Introduction
The prevalence of colorectal cancer (CRC) is alarming, being the main cause of morbidity and mortality in oncology (International Agency for Research on Cancer [GLOBOCAN] 2020) and constituting a relevant public health problem (García-Espinosa et al. Reference García-Espinosa, García-Villacampa and Pérez-González2020). In its evolution, CRC can be asymptomatic, but once the disease is advanced, the symptoms may vary depending on the location of the tumor (Cubiella et al. Reference Cubiella, Marzo-Castillejo and Mascort-Roca2018).
Quality of life (QoL) is an important indicator in the evaluation of health outcomes in cancer patients, being a multidimensional construct that includes physical, social, and psychological domains, as well as symptoms associated with the disease and its treatment (Aaronson Reference Aaronson1987; Moinpour et al. Reference Moinpour, Feigl and Metch1989; Testa and Simonson Reference Testa and Simonson1996). Since a large number of patients with CRC experience a variety of side effects and symptoms related to the disease and its treatment, their QoL may be affected at different points in cancer care (Kristensen et al. Reference Kristensen, Thyø and Christensen2019; Lapinsky et al. Reference Lapinsky, Man and MacKenzie2019).
Among the most common physical side effects in CRC patients are diarrhea and chronic pain, urinary incontinence, fatigue, and sexual difficulties (El‐Shami et al. Reference El‐Shami, Oeffinger and Erb2015; Simard et al. Reference Simard, Kamath and Kircher2019; Yde et al. Reference Yde, Larsen and Laurberg2018). These symptoms can persist for years after diagnosis, as in the case of intestinal problems (Jansen et al. Reference Jansen, Koch and Brenner2010; Ramsey et al. Reference Ramsey, Berry and Moinpour2002). In addition, psychosocial effects such as anxiety and depression (Mitchell et al. Reference Mitchell, Ferguson and Gill2013; Peng et al. Reference Peng, Huang and Kao2019), emotional distress (Dunn et al. Reference Dunn, Ng and Holland2013; El‐Shami et al. Reference El‐Shami, Oeffinger and Erb2015), risk of suicide (Lloyd et al. Reference Lloyd, Baraghoshi and Tao2019), and sexual dysfunction have been reported (El‐Shami et al. Reference El‐Shami, Oeffinger and Erb2015).
Therefore, having an instrument to assess HRQoL specifically for patients with CRC is essential in the clinical, research, and institutional setting, as it is an outcome measure based on the patient’s perception of the impact of the disease on their life. To assess the QoL of cancer patients, the European Organization for Research and Treatment of Cancer (EORTC) has developed measurement instruments. In the specific case of CRC, the EORTC group developed a specific module to assess QoL in this neoplasm; the EORTC QLQ-CR29 (Whistance et al. Reference Whistance, Conroy and Chie2009). The advantage of this instrument is that it contains a large number of specific items and subscales to measure side effects, symptoms, and key functional areas in this cancer group, as well as a specific section for patients with or without stoma. This increases the content validity of the EORTC QLQ-CR29 in comparison with other scales.
The evaluation of QoL in patients with CRC is fundamental in the improvement of medical and psychological care. It is not only a relevant indicator for predicting morbidity and mortality, but it is also a significant parameter in therapeutic decision-making and an important outcome measure in the long term (Fallowfield Reference Fallowfield2002; Kristensen et al. Reference Kristensen, Thyø and Christensen2019; Mansano-Schlosser and Ceolim Reference Mansano-Schlosser and Ceolim2012). In this sense, its evaluation becomes more relevant in the context of Latin America because it is predicted that the increase in its incidence rate will not cease, mainly due to the standard of living of people (a more sedentary lifestyle and unhealthy eating habits), as well as the aging of its population (Torres-Román et al. Reference Torres-Román, Toro-Huamanchumo and Grados-Sánchez2015). Additionally, many factors stand in the way of early diagnosis and timely treatment of CRC in Latin America, including a lack of specialists and insufficient health system infrastructure. The current infrastructure, in many areas of Latin America, often lacks the capacity to provide screening, chemotherapy, and surgery for all patients in a timely manner (Pharma et al. Reference Pharma, Brown and Mellon2014; Torres-Román et al. Reference Torres-Román, Toro-Huamanchumo and Grados-Sánchez2015).
