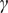Introduction
Bipolar disorder (BD) is a chronic mental illness characterized by recurrent periods of mania and depression (BDI) or hypomania and depression (BDII) (Phillips & Kupfer, Reference Phillips and Kupfer2013). The depressive episodes in BDII are more persistent and lead to a higher burden of depression over time compared to BDI, because hypomania is less severe than mania but is easier to overlook (Baek et al., Reference Baek, Park, Choi, Kim, Choi, Ha and Hong2011). Genetic (Lee et al., Reference Lee, Chen, Chen, Huang, Tzeng, Chang and Lu2011), metabolic (Nikolaus, Müller, & Hautzel, Reference Nikolaus, Müller and Hautzel2017), and neuroimaging (Abé et al., Reference Abé, Ekman, Sellgren, Petrovic, Ingvar and Landén2016; Caseras et al., Reference Caseras, Lawrence, Murphy, Wise and Phillips2013) investigations have demonstrated that BDI and BDII differ in their pathophysiological and neurobiological mechanisms. Moreover, inflammation in BD has been shown to be associated with cognitive dysfunction (Barbosa et al., Reference Barbosa, Ferreira, Rocha, Mol, da Mata Chiaccjio Leite, Bauer and Teixeira2018; Dickerson et al., Reference Dickerson, Stallings, Origoni, Vaughan, Khushalani and Yolken2013), abnormal mood states (mania and depression) (Fiedorowicz et al., Reference Fiedorowicz, Prossin, Johnson, Christensen, Magnotta and Wemmie2015), illness activity (Kapczinski et al., Reference Kapczinski, Dal-Pizzol, Teixeira, Magalhaes, Kauer-Sant’Anna, Klamt and Post2011), and different clinical stages (Tatay-Manteiga et al., Reference Tatay-Manteiga, Balanzá-Martínez, Bristot, Tabarés-Seisdedos, Kapczinski and Cauli2017). Given these findings, it is essential to further explore the role of inflammation in BDII.
Systemic inflammation and immune dysregulation have been considered to play a significant role in psychiatric disorders such as BD, major depressive disorder (MDD), schizophrenia, and obsessive-compulsive disorder (Najjar et al., Reference Najjar, Pearlman, Alper, Najjar and Devinsky2013). Specifically, BD has been reported to be a disease associated with chronic inflammatory and immunological alterations mediated by cytokines’ cascades, cellular immune responses, oxidative factors, and hormone regulation (Altamura, Buoli, & Pozzoli, Reference Altamura, Buoli and Pozzoli2014). Cytokines could access the brain and influence many neurobiological processes known to be involved in depression, including neurotransmitter metabolism, neuroendocrine function, and neural plasticity (Miller, Maletic, & Raison, Reference Miller, Maletic and Raison2009), facilitating peripheral-to-central immune crosstalk (Walker et al., Reference Walker, Le Page, Terrando, Duggan, Heneka and Bettcher2023). Cytokines can enter the central nervous system and trigger neuroinflammation through increased blood–brain barrier (BBB) permeability (Hu et al., Reference Hu, Hu, He, Liu, Gui, Zhu and Xu2024) and can also alter cerebrospinal fluid (CSF) composition by affecting the choroid plexus (Balusu et al., Reference Balusu, Van Wonterghem, De Rycke, Raemdonck, Stremersch, Gevaert and Vandenbroucke2016), with previous studies identifying correlations between choroid plexus volume and peripheral cytokine levels in BDII-D (Cao et al., Reference Cao, Lizano, Deng, Sun, Zhou, Xie and Jia2023). Previous review and meta-analyses have reported elevated levels of inflammatory cytokines such as interleukin-4 (IL-4), interleukin-6 (IL-6), interleukin-8 (IL-8), interleukin-10 (IL-10), and tumor necrosis factor-α (TNF-α) in BD (Goldsmith, Rapaport, & Miller, Reference Goldsmith, Rapaport and Miller2016; Modabbernia, Taslimi, Brietzke, & Ashrafi, Reference Modabbernia, Taslimi, Brietzke and Ashrafi2013; Munkholm, Braüner, Kessing, & Vinberg, Reference Munkholm, Braüner, Kessing and Vinberg2013; Solmi et al., Reference Solmi, Suresh Sharma, Osimo, Fornaro, Bortolato, Croatto and Carvalho2021; Tsai, Reference Tsai2021), and cytokine profiles might differ across mood states and BD subtypes (Long et al., Reference Long, Li, Ding, Mei and Li2024; Munkholm et al., Reference Munkholm, Braüner, Kessing and Vinberg2013). Moreover, cytokines have been robustly associated with psychiatric symptoms and regional brain abnormalities, including functional connectivity disruptions (Chen et al., Reference Chen, Chen, Chen, Zhong, Gong, Zhong and Wang2020 ; Tang et al., Reference Tang, Chen, Chen, Zhong, Gong, Zhong and Huang2021; Xiao et al., Reference Xiao, Cao, Lizano, Li, Sun, Zhou and Walter2024), structural gray matter loss (Cao et al., Reference Cao, Sun, Lizano, Deng, Zhou, Xie and Jia2024), and reduced white matter (WM) integrity (Cao et al., Reference Cao, Lizano, Li, Chand, Sun, Zhou and Jia2025) in BDII-D. In our previous study, the disruption of regional FC was inversely correlated with IL-8 level in BD II (Tang et al., Reference Tang, Chen, Chen, Zhong, Gong, Zhong and Huang2021). However, the relationships between cytokine levels and whole-brain functional abnormalities remain unclear.
In recent years, there has been growing interest in understanding the complex relationships between inflammation and brain function, especially in the context of bipolar disorder. Traditional functional connectivity (FC) analysis has provided valuable insights into these relationships; however, it may not fully capture the intricate topological organization of the brain’s functional networks (Gong & He, Reference Gong and He2015). Studying the topological organization can provide additional information on how different brain regions interact and coordinate their activities, which is crucial for understanding the pathophysiology of bipolar disorder. Graph theory-based analysis has been applied to resting-state functional magnetic resonance imaging (rs-fMRI) to study the features of complex brain networks (Bullmore & Sporns, Reference Bullmore and Sporns2009; Liao, Vasilakos, & He, Reference Liao, Vasilakos and He2017). In terms of graph theory, the human brain’s functional networks can be defined as a complex dynamic system consisting of functionally connected nodes (brain regions) and edges (functional connections between these brain regions) (van den Heuvel & Hulshoff Pol, Reference van den Heuvel and Hulshoff Pol2010). This method derives various parameters that reflect the topological properties of the brain’s functional networks, such as clustering coefficient, characteristic path length, small-worldness, global efficiency, and local efficiency, at the global level, and nodal efficiency, at the nodal level (Bullmore & Sporns, Reference Bullmore and Sporns2012; Gozdas, Holland, & Altaye, Reference Gozdas, Holland and Altaye2019; Jiang et al., Reference Jiang, Yao, Zhou, Tan, Huang, Wang and Luo2020; van den Heuvel & Hulshoff Pol, Reference van den Heuvel and Hulshoff Pol2010; Zhou et al., Reference Zhou, Womer, Kong, Wu, Jiang, Zhou and Wang2017). For example, global efficiency reflects the overall efficiency of information transfer in the brain network, and nodal efficiency reflects the efficiency of information transfer in specific brain regions (Bassett et al., Reference Bassett, Bullmore, Meyer-Lindenberg, Apud, Weinberger and Coppola2009). These parameters provide a more comprehensive understanding of the brain’s functional organization and its relationship with inflammation in BD. Previous studies using graph theoretical brain network analysis have reported topological alterations of functional networks in BD (Kim et al., Reference Kim, Bolbecker, Howell, Rass, Sporns, Hetrick and O’Donnell2013; Wang et al., Reference Wang, Wang, Jia, Zhong, Zhong, Sun and Huang2017; Xia et al., Reference Xia, Womer, Chang, Zhu, Zhou, Edmiston and Wang2019). For example, at the global level, BD II individuals showed increased characteristic path length and decreased global efficiency compared to the controls (Wang et al., Reference Wang, Wang, Jia, Zhong, Zhong, Sun and Huang2017); at the nodal level, BD exhibited significant abnormalities and decreased nodal efficiency in the left ventral pallidum (Zhang et al., Reference Zhang, Qiao, Wang, Yang, Wang, Sun and Zhang2023). In MDD, significant correlations were observed between clinical symptoms as well as plasma IL-6 levels with whole-brain functional network connectivity (Liu et al., Reference Liu, Jing, Gao, Li, Qin, Xie and Li2024), suggesting the potential role of whole-brain functional network disruption in the pathophysiological mechanisms of depression.
