Introduction
Population ageing has contributed to a rise in dementia cases, with an estimated 55 million people affected globally; a number projected to grow to 153 million by 2050. According to the Lancet Commission, approximately 45% of dementia cases are attributable to 12 modifiable risk factors (Livingston et al., Reference Livingston, Huntley, Liu, Costafreda, Selbæk, Alladi, Ames, Banerjee, Burns, Brayne, Fox, Ferri, Gitlin, Howard, Kales, Kivimäki, Larson, Nakasujja, Rockwood and Mukadam2024). Sleep disturbances are not included, as evidence remains inconclusive. The Commission proposes impaired sleep quality and circadian rhythm (CR) disturbances as potential causal factors (Livingston et al., Reference Livingston, Huntley, Liu, Costafreda, Selbæk, Alladi, Ames, Banerjee, Burns, Brayne, Fox, Ferri, Gitlin, Howard, Kales, Kivimäki, Larson, Nakasujja, Rockwood and Mukadam2024). The CR – a 24-hour cycle regulating processes such as sleep (Chauhan et al., Reference Chauhan, Norbury, Faßbender, Ettinger and Kumari2023) – is reflected in chronotype, often called an ‘early lark’ or a ‘night owl’. Chronotype is partially influenced by age, with individuals typically shifting to a later chronotype during adolescence, followed by a gradual return to an earlier chronotype with increasing age (Fischer et al., Reference Fischer, Lombardi, Marucci-Wellman and Roenneberg2017). By midlife, chronotype stabilises, though 19.9% remain evening types and 7.1% adapt extreme morning types (Roenneberg et al., Reference Roenneberg, Pilz, Zerbini and Winnebeck2019). Dim light melatonin onset is accurate but rarely used in large studies due to time, cost and participant burden (Kantermann et al., Reference Kantermann, Sung and Burgess2015; Roenneberg et al., Reference Roenneberg, Pilz, Zerbini and Winnebeck2019). As an alternative, self-report questionnaires such as the Munich ChronoType Questionnaire (MCTQ) are commonly used (Roenenberg et al., Reference Roenenberg, Keller, Fischer, Maera, Vetter and Winnebeck2015; Chauhan et al., Reference Chauhan, Norbury, Faßbender, Ettinger and Kumari2023). The MCTQ determines chronotype using sleep and wake times on workdays and free days by calculating Mid-point Sleep on Free days corrected for sleep debt on workdays (MSFSC) (Roenneberg et al., Reference Roenneberg, Kuehnle, Juda, Kantermann, Allebrandt, Gordijn and Merrow2007). Midpoint sleep time is a precise behavioural indicator of circadian phase and has been validated against biomarkers like DLMO (Kantermann et al., Reference Kantermann, Sung and Burgess2015). The MSFSC outcome from the MCTQ provides a continuous chronotype measure, ranging from extremely early (<12:00 am) to extremely late chronotypes (>9:00 am) (Juda, Reference Juda2010).
The pathways through which chronotype may influence dementia risk are not fully understood. Chronotype reflects individual variation in a CR, such as morningness versus eveningness. A disrupted CR has been associated with dementia pathology (Musiek et al., Reference Musiek, Xiong and Holtzman2015). Neurodegeneration within the suprachiasmatic nucleus (SCN) can disrupt circadian regulation (Musiek and Holtzman, Reference Musiek and Holtzman2016). Conversely, CR disruptions may impair memory and cognitive functioning through SCN neurodegeneration (Montaruli et al., Reference Montaruli, Castelli, Mulè, Scurati, Esposito, Galasso and Roveda2021), enhanced amyloid-β