The study of aquatic macroinvertebrates in wetlands typically involves labour-intensive sweep-net sampling in the field and sample processing in the laboratory (Anderson et al. Reference Anderson, Zilli, Montalto, Marchese, McKinney, Park, Anderson and Davis2013; Alberta Biodiversity Monitoring Institute 2019; Armellin et al. Reference Armellin, Baird, Curry, Glozier, Martens and McIvor2019). Consequently, these studies can be costly, and these costs may hinder efforts to obtain information on wetland macroinvertebrate communities, restricting our understanding of these systems and contributing to shortfalls in invertebrate conservation (Cardoso et al. Reference Cardoso, Erwin, Borges and New2011; Wrubleski and Ross Reference Wrubleski, Ross and Floate2011). Sample processing can be time-consuming, often taking several hours per sample and approximately 7–9 times as long as field sample collection (Hanisch et al. Reference Hanisch, Connor, Scrimgeour and Cobbaert2020). Sample processing typically involves two steps: extracting target organisms (sample cleaning) and then sorting the extracted organisms for identification and enumeration. No automated technique has been developed for the second step, but various methods have been applied to the first, including sieving, flotation, and behavioural extraction (Anderson et al. Reference Anderson, Zilli, Montalto, Marchese, McKinney, Park, Anderson and Davis2013). Therefore, it is worthwhile to compare these extraction methods to determine which best balances time cost and extraction effectiveness.
We compared two commonly used methods, elutriation and flotation, and a new behavioural method for extracting macroinvertebrates from wetland net sweep samples. We focused on Coleoptera (adults only), Hemiptera (adults and nymphs), and water boatmen (Hemiptera: Corixidae), all of which were collected for another study targeting water boatmen. Elutriation uses a combination of flowing water, sieves, and manual picking to extract macroinvertebrates (Alberta Biodiversity Monitoring Institute 2015). Flotation is submerging the sample in a solute-water solution to separate material based on density and collecting floating specimens from the surface (Flannagan Reference Flannagan1973; Anderson et al. Reference Anderson, Zilli, Montalto, Marchese, McKinney, Park, Anderson and Davis2013). Salt and sugar solutions are shown to be effective (Pask and Costa Reference Pask and Costa1971; Dhal and Mitchell Reference Dhal and Mitchell2013), although this may vary by habitat (Foth et al. Reference Foth, Straub and Kaminski2012). Water may be an effective flotation medium if organisms can be trapped in the surface film (Wilhelm and Hiebert Reference Wilhelm and Hiebert1996). We tested all three of these flotation media, including two sugar concentrations shown to be effective at floating water boatmen (Flannagan Reference Flannagan1973). Our behavioural method occurs in the field immediately after sample collection and targets air-breathing insects using a submerged inverted funnel, similar to the funnel-trap sampler of Henrikson and Oscarson (Reference Henrikson and Oscarson1978). The samples used in our comparison were part of a parent study, with time and equipment constraints such that we could not equally distribute samples among extraction methods. However, we were able to opportunistically conduct this preliminary assessment of each method’s time cost and extraction effectiveness and objectively provide recommendations.
We collected four samples from a single wetland (pond) in Edmonton, Alberta, Canada on 18 June 2024, during a pilot test of flotation techniques. We then collected another 88 samples from 50 wetlands south and east of Edmonton in Alberta’s Central Parkland Natural Subregion from 22 July to 2 August 2024, for a total of 92 samples. We followed the Canadian Aquatic Biomonitoring Network’s wetland macroinvertebrate collection protocol (Armellin et al. Reference Armellin, Baird, Curry, Glozier, Martens and McIvor2019), which is a two-minute net sweep along a transect in an area representative of the wetland. Most of the samples contained high amounts of aquatic macrophytes or filamentous algae (Fig. 1A). Some samples contained high amounts of coarse organic debris, fine organic debris, or nonorganic material (mud). We placed the net contents of each sweep into a 1-L sample bottle and fixed two samples (one for flotation with saltwater and one for flotation with 300 g/L sugar–water) with 70% ethanol and 84 with propylene glycol. We kept five samples alive for behavioural extraction.
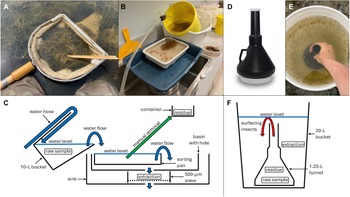
Two methods used to extract Coleoptera adults and Hemiptera from wetland net sweep samples: A, a typical wetland net sweep result; B and C, elutriation with manual removal of noninvertebrate material (= residue) and capture of invertebrate material (= extraction) in a sieve; D, funnel used for behavioural extraction; E and F, behavioural extraction targeting air-breathing, swimming aquatic insects.
