In October 2024, our Clinical Microbiology Lab informed Infection Preventionists that Burkholderia contaminans had been isolated in breast biopsy specimens from two recent patients. B. contaminans is a glucose non-fermenting Gram negative bacillus within the Burkholderia cepacia complex. Reference Nguyen, Phuong Linh and Huong1 It is commonly found in the environment and has an innate ability to survive and grow in water with only minimal nutritional sources. Reference Mangram and Jarvis2 Species within B. cepacia complex are opportunistic pathogens with intrinsic resistance to some preservatives, antiseptics, and antimicrobial agents. Reference Hudson, Park and Mathers3 Outbreaks with B. cepacia complex have been linked to medical devices and contaminated liquids such as medications, saline, and multiuse ultrasound gel. Reference Hudson, Park and Mathers3,Reference Eldridge, Flinchum, Thoroughman and Spicer4 With the availability of new technologies such as matrix-assisted laser desorption/ionization—time of flight (MALDI-TOF, bioMerieux), microbiology labs are now better able to identify these members of B. cepacia complex to the species level.
An initial investigation by Infection Prevention staff revealed that cultures from two additional patients who had breast biopsies collected the previous month were also positive for B. contaminans. All four patients had undergone breast biopsy procedures at the same hospital-based clinic. Different clinicians performed the biopsies. During the investigation, two additional patients grew B. contaminans from breast biopsies obtained in the same clinic, giving a total of six positive patients between September 2024 and November 2024.
Patient histories and procedural details were obtained via chart review. Breast Imaging Clinic leadership provided detailed accounts of the biopsy procedure methods. Inquiries about disinfection of equipment, multi-use vials, and recent changes in processes or supplies were discussed. Infection Prevention staff toured the procedural area of the clinic to observe infection prevention practices. No violations were observed.
Twenty-nine items, including contents of an unopened ultrasound biopsy procedure tray, tubes of non-sterile ultra-sound gel, vials of topical anesthetics, bagged IV saline, and individual wipes, were obtained from the clinic for surveillance cultures. In addition, a screw-capped tube containing 0.9% saline, which was being used to submit breast biopsy specimens for a research study, was cultured. These tubes were prepared and hand-labeled by research lab personnel.
Burkholderia cepacia Selective Agar (BCSA, Hardy Diagnostics) was used to perform the surveillance cultures. Some items were plated directly onto the agar, while others were sampled using a moistened sterile swab. Larger items were washed in sterile saline, then the solution was vacuum filtered through a sterile funnel onto a cellulose nitrate filter (Fisher Scientific), which was then placed on the agar surface. Larger volumes of liquids were directly vacuum filtered in the same fashion. Items containing disinfectants were placed in Dey-Engley Neutralizing Broth (Remel) for 15 minutes to neutralize the disinfectant prior to filtering and plating. All BCSA plates were incubated at 35°C ± 1°C for 72 hours.
