9.1 Introduction
The flux of magmatic CO2 into the oceans and atmosphere contributes to the global surface carbon cycle, and changes in basaltic magma production and associated degassing have been proposed as one of several important forcing mechanisms that have influenced past global climate variations.Reference Zhong1–Reference Huybers and Langmuir4 Despite this degassing, the vast majority of Earth’s carbon is present not at the surface but in Earth’s convecting mantle.Reference Halliday5–Reference Marty and Tolstikhin10 The convecting mantle is the part of the mantle that lies beneath tectonic plates, and its motions over geologic timescales are driven by the sinking of cold oceanic plates into the interior and hot active upwellings originating from great depth. The convecting upper mantle rises, melts, and erupts mid-ocean ridge basalt (MORB) due to seafloor spreading along the 56,000‑km length of the global mid-ocean ridge system. Active upwellings, driven by mantle buoyancy, produce intraplate and near-ridge hot spots (e.g. Hawaii, Iceland) that erupt ocean island basalt (OIB) with a smaller overall flux.Reference Crisp11 Occasionally, however, active upwellings produce large igneous provinces (LIPs; e.g. Ontong–Java Plateau, Caribbean–Columbian Plateau, Manihiki Plateau, Kerguelen Plateau) characterized by enormous outputs of magma erupted over very short timescales. These flood basalt eruptions have often coincided in time with global climate crises and mass species extinctions.Reference Schoene, Guex, Bartolini, Schaltegger and Blackburn12–Reference Self, Widdowson, Thordarson and Jay16 Ridges, hot spots, and LIPs represent the main volcanic expressions of the delivery of heat to Earth’s surface by the ongoing motions of the convecting mantle.
Seafloor dredging and core recovery have produced tens of thousands of individual submarine glass and rock samples. Study of these samples has demonstrated that variations in the major and trace element chemistry of MORBs and OIBs are primarily the result of crystal fractionation following polybaric melting of mantle sources at different depths and variable temperatures.Reference Gale, Langmuir and Dalton17–Reference Langmuir, Klein, Plank, Morgan, Blackman and Sinton21 At the same time, isotopic differences between MORBs and OIBs show that the mantle sources of hot spots are much more diverse and heterogeneous than those of ridges, reflecting their derivation from distinct reservoirs within the convecting mantle consisting of numerous geochemically distinct components with varied origins and evolutions.Reference Hart22–Reference Zindler and Hart25 Some of these reservoirs have retained anomalies in short-lived isotope systems that were active only in the first tens of millions of years of Earth’s history, and thus appear to have been isolated from each other – and from Earth’s surface – for a large fraction of the age of Earth.Reference Horan26–Reference Tucker, Mukhopadhyay and Schilling33 It is also well established that, from segment to segment, the concentrations of highly incompatible trace elements do not correlate with crustal thickness (a proxy for potential temperature) or spreading rate (a proxy for melting dynamics). Instead, highly incompatible trace elements (e.g. Th concentrations, Ba/Nb ratios) covary with radiogenic isotopes (e.g. 87Sr/86Sr) within segments,Reference Donnelly, Goldstein, Langmuir and Spiegelman34 approaching hot spots,Reference Schilling35 and globally;Reference Gale, Dalton, Langmuir, Su and Schilling36 these covariations indicate heterogeneity in the mantle source. Here, we rely on this fundamental observation of MORB and OIB geochemistry to infer mantle carbon concentrations (Section 9.3.2).
Carbon is a trace element in the mantle, and it is present in erupted magmas as CO2-rich vesicles and as carbonate ions dissolved in the melt.Reference Dixon and Stolper37, Reference Javoy and Pineau38 The solubility of CO2 in basaltic magma is low at pressures of eruption on land or on the seafloor,Reference Dixon, Stolper and Holloway39–Reference Shishkina, Botcharnikov, Holtz, Almeev and Portnyagin42 and as a result nearly all magmas have degassed much of their original pre-eruptive budget of carbon during eruption. Degassing has thus complicated the use of MORBs and OIBs for determining the carbon content of their mantle sources and the flux of CO2 from basaltic eruptions. To attempt to circumvent this complication, researchers have used a variety of microbeam analysis methods to study the composition and volatile content of melt inclusions, which are small pockets of melt (tens to hundreds of microns in diameter) that become trapped in phenocryst minerals that grow in the magma prior to eruption at the surface (Figure 9.1) and are consequently less degassed than their host magmas. Fourier-transform infrared spectroscopy (FTIR) and secondary ion mass spectrometery (SIMS) have both been used to make accurate and precise measurements of CO2 and H2O in melt inclusions, providing a means to interrogate collections of MORB and OIB samples to study the origin and evolution of carbon in the convecting mantle. Most melt inclusions (with some exceptions; see below) contain a vapor bubble formed at vapor saturation after inclusion entrapment, during differential cooling of the melt inside the olivine crystals (Figure 9.1). These vapor bubbles typically contain most of the CO2 within the inclusion, but they cannot be measured by SIMS or FTIR.Reference Aster43, Reference Moore44
Illustration of carbon dioxide and nonvolatile trace element behavior in magmas at oceanic volcanoes. Each panel tracks the concentration of CO2 and a nonvolatile trace element during the ascent of magma for three hypothetical cases. (a) A CO2-undersaturated magma that begins with low volatile and trace element concentrations. In this case, the magma ascends and erupts on the seafloor without experiencing CO2 degassing, such that the starting CO2 concentration and that of a nonvolatile trace element remain roughly constant during ascent and eruption. (b) A magma that begins with higher volatile and trace element concentrations and experiences CO2 saturation during ascent to the seafloor. At conditions of saturation, bubbles form as the magma moves upwards to lower pressure, removing CO2 from the magma. The concentration of a nonvolatile trace element in this system, however, remains roughly constant. In the last stages prior to eruption, the magma may move faster than CO2 is able to diffuse toward bubbles, resulting in an erupted magma on the seafloor that contains more CO2 than would be present for equilibrium saturation (i.e. it is supersaturated). This is the most common condition for basalts erupted at mid-ocean ridges. (c) A volatile- and trace element-enriched magma that ascends beneath an oceanic island and experiences CO2 saturation at greater depth. Because of the lower eruption pressure above sea level, the magma loses virtually all of its CO2 to degassing, whereas the concentration of a nonvolatile trace element remains roughly constant. In all cases, olivine-hosted melt inclusions (see inset, sample NMNH 116111-5 melt inclusion is 100 µm in diameter) may trap melts during various stages of ascent. If trapped at pressures higher than the CO2 saturation curve, melt inclusions may preserve the undegassed magma composition even if the external magma has experienced degassing.

In this chapter, we review the types of samples that are best studied to assessing the primary CO2 content of magmas from mid-ocean ridges and hot spots, estimate volcanic fluxes, and estimate the CO2 content of mantle sources present in the convecting mantle. While we are unable to assess the primary CO2 content of LIP magmas, we review CO2 flux estimates from the literature. The volcanism that we consider here does not include subduction zones or continental intraplate volcanism, which are covered in Chapter 8. We also do not consider petit-spot volcanism – small, young edifices discovered outboard of the Japan trench.Reference Hirano45 These lavas may originate from below the lithosphere–asthenosphere boundary and erupt on the seafloor due to plate flexure. They are highly alkalic and highly vesicular, such that they may carry significant CO2, although it is unclear whether this CO2 originated from the mantle or from crustal contamination. Their occurrence may be restricted to a few edifices outboard of the Japan trench, or could be ubiquitous. Their contribution to CO2 outgassing is presently highly unconstrained.
9.2 Sampling
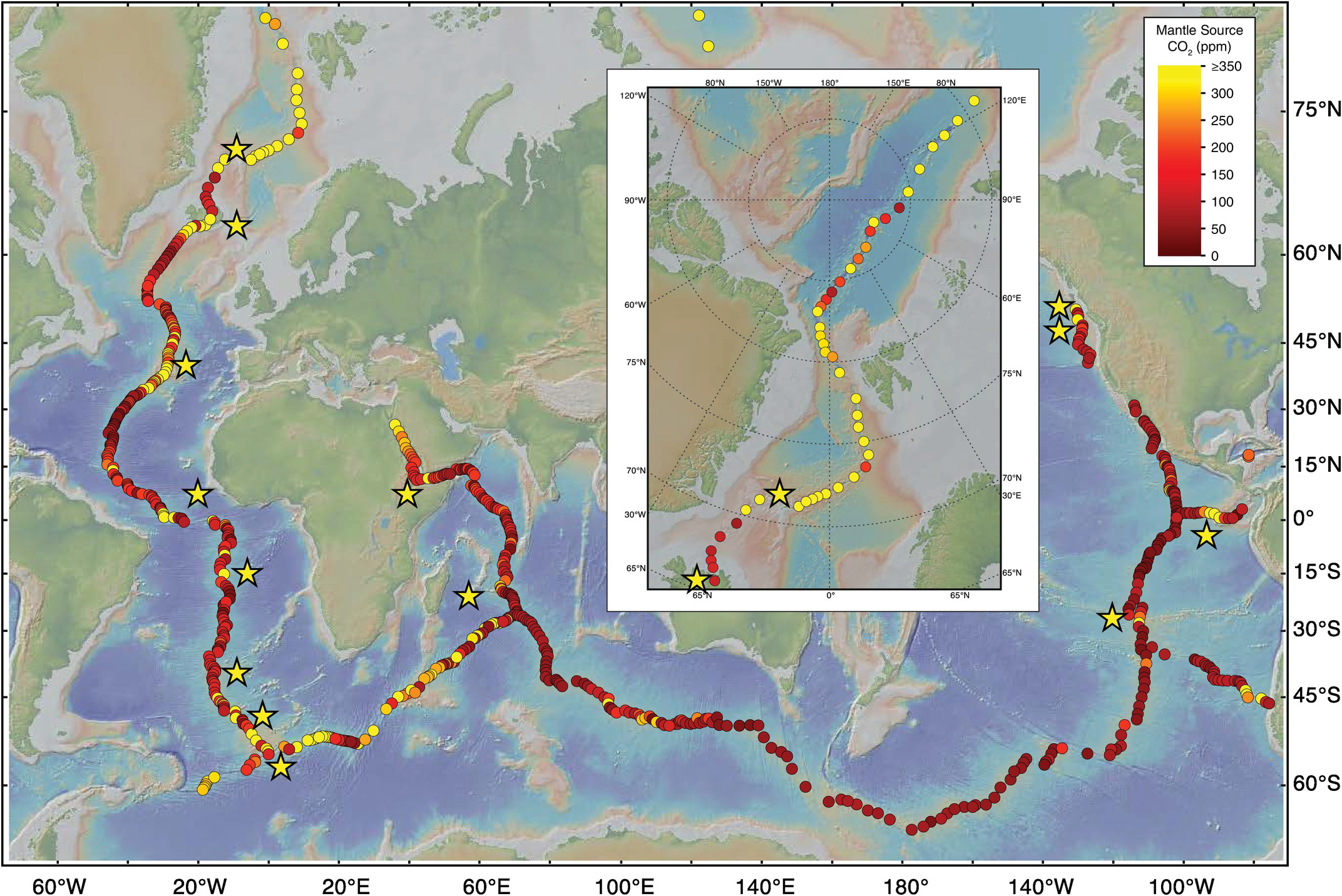