In Mexico there is no valid, reliable, and psychometrically adequate instrument to measure this construct, which makes it difficult to understand the impact of CRC on the lives of patients and, therefore, make informed decisions for their treatment. Therefore, the purpose of this study was to validate the EORTC QLQ-CR29 questionnaire in the Mexican cancer population.
Method
Participants
The present study had a sample obtained by availability in the functional unit of the Instituto Nacional de Cancerología (INCan) Gastroenterology service, during the period from May 9 to August 3, 2022. An instrumental, nonexperimental, cross-sectional design was used (Montero and León Reference Montero and León2005). The inclusion criteria established for participation in the research were as follows:
- Inclusion criteria: Have a confirmed diagnosis of CRC, present any clinical stage, be in active treatment or under follow-up, and have a Karnofsky score ≥70.
- Exclusion criteria: Exclusion criteria were established for those participants with visual or hearing impairment that prevented them from responding to the surveys, patients with a second primary cancer, and patients with severe physical problems (such as nausea, vomiting, dyspnea, pain, and fatigue) that prevented them from participating.
- Elimination criteria: Likewise, elimination criteria were established for those participants who, during the process of filling out the questionnaires, decided not to continue participating in the study.
Ethical aspects
A request was made to the EORTC group for permission to use the QLQ-C30 and CR29 instruments in the study. The protocol was approved by Mexico’s Instituto Nacional de Cancerología Research and Ethics Committees with approval number: (022/003/OMI) (CEI/1600/21). This study conforms to the standards of the Declaration of Helsinki. All participants who agreed to participate in the study did so after having read and understood the information provided in the informed consent, and voluntarily signed it.
Instruments
• Health-related Quality of Life Scale EORTC QLQ-30. Designed by Aaronson et al. (Reference Aaronson, Ahmedzai and Bergman1993) is used to measure the global QoL of patients through 30 items distributed in 3 dimensions: functional, which includes physical, role, cognitive, emotional, and social functioning; symptoms, including fatigue, pain, nausea, and vomiting; and overall QoL. Cronbach’s alpha coefficient ranges from α = .52 to α = .89, with a Likert-type scale from 1 to 4 and 2 items from 1 to 7. It has been validated in the Mexican cancer population by Onate-Ocana et al. (Reference Onate-Ocana, Alcantara-Pilar and Vilar-Compte2009).
• EORTC QLQ-CR29 questionnaire. This tool is designed to measure health-related QoL in patients with CRC, considering the symptoms and functional areas that are associated with this type of cancer and its treatment. This scale includes 29 items that are divided into 4 subscales: bowel function (α = .70), urinary frequency (α = .75), body image (α = .84), and blood and mucus in stool (α = .69). The alpha coefficient of these subscales ranges between .69 and .84, which indicates adequate internal consistency. In addition, the questionnaire has separate sections for patients with and without stoma and individual questions that assess functional areas, such as sexual desire in men and women. The scale uses a Likert-type score from 1 to 4, and the questions related to symptoms refer to the previous week, with the exception of the question on sexuality, which addresses the last 4 weeks. The instrument has demonstrated adequate test–retest reliability with r values >.68 and has been validated for use in the Mexican cancer population (Whistance et al. Reference Whistance, Conroy and Chie2009).
Procedure
Cultural adaptation
Initially, an evaluation of the EORCT CR29 (version 2.1) in the Spanish language of Spain, provided by the EORCT group, was carried out by 11 oncology experts belonging to the disciplines of psychology and nursing, to receive feedback and recommendations on wording and language appropriate to the target population. Changes were made to the instrument to improve comprehension of the instructions and questions.
