{C_0} - \left( {{C_0} - {C_{\text{b}}}} \right) - {C_{\text{e}}}} \right) \times V}}{m}\end{equation}
\begin{equation}{q_{\text{e}}} = {\text{ }}\frac{{\left( {{C_0} - \left( {{C_0} - {C_{\text{b}}}} \right) - {C_{\text{e}}}} \right) \times V}}{m}\end{equation}where C 0 (mg L–1) is the initial NH4+ concentration, C b (mg L–1) is the NH4+ concentration in blanks after 24 h, C e (mg L–1) is the concentration at equilibrium, V (L) is the volume of digestate and m (g) is the mass of zeolitic tuff. Blank samples without zeolitic tuff were considered to assess the possible effects of ammonia stripping or NH4+ adsorption on the surface of the plastic containers.
Isotherms were generated at 25°C using 12 different S/L ratios, expressed as mass of zeolitic tuff per volume of digestate (w/v%). These ratios ranged from 1% to 50%, meaning that variable amounts of zeolitic tuff (0.8–40 g) were added to 80 mL of digestate in 100 mL sealed plastic bottles, which were shaken at 200 rpm for 24 h (assumed equilibrium time). After shaking, the NH4+ concentration in the liquid phase was measured immediately. According to previous studies, zeolite adsorption of NH4+ remains constant within a pH range of 2–8 but declines significantly at pH values above 9 (Galamini et al., Reference Galamini, Ferretti, Medoro, Tescaro and Faccini2020; Guo et al., Reference Guo, Zeng, Li and Park2008). Given that the pH of all tested digestates ranged between 7.7 and 8.5, pH variation was not specifically investigated so as to avoid additional chemical inputs.
Kinetic experiments
Kinetic adsorption models of NH4+ onto zeolitic tuff were determined for each digestate. A fixed amount of zeolitic tuff corresponding to 5% S/L ratio (w/v) was added (i.e. 25 g of zeolitic tuff in 500 mL of each digestate), and the suspensions were shaken in an orbital shaker at 25°C and 200 rpm for 420 min. At 12 time intervals, corresponding to 5, 10, 15, 20, 30, 45, 60, 90, 120, 180, 360 and 420 min, 10 mL aliquots were sampled and immediately analysed for NH4+ concentration. The progressive volume reduction due to sampling was considered in the subsequent calculations of adsorption capacity over time. The amount of NH4+ adsorbed at any time (qt; mg g⁻1) was determined using Equation 2, substituting q e and C e with qt and Ct, respectively.
Isotherm and kinetic models
Several isotherm models have been proposed in the literature to describe the equilibrium adsorption of NH4+ (Wasielewski et al., Reference Wasielewski, Rott, Minke and Steinmetz2018; Fu et al., Reference Fu, Li, Yu, Shen, Li and Zhang2020; Galamini et al., Reference Galamini, Ferretti, Medoro, Tescaro and Faccini2020; Eberle et al., Reference Eberle, Börnick and Stolte2022; Muscarella et al., Reference Muscarella, Laudicina, Cano, Badalucco, Conte and Mannina2023; Zhou et al., Reference Zhou, Wang, Wang, Xu and Zhu2024). Among these, the Langmuir and Freundlich models are frequently identified as the most suitable for describing NH4+ adsorption on natural zeolites. Consequently, these models were selected in this study as well.
The isotherm data were processed using RStudio (version 2023-3.0-386; R Core Team, 2021) with the PUPAIM package (version 0.3.1; Deocaris & De Osio, Reference Deocaris and De Osio2020). Model performance was assessed based on the coefficient of determination (R 2), Akaike information criterion (AIC) and p-value. As highlighted by Wang & Guo (Reference Wang and Guo2020a), nonlinear models provide more accurate parameter estimations. Therefore, the parameters of the Langmuir and Freundlich models were determined using their nonlinear formulations.
The Freundlich adsorption isotherm describes multilayer adsorption on heterogeneous surfaces, where adsorption is non-ideal and reversible (Freundlich, Reference Freundlich1906). It follows Equation 3:
 \begin{equation}{q_{\text{e}}} = {\text{ }}{{\text{K}}_{\text{F}}}C_{\text{e}}^{1/n}\end{equation}
\begin{equation}{q_{\text{e}}} = {\text{ }}{{\text{K}}_{\text{F}}}C_{\text{e}}^{1/n}\end{equation}where q e is the equilibrium adsorption capacity (mg g–1), KF is the Freundlich constant, C e the equilibrium concentration (mg L–1) and n is an empirical parameter related to adsorption intensity.
The Langmuir isotherm assumes monolayer adsorption on homogeneous sites, meaning each site can hold only one adsorbate molecule, with no interactions occurring between them (Wang & Guo, Reference Wang and Guo2020a). It follows Equation 4:
 \begin{equation}{{\text{q}}_{\text{e}}} = {\text{ }}\frac{{{q_{{\text{max}}}}{{\text{K}}_{\text{L}}}{C_{\text{e}}}}}{{1 + {{\text{K}}_{\text{L}}}{C_{\text{e}}}{\text{ }}}}\end{equation}
\begin{equation}{{\text{q}}_{\text{e}}} = {\text{ }}\frac{{{q_{{\text{max}}}}{{\text{K}}_{\text{L}}}{C_{\text{e}}}}}{{1 + {{\text{K}}_{\text{L}}}{C_{\text{e}}}{\text{ }}}}\end{equation}where q e is the equilibrium adsorption capacity (mg g–1), q max represents the maximum adsorption capacity (mg g–1), Ce is the concentration at equilibrium (mg L–1) and KL is the Langmuir constant (L mg–1). Adsorption favourability is assessed via the separation factor R L (Weber & Chakravorti, Reference Weber and Chakravorti1974), calculated using Equation 5:
 \begin{equation}{R_{\text{L}}} = {\text{ }}\frac{1}{{1 + {{\text{K}}_{\text{L}}}{C_0}{\text{ }}}}\end{equation}
\begin{equation}{R_{\text{L}}} = {\text{ }}\frac{1}{{1 + {{\text{K}}_{\text{L}}}{C_0}{\text{ }}}}\end{equation}where C 0 is the initial concentration. Adsorption is favourable when 0 < R L < 1, unfavourable if R L > 1, linear if R L = 1 and irreversible if R L = 0.
The spontaneity of adsorption is evaluated through the Gibbs free energy (ΔG°), calculated using Equation 6 (Jha & Hayashi, Reference Jha and Hayashi2009):
where R is the gas constant (8.314 J mol–1⋅K–1), T is the absolute temperature and Ke is the thermodynamic equilibrium constant. Ke is determined by extrapolating the intercept in a plot of q e/C e vs C e as C e approaches zero (Khan & Singh, Reference Khan and Singh1987). A negative ΔG° indicates a spontaneous and energetically favourable adsorption process.
Several kinetic models have been used in the past to describe the adsorption of NH4+ onto zeolitic tuff, including the pseudo-first-order (PFO), pseudo-second-order (PSO) and Elovich models (Wen et al., Reference Wen, Ho and Tang2006; Pan et al., Reference Pan, Zhang, Zou, Zhao, Deng, Chen and Huang2019; Galamini et al., Reference Galamini, Ferretti, Medoro, Tescaro and Faccini2020; Damodara & Parameswaran, Reference Damodara and Parameswaran2021; Duyen et al., 2022; Eberle et al., Reference Eberle, Börnick and Stolte2022). However, the PFO and PSO models are generally more efficient at fitting experimental data; therefore, they were selected for this study. Adsorption kinetics should be analysed using data far from equilibrium (Simonin, Reference Simonin2016). Therefore, only data with a fractional uptake F(t) ≤ 0.90 were considered.
