Introduction
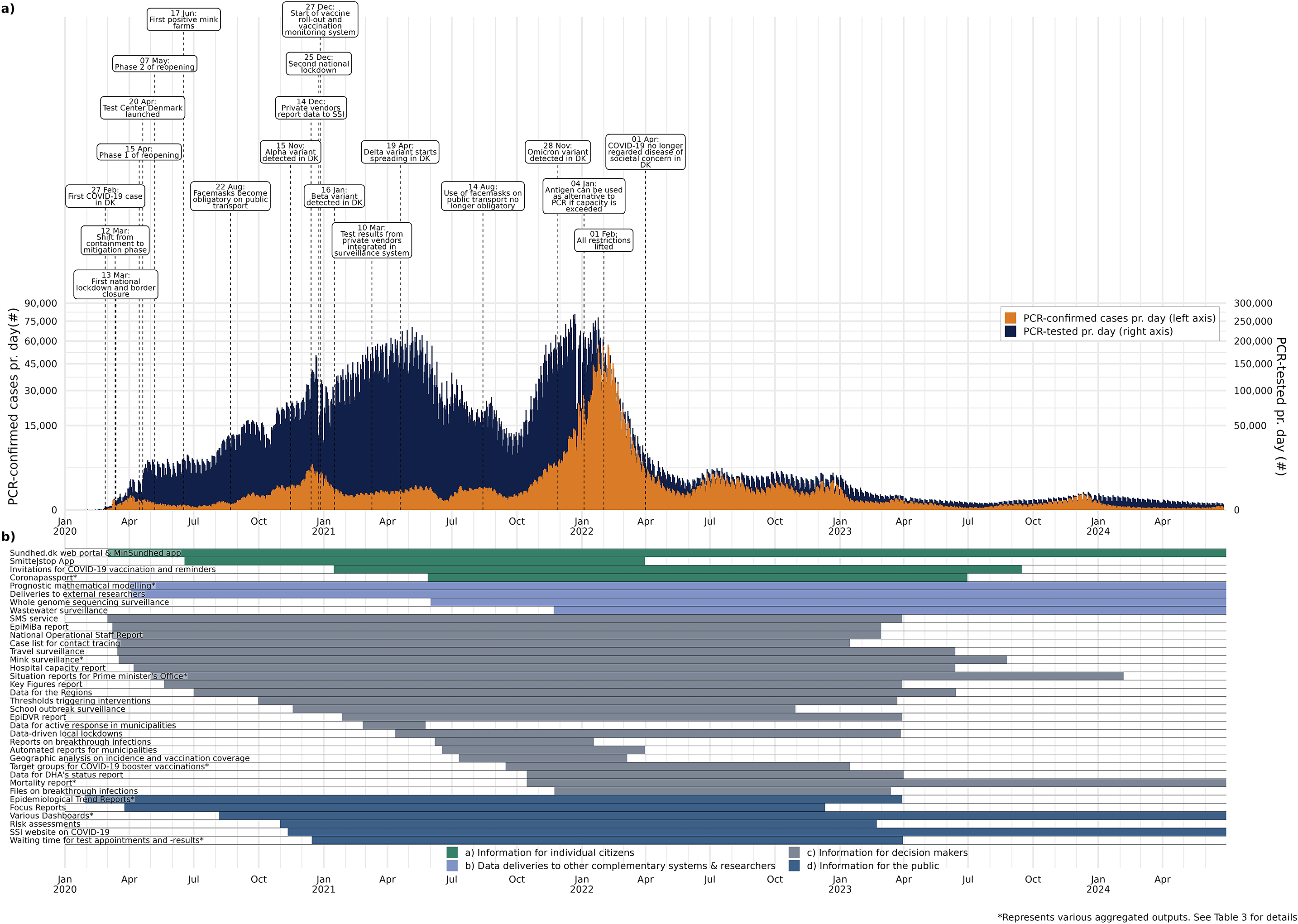
Infectious disease surveillance allows monitoring trends, describing the epidemiology and burden of disease in a population, enables the early detection of outbreaks, epidemics, and new pathogens, and thereby informs data-driven public health responses [Reference Murray and Cohen1]. When the new SARS-CoV-2 virus started to emerge in Denmark, with the first case detected on 26 February 2020, Statens Serum Institut (SSI), which is responsible for the national infectious disease preparedness, rapidly set up an automated, real-time surveillance system for COVID-19 within days. Figure 1a shows key milestones of the epidemic in Denmark that unfolded in form of several waves caused by various variants, as well as response measures taken. Two national lockdowns were implemented, and the COVID-19 vaccine roll-out started at the end of December 2020. Denmark’s epidemic response was also characterized by a high test capacity.
(a) Epidemic curve showing the number of PCR-confirmed COVID-19 cases and the number of persons tested by PCR per day as of January 2020 in Denmark including key events and (b) timespan indicating when outputs were produced in relation to the epidemic.
Outputs are coloured according to the four main stakeholder groups as described in Table 3. The number of tests and confirmed cases reflects persons tested by PCR. Antigen tests for SARS-CoV-2 played an important role in the epidemic response in Denmark, with the number of antigen tests performed per day ranging from ~100.000–500.000 between March 2021 and March 2022. Persons testing positive by antigen tests were advised to get confirmation via PCR due to the superior sensitivity and specificity of the latter. Therefore, the majority of outputs are based on PCR-confirmed cases (Table 3).
In this article, we outline the setup of the automated, real-time integrated surveillance and vaccination monitoring system for COVID-19 in Denmark, describe how outputs for various stakeholders were used for action and reflect on challenges and lessons learnt. This surveillance system also constitutes a model for the future infectious disease surveillance system in Denmark.
Methods
Country profile
Denmark is part of the European Union (EU) and has a population of roughly 5.9 million people and a geographical size of 42 925 km2 [2].