Major collections of seafloor samples (Figure 9.2) are discoverable and searchable through the Index to Marine and Lacustrine Geological Samples (IMLGS) at the National Centers for Environmental Information (NCEI; formerly National Geophysical Data Center, NGDC) at the US National Oceanographic and Atmospheric Association.46 Once identified, samples can be requested from the individual sample repositories. In this chapter, we rely heavily on the MORB study of Le Voyer et al.Reference Le Voyer47 (and references therein), who reported new data on 753 geographically distributed MORB samples, combined with a quality-controlled database of published data from an additional 2446 samples, describing CO2 fluxes and estimated primary magma CO2 contents for 387 of the 458 mid-ocean ridge segments with major element data (out of a total of 711 geophysically defined MORB ridge segments).Reference Gale, Langmuir and Dalton17, Reference Gale, Dalton, Langmuir, Su and Schilling36 Among these MORB samples, there exist samples with sufficiently low pre-eruptive carbon contents and high eruptive pressures that they are thought to have avoided CO2 degassing (e.g. Figure 9.1a); these samples display correlations of CO2 with nonvolatile trace elements, as described by Cartigny et al.Reference Cartigny, Pineau, Aubaud and Javoy48 and Javoy and PineauReference Javoy and Pineau38 (CO2-rich samples), and by Saal et al.,Reference Saal, Hauri, Langmuir and Perfit49 Michael and GrahamReference Michael and Graham50, and Shimizu et al.Reference Shimizu51 (undegassed samples; Figure 9.3). Additionally, three suites of melt inclusions from MORB sources show similar correlations of CO2 with nonvolatile trace elements characteristic of vapor undersaturation, which have been described from the Siqueiros Transform on the East Pacific Rise,Reference Saal, Hauri, Langmuir and Perfit49 the equatorial Mid-Atlantic Ridge,Reference Le Voyer, Kelley, Cottrell and Hauri52 and northern Iceland (Figure 9.3).Reference Hauri53 Importantly, these three melt inclusion suites are dominated by inclusions that have not formed post-entrapment vapor bubbles, such that the pre-eruptive magmatic CO2 contents are preserved in the glass. These data are supplemented by an additional 113 melt inclusions that have been measured for CO2 but show evidence of degassing; these come from the Gakkel Ridge,Reference Shaw, Behn, Humphris, Sohn and Gregg54, Reference Wanless, Behn, Shaw and Plank55 the Juan de Fuca Ridge,Reference Wanless and Shaw56 the Lucky Strike segment of the Mid-Atlantic Ridge,Reference Wanless57 and selected melt inclusions from Axial Seamount on the Juan de Fuca Ridge.Reference Helo, Longpré, Shimizu, Clague and Stix58 This entire set of new and published data, from the paper of Le Voyer et al.,Reference Le Voyer47 is available as an electronic supplement to that paper, as well as for download at the EarthChem Library.Reference Le Voyer59
GeoMapApp figures showing MORB sample locations from the study of Le Voyer et al.Reference Le Voyer47 (red circles) and locations of previously published MORB samples (gray circles). Hot spots with estimated CO2 fluxes are shown as yellow stars.
Arctic Ocean basin;

Pacific Ocean basin;

Atlantic Ocean basin;

Indian Ocean basin.

CO2–Nb data for (a) vapor-undersaturated sample suites of submarine glasses and melt inclusions and (b) individual MORBs, with undersaturated sample suites from (a) represented by average compositions (boxes). The CO2/Nb = 398 line represents the average of the individual average CO2/Nb ratio for each of the undersaturated samples suites. For any given sample, one can gain a sense of the extent of CO2 degassing by comparing the sample’s CO2/Nb ratio to the average CO2/Nb ratio of undegassed samples.

For hot spots, we rely heavily on an extensive new data set for Hawaiian melt inclusions reported by Marske and HauriReference Marske and Hauri60 consisting of major, trace, and volatile element data on 915 olivine-hosted melt inclusions in 29 samples of submarine-erupted pillow lavas and subaerial tephra deposits from the shield-building stage of five Hawaiian volcanoes (Figure 9.4). Tucker et al.Reference Tucker61 estimated bulk (dissolved + bubble) CO2 contents in 437 of the Hawaiian melt inclusions studied by Marske and Hauri.Reference Marske and Hauri60 These data are supplemented by melt inclusion data from other hot spot localities where shrinkage of bubble dimensions are reported, namely the Azores,Reference Métrich62 Canaries,Reference Longpré, Stix, Klügel and Shimizu63 and Iceland.Reference Hartley, Maclennan, Edmonds and Thordarson64, Reference Neave, Maclennan, Edmonds and Thordarson65 The compiled OIB melt inclusion dataset may be downloaded from the EarthChem Library.Reference Tucker66 We emphasize, however, that none of the microbeam and statistical methods applied to melt inclusions can account for CO2 lost from magmas prior to melt inclusion entrapment in their crystal hosts.
Probability distribution of the ratio of vapor bubble volume to total melt inclusion volume for vapor bubbles in melt inclusions from HawaiiReference Tucker61 and Iceland.Reference Hartley, Maclennan, Edmonds and Thordarson64, Reference Neave, Maclennan, Edmonds and Thordarson65 Bubbles in Iceland melt inclusions strongly peak near 1.5 vol.%, whereas bubbles in Hawaiian melt inclusions are more variable and peak around 3 vol.%.

9.3 Fluxes of CO2 from the Global Mid-Ocean Ridge System
The samples compiled by Le Voyer et al.Reference Le Voyer47 were assigned to individual ridge segments based on a comparison of their sampling coordinates with the global spreading center segment catalog of Gale et al.Reference Gale, Dalton, Langmuir, Su and Schilling36 (Figure 9.2). Segment-average volatile concentrations were calculated for all segments that contained an on-axis sample analyzed for H2O, CO2, F, S, and/or Cl. Segment averages for the volatile data were corrected to MgO = 8 wt.% and to compositions in equilibrium with Fo90 olivine in order to compare with the fractionation-normalized database of Gale et al.Reference Gale, Langmuir and Dalton17 We note that correction for crystal fractionation cannot account for the irreversible loss of CO2 by degassing, although we estimate primary magma CO2 contents using Ba and Rb as proxies, as explained below.
MORBs are erupted under hydrostatic pressure on the seafloor, determined by the water depth at the site of MORB collection. The solubility of CO2 in MORBs is strongly dependent on pressure,Reference Dixon and Stolper37, Reference Pan, Holloway and Hervig41 and so dissolved CO2 concentrations in MORBs correlate with the pressure of eruption on the seafloor.Reference Dixon, Stolper and Delaney67 However, many MORBs have dissolved CO2 contents in excess of the equilibrium solubility of CO2 predicted at the eruption pressure. This oversaturation is due to rapid melt ascent during eruption, during which diffusion of carbon in the melt is too slow to maintain equilibrium between the dissolved CO32– in the melt and the CO2 vapor within the vesicles (Figure 9.1b).Reference Dixon and Stolper37, Reference Javoy and Pineau38, Reference Dixon, Stolper and Delaney67–Reference Chavrit, Humler, Morizet and Laporte69
The CO2–H2O solubility model of Dixon et al.Reference Dixon, Stolper and Holloway39 has typically been used to constrain vapor saturation pressure (Psat) in MORBs because they determined CO2 and H2O solubility specifically on MORB melt compositions. When we use measured CO2 and H2O contents in MORBs to calculate Psat using the Dixon et al.Reference Dixon, Stolper and Holloway39 model and compare these with the pressures of MORB sample collection (Psample), a subset of the data scatter around the 1:1 line (line in Figure 9.5) where vapor saturation pressures are close to those corresponding to the sampling location. A large number of samples are also demonstrably oversaturated in a CO2–H2O vapor and thus plot to high values of Psat (above line in Figure 9.5), in good agreement with the presence of vapor bubbles in almost all MORB samples.Reference Chavrit, Humler, Morizet and Laporte69
Pressure of MORB sample collection on the seafloor (Psample) versus pressure of vapor saturation (Psat) calculated from the CO2 and H2O contents of MORB samples using the vapor solubility model of Dixon et al.Reference Dixon, Stolper and Holloway39 The 1:1 line (Psat = Psample) corresponds to equilibrium of CO2–H2O contents at seafloor pressures. The pink field encloses the Psat values calculated from the solubility model of Ghiorso and Gualda,Reference Ghiorso and Gualda71 which produces Psat estimates 20% lower than those of Dixon et al.;Reference Dixon, Stolper and Holloway39 the green field encloses the Psat values calculated from the solubility model of Papale et al.,Reference Papale, Moretti and Barbato70 which are 60% lower than those of Dixon et al.Reference Dixon, Stolper and Holloway39

Two other solubility models are often used as alternatives to the Dixon et al.Reference Dixon, Stolper and Holloway39 model: Papale et al.Reference Papale, Moretti and Barbato70 and Ghiorso and Gualda.Reference Ghiorso and Gualda71 The model of Papale et al.Reference Papale, Moretti and Barbato70 (green field in Figure 9.5) produces Psat values that are 60% lower on average compared with Dixon et al.,Reference Dixon, Stolper and Holloway39 which suggests that most MORBs are vapor undersaturated. Vesicle abundances observed in MORBs, however, contradict this predictionReference Chavrit, Humler, Morizet and Laporte69 because samples that are vapor undersaturated should not have vesicles.Reference Saal, Hauri, Langmuir and Perfit49, Reference Michael and Graham50, Reference Le Voyer, Kelley, Cottrell and Hauri52 The Papale et al.Reference Papale, Moretti and Barbato70 model thus likely underestimates vapor saturation pressures in MORBs. The Papale et al.Reference Papale, Moretti and Barbato70 model was calibrated over a very wide range in melt compositions, whereas the Dixon et al.Reference Dixon, Stolper and Holloway39 model was calibrated specifically on MORB compositions, so disagreement between these models is perhaps not surprising. More recently, the CO2–H2O solubility model of Ghiorso and GualdaReference Ghiorso and Gualda71 (pink field in Figure 9.5) results in Psat values that are 20% lower on average compared with Dixon et al.,Reference Dixon, Stolper and Holloway39 producing good agreement with Psample values. The Ghiorso and GualdaReference Ghiorso and Gualda71 model thus also predicts more samples than Dixon et al.Reference Dixon, Stolper and Holloway39 to be vapor undersaturated. Both models provide reasonable estimates of vapor saturation pressures, but we prefer the model of Dixon et al.Reference Dixon, Stolper and Holloway39 because it is calibrated specifically for MORBs and it predicts that fewer MORBs are vapor undersaturated, which is consistent with the presence of vesicles in the vast majority of MORBs.