Experimental data was processed using RStudio (version 2023-3.0-386) and the PUPAK package (version 0.1.1; Magalong et al., Reference Magalong, DelaCruz, Bumatay and Deocaris2022). The best-fitting kinetic model was assessed based on statistical parameters, including the root mean square error (RMSE), mean square error (MSE), AIC and Bayesian information criterion (BIC), where lower values indicate a better fit.
The nonlinear PFO model, proposed by Lagergen (Reference Lagergren1898), is described by Equation 7:
where q e and qt represent the adsorption capacity at equilibrium and at time t (mg g–1), respectively, while k1 is the PFO rate constant (min–1).
The nonlinear form of the PSO model is given by Equation 8:
 \begin{equation}{q_t} = {\text{ }}\frac{{{{\left( {{q_{\text{e}}}} \right)}^2}{{\text{k}}_2}t}}{{1 + {q_{\text{e}}}{{\text{k}}_2}t{\text{ }}}}\end{equation}
\begin{equation}{q_t} = {\text{ }}\frac{{{{\left( {{q_{\text{e}}}} \right)}^2}{{\text{k}}_2}t}}{{1 + {q_{\text{e}}}{{\text{k}}_2}t{\text{ }}}}\end{equation}where q e and qt represent the adsorption capacity at equilibrium and at time t (mg g–1), respectively, while k2 is the PSO rate constant (g mg–1 min–1).
As the PFO and PSO models do not provide insights into the diffusion mechanism of NH4+, the Intraparticle Diffusion (ID) model (Weber et al., Reference Weber, Walter and Morris1963), described by Equation 9, was applied to the experimental data:
where qt (mg g–1) is adsorption capacity at time t (min), kID (mg g–1 min0.5) is the ID rate constant and C is the intercept of the slope of the curve, which is also indicative of the boundary-layer thickness. If the plot of qt vs t 0.5 intercepts the origin (C ≈ 0), ID is considered to be the rate-controlling step (Wasielewski et al., Reference Wasielewski, Rott, Minke and Steinmetz2018). The presence of multiple linear regions in a plot suggests a multi-stage adsorption process, with the first region corresponding to surface adsorption or macropore diffusion (Halim et al., Reference Halim, Aziz, Johari and Ariffin2010; Widiastuti et al., Reference Widiastuti, Wu, Ang and Zhang2011).
Model-based calculation of nitrogen recovery from digestates
To evaluate the amount of nitrogen recovery at the farm scale, a theoretical calculation was performed based on the average daily volume of digestate produced per biogas plant, which, based on the national reports, was assessed to be 10 m3 per day (Dal Verme et al., Reference Dal Verme, Lipari, Lucido, Maio, Surace and Liberatore2021; European Biogas Association, 2022). Based on this data, the amount of N recoverable per cycle was calculated using Equation 10, and the amount of N recoverable yearly was calculated using Equation 11:
 \begin{align}{\text{kg N recovered year}} & = {\text{ kg N recovered per cycle}}\nonumber\\
& \quad\times {\text{number of cycles}} \times \ 365\end{align}
\begin{align}{\text{kg N recovered year}} & = {\text{ kg N recovered per cycle}}\nonumber\\
& \quad\times {\text{number of cycles}} \times \ 365\end{align}The duration of each treatment cycle, representing the time during which fresh digestate is in contact with the zeolitic tuff, was calculated by considering both the time required to reach equilibrium in the kinetic experiments and the additional time needed for loading and unloading the pilot-scale prototype, estimated as 20 min before and after the equilibrium contact time. The number of cycles was then derived assuming continuous plant operation, with the S/L ratio and the amount of NH4+ adsorbed at equilibrium (q e) corresponding to the optimal values identified in the experimental trials.
Analytical techniques
Electrical conductivity (EC) was determined in digestate samples diluted 1:10 (w/v) using an RS 180-7127 probe (Hanna Instrument, Woonsocket, RI, USA), while pH measurements were performed on undiluted samples with an electrode connected to an 877 Titrino plus automatic titration unit (Methrom, Origgio, Italy). Total Kjeldahl nitrogen (TKN) was quantified after digesting the samples with 98% H2SO4 and catalyst tablets containing TiO2, CuSO4 and K2SO4 using an IR Digester K-425 (Büchi, Flawil, Switzerland). The digested samples were then distilled with a K-360 unit (Büchi, Flawil, Switzerland), employing a 4% H3BO3 acid trap pre-buffered at pH 4.65 ± 0.01 to capture the released NH3. NH4+ was subsequently determined by endpoint titration with 0.25 M H2SO4 using the 877 Titrino plus automatic titration unit (Methrom, Origgio, Italy). NH4+ concentration was measured by directly distilling the samples, bypassing the digestion step. The concentrations of major cations (Na+, K+, Ca2+ and Mg2+) in the digestates were analysed using inductively coupled plasma mass spectrometry (ICP-MS) with a Thermo Electron Corporation X Series mass spectrometer (Thermo Fisher Scientific, Bremen, Germany). Samples were pre-diluted 1:500, filtered through 0.45 μm polytetrafluoroethylene (PTFE) syringe filters and stabilized with 2% HNO3 to maintain pH < 2. TS content was determined by measuring the weight change of a known sample volume after drying overnight at 105°C. The chemical composition of the zeolitic tuff was assessed through X-ray fluorescence (XRF) analysis on powder pellets using a wavelength-dispersive ARL Advant’X spectrometer (Thermo Scientific, Waltham, MA, USA). The estimated accuracy and precision are within 3% for major elements such as Si, Ti, Fe, Ca and K and within 7% for Mg, Al, Mn and Na; for trace elements (above 10 ppm), both accuracy and precision remain below 10%.
The mineralogical characterization of the zeolitic tuff was performed using X-ray powder diffraction (XRPD) with an X’Pert PRO modular powder diffractometer (PANalytical, Malvern, UK). To minimize preferred crystallite orientations, the zeolitic tuff was back-loaded. The amorphous phase content was quantified by adding 20% (w/w) ZnO to the sample. Rietveld refinements and quantitative phase analyses were conducted using the fundamental parameters approach implemented in Profex–BGMN (version 5.0.1; Cheary et al., Reference Cheary, Coelho and Cline2004), with crystal structures sourced from the BGMN database.