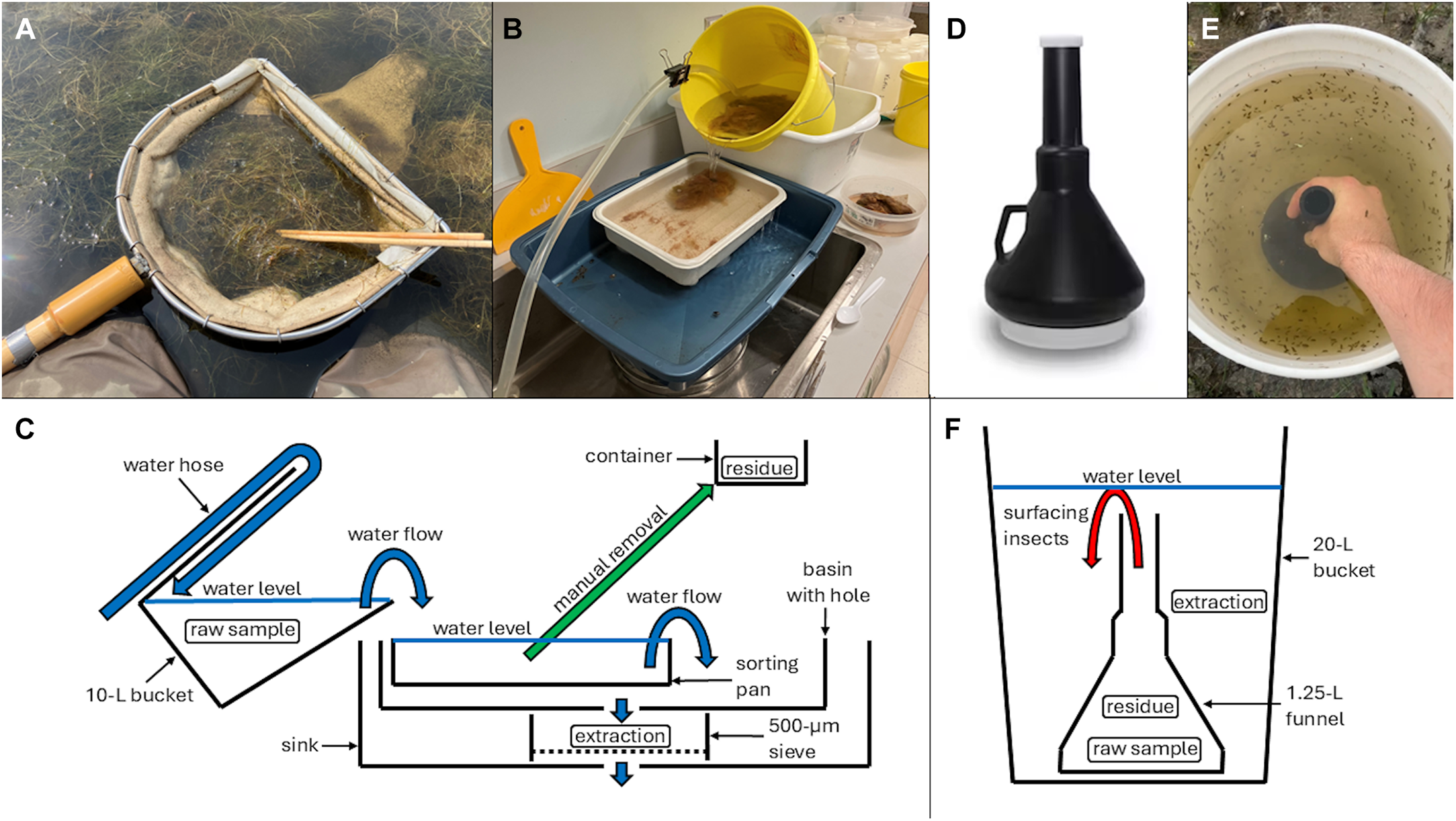
We elutriated 64 samples, floated 13, and behaviourally extracted 4. For elutriation, we followed the Alberta Biodiversity Monitoring Institute’s (2015) method by placing the rinsed sample into a tilted 10-L bucket, then running tap water into the bucket and letting the sample flow out into a white sorting pan where unwanted material (residue) was manually removed and placed into a container. The rest of the material flowed out of the sorting pan into a pet litter pan with a hole cut in the centre, which we placed over a 500-μm stainless steel sieve in a sink (Fig. 1B, C). For flotation, we placed rinsed samples into a 10-L bucket and submerged two in a table salt–tap water solution (357 g/L), nine in a granulated sugar–tap water solution (seven in 600 g/L and two in 300 g/L), and two in tap water. Once the samples were submerged, we stirred them, collected floating invertebrates from the surface using a small strainer, and then repeated this until we did not see any more floating macroinvertebrates. For behavioural extraction, we placed live samples into a WirthCo 1.25-L black plastic funnel (US Plastics, Lima, Idaho, United States of America; Fig. 1D). The funnel is 28 cm tall with a 3.5-cm-wide spout and a screw-on cap at each end that allowed easy filling and transport. We filled the funnel with water, held it inverted inside a 20-L bucket, filled the bucket with water to approximately 2 cm above the funnel spout and removed the spout cap (Fig. 1E, F). We waited 15 minutes for insects to swim out of the funnel into the bucket, with this time based on consideration of field logistics and three pilot tests in which we observed few insects surfacing after this length of time. We then capped and removed the funnel and poured the bucket contents through a 500-μm sieve and preserved them in propylene glycol. We preserved the residual material from flotation and behavioural extraction in propylene glycol.
