Introduction
Understanding wildlife–habitat relations is important for conservation and management (Graf et al. Reference Graf, Mathys and Bollmann2009; Wintle et al. Reference Wintle, Elith and Potts2005). Management of threatened populations will be most effective if range-wide wildlife–habitat relations are considered (Lengyel et al. Reference Lengyel, Kobler, Kutnar, Framstad, Henry and Babij2008). Biodiversity loss through habitat fragmentation, overexploitation, and climate change occurs over a large scale; a landscape-level approach to conservation matches the scale of such threats (Jones Reference Jones2011). Furthermore, the threat status of species is based on population size across their entire range (USFWS 1984), and thus land-use guidelines also need to apply at this level.
Nest-site selection is a component of wildlife–habitat relations that is fundamental to the reproductive success of birds (Clark and Shutler Reference Clark and Shutler1999; Pleszczynska Reference Pleszczynska1978). Nest location can reduce the energetic cost of reproduction (Høyvik Hilde et al. Reference Høyvik Hilde, Pélabon, Guéry, Gabrielsen and Descamps2016), provide cover from predators (Eggers et al. Reference Eggers, Griesser, Nystrand and Ekman2006), and relieve both inter- and intraspecific competition for resources (Cunningham et al. Reference Cunningham, Kesler and Lanctot2016; Quintana and Yorio Reference Quintana and Yorio1998). Examining habitat differences between successful and unsuccessful nests can help determine whether landscape or habitat characteristics correlate with increased reproductive success (Ha et al. Reference Ha, Morton, Ha, Berry and Plentovich2011). This link makes nest-site selection an important consideration for the conservation and recovery of threatened species, particularly with regards to decision-making around land (Poirazidis et al. Reference Poirazidis, Goutner, Tsachalidis and Kati2007).
The Mariana Crow or Åga Corvus kubaryi was federally listed as an endangered species in 1984 and is considered ‘Critically Endangered’ by the IUCN (IUCN 2018; USFWS 1984). It is native to both Guam and Rota, Commonwealth of the Mariana Islands, but was extirpated from Guam in the late 1990s (USFWS 2005) as a result of predation by the introduced brown tree snake Boiga irregularis (Wiles et al. Reference Wiles, Bart, Beck and Aguon2003). The Rota population showed a 93% decline in point-count surveys from 1982 to 2004 (Amar et al. Reference Amar, Amidon, Arroyo, Esselstyn and Marshall2008), despite the absence of the brown tree snake (Rodda and Savidge Reference Rodda and Savidge2007). A variety of causes have been hypothesised for this decline, including predation from introduced predators, habitat loss and degradation, human persecution, inbreeding depression, and a poorly understood inflammatory syndrome (Faegre et al. Reference Faegre, Nietmann, Hubl, Ha and Ha2019; Morton et al. Reference Morton, Plentovich and Sharp1999; T. Work 2022, personal communication). While healthy demographic rates for the species are unknown, the observed nest success rates (25.7%; Zarones et al. Reference Zarones, Sussman, Morton, Plentovich, Faegre and Aguon2015) and first-year survivorship (63%; S. Faegre, unpublished data) are currently limiting species recovery to some degree.
A 2011 study found that nest-sites selected by Åga had a higher canopy cover, contained woody plants associated with limestone forests, and had larger woody vines and stems of papaya Carica papaya compared with random points (Ha et al. Reference Ha, Morton, Ha, Berry and Plentovich2011). A higher canopy cover could be advantageous by hiding nests from predators, protecting nests from sun exposure, and limiting damaging winds caused by typhoons (Bowman and Woolfenden Reference Bowman and Woolfenden2002). Significant canopy damage was sustained on Rota from super-typhoon Roy in 1988 (Fancy and Snetsinger Reference Fancy and Snetsinger2001) and breeding efforts were disrupted by super-typhoon Paka in 1997. Both storms had a negative impact on mature forest island-wide (Morton et al. Reference Morton, Plentovich and Sharp1999), and while flighted Åga appear to suffer little mortality from typhoons, the breeding disruption from these super-typhoons coincided with declining numbers of Åga pairs on Rota (Plentovich et al. Reference Plentovich, Morton, Bart, Camp, Lusk and Johnson2005).