Every resident of Denmark has free access to the Danish healthcare system and receives a health insurance card that includes their name and civil registration number (CPR number) – a unique personal identifier assigned to every resident of Denmark. The majority of treatments and examinations are free [3].
Primary stakeholders in the epidemic response in Denmark
Table 1 shows primary stakeholders in the epidemic response and their roles and responsibilities. SSI falls under the Ministry of the Interior and Health and is responsible for the prevention and control of infectious and congenital diseases through research, surveillance, and advice. Since the legal basis for receiving data differed by stakeholder, outputs, calculations, and visualizations needed to be tailored accordingly.
Primary stakeholders in the epidemic response in Denmark with a focus on roles and responsibilities during the epidemic
Indentations and different font types in the stakeholder column reflect the hierarchy among stakeholders
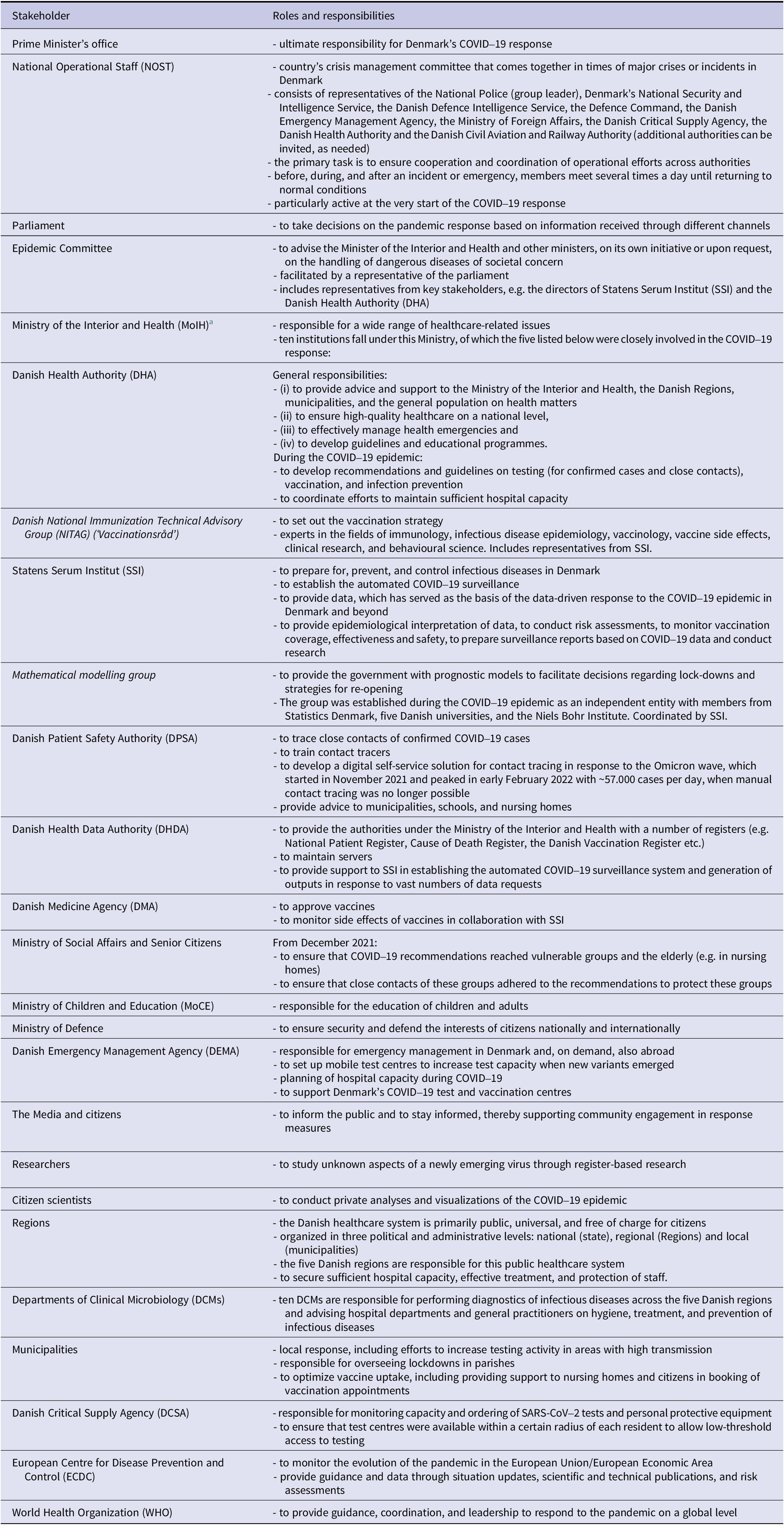
a Between 27 June 2019 and 14 December 2022: ‘Ministry of Health and Elderly Affairs’.