Then, the questionnaire was adapted to Mexican Spanish through a pilot test carried out in 30 patients with CRC from the functional unit of the INCan Gastroenterology service. A structured interview was used to identify possible confusing or offensive expressions, and to assess understanding of the questions, instructions, and response options (Mora-Rios et al. Reference Mora-Rios, Bautista-Aguilar and Natera2013). Therefore, the modifications made consisted of substituting terms that are more common in the Spanish of the Mexican population. For example, the elimination of the word “barriga,” leaving only “estomago,” the substitution of the expression “hinchazón” for “inflamación.” In addition, a definition of colostomy and ileostomy was added at the end of the instrument, since they are technical terms that are sometimes not understood by patients. Finally, the final version of the EORCT CR29 was obtained after receiving few suggestions from patients.
Statistical analysis
Data analysis was carried out using the statistical program SPSS version 26. Since the EORTC QLQ-CR29 comprises 4 scales composed mostly of 2 items, a principal component factor analysis with Varimax rotation was performed and adjusted to 4 factors, corresponding to the 4 functional subscales of the original scale. The 17 items that were answered by all the participants (items 31–47) were considered, regardless of whether they had a stoma or not and of their gender, with the purpose of determining the configuration of the items in the proposed scales. For this analysis, the following criteria were used: (a) factor loadings ≥.40; (b) a minimum number of items per-factor was not established; and (c) internal consistency coefficients for each Cronbach’s alpha factor ≥.50. The relationship between the EORTC QLQ-CR29 instrument and the concurrent measurements with the EORTC QLQ-C30 was evaluated using the Pearson correlation coefficient. Likewise, internal consistency was obtained using Cronbach’s alpha and the Omega coefficient.
We proceeded to assess the fit of the 4-factor model by means of a confirmatory factor analysis using the maximum likelihood method (Abd-El-Fattah Reference Abd-El-Fattah2010; George and Mallery Reference George and Mallery2019; Kline Reference Kline2015): The analysis included the identification and specification of the model, the estimation of standardized parameters, such as R2 correlations, covariances, modification indices, and critical proportions of the differences, followed by the evaluation of the fit by observing the acceptable limits of the estimators. Various fit indices were estimated, including: the X2 index, the X2/df ratio, the goodness-of-fit index (GFI), the Tucker–Lewis index (TLI), and the corrected goodness-of-fit index (AGFI), as well as the comparative goodness-of-fit index (CFI), (Ullman Reference Ullman2006) which is considered the best indicator for samples equal to or greater than 200. Finally, the root mean square of the approximation error (RMSEA) was also calculated.
Results
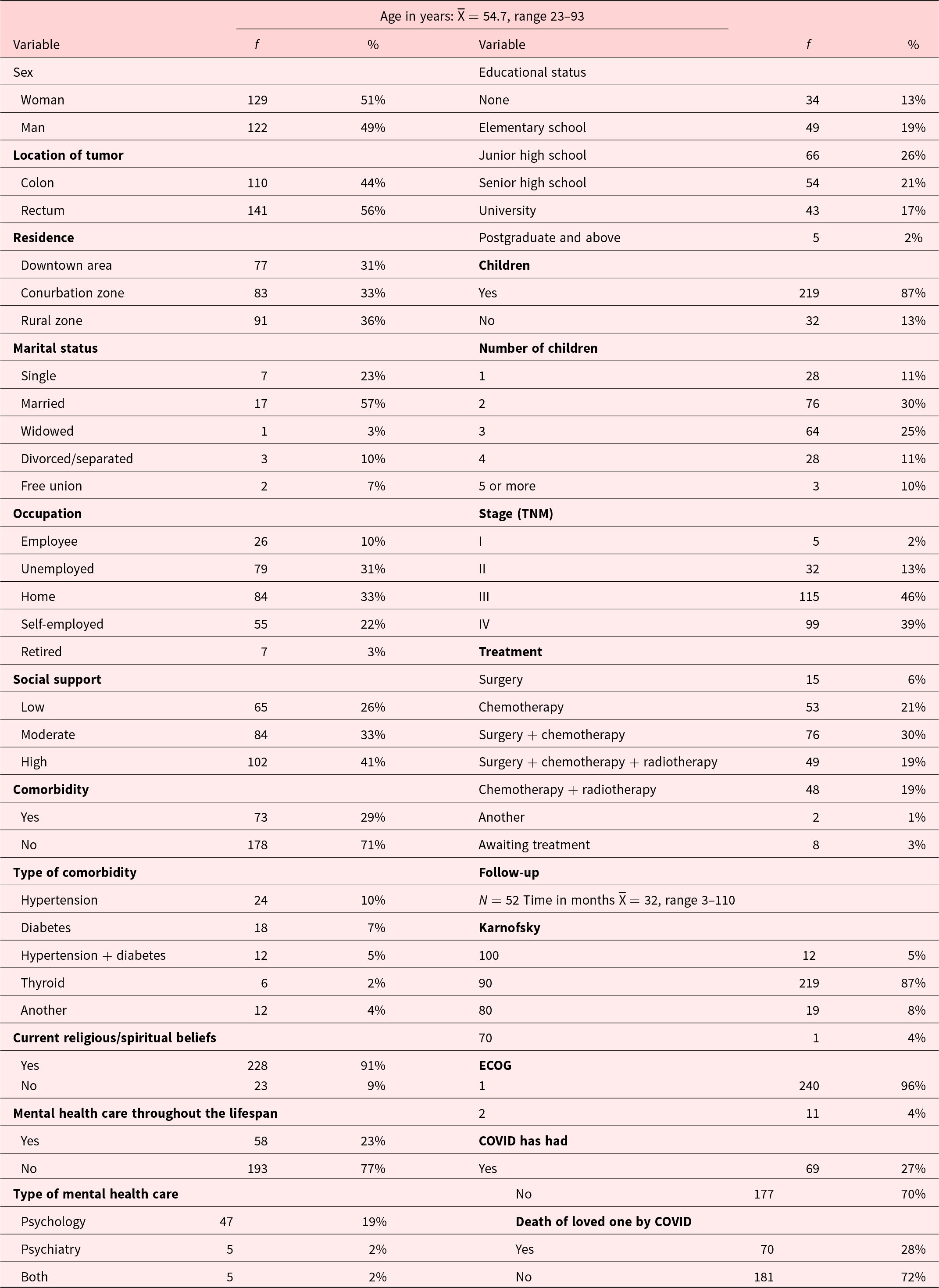
A nonrandom sample composed of 251 individuals, of both genders and with a diagnosis of CRC, whose average age was 54.7 ± 12.28 years (see Table 1), was used.