Results and discussion
Ammonium removal kinetics
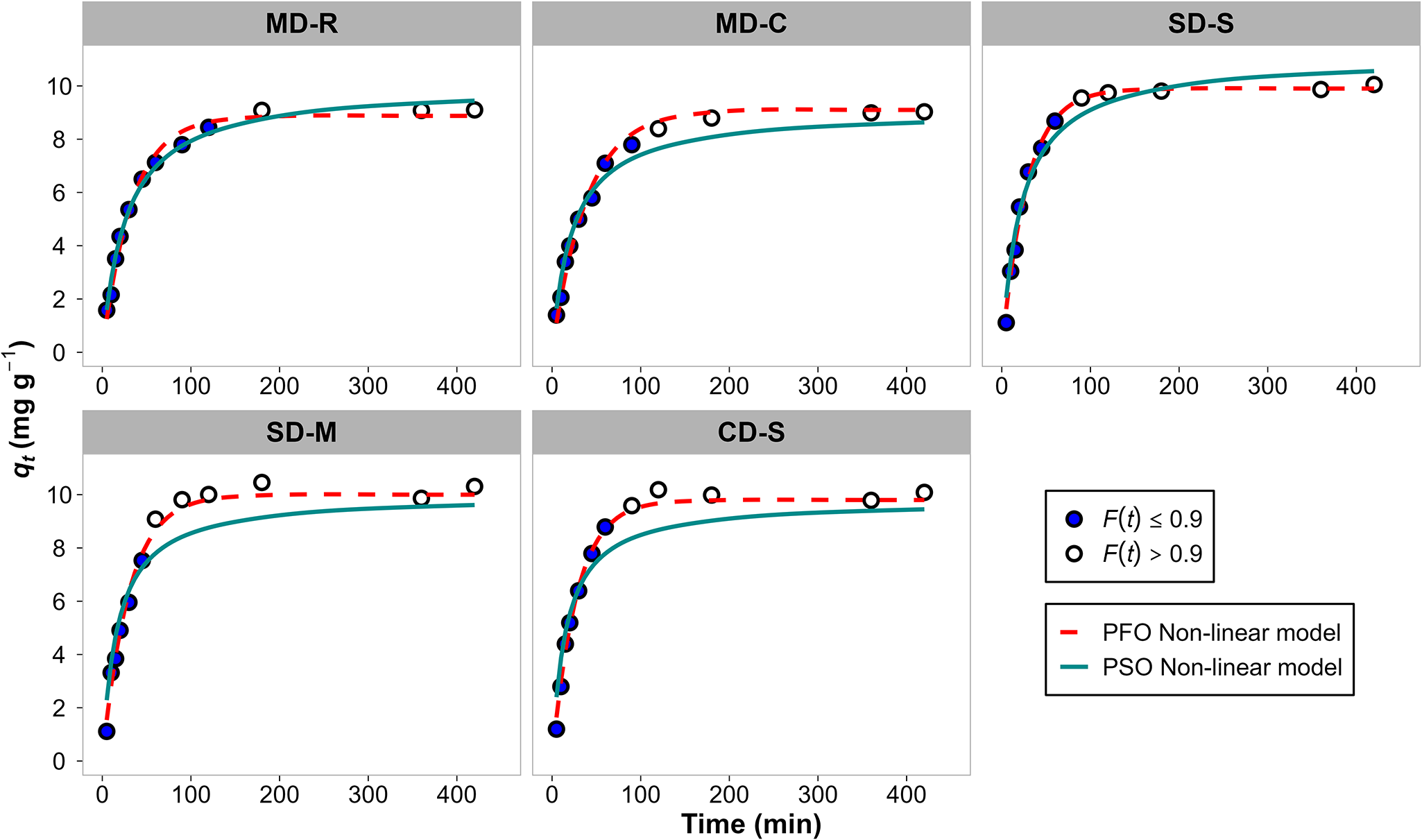
Figure 1 shows the kinetics of NH4+ adsorption up to 420 min. NH4+ adsorption showed a relatively fast initial uptake. The equilibrium stage was attained after 120 min for MD-R and MD-C and within 90 min for SD-S, SD-M and CD-S. The high amount of NH4+ adsorbed by the zeolitic tuff at the beginning of the experiment could be attributed to both the greater availability of negatively charged surface sites and the presence of extra-framework sites available for ion exchange during the early stages of the adsorption process (Sánchez-Hernández et al., Reference Sánchez-Hernández, Padilla and López-Andrés2018; Wang & Guo Reference Wang and Guo2020b; Vamvuka et al., Reference Vamvuka, Loupasis, Chamilaki and Sdoukou2024). A cation may either adsorb onto the external surface of the channels or exchange with the cations originally occupying the zeolitic cages (Chen et al., Reference Chen, Lin, Chen, Yoshiyuki, Liou and Huang2018). However, a detailed investigation of the precise location and distribution of NH4+ within the zeolite framework is beyond the scope of this study, which focuses on evaluating the overall adsorption performance under practical conditions. As the adsorption process continued, the number of available sites decreased, and it approached zero at equilibrium. The differences in the time required to reach equilibrium between the different digestates could be due to the different initial NH4+ concentrations. An increase in NH4+ concentration may enhance the saturation rate of exchange sites, thereby promoting a more rapid establishment of the adsorption equilibrium. Several others studies reported a similar trend (Huang et al., Reference Huang, Xiao, Yan and Yang2010, Reference Huang, Yang, Xue, Liu, Hou and Ding2015; Cheng & Ding, Reference Cheng and Ding2015; Kotoulas et al., Reference Kotoulas, Agathou, Triantaphyllidou, Tatoulis, Akratos, Tekerlekopoulou and Vayenas2019; Galamini et al., Reference Galamini, Ferretti, Medoro, Tescaro and Faccini2020; Tang et al., Reference Tang, Xu, Wang, Lv and Shi2020; Damodara & Parameswaran, Reference Damodara and Parameswaran2021; Liu, et al., Reference Liu, Zhang, Liu, Liu, Liu, Yang and Yang2022; Susilawati et al., Reference Susilawati, Andriayani, Sihombing, Saragi, Masruchin, Nuryawan and Irma2025). For example, Huang et al. (Reference Huang, Xiao, Yan and Yang2010) found that the amount of NH4+ adsorbed increased very quickly within the first 60 min to reach the equilibrium stage after 120 min. In addition, Damodara & Parameswaran (Reference Damodara and Parameswaran2021) and Kotoulas et al. (Reference Kotoulas, Agathou, Triantaphyllidou, Tatoulis, Akratos, Tekerlekopoulou and Vayenas2019) observed a rapid increase in the amount of NH4+ adsorbed by a zeolitic tuff in the early stages of the adsorption process and the attainment of equilibrium after 90–120 min. These aspects are critical for process optimization at the real-world scale, particularly in terms of energy consumption, processing time and overall cost efficiency, as faster attainment of equilibrium enables shorter contact times and reduces equipment operational time.
Given the importance of kinetic analysis for designing processes in water treatment applications, the adsorption kinetics were assessed through nonlinear regression by fitting the PFO and PSO models (Fig. 1).

Figure 1. Kinetics of NH4+ adsorption for the different digestates; data with F(t) ≤ 90% have been considered in kinetic analyses; data with F(t) > 90% were not taken into consideration.