Setup of the Danish COVID-19 surveillance and vaccination monitoring system
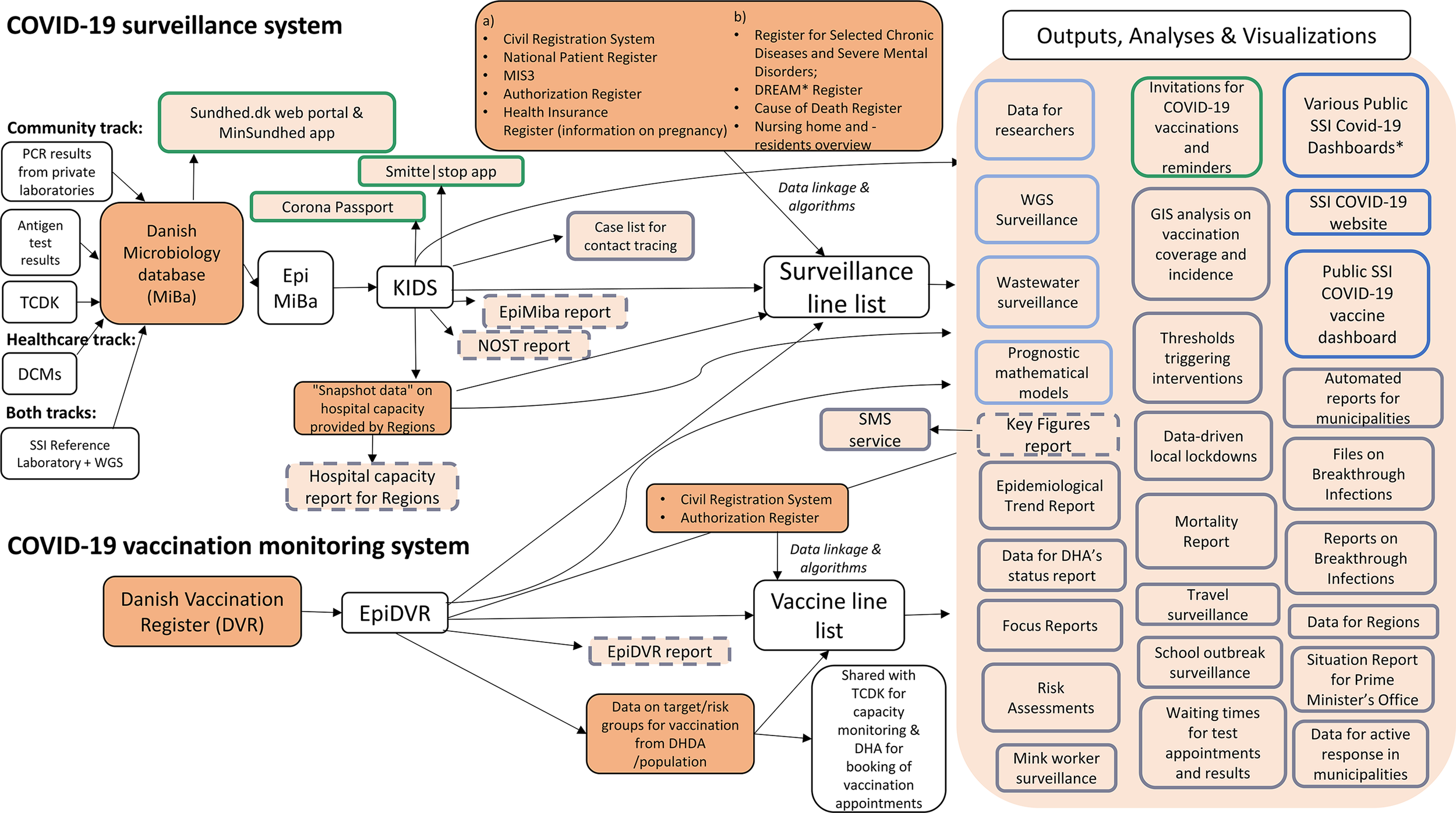
Figure 2 shows an overview of the dataflow of the Danish COVID-19 surveillance and vaccination monitoring system, including data sources used (Table 2) and outputs produced (Table 3). The following sections describe key elements of the system in more detail.
Overview of the dataflow of the Danish COVID-19 surveillance and vaccination monitoring system, including data sources, outputs, analyses, and visualizations.
Data sources and registers are depicted in dark orange and outputs, analyses and visualizations in light orange. Column (a) indicates data sources that SSI already had permission for to use for surveillance purposes prior to the COVID-19 epidemic, and column (b) shows existing data sources to which access was granted for surveillance purposes during the COVID-19 epidemic.
Coloured outlines indicate the four main stakeholder groups, as described in Table 3, to which outputs, analyses, and visualizations were tailored. In green – information for individual citizens, in light blue – data deliveries to other complementary systems and researchers, in grey – information for decision-makers, and in dark blue – information for the public. Automated e-mail reports are depicted with a dashed outline.
Abbreviations: DCMs, Departments of Clinical Microbiology; TCDK, Test Center Denmark; MiBa, Danish Microbiology Database; SSI, Statens Serum Institut; KIDS, Keys to Infectious Disease Surveillance; NOST, National Operational Staff; CRS, Civil Registration System; NPR, National Patient Registry; DHDA, Danish Health Data Authority; DVR, Danish Vaccination Register; DPSA, Danish Patient Safety Authority; DHA, Danish Health Authority; DREAM, database from which type of employment was extracted (see Table 2), WGS, whole genome sequencing.
Overview of registers and other data sources used in the Danish COVID-19 surveillance and vaccine monitoring system, responsible authority and type of information used

a Danish acronym of ‘Den Registerbaserede Evaluering af Marginaliseringsomfanget’ translating into ‘The evaluation of marginalized groups of individuals based on registered social public transfer payments’.
Overview of outputs and data deliveries of the Danish COVID-19 surveillance and vaccination monitoring system for the four major stakeholder groups
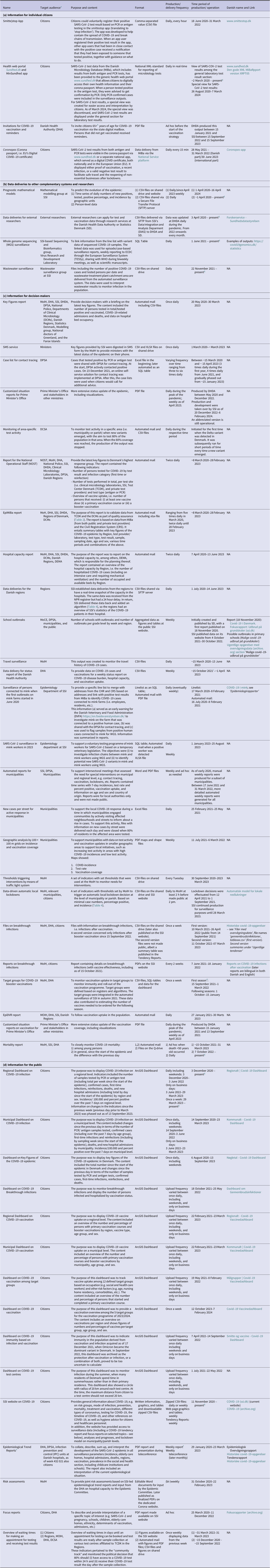
a For abbreviations, see Table 2.
b 50+ of age in season 2020/21.
c DHDA produced the status report before SSI took over the production.
d DHDA defined the target groups based on templates from Regions with an overview of persons eligible to receive the vaccine, before SSI took it over as of summer 2021.
e The name of this output changed during the course of the epidemic from ‘epidemiological report’, signal report from week 45, 2020 to ‘trend report’ from week 43, 2021 onwards. Trend reports became publicly available on the SSI website.
f With a few deviations from the weekly schedule during the summer months.
g Status as of end of June 2024.