Existing studies have primarily focused on either the investigation of brain network alterations in BD (Kim et al., Reference Kim, Bolbecker, Howell, Rass, Sporns, Hetrick and O’Donnell2013; Wang et al., Reference Wang, Wang, Jia, Zhong, Zhong, Sun and Huang2017; Xia et al., Reference Xia, Womer, Chang, Zhu, Zhou, Edmiston and Wang2019), or on the exploration of peripheral inflammation in the context of BD using traditional methods without specifically linking it to the brain network topology (Chen et al., Reference Chen, Chen, Chen, Zhong, Gong, Zhong and Wang2020 ; Tang et al., Reference Tang, Chen, Chen, Zhong, Gong, Zhong and Huang2021; Xiao et al., Reference Xiao, Cao, Lizano, Li, Sun, Zhou and Walter2024). To date, the link between peripheral inflammation and network-level dysfunction in BD remains unclear, particularly regarding whether alterations in inflammatory cytokines are correlated with critical brain network properties from topological analysis. In this study, we hypothesized that unmedicated BDII-D individuals would show abnormal global parameters such as greater characteristic path length, lower clustering coefficient, lower global efficiency, and decreased nodal efficiency compared to healthy controls (HCs). We also expected greater levels of inflammatory cytokines in unmedicated BDII-D. Therefore, we assumed that the abnormal global and nodal parameters would show correlation with peripheral cytokine levels (such as IL-8) and clinical variables in unmedicated BDII-D. By exploring these associations, this study aims to elucidate the potential interplay between peripheral inflammation and brain network disruptions in BDII-D, thereby contributing to a more comprehensive understanding of the pathophysiology of this disorder.
Materials and methods
Participants
Sixty-five unmedicated BDII-D individuals were recruited from the psychiatry department of the First Affiliated Hospital of Jinan University, Guangzhou, China. The BDII-D individuals were aged from 18 to 55 years old, to reduce the potential influence of age-related vascular lesions and late life multimorbidity (Ben Hassen et al., Reference Ben Hassen, Fayosse, Landré, Raggi, Bloomberg, Sabia and Singh-Manoux2022). Participants were recruited between March 2018 and June 2022. The BDII-D participants were diagnosed based on the Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition) (DSM-5). The enrolled participants were diagnosed by two experienced clinical psychiatrists (Y.J. and S.Z., with 27 and 10 years of experience in clinical psychiatry, respectively) based on the Structured Clinical Interview for DSM-IV Patient Edition (SCID-P). During a 3-day period before the neuroimaging acquisition, the 24-item Hamilton Depression Rating Scale (HDRS) and the Young Mania Rating Scale (YMRS) were used to evaluate the clinical state of BD individuals. Individuals who met the conditions of the 24-item HDRS total score more than 20 and YMRS less than 7 were recruited. Exclusion criteria were as follows: (i) currently suffering from any psychiatric disorder or associated symptoms; (ii) history of any organic or neurological brain disorder; (iii) history of any alcohol/substance abuse or dependence; (iv) history of use of any psychotropic medication, psychotherapy, or electroconvulsive therapy; (v) any physical illness demonstrated by clinical or laboratory examinations, or personal history; and (vi) pregnancy or postpartum depression, and any contraindication to magnetic resonance imaging (MRI) scanning. All the individuals have not received medication for at least 6 months at the time of the neuroimaging acquisition.
Fifty right-handed healthy volunteers were recruited in this study through local advertisements at the same period of time. All the HCs were chosen after a diagnostic interview (the SCID Nonpatient Edition) to eliminate the possibility of current or past history of psychiatric illness. Moreover, HCs with the following situations would be excluded from this study: history of psychiatric illness in first-degree relatives, or current or past significant medical or neurological illness.
This study was approved by the Ethics Committee of First Affiliated Hospital of Jinan University, China. All the participants signed informed consent forms after a full written and verbal explanation of the study.
MR imaging data acquisitions
Neuroimaging data were acquired on GE Discovery MR750 3.0 T machine with the eight-channel phased-array head coil. All the subjects were scanned in supine, head-first positions, with cushions placed symmetrically on both sides of their heads to reduce movement. The subjects were told to relax and not close their eyes or fall asleep during the scan. At the end of the experiment, it was confirmed that none of the participants had fallen asleep. The rs-fMRI data were obtained by gradient-echo echo planar imaging sequence, and its parameters were as follows: time repetition (TR)/time echo (TE) = 2000/25 ms; field of view (FOV) = 240 × 240 mm2; flip angle = 90°; voxel size = 3.75 × 3.75 × 3 mm3; slice thickness/gap = 3.0/1.0 mm; matrix = 64 × 64; 35 axial slices covering the whole brain; and 210 volumes acquired in 7 min. Additionally, whole-brain, three-dimensional brain volume imaging (3D-BRAVO) sequence was used to collect brain structural data, and the parameters were as follows: TR/ TE = 8.2/ 3.2 ms; bandwidth = 31.25 Hz; flip angle = 12°; NEX = 1; slice thickness/gap = 1.0/0 mm; FOV = 240 × 240 mm2; matrix = 256 × 256; and acquisition time = 3 min 45 s. Also, conventional MRI data of all the participants were acquired to confirm the absence of any anatomic abnormalities of their brains, which were determined by two experienced neuroradiologists (G.C. and G.T.).