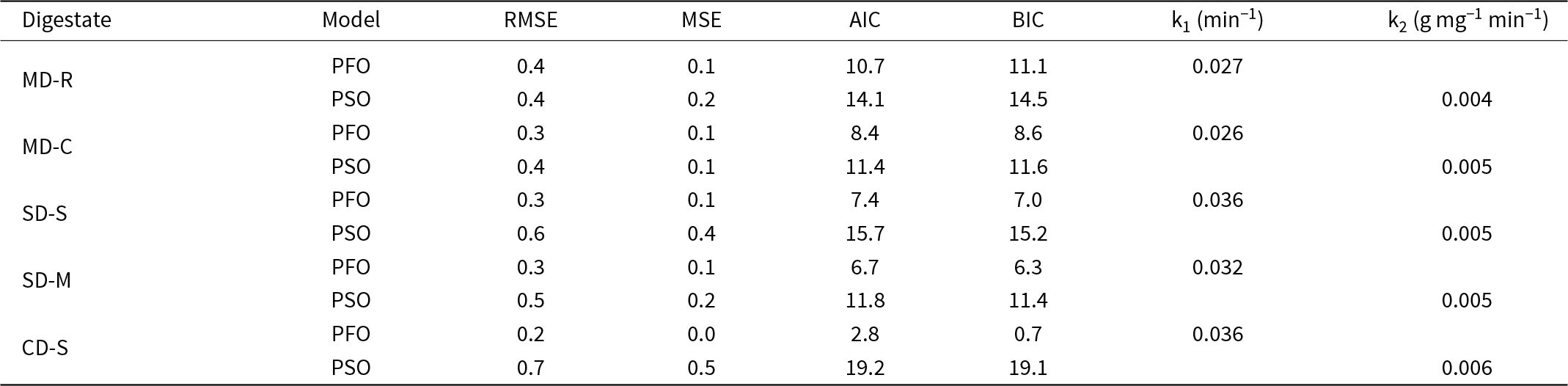
The PFO model provided a better fit than the PSO model for all types of digestate (Fig. 1 & Table 3). This is at odds with most previous works on NH4+ adsorption onto zeolitic tuffs, where the PSO model typically offers a better representation of the kinetic data (Halim et al., Reference Halim, Aziz, Johari and Ariffin2010; Huang et al., Reference Huang, Xiao, Yan and Yang2010; Moussavi et al., Reference Moussavi, Talebi, Farrokhi and Sabouti2011; Widiastuti et al., Reference Widiastuti, Wu, Ang and Zhang2011; Wasielewski et al., Reference Wasielewski, Rott, Minke and Steinmetz2018; Tang et al., Reference Tang, Xu, Wang, Lv and Shi2020; Damodara & Parameswaran, Reference Damodara and Parameswaran2021). However, this study is in agreement with Sánchez-Hernández et al. (Reference Sánchez-Hernández, Padilla and López-Andrés2018), who also observed a better fit with the PFO model under specific experimental conditions. In previous studies, all kinetic data points were considered during modelling. However, including data points close to the equilibrium may mask the actual adsorption behaviour, often leading to a better fit with models such as the PSO model, even when they may not be the most representative (Simonin, Reference Simonin2016). In contrast, focusing on the earlier stages of adsorption (i.e. data farther from the equilibrium) can sometimes reveal a better fit with models such as the PFO model, as observed in this study and in Sánchez-Hernández et al. (Reference Sánchez-Hernández, Padilla and López-Andrés2018). In fact, the PSO equation often provides a good fit at the equilibrium stage but may not accurately describe the adsorption process across the entire time range (Ho & Mckay, Reference Ho and McKay1998). The PFO model describes the NH4+ removal kinetics where the rate of removal is initially fast and proportional to the remaining amount to be removed, then decreasing as equilibrium is approached. This behaviour, which is typical of many adsorbent systems including zeolitic materials, is well described by the PFO model, which empirically represents processes often governed by surface diffusion or external control. The kinetic parameters, including k₁ and k2, were determined using the PUPAK tool. k₁ in particular provides insights into how rapidly the system approaches equilibrium (Plazinski et al., Reference Plazinski, Rudzinski and Plazinska2009). In our study, k1 decreased as the NH4+ concentration increased, implying a greater driving force that accelerates adsorption, particularly in the initial stages (Taddeo et al., Reference Taddeo, Prajapati and Lepistö2017). However, plotting the PSO and PFO models alone is insufficient to fully describe the kinetics of adsorption. Therefore, the ID model was also applied to explore the diffusion mechanism of NH4+. Figure 2 & Table 4 show the qt vs t 0.5 plots and the corresponding ID model parameters, respectively.

Figure 2. ID plots: amount of NH4+ adsorbed at any time (qt) vs t 0.5 plot for the different digestates.
Table 3. Statistical and kinetic parameters of the tested PSO and PFO models.

Table 4. ID parameters for the ID model.

In agreement with several previous studies (Huang et al., Reference Huang, Yang, Xue, Liu, Hou and Ding2015; Galamini et al., Reference Galamini, Ferretti, Medoro, Tescaro and Faccini2020; Damodara & Parameswaran, Reference Damodara and Parameswaran2021; Susilawati et al., Reference Susilawati, Andriayani, Sihombing, Saragi, Masruchin, Nuryawan and Irma2025), the qt vs t 0.5 plots did not pass through the origin, indicating that ID contributed to NH4+ removal but was not the only process affecting the adsorption rate. Three distinct linear regions were observed in all qt vs t 0.5 plots. The first region represents the initial stage of adsorption, where rapid uptake is attributed to external surface adsorption or instantaneous ion exchange. This phase was particularly short for digestates with greater initial NH4+ concentrations, such as those of livestock origin, confirming a strong initial driving force. The second region corresponds to the gradual adsorption phase, where ID contributes to a slower uptake (Hou et al., Reference Hou, Huang, Yang, Zhao and Deng2016). The adsorption rate constant for the first region (kID1) is consistently higher than that of the second region (kID2), and the diffusion boundary-layer thickness in region 2 (C 2) is larger than in region 1 (C 1) for all digestates (Table 4). This