The Danish microbiology database, EpiMiBa and Keys to Infectious Disease Surveillance (KIDS)
MiBa, as described elsewhere [Reference Schønning5, Reference Voldstedlund6], forms the core of the COVID-19 surveillance system. In short, MiBa is the national microbiology database established at SSI in 2010. It is managed by SSI’s Department for Data Integration and Analysis (DIAS), that also is responsible for the development and maintenance of the automated infectious disease surveillance system in Denmark. MiBa receives microbiology results from (i) all 10 Danish Departments of Clinical Microbiology (DCMs) across the five Regions of Denmark and (ii) reference laboratories based at SSI. MiBa serves a dual purpose: to allow healthcare providers access to patients’ microbiology test results and to provide a basis for surveillance of infectious diseases and pathogens [Reference Schønning5, Reference Voldstedlund6]. For COVID-19 surveillance, MiBa also integrated test results from Test Center Denmark (TCDK) [7], as well as PCR and antigen test results from private laboratories [Reference Schønning5] (Figure 2).
EpiMiBa constitutes a mirrored version of MiBa, which is used as a basis for surveillance and research. For EpiMiBa, Central Dynamic Mapping is used to convert local codes submitted by DCMs into shared standard coding and uniform terminology to facilitate data extraction and statistical analysis [Reference Voldstedlund6].
Raw data from EpiMiBa is subsequently standardized in the KIDS, where the following levels are applied to the data through the use of algorithms: (1) demarcation of all potentially relevant test reports, that is all test reports with SARS-CoV-2 mentioned in their requisition or results codes, (2) interpretation of test reports (i.e. categorization into positive, negative, or irrelevant results for SARS-CoV-2) and application of the so-called ‘patient-date’ (Table 4), and (3) application of episode definition, where test results are converted to cases and are used to determine reinfections (i.e. time window of 60 days between two SARS-CoV-2 infections) (Table 4) [Reference Michlmayr8].
Overview of algorithms used in the Danish COVID-19 surveillance and vaccination monitoring system
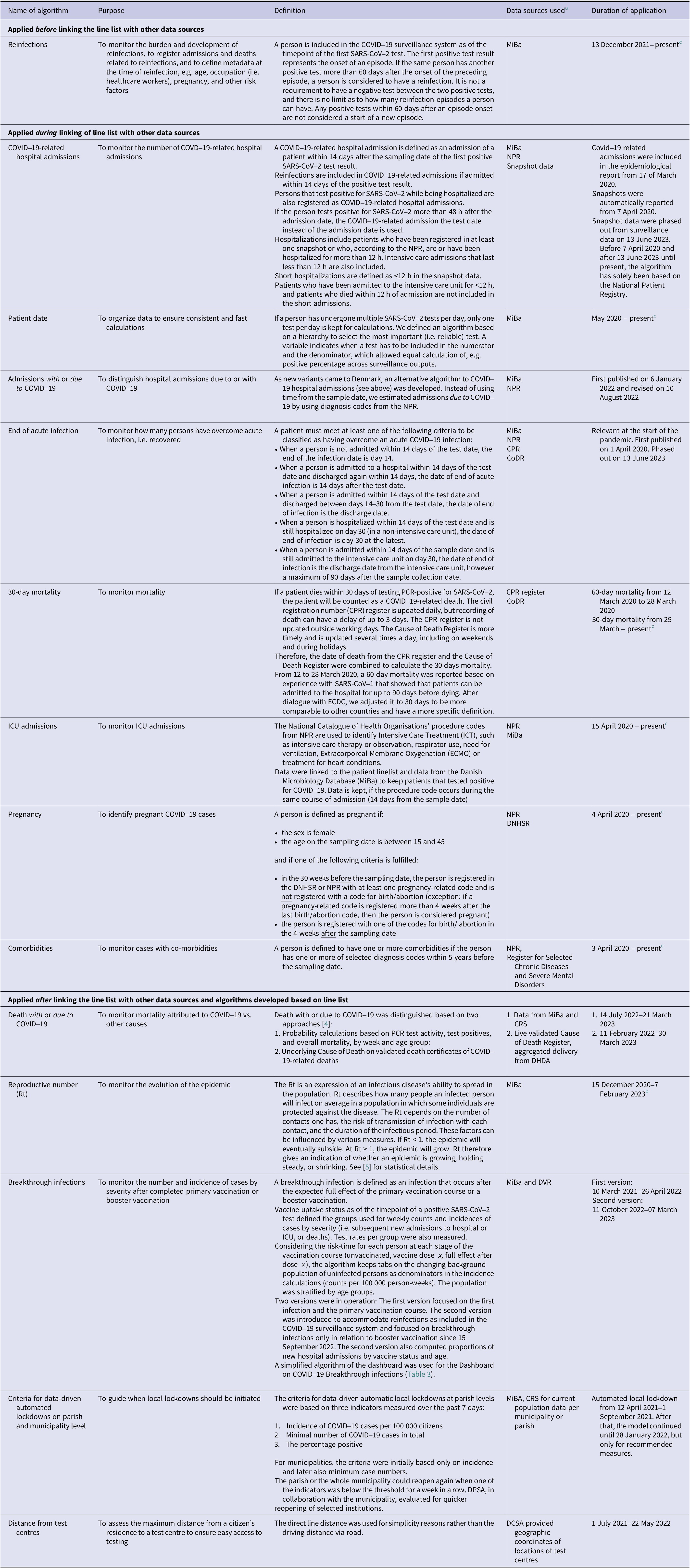
a See Table 2 for details.
b Period of automated production; manually calculated as of July 2020.
c Status as of end of June 2024.
