Introduction
The processes driving population dynamics have intrigued scientists for decades. To unravel these processes it is essential to understand which factors influence population growth and how they affect key demographic variables such as birth, death, and migration rates. Many processes can affect population growth, and these may be extrinsic (e.g. climate, predation, parasitism, food availability) and intrinsic (e.g. territoriality; Krebs,Reference Krebs1995; Reference Krebs2013). Parasitism, in particular, represents a potentially crucial component in population dynamics (Anderson and May Reference Anderson and May1978; May and Anderson Reference May and Anderson1978; Scott and Dobson Reference Scott and Dobson1989), and its study could reveal essential aspects that often remain unexplored. Parasites typically have a detrimental impact on host fitness by increasing mortality or morbidity, decreasing fecundity, or limiting the energy available for other biological functions (either through the activation of the immune system or by impairing nutrient intake and food assimilation; Morand et al., Reference Morand, Krasnov and Poulin2006). As a result, parasites can deeply affect the population dynamics of the host, community structure, and even the functioning of the ecosystem in which they are embedded (Hatcher et al. Reference Hatcher, Dick and Dunn2012; Morand et al. Reference Morand, Krasnov and Poulin2006; Telfer et al. Reference Telfer, Lambin, Birtles, Beldomenico, Burthe, Paterson and Begon2010).
According to theory, investigations into parasite-induced regulation have highlighted the conditions under which parasites can destabilise host populations, potentially generating population cycles (periodic cyclic fluctuations in abundance). These conditions include negative effects on host fitness (through reduced survival or fecundity), high aggregation of parasites among hosts (most individuals carry few parasites, but some many), and delayed density-dependence, that is, parasite abundance increasing with host abundance but with a time delay (Anderson and May Reference Anderson and May1978; May and Anderson Reference May and Anderson1978; Hudson et al. Reference Hudson, Dobson and Newborn1998; Tompkins et al. Reference Tompkins, Dobson, Arneberg, Begon, Cattadori, Greenman, Heerterbeek, Hudson, Newborn, Pugliese, Hudson, Rizzoli, Grenfell, Heesterbeek and Dobson2002). Crucially, parasite-induced regulation requires reciprocal feedback between host and parasite populations, i.e., parasites must affect host demography, and host abundance must in turn influence parasite transmission and abundance (Anderson and May Reference Anderson and May1978; Hudson et al. Reference Hudson, Dobson and Newborn1998). Such feedbacks are most likely to arise in systems involving directly transmitted parasites with a single definitive host, where parasite dynamics closely track host density, behaviour, and contact rates (Tompkins et al. Reference Tompkins, Dobson, Arneberg, Begon, Cattadori, Greenman, Heerterbeek, Hudson, Newborn, Pugliese, Hudson, Rizzoli, Grenfell, Heesterbeek and Dobson2002).
Among macroparasites, gastrointestinal helminths have been proposed as potential regulators of vertebrate populations because of their widespread occurrence and capacity to affect host physiology and reproduction (Morand et al. Reference Morand, Krasnov and Poulin2006). However, empirical evidence for their role in population regulation remains mixed. Several studies find no clear relationship between helminths burden and host fitness (Forbes et al. Reference Forbes, Stuart, Mappes, Henttonen and Huitu2014), whereas others have demonstrated negative effects on host population parameters (Deter et al. Reference Deter, Charbonnel, Cosson and Morand2008; Pedersen and Greives Reference Pedersen and Greives2008; Winternitz et al. Reference Winternitz, Yabsley and Altizer2012). Helminth infections can cause physiological pathologies and have been associated with hypertrophy of organs involved in immune response, such as the spleen, adrenal glands, or liver (Gotardo et al. Reference Gotardo, Andrade and Andrade2000; Ponlet et al. Reference Ponlet, Chaisiri, Claude and Morand2011; Spratt and Singleton Reference Spratt and Singleton1986; Tenora et al. Reference Tenora, Wiger and Barus1979; Wiger Reference Wiger1977). High parasite abundance has been associated with host population crashes (e.g. Deter et al. Reference Deter, Charbonnel, Cosson and Morand2008; Pedersen and Greives Reference Pedersen and Greives2008), consistent with delayed density-dependent patterns. Notwithstanding, other authors support the hypothesis that parasitism usually plays a secondary rather than a main driving role, acting synergistically with other factors (Keymer and Dobson Reference Keymer and Dobson1987; Marcogliese and Pietrock Reference Marcogliese and Pietrock2011; Pedersen and Greives Reference Pedersen and Greives2008; Redpath et al. Reference Redpath, Mougeot, Leckie, Elston and Hudson2006). This variability highlights the importance of focusing on specific host-parasite systems and parasite life histories, particularly transmission mode and host specificity, when evaluating theoretical predictions.