9.3.1 MORB Melt Inclusions and the Usefulness of Volatile/Nonvolatile Element Ratios
Because CO2 has degassed irreversibly from most MORBs, the flux of CO2 from the MORB mantle is often determined by establishing the mantle ratio of CO2 to another element whose flux is well constrained. An element often used this way is helium, as the upper mantle 3He flux is determined from oceanographic measurementsReference Craig, Clarke and Beg72–Reference Holzer74 or measurements in MORBs.Reference Tucker, Mukhopadhyay and Gonnermann75 CO2/3He ratios measured in MORBs must be corrected for fractionation during magmatic degassing, and an equilibrium degassing model is often used for this purpose.Reference Marty and Tolstikhin10, Reference Burnard76, Reference Colin, Burnard and Marty77 However, Tucker et al.Reference Tucker, Mukhopadhyay and Gonnermann75 showed that MORB degassing is influenced by kinetic effects not accounted for in equilibrium degassing models. Using a disequilibrium degassing model, Tucker et al.Reference Tucker, Mukhopadhyay and Gonnermann75 estimated a mantle CO2 flux of 59 Tg/yr, although the extent of CO2 fractionation from other volatile elements during degassing and the total mantle CO2 flux strongly depend on poorly constrained diffusivities of carbon in basaltic melt.
To avoid reliance on degassing models and uncertainties in their critical parameters, we seek undegassed samples to develop nonvolatile proxies for the CO2 contents of primary mantle melts (melts in equilibrium with mantle olivine of Fo90). We therefore turn to melt inclusions. By virtue of being encapsulated in rigid host minerals, melt inclusions gain additional protection from degassing, and their volatile concentrations are not constrained by eruption depth as described in the preceding section. Indeed, some suites of melt inclusions lack vapor bubbles and are well established as being vapor (and hence carbon) undersaturated. For a proxy, we turn to highly incompatible nonvolatile trace elements with similar behavior to carbon during partial melting of the mantle (Figure 9.1). Analyses of rare undegassed melt inclusions,Reference Saal, Hauri, Langmuir and Perfit49, Reference Le Voyer, Kelley, Cottrell and Hauri52, Reference Hauri53 rare undegassed MORBs,Reference Michael and Graham50, Reference Shimizu51 and experimental studiesReference Rosenthal, Hauri and Hirschmann78 establish that the nonvolatile elements Rb, Ba, Nb, and Th behave similarly to CO2 during mantle melting. In undegassed samples, CO2 concentrations correlate with Rb, Ba, Nb, and Th. Moreover, the CO2/Ba and CO2/Rb ratios of undegassed mantle-derived melts are approximately constant.Reference Hauri53 Two fundamental assumptions follow from these observations and underpin this contribution: (1) mantle processes do not fractionate carbon from Ba and Rb; and (2) MORB Ba and Rb concentrations can be used as proxies for CO2 in partially degassed MORBs and OIBs. Furthermore, we suggest that loss of CO2 by degassing can be detected by the presence (or lack thereof) of a correlation of CO2 with incompatible trace elements.
The first such correlation was observed for MORB melt inclusions from the Siqueiros Fracture Zone on the East Pacific Rise, where depleted MORB (D-MORB) is erupted at great water depth, inhibiting CO2 loss even from the erupted magmas.Reference Saal, Hauri, Langmuir and Perfit49 Subsequent studies of MORB melt inclusions, from less-depleted ridge segments and containing shrinkage bubbles, generally observed degassed magmas with no correlations of CO2 with other trace elements.Reference Shaw, Behn, Humphris, Sohn and Gregg54, Reference Wanless, Behn, Shaw and Plank55 Recently, two other suites of melt inclusions, showing CO2 correlating with Rb, Ba, Nb, Th, and other trace elements, have been reported from the Mid-Atlantic RidgeReference Le Voyer, Kelley, Cottrell and Hauri52 and northern Iceland.Reference Hauri53 These recently reported melt inclusion suites were free of vapor bubbles within the melt inclusions, with the exception of the most enriched of the Iceland melt inclusions, for which bubble measurements were made to account for the influence of their CO2 budget on the overall CO2 content of the inclusions.Reference Hauri53 Additionally, Michael and GrahamReference Michael and Graham50 and Shimizu et al.Reference Shimizu51 identified limited suites of deeply erupted D-MORB glasses that also show correlations of CO2 with incompatible trace elements. Hauri et al.Reference Hauri53 showed that vapor-undersaturated MORBs have average CO2/Rb (991 ± 26%) and CO2/Ba (81.3 ± 56%) ratios that are more uniform than CO2/Nb or CO2/Th ratios. The ratio of the measured CO2/Ba ratio to the average CO2/Ba ratio for undegassed MORBs indicates that a majority of MORB samples have degassed >80% of their initial CO2 (Figure 9.3).
Although vapor-undersaturated MORBs with relatively uniform CO2/Ba, CO2/Rb, CO2/Th, and CO2/Nb ratios have mostly been interpreted to be undegassed, alternative interpretations have been explored that bear on how accurately these ratios record those in the mantle sources of MORB. Rosenthal et al.,Reference Rosenthal, Hauri and Hirschmann78 argued that scattering in observed CO2/Ba and CO2/Nb ratios in vapor-undersaturated MORBs could be due to undetected degassing of CO2 in amounts small enough to preserve CO2–Ba–Nb correlations. Matthews et al.Reference Matthews, Shorttle, Rudge and Maclennan79 modeled this undetected degassing as a partial degassing and mixing process, where near-fractional melts of the mantle are partially degassed at a specified depth and partially mixed. Despite being partially degassed, modeled melt mixtures were shown to retain positive correlations between CO2 and nonvolatile trace elements while delineating average CO2/nonvolatile trace element ratios that underestimated those of the mantle source. A defining characteristic of the model results from Matthews et al.Reference Matthews, Shorttle, Rudge and Maclennan79 is the prediction that sample suites that partially degas will show a gradual as opposed to a sharp transition from positive to negative Pearson correlation coefficients between CO2/El and 1/El ratios (where El is a nonvolatile trace element) with increasing compatibility of the El. Applying their model results to natural melt inclusion populations, Matthews et al.Reference Matthews, Shorttle, Rudge and Maclennan79 argued that Pearson correlation coefficients in the equatorial Mid-Atlantic Ridge melt inclusions reported by Le Voyer et al.Reference Le Voyer, Kelley, Cottrell and Hauri52 show a gradual transition from positive to negative correlation coefficients, and that the melt inclusions were partially degassed. They also argued that other MORB data sets are consistent with partial degassing, and that rather than the average CO2/Ba ratio in CO2-undersaturated MORBs, the maximum CO2/Ba ratio of ~140 provides a more reliable estimate of the CO2/Ba ratio of the mantle source.
The study of Shimizu et al.Reference Shimizu, Saal, Hauri, Perfit and Hékinian80 evaluated whether the partial degassing and mixing process described by Matthews et al.Reference Matthews, Shorttle, Rudge and Maclennan79 can reproduce the data for the olivine-hosted MORB melt inclusions from the Garrett and Siqueiros transform faults on the East Pacific Rise and found that it is difficult to fully explain their Pearson correlation coefficients between the CO2/El and 1/El ratios (Figure 9.6). To explain the observed coefficients, Shimizu et al.Reference Shimizu, Saal, Hauri, Perfit and Hékinian80 considered the effect of analytical uncertainties in the model by adding noise to the modeled concentrations of CO2 and EI, simulating the analytical uncertainties during measurements of the Siqueiros and Garrett melt inclusions. They demonstrated that when analytical uncertainties are considered, the correlations between the CO2/El and 1/El ratios of an undegassed model show a gradual transition from positive to negative similar to a partially degassed model (Figure 9.6). They argued that although partial degassing cannot be ruled out, it may not be required to explain the Pearson correlation coefficients in the Siqueiros and Garrett melt inclusions (Figure 9.6).
Pearson correlation coefficients for 1/El versus CO2/El (where El is an incompatible trace element) for Siqueiros and Garrett melt inclusions (MIs) plotted against the bulk partition coefficients of the EI.Reference Shimizu, Saal, Hauri, Perfit and Hékinian80 Garrett MIs have been filtered for anomalously depleted MIs with high analytical uncertainties. Bulk partition coefficients are from Rosenthal et al.Reference Rosenthal, Hauri and Hirschmann78 for C and Kelemen et al.Reference Kelemen, Yogodzinski, Scholl and Eiler173 for nonvolatile trace elements. Solid gray and black lines show correlation coefficients generated by the model of Matthews et al.Reference Matthews, Shorttle, Rudge and Maclennan79 conducted using the pHMELTS modelReference Ghiorso, Hirschmann, Reiners and Kress174–Reference Smith and Asimow176 to generate near-fractional melts of the DDMM from Workman and Hart,Reference Workman and Hart177 which were then mixed using the model of Rudge et al.Reference Rudge, Maclennan and Stracke178 The black and gray lines are the undegassed and partially degassed models, respectively, both of which do not include the analytical uncertainties. The black and gray dots are mean and 2σ uncertainties of Pearson correlation coefficients generated by the undegassed and partially degassed models ran 1000 times, both of which include analytical uncertainties ranging from 6% to 32% (2σ) depending on the element. In the partial degassing model, partial degassing is done using the CO2 solubility model of Shishkina et al.Reference Shishkina179 at 7 km in the oceanic crust underneath 4 km of seawater.

Isotopic heterogeneity within melt inclusion populations derived from individual samples has been reported by several studies,Reference Koornneef81–Reference Saal84 which can be explained only if the melt inclusion suite represents mixtures of melts from a heterogeneous source. Indeed, chemical and isotopic heterogeneity has been observed in MORBs by numerous studies, suggesting generation through melting of a heterogeneous mantle sourceReference Zindler and Hart25, Reference Schilling35, Reference Dupré and Allégre85–Reference Arevalo and McDonough87 (and see expanded discussion in Section 9.3.2). This heterogeneity typically takes the form of mixing between melts from depleted and enriched sources with low and high concentrations of trace elements, respectively. In this mixing scenario, trace element ratios like CO2/Ba and CO2/Nb are dominated by melts from the more enriched component with higher trace element abundances, while melts from the depleted source act to dilute the signature of melts from the enriched source. As a result, the population average of a ratio of two highly incompatible elements (such as CO2/Ba) might also be modified by mixing of melts from heterogeneous sources, having an important and unexplored effect on the modeling results of Matthews et al.Reference Matthews, Shorttle, Rudge and Maclennan79 While it is beyond the scope of this chapter to provide detailed discussion of all of the processes that lead to compositional heterogeneity at ridges and hot spots, we note that the Pearson correlation coefficients between the CO2/El and 1/El ratios for each of the vapor-undersaturated sample suites is not completely explained by either partial degassing and mixing or melting of a single mantle source (e.g. La, Ce, Nd, and Hf in Figure 9.6). Given this and the result that gradual changes in Pearson correlation coefficients between CO2/El and 1/El ratios from positive to negative may not require partial degassing (Figure 9.6), we postulate that average CO2/El ratios observed in vapor-undersaturated melt inclusions reflect those of the mantle source(s) involved in melting, and that these ratios apply to the entire global ridge system.
