Introduction
The Southern Ocean (SO) remains one of the most unexplored regions on the globe due to its remote location and sampling difficulty. The geological and hydrographic isolation, a persistent cold climate and intense seasonal primary production experienced on the Antarctic shelf are responsible for a high level of endemism in this ocean (Clarke & Johnston Reference Clarke and Johnston2003). Of the estimated global number of gastropod species (~63 000; Bouchet et al. Reference Bouchet, Rocroi, Hausdorf, Kaim, Kano and Nützel2017), ~600 species of shelled gastropods have been described from the SO to date (Clarke et al. Reference Clarke, Griffiths, Linse, Barnes and Crame2007), 75% of them endemic to this ocean (Linse et al. Reference Linse, Griffiths, Barnes and Clarke2006). Yet, recent studies have revealed even greater hidden diversity and a high incidence of cryptic speciation in Antarctic marine gastropods (Wilson et al. Reference Wilson, Schrödl and Halanych2009, Fassio et al. Reference Fassio, Modica, Alvaro, Buge, Salvi, Oliverio and Schiaparelli2019, Moles et al. Reference Moles, Avila and Malaquias2019).
Among gastropods, cephalaspideans (commonly known as head-shield snails) can be found in marine ecosystems worldwide, ranging from shallow to abyssal waters (e.g. Schrödl et al. Reference Schrödl, Bohn, Brenke, Rolán and Schwabe2011, Ohnheiser & Malaquias Reference Ohnheiser and Malaquias2013, Moles et al. Reference Moles, Avila and Malaquias2017, Chaban et al. Reference Chaban, Ekimova, Lubin, Nikitenko and Schepetov2023). Philinoid snails, one of the most diverse groups of cephalaspideans, have been intensively studied in recent years due to the high misidentification of taxa and new possibilities for clarifying current diversity with molecular tools (e.g. Oskars et al. Reference Oskars, Bouchet and Malaquias2015, Moles et al. Reference Moles, Avila and Malaquias2017, Chaban et al. Reference Chaban, Ekimova, Schepetov, Kohnert, Schrödl and Chernyshev2019). Originally classified within the genus Philine Ascanius, 1772, philinoids in the SO have now been encompassed in three recently erected genera: Spiraphiline Moles, Avila & Malaquias, Reference Moles, Avila and Malaquias2019 from the family Philinidae Gray, 1850 (1815); Waegelea Moles, Avila & Malaquias, Reference Moles, Avila and Malaquias2019; and Antarctophiline Chaban, Reference Chaban2016 from the family Antarctophilinidae Moles, Avila & Malaquias, Reference Moles, Avila and Malaquias2019.
Antarctophiline was recently established by Chaban (Reference Chaban2016) based on morphological differences in the male reproductive and digestive systems (i.e. gizzard plates). They also evaluated the species Antarctophiline gibba (Strebel, Reference Strebel1908) and Antarctophiline alata (Thiele, Reference Thiele1912) and suggested Antarctophiline amoena (Thiele, Reference Thiele1925) as a synonym of A. alata, a finding later corroborated based on molecular data and more comprehensive taxon sampling by Moles et al. (Reference Moles, Avila and Malaquias2019). Among the six Antarctophiline species reinstated in the latter study, five are distributed solely in shallow water, while one is also found in deeper waters. A. alata has a circumpolar distribution and can be found in numerous locations such as the Davis Sea, Adélie Land in East Antarctica, South Orkney Islands, South Sandwich Islands, Palmer Archipelago in the western Antarctic Peninsula, South Shetland Islands, Peter I Island, Bellingshausen Sea, eastern Weddell Sea and Bouvet Island (Thiele Reference Thiele1912, Powell Reference Powell1951, Vicente & Arnaud Reference Vicente and Arnaud1974, Hain Reference Hain1990, Troncoso et al. Reference Troncoso, García, Backeljau and Urgorri1996, Aldea & Troncoso Reference Aldea and Troncoso2008, Engl Reference Engl2012, Moles et al. Reference Moles, Avila and Malaquias2019, Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021). A. gibba (Strebel, Reference Strebel1908) is known from the Argentinian Sea in the South Atlantic, as well as the South Orkney Islands, South Georgia, King George Island, South Shetland Islands, McMurdo Sound and the Ross Sea (Strebel Reference Strebel1908, Odhner Reference Odhner1926, Powell Reference Powell1951, Marcus & Marcus Reference Marcus and Marcus1969, Rudman Reference Rudman1972, Seager Reference Seager1978, Reference Seager1982, Zelaya Reference Zelaya2005, Moles et al. Reference Moles, Avila and Malaquias2019). Antarctophiline falklandica (Powell, Reference Powell1951) is exclusive to the Falkland Islands and the Ross Sea, while Antarctophiline apertissima (E.A. Smith, Reference Smith1902) primarily occupies Cape Adare within the Ross Sea region. Antarctophiline easmithi Moles, Avila & Malaquias, Reference Moles, Avila and Malaquias2019 is confined to the Eastern Weddell Sea, while Antarctophiline amundseni Moles, Avila & Malaquias, Reference Moles, Avila and Malaquias2019 is also restricted to the eastern Weddell Sea but is found in deeper waters.
In this study, we had access to additional material from the South Shetland Islands, spanning deep waters from the South Atlantic Antarctic Ridge to Bouvet Island, as well as from the South Sandwich Islands and Bransfield Strait, at depths ranging from 134 to 4548 m. These specimens were recently sequenced, and the identities of two new species were confirmed (Moles et al. Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021). Here, we explore the species identification employing a fragment of the cytochrome c oxidase subunit I (COI) gene and by applying multiple species delimitation tests. Additionally, we examine the morphology and anatomy of these specimens in a comparative framework, providing detailed taxonomical descriptions of the two newly discovered species of Antarctophiline. Furthermore, we describe the complete reproductive, digestive and excretory systems based on multiple specimens of A. alata from its distributional range.
Materials and methods
Sampling strategy
Specimens were collected aboard the German R/V Polarstern during the expeditions ANT-XIX (2002), ANT-XXIV/2 (2007–2008) and ANT-XXVIII/3 (2012), spanning from the South Shetland Islands across the South Atlantic Antarctic Ridge to Bouvet Island. Additional material was gathered aboard the US R/V Nathaniel B. Palmer in 2011 (NBP11-05) in the vicinity of the South Sandwich Islands and Bransfield Strait. Samples were collected using the Agassiz trawl, Blake trawl and epibenthic sledge methods. On board, specimens were sorted by morphotypes, fixed in 70% or 96% EtOH and deposited in the SNSB-Zoologische Staatssammlung München (ZSM) and the Scripps Institution of Oceanography (SIO-BIC).
Molecular data treatment
All analysed specimens were sequenced in Moles et al. (2020) either using ddRADseq or by Sanger sequencing of the COI marker. All COI sequences were obtained from GenBank and aligned using MAFFT (Katoh & Standley Reference Katoh and Standley2013) under the G-INS-I algorithm implemented in Geneious Prime® v2019.2.3. Alignment ends were trimmed to remove primer overlapping regions.