SARS-CoV-2 test results formed the core of the surveillance system, which was therefore heavily influenced by the test strategy and capacity. In addition to the public testing system as described elsewhere [Reference Schønning5, Reference Gram9], private vendors provided SARS-CoV-2 PCR and antigen tests as of summer 2020. These became publicly funded at the end of 2020 to expand the free-of-charge test capacity. By March 2021, test results from private providers, including antigenic tests, were fully integrated in the surveillance system and were used for screening, contact tracing, and the Digital COVID-19 certificate (Figure 1a, Table 3).
The Danish vaccination register (DVR) and EpiDVR
The DVR was implemented in 2012 to support monitoring of vaccination coverage and estimating vaccine effectiveness. It is based on mandatory registration of all administered vaccinations, including those not included in the national vaccination programme [Reference Krause10].
EpiDVR is a mirrored version of DVR used for monitoring vaccination coverage and the childhood vaccination programme. This system includes a function for sending out electronic invitations to vaccination appointments, reminders before appointments, and reminders if the recommended vaccination time is exceeded by 4 weeks.
The roll-out of the COVID-19 vaccination programme started on 27 December 2020 and coincided with the second national lockdown (Figure 1a). SSI, together with the Danish Health Data Authority (DHDA), developed a monitoring system to track COVID-19 vaccine uptake, coverage, and effectiveness, as well as the capacity of vaccination centres.
Line lists, data sources, data linkage and algorithms
Standardized test results have been subsequently linked with data from national registers and other data sources through CPR number to create a line list. A line list is a table with key information, where each row represents a person and each column represents metadata, such as demographics, clinical and epidemiological information. Dedicated surveillance and vaccination line lists have formed the basis for the majority of outputs described hereafter (Table 3).
Table 2 shows an overview of the data sources, the responsible authorities, and including descriptions of the types of data used from the data sources. Before, during, and after data linkage, various algorithms have been applied (Table 4). These vary in complexity and consist of business rules used for automation processes. Each algorithm was developed and documented by a multidisciplinary team using available international literature and guidelines, as well as clinical expertise. Algorithms were approved on director’s level and in some cases ministerial level. The algorithms were validated with the available means, such as clinical data. Where validation based on clinical data was not possible, algorithms were validated through thorough data analysis, investigating outliers, and scientific discussions. Later in the epidemic, an algorithm for reinfections was developed, and the line list was changed to represent episodes of infection rather than unique persons. Documentation on the algorithms and limitations for interpretation was published on SSI’s website.
Results
Cohort included in the COVID-19 surveillance and vaccination monitoring system
Based on the Civil Registration System (CRS) (Table 2), the Danish population counts ~5.9 million by 2023. Differences in testing patterns during the three epidemic waves of SARS-CoV-2 in Denmark by demography have been described elsewhere [Reference Gram9]. Confirmed cases were registered based on their first SARS-CoV-2 PCR test. As of 27 June 2024, 3 438 887 COVID-19 cases, including reinfections, and 9863 COVID-19-related deaths have been recorded since the beginning of the epidemic in Denmark.
To track vaccine uptake, the vaccination monitoring system comprised the entire population of Denmark. Since the start of the vaccine roll-out, 4 737 718 persons have received at least one vaccination.
Outputs and use of data by various stakeholders
Outputs from the automated surveillance system can be divided into four stakeholder categories listed below. Table 3 shows an overview of outputs by stakeholder group, name, target audience, purpose, content, frequency, and time period of production and links where available. Figure 1b shows when various outputs were produced in relation to the COVID-19 epidemic in Denmark.
Information for individual citizens
Information for individual citizens (Table 3a) entailed online access to their own SARS-CoV-2 test results and their COVID-19 vaccinations through a generic web portal sundhed.dk, including the corresponding app MinSundhed (‘MyHealth’). This platform, which already existed before the COVID-19 epidemic, provides access to any kind of diagnostic test result from all clinical laboratory-specialities, vaccinations, and other health information. Displayed microbiological test reports are obtained from MiBa (Figure 2). Physicians can access the same information through a patient’s electronic health record [Reference Schønning5].
Analysed data from KIDS were automatically provided to the Digital COVID-19 Certificate (in Danish: Coronapas App) that documented vaccination, previous infection, or recent negative test results. It offered a ‘two-in-one solution’: (a) the International Coronapas was mandatory for travelling abroad [11] and (b) the Domestic Coronaspas – based on automatic business rules – was mandatory for entering events in Denmark. Large-scale testing and a fast turnaround (15–20 min from the test result was available at the laboratory until the Coronapas was automatically updated) contributed – among others – to keeping society as open and safe as possible. KIDS data was also fed to the Smitte|stop app (‘transmission stop’) that was used to inform citizens if they had had close contact with a SARS-CoV-2-positive person.
Also, in 2020, DVR’s electronic invitation-functionality was expanded to send invitations to citizens in the target groups for COVID-19 vaccination.
Data deliveries to complementary systems and researchers
In April 2020, the Ministry of the Interior and Health (MoIH) tasked SSI with forming an expert group for mathematical modelling, which has since received data from the surveillance system. The purpose was to support predictions about the evolution of the epidemic in Denmark, the burden on hospitals, and regional predictions for the number of new COVID-19 cases, including the effect of actions for gradual reopening after the lockdowns [12].