indicates that the removal efficiency was initially greater due to the abundance of available exchange sites and greater surface accessibility (Zhou et al., Reference Zhou, Wang, Wang, Xu and Zhu2024). As adsorption progressed, a boundary layer formed due to interactions between the zeolitic surface and NH4+ ions. Once external sites were saturated, further uptake depended on the diffusion of ions into the internal structure of the adsorbent. However, the non-zero intercepts of the qt vs t 0.5 plots confirm that ID was not the sole process involved (Karaçetin et al., Reference Karaçetin, Sivrikaya and Imamoğlu2014). Finally, the third region reflects the equilibrium phase, in which all active sites are saturated and the adsorbent attains maximum capacity (Damodara & Parameswaran, Reference Damodara and Parameswaran2021).
Effects of the S/L ratio and digestate properties
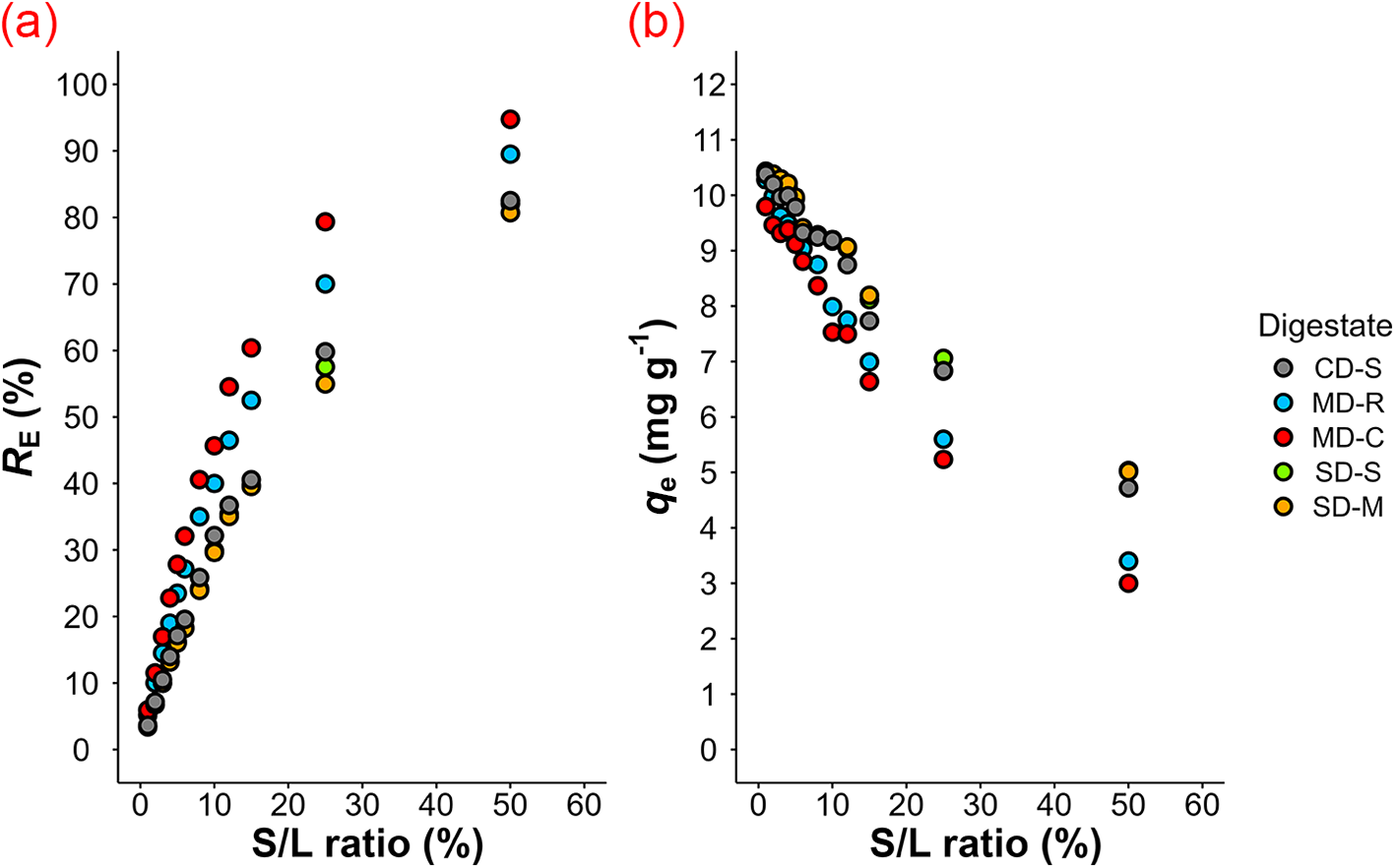
As expected, increasing the S/L ratio led to greater removal efficiency but simultaneously to lower q e values (Fig. 3a,b) because a larger mass of zeolitic tuff provides more exchangeable sites, allowing for a greater total amount of NH4+ to be removed from the digestate (Sánchez-Hernández et al., Reference Sánchez-Hernández, Padilla and López-Andrés2018). NH4+ ions migrate from the digestate towards the adsorbent surface mainly due to electrostatic attraction and occupy the active adsorption sites. At lower S/L ratios, adsorption sites become saturated faster, resulting in higher q e values, whereas at higher S/L ratios, many active sites remain unoccupied, leading to lower NH4+ adsorption per unit mass of zeolite (Mazloomi & Jalali, Reference Mazloomi and Jalali2016; Kotoulas et al., Reference Kotoulas, Agathou, Triantaphyllidou, Tatoulis, Akratos, Tekerlekopoulou and Vayenas2019). As the exchangeable sites on the zeolite surface become fully occupied, equilibrium is reached, limiting further NH4+ removal (Susilawati et al., Reference Susilawati, Andriayani, Sihombing, Saragi, Masruchin, Nuryawan and Irma2025). Among the tested digestates, the removal efficiency followed the trend MD-C > MD-R > SD-S > CD-S > SD-M, highlighting a generally greater performance for digestates derived from municipal solid waste compared to those of swine or cattle origins. These findings are consistent with previous studies reporting that NH4+ removal efficiency tends to decrease significantly at higher S/L ratios (Huang et al., Reference Huang, Xiao, Yan and Yang2010; Sánchez-Hernández et al., Reference Sánchez-Hernández, Padilla and López-Andrés2018; Tang et al., Reference Tang, Xu, Wang, Lv and Shi2020; Susilawati et al., Reference Susilawati, Andriayani, Sihombing, Saragi, Masruchin, Nuryawan and Irma2025). Our observation is especially relevant for industrial applications, where achieving high NH4+ removal with minimal zeolitic tuff usage is essential to reduce operational costs and enhance process sustainability. From an industrial perspective, the use of very high S/L ratios is generally avoided, as they would entail excessive costs in terms of both the adsorbent material and operation processes. Nevertheless, it should be considered that higher adsorbent dosages are required when the objective is to maximize the reduction of the NH4+ concentration in the wastewater, whereas lower dosages are more suitable when the aim is to maximize the nitrogen load per unit of adsorbent.

Figure 3. (a) NH4+ removal efficiency (R E) as a function of the S/L ratio; (b) amount of NH4+ adsorbed at equilibrium (q e) as a function of the S/L ratio.
Adsorption isotherms and fitting models
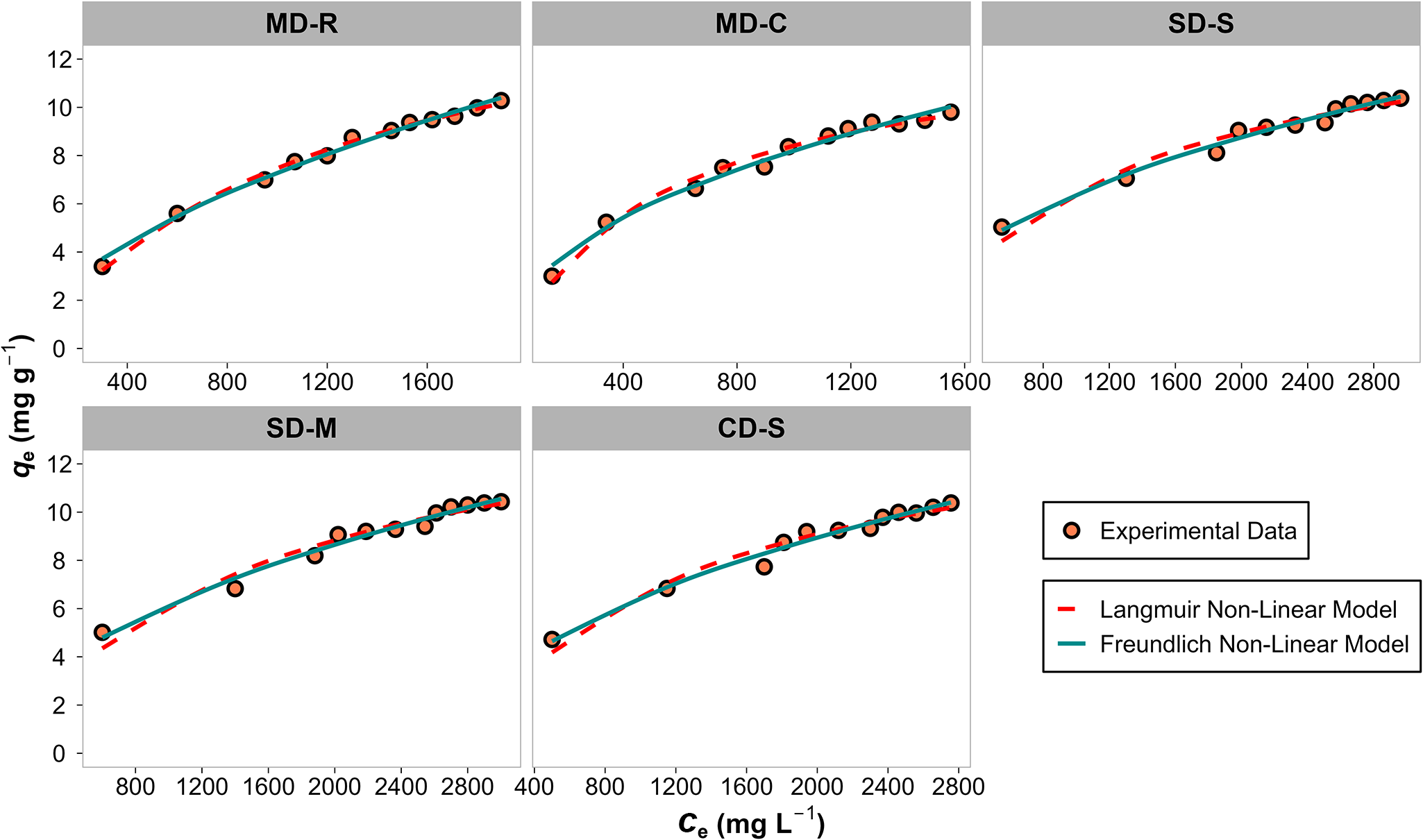
The adsorption equilibrium data were analysed using Langmuir and Freundlich isotherm models (Fig. 4), and the estimated parameters are listed on Table 5. Due to its intrinsic complexity, the adsorption process cannot be univocally defined as homogeneous or heterogeneous, nor exclusively attributed to physical or chemical interactions (Eberle et al., Reference Eberle, Börnick and Stolte2022). Most previous studies applied Langmuir and Freundlich isotherm models to describe the adsorption process (Lin et al., Reference Lin, Lei, Wang, Liu, Zhang and Wan2013; Taddeo et al., Reference Taddeo, Prajapati and Lepistö2017; Kotoulas et al., Reference Kotoulas, Agathou, Triantaphyllidou, Tatoulis, Akratos, Tekerlekopoulou and Vayenas2019; Eberle et al., Reference Eberle, Börnick and Stolte2022; Zhou et al., Reference Zhou, Wang, Wang, Xu and Zhu2024).