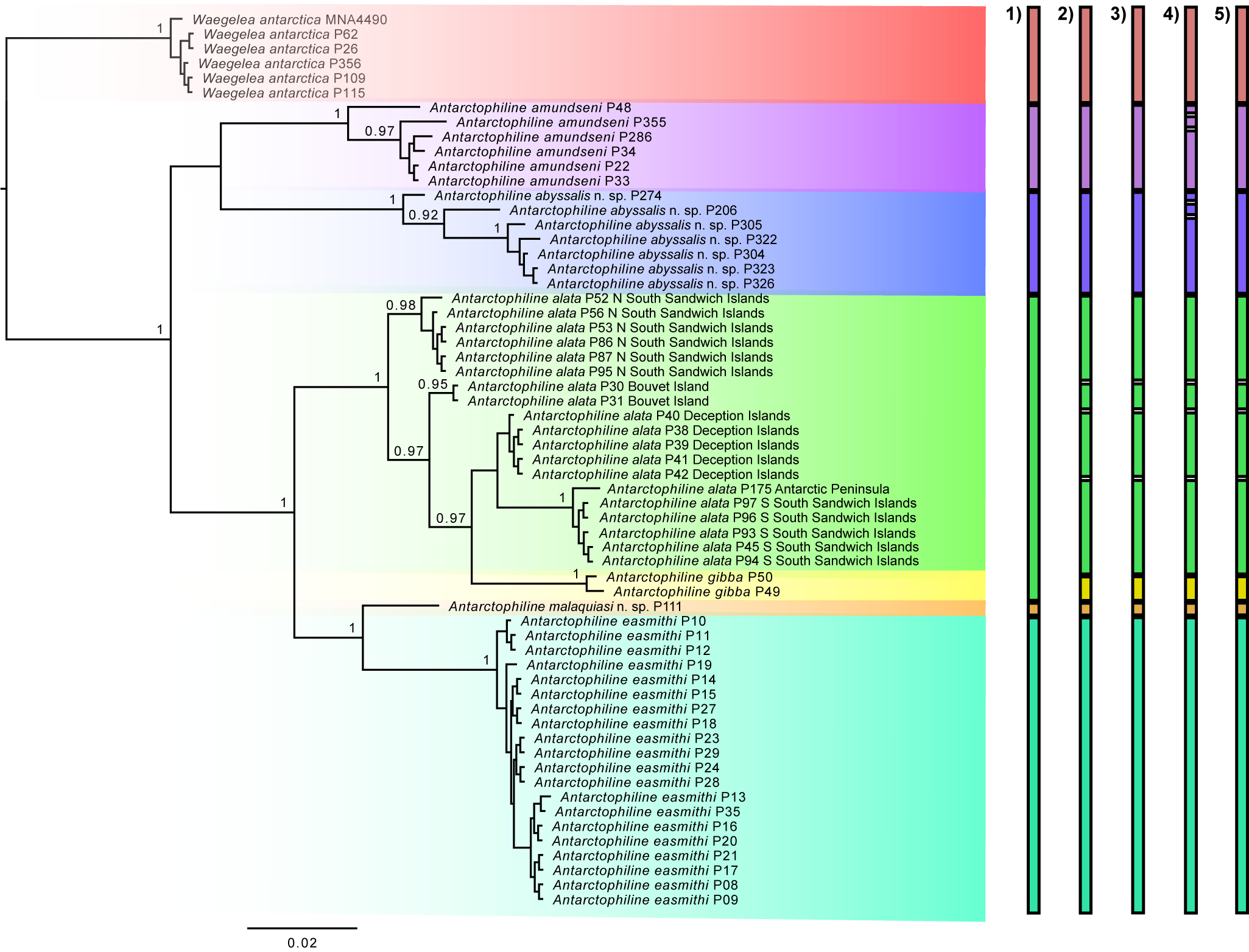
Phylogenetic analysis was conducted in CIPRES Science Gateway v3.3 (at http://www.phylo.org/) using a maximum-likelihood (ML) inference implemented in IQ-TREE v 1.6.10 (Nguyen et al. Reference Nguyen, Schmidt, Von Haeseler and Minh2015), with ModelFinder for the automatic evolutionary model selection (Kalyaanamoorthy et al. Reference Kalyaanamoorthy, Minh, Wong, Von Haeseler and Jermiin2017), and an Ultrafast Bootstrap approximation with 1500 replicates (Hoang et al. Reference Hoang, Chernomor, Von Haeseler, Minh and Vinh2018). The tree was visualized in FigTree v 1.4.4 and edited in Inkscape v0.92.5 (Fig. 1).
Phylogenetic relationships of antarctophilinids based on the COI marker using maximum likelihood (ML), rooting with Waegelea antarctica. Support values listed on nodes refer to Bootstrap. Species delimitation tests representing species concepts including 1) mPTP, 2) ABGD, 3) ASAP, 4) bPTP and 5) GMYC are depicted on the right. The scale bar indicates substitutions per site.
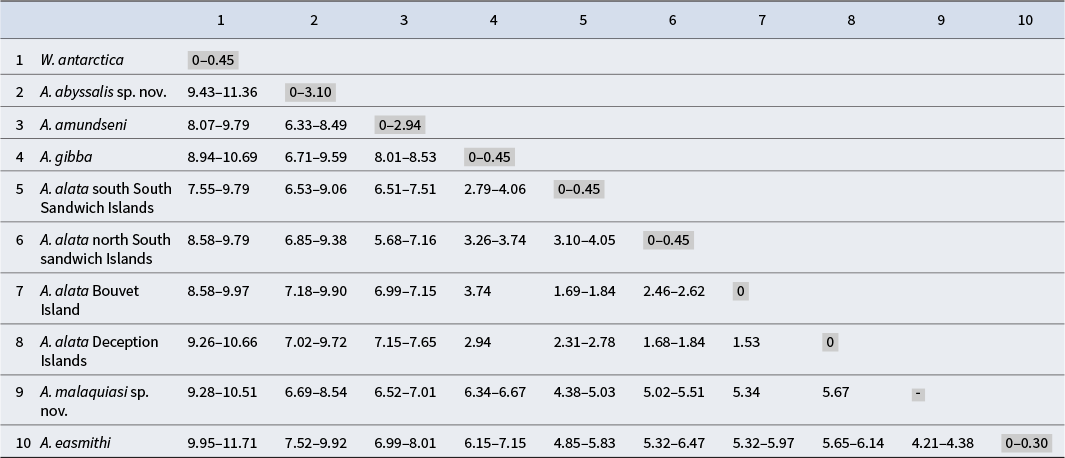
Automatic Barcode Gap Discovery (ABGD) was used as a first approximation for species delimitation by the web server (Puillandre et al. Reference Puillandre, Lambert, Brouillet and Achaz2012) under default parameters (P min = 0.001, P max = 0.1, relative gap width = 1.5, transition/transversion ratio = 2) with a Kimura (K80) measure of distance. A matrix of intra- and interspecific p-distances is depicted in Table I. Assemble Species by Automatic Partitioning (ASAP), a web server for species delimitation (https://bioinfo.mnhn.fr/abi/public/asap/), was used with the Kimura (K80) distance model and default parameters (Puillandre et al. Reference Puillandre, Brouillet and Achaz2021). Generalized Mixed Yule Coalescent (GMYC) species delimitation was performed using BEAUti v1.10.4 to set a lognormal relaxed clock and the best-fitting model, which was HKY + F + I + G4 according to the Bayesian information criterion (BIC). BEAST v.1.10.4 was used to run the trees, and TreeAnotator v.1.10.4 was used to burn the first 2500 trees and generate a single tree. The tree was visualized in FigTree v 1.4.4 and saved as newick files to run in the GMYC web server (https://species.h-its.org/gmyc/). A Bayesian implementation of the Poisson tree processes (bPTP model; Zhang et al. Reference Zhang, Kapli, Pavlidis and Stamatakis2013) for species delimitation was calculated through the web server (https://species.h-its.org/) using the ML tree obtained with IQ-TREE. The mPTP test was performed in the mPTP web server using the ML tree generated in CIPRES.
P-distance matrix based on the mitochondrial gene COI calculated in Automatic Barcode Gap Discovery (ABGD). This includes Waegelea antarctica, Antarctophiline abyssalis sp. nov., Antarctophiline alata species complex, Antarctophiline amundseni, Antarctophiline easmithi, Antarctophiline gibba and Antarctophiline malaquiasi sp. nov.