It has also been suggested that Åga avoid human habitation (Baker Reference Baker1951; NRC 1997) and prefer not to nest within 300 m of a building (Ha et al. Reference Ha, Morton, Ha, Berry and Plentovich2011). Habitat fragmentation, human persecution, and predators such as cats may increase near human habitation (Zarones et al. Reference Zarones, Sussman, Morton, Plentovich, Faegre and Aguon2015). However, these findings were based on nests in six 1-km2 plots, chosen based on the large amount of contiguous native forest relative to the rest of the island (Ha et al. Reference Ha, Morton, Ha, Berry and Plentovich2011). Only 32% of breeding pairs in the population occurred inside these study blocks and they were not selected to be representative of the forests of Rota (Ha et al. Reference Ha, Morton, Ha, Berry and Plentovich2011) or those being considered for U.S. Fish and Wildlife Service (USFWS) special habitat protection (Morton et al. Reference Morton, Plentovich and Sharp1999). The small scale of the study area and the remoteness of the study blocks from developed areas may have biased the results, particularly in relation to nest-site selection in close proximity to buildings, roads, and human habitation.
In this study, we used remotely sensed vegetation data and nest locations from 2014 to 2021 to examine Åga nest-site selection across the species’ extant range. We compared nest-sites to random points in forested habitat to address two main research questions: (1) what habitat characteristics are Åga selecting at nest-sites and (2) does nest success vary with these habitat characteristics? To answer our first question, we tested (1a) if nest-sites are further from human developments than random points, (1b) if nest-sites occur in native limestone forest more often than would be expected based on the availability of forest types on Rota, and (1c) if nest-sites contain higher canopy cover and canopy height than random points. To answer our second question, we provide an updated estimate of Åga nest success and ask whether this success varies with canopy cover and height, proximity to human habitation, or cover type.
Methods
Location
The study was conducted on the island of Rota, the southernmost island in the Commonwealth of the Northern Mariana Islands (Figure 1). Rota is 86 km2 and 491 m at its highest point. The island is primarily limestone, but contains some exposed areas created by volcanic activity (Falanruw Reference Falanruw1989). Rota’s upper limestone terraces are described as rainforest, while its lower limestone terraces and volcanic slopes are described as grassland and scrub. The other inhabited islands within the Northern Marianas Islands have experienced extensive deforestation and secondary regrowth, whereas more than half (52%) of Rota is covered by native limestone forest (Falanruw Reference Falanruw1989).
The study area, which encompassed forested areas on the entire island of Rota, Commonwealth of the Northern Mariana Islands.

Nest-sites
From 2014 to 2021, we located and monitored 370 Åga nests with contents (eggs or nestlings). The search areas included all forested public and conservation land, and over 90% of Åga habitat on private land, for which we were given access permission. Åga can nest year-round, but peak nesting occurs from October to March (Lusk and Taisacan Reference Lusk and Taisacan1996) and our nest searching and monitoring occurred from August to June yearly. After a nest was found it was checked weekly to determine its status and stage. Nest stage was determined either behaviourally or with a camera pole. Åga sometimes build dummy nests prior to incubation (Morton et al. Reference Morton, Plentovich and Sharp1999); however, only nests that were observed with confirmed contents were included in the study. While all 370 nests were used for the nest-site selection analyses, only 342 of those nests were used in the nest success analyses. The 28 nests that were excluded from the analyses were artificially failed when the eggs were collected for a captive rear and release programme administered jointly by the University of Washington and San Diego Zoo Wildlife Alliance. We define successful nests as those that produced at least one fledgling. For this study, we defined a nest as ‘fledged’ if the fledgling had left the nest and moved away from it by a distance of at least 30 cm.
Habitat covariates
Habitat covariates of interest included canopy height, canopy cover, land-cover type, distance to road, and distance to building. Land-cover values on Rota were obtained from U.S. Department of Agriculture Forest Service (USFS) land-cover data (2016; 30 m resolution) from the Pacific Islands Ocean Observing System data portal. Canopy height and canopy cover data were obtained from the Landfire 2.0.0 insular areas data set (2017; 30 m resolution) using the Landfire data distribution site. Roads were created from converting road GPS points in ArcGIS to line shapefiles. A buildings layer was digitised in ArcGIS using Environmental Systems Research Institute (ESRI) world imagery.
Random points
We used ArcGIS to create 400 random points in forested habitat on Rota. Forested habitat included five different land-cover classes: agroforestry, agroforestry–coconut, mixed introduced, native limestone, and ravine. These classes included 100% of nest-sites between 2014 and 2021. The number of random points assigned within each land-cover type was proportional to its total area (Table 1).