Figure 4. Amount of NH4+ adsorbed at equilibrium (q e) vs equilibrium concentration (C e) plot for the different digestates after a contact time of 24 h.
Table 5. Parameters calculated according to the Freundlich (Equation 3) and Langmuir isotherms (Equation 4) at 298.15 K (25°C).

Both the Freundlich and Langmuir models adequately describe the experimental data (c.f. R2, AIC and p-values; Table 5), indicating that NH4+ adsorption onto the zeolitic tuff occurs with both homogeneous and heterogeneous characteristics (Sánchez-Hernández et al., Reference Sánchez-Hernández, Padilla and López-Andrés2018). However, among the two, the Freundlich model generally yielded slightly better fits across most experimental conditions, highlighting the multilayer and heterogeneous nature of the adsorption process. This interpretation aligns with the kinetic studies, which also indicated a complex adsorption process. Further evidence supporting the heterogeneous nature of the process is provided by the Freundlich constant 1/n, which remained consistently below 1 across all tested digestates. Lower 1/n values indicate higher surface heterogeneity, consistent with adsorption occurring on sites with varying affinities (Haghseresht & Lu, Reference Haghseresht and Lu1998). In our study, 1/n values ranged between 0.4 and 0.6, reinforcing the suitability of the Freundlich model to describe the system. Additionally, the n values (commonly used to assess the favourability of adsorption) ranged between 2.04 and 2.28, indicating that the process was favourable in all cases (Wang & Guo, Reference Wang and Guo2020a). Although the differences in n values between digestates were not particularly pronounced, those derived from municipal solid waste (MD) exhibited the highest values, particularly for MD-C, suggesting a more favourable adsorption environment in this matrix. The Freundlich constant KF, which reflects the adsorption capacity, was also higher for MD-derived digestates, confirming their better performance. Moreover, the separation factor (R L) and Gibbs free energy change (ΔG°) further support this conclusion. The R L values ranged between 0.2 and 0.8 across all systems (Fig. 5), indicating favourable adsorption (Ivanova et al., Reference Ivanova, Karsheva and Koumanova2010). However, as the NH4+ concentration decreased (corresponding to near saturation of the zeolitic tuff), R L increased and approached 1, suggesting a gradual decline in adsorption favourability under these conditions. In terms of thermodynamic behaviour, ΔG° values (Table 6) were consistently negative, confirming the spontaneous nature of NH4+ adsorption on the zeolitic tuff. The most negative ΔG° values were observed for MD-C and MD-R, again pointing to the improved performance of MD-derived digestates. These results are in line with other studies using similar materials and experimental conditions (Galamini et al., Reference Galamini, Ferretti, Medoro, Tescaro and Faccini2020). Taken together, the combination of kinetic, isotherm and thermodynamic data indicates that NH4+ adsorption onto the zeolitic tuff is a spontaneous, favourable and surface-heterogeneous process that is particularly efficient in matrices derived from municipal organic waste.

Figure 5. Separation factor (R L) vs equilibrium concentration (C e) for the different digestates.
Table 6. Gibbs free energy (ΔG°; Equation 6) and the thermodynamic equilibrium constant (Ke) values determined at 298.15 K (25°C).

Effects of digestate composition and pre-treatment methods
One of the key elements influencing the efficiency of NH4+ adsorption onto the zeolitic tuff is the physical and chemical composition of the digestates, particularly their TS content and cation concentrations. A high TS content in the digestates can hinder adsorption by promoting pore blockage or competition for adsorption sites due to the presence of suspended organic and inorganic matter (Taddeo et al., Reference Taddeo, Prajapati and Lepistö2017; Eberle et al., Reference Eberle, Börnick and Stolte2022). A similar pattern was observed in this study (Fig. 6a): the digestate derived from MD-C, which exhibited the lowest TS content, showed the highest NH4+ removal, whereas the cattle-derived digestate CD-S, characterized by the highest TS concentration, resulted in the lowest NH4+ reduction. These findings are consistent with observations by Eberle et al. (Reference Eberle, Börnick and Stolte2022), who also reported that dissolved organic carbon (DOC) may interfere with NH4+ adsorption due to competitive occupation of active sites. Although DOC was not measured in this study, it is expected to be partly correlated with TS, especially in clarified digestate samples. However, the relationship is strongly influenced by the solid–liquid partitioning and specific organic matter composition.

Figure 6. Linear regression between the maximum NH4+ removal efficiency (R E) and (a) TS content and (b) potassium (K+) content for the different digestates. The equilibrium NH4+ R E was used as the response variable. In (b), the dashed grey regression line includes all five digestate types, whereas the solid black line excludes CD-S, which exhibits a distinct behaviour.
In addition to TS, the chemical composition of the digestate (especially the concentration of monovalent cations) plays a crucial role. Ions such as K+ and Na+, due to their similar size and charge to NH4+, can strongly compete for adsorption sites in zeolites – often more than divalent cations such as Mg2+ and Ca2+ (Farkaš et al., Reference Farkaš, Rožić and Barbarić-Mikočević2005; Huang et al., Reference Huang, Yang, Xue, Liu, Hou and Ding2015; Ferretti et al., Reference Ferretti, Galamini, Medoro, Coltorti, Di Giuseppe and Faccini2020). Potassium in particular has been identified as such a key competitor (Kong et al., Reference Kong, Liu, Vogt and Lee2016). When the K+ concentrations (Table 1) are compared across the digestates, an inverse relationship between initial K+ levels and NH4+-N removal efficiencies is observed for the MD and swine digestates (Fig. 6b), with lower K+ (MD-R, MD-C) corresponding to higher NH4+ removal and higher K+ (SD-S, SD-M) corresponding to lower removal, in agreement with Guo et al. (Reference Guo, Zeng, Li and Park2008). Interestingly, the cattle digestate (CD-S, dashed line in Fig. 6b) does not follow this trend: despite its greater K+ content, it exhibits greater NH4+ removal than the swine digestates. This unexpected behaviour may be influenced by other cations, such as the lower Na+ and Ca2+ concentrations in CD-S, which could modulate NH4+ adsorption.
Our findings are at odds with the classical expectation that a greater initial NH4+concentration increases adsorption by increasing the mass transfer driving force (Demir et al., Reference Demir, Günay and Debik2002). In this study, however, greater NH4+ reduction occurred in matrices with lower initial NH4+ concentrations – an effect also noted by Kotoulas et al. (Reference Kotoulas, Agathou, Triantaphyllidou, Tatoulis, Akratos, Tekerlekopoulou and Vayenas2019) and Taddeo et al. (Reference Taddeo, Prajapati and Lepistö2017). This deviation suggests that, in complex matrices such as digestates, other limiting factors such as TS content and ion competition may outweigh the driving force effect. Digestates with more favourable physical and chemical profiles, such as MD-derived ones, consistently demonstrated superior performance in terms of NH4+ reduction, despite their lower starting concentrations.
To mitigate the negative impact of solids and ionic competition, various pre-treatments could be applied, such as clarification, microfiltration and screw compression (Chini et al., Reference Chini, Hollas, Bolsan, Antes, Treichel and Kunz2021; Nowak & Czekała, Reference Nowak and Czekała2024). Pre-treated samples displayed lower 1/n values, more negative ΔG° values (Table 6) and R L values closer to zero (Fig. 5), suggesting the increased affinity, spontaneity and favourability of the NH4+ adsorption process. These outcomes were also reflected in faster equilibrium times and greater overall NH4+ reductions.