Morphological analysis
All specimens were photographed dorsally, ventrally and laterally using a Keyence 125 VHX6000 Digital Microscope system at the Museum of Comparative Zoology (MCZ) and a Nikon V1 aided by a Leica Z16 APO macro lens at the ZSM before dissection. The length (L) and width (W) of all specimens were measured with a millimetric ruler under the stereomicroscope. The shell was gently removed with the aid of forceps and photographed. Animals were dissected dorsally, and the digestive and male reproductive systems were removed. The excretory and the female reproductive systems were also dissected and schematically drawn. Hard structures such as the shell, gizzard plates, radula and gut contents were extracted, cleaned with a 10% NaOCl solution and subsequently rinsed with distilled water using gentle ultrasonic baths. All structures were mounted on metal stubs with carbon sticky tabs and coated with gold for scanning electron microscopy (SEM) using a Carl Zeiss Leo 1430VP device. The penial papilla and gizzard were transferred to 100% EtOH and subsequently dried in an increasing series of hexamethyldisilazane (HMDS):100% EtOH solutions (i.e. 1:4, 1:3, 1:2). These were incubated for 30 min each and exchanged to a final concentration of 100% HMDS, then these were left overnight for slow evaporation before SEM.
Results
Molecular analyses
The ML tree divides Antarctophiline specimens into six different species with strong Bootstrap support values (BS), including Waegelea antarctica, A. amundseni, A. alata + A. gibba, A. easmithi and two undescribed species of Antarctophiline. These results differ in the recovery of some of the species (Fig. 1). According to prior works using morphology and many nuclear loci (Moles et al. Reference Moles, Avila and Malaquias2019, Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021), A. gibba can be considered as the sister species to the A. alata complex, and part of our results, including GMYC, bPTP, ABGD and ASAP, recover the same pattern. The tree and the species delimitation tests show a complex of species in A. alata, with four species hypotheses recognized and A. gibba as part of the complex, by ABGD, ASAP, bPTP and GMYC but not mPTP. The specimens belonging to the two undescribed species of Antarctophiline differed topologically from the rest of the sequenced specimens. bPTP recovers the same groupings but splits A. amundseni and one of the undescribed species into three different groups each. The A. alata complex of species is differentiated from the specimens from the South Sandwich Islands, Bouvet Island and Deception Island but has lower p-distance values (1.68–4.05%) than the rest of the species (Table I). The A. alata specimens from the complex had highly different nucleotide proportions compared to the other five species analysed (4.38–9.90%) but a lower proportion compared to A. gibba (2.79–4.06%). Based on our results and those of previous studies, we are going to use as reference the results obtained from ABGD, ASAP and GMYC.
Systematic descriptions
Class Gastropoda Cuvier, 1795
Order Cephalaspidea Fischer, 1883
Superfamily Philinoidea Gray, 1850 (1815)
Family Antarctophilinidae Moles, Avila & Malaquias, Reference Moles, Avila and Malaquias2019
Antarctophiline Chaban, Reference Chaban2016
Antarctophiline alata (Thiele, Reference Thiele1912)
Diagnosis
Body oval, arched dorsally, white or ivory coloured. Cephalic shield square-oval. Shell internal, subquadrate, slightly angled dorsally. Radular formula 12–13 × 2.1.1.1.2. Gizzard plates (3), elongated, oval, symmetrical; internal surface, chitinous, flattened, holes or slits absent, with concentric amber and brown bands; external surface flattened.
Material examined
North South Sandwich Islands, SS1A/30, 56°42′60.6″S, 27°1′35.8″W, 134–142 m depth: 1 specimen, sequenced and dissected, SIO-BIC M13656 (3 September 2011), COI barcode MN486291, L = 11 mm, W = 7 mm; SS2A/38, 58°22′42″S, 26°17′W, 134–260 m depth: 23 specimens, 11 sequenced and 1 dissected, SIO-BIC M12941 (6 October 2011), L = 10 mm, W = 7 mm; SS3/42, 56°42′60.6″S, 27°1′35.8″W, 103–221 m depth: 4 specimens, 1 sequenced and 1 dissected, SIO-BIC M12993 (7 October 2011), barcode MN486273, L = 15 mm, W = 8 mm. South Sandwich Islands, St. A6-2: 12 specimens, 1 dissected (not sequenced), MCZ393943 (February 1995), L = 15 mm, W = 9 mm; St. 136-1, 70°50′12.0″S, 10°35′24.0″W, 271 m depth: 6 specimens, 1 dissected (not sequenced), ZSM20010079 (10 April 2000), L = 13 mm, W = 7 mm.
External morphology (Fig. 2a)
Maximum L = 11 mm, W = 7 mm. Body oval, arched dorsally; white or ivory coloured. Mantle thin, translucent white. Cephalic shield oval, extending approximately half of body, slightly extended over shell; extending longitudinally from second third until posterior edge. Parapodial lobes triangular, slightly extended over shell. Foot smooth, extending from mouth to approximately three-quarters of animal.
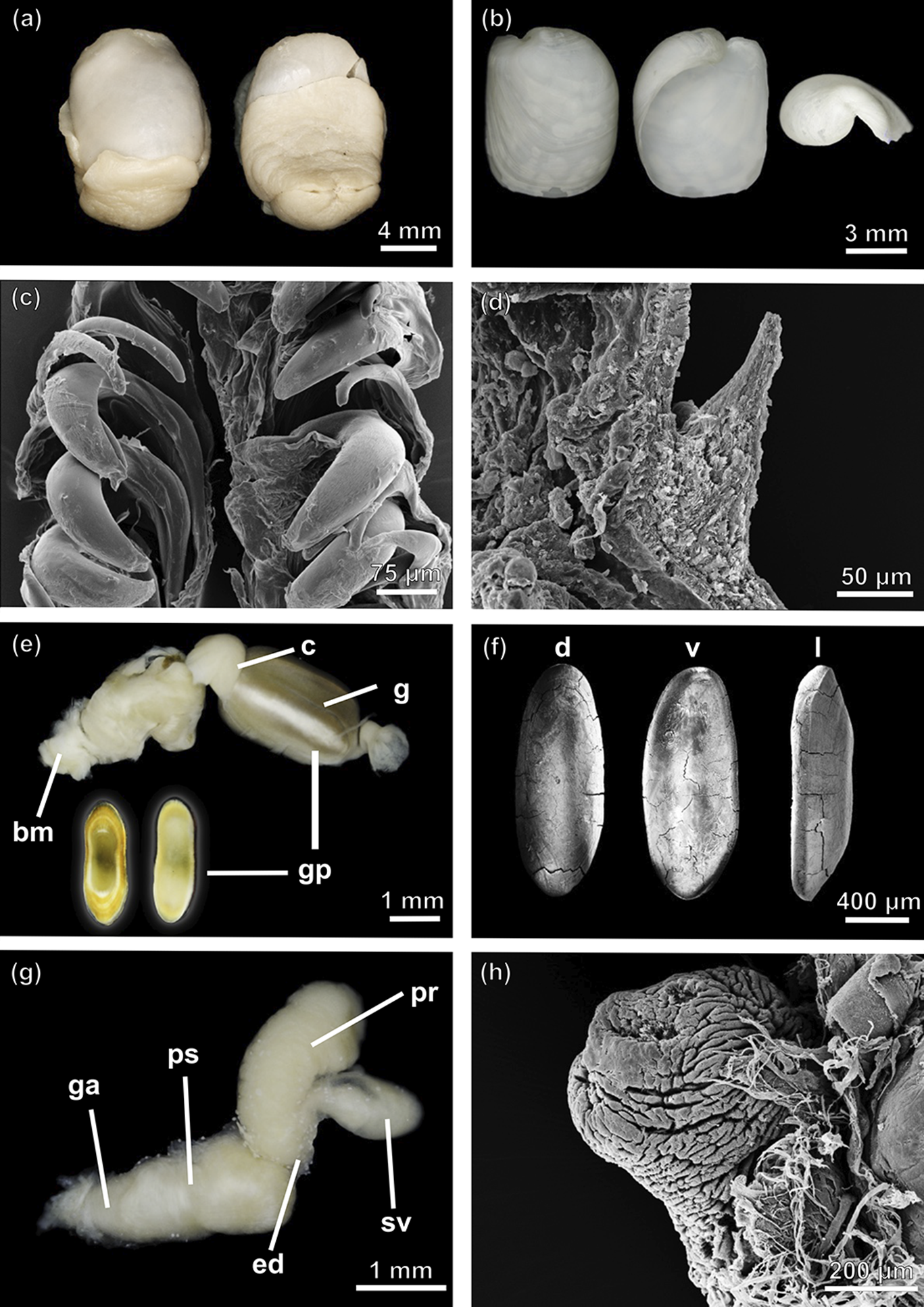
Antarctophiline alata specimens from northern South Sandwich Islands (SIO-BIC M13656 and SIO-BIC M17802), southern South Sandwich Islands (SIO-BIC M12993) and South Shetland Islands (MCZ393943). a. Dorsal (left) and ventral (right) view of the complete animal (SIO-BIC M17802). b. Adapertural (left), apertural (middle) and apex (right) view of the shell (SIO-BIC M17802). c. Scanning electron microscopy (SEM) image of the radula (SIO-BIC M12993). d. Detail of the gizzard spines (SEM; SIO-BIC M13656). e. Anterior part of the digestive system, dorsal (left) and ventral (right) view of a gizzard plate (SIO-BIC M17802). f. Detail of gizzard plates in dorsal (d), ventral (v) and lateral (l) view (SEM; SIO-BIC M17802). g. Male reproductive system (MCZ393943). h. Penial papilla (SEM; MCZ393943). bm = buccal mass; c = crop; ed = ejaculatory duct; g = gizzard; ga = genital aperture; gp = gizzard plate; pr = prostate; ps = penial sheath; sv = seminal vesicle.