Comparison of the percentage of acreage and percentage of total nests for each of the land-cover types where nests have been confirmed

Habitat validation
To assess accuracy for land-cover type, canopy height, and canopy cover, 50 randomly selected points were each ground-truthed for both Åga nest points and random points. We measured canopy cover (using a spherical densitometer) and canopy height (using a range finder) at five places per point: one at the point’s centre and four points 5 m away from the centre to the north, south, east, and west. Land-cover types were determined visually using Falanruw et al. (Reference Falanruw1989) land-cover descriptions. Upper and lower 95% confidence intervals of accuracy for each category were determined using the Hord and Brooner (Reference Hord and Brooner1976) table for map accuracy (see Supplementary material Table S1).
Analyses
To address our nest-site selection questions, we first used a linear discriminant analysis (LDA) as per Ha et al. (Reference Ha, Morton, Ha, Berry and Plentovich2011) to determine if there were differences between actual nest-sites and random points. LDAs reduce dimensionality in data sets, maximising the variability between groups of interest (actual nest-sites and random points). We further explored the covariates that drove group separation individually using the independent two-sample Wilcoxon test (Mann–Whitney U test) and a chi-square test to look at nest-site selection by land-cover type based on availability. Aga pairs usually renest after nest failure, with a mean of 2.05 nests per season (Zarones et al. Reference Zarones, Sussman, Morton, Plentovich, Faegre and Aguon2015). To avoid over-weighting nesting pairs that failed to nest more often and thus represented more nests in the data set, we also ran the Mann–Whitney U and chi-square analyses with the habitat covariates averaged for all nests for a given pair of birds each breeding season. Thus, the nest-site selection for a pair that nested multiple times in a year was given the same weight as a pair that nested once during the year. This resulted in 253 pair nest-site selection events that were used for nest-site selection analyses (from 370 individual nests). Finally, we also re-ran these latter two analyses with the top of the Sabana plateau removed from the random points, as this large area of high elevation does not have any recorded nests and is generally considered to be an area that does not hold any Åga, despite containing primarily limestone forest (Jones Reference Jones2011).
We used the Mayfield method (Mayfield Reference Mayfield1975) to estimate success of the monitored 342 nests. To determine if nest success correlated with any of the habitat covariates, we used logistic regressions with fledging success as the binary response variable.
Results
Nest-site selection
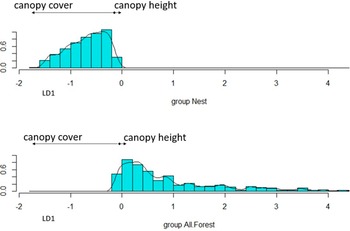
The LDA was fair at discriminating (Landis and Koch Reference Landis and Koch1977) between actual nest-sites and random points (overall accuracy = 0.643, kappa = 0.289). Nest-sites and random points had a positive predicted value of 47% and 46.9%, respectively. Canopy cover had the highest coefficient of linear discriminant (-8.252) and had the highest effect on group separation along the first linear discriminant (LD1 in Figure 2), followed by canopy height (0.240). Distance to road and distance to building contributed little to group separation (0.001 and <0.0001, respectively). There was little overlap between the actual nests and random points along the LD1 (Figure 2).
The two groups (nest-sites and random points) along the first linear discriminant (LD1). Arrows indicate degree and direction of separation from the coefficients of linear discriminant for land-cover covariates (distance from road and buildings not shown because of small coefficients <0.01).

The 95% confidence intervals for canopy cover and distance to building did not overlap when compared between nest-sites and random points, while distance to road and canopy height were similar between the two groups (Figure 3). The Mann–Whitney U test further corroborated this result, revealing significant differences in canopy cover and distance to building between the two groups, although the magnitude of the effect sizes was small (Figure 4). Overall, our results indicated that Åga are not avoiding areas of human habitation, as nest-sites were on average closer to buildings than random points (although the association was weak). Removing the Sabana plateau (where Åga do not nest) from the random points reduced the mean distance to building to be nearly identical to that of the nest points, resulting in non-significance of the distance to building. The shortest nest-to-building distance in this study was 32 m.
Differences in mean canopy height, canopy cover, distance to road, and distance to building with 95% confidence intervals for actual nests and all random points.

Mann–Whitney U effect size of canopy height, canopy cover, distance to road, and distance to building with 95% confidence intervals.