accumulation (C. Wang and Holtzman, Reference Wang and Holtzman2020) and increased oxidative stress from free radicals and reactive oxygen species (Wilking et al., Reference Wilking, Ndiaye, Mukhtar and Ahmad2013), all linked to elevated dementia risk. This suggests a bidirectional relationship between chronotype and dementia. CR disruption may arise from shift work (Boivin et al., Reference Boivin, Boudreau and Kosmadopoulos2022) or repeated jet lag (Vosko et al., Reference Vosko, Colwell and Avidan2010), thereby contributing to neurodegeneration (Moeller and Kriegsfeld, Reference Moeller, Kriegsfeld, Fonken and Nelson2023). Several studies have shown that a late chronotype is associated with negative health outcomes, independent of sleep debt or social jetlag (Didikoglu et al., Reference Didikoglu, Maharani, Payton, Pendleton and Canal2019; Partonen, Reference Partonen2023). Recently, variations in chronotype have been linked to cognitive functioning (Thapa et al., Reference Thapa, Kim, Yang, Park, Jang, Son, Kim and Park2020; Moeller and Kriegsfeld, Reference Moeller, Kriegsfeld, Fonken and Nelson2023), cognitive decline (Wenzler et al., Reference Wenzler, Liefbroer, Voshaar and Smidt2025) and dementia (Guo et al., Reference Guo, Harshfield and Markus2024). The only longitudinal study had respondents categorised as either early or late chronotype, without acknowledging intermediate chronotypes (Guo et al., Reference Guo, Harshfield and Markus2024). Additionally, the direction of the results was unclear. Many studies have relied on the Morningness-Eveningness Questionnaire (MEQ), which assesses a personality trait rather than the phase of entrainment (i.e., the timing of your biological clock) (Roenneberg, Reference Roenneberg2015). The limited evidence on this topic, combined with the Lancet Commission’s call for more research (Livingston et al., Reference Livingston, Huntley, Liu, Costafreda, Selbæk, Alladi, Ames, Banerjee, Burns, Brayne, Fox, Ferri, Gitlin, Howard, Kales, Kivimäki, Larson, Nakasujja, Rockwood and Mukadam2024), highlights the importance of examining chronotype as a potential risk factor for dementia.
The primary aim of this study is to investigate the association between chronotype and dementia among older adults in the general population. We hypothesise that a late chronotype is associated with an increased risk of dementia. Given the evidence that dementia risk is higher among women and individuals with lower educational attainment, and increases with age, moderation by age, sex and educational attainment will be examined.
Methods
Study design and population
For this study, we linked national register data on dementia healthcare claims to the Lifelines cohort study. Lifelines is a multi-disciplinary prospective population-based cohort study examining in a unique three-generation design the health and health-related behaviours of 167,729 persons living in the North of the Netherlands. It employs a broad range of investigative procedures in assessing the biomedical, socio-demographic, behavioural, physical and psychological factors which contribute to the health and disease of the general population, with a special focus on multi-morbidity and complex genetics, as extensively described elsewhere (Scholtens et al., Reference Scholtens, Smidt, Swertz, Bakker, Dotinga, Vonk, Van Dijk, Van Zon, Wijmenga, Wolffenbuttel and Stolk2015). All included