Clinical and sociodemographic characteristics of a sample of 251 participants with CRC

Factor structure
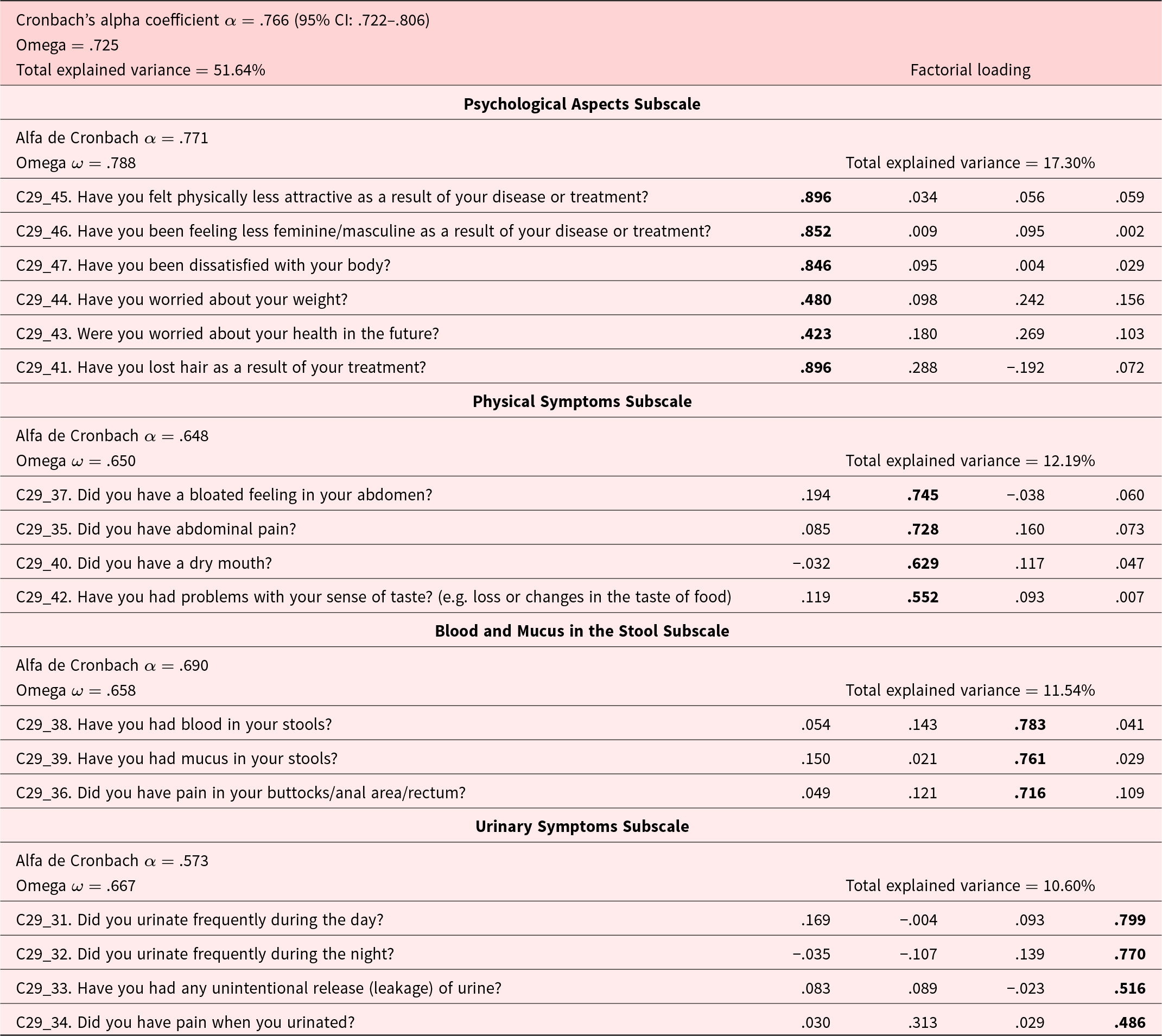
The sample used in the analysis was considered adequate, according to the value of the Kaiser–Meyer–Olkin test, which was .732 (p = .001). An exploratory factorial analysis was performed using the principal components method, and varimax rotation and adjusted to 4 factors with the 17 items. An explained variance of 51.64% was found, and 4 factors were identified and named: Psychological affectations, Physical symptoms, Blood and mucus in the stool, and Urinary symptoms, as seen in Table 2.
Psychometric properties and exploratory factor analysis of the EORTC QLQ-CR29

Internal consistency
An internal consistency analysis of the EORTC QLQ-CR29 instrument was performed, obtaining a Cronbach’s alpha coefficient of .766 (95% CI: .722–.806) and an Omega coefficient of ω = .788, for the 17 items of the test questionnaire. Likewise, alpha coefficients of α = .573 to .771 were obtained in the 4 factors identified in the factor analysis, with Omega coefficients of ω = .650–.788.