Functional data preprocessing
The preprocessing was carried out using Statistical Parametric Mapping (SPM 12, http://www.fil.ion.ucl.ac.uk/spm/) and Data Processing & Analysis of Brain Imaging (DPABI_V3.0, http://restfmri.net/forum/DPABI) (Yan, Wang, Zuo, & Zang, Reference Yan, Wang, Zuo and Zang2016). For each subject, the first 10 volumes of the rs-fMRI dataset were removed; the remaining 200 volumes were slice timing corrected; and all images were realigned to the first image for head motion correction. Individuals who had more than 2 mm maximum displacement, 2°of angular motion, and 0.2 mm in mean frame-wise displacement were excluded (Jenkinson, Bannister, Brady, & Smith, Reference Jenkinson, Bannister, Brady and Smith2002). Several spurious covariates (global signal, white matter, cerebrospinal fluid signals, and Friston-24 parameters) were removed from the data. The Diffeomorphic Anatomical Registration Through Exponentiated Lie algebra (Shen & Sterr, Reference Shen and Sterr2013) segment toolbox was utilized to create templates for spatial normalization to the Montreal Neurological Institute (MNI) space with 3 × 3 × 3 mm3 voxels, which was followed by signal linear trend removing and temporal band-pass filtering (0.01–0.1 Hz).
Constructing brain functional networks
The brain functional network of each subject was constructed using the GRETNA toolbox (http://www.nitrc.org/projects/gretna/) (Wang et al., Reference Wang, Wang, Xia, Liao, Evans and He2015) according to the automated anatomical labeling (AAL) template (Tzourio-Mazoyer et al., Reference Tzourio-Mazoyer, Landeau, Papathanassiou, Crivello, Etard, Delcroix and Joliot2002). First, the AAL template was parcellated into 116 brain regions as the regions of interest (ROIs); each ROI was defined as a node of the network. The time series for each ROI was calculated by extracting and averaging the time series of all the voxels within the ROI. Second, to determine the edges of brain networks, for each subject, Pearson’s correlation coefficients were computed between the given ROI and the other 115 ROIs, and we repeated this process for all given ROIs. Then, a 116 by 116 symmetric partial correlation matrix (R) for each subject was obtained. Of note, in the matrix, because of detrimental effects on test–retest reliability and ambiguous interpretation (Fox, Zhang, Snyder, & Raichle, Reference Fox, Zhang, Snyder and Raichle2009; Murphy et al., Reference Murphy, Birn, Handwerker, Jones and Bandettini2009; Weissenbacher et al., Reference Weissenbacher, Kasess, Gerstl, Lanzenberger, Moser and Windischberger2009), negative correlations were excluded. Finally, binary graphs were obtained from the sparsity threshold from 0.1 to 0.4 with intervals of 0.01, which was used in subsequent analyses. Sparsity value was equal to the ratio of the actual total number of edges to the maximum possible number of edges.
Small-world network properties
At each sparsity threshold, we calculated global and nodal network properties. Several global network properties involving the small-world parameters and network efficiency were computed. The small-world parameters included normalized clustering coefficient
![]() $ \gamma $
, normalized characteristic path length
$ \gamma $
, normalized characteristic path length
![]() $ \lambda $
, and small-worldness
$ \lambda $
, and small-worldness
![]() $ \sigma $
(the ratio of
$ \sigma $
(the ratio of
![]() $ \gamma\;\mathrm{to}\;\lambda $
). The network efficiency indexes included global efficiency E
glob and local efficiency E
loc. We also calculated nodal efficiency E
nodal, one of the parameters of nodal properties. Detailed interpretations of these properties are shown in the previous study (Rubinov & Sporns, Reference Rubinov and Sporns2010).
$ \gamma\;\mathrm{to}\;\lambda $
). The network efficiency indexes included global efficiency E
glob and local efficiency E
loc. We also calculated nodal efficiency E
nodal, one of the parameters of nodal properties. Detailed interpretations of these properties are shown in the previous study (Rubinov & Sporns, Reference Rubinov and Sporns2010).
Thus, we applied five global parameters (
![]() $ \gamma $
,
$ \gamma $
,
![]() $ \lambda $
,
$ \lambda $
,
![]() $ \sigma, $
E
glo, and E
loc) and one regional nodal parameter (E
nodal) to characterize functional network properties. For each network index, we further calculate the area under the curve (AUC) to obtain the summary scalar of the brain function network topology representation (Zhang et al., Reference Zhang, Wang, Wu, Kuang, Huang, He and Gong2011).
$ \sigma, $
E
glo, and E
loc) and one regional nodal parameter (E
nodal) to characterize functional network properties. For each network index, we further calculate the area under the curve (AUC) to obtain the summary scalar of the brain function network topology representation (Zhang et al., Reference Zhang, Wang, Wu, Kuang, Huang, He and Gong2011).
Inflammatory cytokine measures
Peripheral blood samples from 34 unmedicated BDII-D individuals and 30 HCs were collected in fasting states in the morning (all the participants without alcoholic beverages intake for at least 1 day before testing) and were processed (then frozen) by technicians. Details on processing blood samples can be found in the Supplementary Materials. Serum levels of 17 inflammatory cytokines, including interleukin 1β (IL-1β), interleukin 2 (IL-2), IL-4, interleukin 5 (IL-5), IL-6, interleukin 7 (IL-7), IL-8, IL-10, interleukin 12 (IL-12), interleukin 13 (IL-13), interleukin 17 (IL-17), granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), interferon-γ (IFN-γ), monocyte chemoattractant protein-1/monocyte chemotactic and activating factor (MCP-1/MCAF), macrophage inflammatory protein 1β (MIP-1β), and TNF-α were measured using the Bio-Plex Human Cytokine 17-Plex panel in combination with the Bio-Plex Suspension Array System (Bio-Rad Laboratories Inc., Hercules, CA, USA) (Gauglitz et al., Reference Gauglitz, Finnerty, Herndon, Mlcak and Jeschke2008). Bio-Plex Manager Software, version 6.1, was used for data acquisition (Tang et al., Reference Tang, Chen, Chen, Zhong, Gong, Zhong and Huang2021).
Statistical analysis
Comparison of demographic variables
Independent sample t-tests and chi-square tests were used to compare the demographic data between the BDII-D and HC groups using SPSS 19.0 software (SPSS, Chicago, IL, USA). All tests were two-tailed, and p < 0.05 was considered statistically significant.
Comparison of network parameters and cytokine levels
The Kolmogorov–Smirnov tests were used to determine whether network parameters including global network topological parameters (
![]() $ \gamma $
,
$ \gamma $
,
![]() $ \lambda $
,
$ \lambda $
,
![]() $ \sigma, $
E
glo, and E
loc), nodal parameter (E
nodal), and cytokine levels including IL-1β, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12, IL-13, IL-17, G-CSF, GM-CSF, IFN-γ, MCP-1, MIP-1β, and TNF-α fit within normal distributions. If the network parameters and cytokine levels exhibited normal distributions, the data would be expressed as the mean and standard deviation, and independent sample t-tests were used to compare data between two groups. If a normal distribution was not observed, the median values and interquartile range would be reported, and subsequent comparisons of network parameters and cytokine levels between groups were performed using the Mann–Whitney U-tests. To stabilize variance and normalize the distribution, all cytokine levels were normalized (base 10 log-transformed) before analysis. False discovery rate (FDR) was applied for multiple comparison corrections, and p < 0.05 after FDR correction was considered statistically significant.