9.3.2 Variations in Primary MORB CO2 Contents and CO2 Fluxes
We describe here the central methodology of this contribution. Trace elements vary among MORB segments by a factor of 20, and this cannot be explained by variations in the extent of melting,Reference Gale, Langmuir and Dalton17, Reference Klein and Langmuir20, Reference Langmuir, Klein, Plank, Morgan, Blackman and Sinton21 but instead requires mantle heterogeneity. At the segment,Reference Donnelly, Goldstein, Langmuir and Spiegelman34 basin,Reference Schilling35 and global scales,Reference Gale, Dalton, Langmuir, Su and Schilling36 trace element ratios correlate with some, but not all, radiogenic isotopes. For example, segment average Ba/Nb ratios correlate with 87Sr/86Sr.Reference Gale, Dalton, Langmuir, Su and Schilling36 While correlations between trace elements and isotopes can be complex because isotopes represent time-integrated histories and trace elements are highly sensitive to recent fractionation events, trace elements are highly correlated among themselves. Carbon – another trace element – has been shown to correlate with the highly incompatible trace elements and varies on an order that exceeds that which can be produced by ridge processes. By analogy with trace elements, we consequently infer large variations in primary mantle CO2 concentrations due to mantle heterogeneity, which we calculate on the segment scale below.
The restricted ranges in CO2/Ba and CO2/Rb in undegassed MORBs permit an estimation of the initial primary CO2 concentrations of MORB magmas, given an estimate of their primary trace element abundances. Gale et al.Reference Gale, Langmuir and Dalton17, Reference Gale, Dalton, Langmuir, Su and Schilling36 provide such estimates based on the compositions of MORB magmas averaged by ridge segment, and corrected for shallow-level crystal fractionation to MgO = 8 wt.% and also further corrected to be in equilibrium with Fo90 olivine. Le Voyer et al.Reference Le Voyer47 estimated primary MORB magma CO2 for 387 segments that contain data for Ba and/or Rb out of the 711 MORB spreading ridge segments identified by Gale et al.Reference Gale, Dalton, Langmuir, Su and Schilling36 These segment-average concentrations were then multiplied by the undegassed-average ratios from Hauri et al.Reference Hauri53 of CO2/Rb (991) and CO2/Ba (81.3) to calculate segment-averaged primary CO2 concentrations. Unsurprisingly, the segment-average primary CO2 contents (as derived from Ba and Rb) correlate well with segment-averaged primary Th and K2O (Figure 9.7). These CO2 abundances range widely, from a low of 104 ppm CO2 at a depleted segment in the Galapagos Spreading Center to a high of 1.90 wt.% CO2 at a highly enriched segment of the Juan de Fuca Ridge. The distribution of primary melt CO2 contents is strongly skewed with a near log-normal distribution. The median CO2 content is 1107 ppm and the mean is 2139 ppm. To determine the mode (“most likely” CO2 content), we applied a kernel density analysis with an optimal bandwidth of 318 (Bowman & Azzalini, 1997Reference Bowman and Azzalini182), and obtained 621 ppm. This preferred value for “typical primary MORB” (621 ppm CO2) is similar to the analytical mode (486 ppm), which assumes a true log-normal distribution. (Note that the original printing of Le Voyer et al. (2019)59 incorrectly referred to 2100 ppm as median primary CO2. This contribution revises that error.)
Segment-averaged primary MORB compositions corrected for low-pressure fractionation to equilibrium with Fo90 olivine. CO2(Ba)90 versus (a) Th90 and (b) K2O90 from the study of Le Voyer et al.,Reference Le Voyer47 who estimated primary magma CO2 contents from the average MORB CO2/Ba ratio (81.3) and segment-average Ba90 concentrations.

Nearly five decades of deep-water oceanographic research, coupled with ocean drilling by the Deep Sea Drilling Program (DSDP) and the International Ocean Drilling Program (IODP), have demonstrated a limited range of crustal thickness of 6 ± 1 km,Reference Klein and Langmuir20, Reference Behn and Grove88, Reference White, McKenzie and O’Nions89 and at variable spreading rates that are well determined globally.Reference DeMets, Gordon, Argus and Stein90 Given the relationship between crustal thickness and seafloor depth,Reference Klein and Langmuir20, Reference Behn and Grove88, Reference Asimow, Hirschmann and Stolper91 magma production rates at mid-ocean ridges are readily calculated from the product of ridge length, crustal thickness, and spreading rate.Reference Crisp11 Behn and GroveReference Behn and Grove88 and Van Avendonk et al.Reference Van Avendonk, Davis, Harding and Lawver92 summarized seismically determined measurements of crustal thickness at ridge axes, which varies from 2 km at the slow-spreading Gakkel Ridge to 30 km at central Iceland and is a product of the local magma production rate and spreading rate. The crustal thickness of ridge segments correlates with ridge depth (reflecting isostatic compensation of MORB crust at the ridge axis). With this, Le Voyer et al.Reference Le Voyer47 estimated the crustal thickness at all of the 711 MORB segments defined by Gale et al.Reference Gale, Dalton, Langmuir, Su and Schilling36 based on the axial depth. Le Voyer et al.Reference Le Voyer47 then determined a segment-average magma production rate by multiplying the crustal thickness by the segment spreading rate, which was then used to calculate the fluxes of CO2 (and other elements) on a segment-by-segment basis.
CO2 fluxes, normalized by ridge length, vary by a factor of >100 because they are the product of local primary magma CO2 content and local magma production rate, which are uncorrelated. Fluxes of CO2 vary from 1.52 × 106 to 4.74 × 108 mol/yr/km. High CO2 fluxes are typically correlated with high primary CO2 in the magma (Figure 9.8), reinforcing the observations made above that variations in the MORB primary magma CO2 content are mostly driven by variations in mantle composition rather than magma production dynamics or mantle temperature.Reference Le Voyer47
MORB segment-average CO2 flux normalized by ridge length (mol/yr/km) versus (a) crustal thickness and (b) primary CO2 concentration estimated from the average MORB CO2/Ba ratio (81.3) and segment-average Ba90 concentrations.Reference Le Voyer47 Estimated CO2 fluxes are more strongly correlated with mantle composition than with crustal thickness.

Integrated over the length of the global mid-ocean ridge system, the global MORB magma production rate is 16.5 km3/yr (lower than the estimate of 21 km3/yr from CrispReference Crisp11), and the total flux of CO2 is 1.32 × 1012 mol/yr.Reference Le Voyer47 This estimate is significantly higher than that of Saal et al.,Reference Saal, Hauri, Langmuir and Perfit49 which is based on depleted Siqueiros Fracture Zone melt inclusions. Our estimate is lower than the estimate of Cartigny et al.,Reference Cartigny, Pineau, Aubaud and Javoy48 which is based on enriched MORBs from 14°N and 34°N on the Mid-Atlantic Ridge. Marty and Tolstikhin,Reference Marty and Tolstikhin10 Michael and Graham,Reference Michael and Graham50 and Hauri et al.Reference Hauri53 give CO2 fluxes that are nearly a factor of two higher than the integrated value summed over the detailed fluxes determined on a segment-by-segment basis by Le Voyer et al.Reference Le Voyer47
The MORB flux of CO2 degassed at the ridge axis is ultimately dissolved into the overlying water column, either instantly by eruptive degassing (~80%) or later stripped out by hydrothermal circulation, where it becomes part of the marine bicarbonate cycle. Short-term global effects of large variations in the ridge CO2 flux are thus muted by the ~100,000 year residence time of carbon dissolved in seawater,Reference Broecker and Peng93 and thus variations in the ridge CO2 flux are unlikely to influence short-term surface climate conditions while the ridge is submerged. Only when the mid-ocean ridge system is exposed above sea level, such as at Iceland today, will there be a significant delivery of CO2 from the mid-ocean ridge mantle directly into the atmosphere. On longer timescales, the ridge CO2 flux is dwarfed by the deposition of carbonate sediments and organic carbon on the seafloor and of carbonate alteration veins within the crustal and mantle sections of the oceanic lithosphere, which make the seafloor a net sink for carbon by a large factor compared with its initial magmatic budget.Reference Kelemen and Manning8, Reference Alt and Shanks94–Reference Hayes and Waldbauer96
9.4 Fluxes of CO2 from Mantle Plumes
The next most important flux of CO2 from the convecting mantle occurs at intraplate hot spots, where deeper sources of carbon penetrate through the upper mantle and oceanic lithosphere to produce seamounts and volcanic chains (Figure 9.1c). The flux of carbon from the deep mantle is of critical importance to mantle geochemistry and the study of planetary volatiles. Whereas mid-ocean ridges sample the uppermost mantle, some ocean intraplate volcanics sample the deeper mantle via upwelling plumes,Reference French and Romanowicz97, Reference Montelli, Nolet, Dahlen and Masters98 which could represent a significant reservoir of terrestrial carbon and other volatile elements. Hot spot magma fluxes are similarly calculated from the volumes of the individual volcanoes in the chain; however, weathering of the volcanic edifice and poorer chronologic information on the duration of volcanism result in greater relative uncertainties for these fluxes. The flux of CO2 from oceanic intraplate volcanics is generally considered to be much smaller than the mid-ocean ridge flux.Reference Dasgupta and Hirschmann7, Reference Marty and Tolstikhin10,99 However, many oceanic intraplate volcanics erupt subaerially, so their CO2 emissions are injected directly into the atmosphere. Furthermore, many modern-day plumes are linked to LIPs,Reference Richards, Duncan and Courtillot100 and gas emissions from eruption of these OIB progenitors can have profound and devastating environmental effects.Reference Kiehl and Shields101
Modern-day volatile fluxes can be directly measured at active volcanic centers by analysis of volcanic gas, and this subject is discussed in depth in Chapter 8. Here, we focus on quantification of long-term integrated fluxes from oceanic intraplate volcanics. These are most typically quantified by combining estimates of primary magmatic CO2 concentrations with magmatic fluxes. Two broad strategies are used to estimate pre-degassing CO2 concentrations: using maximum measured CO2 concentrations; and using a degassing model to calculate pre-degassing concentrations. The disadvantage of using degassing models is that they require additional constraints such as noble gas or carbon isotopic measurements to quantify the extent of gas loss, and the results may be sensitive to the assumed degassing model and its physical parameters. Using maximum measured values estimated from melt inclusions does not require assumptions about degassing processes, but can only provide lower limits on pre-degassing concentrations.