Other surveillance applications that used data from the surveillance system included whole genome sequencing (WGS) surveillance and wastewater surveillance (Figure 2). For WGS surveillance, data from the line list was linked to sequencing results. For wastewater surveillance, the surveillance system provided information on the daily PCR testing rate and incidence within the catchment area of each wastewater treatment plant [Reference McManus13]. Researchers have also had the opportunity to request daily updated data through dedicated application processes set up by DIAS and the DHDA (Tables 1 and 3b).
Information provided to decision makers
Recipients of data deliveries included SSI-internal and external national and international stakeholders (Table 3c).
Data for decision-makers was provided in various ways, for example as automated e-mails and comma-separated value (CSV) files that included, among others, key numbers for daily meetings on preparedness and hospital capacity planning. Data were provided close to real-time and multiple times daily, which also required rapid epidemiological interpretation by SSI-internal stakeholders.
At the MoIH, this information was converted into an SMS-service to ensure continuous status updates for ministers. In addition, the Prime Minister’s office required a more extensive PDF file with visualizations of the current status.
The Danish Patient Safety Authority (DPSA) received data on new PCR and antigen test-positive persons, including key metadata for contact tracing (Table 1).
Regions and municipalities formed a new group of stakeholders during the pandemic. Several outputs were developed to directly support the regional and local level in managing the COVID-19 response (Table 3c).
Information for the public
For the public, SSI developed various interactive COVID-19 dashboards (Table 3d), among others, for infection and vaccination on a regional and municipal level, as well as a dashboard with key figures of the epidemic. Between August 2020 and March 2023, the dashboards were updated daily. Prior to uploading, underlying data from the dashboards were shared with authorities under embargo. By April 2023, when COVID-19 was no longer considered a disease of societal concern in Denmark, the frequency of updating the regional dashboard decreased to once a week, and vaccine dashboards were archived but still remain accessible. For the COVID-19 vaccination programme of 2023/2024, a new vaccine dashboard was launched. CSV-files with data underlying the dashboards and other supplementary calculations were uploaded daily until 29 March 2023. Afterwards, the frequency of data uploads had been reduced to weekly. In addition, SSI published data on its website in the form of figures and tables once a week (Table 3d).
Discussion
This article describes how the Danish COVID-19 surveillance and vaccine monitoring system was built and how key outputs were used to support the epidemic response of various stakeholders. The emergence of the COVID-19 epidemic constituted the first time in Denmark that close to real-time infectious disease data were provided to decision-makers and the public [Reference Schønning5]. Data on (re)infection and vaccination not only guided the national COVID-19 response but also supported interventions and outbreak investigations in special settings, for example long-term care facilities [Reference Espenhain14], schools [Reference Funk15], mink farms [Reference Larsen16], and large-scale events, such as the European Soccer Championships [Reference Bennedbæk17]. Data also enabled addressing public health research questions, for example to investigate SARS-CoV-2 transmission within households [Reference Lyngse18], protection against reinfection after primary infection [Reference Michlmayr8, Reference Hansen19], risk of hospitalizations associated with different SARS-CoV-2 variants [Reference Bager20, Reference Bager21], COVID-19 vaccine effectiveness and uptake [Reference Gram22, Reference Nielsen23], and long COVID [Reference Sørensen24].
Building on existing foundation
Denmark is one of the internationally leading countries with respect to establishing integrated digital solutions in the health sector [25], and earlier investments in digitalization in the health field have laid a solid basis for surveillance. Before SARS-CoV-2 emerged in Denmark, the prerequisites for real-time, automated reporting, including MiBa and DVR, had been in place for more than a decade. This allowed rapid integration and upscaling of surveillance for COVID-19. The system has proven highly flexible and adaptable, was supported by a high test capacity and a low threshold to access testing, and covered the entire population of Denmark. Having had the infrastructure in place to accommodate the strategy of mass testing and to digitally handle the associated high amounts of real-time data can be considered a major achievement in infectious disease surveillance. Although developed under high time pressure, the system proved stable and largely ran effectively throughout the pandemic. System breakdowns were rapidly addressed and therefore resulted in very limited downtime.
The availability of the population-wide number of tests performed allowed representative outputs, e.g. calculation of percent positive and the reproductive number (Rt). The high degree of completeness (i.e. all data on tests, vaccinations, and hospital admissions were captured), accuracy, and timeliness in both the numerator and denominator was, among others, a prerequisite to enable local responses, for instance, for setting sensitive data-thresholds to initiate lockdowns on a parish level that were linked with high political, social, and economic consequences [Reference Espenhain26].
Transparency and data sharing
In Denmark, there is a high level of trust between authorities and the public [Reference Gamerdinger27], which has been conducive to compliance with the testing and vaccination system. Data were continuously shared with authorities and researchers throughout the epidemic. Data shared on the SSI website were used by the media and citizen scientists for customized visualizations. In addition, SSI actively addressed questions by the media and the public on the SSI website and social media.
The availability of timely data had also sparked an interest of various national and international stakeholders and societies, which led to a substantial increase in requests for data and access to documents. To support SSI, the DHDA assisted with selected data deliveries. To ensure close collaboration regarding surveillance data between authorities under the MoIH, a data coordination group was established, which was chaired by SSI. The group included representatives of MoIH, the Danish Health Authority (DHA), DPSA, and DHDA (Table 1).
Human resource challenges and organizational considerations
Human resources were an important aspect that challenged the automated COVID-19 surveillance. The close-to-real-time access to data and high level of transparency required tailoring outputs for multiple stakeholders with competing timelines and priorities, responding to ad hoc requests, and predicting the needs of decision-makers. In addition, epidemiological interpretation of outputs and communication had to be pursued under high time pressure and political and media attention. To meet the demand, SSI expanded significantly in the course of the epidemic. Onboarding new staff, coordinating across fast-growing departments and recruiting data scientists posed particular challenges. In 2017, SSI established DIAS, a multidisciplinary department encompassing specialists in microbiology, epidemiology, IT architecture, data science and engineering, project leadership, and communication to facilitate the needed crosscutting working environment for the development and management of these systems and enable in-depth collaboration with other departments at SSI.