$ \sigma, $
E
glo, and E
loc), nodal parameter (E
nodal), and cytokine levels including IL-1β, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12, IL-13, IL-17, G-CSF, GM-CSF, IFN-γ, MCP-1, MIP-1β, and TNF-α fit within normal distributions. If the network parameters and cytokine levels exhibited normal distributions, the data would be expressed as the mean and standard deviation, and independent sample t-tests were used to compare data between two groups. If a normal distribution was not observed, the median values and interquartile range would be reported, and subsequent comparisons of network parameters and cytokine levels between groups were performed using the Mann–Whitney U-tests. To stabilize variance and normalize the distribution, all cytokine levels were normalized (base 10 log-transformed) before analysis. False discovery rate (FDR) was applied for multiple comparison corrections, and p < 0.05 after FDR correction was considered statistically significant.
Correlations between the network properties, inflammatory cytokines, and clinical variables
Partial Pearson correlation analyses (controlled for age, gender, and education) were performed to explore the relationships between network properties and log-transformed inflammatory cytokine levels that showed significant group differences after FDR corrections and their relationships with clinical variables in BDII-D. The clinical variables included the 24-item HDRS score, the YMRS score, onset age of illness, number of episodes, and durations of illness.
Validation analyses
The complementary utility of binary and weighted functional network (Cole, Pathak, & Schneider, Reference Cole, Pathak and Schneider2010) has been reported previously, as binary networks show strengths of reduction of the computational complexity and clearness of network metric definitions, while connectivity strength is taken into account in weighted networks (Wang et al., Reference Wang, Wang, Xia, Liao, Evans and He2015). To determine whether the observed group differences in the topological parameters reflect true differences rather than artifacts, we repeated the network analyses using weighted graphs from the sparsity threshold from 0.1 to 0.4 with intervals of 0.01 to validate the main results obtained from binary graphs. Furthermore, we repeated the correlation analyses between weighted functional network topological parameters, clinical variables, and inflammatory cytokine levels to validate the correlation results of the main findings.
Results
Demographic and clinical characteristics
Demographic and clinical characteristics of all the participants are displayed in Table 1. No significant differences were found in age, sex, and education levels between the BDII-D and HC groups.
Demographic data and clinical variables of the two groups

Note: Means (with standard deviations in parentheses) are reported unless otherwise noted.
Abbreviations: BDII-D, bipolar II depression; HCs, healthy controls; HDRS, Hamilton Depression Rating Scale; YMRS, Young Mania Rating Scale.
a The p values were obtained by independent sample t-tests.
b The p value for gender distribution was obtained by the chi-square test.
Global and nodal parameters of the brain functional network
Figure 1 and Table 2 show the results of global parameters (normalized clustering coefficient
![]() $ \gamma $
, normalized characteristic path length
$ \gamma $
, normalized characteristic path length
![]() $ \lambda $
, small-worldness
$ \lambda $
, small-worldness
![]() $ \sigma $
, global efficiency E
glob, and local efficiency E
loc) for the BDII-D and HC groups. Statistical analyses revealed that the BDII-D group showed significantly higher
$ \sigma $
, global efficiency E
glob, and local efficiency E
loc) for the BDII-D and HC groups. Statistical analyses revealed that the BDII-D group showed significantly higher
![]() $ \lambda $
(z = −4.028, p < 0.001, FDR-p < 0.001) and significantly decreased
$ \lambda $
(z = −4.028, p < 0.001, FDR-p < 0.001) and significantly decreased
![]() $ \gamma $
(t = −3.285, p = 0.001, FDR-p = 0.001),
$ \gamma $
(t = −3.285, p = 0.001, FDR-p = 0.001),
![]() $ \sigma $
(t = −3.721, p < 0.001, FDR-p < 0.001), Eglo (z = −3.769, p < 0.001, FDR-p < 0.001), and Eloc (t = −1.993, p = 0.049, FDR-p = 0.049) compared to HCs.
$ \sigma $
(t = −3.721, p < 0.001, FDR-p < 0.001), Eglo (z = −3.769, p < 0.001, FDR-p < 0.001), and Eloc (t = −1.993, p = 0.049, FDR-p = 0.049) compared to HCs.
Group differences of the inflammatory cytokine levels and the global topological parameters between the BDII-D and HCs group. (A) The BDII-D group showed significantly higher levels of IL-8, IL-10, G-CSF, MCP-1 and MIP-1β compared to HCs (FDR corrected, p < 0.05). (B) The BDII-D group showed significantly higher λ, and significantly decreased γ, σ, Eglo, and Eloc compared to HCs (FDR corrected, p < 0.05). BDII-D, bipolar II depression; HCs, healthy controls; FDR, false discovery rate; IL-8, interleukin 8; IL-10, interleukin 10; G-CSF, granulocyte colony-stimulating factor; MCP-1, monocyte chemoattractant protein-1; MIP-1β, macrophage inflammatory protein 1β; γ, normalized clustering coefficient; λ, normalized characteristic path length; σ, small-worldness; Eglo, global efficiency; Eloc, local efficiency. Log, base 10 log-transformed. *: p < 0.05. **: p < 0.01. ***: p < 0.001.

Statistical results for the global and nodal parameters between the BDII-D group and the HC group

Note: Means (with standard deviations in parentheses) and median [25% quartile-75% quartile] are reported unless otherwise noted. BDII-D, bipolar II depression; HCs, healthy controls; FDR, false discovery rate;
![]() $ \lambda $
, normalized characteristic path length;
$ \lambda $
, normalized characteristic path length;
![]() $ \gamma $
, normalized clustering coefficient; σ, small-worldness; Eglo, global efficiency; Eloc, local efficiency; R, right; L, left.
$ \gamma $
, normalized clustering coefficient; σ, small-worldness; Eglo, global efficiency; Eloc, local efficiency; R, right; L, left.
a The p values were obtained by independent sample t-tests.
b The p values were obtained by Mann–Whitney U tests.
Figure 2 and Table 2 show the brain regions of significantly different nodal parameters (E nodal) between the BDII-D group and the HC group. We found decreased Enodal in the right olfactory cortex (z = −3.921, p < 0.001, FDR-p < 0.001), left pallidum (t = −3.628, p < 0.001, FDR-p < 0.001), and vermis (z = −3.523, p < 0.001, FDR-p < 0.001) in the BDII-D group when compared with the HC group.
Results of between group differences of the nodal parameter (Enodal) for the BDII-D and HCs group. The BDII-D patients showed significantly decreased Enodal in the right olfactory cortex, left pallidum and the vermis (FDR corrected, p < 0.05). BDII-D, bipolar II depression; HCs, healthy controls; FDR, false discovery rate; Enodal, nodal efficiency; L, left; R, right. ***: p < 0.001.

Inflammatory cytokine levels between two groups
The results of comparison of log-transformed cytokine levels are shown in Figure 1 and Table 3. The BDII-D group showed higher levels of IL-8 (z = −3.220, p = 0.001, FDR-p = 0.009), IL-10 (z = −3.698, p = <0.001, FDR-p = <0.001), G-CSF (z = −2.664, p = 0.008, FDR-p = 0.031), MCP-1 (t = −2.689, p = 0.009, FDR-p = 0.031), and MIP-1β (t = −2.773, p = 0.008, FDR-p = 0.031) compared with the HC group.