Shell (Fig. 2b)
Maximum L = 7.2 mm, W = 5.7 mm. Internal, subquadrate, white, thin, slightly angled dorsally, flattened. Periostracum thin, pellucid. Aperture almost full extension of shell, with thin parietal callus. Outer lip straight, in right angle posteriorly. Columellar wall concave. Apex superficial, umbilicated, left-sided. Growth lines marked.
Radula (Fig. 2c)
Radular formula 12–13 × 2.1.1.1.2. Rachidian teeth not detected. Later teeth hook-shaped, inner lateral thick, with broad base, tip rounded or pointed, inner edge with faint dentition not always detected. Outer lateral teeth hook-shaped, with broad base, tip rounded.
Digestive system (Figs 2d & 3)
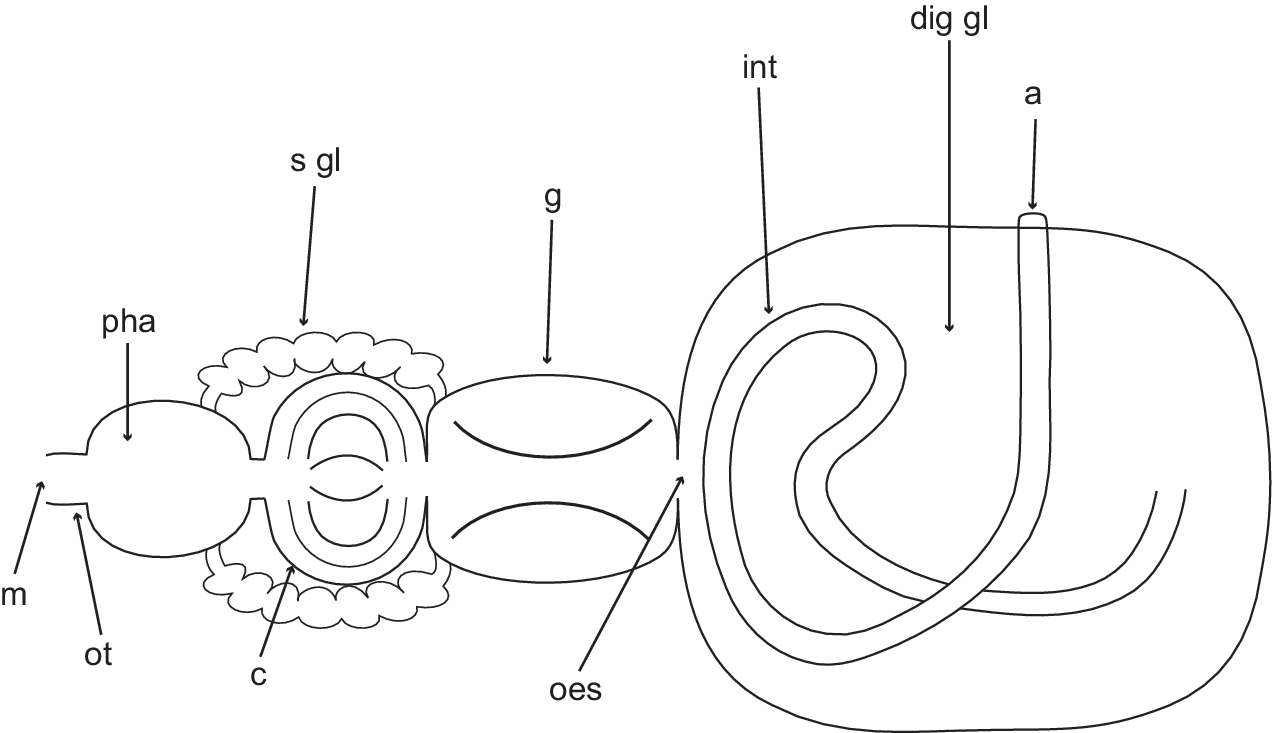
Buccal mass very muscular. Salivary glands short, entering pharynx from posterior end, extending over crop to beginning of gizzard. Crop globose, saccular, thin-walled. Gizzard elongated, surrounded by circular muscle fibres. Gizzard plates three (maximum L = 1.5 mm, W = 0.6 mm), elongated, oval, symmetrical; internal surface, chitinous, flattened, holes or slits absent, with concentric amber and brown bands; external surface flattened (Fig. 2e,f); spines present in gizzard (Fig. 2d). Oesophagus thin, internally plicate. Stomach embedded in digestive gland. Digestive gland large, brownish, granulose; occupying most of viscera. Intestine rising posteriorly towards the anterior part of the shell, creating a loop, turning backwards to right side; ending in external anal papilla situated in mantle cavity.
Dorsal schematic representation of the digestive system of Antarctophiline alata. a = anus; c = crop; dig gl = digestive gland; g = gizzard; int = intestine; m = mouth; oes = oesophagus; ot = oral tube; pha = pharynx; s gl = salivary glands.
Reproductive system (Figs 2g,h & 4)
Male penial sheath short, pyriform. Penial papilla short. Ejaculatory duct displayed in an S shape, surrounded by musculature tissue. Prostate granulose, saccular, shorter than penial sheath, attached to seminal vesicle. Seminal vesicle saccular, semi-translucent, slightly smaller than prostate, containing autosperm. Female gonad orange in colour (ovules), presenting white marks (sperm), lumpy, surrounding the digestive gland posteriorly. Ampulla cylindrical, isodiametric, displayed in zigzag. Seminal receptacle oval, short, connected by a thin duct. Albumen gland (= capsule gland; Klussmann-Kolb Reference Klussmann-Kolb2001) rugose, relatively small, covering end of ampulla and seminal receptacle duct. Membrane gland bean-shaped, internally attached to mantle cavity. Mucous gland white, large, folded, extending ventrally above viscera towards left edge. Bursa copulatrix rounded, distally connected to vagina through a thin, long duct. Vagina muscular, with ample atrium.
Schematic representation of the reproductive system of Antarctophiline alata. amp = ampulla; bc = bursa copulatrix; cap gl = capsule gland; go = gonad; gon = genital opening = mem gl = membrane gland; muc gl = mucous gland; pr = prostate; ps = penial sheath; sr = seminal receptacle; sv = seminal vesicle; vag = vagina.