We measured extraction time as the time it took to extract all perceived macroinvertebrates from a sample, because it was simpler and more time efficient than extracting only Coleoptera and Hemiptera. We measured the extraction time for each sample processed with elutriation and flotation (600 g/L sugar and water only). To measure specimen extraction effectiveness, we examined the extracted and residual sample material from all extraction methods under 10–40× stereo dissecting microscopes, manually counted adult Coleoptera, Hemiptera, and water boatmen adults and nymphs, then calculated the proportion of specimens extracted for each method (extracted count/(extracted count + residual count)). We did not measure extraction effectiveness for elutriation because we examined the residual material from the elutriated samples immediately after finishing extraction and returned unextracted macroinvertebrates to their samples, without counting them, to maximise species capture for the parent study. However, we provide a mean proportion of Coleoptera (adults) and Hemiptera (adults and nymphs) specimens extracted, as well as a mean extraction time for elutriation from quality control data collected by the Alberta Biodiversity Monitoring Institute (provided by Robert Hinchliffe). These data were from wetland samples also collected with the Canadian Aquatic Biomonitoring Network protocol in Alberta’s Central Parkland. We counted extracted and unextracted (residual) Coleoptera and Hemiptera from the floated and behaviourally processed samples separately. We descriptively compared extraction times and extracted proportions to determine which method was least time-consuming and most effective.
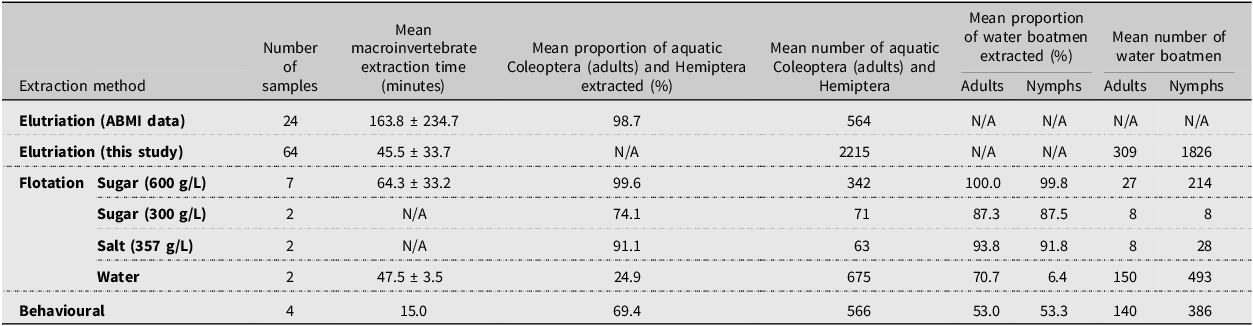
At a fixed 15 minutes, behavioural extraction was the fastest method, followed by elutriation, then flotation with water and the 600 g/L sugar solution (Table 1). Elutriation (based on Alberta Biodiversity Monitoring Institute data) and flotation with the 600 g/L sugar solution were the most effective at extracting Coleoptera adults and Hemiptera (Table 1). Flotation with salt and the 300 g/L sugar solution was also highly effective; however, this may be partly due to lower numbers of specimens in these samples, and we note that fewer samples were tested for flotation than elutriation (Table 1). The water float extracted 25% of Coleoptera and Hemiptera, 71% of adult water boatmen, and only 6% of water boatman nymphs. The behavioural method extracted 69% of Coleoptera and Hemiptera and about half of both adult and nymphal water boatmen (Table 1). Using Alberta Biodiversity Monitoring Institute quality control data from 24 samples collected in 2022, mean macroinvertebrate extraction time for elutriation was 163.8 ± 234.7 minutes, and the overall proportion of Coleoptera and Hemiptera specimens extracted was 98.7%. Combining our dataset with that of the Alberta Biodiversity Monitoring Institute, the overall mean extraction time was 77.7 ± 134.9 minutes.