participants signed informed consent. Dementia diagnoses were identified using healthcare claims from Vektis, a registry of all healthcare claims in the Netherlands. These data, along with mortality information, are available within the System of Social Statistical Databases (SSB) of Statistics Netherlands. Lifelines participants were securely linked to the SSB by Statistics Netherlands, enabling the integration of dementia data within Lifelines. All analyses were conducted within this secure environment. A study timeline is provided in Figure S1 Supplementary Material.
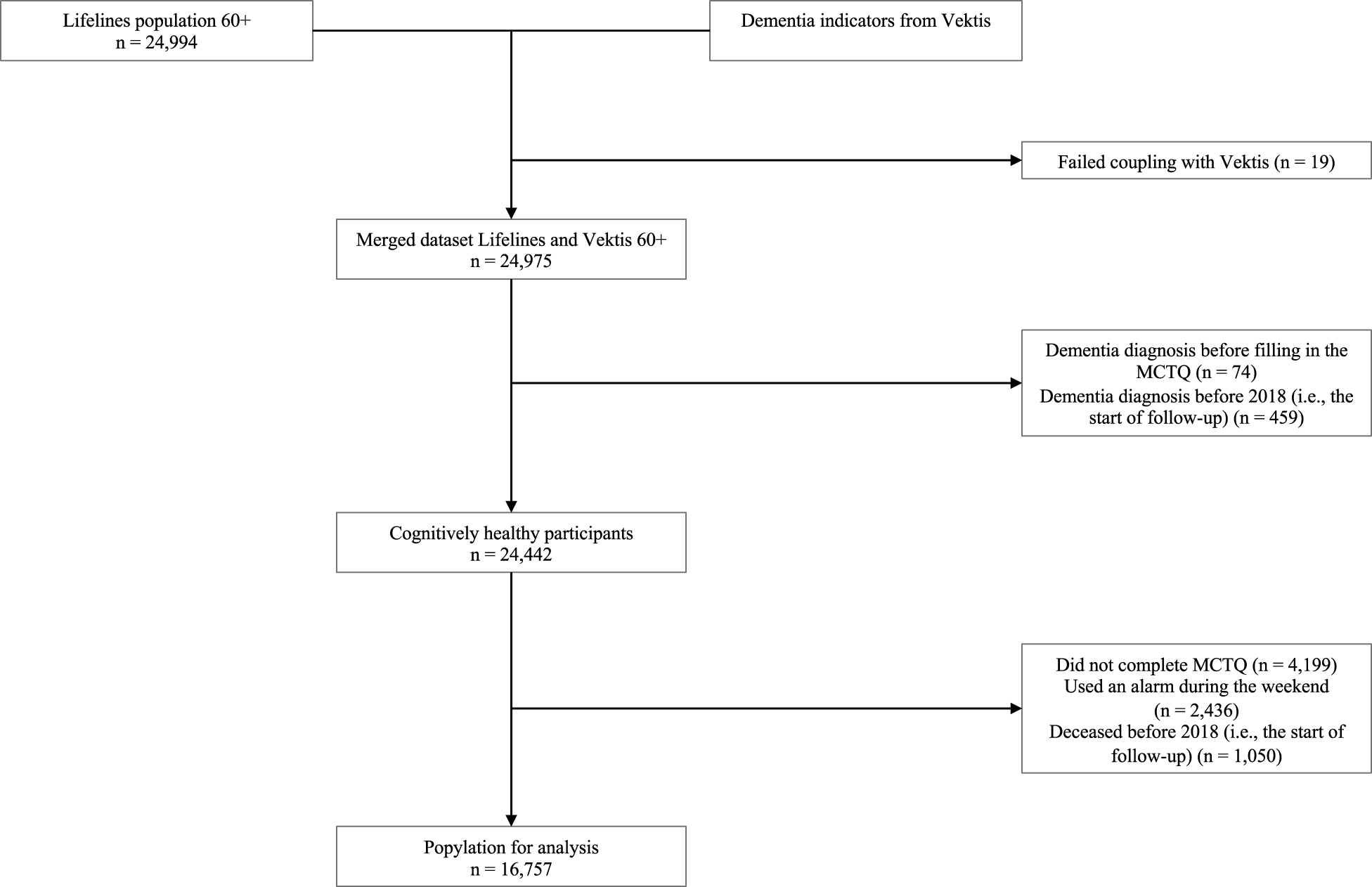
All Lifelines participants aged ≥60 years (n = 24,994) were linked to CBS data; after excluding 19 mismatches, 24,975 remained. Next, we excluded all participants who had developed dementia before 2018, based on information in Lifelines and data obtained from Vektis before 2018 (n = 533). This resulted in a cognitively healthy study sample of 24,442 participants. Participants without chronotype data (missing MCTQ, n = 4,199 or weekend alarm use, n = 2,436) or who died before 2018 (n = 1,050) were excluded, yielding a final sample of 16,757 (Fig. 1).
Flowchart of study participants.

Measurements
Dementia
Incident dementia defined using healthcare claims from Vektis, including (1) dementia-related medication, (2) dementia-related visits to neurology, geriatric or internal medicine clinics, (3) dementia-related mental healthcare, (4) community nursing for dementia and (5) Long-term Care Act (WLZ) eligibility assessments for dementia. The availability and completeness of these indicators have improved over time; since 2017, their combined use provides a realistic representation of dementia cases (van den Pol, Reference van den Pol2020) (Table S1 Supplementary Material), giving the opportunity to calculate the incidence of dementia from 2018 onwards. Although informative, these claims serve as an indirect proxy for dementia. Dementia was modelled as a time-varying dummy variable, coded 1 in the year the participant had ≥1 indicator and 0 in all preceding years.
Chronotype
Chronotype was assessed at baseline with the Dutch version of the MCTQ (Zavada et al., Reference Zavada, Gordijn, Beersma, Daan and Roenneberg2005). Chronotype was defined as the MSFSC. Adjusting for sleep debt avoids misclassifying individuals as having a stronger evening preference then they actually do (Roenneberg et al., Reference Roenneberg, Wirz-Justice and Merrow2003). Each 1-hour increase in mid-point of sleep reflects a 1-hour shift in chronotype, ranging from extremely early to extremely late. Participants who reported using an alarm clock during the weekend were excluded, as the MSFSC requires natural waking times. Because category cut-offs for the MSFSC can be arbitrary, chronotype was included as a continuous variable (Juda, Reference Juda2010). For categorical analysis, the following groups based on MSFsc cut-offs were used: extremely early (≤1:30), moderately early (>1:30 and ≤2:30), slightly early (>2:30 and ≤3:30), intermediate (>3:30 and ≤4:30), slightly late (>4:30 and 5:30), moderately late (>5:30 and ≤6:30) and extremely late (>6:30) (Roenneberg et al., Reference Roenneberg, Pilz, Zerbini and Winnebeck2019). Due to the small subgroup sizes, the extremely early (n = 57) and extremely late (n = 73) groups were combined with the moderately early or moderately late chronotype, respectively.