Excretory, circulatory and respiratory systems
Kidney flattened, in anterior right side of viscera, attached to mantle, internally highly ramified, connected to pericardium anteriorly. Pericardial complex arranged transversally in an angle of 45°. Auricle thin-walled. Ventricle triangular, muscular. Aorta thick, bifurcating into two branches that run parallel to edge of shell.
Ecology
Occurs in sandy bottoms at depths from 4 m (this study) down to 640 m (Hain Reference Hain1990). Sponge spicules, diatoms and foraminifera were found in the gut contents of the specimens dissected in this study (Fig. 5a–c).
Gut content from a.–c. Antarctophiline alata, d.–f. Antarctophiline abyssalis sp. nov. and g.–i. Antarctophiline malaquiasi sp. nov., including sponge spicules (a., d., g.), diatoms (b., h.) and foraminifera (c., e., f., i.).

Distribution
Known from the Davis Sea and Adélie land in East Antarctica (Thiele Reference Thiele1912, Vicente & Arnaud Reference Vicente and Arnaud1974); South Orkney Islands, South Sandwich Islands in the Scotia Sea and Palmer Archipelago in the western Antarctic Peninsula (Powell Reference Powell1951, Aldea & Troncoso Reference Aldea and Troncoso2008, Moles et al. Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021); Deception, Livingston and King George islands in the South Shetland Islands (Powell Reference Powell1951, Troncoso et al. Reference Troncoso, García, Backeljau and Urgorri1996, Engl Reference Engl2012, Moles et al. Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021); Peter I Island, Bellingshausen Sea (Aldea & Troncoso Reference Aldea and Troncoso2008); ? eastern Weddell Sea (Hain Reference Hain1990, Engl Reference Engl2012); and Bouvet Island (Moles et al. 2019).
Remarks
This species was described by Thiele (Reference Thiele1912) based on a single shell record from the Davis Sea characterized by an almost squarish, smooth shell with growth lines, a straight outer lip and a somewhat straight base. This type of shell has been found all over Antarctica and resembles our material. Although slight intraspecific differences in the shape of the gizzard plates exist (e.g. more convex, sometimes slightly pointy, the colour on the bands), they are oval, chitinous and flattened in the interior and exterior parts, with concentric amber and brown bands on the interior side. Most records in the distribution area have been revised by Moles et al. (Reference Moles, Avila and Malaquias2019), and here we add specimens from the northern and southern South Sandwich Islands. There, we found specimens showing no remarkable morphological differences from those from the remaining areas of distribution. Overall, although molecular data recognize several lineages (Fig. 1; Moles et al. Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021), the dissected specimens from Bouvet Island, the South Shetland Islands and the South Sandwich Islands present no clear morphological differences (Moles et al. Reference Moles, Avila and Malaquias2019; this study) and thus are retained here as a single species.
Antarctophiline abyssalis sp. nov.
Diagnosis
Body oval, large, arched sagittally, white nacreous coloured. Cephalic shield sub-rectangular. Shell internal, ovate-subquadrate, slightly angled dorsally, wide dorsoventrally. Radular formula 15 × 2.1.0.1.2. Gizzard plates (3) elongated, oval, ventral plate smaller than dorsal ones; internal surface highly chitinous, convex, holes or slits absent; external surface flattened, with concentric amber and dark brown bands.
ZooBank registration
urn:lsid:zoobank.org:act:2B7C5D1E-C187-47CE-8897-250E4812071F.
Etymology
The species’ name derives from its depth distribution encompassing the abyssal plain from ~3000 to 4550 m depth.
Type locality
Transition Weddell Sea to south of South Sandwich Islands, 2892.8–4547.6 m depth.
Material examined
Transition Weddell Sea to south of South Sandwich Islands, St. PS61/1384, 62°57′48″S, 27°52′8.4″W, 4547.6 m depth: 1 specimen, ZSM20021046 (holotype; 16 March 2002), COI barcode MN486283, L = 27 mm, W = 10 mm. North Elephant Island, St. PS61/046-7, 60°39′11.4″S, 53°56′51″W, 2892.8 m depth: 1 specimen, sequenced, ZSM20021211 (paratype; 30 January 2002), COI barcode MN486284, L = 5 mm, W = 2 mm. North-west Bouvet Island, South Atlantic Ocean, St. PS71/013-15, 52°2′31.8″S, 0°0′36″E, 2996 m depth: 2 specimens, 2 sequenced, ZSM20081179 (paratypes; 6 December 2007), COI barcode MN486285, L = 10 mm, W = 5 mm, COI barcode MN486286, L = 8 mm, W = 3 mm; St. PS79/081-18, 52°21″S, 10°1′28″E, 3705–3757 m depth: 1 specimen, ZSM20130151 (paratype; 20 January 2012), COI barcode MN486287, L = 16 mm, W = 5 mm; PS79/081-17, 52°11″S, 10°43″E, 3743–3763 m depth: 1 specimen, dissected, ZSM20130153 (paratype; 20 January 2012), COI barcode MN486288, L = 27 mm, W = 11 mm.
External morphology (Fig. 6a)
Maximum L = 27 mm, W = 11 mm. Body oval, large, arched sagittally. Shell slightly wide, white nacreous coloured; surrounded by translucent white thin mantle. Cephalic shield sub-rectangular, representing 45% body extension, slightly over shell; posteriorly slightly bilobed. Parapodia triangular, extended over shell. Foot smooth, extending from mouth approximately three-fifths of the animal.
Antarctophiline abyssalis sp. nov. a. Ventral (left) and lateral (right) view of the complete animal. b. Adapertural (left), apertural (middle) and apex (right) view of the shell. c. Scanning electron microscopy (SEM) image of the radula. d. Detail of the gizzard spines (SEM). e. Anterior part of the digestive system, dorsal (left) and ventral (right) view of a gizzard plate. f. Detail of gizzard plates in dorsal (d), ventral (v) and lateral (l) view (SEM). g. Male reproductive system. h. Penial papilla (SEM). bm = buccal mass; c = crop; ed = ejaculatory duct; g = gizzard; ga = genital aperture; gp = gizzard plate; pr = prostate; ps = penial sheath; sgl = salivary glands; sv = seminal vesicle.