Streamlining operation of the surveillance system
As the COVID-19 systems became more complex, incident- and change-management processes were introduced to further streamline maintenance, development, and communication with users. In addition, duty rosters were implemented. Since the start of the epidemic, the following teams with different capabilities have been on duty simultaneously to ensure the functioning of the system: incident management, data management, IT-infrastructure, and leadership. Since January 2023, the weekend duties have been discontinued, retaining a minimal run of the surveillance system to display only key figures. In April 2023, further downscaling took place.
Legal challenges
Data sharing and legal aspects also posed challenges. Despite guidance through the General Data Protection Regulation (GDPR) [28], various issues had to be evaluated on a case-by-case basis, and national legislation changed several times according to the assessed level of threat SARS-CoV-2 posed to society. This influenced the COVID-19 surveillance system, both in terms of the legal basis for receiving and sharing data as a health authority and in terms of the GDPR. For instance, outputs with personally identifiable information (PII) needed to be further aggregated or blacked out, when necessary. However, as information sharing with the public in the context of a high threat was important, SSI considered it proportionate on a case-by-case basis to show PII in selected outputs, for example showing the number of deaths in younger age groups or low vaccination coverage in a specific area and age group. Every time legislation changed, case-by-case judgements needed to be re-evaluated. An additional challenge was that authorities under the MoIH constitute separate legal entities with separate legal permissions for data sharing. This meant that data sharing between authorities under the MoIH was more complicated than sharing solely within SSI, as the interpretation of the Data Protection Act [29] – in terms of what is considered PII in aggregate data – differed between authorities. In addition, the legal basis for data sharing with certain stakeholders was not yet established for some authorities. For instance, SSI was not allowed to directly share certain outputs with municipalities. Consequently, data had to be sent via the DPSA, which had the required legal basis to share.
Limitations of existing data sources and IT infrastructure
Denmark has a long history of high-quality national registers that proved valuable during the COVID-19 epidemic. However, the epidemic also highlighted some gaps. For instance, there was no register of nursing homes and nursing home residents or schools and school pupils available for the health authorities. This required temporary solutions specifically developed for the surveillance of COVID-19. In case of a new threat, these solutions would need to be reinstated. Similarly, a register of residences for vulnerable groups, such as psychiatric patients, was lacking. Developing new registers for these groups would be beneficial to more effectively monitor infections in these populations in the future.
Varying timeliness between the different data sources posed another challenge. For instance, MiBa provides real-time data, whereas the National Patient Register (NPR; Table 2) – providing information on hospitalizations – has a delay of a few days varying by region. As this challenges timely monitoring of and responding to fast-spreading diseases, improving the timeliness of the NPR is recommended. During the COVID-19 epidemic, this current limitation was solved by setting up a new and temporary active reporting-system, the so-called ‘snapshot-data’ (Table 2). Through this solution, the Danish Regions reported twice daily. Also, the CRS (Table 2) – from which, among others, the date of death was extracted – is not updated on weekends. To address this limitation, an additional connection was established to the Cause of Death Register (CoDR) (Table 2), which records the date of death also on weekends. The dynamic nature of real-time data from MiBa was dealt with by agreeing to freeze data at set time points per day to ensure matching figures between outputs. Another challenge was a lack of server capacity. Upscaling SARS-CoV-2 surveillance was achieved by migrating servers at the expense of other surveillance systems that needed to be shut down at the time. Additional human and financial resources are needed to ensure a stable and flexible surveillance system capable of monitoring multiple threats concomitantly.
Aspects influencing the data
Data entering the surveillance system depended on the test strategy. At the same time, data from the surveillance system formed the basis for decision-making and developing the strategy. Due to this interdependence, data needed to be explained and interpreted with caution. For instance, a decrease in incidence did not necessarily reflect decreasing transmission, but a change in the test strategy or test seeking behaviour of the population influenced by information from the government or media. We also observed an interplay between data and behaviour in the effective reproductive number Rt (Table 4), which describes how many persons – in a population where some individuals are protected against the disease – an infectious person on average can infect altogether during their infectious period. Anecdotal evidence suggested that some people tended to use the Rt to judge whether it could be regarded as safe to attend public events.
The outputs from the surveillance system have been largely driven by ministerial requests. This often required navigating the interface between public health and politics, as the decision-making process during the epidemic response also took political, economic, and social interests into account. At times this resulted in challenges regarding prioritization of requests and development of meaningful algorithms and calculations.
Conclusions
To conclude, the COVID-19 epidemic constituted a pressure test that led to useful insights regarding the possibilities and limitations of the system, as well as valuable learning points for the future. It confirmed that future surveillance systems need to be (i) sufficiently agile for rapid up- and downscaling and responding to newly emerging health threats, (ii) require sustainable financial investments, and (iii) depend on the attraction and retention of highly specialized staff, including data science talent [30].
The strong digital data infrastructure allowed stakeholders to navigate the COVID-19 epidemic based on high-quality, transparent data. In line with the goal of the EU4Health Programme that supports EU Member States in the implementation of digitalised, integrated surveillance systems [31], several developments are ongoing to further improve and prepare for future threats, for example the integration of standardized conclusions from WGS into MiBa and algorithm-based interpretation of MiBa data to facilitate automated linkage with other data sources to achieve truly integrated surveillance.
This surveillance system constitutes a model for the future infectious disease surveillance system in Denmark, including surveillance of more than 80 infections and resistant microorganisms. Denmark is in the process of a digital transformation, which not only includes technical developments and targeted data outputs but also changes in workflows, responsibilities, and organization. This process will draw on experiences gained from the COVID-19 epidemic and will build on collaborations with authorities and other stakeholders established over the past years, as effective prevention and control of infectious diseases is dependent on strong intersectoral partnerships. With the current pandemic threat posed by avian influenza viruses and the global concern of antimicrobial resistance, lessons learnt from the COVID-19 pandemic as described here still remain topical and relevant [Reference Okeke32, 33].