Such results align with similar previous studies (Proskynitopoulou et al., Reference Proskynitopoulou, Garagounis, Vourros, Dimopoulos Toursidis, Lorentzou and Zouboulis2024) that affirm that removing solids through centrifugation or filtration can improve accessibility to zeolite-exchange sites, thus improving performance. The greatest difference in NH4+ reduction (Fig. 3a) was observed between MD-R and MD-C, suggesting that performing a centrifugation step prior to NH4+ adsorption is the most effective strategy for improving NH4+ removal from a digestate. In contrast, the microfiltration step also increased NH4+ adsorption by the zeolitic tuff, but this improvement was less pronounced. From an applied perspective, integrating pre-treatment strategies into the treatment chain could significantly improve the efficiency of zeolite-based NH4+ recycling systems, particularly when dealing with livestock-derived effluents or complex digestates.
Effects of zeolite type and amount: comparative evaluation
To assess the role of zeolite mineralogy in NH4+ recovery from digestates, the performance of the chabazite-rich tuff tested in this study was directly compared with the data from Alberghini et al. (Reference Alberghini, Ferretti, Galamini, Botezatu and Faccini2025), who employed a zeolitic tuff composed of both phillipsite and chabazite. The comparison was carried out under identical experimental conditions, using the same digestates (MD, swine and cattle digestates), experimental setup and data analysis methods.
The kinetic analysis revealed substantial differences in the NH4+ uptake behaviour between the two zeolitic tuffs, attributable to their distinct mineralogical composition and cation-exchange properties. The NH4+ removal kinetics were well described by the PFO model, where the rate is initially fast and slows down as equilibrium is approached. This behaviour, which is common in zeolitic adsorbents, reflects processes often governed by surface diffusion or external mass transfer. In contrast, the chabazite–phillipsite-rich tuff showed a better fit with the PSO model, implying a more complex process probably involving multilayer adsorption or chemisorption interactions, consistent with a more heterogeneous surface environment (Halim et al., Reference Halim, Aziz, Johari and Ariffin2010; Shaban et al., Reference Shaban, AbuKhadra, Nasief and Abd El-Salam2017).
The faster initial uptake rates observed for the chabazite-rich tuff are indicative of a strong driving force for ion exchange in the early phase, followed by rapid attainment of equilibrium (typically within 90–180 min). In contrast, the chabazite–phillipsite-rich tuff generally reached equilibrium within 45–120 min, probably due to the enhanced surface heterogeneity and microporosity conferred by the dual-zeolite phases and greater glass content. This difference in equilibrium time was particularly marked in the MD-R digestate, where the chabazite–phillipsite-rich tuff achieved equilibrium significantly faster than the chabazite-rich tuff (Fig. 7a). Moreover, in the same digestate, the difference between the two materials was also more evident in terms of R E and q e (Fig. 7b,c). Nonetheless, the differences observed for these two parameters were not pronounced, highlighting that the chabazite-rich tuff still performed very well and represents a highly suitable option for NH4+ recovery from digestates.

Figure 7. Comparison between chabazite-rich and phillipsite–chabazite-rich tuffs in terms of (a) adsorption kinetics (qt vs time), (b) NH4+ removal efficiency (R E) as a function of S/L ratio and (c) amount of NH4+ adsorbed at equilibrium (q e) as a function of S/L ratio. Results refer to the MD-R digestate, which was selected as a representative case due to the more pronounced differences observed between the two materials in terms of adsorption kinetics, NH4+ removal and capacity. Data for the chabazite–phillipsite-rich tuff are from Alberghini et al. (Reference Alberghini, Ferretti, Galamini, Botezatu and Faccini2025).
The application of the ID model further supported a multi-step adsorption process in both materials, characterized by an initial external surface sorption phase, a gradual ID stage and final equilibration (Zhou et al., Reference Zhou, Wang, Wang, Xu and Zhu2024). However, the deviations from linearity and the absence of plot intercepts through the origin indicate that, although ID contributes to NH4+ uptake, it is not the sole rate-controlling process. This is especially evident for the chabazite–phillipsite-rich tuff, for which diffusion into micropores may be more influential due to the presence of a more heterogeneous pore structure.
These kinetic insights align well with the equilibrium results obtained from Freundlich isotherm modelling, which further emphasized differences in sorption performance between the two rocks. The chabazite-rich tuff generally exhibited lower KF values across all digestates, except for MD-C, where the values were similar (8.20 vs 8.47 mg g⁻1(L g⁻1)1/n). The highest KF value observed for the chabazite–phillipsite-rich tuff was 9.06 (MD-R), compared to 8.20 for the chabazite-rich tuff, confirming a slightly reduced adsorption capacity when phillipsite is absent. Despite these differences, the Freundlich n values remained within a comparable range (1.78–2.59), indicating similar adsorption intensities and surface heterogeneities between the two materials (Haghseresht & Lu, Reference Haghseresht and Lu1998).
Thermodynamic parameters further enriched the comparative analysis. In both materials, ΔG° values ranged from approximately –5000 to –7200 J mol⁻1, confirming the spontaneous nature of the process and its predominantly physical character (Jha & Hayashi, Reference Jha and Hayashi2009). Interestingly, the chabazite-rich tuff exhibited slightly more negative ΔG° values than the phillipsite–chabazite-rich tuff for certain digestates (e.g. MD-C: –7163 vs –7024 J mol⁻1; CD-S: –5509 vs –5075 J mol⁻1), suggesting a marginally more favourable energetic profile for NH4+ sorption on pure chabazite.
These results can be rationalized by considering the distinct physicochemical properties of the two tuffs. The chabazite–phillipsite-rich tuff had a slightly higher CEC (233 cmol kg⁻1) compared to the chabazite-rich tuff (217 cmol kg⁻1), which is consistent with the greater sorption capacity observed. Moreover, the mineralogical composition differed markedly: the former contained ∼20% chabazite, 10% phillipsite and 44% volcanic glass, whereas the latter consisted predominantly of chabazite (68.5%), with only 1.8% phillipsite and 11.0% volcanic glass. The greater glass content and dual-zeolite phase in the previous material probably increased surface heterogeneity and microporosity, promoting multilayer interactions and diffusion-limited uptake. Moreover, the presence of nanocrystalline domains within the amorphous phase (potentially of a zeolitic nature; Leggo et al., Reference Leggo, Cochemé, Demant and Lee2001; Holder & Schaak, Reference Holder and Schaak2019) could further contribute to NH4+ adsorption, thereby explaining the greater sorption capacity observed for this zeolitic tuff. Conversely, the chabazite-rich tuff, with its simpler phase composition and greater abundance of exchange sites per unit mass, favoured faster, surface-dominated ion exchange, but with a slightly reduced capacity.
This comparative evaluation underscores how mineralogy and CEC critically shape both the kinetics and thermodynamics of NH4+ adsorption. Although phillipsite improves overall sorption capacity and introduces more complex uptake mechanisms, chabazite alone provides an energetically favourable and kinetically efficient pathway for NH4+ removal from digestates. These findings are pivotal for guiding the selection and optimization of natural zeolitic materials in nutrient recovery applications.
Scalability assessment of nitrogen recovery from anaerobic digestate
Using the same calculation method described in our previous work (Alberghini et al., Reference Alberghini, Ferretti, Galamini, Botezatu and Faccini2025), the potential nitrogen recovery (see earlier) was estimated for each digestate matrix under realistic operational conditions at the farm scale. Assuming an average daily production of 10 m3 of digestate per plant with continuous operation throughout the year, the annual nitrogen recovery was calculated as a function of S/L ratio, up to a maximum of 15%, a value considered feasible for practical application at the farm level.
Figure 8 shows that the amount of nitrogen potentially recoverable on an annual basis varies significantly depending on the digestate type and S/L ratio. As expected, livestock-derived digestates (particularly SD-S and SD-M) showed greater recovery values due to their greater NH4+ concentrations and faster adsorption kinetics, which enable a higher number of treatment cycles per day. At an S/L ratio of 15%, the maximum nitrogen recovery reached values exceeding 3000 kg N year–1 per plant for these matrices.