Confounders
Age was used as the underlying time scale in the discrete-time logistic survival analysis. Age at baseline and age at the end of follow-up were included, the latter defined as age at dementia diagnosis, death, age 95 years or last observation. Sex was included as a binary variable, female or male. Educational attainment was assessed with the question ‘What is the highest level of education you have completed?’ and categorised in three groups: low (junior general secondary education or lower), middle (secondary vocational education, work-based learning or higher general secondary education) and high (higher vocational education or university) educational attainment.
Statistical analysis
Descriptive statistics were calculated to describe baseline characteristics. Age was presented by median and interquartile range (IQR) because the variable was non-normally distributed. Sex, educational attainment and chronotype categories were presented as number and percentages (n, %).
A discrete-time survival model was estimated to examine the association between chronotype and dementia. The analysis was right censored at 95 years old, as the sample size above this age was too small to extract reliable estimates. Model 1 assessed the underlying hazard of age on dementia risk, including a quadratic term to account for potential non-linearity. Model 2 examined the linear association between chronotype and dementia risk, additionally adjusting for sex. If no linear association was observed, a spline model was used to explore non-linearity. When non-linearity was indicated, chronotype was categorised. Exact p-values were presented, with a p < 0.05 deemed statistically significant. Effect modification by age, sex and educational attainment was assessed using log-likelihood ratio tests (LRT) comparing models with and without the interaction terms. Because the MCTQ was administered between 2011 and 2015, the time to the first possible dementia diagnosis in 2018 varied across participants. This interval, referred to as lag time, was included as a covariate in all analysis (Figure 1, Supplementary Material).
As a sensitivity analysis, chronotype was re-categorised into three groups: early (≤3:30), intermediate (>3:30 & ≤4:30) and late (>4:30), to provide a simplified representation of its association with dementia risk. The model used the intermediate chronotype as the reference and was adjusted for sex and lag time. In a second sensitivity analysis, there was examined whether sleep duration explained the association between chronotype and dementia. Sleep duration was included as a continuous variable, measured in hours using the MCTQ.
Statistical analyses were performed in R, version 4.3.1. The discSurv package was used for the discrete time analysis.
Results
Table 1 presents the characteristics of the study sample (n = 16,757). Participants had a median age of 65 years (IQR, 62–69), and 54% of whom were women. The mean follow-up time was 6.6 years, during which 7% developed dementia and 9% died. Most participants had an intermediate chronotype (56%), followed by slightly early (20%) and slightly late (19%) chronotypes. Extremely early (2%) and late (3%) chronotypes were rare. Over half of the participants had low educational attainment (51%) while the middle and high groups were equally represented (23%).
Population characteristics

Model 1 assessed whether age was associated with dementia risk and showed a curve-linear relationship (Fig. 2). Dementia risk increased with age, while the small but statistically significant quadratic term indicated a slight decrease in hazard over time. Lag time, ranging from 3 to 7 years, had no effect on this association (Table S2 Supplementary Material).
Predicted hazard of dementia by age.