External validity
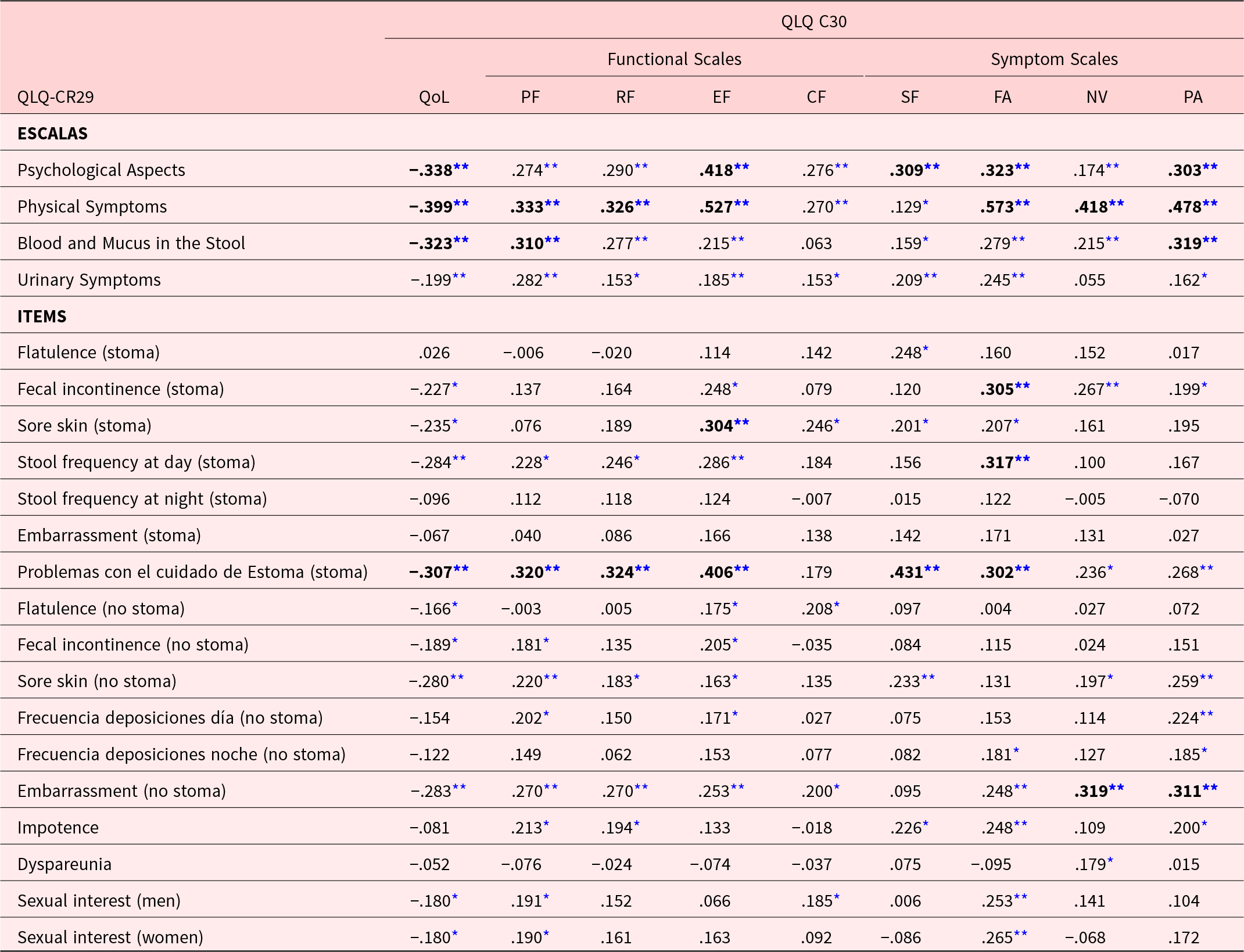
Regarding the correlations between the EORTC QLQ-CR29 and the EORTC QLQ-C30, it was observed that in most cases these were low (r < .40), however, in some areas with more related contents, higher correlations were found to be high (r ≥ .40) (see Table 3). In relation to the 4 subscales of the EORTC QLQ-CR29, statistically significant negative correlations were found with the overall QoL of the EORTC QLQ-C30 (Pearson’s r from −.199 to −.399, p < .01). Table 3 is a breakdown of the highest correlations identified in each subscale.