Number of samples, mean processing times for extracting macroinvertebrates, mean proportion of select macroinvertebrates extracted, and mean number of specimens per sample for six methods of extracting macroinvertebrates from wetland net sweep samples. ABMI, Alberta Biodiversity Monitoring Institute
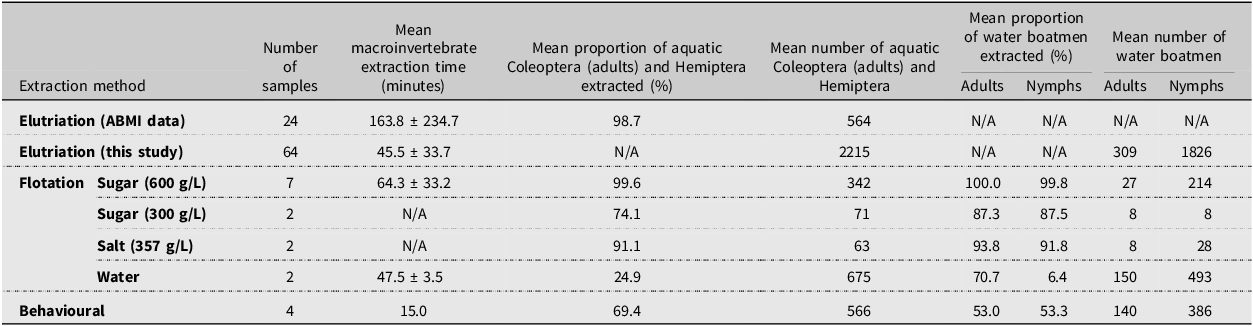
Based on our samples, elutriation was generally the best method for extracting Coleoptera adults and Hemiptera from wetland net sweep samples because it was the least time-consuming method that yielded a proportion of specimens extracted above 95%. However, flotation using the thicker sugar solution (600 g/L) and salt solution (357 g/L) was also highly effective and faster than elutriation given Alberta Biodiversity Monitoring Institute quality control data (Table 1). We found that repeated stirring was necessary because some specimens would remain trapped in the sample matrix until the sample was adequately turned over. Floating samples in a wide container allowing for horizontal spread may reduce the need to stir the sample. We found that flotation was better suited than elutriation to samples with fine organics and inorganics because this material tended to plug the sieve. Our behavioural method extracted only about 70% of Coleoptera and Hemiptera and about half of the water boatmen. However, this first test is a proof of concept, and it likely would perform better if given more time to operate, considering some water boatmen can remain underwater for up to 80 minutes at 12 °C (Henrikson and Oscarson Reference Henrikson and Oscarson1978). However, the sample must not be so tightly packed into the funnel as to prevent insects from moving through the sample matrix. Some preliminary sample cleaning in the net to remove excess vegetation is beneficial for this, but double-capped funnels larger than 1.25 L may be better for samples with high amounts of vegetation. The behavioural extraction method was well suited to samples with high amounts of filamentous algae, which make sample cleaning with the other extraction methods and subsequent sample sorting difficult. Behavioural extraction also produced much cleaner samples for sorting than any of the other methods did, reducing sorting time in the laboratory.
For macroinvertebrates other than Coleoptera adults and Hemiptera, the choice of extraction method depends on target taxa, time constraints, and equipment availability. In our case, elutriation required pans, a hose, and a large sink. We observed that flotation failed to extract snails (Gastropoda) and caddisflies (Trichoptera), which elutriation successfully extracts (Alberta Biodiversity Monitoring Institute 2015), due to their dense shells and cases. Our behavioural extraction method targeted only air-breathing, swimming insects. We recommend elutriation for taxonomically broad wetland macroinvertebrate surveys for its reliability. We recommend our behavioural extraction method for surveys targeting air-breathing, swimming aquatic insects if given longer than 15 minutes to operate, because it produces very clean samples for subsequent sorting.
Acknowledgements
The authors thank Solomiya Kucharyshyn, Victoria Berg, and Samuel Bennett for their assistance in the field and laboratory, and Robert Hinchliffe for providing laboratory space, equipment, and data. This project was generously supported by the Alberta Conservation Association, Mitacs, InnoTech Alberta, Alberta Innovates, Ducks Unlimited Canada, and the National Science and Engineering Research Council.
Competing interests
The authors declare that they have no competing interests.