The use of land-cover type for nesting was significantly different than expected based on availability (X2 =42.351, P <0.00001). Post hoc tests revealed that ravine forest was used for nesting more often than expected based on availability (P <0.0001), while mixed introduced forest was used less often (P <0.0001). Removing the Sabana plateau maintained these results, while also revealing that Åga use limestone forest that is not on Sabana more often than expected based on availability (P <0.001).
Nest success
Of the 342 nests, 119 fledged successfully (34.8%) with a Mayfield nest success rate of 23.9% (95% confidence interval: 23.6–24.2). None of the habitat covariates significantly correlated with nest success. Similarly, there was no land-cover type in which nest success was higher (Figure 5). Furthermore, model ranking revealed that the null model was the best supported model, further indicating that the habitat covariates we used described patterns of nesting success poorly.
Probability of nest success by forest cover type with 95% confidence intervals.

Discussion
Large-scale studies of habitat needs for threatened species are valuable for shaping effective and reasonable conservation guidelines. In the case of the Åga, our study can be used to more effectively protect or improve nesting habitat and to aid in the selection of locations suitable for species translocations. One of the criteria for down-listing threat status is the existence of two populations, in this case one on Rota and another elsewhere (USFWS 2005); the suitability of other islands is currently being considered.
Ha et al. (Reference Ha, Morton, Ha, Berry and Plentovich2011) found that Åga preferred to nest in native limestone forest over secondary vegetation comprising mainly introduced species; however, their study plots did not include the full range of forest types used by breeding Åga. We found that Åga nested in all forest types but preferred to nest in native limestone, ravine, and agroforestry-coconut over agroforestry and mixed introduced forest, with ravine forests being used significantly more relative to their availability (Table 2). Generally, our results suggest that the species selects a broader variety of habitat types for nesting than previously thought. In particular, the finding of many nests within agroforestry-coconut (old coconut plantations that are now interspersed with native limestone forest) is informative, given that this forest type is common on some of the uninhabited Northern Mariana Islands that are being considered for translocation.
Percentage of nests and all forest points that fall within different thresholds of the explanatory covariates, including summary statistics

It should be noted that although our analysis shows native limestone was not used significantly more given availability, there were two large swathes of limestone forest that were considered available but not used by Åga. The Mt. Taipingot conservation area in the south-west corner of the island (2.2% of all limestone forest) historically may have held Åga, but has not been recorded since surveys began, likely because these surveys began well after the population declined and this peninsular forest is isolated from other forested areas by Songsong village. The Sabana plateau also holds a large amount (35%) of limestone forest but does not have any records of Åga. It should be noted that Sabana was not systematically searched for nests because there have been no records of Åga in the area; the area is regularly visited for other reasons (e.g. wildlife surveys, tourism, farming, collecting of medicinal plants) and we believe it would be known if Åga were present there. Radio-telemetry studies corroborate the evidence that Åga use the edge of the plateau, but not the top. Excluding the area from available nesting habitat in our analysis caused limestone forest to stand out as used significantly more proportionately to its availability. We found no significant differences in nest success based on any of the habitat covariates in our study. Other forest characteristics, such as understorey cover and composition or canopy structure, were not explored in this study but may be important for both prey availability and nest-site selection (Newell and Rodewald Reference Newell and Rodewald2011; Richards and Windsor Reference Richards and Windsor2007).
Amongst the habitat covariates we measured, we found that canopy cover was a driver for nest-site selection. While the Åga does not have resident aerial predators, canopy cover may protect nests from conspecifics and occasional migratory raptors. It also may serve as an important form of protection from extreme weather, including intense heat and high winds which are common on Rota. The canopy land-cover layer we used (and the period during which the ground-truthing was carried out) represented a period during which the canopy was representative of a recovered canopy rather than a storm-damaged one. While increased canopy cover was selected by nesting pairs in this study, canopy cover did not correlate with nest success suggesting it may not be a limiting factor in habitat suitability. Repeating this study shortly after a large typhoon could provide additional insight into the function and importance of canopy cover.