Group differences of 17 cytokine levels between the BDII-D and HC groups

Note: Means (with standard deviations in parentheses) and median [25% quartile, 75% quartile] are reported unless otherwise noted. BDII-D, bipolar II depression; HCs, healthy controls; FDR, false discovery rate; IL-1β, interleukin 1β; IL-2, interleukin 2; IL-4, interleukin 4; IL-5, interleukin 5; IL-6, interleukin 6; IL-7, interleukin 7; IL-8, interleukin 8; IL-10, interleukin 10; IL-12, interleukin 12; IL-13, interleukin 13; IL-17, interleukin 17; G- CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; IFN-γ, interferon-γ; MCP-1(MCAF), monocyte chemoattractant protein-1 (monocyte chemotactic and activating factor); MIP-1β, macrophage inflammatory protein 1β; and TNF-α, tumor necrosis factor alpha. Bold entries indicate statistical significance at p < 0.05 after FDR correction for multiple comparisons.
a The p values were obtained by Mann–Whitney U tests. Log, base 10 log-transformed.
b The p values were obtained by independent sample t-tests.
Correlation analyses
For the correlations between abnormal global parameters and clinical variables,
![]() $ \gamma $
(r = −0.425, p = 0.043) and
$ \gamma $
(r = −0.425, p = 0.043) and
![]() $ \sigma $
(r = −0.424, p = 0.044) were negatively correlated with the 24-item HDRS scores. Besides,
$ \sigma $
(r = −0.424, p = 0.044) were negatively correlated with the 24-item HDRS scores. Besides,
![]() $ \gamma $
(r = −0.464, p = 0.030) and
$ \gamma $
(r = −0.464, p = 0.030) and
![]() $ \sigma $
(r = −0.449, p = 0.036) were negatively correlated with number of episodes in BDII-D (Figure 3A). The non-significant results are shown in Table S1 of the Supplementary Materials.
$ \sigma $
(r = −0.449, p = 0.036) were negatively correlated with number of episodes in BDII-D (Figure 3A). The non-significant results are shown in Table S1 of the Supplementary Materials.
Correlation results between clinical variables, functional network topology and inflammatory cytokine levels. (A) The correlations between abnormal global parameters and 24-item HDRS scores and number of episodes in BDII-D. (B) The correlations between abnormal global and nodal parameters and log IL-8 (base 10 log-transformed) in BDII-D. BDII-D, bipolar II depression; γ, normalized clustering coefficient; σ, small-worldness; Enodal, nodal efficiency; L, left; HDRS, Hamilton Depression Rating Scale.

For the correlations between abnormal inflammatory cytokine levels (log-transformed) and functional network topology in BDII-D, IL-8 levels were negatively correlated with global parameters including
![]() $ \gamma $
(r = −0.387, p = 0.031) and
$ \gamma $
(r = −0.387, p = 0.031) and
![]() $ \sigma $
(r = −0.392, p = 0.029), and negatively correlated with Enodal of the left pallidum (r = −0.379, p = 0.036) in BDII-D (Figure 3B). The remaining correlation results in the BDII-D and HC groups are shown in Tables S2–S3 of the Supplementary Materials.
$ \sigma $
(r = −0.392, p = 0.029), and negatively correlated with Enodal of the left pallidum (r = −0.379, p = 0.036) in BDII-D (Figure 3B). The remaining correlation results in the BDII-D and HC groups are shown in Tables S2–S3 of the Supplementary Materials.
Validation results
Table S4 of the Supplementary Materials lists the statistical results for the global parameters of weighted network between the BDII-D group and the HC group. The BDII-D group showed significantly decreased
![]() $ \gamma $
(t = −3.266, p = 0.001, FDR-p = 0.004) and
$ \gamma $
(t = −3.266, p = 0.001, FDR-p = 0.004) and
![]() $ \sigma $
(t = −3.756, p < 0.001, FDR-p < 0.001) compared to HCs. These results indicate that the results of global parameters including normalized clustering coefficient
$ \sigma $
(t = −3.756, p < 0.001, FDR-p < 0.001) compared to HCs. These results indicate that the results of global parameters including normalized clustering coefficient
![]() $ \gamma $
and small-worldness
$ \gamma $
and small-worldness
![]() $ \sigma $
reported above were relatively reliable across the different network type strategies.
$ \sigma $
reported above were relatively reliable across the different network type strategies.
Figure S1 of the Supplementary Materials shows the robustness of the correlation results between clinical variables, weighted functional network topological parameters, and inflammatory cytokine levels. The correlations between abnormal global parameters including
![]() $ \gamma $
(r = −0.426, p = 0.034) and
$ \gamma $
(r = −0.426, p = 0.034) and
![]() $ \sigma $
(r = −0.483, p = 0.017) and the 24-item HDRS scores in BDII-D, the correlations between abnormal global parameters including
$ \sigma $
(r = −0.483, p = 0.017) and the 24-item HDRS scores in BDII-D, the correlations between abnormal global parameters including
![]() $ \gamma $
(r = −0.399, p = 0.048) and
$ \gamma $
(r = −0.399, p = 0.048) and
![]() $ \sigma $
(r = −0.423, p = 0.040) and number of episodes in BDII-D, and the correlations between abnormal global parameters including
$ \sigma $
(r = −0.423, p = 0.040) and number of episodes in BDII-D, and the correlations between abnormal global parameters including
![]() $ \gamma $
(r = −0.377 p = 0.037) and
$ \gamma $
(r = −0.377 p = 0.037) and
![]() $ \sigma $
(r = −0.409, p = 0.022) and IL-8 level in BDII-D still exist in the validation analyses.
$ \sigma $
(r = −0.409, p = 0.022) and IL-8 level in BDII-D still exist in the validation analyses.
Discussion
The main findings of the present study are as follows: (1) altered global parameters including greater normalized characteristic path length λ, lower normalized clustering coefficient
![]() $ \gamma $
, small-worldnes
$ \gamma $
, small-worldnes
![]() $ \sigma $
, global efficiency E
glob, and local efficiency E
loc in unmedicated BDII-D; (2) decreased nodal efficiency E
nodal in the right olfactory cortex, left pallidum, and vermis in unmedicated BDII-D; (3) elevated levels of IL-8, IL-10, and G-CSF in unmedicated BDII-D; (4) elevated IL-8 level was negatively correlated with decreased
$ \sigma $
, global efficiency E
glob, and local efficiency E
loc in unmedicated BDII-D; (2) decreased nodal efficiency E
nodal in the right olfactory cortex, left pallidum, and vermis in unmedicated BDII-D; (3) elevated levels of IL-8, IL-10, and G-CSF in unmedicated BDII-D; (4) elevated IL-8 level was negatively correlated with decreased
![]() $ \gamma $
and
$ \gamma $
and
![]() $ \sigma $
, and Enodal of the left pallidum in BDII-D; and (5) decreased
$ \sigma $
, and Enodal of the left pallidum in BDII-D; and (5) decreased
![]() $ \gamma $
and
$ \gamma $
and
![]() $ \sigma $
were correlated with higher HDRS scores in BD. This study is among the first to investigate the relationship between functional network topology and inflammation markers in unmedicated BDII-D individuals. Notably, the highly homogeneous samples included in this study are unmedicated individuals with BDII-D, eliminating confounding factors such as medication, BD subtypes, and mania or hypomania state, which may provide more reliable evidence of the neuropathological mechanisms behind depressive symptoms of BD individuals from the perspective of inflammation.