The heat flux produced by oceanic intraplate hot spots amounts to only 6–10% of the total surface heat flux;Reference Sleep102, Reference Davies103 however, OIB magmas produced at hot spots are known to have enriched abundances of volatile elementsReference Dixon and Clague104–Reference Workman, Hauri, Hart, Wang and Blusztajn107 that may contribute to a CO2 flux in excess of that inferred from their relative heat fluxes. However, enriched CO2 contents can also result in deeper degassing of CO2 and thus many submarine-erupted OIBs have likely lost a greater fraction of their initial CO2 compared with MORBs erupted under the same submarine water pressures.Reference Hilton, Thirlwall, Taylor, Murton and Nichols108 As a result, like the situation for MORBs, submarine glasses at hot spots have lost the majority of their CO2.
Melt inclusions once again provide the means to examine the CO2 content of OIB magmas. The recent study of Marske and HauriReference Marske and Hauri60 provides a definitive data set of volatiles in Hawaiian melt inclusions, adding to the smaller datasets of Refs. Reference Hauri109–Reference Sides, Edmonds, MacLennan, Swanson and Houghton111. However, for CO2, these prior studies were all complicated by the presence of vapor bubbles within melt inclusions, which were not accounted for in determining inclusion CO2 concentrations. Tucker et al.Reference Tucker61 examined hundreds of bubbles in Hawaiian melt inclusions and quantified the uncertainties associated with reconstructing total (bubble + dissolved) CO2 in the melt inclusions. Given the proportion of bubble/inclusion volume (which strongly peaks at 3.3% for Hawaiian inclusions; Figure 9.4), an estimate of the bubble formation pressure determined from the dissolved H2O and CO2 contents of the glass phase, a bubble closure temperature, and an equation of state for CO2 vapor, it is possible to reconstruct the total CO2 content of the inclusion. At Hawaii, these CO2 contents range over hundreds of ppm to several wt.% CO2; however, much of this range is likely due to degassing, and only the highest CO2 contents are likely to be representative of primary (undegassed) CO2 contents. Tucker et al.Reference Tucker61 estimated an average primary Hawaiian magma CO2 content of 7000 ppm (Table 9.1) and a modern CO2 flux for Hawaii of 8.6 × 1010 mol/yr. This flux represents approximately 5% of the MORB CO2 flux,Reference Le Voyer47 while the magma flux at Hawaii (0.2 km3/yr) represents only 1% of the MORB magma flux, a comparison that highlights the enriched nature of CO2 in Hawaiian magmas.
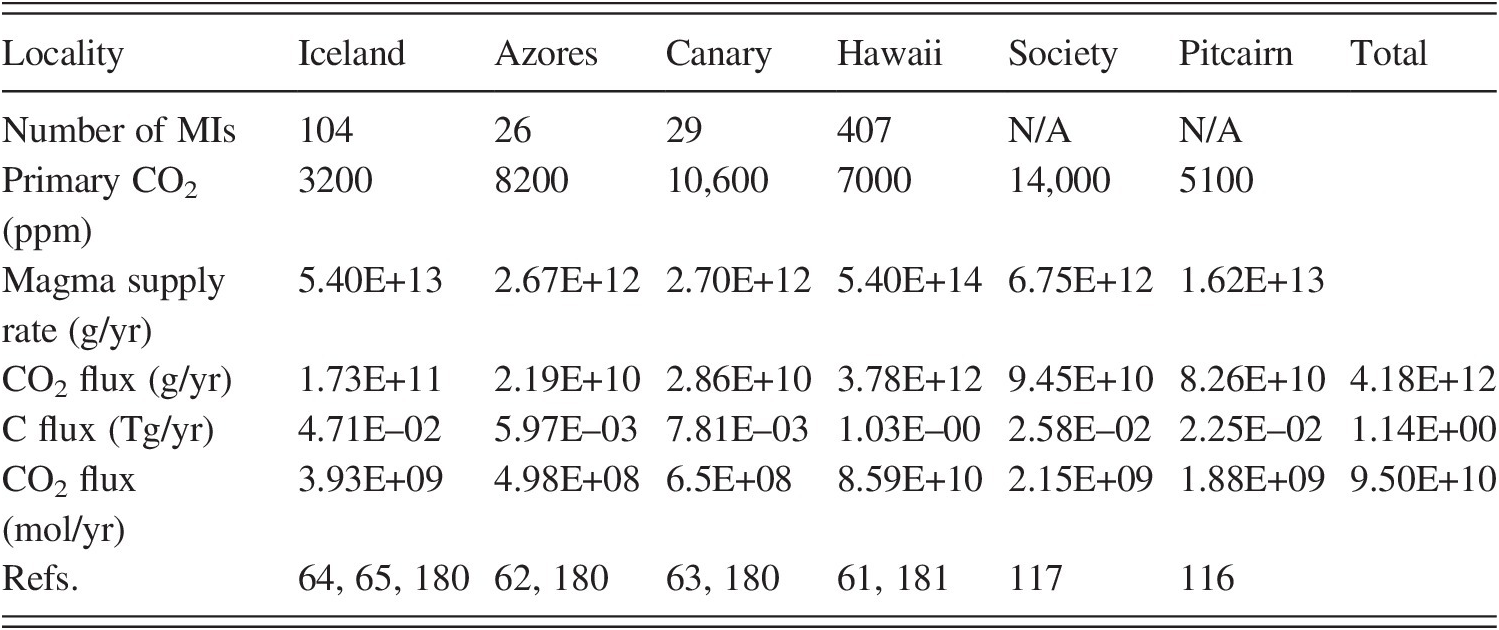
MI = melt inclusion; N/A = not applicable.
The melt inclusion-based estimates compare well to prior published estimates of Hawaiian magma primary CO2 contents and volcanic fluxes. By combining dissolved and vesicular CO2 concentrations, Dixon and ClagueReference Dixon and Clague104 found maximum CO2 concentrations of up to 6300 ppm in submarine Loihi basalts. Using a noble gas-based disequilibrium degassing model, Gonnermann and MukhopadhyayReference Gonnermann and Mukhopadhyay112 estimated pre-degassing concentrations of between 1100 and 6300 ppm in basalts from Loihi and Kilauea. However, those results may be sensitive to model parameters such as the magmatic carbon diffusivity. The long-term rate from melt inclusions is similar to modern-day emission rates measured from the Kilauea summit.Reference Anderson and Poland113–Reference Poland, Miklius, Sutton and Thornber115
Given knowledge of the H2O and CO2 contents of glassy melt inclusions and vapor bubble sizes, the same methodology as applied to the Hawaiian inclusions can also be applied to published data sets from other hot spots in order to calculate their primary magma CO2 contents and CO2 fluxes. Melt inclusion volatile and bubble size data are available from the Azores,Reference Métrich62 Canary,Reference Longpré, Stix, Klügel and Shimizu63 and IcelandReference Hartley, Maclennan, Edmonds and Thordarson64, Reference Neave, Maclennan, Edmonds and Thordarson65 hot spots; primary magmatic CO2 contents of these hot spots are conservatively estimated as the 95th percentile of CO2 contents from the data sets. Additionally, we include estimates based on analyses of submarine glasses of the Society and Pitcairn hot spots.Reference Aubaud, Pineau, Hekinian and Javoy116, Reference Aubaud, Pineau, Hekinian and Javoy117 The primary CO2 contents of these hot spot OIBs range from 3200 ppm at Iceland to 10,600 ppm CO2 at the Canaries, suggesting a large degree of heterogeneity in primary CO2 contents between hot spots (Table 9.1). However, each of the individual CO2 fluxes of the smaller hot spots is more than an order of magnitude lower than the CO2 flux from Hawaii.
In their bathymetric studies of Pacific seamounts and islands, Batiza,Reference Batiza118 Wessel and Lyons,Reference Wessel and Lyons119 and Kim and WesselReference Kim and Wessel120 estimated the total volume of oceanic intraplate magmatism to be 1–2 × 107 km3, which equates to an extra 100–200 m (1.5–3.0%) of crustal thickness across the entirety of the Pacific plate. If this relationship holds for all of the ocean basins, then even with a primary magma CO2 content of 1 wt.% (similar to the highest OIB primary CO2 contents; Table 9.1), this would represent an additional 12–25% atop the ridge flux, or 0.17–0.35 × 1012 mol/yr (or two to four times the output of Hawaii; Table 9.1). Thus, while the overall intraplate CO2 output is not insignificant compared with the ridge flux, long-term global variations in the CO2 flux from ridges alone are no doubt of a similar magnitude given the spreading rates and total ridge length changes during plate tectonic evolution. And because most of the intraplate volcanism in the oceans is below sea level, the CO2 outgassed at all but the largest hot spots is fated for dissolution into seawater, and, like the MORB flux, it is ultimately precipitated as carbonate sediments and carbonate vein fillings on the seafloor.
LIPs represent an exceptional source of hot spot-related magmatism due to their punctuated eruptions of enormous volumes over very short periods of time (~1 Myr) that approach the global MORB production rate and the well-known correlation of ages of LIP eruption events with the timing of mass species extinctions.Reference Black, Hauri, Elkins-Tanton and Brown121–Reference Black and Manga122 Such eruptions have great potential to influence global climate on short timescales due to the likelihood that their volatile inventory was delivered directly to the atmosphere; this is particularly acute for the emission of SO2 during LIP volcanism, as the atmospheric abundance of SO2 is typically low and its ability to absorb or reflect UV radiation is very high. Thus, LIP SO2 emissions (of the order 1013 mol/yr over decadal timescales) are enormous compared with the SO2 budget of the atmosphere (of the order 1010 mol/yr) and can result in severe short-term cooling effects on Earth’s surface.Reference Self, Widdowson, Thordarson and Jay16, Reference Black, Hauri, Elkins-Tanton and Brown121 If flood basalts contain primary CO2 contents similar to OIBs, the emission of CO2 in a single decade-long eruption potentially represents a magnitude of the order 1013 mol/yr,Reference Self, Widdowson, Thordarson and Jay16 approximately ten times the modern emission rate from mid-ocean ridgesReference Le Voyer47 and representing an instantaneous decadal addition to atmospheric CO2 on the order of 1000 ppm. The greenhouse effect of such LIP CO2 emissions would be concurrent with LIP SO2 emissions. Increased SO2 would push conditions toward global cooling, but would have a shorter residence time in the atmosphere than CO2. Thus, the overall impact of LIP volcanism might be expected to strongly drop global temperatures while the volcanism is active and to strongly increase global temperatures following the cessation of eruptions. The evolution of end-member “hothouse” or “icehouse” climate conditions, triggered by LIP eruptions, could depend on whether the atmospheric system was already close to a tipping point in one direction or the other.Reference Self, Widdowson, Thordarson and Jay16, Reference Black and Manga122, Reference Ward126 Nevertheless, both SO2 and CO2 emissions from LIPs would ultimately lead to intense ocean acidification that could be an additional driver in triggering mass extinctions associated with LIP volcanism.