Figure 8. Values of kg N recovered year–1 as a function of the S/L ratio of the zeolitic tuff used during the experiment.
Conversely, although municipal solid waste-derived digestates (MD-R and MD-C) allowed for a lower absolute nitrogen recovery (due to lower initial NH4+-N concentrations and slower kinetics), they showed higher percentages of NH4+ reduction during treatment, as discussed in previous sections. These matrices may therefore be more suitable in contexts in which the treatment objective is primarily the reduction of nitrogen load rather than its recovery.
Compared to the study with a chabazite–phillipsite-rich tuff (Alberghini et al., Reference Alberghini, Ferretti, Galamini, Botezatu and Faccini2025), the chabazite-rich tuff of the present study provided comparable or slightly lower nitrogen recovery yields across most conditions. For a consistent comparison, the previous data were recalculated to account for the contact time needed to reach equilibrium, as well as the additional time required for loading and unloading the pilot-scale prototype. Despite slightly lower total yields in some cases, the chabazite-rich tuff consistently achieved higher annual nitrogen recovery, particularly in more complex matrices such as swine and cattle digestates. This improved performance is probably related to its robustness under high organic loads and varying ionic strengths, suggesting that chabazite may be more adaptable under real operational conditions. A detailed comparison of these parameters is provided in Table S1.
Overall, even at relatively low S/L ratios, the annual nitrogen recovery potential remains relevant across all digestate types. This reinforces the potential of NH4+-enriched zeolitic tuff to be reused as a slow-release fertilizer in agriculture, partially replacing synthetic fertilizers such as urea. This substitution would align with the sustainability goals promoted by the European Green Deal by reducing both the environmental impacts and the energy demands associated with synthetic nitrogen fertilizer production (Di Giuseppe et al., Reference Di Giuseppe, Ferretti, Faccini, Blasi, Passeri, Bianchini and Coltorti2016; Ali et al., Reference Ali, Nakhli and Delkash2017; Eroglu et al., Reference Eroglu, Emekci and Athanassiou2017; Ghasemi et al., Reference Ghasemi, Sourinejad, Kazemian and Rohani2018; Mpanga et al., Reference Mpanga, Braun and Walworth2020; Cataldo et al., Reference Cataldo, Salvi, Paoli, Fucile, Masciandaro and Manzi2021; Jarosz et al., Reference Jarosz, Szerement, Gondek and Mierzwa-Hersztek2022; Galamini et al., Reference Galamini, Ferretti, Rosinger, Huber, Medoro and Mentler2023; Ferretti et al., Reference Ferretti, Rosinger, Diaz-Pines, Faccini, Coltorti and Keiblinger2024).
To validate the theoretical framework under practical conditions, a preliminary field application was performed in a test plant belonging to the project ‘STRUVITE’ (funded through the PR-FESR Emilia-Romagna 2021–2027 programme). The prototype machine could treat a maximum of 2 m3 per cycle. The tests were performed in May 2025 using microfiltered swine digestate (SD-M) at a zeolite S/L ratio of 3% (dry basis). The treatment was conducted in a discontinuous batch mode with three cycles per day, each processing 2 m3 of digestate (total 6 m3 day–1). Each cycle included ∼1.5 h of effective contact time between the zeolite and the digestate, plus an additional ∼20 min before and after the cycle for prototype loading and unloading. Each batch resulted in an average NH4+ removal of ∼10% of the initial concentration, corresponding to a zeolite loading of ∼11 g NH4+-N per kg of dry zeolite. This value is consistent with the results obtained in laboratory tests, confirming the reliability of the proposed batch setup and the practical relevance of the findings. During the treatment, the pH of the digestate increased slightly, from an average of 7.6 to 7.8 before and after contact with the zeolite, indicating a mild alkalinization of the system, in line with previous studies reporting similar effects of zeolite application on wastewater and digestate treatment (Ferretti et al., Reference Ferretti, Galamini, Medoro, Coltorti, Di Giuseppe and Faccini2020; Myrzalieva et al., Reference Myrzalieva, Putra Pratama and Khamidulla2021; Teymouri et al., Reference Teymouri, Wong, Tan and Mohd Pauzi2024). Based on this configuration, the estimated annual nitrogen recovery was ∼715 kg N year–1. Future scale-up is planned in terms of using a continuous-flow system, which is expected to significantly increase the daily throughput and improve the overall nitrogen recovery efficiency.
Conclusions
This study highlights the potential of a chabazite-rich tuff as a sustainable and efficient adsorbent for NH4+ recovery from various anaerobic digestates, with direct implications for farm-scale applications. The results confirm that the chabazite-rich zeolitic tuff is a promising material for ammonium recovery from various digestates. Adsorption followed a multilayer and heterogeneous process (Freundlich isotherm), with its kinetics best described by the PFO model. Digestate composition (especially K+ levels and TS content) significantly influenced NH4+ uptake. Livestock-derived digestates ensured the greatest recovery, while solid–liquid separation (particularly centrifugation) improved performance by increasing site accessibility. A preliminary field-scale trial with microfiltered swine digestate demonstrated process feasibility, with a potential recovery of up to ∼715 kg N year–1 per plant, supporting its integration into circular nutrient management strategies. Compared to previous findings involving a zeolitic tuff containing both phillipsite and chabazite, the chabazite-rich tuff used in this study showed distinct adsorption behaviour under the same operating conditions. These differences, related to mineralogical composition and CEC, will be the basis for future comparative studies aiming to optimize zeolite selection based on kinetic performance, adsorption capacity and process simplicity. It is considered that by enhancing our understanding of the process through exploring the behaviour of other commercially available zeolite tuffs (e.g. clinoptilolite-rich tuffs), it will be possible to increase the applicability of this technology under diverse operational scenarios. Overall, these findings highlight the potential of zeolite-based systems for circular nitrogen management in agriculture, enabling the recovery of valuable nutrients from organic wastes while reducing reliance on synthetic fertilizers, in line with European Green Deal objectives and supporting more resilient and sustainable food systems. The NH4+-saturated tuff produced through this process can be applied directly as a slow-release soil amendment, and although the chabazite-rich tuff studied here showed comparable nitrogen recovery to the chabazite–phillipsite-rich tuff, it stands out for its consistently robust and adaptable NH4+ removal performance, even in complex matrices.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1180/clm.2026.10025.
Author contributions
Conceptualization: Matteo Alberghini, Giacomo Ferretti; Methodology: Matteo Alberghini, Giacomo Ferretti; Investigation: Matteo Alberghini, Giacomo Ferretti; Writing – Original draft: Matteo Alberghini; Writing – Review and editing: Matteo Alberghini, Giacomo Ferretti; Supervision: Matteo Alberghini, Giacomo Ferretti; Formal analysis: Matteo Alberghini, Cristina Botezatu; Data curation: Matteo Alberghini, Giacomo Ferretti; Project administration: Giacomo Ferretti, Barbara Faccini; Data analysis: Matteo Alberghini, Giacomo Ferretti; Validation: Giacomo Ferretti; Resources: Giacomo Ferretti, Barbara Faccini. All authors have read and agreed to the published version of the manuscript.
Acknowledgements
We acknowledge the support of CRPA (Emilia-Romagna) and WAM Group for their technical assistance. We also extend our gratitude to Biorg and Colombaro for supplying the various digestate samples utilized in this research. We further thank Verdi S.p.A. for providing the zeolitic tuff used in this study. We gratefully acknowledge Renzo Tassinari for performing the ICP-MS analyses.
Financial support
This work was supported by the project ‘STRUVITE – Trattamento dei digestati per ridurre le emissioni e recuperare un fertilizzante, la Struvite’, funded through the PR-FESR Emilia-Romagna 2021–2027 programme.
Competing interests
The authors declare none.
Data availability
All relevant data for this study are available in the manuscript and accompanying Supplementary Material.