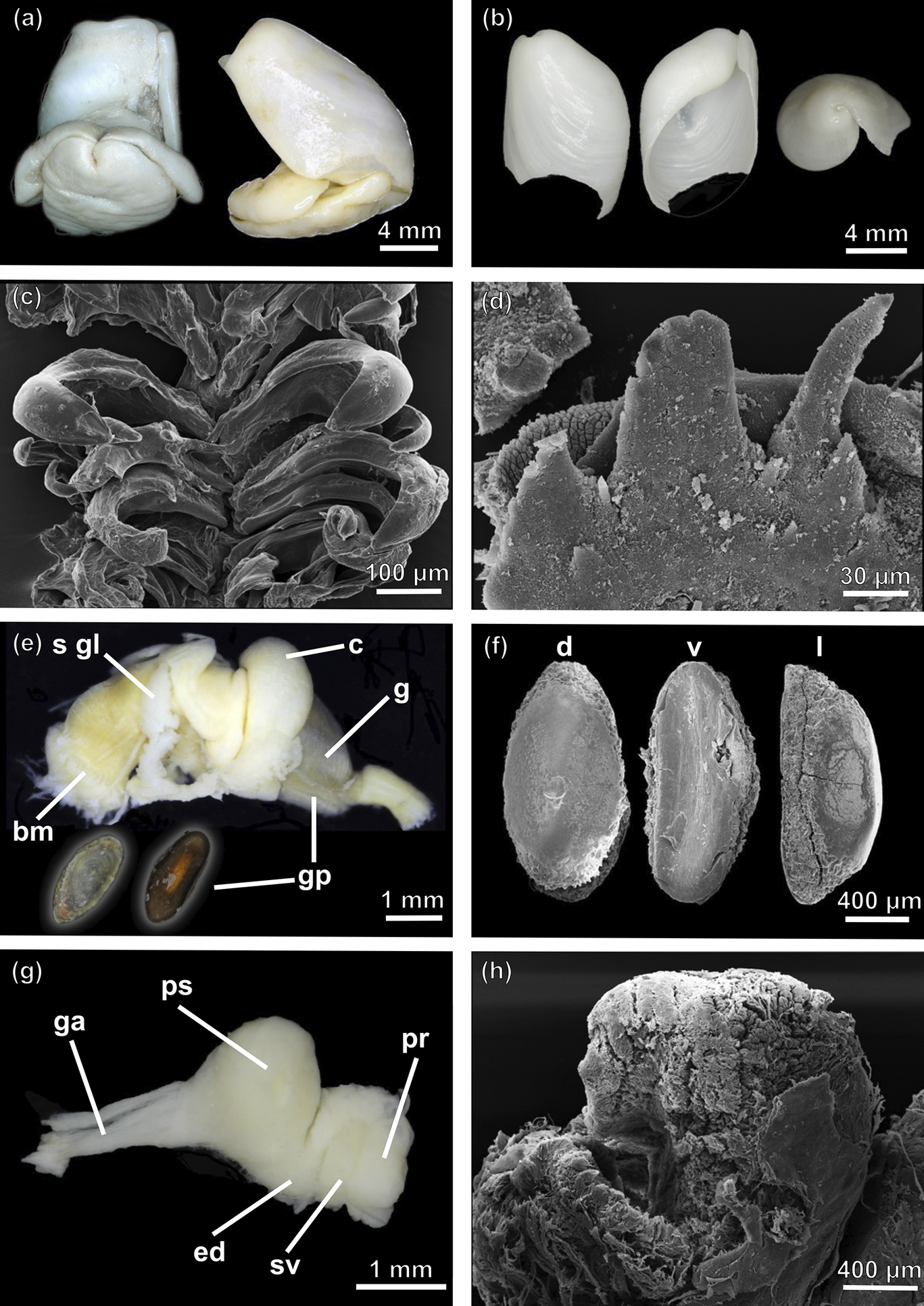
Shell (Fig. 6b)
Maximum L = 12.6 mm, W = 8.6. Internal, ovate-subrectangular, white, thin, slightly angled dorsally, wide dorsoventrally. Periostracum thin, pellucid. Aperture almost representing full extension of shell. Columellar wall concave. Parietal callus thin; outer lip directed towards body whorl, extending downwards. Body whorl wide. Apex bulky, umbilicated, sunken, left-sided. Growth lines marked.
Radula (Fig. 6c)
Radular formula 15 × 2.1.0.1.2. Rachidian not detected. Lateral teeth hook-shaped, inner lateral thick, with broad base, tip rounded or pointed, inner edge with very faint denticulation. Outer lateral teeth hook-shaped, with broad base, tip rounded or pointed.
Digestive tract (Fig. 6e)
Buccal mass highly muscular. Salivary glands long, connected ventrally, entering pharynx from dorsal end, extending under pharynx, going up over crop to beginning of gizzard. Crop globose, saccular, thin walled. Gizzard elongated, surrounded by circular muscle fibres. Gizzard plates (3; maximum L = 1.5 mm, W = 0.8 mm), elongated, oval, ventral plate smaller than dorsal ones; internal surface highly chitinous, convex, translucid, holes or slits absent; external surface flattened, with concentric amber and dark brown bands (Fig. 6e,f); spines present in gizzard (Fig. 6d).
Male reproductive system (Fig. 6g,h)
Penial sheath short, pyriform. Penial papilla small, ~4 mm in length. Ejaculatory duct surrounded by musculature tissue. Prostate granulose, saccular, shorter than penial sheath, displayed attached to seminal vesicle. Seminal vesicle saccular, slightly smaller than prostate, containing autosperm.
Ecology
Found in abyssal plains from 2892.8 to 4547.6 m depth. Sponge spicules and foraminifera were found in the gut contents of the single specimen studied (Fig. 5d–f).
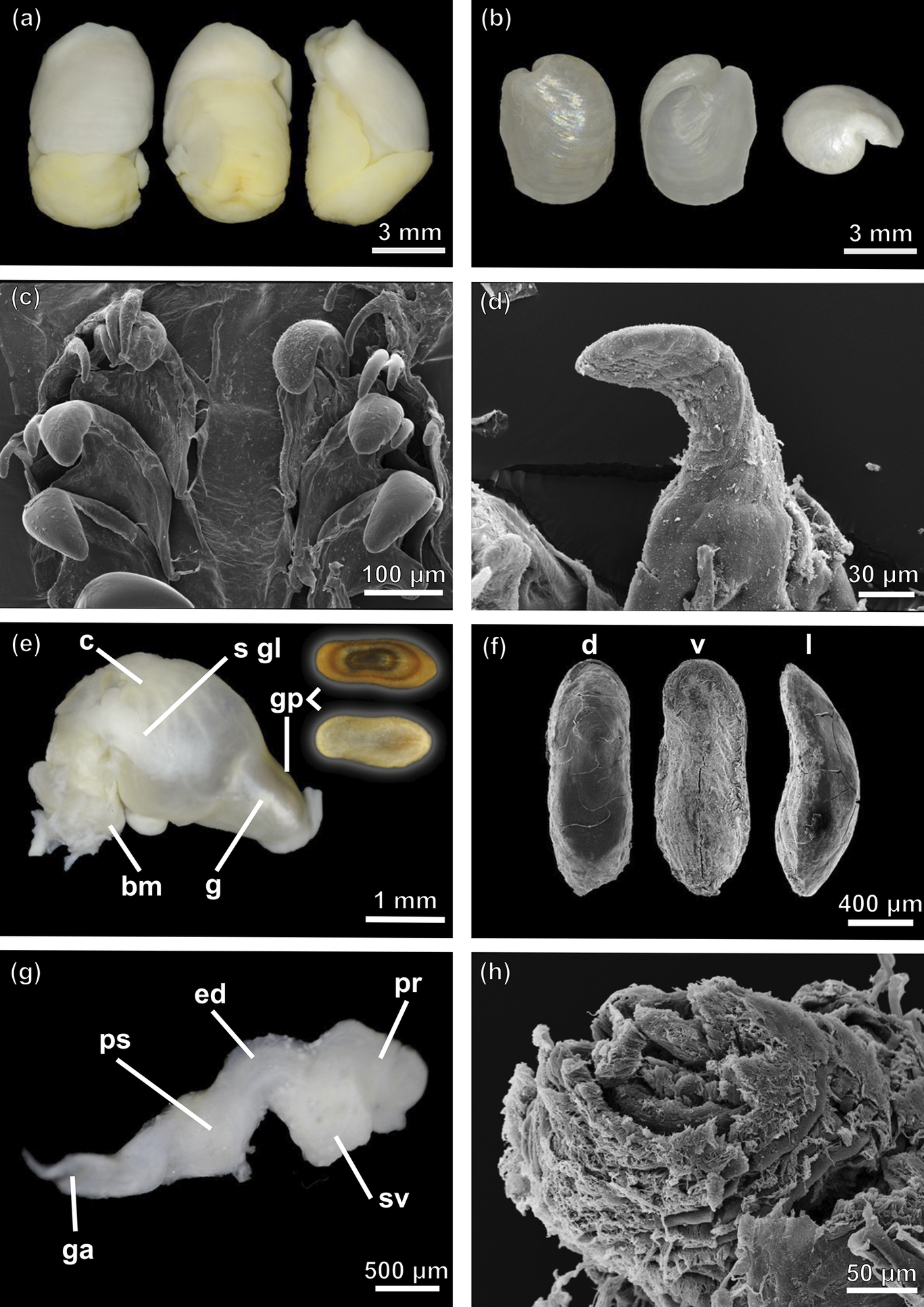
Antarctophiline malaquiasi sp. nov. a. Dorsal (left), ventral (middle) and lateral (right) view of the complete animal. b. Adapertural (left), apertural (middle) and apex (right) view of the shell. c. Scanning electron microscopy (SEM) image of the radula. d. Detail of rachidian tooth (SEM). e. Anterior part of the digestive system, dorsal (left) and ventral (right) view of a gizzard plate. f. Detail of gizzard plates in dorsal (d), ventral (v) and lateral (l) view (SEM). g. Male reproductive system. h. Penial papilla (SEM). bm = buccal mass; c = crop; ed = ejaculatory duct; g = gizzard; ga = genital aperture; gp = gizzard plate; pr = prostate; ps = penial sheath; s gl = salivary glands; sv = seminal vesicle.