$ \sigma $
were correlated with higher HDRS scores in BD. This study is among the first to investigate the relationship between functional network topology and inflammation markers in unmedicated BDII-D individuals. Notably, the highly homogeneous samples included in this study are unmedicated individuals with BDII-D, eliminating confounding factors such as medication, BD subtypes, and mania or hypomania state, which may provide more reliable evidence of the neuropathological mechanisms behind depressive symptoms of BD individuals from the perspective of inflammation.
In this study, the whole-brain functional networks in BDII-D and HC groups showed small-world properties that were consistent with previous findings (Bassett & Bullmore, Reference Bassett and Bullmore2006; Suo et al., Reference Suo, Lei, Li, Li, Dai, Wang and Gong2018). Additionally, we found significant decreased
![]() $ \gamma $
,
$ \gamma $
,
![]() $ \sigma $
, E
glo, and E
loc, and significant increased λ in BD, which partly validate the topological disruption among BDII-D in our previous study (Wang et al., Reference Wang, Wang, Jia, Zhong, Zhong, Sun and Huang2017) with a larger sample size and different templates for constructing brain network, and further suggest its correlation with inflammation. Global efficiency E
glo quantifies parallel information transfer capacity across distributed networks, while local efficiency E
loc reflects specialized processing within modular subsystems (Bullmore & Sporns, Reference Bullmore and Sporns2009). Characteristic path length λ inversely correlates with integration efficiency, with higher values indicating suboptimal long-range communication. Small-world coefficients (σ = γ/λ) balance local segregation and global integration – a configuration evolutionarily optimized for cognitive flexibility (Bassett & Sporns, Reference Bassett and Sporns2017). The observed changes in network parameters collectively suggest that the brain’s functional organization in BD is characterized by impaired segregation and integration (Yoon, Kim, Kim, & Lyoo, Reference Yoon, Kim, Kim and Lyoo2021), which is supported by some previous graph theoretical studies using rs-fMRI in BD (Spielberg et al., Reference Spielberg, Beall, Hulvershorn, Altinay, Karne and Anand2016; Wang et al., Reference Wang, Wang, Jia, Zhong, Zhong, Sun and Huang2017; Yu et al., Reference Yu, Qin, Xiong, Xu, Wang, Hou and Yang2020). These functional network topological abnormalities might lead to impaired cognitive integration, disrupted local processing, and reduced network flexibility, which in turn contribute to emotional dysregulation, executive dysfunction, and cognitive rigidity in BD. Notably, we found decreased
$ \sigma $
, E
glo, and E
loc, and significant increased λ in BD, which partly validate the topological disruption among BDII-D in our previous study (Wang et al., Reference Wang, Wang, Jia, Zhong, Zhong, Sun and Huang2017) with a larger sample size and different templates for constructing brain network, and further suggest its correlation with inflammation. Global efficiency E
glo quantifies parallel information transfer capacity across distributed networks, while local efficiency E
loc reflects specialized processing within modular subsystems (Bullmore & Sporns, Reference Bullmore and Sporns2009). Characteristic path length λ inversely correlates with integration efficiency, with higher values indicating suboptimal long-range communication. Small-world coefficients (σ = γ/λ) balance local segregation and global integration – a configuration evolutionarily optimized for cognitive flexibility (Bassett & Sporns, Reference Bassett and Sporns2017). The observed changes in network parameters collectively suggest that the brain’s functional organization in BD is characterized by impaired segregation and integration (Yoon, Kim, Kim, & Lyoo, Reference Yoon, Kim, Kim and Lyoo2021), which is supported by some previous graph theoretical studies using rs-fMRI in BD (Spielberg et al., Reference Spielberg, Beall, Hulvershorn, Altinay, Karne and Anand2016; Wang et al., Reference Wang, Wang, Jia, Zhong, Zhong, Sun and Huang2017; Yu et al., Reference Yu, Qin, Xiong, Xu, Wang, Hou and Yang2020). These functional network topological abnormalities might lead to impaired cognitive integration, disrupted local processing, and reduced network flexibility, which in turn contribute to emotional dysregulation, executive dysfunction, and cognitive rigidity in BD. Notably, we found decreased
![]() $ \gamma $
and
$ \gamma $
and
![]() $ \sigma $
were correlated with the HDRS scores and number of episodes in BDII-D, suggesting more serious depressive symptoms and more episodes are related to more pronounced disrupted functional networks in unmedicated BDII-D. Specifically, a previous rs-fMRI study reported that altered resting-state functional network connectivity was significantly associated with symptom severity on depression rating scales in unmedicated BD (He et al., Reference He, Yu, Du, Vergara, Victor, Drevets and Calhoun2016). However, this previous study had the limitation of a relatively small sample size and inclusion of both BD type I and type II individuals. In the context of cognitive therapy, brain functional network change was associated with the improvement of emotion regulation in mood-dysregulated adolescents at familial risk for BD (Qin et al., Reference Qin, Lei, Yang, Li, Tallman, Duran and DelBello2021), which suggests abnormal brain functional network might underline the mechanism of mood dysregulation. Furthermore, we found the correlation between brain functional network topology and number of episodes in BD. Although few studies have reported this correlation in BD, studies in MDD have reported the number of depressive episodes was significantly positively associated with altered functional network connectivity (Goya-Maldonado et al., Reference Goya-Maldonado, Brodmann, Keil, Trost, Dechent and Gruber2016; Lu et al., Reference Lu, Chen, Cui, Guo, Pang, Luo and Chen2023; Morgan et al., Reference Morgan, Shaw, Olino, Musselman, Kurapati and Forbes2016). Therefore, our findings shed lights on the importance of functional network topological abnormalities in the neuropsychological processes and neuropsychiatric symptoms of BD.
$ \sigma $
were correlated with the HDRS scores and number of episodes in BDII-D, suggesting more serious depressive symptoms and more episodes are related to more pronounced disrupted functional networks in unmedicated BDII-D. Specifically, a previous rs-fMRI study reported that altered resting-state functional network connectivity was significantly associated with symptom severity on depression rating scales in unmedicated BD (He et al., Reference He, Yu, Du, Vergara, Victor, Drevets and Calhoun2016). However, this previous study had the limitation of a relatively small sample size and inclusion of both BD type I and type II individuals. In the context of cognitive therapy, brain functional network change was associated with the improvement of emotion regulation in mood-dysregulated adolescents at familial risk for BD (Qin et al., Reference Qin, Lei, Yang, Li, Tallman, Duran and DelBello2021), which suggests abnormal brain functional network might underline the mechanism of mood dysregulation. Furthermore, we found the correlation between brain functional network topology and number of episodes in BD. Although few studies have reported this correlation in BD, studies in MDD have reported the number of depressive episodes was significantly positively associated with altered functional network connectivity (Goya-Maldonado et al., Reference Goya-Maldonado, Brodmann, Keil, Trost, Dechent and Gruber2016; Lu et al., Reference Lu, Chen, Cui, Guo, Pang, Luo and Chen2023; Morgan et al., Reference Morgan, Shaw, Olino, Musselman, Kurapati and Forbes2016). Therefore, our findings shed lights on the importance of functional network topological abnormalities in the neuropsychological processes and neuropsychiatric symptoms of BD.