Correlations between the EORTC QLQ-CR29 and EORTC QLQ-C30 instruments

* Correlation is significant at the .05 level. **Correlation is significant at the .01 level.
Confirmatory factor analysis
The relative comparative goodness-of-fit indices (CFI, TLI, and AGFI) are significantly close to the ideal value of .95, confirming the acceptability of the model compared to the null. The PCFI value, which is close to 1 and is greater than .5, indicates that the proposed model is more parsimonious and efficient than the null model (Hu and Bentler Reference Hu and Bentler1998).
Furthermore, the root mean square residual (RMR) index is close to 0 and less than .06 (RMR = .044), confirming that there is virtually no difference between the observed and the predicted covariance matrix, suggesting that the discrepancy between the proposed model and the actual data is almost nonexistent or very low (Abd-El-Fattah Reference Abd-El-Fattah2010).
In general, the adjusted chi-square value at 111 degrees of freedom was much less than 3 (ideally less than 3) (chi-square = 1.410), with a p-value less than .05, confirming an excellent absolute fit of the model to the observed data (Abd-El-Fattah Reference Abd-El-Fattah2010). In addition, a value close to 0 is observed in the RMSEA index (RMSEA = .040 [.024–.055]), which indicates that the model has almost 0 error (Abd-El-Fattah Reference Abd-El-Fattah2010). These results are summarized in Figure 1 and Table 4of the final model.
Four-factor first-order confirmatory factor analysis model with 17 items of the EORTC QLQ-CR29.

Indices obtained from confirmatory factor analysis confirmatory factor analysis model with 17 items of the EORTC QLQ-CR29

Discussion
The incidence of cancer is not distributed evenly throughout the world, as infection-related malignancies are gradually being replaced by those related to rapid social, economic, and demographic changes. These changes are due to the adoption of lifestyles and behaviors commonly associated with Westernization (Fidler et al. Reference Fidler, Bray and Vaccarella2017), which is reflected in the increase in CRC, which is one of the clearest markers of this oncological transition (Fidler et al. Reference Fidler, Bray and Vaccarella2017).
In Latin America and the Caribbean, CRC occupies third place with the highest incidence and is expected to continue to increase in the future (GLOBOCAN, 2020). This increase is partly due to the adoption of unhealthy lifestyles, characterized by dietary patterns low in fiber and high in animal fats, red and processed meats, high alcohol consumption, smoking, and physical inactivity (Figuero et al. Reference Figuero, Tocino and Fonseca2021; Granados and Herrera Reference Granados and Herrera2016; Kolligs Reference Kolligs2016). In 2017, approximately two thirds of the population of Mexico (more than 50 million people) were overweight or obese, and in 2018 approximately 10,000 cases of colon cancer were diagnosed, of which approximately 15% could be attributed to a high body mass index (Soerjomataram and Bray Reference Soerjomataram and Bray2021).
Due to the increase in the incidence of CRC, the health system must be resized to face this challenge in the future (Álvarez-Escobar 2019). The evaluation of the QoL of patients with CRC is essential to know their perspective on the impact of the disease and the treatment on their lives, which helps to make therapeutic decisions and becomes a primary outcome measure.