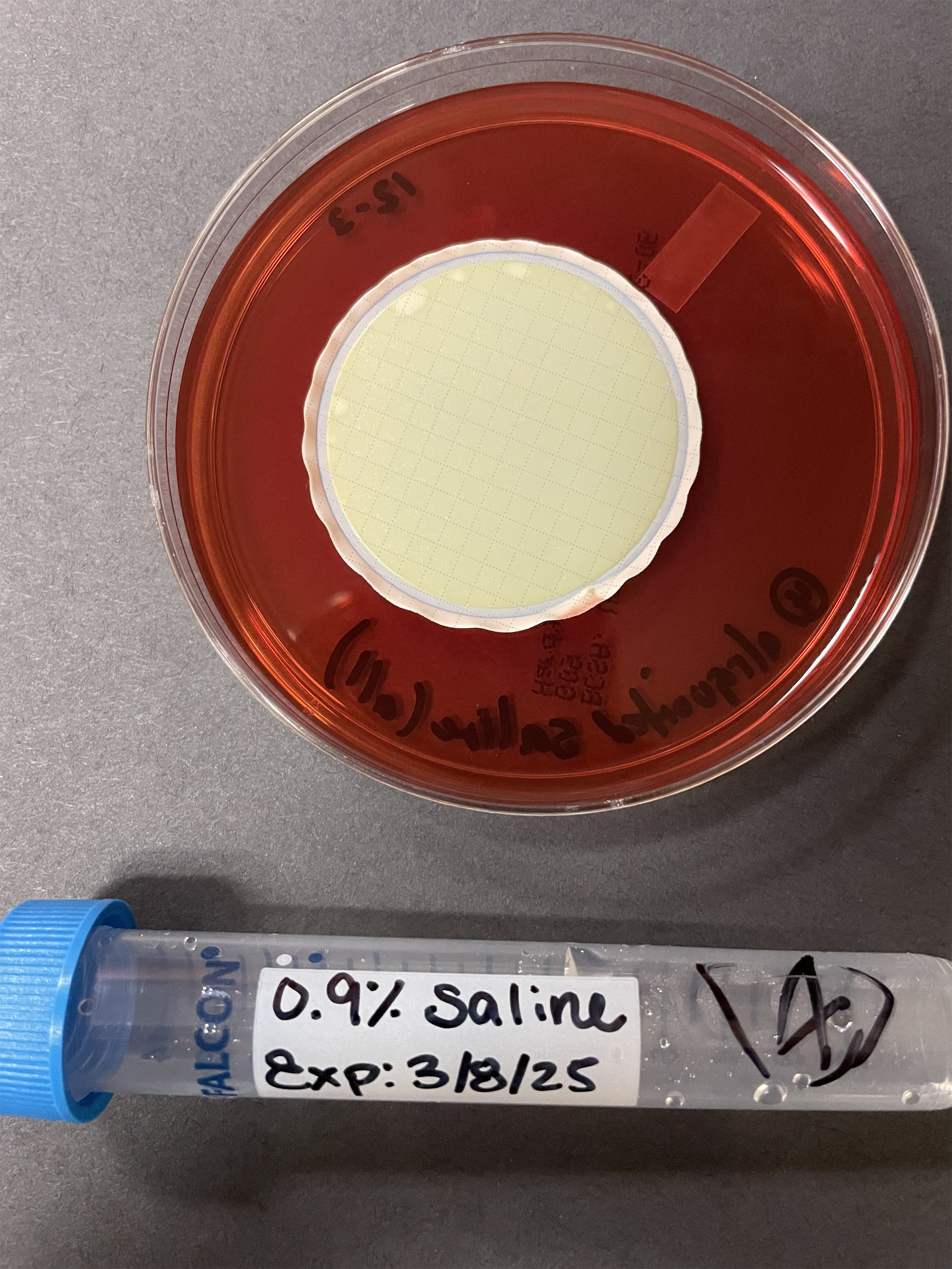
Of the 29 items cultured, only one culture was positive. Growth was seen on the filtered sample of 0.9% saline taken from the screw-capped research tube (see Figure 1). Testing performed by MALDI-TOF identified the isolate as B. contaminans with 99.9% accuracy.
One of the research saline tubes found to be the apparent source of the pseudo-outbreak, along with its corresponding positive culture plate growing Burkholderia contaminans.

Upon detection of bacteria in the research saline tube, seven remaining research saline tubes were immediately removed from the clinic’s procedural areas for further testing. Aliquots of saline from each of these tubes were plated to BCSA agar. All seven cultures were positive, with growth that was identical to the morphology seen from the original positive tube.
Culture results were shared with the clinic providers and leadership. Providers stated that they thought the saline tubes were available for general clinical use, not just for the research study. Biopsy specimens were being placed into these tubes and submitted to the Clinical Microbiology Lab for culture. Microbiology staff did not have a reason to consider these specimens unacceptable for culture since the screw-capped tubes looked identical to the sterile tubes stocked in their lab and since each specimen tube was clearly labeled as containing “0.9% saline.” The research tubes in question were aliquoted by research lab staff, not commercially obtained. Details about how these tubes had been made were not available, but the contents were presumed to be sterile. These tubes were to be used for research specimens only, not to submit biopsy specimens for clinical cultures. Failure to clearly follow research protocol resulted in biopsy specimens being contaminated with B. contaminans after collection.
After the apparent source of B. contaminans was identified, a new batch of research saline tubes was obtained. Research tubes are now being stored in the clinic supervisor’s office to prevent inappropriate use. Each research saline tube is now clearly labeled as “research use only” to avoid confusion. Newly purchased sterile saline tubes for biopsy cultures are now being stocked in the procedural rooms. Education was sent to all Breast Imaging staff and providers regarding proper usage of both types of saline tubes. No additional patients with positive breast cultures for B. contaminans were found once the contaminated saline tubes were identified and removed.
Pseudo-outbreaks, outbreaks not causing infections, can increase the risk of patient harm and lead to unnecessary treatments and procedures. Reference Eldridge, Flinchum, Thoroughman and Spicer4 In this pseudo-outbreak, each of the six patients were placed on antibiotics after B. contaminans was reported in their biopsy specimens and prior to the discovery of the contaminated saline tubes. Delayed notification when unusual isolate patterns are detected can result in unnecessary antibiotic treatment for the patients involved. This pseudo-outbreak highlights the importance of timely trend recognition and notification of Infection Prevention by the Clinical Microbiology Lab.
Acknowledgements
None.
Financial support
None received.
Competing interests
None.