9.5 Carbon Content of Convecting Mantle Sources
The large range of primary MORB and OIB magma CO2 contents at the segment scale is due to a combination of variations in degree of melting, as well as variations in mantle CO2 abundance. The mean degree of melting at a given ridge segment can be evaluated by consideration of crustal thickness. For the standard model of passive upwelling at oceanic spreading centers, Klein and LangmuirReference Klein and Langmuir20 describe the relationships between crustal thickness (Dc), mean degree of melting (![]() ),mean pressure of melting (
),mean pressure of melting (![]() ), and final pressure of melting (Pf). The mean degree of melting can be calculated from the crustal thickness using the following relation:
), and final pressure of melting (Pf). The mean degree of melting can be calculated from the crustal thickness using the following relation:
 (9.1)
(9.1)where crustal thickness Dc has units of kilometers and ρc is the density of the oceanic crust in units of Tg/km3 (equivalent to g/cm3). With estimates of crustal thickness for each ridge segment derived from its relationship with ridge depth, we can use (9.1) to calculate the degree of melting at each segment and then invert the primary CO2 content for mantle CO2 using the batch melting equation:
 (9.2)
(9.2)where Cmantle is the mantle concentration, Cmelt is the melt concentration (in equilibrium with Fo90 olivine), and Dcarbon is the carbon bulk solid/melt partition coefficient. Because Dcarbon is much lower than ![]() , (9.2) reduces to a simple relationship between the degree of melting and mantle-melt concentrations:
, (9.2) reduces to a simple relationship between the degree of melting and mantle-melt concentrations:
Bearing in mind that the accuracy of the primary CO2 in MORB contents relies on our assumption that CO2/Ba and CO2/Rb are relatively invariant, we can use (9.3) to calculate mantle source concentrations of CO2 at each ridge segment. The calculations, carried out in the dataset of Cottrell et al. (2019),Reference Cottrell, Kelley, Hauri and Le Voyer183 predict that MORB mantle CO2 content ranges over two orders of magnitude from 10 ppm (segment GALA16) to 1980 ppm (segment JUAN1; Figure 9.9) and follows a log-normal distribution. For the 387 segments with trace element constraints, we estimate that the mean mantle CO2 content is 205 ppm, the median is 99 ppm, and the mode (estimated using a kernel density analysis as above), is 73 ppm. Segment-specific values are tabulated in Cottrell et al. (2019).Reference Cottrell, Kelley, Hauri and Le Voyer183 If we assume that the segments without trace element constraints have “typical” primary melt CO2 contents, constrained by either the distribution mode (621 ppm) or the median (1107 ppm), then the mode of mantle CO2 distribution is 50 or 104 ppm, respectively. The Arctic ridges are noteworthy in having uniformly high estimates of mantle CO2. There are additional isolated ridge segments with high estimated mantle CO2 concentrations that are far from hot spots, notably just north of the equatorial Mid-Atlantic Ridge (MARR168), much of the American–Antarctic Ridge, several slow-spreading segments of the Southwest Indian Ridge, and the northernmost Red Sea. Apart from these scattered locations, there is a strong geographic correlation of high mantle CO2 at ridge segments that occur near hot spots (Iceland, Azores, South Atlantic hot spots, Reunion, Afar, Easter Island, Galapagos). All of the ridge segments with >700 ppm mantle CO2 are located within 1000 km of the nearest hot spot, which may indicate that hot spots have elevated concentrations of CO2 in their mantle sources. This inference is predicated on our underlying assumption that the mantle has uniform CO2/Ba and CO2/Rb ratios.Reference Le Voyer47, Reference Hauri53 If hot pots sample recycled mantle that has had carbon added to it or stripped from itReference Dasgupta and Hirschmann7, Reference Kelemen and Manning8 relative to other trace elements, our assumption would not hold and our inferences about primary CO2 at hot spots would not be valid. While this is an important topic for future inquiry, at this point there is no evidence to suggest that the CO2/Ba or CO2/Rb ratios at hot spots are significantly different from those of the MORB mantle.Reference Hauri53
Map of segment-average mantle source CO2 concentrations along the global mid-ocean ridge system. Mantle source CO2 concentrations were derived from segment-average primary CO2 abundances by estimating the degree of melting at each ridge segment and assuming batch melting (see Eqs. (9.1)–(9.3) in the text). High concentrations are observed near hot spots (yellow stars show approximate localities but are off-set so as to not obscure data), but also at isolated sections of ridge far from hot spots, particularly the Arctic ridges and the southern Indian Ocean ridges.
At hot spots, no simple relationship translates between volcano volume and degree of melting, as hot spots have widely different temperatures and buoyancy fluxesReference Sleep127, Reference Davies and Richards128 and erupt through oceanic lithosphere of widely variable age and thickness. Thus, the degree of melting must be determined in a different way from MORBs. Estimates of the degree of melting for the major Hawaiian volcanoes range from ~4% to 10%,Reference Norman and Garcia129, Reference Pietruszka, Norman, Garcia, Marske and Burns130 which, when combined with estimates of primary CO2 contents of individual volcanoes, yield mantle CO2 concentrations ranging from ~400 to 500 ppm.Reference Tucker61
Other hot spots appear to produce magmas that have high primary CO2 concentrations similar to Hawaii. Barry et al.Reference Barry, Hilton, Füri, Halldórsson and Grönvold131 estimated a CO2 concentration for the Iceland plume of 530 ppm, which is similar to the 590 ppm CO2 estimated by Le Voyer et al.Reference Le Voyer47 for the most CO2-rich ridge segment at Iceland. Similarly, high mantle CO2 contents are observed at hot spot-proximal ridge segments near Jan Mayen (1800 ppm), the Azores (1490 ppm), Cape Verde (830 ppm), Ascension (415 ppm), Discovery/Meteor (910 ppm), Shona (480 ppm), Bouvet (1160 ppm), Reunion (415 ppm), Afar (1420 ppm), Easter Island (350 ppm), Galapagos (850 ppm), and the Bowie hot spot on the Juan de Fuca Ridge (1980 ppm). At intraplate hot spots, mantle CO2 concentrations in the Society and Pitcairn hot spots have been estimated to be 700 ppmReference Aubaud, Pineau, Hekinian and Javoy117 and between 260 and 510 ppm,Reference Aubaud, Pineau, Hekinian and Javoy116 respectively. Despite the highly variable methods used, these hot spot mantle CO2 concentrations are distinctly higher than mantle concentrations at nearly all MORB ridge segments located >1000 km away from hot spots.Reference Le Voyer47
9.6 Carbon and Mantle Melting
Partial melting of the convecting mantle beneath ridges and hot spots is the key process and pathway by which carbon moves from Earth’s interior to the surface.Reference Dasgupta and Hirschmann7, Reference Kelemen and Manning8, Reference Dasgupta and Hirschmann132, Reference Keller, Katz and Hirschmann133 Thus far in this chapter, we have addressed the quantity and dynamic range of carbon present in the mantle and how it partitions between the crust and hydrosphere. We now explore carbon’s role in mantle melting and how it affects the flux of carbon reaching the ridge axis.
Decompression melting above the anhydrous solidus dominates the production of melt and oceanic crust at ridges.Reference Hirschmann134 However, the volume of mantle that experiences low degrees of partial melting far exceeds the volume above the anhydrous solidus and depends on the power of volatiles, primarily H2O and CO2, to flux mantle melting.Reference Dalton and Presnall135–Reference Dasgupta137 DasguptaReference Dasgupta138 provides an in-depth review and further exploration of near-solidus mantle melting in the presence of H2O and CO2. Here, we keep the focus on carbon and how its dynamic range in the mantle source relates to the flux of carbon reaching the ridge axis.
Three fundamental properties of carbon affect its fate during partial melting: (1) carbon is highly incompatible in mantle silicates;Reference Rosenthal, Hauri and Hirschmann78, Reference Shcheka, Wiedenbeck, Frost and Keppler139, Reference Keppler, Wiedenbeck and Shcheka140 (2) carbon is likely to exist in both oxidized and reduced forms within Earth’s mantle;Reference Dasgupta and Hirschmann7, Reference Stagno and Frost141–Reference Tsuno and Dasgupta147 and (3) oxidized carbon stabilizes melts, influencing the partial melting process itself.Reference Dasgupta and Hirschmann132, Reference Keller, Katz and Hirschmann133, Reference Dasgupta137, Reference Eggler, Baker, Akimoto and Manghnani148–Reference Massuyeau, Gardés, Morizet and Gaillard151 In mantle where oxygen fugacity (fO2) is low enough to stabilize graphite, carbon concentration in silicate melts, and hence the flux of carbon to the surface, will vary as a function of fO2, temperature, and pressure.Reference Armstrong, Hirschmann, Stanley, Falksen and Jacobsen145, Reference Holloway, Pan and Gudmundsson152–Reference Eguchi and Dasgupta154 Depending on the concentration of carbon in the source and degree of melting, graphite may be exhausted from the source during decompression melting under reduced conditions at quite low melt fractions.Reference Balhaus155 Such conditions may be relevant on reduced terrestrial bodies, such as Mars, but on Earth, basaltsReference Haggerty156–Reference Zhang, Cottrell, Solheid, Kelley and Hirschmann159, Reference O’Neill, Berry and Mallmann184 and their residuesReference Birner, Cottrell, Warren, Kelley and Davis160 record fO2 near the quartz–fayalite–magnetite (QFM) buffer and are too oxidized to stabilize graphite in their residues.