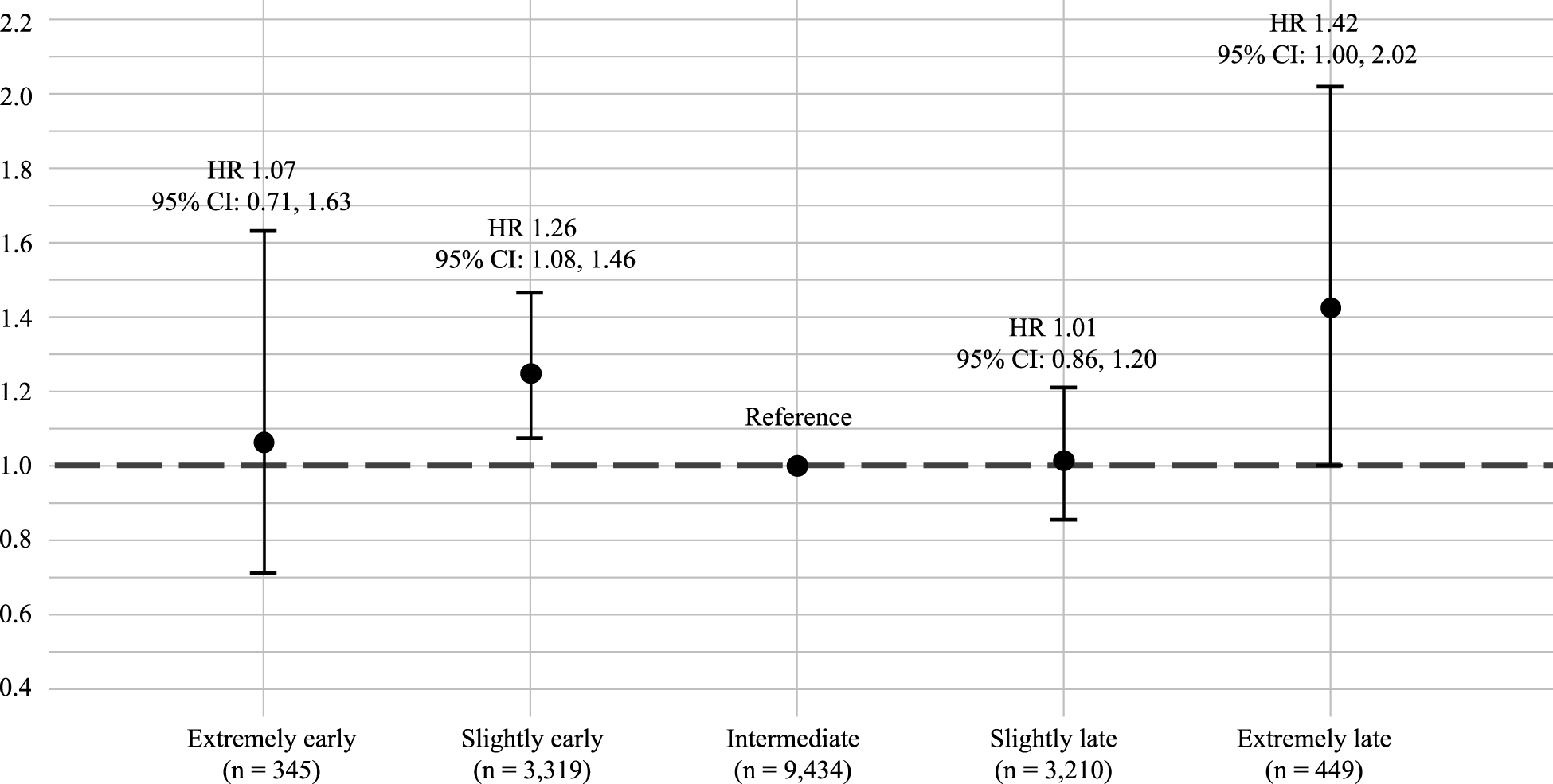
Model 2 examined the association between chronotype and dementia risk. No linear association was found between continuous chronotype (MSFSC) and dementia (HR, 0.95 [95% CI: 0.88, 1.03]) (Table S3, Supplementary Material). Sex and lag time were not associated with dementia risk. The distribution of the MSFSC is presented in Figure S2 Supplementary Material. A spline model suggested some curvature, although the non-linear term was not significant (effective degrees of freedom 1.84, p = 0.43). Chronotype was, therefore, categorised into five groups. A log-likelihood ratio test (LRT) showed improved model fit when adding chronotype on top of age (p = 0.02). Compared to an intermediate chronotype, an extremely late chronotype was associated with a 42% increased hazard of dementia (HR, 1.42 [95% CI: 1.00, 2.02]) and a slightly early chronotype with a 26% higher hazard of dementia (HR, 1.26 [95% CI: 1.08, 1.46]) (Fig. 3). Extremely early and slightly late chronotypes were not associated with increased dementia risk (Table S4 Supplementary Material).
Risk of dementia (hazard ratio [HR]) by chronotype during average 6.6 years of follow-up.

Interaction analyses showed no significant effect modification by age (p-value LRT-test 0.89), educational attainment (p-value LRT-test 0.75) or sex (p-value LRT-test 0.41) (Tables S5–S7 Supplementary Material).