The Ha et al. (Reference Ha, Morton, Ha, Berry and Plentovich2011) nest-site selection study for Åga found that nest-sites were further from human infrastructure such as roads and buildings than random points, and that native forest was associated with higher nest success (Ha et al. Reference Ha, Morton, Ha, Berry and Plentovich2011). In contrast, we found that distance to human infrastructure did not contribute to nest-site selection, and that on average Åga nested only 191.9 m to roads, with 13.7% of nests found less than 50 m from a road. While all nests in the Ha et al. (Reference Ha, Morton, Ha, Berry and Plentovich2011) study were further than 300 m from buildings, nearly a third of our nests were within 300 m of a building; four nests were within 50 m of a building (Table 2) and three of these successfully fledged, indicating the close proximity to occupied human dwellings may not be inherently problematic for the species. Several factors may contribute to this contrast between Ha et al. (Reference Ha, Morton, Ha, Berry and Plentovich2011) and the current study. (1) The Ha et al. (Reference Ha, Morton, Ha, Berry and Plentovich2011) study had a smaller sample size and the study area was limited to primarily contiguous native forest blocks, which tends to occur further from human habitation. (2) The difference between actual nests and random points in the Ha et al. (Reference Ha, Morton, Ha, Berry and Plentovich2011) study may not have been biologically meaningful (226 m for random points and more than 300 m for nests). (3) The effects of human persecution during the 1990s and early 2000s, a factor which has been proposed as a driver of Åga decline (Plentovich et al. Reference Plentovich, Morton, Bart, Camp, Lusk and Johnson2005), could have been more salient during the Ha et al. (Reference Ha, Morton, Ha, Berry and Plentovich2011) study, which used nest-sites from 1997 to 1999. If this were the case, the Ha et al. (Reference Ha, Morton, Ha, Berry and Plentovich2011) study may have captured habitat characteristics influenced by human activity rather than what may comprise suitable nesting habitat in the absence of persecution. Data from 2014 to 2021 suggested that Åga successfully nest in most forested habitat, regardless of proximity to humans. This result is in line with what we would expect from corvids in general, which as a family tend to be behaviourally plastic and tolerant of humans (Benmazouz et al. Reference Benmazouz, Jokimäki, Lengyel, Juhász, Kaisanlahti-Jokimäki and Kardos2021; Cornell et al. Reference Cornell, Marzluff and Pecoraro2012).
It is unclear what caused the initial major decline of the Åga over the two decades from 1982 until the early 2000s, but it has been suggested that human persecution may have been the driving factor (Sussman et al. Reference Sussman, Ha and Henry2015). While it was previously believed that first-year survival had declined dramatically, leading the population towards extinction (Ha et al. Reference Ha, Butler and Ha2010), more recent analyses of an expanded data set based on increased effort, land-owner access, and proportion of banded birds (~95%) suggest that first-year survival has been stable from the 1990s to the present (S. Faegre, unpublished data). Following the listing of the Åga as an endangered species, the resulting regulatory changes for Åga habitat, as well as conflicts between state and federal agencies, turned the Åga into a symbol of federal overreach and an obstacle to land development. Negative sentiment towards the Åga escalated in documented cases of persecution (Plentovich et al. Reference Plentovich, Morton, Bart, Camp, Lusk and Johnson2005). To secure the future of the Åga, conservation efforts must focus on maintaining outreach and building local capacity for stewardship.
While adult survival is generally high (Ha et al. Reference Ha, Morton, Ha, Berry and Plentovich2011), the low nest success rate is of concern. Our finding of 23.9% Mayfield nest success from 2014 to 2021 is similar to the historical nest success estimates of 25.7%, suggesting that threats to nests may have remained similar between the two studies. Little data exist on nest success of tropical crows, with the Åga representing the only tropical corvid to our knowledge for which there is a long-term data set on nesting. Thus, it is unclear if the low nest success rate for the Åga is the norm, or lower than expected. What is clear from this study is that, based on the variables we measured, suitable nesting habitat is unlikely to be a limiting factor on Rota at this time. Future research efforts would be most fruitfully allocated towards determining the factors driving low nest success and first-year survival and to mitigating these to improve reproductive output.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0959270925100361.
Acknowledgements
We thank the many field biologists who were involved in searching for and observing nests over the years, especially Dylan Hubl, Lena Ware, Jonny Shepherd, and Dacia Wiitala who found a large proportion of the nests in this data set. Renee and James Ha provided useful comments on drafts of this manuscript. This work was supported by the Commonwealth of the Northern Mariana Islands Department of Lands and Natural Resources-Division of Fish and Wildlife and the U.S. Fish and Wildlife Section 6 Endangered Species funds. Work was conducted in accordance with Federal Fish and Wildlife Recovery Permit ES09155B, Federal Bird Marking and Salvage Permit 24329, and University of Washington Institutional Animal Care and Use Committee protocol number 4530-01.