Acknowledgements
The following members of the Statens Serum Institut COVID-19 Automated Surveillance Group contributed to building and maintaining the surveillance system and co-authored this paper (in alphabetical order with affiliations in superscript): Nour Abushalleeh1, Sophie Agger1, Niels Bindslev1, Karsten Dalsgaard Bjerre1, Palle Valentiner-Branth2, Manon Chaine1, Sisse Drotner1, Hanne-Dorthe Emborg2, Laura Espenhain2, Michael Galle1, Julie Grau2, Louise Hallundbæk2, Irfatha Irshad1, Lars Hervig Jacobsen1, Dora Kovacs1, Tyra Grove Krause3, Ida Suwalski1, Jonas Kähler1, Janni Lam1, Tomas Martin-Bertelsen1, Melea Menzinger1, Jacob Elskær Mollerup1, Ida Rask Moustsen-Helms2, Manisha Sammani Mudalige1, Frederik Trier Møller2, Jens Nielsen2, Lisbeth Nielsen1, Stefan Schytte Olsen1, Anne Provis3, Camilla Rasmussen1, Steen Hulthin Rasmussen1, Torben Rasmussen1, Signe Fischer Ravn1, Amalie Doctor Sejling1, Lotte Flink Sørensen1, Emily Dibba White2.
The authors would also like to thank and acknowledge the contribution of the following persons (in alphabetical order): Firstly we would like to thank our colleagues in the Department of Data Integration and Analysis (DIAS), who supported the development and maintenance of the automated surveillance system: Bettina Heinesen, Julie Håkansson, Dennis Karsten Kristensen, Claes Ottzen Laurentiussen, Jacob Lind, Lærke Lindholm, Martin Nguyen, Rikke Thoft Nielsen and Torben Møller Weide, as well as former DIAS colleagues: Michael Asger Andersen, Simon Bak, Ann-Sofie Nicole Berthelsen, Sara Birksø, Giulia Corn, Jørgen Vinsløv Hansen, Sofie Theisen Honoré, Julie Jakobsen, Marianne Karakis, Kjetil Erdogan Lavik, Marianna Meaidi, Finn Nielsen, Kenn Schultz Nielsen, Malene Nørregaard Nielsen, Anne Katrine Bjørkholt Sørensen, Nicklas Myrthue Thorsen, Malik Thrane, Pernille Westphall, Søren Steenstrup Zeeberg and Ditte Rechter Zenas. In addition, we would like to recognize the contributions of additional former and current colleagues from other departments at SSI: Nanna Bach Andersen, René Ravnsbæk Bode, Dalila Lepirica Brødbæk, Sissel Kristensen Buch, Mia Sarah Fischer Button, Lasse Engbo Christiansen, Arieh Cohen, Susan Cowan, Sofie Marie Edslev, Steen Ethelberg, Anders Fomsgaard, Jannik Fonager, Anna Cäcilia Ingham, Anders Jensen, Gitte Konieczny, Brian Kristensen, Rebecca Legarth, Cyril Jean-Marie Martel, Kåre Mølbak, Camilla Holten Møller, Luise Müller, Tam Nguyen, Flemming Platz, Morten Rasmussen, Pernille Sartvin, Raphael Sieber, Robert Leo Skov, Nina Steenhard, Marc Stegger, Bolette Søborg, Siri Tribler, Henrik Ullum, Lasse Skafte Vestergaard, Sidsel Skou Voss, Bartlomiej Wilkowski. We thank the representatives in the Data Coordination Group for the relevant discussions and thorough questions regarding our outputs: Jakob Schmidt Andersen, Susanne Lausten Brogaard, Birgitte Drewes, Lars Grunnet, Mads Hansen, Anne Hempel-Jørgensen, Dorte Vesterholm Lind, Karen Marie Lyng, Søren Mikael Neermark, Joanna Phermchai-Nielsen, Sebastian Pehn Slot, Dorte Hansen Thrige, Anna Sophie Tvermosegaard. The following persons from the Danish Health Data Authority are gratefully acknowledged for their support on accessing data sources and data management: Susanne Lausten Brogaard, Lucas Benjamin Christensen, Christoffer Grubb, Christopher Møller Hansen, Henrik Mulvad Hansen, Line Hansen, Ulrik Hesse, Lars Seidelin Knutsson, Anders Muldbjerg Kruse, Mette Lange, Marie Frankov Nissen, Mikkel Wermer Steen, Maria Svendsen and Sarah Aaby Toftlund. From the Danish Patient Safety Authority we want to thank Jane Christiansen, Britta Bjerrum Mortensen and Anders Welander-Haahr for the collaboration on data from contact tracing and outputs to the municipalities. From the Danish Health Authority we thank Frederik Løgstrup Magnusson and Emilie Sofie Esbirk for their collaboration on the surveillance of COVID-19 vaccination. Last but not least we would like to thank the Danish Clinical Microbiological Departments for their collaboration and data on the tests performed.
Author contribution
SG, MV and KLM conceptualised this work. GWF and SG drafted the original manuscript including tables. KLM analysed data and contributed to the collection of information listed in tables. MV and KLM revised the first draft. The Statens Serum Institut COVID-19 Automated Surveillance Group contributed to building and maintaining the surveillance system, the collection of information displayed in tables, critical reviewing and editing the manuscript. SA created Figure 1. All authors have reviewed and approved the final manuscript.
Funding statement
This work received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interest
The authors declare none.
Ethical standards
No ethical approval was required for this work, as surveillance is conducted in accordance with §222 of the Danish Health Act.