Distribution
Known from the South Atlantic Antarctic Ridge, from the vicinities of Bouvet Island to Elephant Island.
Remarks
The new species is differentiated from its counterparts (i.e. A. alata, A. amundseni, A. apertissima, A. easmithi, A. falklandica, A. gibba, A. malaquiasi sp. nov.) by the larger size and wider shell. The shell has a large columellar callus and presents a greater length/width ratio and larger body compared to the other species. In addition, the dark brown, highly chitinous, small gizzard plates, pronouncedly convex internally and flattened externally, are characteristic of this species. The bathymetric range of A. abyssalis sp. nov. encompasses bathyal and abyssal depths in the South Atlantic Antarctic Ridge, where no previous records of any other species are found. The most closely related species is A. amundseni, known from 196 to 1048 m depth in the Weddell Sea.
Antarctophiline malaquiasi sp. nov.
Diagnosis
Body oval, large, arched in sagittal plain, white or ivory coloured. Cephalic shield oval. Shell internal, subquadrate, slightly angled dorsally, flattened. Radular formula 12 × 2.1.0.1.2. Gizzard plates (3) elongated, oval, ventral plate smaller than dorsal ones; internal surface, chitinous, slightly convex on one side, holes or slits absent, with concentric amber and dark brown bands; external surface highly convex, sometimes asymmetrically pointy dorsally.
ZooBank registration
urn:lsid:zoobank.org:act:963E806A-B353-472D-A3E9-29D690CB00B7.
Etymology
This species is named in honour of Dr Manuel António E. Malaquias for his contribution to the systematics of cephalaspidean molluscs.
Type locality
Bransfield Strait, 150–247 m depth.
Material examined
Bransfield Strait, St. BS1/86, 62°52′20.7″S, 57°11′32.5″W, 150–247 m depth: 2 specimens, sequenced and dissected, SIO-BIC M17786 (holotype; 24 October 2011), L = 9.6 mm, W = 5.4 mm, COI barcode MN486290, SIO-BIC M17787 (paratype; 24 October 2011), L = 6 mm, W = 4 mm.
External morphology (Fig. 7a)
Body oval, arched in sagittal plane; maximum L = 8 mm, W = 4.5 mm, white or ivory coloured; surrounding translucent white thin mantle. Cephalic shield oval, half of body extension, slightly over shell; central grove slightly marked, last two-thirds extension. Parapodia triangular, slightly extended over shell; gill visible from above. Foot smooth, extending from mouth approximately three-quarters of the animal.
Shell (Fig. 7b)
Maximum L = 6.6 mm, W = 5.3. Internal, subquadrate, white, thin, slightly angled dorsally, flattened. Periostracum thin, pellucid. Aperture almost full extension of shell, with thin, parietal callus. Outer lip straight, in right angle on posterior side. Columellar wall concave. Apex superficial, umbilicated, left-sided. Growth lines marked.
Radula (Fig. 7c)
Radular formula 12 × 2.1.0.1.2. Rachidian not detected. Lateral teeth hook-shaped, inner lateral thick, with broad base, tip rounded, inner edge without denticulation. Outer lateral teeth hook-shaped, with broad base, tip rounded or pointed.
Digestive tract (Fig. 7e)
Buccal mass highly muscular. Salivary glands short, entering pharynx from posterior end, extending over crop to beginning of gizzard. Crop globose, saccular, thin walled. Gizzard elongated, surrounded by circular muscle fibres. Gizzard plates (3; maximum L = 1.5 mm, W = 0.6 mm) elongated, oval, symmetrical; internal surface chitinous, slightly convex on one side, holes or slits absent, with concentric amber and brown bands; external surface highly convex, sometimes asymmetrically pointy dorsally (Fig. 7e,f); spines present in gizzard (Fig. 7d).
Male reproductive system (Fig. 7g,h)
Penial sheath short, pyriform. Penial papilla small. Ejaculatory duct surrounded by musculature tissue. Prostate granulose, saccular, shorter than penial sheath, displayed attached to seminal vesicle. Seminal vesicle saccular, semitranslucent, slightly bigger than prostate, containing autosperm.
Ecology
Found between 150 and 247 m depth. Sponge spicules, diatoms and foraminifera were found in the gut contents of the single specimen dissected in this study (Fig. 5g–i).
Distribution
Only known from the Bransfield Strait.
Remarks
Although not closely related phylogenetically to A. alata and A. gibba, the new species can hardly be differentiated from them externally. Only the shape of the gizzard plates, being highly convex and pointy dorsally, and a slightly more rounded shell are differential characters compared to the ones from A. alata and A. gibba. These are characters subject to intraspecific variation, thus more specimens are needed to further explore these differences. Unfortunately, only two specimens have been collected so far. Its known distribution also seems to overlap with A. alata and A. gibba, matching the same bathymetrical distribution, indicating that this species may coexist in the same locality.
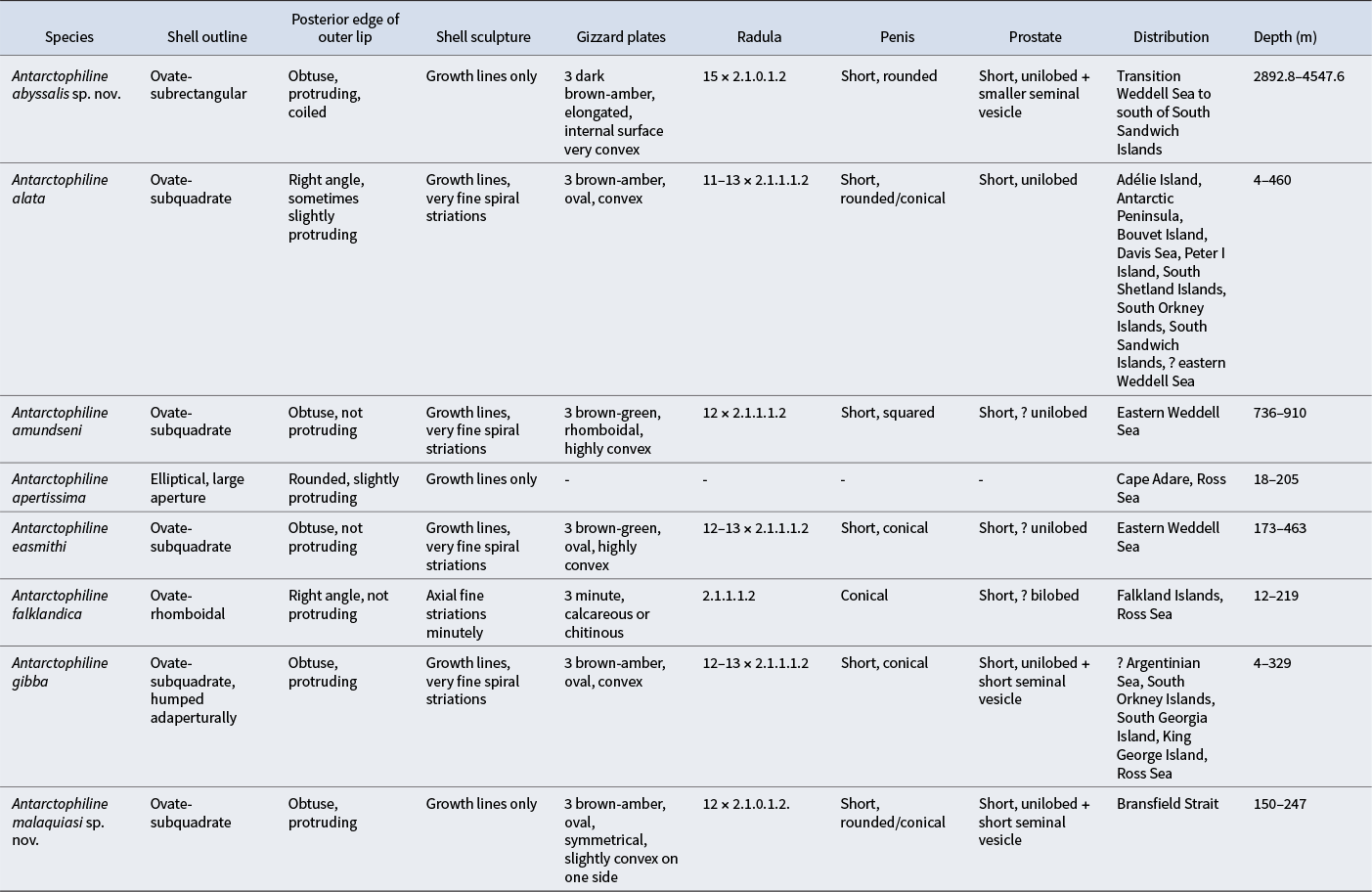
Summary of most useful characters for diagnosis of all species of Antarctophiline based on the previous literature and our data.