At the nodal level, the BDII-D group showed lower nodal efficiency in the left pallidum, right olfactory cortex, and cerebellum vermis, suggesting impaired regional connectivity of these regions. These findings were partly consistent with previous studies showing decreased nodal efficiency of the left pallidum (A. Zhang et al., Reference Zhang, Qiao, Wang, Yang, Wang, Sun and Zhang2023) and lower nodal characteristic path length in the right olfactory cortex in BD (Dvorak et al., Reference Dvorak, Hilke, Trettin, Wenzler, Hagen, Ghirmai and Oertel2019). Emerging evidences proved the basal ganglia including the pallidum regulate affect (Caligiuri et al., Reference Caligiuri, Brown, Meloy, Eberson, Niculescu and Lohr2006) and carry non-motor information such as reward and sensory stimuli (Howell et al., Reference Howell, Prescott, Lozano, Hodaie, Voon and Hutchison2016). In healthy individuals, reduced incentive cue-related activation in the pallidum was associated with higher anhedonia (Chung & Barch, Reference Chung and Barch2015), which is a core symptom of depression (Pizzagalli, Reference Pizzagalli2014). Also, decreased cerebral blood flow was found in the left pallidum in adolescents with depression (Fu et al., Reference Fu, Liu, Wang, Li, Dai, Ren and Yu2023). The olfactory cortex has been reported to be involved in sensory functions, emotional regulation, and memory formation (Yuan & Slotnick, Reference Yuan and Slotnick2014). In animal models, bilateral olfactory bulbectomy results in changes in behavior, and in the endocrine, immune, and neurotransmitter systems that simulate many of those seen in individuals with depression (Coppola & Parrish Waters, Reference Coppola and Parrish Waters2021; Song & Leonard, Reference Song and Leonard2005). Neuroimaging studies have demonstrated structural abnormalities of the olfactory sulci in BD (Kocakaya, Bayar Muluk, & Bekin Sarikaya, Reference Kocakaya, Bayar Muluk and Bekin Sarikaya2023; Takahashi et al., Reference Takahashi, Malhi, Nakamura, Suzuki and Pantelis2014). The vermis is considered to be a part of the limbic cerebellum (Bodranghien et al., Reference Bodranghien, Bastian, Casali, Hallett, Louis, Manto and van Dun2016; Schmahmann, Reference Schmahmann2004), which is critical in the modulation of cognition and emotion (Schmahmann, Weilburg, & Sherman, Reference Schmahmann, Weilburg and Sherman2007). Several previous studies have reported decreased amplitude of low-frequency fluctuation (Chen et al., Reference Chen, Gong, Chen, Chen, Zhong, Tang and Wang2022; Gong et al., Reference Gong, Wang, Qiu, Chen, Luo, Wang and Wang2020; Lai et al., Reference Lai, Zhong, Shan, Wang, Chen, Luo and Jia2019; Shunkai et al., Reference Shunkai, Chen, Zhong, Chen, Zhang, Zhao and Wang2023; Zhao et al., Reference Zhao, Luo, Qiu, Jia, Zhong, Chen and Wang2020) in the cerebellum in unmedicated BD II. A recent study has demonstrated that the baseline functional connectivity between the vermis and temporal lobes was associated with antidepressant treatment outcomes in individuals with mood disorders (Wang et al., Reference Wang, Zhao, Zhang, Zhang, Liu, Duan and Wang2023). On the basis of these studies, this study further shows the important roles the pallidum, olfactory cortex, and vermis play in the neurophysiological mechanism of BDII-D.
In this study, we found significantly elevated inflammatory cytokine levels such as IL-8, IL-10, and G-CSF in BDII-D, which were largely consistent with previous findings in meta-analyses of BD (Misiak et al., Reference Misiak, Bartoli, Carrà, Małecka, Samochowiec, Jarosz and Stańczykiewicz2020; Modabbernia et al., Reference Modabbernia, Taslimi, Brietzke and Ashrafi2013). Notably, we found a negative correlation between IL-8 and functional brain network topology in BD, suggesting that inflammation might play a role in the shift of functional network topology toward randomization in BD. Previous neuroimaging studies have shown the relationship between inflammation and regional functional connectivity abnormalities in BD (Chen et al., Reference Chen, Chen, Chen, Zhong, Gong, Zhong and Wang2020 ; Tang et al., Reference Tang, Chen, Chen, Zhong, Gong, Zhong and Huang2021; Tseng et al., Reference Tseng, Chang, Wei, Lu, Hsieh, Yang and Chen2021). As a continuation and complement to previous research, this study further reflects the involvement of inflammation in the mechanisms of BD from the perspective of the whole-brain functional network. IL-8, a proinflammatory cytokine produced by many cell types including macrophage and microglia, mainly functions as a neutrophil chemoattractant not only in the bloodstream but also in the brain, and represents chronic inflammatory changes in neurodegenerative and neuropsychological alterations in the brain (Tsai, Reference Tsai2021). According to previous evidence, chronic neuroinflammation caused by the activation of microglia and astrocytes in the brain contributes to neuronal loss and disease progression in neurodegenerative diseases (Basurco et al., Reference Basurco, Abellanas, Ayerra, Conde, Vinueza-Gavilanes, Luquin and Aymerich2023). Also, neuroinflammation may disrupt the delicate balance needed for neurophysiological activities and exert direct harmful impacts on neural plasticity and neurogenesis, facilitating many kinds of neuropathologies associated with neuropsychiatric diseases including BD (Yirmiya & Goshen, Reference Yirmiya and Goshen2011). In cell culture studies, reactive microglia can inhibit neuronal autophagy (Festa et al., Reference Festa, Siddiqi, Jimenez-Sanchez, Won, Rob, Djajadikerta and Rubinsztein2023) and induce different levels of neuronal network dysfunction (Schilling et al., Reference Schilling, Chausse, Dikmen, Almouhanna, Hollnagel, Lewen and Kann2021); GM-CSF induces the proliferation of microglia and disturbs electrical neuronal network rhythms in situ (Dikmen et al., Reference Dikmen, Hemmerich, Lewen, Hollnagel, Chausse and Kann2020). In animal studies, inflammation at early developmental stages is sufficient to exert a long-lasting effect on glutamatergic synaptogenesis and brain connectivity (Mirabella et al., Reference Mirabella, Desiato, Mancinelli, Fossati, Rasile, Morini and Pozzi2021), and targeting neuroinflammation by pharmacologic downregulation of inflammatory pathways is neuroprotective in protein misfolding disorders (Risen et al., Reference Risen, Boland, Sharma, Weisman, Shirley, Latham and Moreno2024). In individuals with depression, inflammation was associated with reduced functional connectivity in a widely distributed network centralized in the ventral medial prefrontal cortex (Yin et al., Reference Yin, Xu, Chen, Mehta, Haroon, Miller and Felger2019). A recent systematic review has shown the association of blood biomarkers including IL-8 with cerebral white matter and myelin content in BD (Ali, Husnudinov, Wollenhaupt-Aguiar, & Frey, Reference Ali, Husnudinov, Wollenhaupt-Aguiar and Frey2024). Interestingly, a recent study shows that regional global brain connectivity changed after pro- and anti-inflammatory therapies (Martins et al., Reference Martins, Dipasquale, Davies, Cooper, Tibble, Veronese and Harrison2022). Based on the previous findings, the potential influence of IL-8 on brain network topology may be mediated through the activation of microglia and the subsequent disruption of neurotransmitter metabolism. These processes could exert both direct and indirect effects on neural network architecture. Specifically, the activation of microglia by IL-8 may lead to the release of pro-inflammatory cytokines, which in turn can impair neuronal function, disrupt neural connectivity, and attenuate neuroplasticity. Collectively, these alterations may contribute to the observed changes in brain network topology, highlighting the multifaceted role of IL-8 in modulating neuroinflammatory processes and their downstream effects on neural network integrity. Future research is needed to further elucidate the mechanisms underlying the role of IL-8 in neuroinflammation and to explore its potential as a biomarker and therapeutic target.