The original QLQ-CR29 incorporates 4 multi-thematic scales and 19 individual items that assess the range of symptoms and common problems in patients with CRC. However, some previous validations have not included exploratory factor analysis (EFA) (Arraras et al. Reference Arraras, Suárez and de la Vega2011; El Alami et al. Reference El Alami, Bachri and Essangri2020; Ihn et al. Reference Ihn, Lee and Son2015; Lin et al. Reference Lin, Zhang and Wu2017; Magaji et al. Reference Magaji, Moy and Roslani2016; Montazeri et al. Reference Montazeri, Emami and Sadighi2017; Nowak et al. Reference Nowak, Tobiasz-Adamczyk and Brzyski2011; Shen et al. Reference Shen, Chen and Ho2018; Wickramasinghe et al. Reference Wickramasinghe, Dayasena and Seneviratne2020). In this sense, the German validation (Stiggelbout et al. Reference Stiggelbout, Kunneman and Baas-Thijssen2016) has identified 7 factors through an EFA, although some of them were not theoretically interpretable.
On the other hand, in the present validation in the Mexican population, an EFA and a confirmatory factor analysis were performed, which allowed grouping a greater number of items in the 4 factors of the QLQ-CR29. In the Psychological Aspects subscale, image and concern for weight/health were grouped; in the Physical Symptoms subscale, pain, abdominal swelling, and taste disturbances were grouped together; in the Urinary Symptoms subscale, urinary frequency, painful urination, and urinary incontinence were grouped; and in the subscale Blood and mucus in the stool, pain in the rectum was incorporated. This grouping of items decreases the number of unique items, improves the reliability of the scale, and provides evidence of the validity of the structure by integrating the items in a theoretically interpretable manner.
The QLQ-CR29 questionnaire was shown to have acceptable psychometric properties, similar to previous validations carried out in other studies (Arraras et al. Reference Arraras, Suárez and de la Vega2011; El Alami et al. Reference El Alami, Bachri and Essangri2020; Ihn et al. Reference Ihn, Lee and Son2015; Stiggelbout et al. Reference Stiggelbout, Kunneman and Baas-Thijssen2016). The results of the confirmatory factor analysis suggest that the proposed theoretical model adjusted almost perfectly to the data of the sample used in this research and the structural indicators (CFI, RMR, and RMSEA) indicate that it is a balanced model with minimal error that measures in a pertinent way the QoL of patients with CRC.
Small modifications were made in the wording of the questionnaire to make it more easily understandable for the Mexican population, because the way of asking questions and the language used are sources of bias, as well as cultural factors that lead to the same question being valid or not in one language or another, or even in different countries that share the same language (Carvajal et al. Reference Carvajal, Centeno and Watson2011). These modifications focused on improving the acceptance of the questionnaire by the participants, reducing attrition and minimizing the amount of data lost.
The EORTC QLQ-CR29 stands out from other QoL assessment instruments in oncology due to its specific focus on patients with CRC, covering relevant aspects such as intestinal problems, urinary symptoms, and sexual difficulties. Although there is an alternative to the Functional Assessment of Cancer Therapy-Colorectal (FACT-C) scale (Ward et al. Reference Ward, Hahn and Mo1999), the latter has fewer items and specific subscales to measure side effects, symptoms, and key functional areas in this cancer group, in addition to lacking a specific section for patients with or without stoma. Therefore, the EORTC QLQ-CR29 has superior content validity compared to the FACT-C.
In summary, the EORTC QLQ-CR29 is recommended for the evaluation of the QoL of Mexican cancer patients with CRC in different settings, including clinical, research, and institutional care. The use of this instrument allows reliable and valid results, to be obtained and allows the comparison of results at the national and international levels.
In future studies, it is suggested to evaluate the stability of the instrument over time, analyzing whether the instrument is sensitive to the effects of medical and/or psychosocial interventions, as well as conduct additional research to explore the impact of QoL in patients with CRC in relation to other constructs such as overall disease survival, symptomatology emotional and therapeutic adherence.
Acknowledgments
Jazmín Hernández-Marín is a doctoral student from Programa de Doctorado en Psicología y Salud, Universidad Nacional Autónoma de México (UNAM) and received fellowship 1084046 from CONAHCYT.
Funding
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
The author(s) declare none.