Discussion
Based on the molecular, morphological and anatomical data obtained here, we propose the establishment of two new species. A. abyssalis sp. nov. is described from the transition of Weddell Sea to the south of the South Sandwich Islands, at depths of 2996.0–4547.6 m, and A. malaquiasi sp. nov. is described from the Bransfield Strait at depths of 150–247 m. These species present subtle morpho-anatomical differences that are supported by the results of the molecular analyses (see Table II). A. abyssalis sp. nov. presents a larger body size and greater length/width ratio compared to the other species. The shell is higher, has a larger columellar callus and presents a greater length/width ratio. We observed some differences in the anterior part of the digestive system, such as longer salivary glands and a different shape and colour in the gizzard plates. A. malaquiasi sp. nov. is morphologically different in the shape of the gizzard plates, being highly convex and pointy dorsally, and it has a slightly more rounded shell compared to A. alata. Yet, the external morphology of A. malaquiasi sp. nov. is quite similar to those of A. alata and A. gibba. Overall, the species from the genus Antarctophiline are differentiated by subtle morphological differences, usually found in the shell and the gizzard plates, although cryptic speciation is also observed in the genus (Moles et al. Reference Moles, Avila and Malaquias2019, Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021). Common characters used for cephalaspideans’ taxonomy, related to the male reproductive system or shell ultrastructure, have proven unsuitable for differentiating most species.
A. alata is considered to encompass several independent taxonomic units using both mitochondrial and nuclear markers (Moles et al. Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021). Yet, the newly analysed specimens of A. alata from the southern and northern South Sandwich Islands reveal few morphological differences from specimens from the rest of the species distribution (see Moles et al. Reference Moles, Avila and Malaquias2019). The complex of species of A. alata is distributed in the South Sandwich Islands, South Georgia Island, South Shetland Islands and Bouvet Island. The low character displacement of these molecular taxonomic units defines a clear case of cryptic speciation. With the absence of morphological differences, even though molecular characters are present, we decided to retain the same taxonomical identity for all specimens belonging to the complex A. alata (except for A. gibba). This might be a case of incipient speciation among the different populations (Moles et al. Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021) or a case of parallel evolution due to the morphological constraints in the adaptability of the species to the same environment. Another similar-looking species, A. gibba, was revised morphologically by Seager (Reference Seager1978) and Moles et al. (Reference Moles, Avila and Malaquias2019) and sequenced by Moles et al. (Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021). The molecular analyses made in the latter study using thousands of molecular markers support the identity of A. gibba as a different species from A. alata, as was already suggested morphologically by Moles et al. (Reference Moles, Avila and Malaquias2019).
Most of the molluscs in the SO are only known by their shells, and this has created taxonomic confusion and misidentification of taxa (Price et al. Reference Price, Gosliner and Valdés2011). New morphological and molecular analyses have enabled us to differentiate and revise the species described during the early and mid-twentieth century (Moles et al. Reference Moles, Avila and Malaquias2017, Reference Moles, Avila and Malaquias2019, Fassio et al. Reference Fassio, Modica, Alvaro, Buge, Salvi, Oliverio and Schiaparelli2019, Layton et al. Reference Layton, Rouse and Wilson2019, Schächinger et al. Reference Schächinger, Schrödl, Wilson and Moles2022). Gastropods in Antarctica generally show a eurybathic (wide) depth range, usually with species numbers decreasing from shallow to deep waters (Brandt et al. Reference Brandt, Linse and Schüller2009). Antarctophilinids display this eurybathic distribution, being particularly diverse in shallow waters, except for two deep-sea species, one being present in the abyssal plains. This family has a circum-Antarctic distribution, with most of the species and records found in West Antarctica (Moles et al. Reference Moles, Avila and Malaquias2019, Reference Moles, Derkarabetian, Schiaparelli, Schrödl, Troncoso, Wilson and Giribet2021). However, due to high morphological similarities, historical misidentifications of taxa, overlap in bathymetric and geographical distribution and high sampling efforts in specific regions, determining the current distribution of the species remains challenging. Even at the superfamily level they are very difficult to differentiate, as they share a similar morphology and ecological preferences (Price et al. Reference Price, Gosliner and Valdés2011, Ohnheiser & Malaquias Reference Ohnheiser and Malaquias2013, Moles et al. Reference Moles, Avila and Malaquias2019). They are sediment-dwelling organisms inhabiting sandy or muddy substrates with an omnivorous diet, feeding on detritus, small invertebrates or organic matter found within the sediment (Malaquias et al. Reference Malaquias, Ohnheiser, Oskars and Willassen2016). That matches with the gut contents of our specimens, in which we found foraminifera, diatoms and sediment remnants, including sponge spicules, which are present due to their burrowing behaviour. These discoveries add to the growing body of evidence supporting the high level of endemism in this region, improving our knowledge of the hidden diversity of the SO, even if many instances of cryptic species exist (Wilson et al. Reference Wilson, Schrödl and Halanych2009, Reference Wilson, Maschek and Baker2013, Maroni et al. Reference Maroni and Wilson2022).
Acknowledgements
We thank the crews and participants of expeditions undertaken by the German R/V Polarstern and the US R/V Nathaniel B. Palmer. We also thank Enrico Schwabe from the ZSM, as well as Greg Rouse and Charlotte Seid for their assistance with loans at the Scripps Institution of Oceanography Benthic Invertebrates collection. The comments of two anonymous reviewers are also acknowledged.
Financial support
MP-S thanks the Erasmus+ programme of the European Union and UNITAS Malacologica for funding his participation in the World Congress of Malacology 2022. JM’s postdoctoral fellowships were supported by Ramón Areces (Spain) and the Alexander von Humboldt (Germany) Foundations. NGW acknowledges the funding provided by the NSF Office of Polar Programs (ANT-1043749). JM is indebted to the Ministry of Science and Innovation through the HETGEN1000 project (PID2021-127037NA-I00/MCIN/AEI/10.13039/501100011033/ and by FEDER una manera de hacer Europa).
Author contributions
JM was involved in the conception and design of the study. MS, NGW and JM contributed samples and funding. MP-S and JM performed morpho-anatomical and molecular analyses and wrote the first draft of the article. All authors reviewed and edited the final manuscript.