Furthermore, in this study, we found elevated IL-8 level was correlated with decreased nodal efficiency in the left pallidum in BDII-D, which suggests inflammation might contribute to impaired regional connectivity in the basal ganglia in BD. Decreased nodal efficiency in the left pallidum has been previously reported in BD individuals (Zhang et al., Reference Zhang, Qiao, Wang, Yang, Wang, Sun and Zhang2023). In the present study, we validated this finding in the BDII-D subtype and further revealed a potential association between this neuroimaging alteration and peripheral inflammation. Emerging evidences have demonstrated that inflammatory cytokines are associated with abnormal basal ganglia function and neurotransmitter activities, leading to symptoms of depression. For example, in nonhuman primates, a positron emission tomography study indicated that chronic peripheral cytokine exposure reduces striatal dopamine release in association with anhedonia-like behavior (Felger et al., Reference Felger, Mun, Kimmel, Nye, Drake, Hernandez and Miller2013). In individuals with major depression, increased inflammation is associated with increased basal ganglia glutamate activity (Haroon et al., Reference Haroon, Fleischer, Felger, Chen, Woolwine, Patel and Miller2016). Also, the administration of the inflammatory cytokine has been shown to increase brain glutamate in the basal ganglia as measured by magnetic resonance spectroscopy in depression (Haroon & Miller, Reference Haroon and Miller2017). Besides, inflammation was consistently found to affect basal ganglia and cortical reward and motor circuits to drive reduced motivation and motor activity, which may result from cytokine effects on monoamines and glutamate (Felger, Reference Felger2018). A task-fMRI study showed the associations between inflammatory processes and activation of basal ganglia regions during multiple phases of reward processing in psychotropic medication-free youth with psychiatric symptoms (Bradley et al., Reference Bradley, Stern, Alonso, Xie, Kim-Schulze and Gabbay2019). Based on the previous findings, our result suggests that the pallidum may serve as a potential neuroimaging biomarker and anti-inflammation target in BD in future research. Moreover, the role of IL-8 in neuroinflammation highlights its potential as a therapeutic target. By modulating IL-8 signaling, it may be possible to reduce neuroinflammatory processes that contribute to neuronal dysfunction and network abnormalities, as observed in conditions like BD. Targeting IL-8 could also help in mitigating the downstream effects of chronic inflammation on brain connectivity and plasticity, potentially improving clinical outcomes.
The association between aberrant network topology and IL-8 level underscores the necessity of investigating IL-8 as a therapeutic target to restore normal brain network function and enhance clinical outcomes in individuals with BDII-D. Additionally, the correlation between abnormal brain network topology and the HDRS scores and number of episodes in BDII-D suggests the potential to develop personalized treatment strategies that address both inflammation and network connectivity, ultimately improving symptom management and reducing the recurrence of depressive episodes in BDII-D individuals.
Limitations
There are several limitations to this study that need to be mentioned. First, the sample size was relatively small. Additional studies with a bigger sample size from multi-centric cohorts or data-sharing initiatives are needed in future research. Second, since this was a cross-sectional study, we were unable to further investigate the causal relationship between topological organization disruption and inflammation. Therefore, longitudinal studies are necessary to confirm the role of cytokines in brain function. Third, we were unable to collect the cytokine levels in CSF in this study. However, peripheral cytokine levels remain the primary basis for evaluation due to ethical considerations for acquiring CSF or brain tissue. Peripheral cytokine levels may not fully reflect brain cytokine levels, but may affect brain cytokine production. Besides, cytokine levels may also be influenced by stress. More research is needed to explore the role of pathological “states” (i.e. current mental disorders) and their impact on brain immune relationships under socially stressful conditions. Fourth, the correlation results did not withstand after multiple comparison corrections. Therefore, our findings in this study should be regarded as preliminary and should be cautiously inferred, which should be further replicated in a larger sample size or multi-center study. Fifth, chronic inflammation is known to be associated with a wide range of metabolic and immune changes, which could potentially confound the interpretation of our results. Future studies should consider incorporating more comprehensive assessments of inflammatory status to better control for this potential confounding factor.
Conclusion
To summarize, this study displayed disrupted brain functional network topology and elevated peripheral cytokines in BD. Elevated pro-inflammatory cytokine IL-8 level might contribute to both decreased global brain network integration and segregation, and regional efficiency of the basal ganglia in unmedicated BDII-D.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0033291725100834.
Data availability statement
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Acknowledgments
Ying Wang designed the study; Guixian Tang, Guanmao Chen, and Pan Chen contributed to data selection; Guixian Tang, Guanmao Chen, Pan Chen, Yanbin Jia, Shuming Zhong, Feng Chen, Jurong Wang, Zhenye Luo, and Zhangzhang Qi contributed to data acquisition; Guixian Tang, Guanmao Chen, Pan Chen, Hui Zhong, and Hengwen Yang contributed to data analysis; Guixian Tang and Guanmao Chen wrote the manuscript; Guixian Tang, Guanmao Chen, Pan Chen, Li Huang, and Ying Wang revised the manuscript. All authors contributed to and had approved the final manuscript.
Funding statement
The study was supported by grants from the National Natural Science Foundation of China (82472057, 81971597), Science and Technology Projects in Guangzhou (2024B03J1299). The funding organizations play no further role in study design, data collection, analysis and interpretation, and paper writing.
Competing interests
The authors have declared that no competing interest exists.
Ethical standard
This study was approved by the Ethics Committee of First Affiliated Hospital of Jinan University, Guangzhou, China. All subjects were right-handed and signed a written informed consent form after a full written and verbal explanation of the study. Two senior clinical psychiatrists confirmed that all subjects had the ability to consent to participate in the examination.