The first sensitivity analysis using three groups of chronotype showed that an early chronotype was associated with a 24% higher risk to develop dementia compared to an intermediate chronotype (HR, 1.24 [95% CI: 1.07, 1.43]) (Table S8 Supplementary Material). The second sensitivity analysis showed that after adjustment of sleep duration the associations between chronotype and dementia remain the same (Table S9 Supplementary Material).
Discussion
This prospective cohort study found that individuals aged 60 years and older with a slightly early (i.e., midpoint sleep >2:30 and ≤3:30), and an extremely late chronotype (i.e., midpoint sleep >5:30) had a higher risk of dementia than those with an intermediate chronotype. These associations were not moderated by age, sex or educational attainment.
Current evidence chronotype and dementia
Individuals aged 60 years and older with an extremely late chronotype had a 42% higher risk of dementia compared to those with an intermediate chronotype in our study. However, the confidence interval was wide, limiting the precision and making it difficult to draw firm conclusions. Previous studies examining chronotype in relation to dementia risk or cognitive decline mostly conceptualised chronotype as a personality trait (i.e., using the Morningness-Eveningness Questionnaire (MEQ), which aims to identify morning and evening persons (Horne and Ostberg, Reference Horne and Ostberg1976)), rather than a phase of entrainment measured with the MCTQ (Roenneberg, Reference Roenneberg2015). As the MEQ and MCTQ have different aims, they are not interchangeable (Roenneberg, Reference Roenneberg2015). Previous studies have linked chronotype to cognitive outcomes (Ahn et al., Reference Ahn, Yoon and Park2024; Cox et al., Reference Cox, Ritchie, Allerhand, Hagenaars, Radakovic, Breen, Davies, Riha, Harris, Starr and Deary2019; Thapa et al., Reference Thapa, Kim, Yang, Park, Jang, Son, Kim and Park2020; C. Wang and Holtzman, Reference Wang and Holtzman2020; Wenzler et al., Reference Wenzler, Liefbroer, Voshaar and Smidt2025). For example, being an evening person based on the MEQ was cross-sectionally associated with dementia risk based on the Mini-Mental State Examination (MMSE) score (Thapa et al., Reference Thapa, Kim, Yang, Park, Jang, Son, Kim and Park2020). Longitudinal studies using the MCTQ have shown that a later chronotype is associated with greater decline in executive functioning (Wenzler et al., Reference Wenzler, Liefbroer, Voshaar and Smidt2025) and increased subjective memory complains (Ahn et al., Reference Ahn, Yoon and Park2024), although other studies, including those incorporating genetic measures of chronotype, reported no association (Cox et al., Reference Cox, Ritchie, Allerhand, Hagenaars, Radakovic, Breen, Davies, Riha, Harris, Starr and Deary2019; Q. Wang et al., Reference Wang, Xu, Liu, Liu, Chen, Huang, Xu and Liu2023). In addition to the late chronotype findings, our results also suggest a potential risk among individuals with a (slightly) early chronotype. While most studies only report adverse effects for late chronotypes, some also indicate the negative cognitive outcomes for early chronotypes. Guo et al., using UK Biobank data, examined the association between chronotype and dementia, using Mendelian randomisation. They found no association between the chronotype based on PRS and dementia, but chronotype based on MCTQ was associated with all-cause dementia (Guo et al., Reference Guo, Harshfield and Markus2024). In this study chronotype was dichotomised, potentially obscuring effects in extreme groups because individuals with intermediate chronotypes were divided between categories. Our study used a more detailed categorisation of chronotype, allowing detection of effects in smaller subgroups. In line with our findings, Kim et al., also found that an earlier chronotype measured with the MCTQ, was associated with faster temporal lobe atrophy and poorer verbal learning and visual memory compared to an intermediate chronotype (Kim et al., Reference Kim, Kim, Kim, Lee, Lee and Shin2023). Lastly, Suh et al., reported a protective effect of a late chronotype for global cognitive decline and a higher, though non-significant, risk among early chronotypes (Suh et al., Reference Suh, Han, Lee, Byun, Kwon, Oh, Lee, Han, Hong, Kwak, Kim, Kim, Kim, Kim, Ryu, Moon, Park, Seo, Youn and Kim2018). Although some evidence supports associations between chronotype and cognitive outcomes, future studies should clearly distinguish between chronotype as a personality trait and as a phase of entrainment (i.e., the timing of your biological clock), as these have different clinical implications (Roenneberg, Reference Roenneberg2015). Further research should also examine the biological and behavioural mechanisms underlying these associations.