In a uniformly oxidized mantle, the stable form of carbon is carbonate. The solidus of carbonated peridotite is depressed by hundreds of degrees relative to the anhydrous solidus,Reference Dasgupta and Hirschmann132 is nearly independent of carbon concentration,Reference Dasgupta and Hirschmann161 and is sufficiently steep to allow carbonatitic melts to form all the way to the transition zone along convecting mantle geotherms.Reference Ghosh, Litasov and Ohtani162 The flux of carbon from the mantle to the surface in this scenario depends on the concentration of carbon in the source, with carbonatitic (~40 wt.% CO2) melts forming at melt fractions proportional to carbon concentration. The melt fraction across the carbonated solidus for a MORB mantle with median CO2 (~100 ppm) would be 0.03%, but could range from 0.0025% to 0.5% spatially, consistent with the dynamic range of mantle CO2 contents predicted here (10–2000 ppm). The addition of water to the system may stabilize carbonated silicate melts over carbonatitic melts to hundreds of kilometers;Reference Dasgupta137, Reference Dasgupta138 however, consideration of silica activity–composition relationships at greater depths predicts an abrupt transition from carbonatitic to carbonated silicate melts as shallow as 75–100 km along a 1350°C geotherm.Reference Massuyeau, Gardés, Morizet and Gaillard151 Regardless of the melt composition, in a uniformly oxidized mantle, deep carbon-rich melts would be widespread, but heterogeneously distributed at depth if mantle carbon concentrations are as variable as suggested by Le Voyer et al.Reference Le Voyer47
A uniformly oxidized mantle is unlikely on Earth, however. Theoretical and experimental considerations and observational evidence from continental xenoliths all suggest that the mantle becomes more reducing with depth relative to the QFM buffer.Reference Frost and McCammon142, Reference Rohrbach and Schmidt146, Reference Gudmundsson and Wood163–Reference Stagno, Ojwang, McCammon and Frost167 When fO2 becomes low enough to stabilize graphite/diamond over carbonate, carbonate-fluxed melting cannot proceed. The depth of the carbonate–graphite/diamond transition is uncertain, but may lie as shallow as 120 km due to increasing stabilization of the ferric iron component in garnet with pressure,Reference Stagno, Ojwang, McCammon and Frost167 or perhaps at depths closer to 250 km due to increasing modes of majoritic garnet and stabilization of Fe metal.Reference Rohrbach166 It should be noted that the xenolith record of iron redox comes entirely from continental lithospheric xenoliths rather than oceanic samples; thus, our limited knowledge of the redox-depth profile of the convecting mantle stands as a major limitation to our ability to predict the extent and consequences of carbon-fluxed melting at depth.
If we assume that the mantle becomes reduced quickly with depth and that the carbonate to graphite transition takes place at 120 km depth, then this depth will correspond to the depth of incipient melting. The depth at which melts are not only present, but interconnected and detectable with geophysical probes, will depend on carbon concentration. If we assume a threshold for melt interconnectivity and detection of 0.05 wt.% melt,Reference Minarik and Watson168 then Le Voyer et al.Reference Le Voyer, Kelley, Cottrell and Hauri52 showed how minimum CO2 concentrations of ~85 ppm are required at 120 km depth to generate detectable melts in anhydrous mantle with a potential temperature of 1345°C. The depth of interconnected, detectable melting will decrease as carbon concentration decreases. As the depth of the carbonate–graphite/diamond transition deepens, the importance of carbon concentration to the volume of mantle interconnected by melts increases. However, so long as mantle carbon concentrations are above 220 ppm, melts will be interconnected and detectable at all depths and carbon will be extracted as soon as the mantle becomes oxidized enough to stabilize carbonate, provided those melts can reach the ridge axis (see below). Thus, the depth range over which carbon-fluxed melting creates important geophysical signatures and actively increases the flux of carbon to the surface by enabling melting itself may be narrow. Indeed, in a mantle with only 140 ppm water, there may be no depth range over which carbon concentration strongly influences such signatures because of water’s ability to contribute to melt stabilizations.Reference Dasgupta138, Reference Sarafian, Gaetani, Hauri and Sarafian169
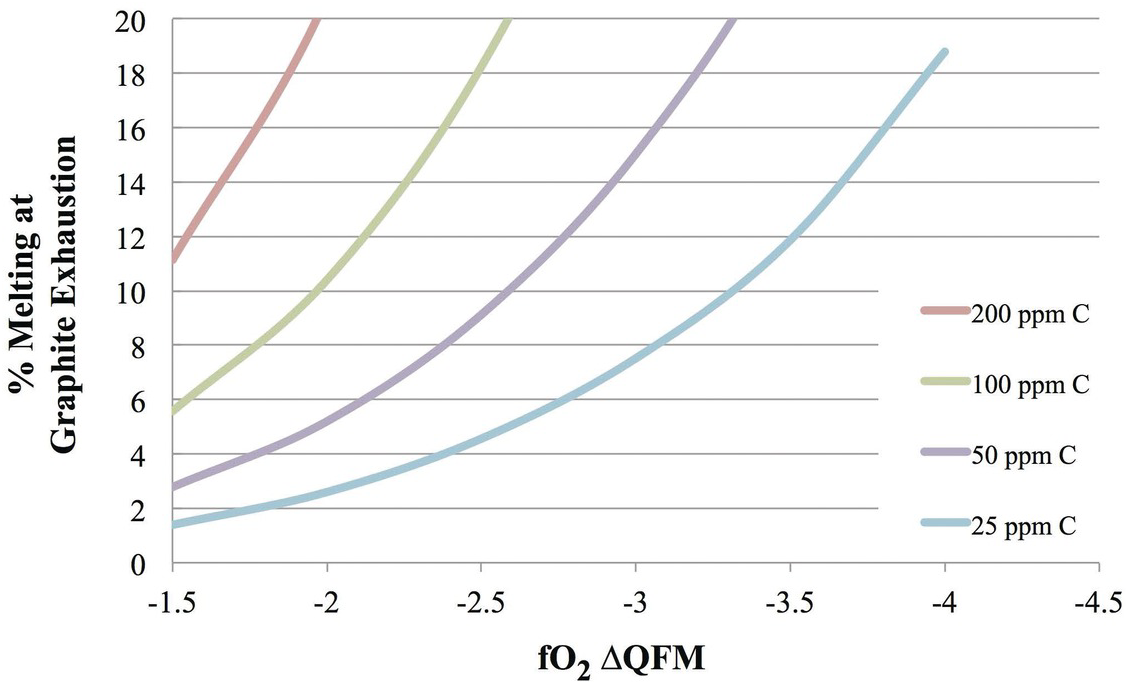
While the fO2 recorded by ridge lithologies suggests that the MORB source is too oxidized to retain graphite as a stable residual phase, the high concentration of CO2 dissolved in undegassed MORB provides further independent evidence of this. Figure 9.10 shows the extent of melting required to exhaust graphite during peridotite melting in the graphite stability field as a function of fO2 and the carbon content of the mantle (100 ppm CO2 corresponds to 27 ppm C). The fO2 range for graphite/diamond stability in the mantle corresponds to depths greater than 100 km and is taken from Stagno et al.Reference Stagno, Ojwang, McCammon and Frost167 For typical mantle with ~50–100 ppm CO2 (= 14–27 ppm C), graphite would be exhausted at reasonable melt fractions for MORBs (6–20%). In this scenario, we would expect carbon to behave as a highly incompatible element, just as we observe in some submarine glass and melt inclusion suites that are undersaturated in CO2–H2O vapor.Reference Saal, Hauri, Langmuir and Perfit49–Reference Hauri53 However, at higher mantle C contents, such as are evidenced by Le Voyer et al.Reference Le Voyer47 and presented here, melt fractions far in excess of 20% would be required to exhaust C from the residue and produce incompatible behavior for carbon. Such high melt fractions are unreasonable for MORB. Lower melt fractions would leave graphite (or diamond) present in the residue, and carbon would appear to behave as a moderately incompatible element, quite unlike the highly incompatible behavior seen in vapor-undersaturated MORB samples. Although there is a lack of detailed understanding of the fO2 with depth in the convecting mantle, the discovery of vapor-undersaturated samples at higher total carbon contents might illuminate whether or not graphite/diamond-saturated melting is an important process to consider at the deepest parts of the melting regime beneath ridges and hot spots.
Extent of melting required to exhaust graphite from the mantle as a function of the fO2 and the C content of the mantle. The solubility of total C in graphite-saturated silicate melt was taken from experimental studies.Reference Armstrong, Hirschmann, Stanley, Falksen and Jacobsen145, Reference Stanley, Hirschmann and Withers153, Reference Eguchi and Dasgupta154 At low mantle C contents, only small degrees of melting would be required to exhaust graphite, and C would behave similarly to highly incompatible elements (D ~0.02–0.04). At higher mantle C contents, C would appear only mildly incompatible (D ~0.1–0.2). For typical MORB melt fractions, we observe that C behaves as a highly incompatible element in vapor-undersaturated melt inclusions. This provides independent evidence that the MORB source is not graphite saturated; however, these curves could be relevant when considering more reduced planetary bodies, such as Mars.
Thus far, we have considered uniform partial melting, melt interconnectivity, and melt extraction. Keller et al.Reference Keller, Katz and Hirschmann133 show how melt focusing is only effective within and below the width defined by the anhydrous melting triangle, and that this may impact the fraction of mantle carbon in the melting regime that reaches the ridge axis. They show that deep volatile-fluxed melting may not influence melt focusing to the extent previously assumed,Reference Asimow and Langmuire136 but instead introduces spatial and temporal variability in the volatile flux reaching the ridge on timescales of 10,000 years. Keller et al.Reference Keller, Katz and Hirschmann133 predict that the concentrations of carbon (or any incompatible element) in primary MORB melts could vary as a function of time for any given source concentration due to reactive channelization. Furthermore, melts generated in the “wings” of the melt triangle collect along the base of the lithosphere and ultimately freeze, and up to half the melt volume produced within the melting regime may never make it to the surface at the ridge axis.Reference Keller, Katz and Hirschmann133 Because such melts do not contribute to oceanic crust production, their contribution does not factor into estimates of CO2 fluxes derived from geochemical and geophysical observations. The important implication for what we have presented here is that much of the heterogeneity we observe in incompatible trace elements on the dredge scale (hundreds of meters) within MORB may be generated by the melting process, rather than very-fine-scale variations in mantle source.Reference Keller and Katz170 Thus, when interpreting our segment-scale variations in mantle source globally, we must keep in mind how representative the concentrations presented here may be for poorly sampled segments and recognize that within segments it can be difficult to distinguish time-dependent variations in process from small-scale variations in composition.
Keller et al.Reference Keller, Katz and Hirschmann133 and Le Voyer et al.Reference Le Voyer47 provide the only estimates of segment-specific carbon flux. MORB mantle from Le Voyer et al.Reference Le Voyer47 has ~73 ppm (mode), 99 ppm (median) CO2 in the sources.Reference Cottrell, Kelley, Hauri and Le Voyer183 For such sources, the model of Keller et al.Reference Keller, Katz and Hirschmann133 returns global fluxes of 9.4 × 106 to 1.3 × 107 mol/yr/km (normalized to ridge length), which is comparable to the 1.4 × 107 mol/yr/km estimated in Le Voyer et al.Reference Le Voyer47 and this study. These length-normalized ridge fluxes are in excellent agreement despite the facts that Keller et al.Reference Keller, Katz and Hirschmann133 use a constant-source CO2 content and that the two models calculate crustal thicknesses differently. The average crust is 0.83 km thicker in the model output of Keller et al.,Reference Keller, Katz and Hirschmann133 who modeled magma production at variable potential temperatures constrained from a global-scale seismic tomography model,Reference Dalton, Langmuir and Gale171 producing a mean crustal thickness of 6.67 km. Le Voyer et al.Reference Le Voyer47 used the relationship of crustal thickness versus ridge depth observed at ridges with seismically determined crustal thickness measurements and extrapolated this relationship to calculate crustal thickness at all ridge segments from their axial depth, producing an average crustal thickness of 5.75 km despite calculating larger crustal thicknesses at hot spots compared with Keller et al.,Reference Keller, Katz and Hirschmann133 who capped their temperature input values at 1450°C.