Late chronotype and risk of dementia
The association between a late chronotype and increased dementia risk may be partially explained by biological mechanisms related to CR disruption. With ageing, the amplitude of rhythmic gene expression by the circadian clock is reduced, affecting cortisol, melatonin and body temperature regulation. At the same time, there is a gradual loss of brain cells, including those in the SCN the brains primary pacemaker (Ghorbani Shirkouhi et al., Reference Ghorbani Shirkouhi, Karimi, Khatami, Asgari Gashtrodkhani, Kamari, Blaabjerg and Andalib2025). Declines in gene expression and SCN cell volume can cause subtle circadian misalignment, which may impair protein expression (Verma et al., Reference Verma, Singh and Rizvi2023), weaken defences against oxidative stress (Verma et al., Reference Verma, Singh and Rizvi2023), and compromise blood–brain barrier function (Schindler et al., Reference Schindler, Torices, Schurhoff, Gallo and Toborek2025). Minor circadian disruptions can reduce process effectiveness, leaving the brain more vulnerable. Collectively, these changes increase susceptibility to cognitive dysfunction and neurodegenerative diseases. In addition to biological mechanisms, behavioural factors associated with chronotype may contribute to dementia risk. Although older adults might experience less social jetlag due to more flexible schedules (Roenneberg et al., Reference Roenneberg, Pilz, Zerbini and Winnebeck2019), late chronotypes may have been exposed to prolonged circadian misalignment during midlife. Social jetlag occurs when fixed schedules force individuals to wake before obtaining sufficient sleep. People experiencing social jetlag are more likely to consume alcohol, smoke and have a higher body mass index, all established risk factors for cognitive decline and dementia (Livingston et al., Reference Livingston, Huntley, Liu, Costafreda, Selbæk, Alladi, Ames, Banerjee, Burns, Brayne, Fox, Ferri, Gitlin, Howard, Kales, Kivimäki, Larson, Nakasujja, Rockwood and Mukadam2024). One study found that sleep quality and smoking partially mediated the relationship between a late chronotype and cognitive decline (Wenzler et al., Reference Wenzler, Liefbroer, Voshaar and Smidt2025). Moreover, even when sleep duration is similar, late chronotypes report higher subjective sleepiness (Zou et al., Reference Zou, Zhou, Yan, Yao and Lu2022), which may prompt compensatory behaviours that negatively impact long-term health. Together, the combined impact of circadian misalignment, lifestyle factors and increased vulnerability may increase dementia risk.
Early chronotype and risk of dementia
Although late chronotypes are typically associated with adverse health outcomes, our findings suggest that a slightly early chronotype may also be linked to increased dementia risk. Several pathways may explain this unexpected association. Participants with a slightly early chronotype had a sleep midpoint between 2:30 and 3:30 am, corresponding to a sleep window of approximately 22:30–23:30 to 6:30–7:30, assuming 8 hours of sleep. Although this schedule is not extremely early, evening social events such as sports classes, parties or dinners may conflict with their preferred sleep rhythm. Consequently, they may either skip such events, limiting social contacts or attend them at the cost of a reduction in either sleep duration or quality (acknowledging that sensitivity analyses found no association between sleep duration and dementia risk). Another factor may be hearing loss. Gao et al., found a suggestive association between morning chronotype and hearing loss (Gao et al., Reference Gao, Qiu and Lu2024), which is itself a risk factor for dementia. Individuals with an extreme early chronotype were not at higher risk compared with intermediate chronotypes. However, the small sample size (2%) and wide confidence interval indicate limited precision, meaning a true association cannot be excluded. When all early chronotypes were combined in a sensitivity analysis, early chronotype was associated with higher dementia risk, likely due to increased statistical power after merging categories. Lifestyle differences may also play a role. Individuals with a slightly early chronotype may have adjusted their schedule to societal norms, whereas extremely early chronotypes may have aligned their life more closely with their biological clock, reducing negative indirect effects. Genetic differences may contribute as well. Polymorphisms in chronotype-related genes have been linked to psychiatric disease risk (Zou et al., Reference Zou, Zhou, Yan, Yao and Lu2022). Research investigating these pathways is limited. Future studies could investigate both modifiable and non-modifiable risk factors to better understand how chronotype influences dementia risk.
Operationalisation and categorisation of chronotype
In the current study, only 21% of the individuals had a late chronotype, with just 3% having an extremely late chronotype. This small proportion limits the health implications in older adults. By contrast, the MCTQ database with 300,000 entries worldwide, including the Netherlands, shows about 40% of individuals having a late chronotype, with 20% classified as moderately or extreme late (Roenneberg et al., Reference Roenneberg, Pilz, Zerbini and Winnebeck2019). Our sample had less individuals in the extreme, moderate and slightly early categories, and roughly twice the proportion of intermediate chronotypes. This discrepancy raises the question of whether older adults are underrepresented in early and late chronotypes, or if traditional cut-offs are unsuitable for ageing populations. Chronotypes shift earlier with age due to SCN neuron loss, reduced rhythmic gene expression and declining sleep pressure, making late types less common and current categories less representative (Hood and Amir, Reference Hood and Amir2017). Refining chronotype categories for age-specific distributions could improve study accuracy and clarify what constitutes morning or evening types in later life. Another challenge is the lack of consensus on categorising chronotype. Some studies use only early versus late groups, splitting intermediates who often have the lowest health risk (Thapa et al., Reference Thapa, Kim, Yang, Park, Jang, Son, Kim and Park2020; Guo et al., Reference Guo, Harshfield and Markus2024). Others use sample-specific cut-offs, such as median plus or minus one standard deviation. Our findings show intermediate chronotypes had the lowest dementia risk, whereas slightly early or extreme late chronotypes were at higher risk. These inconsistencies highlight the need for validated and standardised chronotype measures in health research.
Strengths and limitations
The current study has several strengths. Chronotype was measured using the validated MCTQ, facilitating the comparison with other studies. The large sample size allowed a detailed categorisation of chronotype. Additionally, there was no loss to follow-up, as all participants who completed the MCTQ at baseline were tracked until the study’s end in 2024 using registry data. Only cognitively healthy individuals at baseline were included as participants diagnosed with dementia before baseline or during the 3–7 years between baseline and start follow-up were excluded. These strengths enhance the study’s validity, though several limitations must be acknowledged. The registry data are based on healthcare declarations, which serve as proxies rather than formal clinical dementia diagnoses. Some indicators, such as dementia-specific medication, strongly suggest a diagnosis, whereas others, including referrals to memory clinics or internal medicine for dementia-related symptoms, may also capture individuals without dementia. Follow-up was relatively short because dementia indicators from Vektis were only reliably available from 2017 onwards, resulting in a maximum of 7 years. Moreover, lag time varied between individuals, although adjusted for in the analysis. Finally, despite the large sample size, the number of participants with extreme chronotypes was limited, requiring merging of extreme early or late chronotypes with moderately early or late groups, which may reduce granularity for the extremes.
Conclusion
To conclude, individuals aged 60 years and older with slightly early or extremely late chronotypes were at higher risk for dementia over 6.6 years. Chronotype can be assessed with short questionnaires and may serve as a low-burden risk marker complementing existing assessments. Future studies should employ tools that capture more extreme chronotypes beyond the simple early-late dichotomy, investigate whether modifiable risk factors mediate the association and replicate findings in independent cohorts. Longitudinal studies with longer follow-up (≈30 years) are needed to explore the impact of midlife chronotype on late-life dementia. Elucidating on these pathways could position chronotype as an entry point for personalised preventive strategies.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S2045796026100687.
Availability of data and materials
Data are not publicly available and may be obtained from third parties. Researchers can apply to access the lifelines data used in this study (project number OV23_00871) via http://www.lifelines-biobank.com. Dementia indicator and mortality data can be requested from the System of Social Statistical Databases (SSB) of Statistics Netherlands.
Acknowledgements
The Lifelines initiative has been made possible by subsidy from the Dutch Ministry of Health, Welfare and Sport, the Dutch Ministry of Economic Affairs, the University Medical Center Groningen (UMCG), Groningen University and the Provinces in the North of the Netherlands (Drenthe, Friesland, Groningen). Lifelines had no role in the conceptualisation, design, analysis, decision to publish or preparation of the manuscript.
Financial support
This work is part of the BIRD-NL consortium funded by the Dutch Medical Research Council (ZonMw) as part of the National Dementia Strategy 2021–2030 by the Dutch Ministry of Health, Welfare and Sport (grant number: 1051003210005). Open access funding provided by University of Groningen and University Medical Center Groningen.
Competing interests
We declare no conflict of interest or competing interest.
Ethical standards
The Lifelines Cohort is conducted in line with the Declaration of Helsinki and received approval by the Medical Ethics Committee of the University Medical Centre Groningen (approval number: 2007/152).