When integrated over the entire length of ridges, weReference Cottrell, Kelley, Hauri and Le Voyer183 calculate a global CO2 flux of 1.3 to 1.5 × 1012 mol/yr, depending on whether the global mode or median CO2 content is applied to segments unconstrained by trace element data. Keller et al.Reference Keller, Katz and Hirschmann133 calculate a global flux of 1.20 × 1012 mol/yr. Despite the differences in methodology, these two studies are in excellent agreement on the integrated ridge CO2 flux, and this agreement emphasizes the depth of our understanding of CO2 fluxes at ridges.
At the same time, mantle heterogeneity must be considered at the more granular scale of individual ridge segments. Figure 9.11 highlights the geographic distribution of differences in segment-scale CO2 fluxes between our estimates, with variable-source CO2, and the Keller et al.Reference Keller, Katz and Hirschmann133 estimates. Observations of Sr, Nd, Pb, and Hf isotopic heterogeneity within individual ridge segments (length scales of tens to hundreds of kilometers) are often observed where measurements are sufficiently dense (see data sets of Gale et al.Reference Gale, Dalton, Langmuir, Su and Schilling36 and Class and LehnertReference Class and Lehnert172). It has long been recognized that mantle heterogeneity is the primary source of segment-scale variations in isotopes and highly incompatible trace elements (e.g. Langmuir et al.Reference Langmuir, Klein, Plank, Morgan, Blackman and Sinton21). Keller et al.Reference Keller, Katz and Hirschmann133 show that, for a homogeneous mantle, spreading rate and potential temperature dominate the flux of carbon at ridges, as these parameters dominate the production rate of magma itself. But the ridge CO2 flux is more sensitive to mantle carbon concentration than to mantle temperature, spreading rate, compaction length, or mantle fertility. The segment-scale CO2 fluxes that we have calculated (normalized by segment length) exhibit a variability that is twice that observed in the model of Keller et al.;Reference Keller, Katz and Hirschmann133 in our calculations of crustal thickness, we have implicitly included the effects of mantle temperature and magma production rate, since they also influence crustal thickness. In this rendering, it is clear that consideration of variability in mantle source CO2 concentrations results in much higher carbon fluxes near hot spots and lower carbon fluxes for the D-MORB mantle than those of the Keller et al.Reference Keller, Katz and Hirschmann133 model with a single-source composition. It is also clear that mantle source concentration has more influence on CO2 flux at the ridge than any other parameter under consideration.
Map of differences in segment-scale CO2 fluxes as estimated compared with fluxes calculated from the model of Keller et al.Reference Keller, Katz and Hirschmann133 The main differences are that Keller et al.Reference Keller, Katz and Hirschmann133 use a uniform mantle CO2 content of 100 ppm; while weReference Cottrell, Kelley, Hauri and Le Voyer183 estimate mantle CO2 contents by calculating primary magma CO2 and degree of melting at each segment, resulting in variations of mantle CO2 of more than three orders of magnitude (Figure 9.8).

9.7 Conclusions
The convecting mantle holds the vast majority of the carbon in the silicate Earth, and melting of this mantle at ridges and hot spots provides a means to assess the behavior of carbon in Earth’s interior. Studies of large numbers of samples of submarine volcanic glasses and melt inclusions utilizing accurate and precise high-throughput microbeam methods have uncovered suites of MORB samples that are undersaturated in CO2 and display correlated variations of CO2 with nonvolatile incompatible trace elements, with limited variations in CO2/Ba and CO2/Rb. These valuable sample suites have provided a means to assess the primary CO2 concentrations in MORBs at each ridge segment and to quantify, with stated uncertainties, the segment-scale and total flux of CO2 from mid-ocean ridges. We suggest that CO2 fluxes from the mantle to the surface vary by over three orders of magnitude at the segment scale.
9.8 Limits of Knowledge and Unknowns
A key advance in the future will be the discovery of more populations of CO2-undersaturated samples so that the assumed limited variability in CO2/Ba and CO2/Rb ratios and the origin of the variability of CO2/Nb and CO2/Th ratios can be critically assessed. It will likewise be advantageous to conduct rare gas and carbon isotope measurements on CO2-undersaturated sample suites in order to assess the degree of possible carbon isotope heterogeneity in the convecting upper mantle. While the knowledge of MORB mantle CO2 is now quite mature, we still lack the number of demonstrably vapor-undersaturated samples with which to assess one of our primary assumptions regarding MORB magmatism, namely the assumed homogeneity of MORB CO2/Ba and CO2/Rb ratios.
A critical examination of melt inclusions at Hawaii and other enriched hot spots requires knowledge of the CO2 content of ubiquitous vapor bubbles within these inclusions. We estimate that hot spot CO2 fluxes comprise 10–50% of the total MORB CO2 flux. The hot spot flux is large compared to the additional 1.5–3.0% magma flux from hot spot volcanism, emphasizing the significant enrichment of CO2 in hot spot mantle sources compared with the MORB mantle. Our understanding of hot spot CO2 is currently limited by the lack of vapor-undersaturated melt inclusion suites; it is not yet apparent whether melts that were undersaturated at the time of entrapment will be recognized once we can precisely account for the CO2 in melt inclusion shrinkage bubbles. However, this is presently a very active area of investigation not only at hot spots, but also at arcs, and thus conclusions (one way or the other) are likely not far behind the publication of this chapter.
LIPs can deliver enormous volatile fluxes over short time periods, although their climate impact is driven by the relative balance of SO2 over shorter timescales (years to decades) versus CO2 in the longer term. The pace of volcanism and relative fluxes of volatile species from LIPs remain largely unknown. So, too, does our knowledge of mantle CO2 fluxes going back in time.
Mantle carbon can, in its oxidized form, lower the solidus of silicates and generate mantle melts. At great depths in the mantle, fO2 is likely low enough to stabilize reduced forms of carbon that do not facilitate silicate melting. The shallow reaches of the upper mantle, however, are everywhere sufficiently oxidizing that carbonate is the stable phase. Thus, at some point during adiabatic ascent, the oxidation of carbon will initiate melting, and large variations in the CO2 content of the mantle will translate into variations in the melt fraction that may be observable by geophysical methods. In the future, large-scale geophysical experiments involving both seismic and electromagnetic studies at CO2-depleted and CO2-enriched ridge segments could reveal whether a redox-melting boundary exists in the sub-ridge mantle. At the same time, improved understanding of the variation of fO2 with depth in the convecting mantle should provide further tests of the behavior of the upper mantle in regions where melting is expected.
Acknowledgments
Erik Hauri worked relentlessly on this contribution, and many associated contributions, in the months, and even in the days, before he passed. We, his coauthors, dedicate this chapter to Erik – to his scientific vision, his insights, his magnificent data, his leadership, his spirit of discovery, his mentorship, and his friendship. We miss him terribly.
Many thanks to Jianhua Wang for expert assistance negotiating with cranky instruments in the Carnegie SIMS lab, to the Smithsonian Department of Mineral Sciences for assistance with sample preparation, to Mike Garcia and Aaron Pietruszka for provision of submarine Hawaii samples, and to Frank Trusdell and Jim Kauahikaua of the Hawaii Volcano Observatory (HVO) for going above and beyond the call of duty in making possible our sampling of Hawaiian cinder cones. We thank the geochemistry group at IPGP, University of Paris, for sharing data and samples from the Pacific–Antarctic Ridge, and the captain, crew, and scientific parties of the PACANTARCTIC 1 and 2 cruises (PAR), the GIMNAUT, CD127, and KNOX11RR cruises (CIR), the PROTEA-5, MD34, AG22, and AG53 cruises (SWIR), and the CHEPR and PANORAMA-1 cruises (EPR). We also acknowledge support from the University of California Ship Funds, and NSF grant OCE-0726573 (DRH). Conversations with Mark Behn, Pierre Cartigny, Raj Dasgupta, Marc Hirschmann, Peter van Keken, Peter Kelemen, Charles Langmuir, Terry Plank, Tobias Keller, and Richard Katz have all led to improvements in this manuscript. We thank Josh Wood for drafting Figure 9.1 and Glenn Macpherson for the Kilauea Iki melt inclusion photograph. We benefited from thorough and constructive reviews from Simon Matthews and Cyril Aubaud and from Raj Dasgupta’s editorial handling. This is a contribution to the Deep Carbon Observatory.
Questions for the Classroom
1 This chapter puts forth a methodology for quantifying global fluxes based on the assumption that CO2/Ba and CO2/Rb ratios are constant in Earth’s mantle. What processes might fractionate these ratios? How might the hypothesis that these ratios are relatively invariant be tested?
2 Other than identifying sample suites with constant CO2/Ba or CO2/Rb ratios, are there other ways to test whether samples have experienced pre-eruptive degassing?
3 How could new, promising samples to determine mantle carbon concentrations be discovered?
4 What factors limit our ability quantify the amount of carbon stored in the mantle?
5 What factors limit our ability to quantify the flux of carbon out of the mantle?
6 From the current mid-ocean ridge outgassing rate, how long is required to accumulate the mass of carbon in the crust, ocean, and atmosphere (~1 × 1023 g C)? How would this number change if carbon is returned to the mantle in subduction zones?
7 How might the carbon outgassing rate from mid-ocean ridges and hot spots have changed throughout Earth’s history?
8 Use the batch melting equation (9.2) to calculate the CO2 and Ba concentrations and CO2/Ba ratio of partial melts formed by melting a mantle with 100 ppm CO2 and 1 ppm Ba (using peridotite-melt partition coefficients of 0.00055 and 0.00012 for CO2 and Ba, respectively, and varying the degree of melting from 0% to 20%). At what degree of melting does the CO2/Ba ratio of the partial melt equal to that of the mantle source (100)? What can this tell us about the usefulness of CO2/Ba ratios measured in mid-ocean ridges and OIBs?
9 Look at the variation of other volatile species included in this chapter (H2O, Cl, F, or S). How do they vary as a function of major elements, as a function of incompatible trace elements, and as a function of location along the ridge system? How do these trends differ from those of CO2? How is their solubility in basalt different from CO2?
10 Given the potential for carbon to precipitate on melt inclusion bubble walls and thus be missed by quantification with Raman spectroscopy, what is the future promise of quantifying CO2 in melt inclusions that have nucleated bubbles?