Studying population regulation is especially relevant in hosts with cyclic population dynamics, which undergo large interannual abundance fluctuations (Elton Reference Elton1924; Hudson and Dobson Reference Hudson and Dobson1995; May and Anderson Reference May and Anderson1978; Tompkins et al. Reference Tompkins, Dobson, Arneberg, Begon, Cattadori, Greenman, Heerterbeek, Hudson, Newborn, Pugliese, Hudson, Rizzoli, Grenfell, Heesterbeek and Dobson2002). Population cycles in rodents display large-scale multiannual fluctuations (Jacob and Tkadlec Reference Jacob, Tkadlec, Singleton, Belmain, Brown and Hardy2010; Krebs Reference Krebs2013), typically comprising increase, peak, and crash phases (Boonstra et al. Reference Boonstra, Krebs and Stenseth1998; Oli Reference Oli2019). During these phases, body mass, social behaviour, age structure, and age at sexual maturation differ markedly (Oli Reference Oli2019), as well as recruitment through delayed-density dependent changes in reproduction rather than survival (Lambin et al. Reference Lambin, Begon, Burthe, Graham, MacKinnon, Telfer and Oli2025). Parasite transmission and host susceptibility are also expected to differ between phases, potentially creating conditions for delayed density-dependent parasite effects on the condition, survival, or reproduction of small rodents (Hudson et al. Reference Hudson, Dobson and Newborn1998).
In north-western Spain, the common vole (Microtus arvalis) is the most abundant rodent inhabiting the intensified farming landscapes (Luque-Larena et al. Reference Luque-Larena, Mougeot, Viñuela, Jareno, Arroyo, Lambin and Arroyo2013; Rodríguez-Pastor et al. Reference Rodríguez-Pastor, Luque-Larena, Lambin and Mougeot2016). There, vole populations have been shown to display cyclic dynamics with abundance outbreaks every 3 years (Herrero-Cófreces et al. Reference Herrero-Cófreces, Mougeot, Lambin and Luque-Larena2021b; Mougeot et al. Reference Mougeot, Lambin, Rodríguez-Pastor, Romairone and Luque-Larena2019) with densities exceeding 1,000 individuals per hectare, followed by sharp declines. Despite extensive research on vole population dynamics, the role of helminths in shaping these cycles remains unknown. Previous studies on voles and other small mammals have reported diverse helminth communities, often dominated by a small number of species, with infection patterns varying according to host sex, season, and population density (Haukisalmi and Henttonen Reference Haukisalmi and Henttonen2000; Tenora et al. Reference Tenora, Murai and Mészáros1973). Sex-specific differences in infection are frequently observed and may arise from behavioural differences, hormonal effects on host susceptibility, or differential exposure to infective stages (Morand et al. Reference Morand, Krasnov and Poulin2006).
In this study, we first provide a descriptive overview of the gastrointestinal helminth community infecting common voles in north-western Spain, quantifying the prevalence, abundance, and aggregation patterns of the different helminth taxa in order to provide ecological context. However, because this community was overwhelmingly dominated by a single nematode species, Syphacia sp., a directly transmitted helminth with a simple life cycle, subsequent inferential analyses focused on this species.
We evaluated whether patterns of infection by Syphacia sp. were consistent with theoretical conditions associated with parasite-induced host population regulation in a cyclic rodent system. Specifically, we aimed to (i) examine whether the prevalence and abundance of Syphacia sp. vary according to host sex, season, and population phase, and (ii) test for associations between the prevalence of Syphacia sp. and host fitness-related traits, i.e. vole condition (body mass and immune-related organ mass) and female fecundity (litter size).
Materials and methods
Study area, vole trappings, and sampling design
We studied the helminth community of common voles in NW Spain. The 80 Km2 study area is located in the province of Palencia (42°01´N, 4°42´), a region mostly influenced by continental-Mediterranean climate (Rivas-Martínez et al., Reference Rivas-Martínez, Penas, del Río, Díaz González, Rivas-Sáenz and Loidi2017). Fieldwork (vole live-trapping) was carried out three times a year (March, July, and November, spring, summer, and autumn, respectively) between July 2010 and March 2015. Our time series (2010–2015) of sample collection included two population peaks (2011, 2014), two years of population increase (2010, 2013), and two years of population crash (2012, 2015; see Mougeot et al. Reference Mougeot, Lambin, Rodríguez-Pastor, Romairone and Luque-Larena2019 and Herrero-Cófreces et al. Reference Herrero-Cófreces, Mougeot, Lambin and Luque-Larena2021b for more details on vole dynamics). Sampling included 15 trapping sessions with a total of 12,600 traps/night (840 traps/night per trapping session) following the same methodology of Rodríguez-Pastor et al. (Reference Rodríguez-Pastor, Luque-Larena, Lambin and Mougeot2016). Traps (8 × 9 × 23 cm; LFAHD Sherman©) were set open for 24 hours, with carrot and apple used as bait. Each animal captured was individually identified with a unique code, and date and location were noted when trapped. All trapped voles, live or dead in traps, were collected. After trapping, live voles were placed in individual cages (29 × 22 × 14 cm; Panlab®) provided with food, water, and bedding material and transported to the laboratory.
Laboratory procedure
At the lab, voles were euthanized with CO2, sexed, weighed (with an electronic balance, to the nearest 0.1 g), and measured with a ruler (total length without tail, nearest 1 mm). Animals were stored at –23°C until dissection, which followed standard protocols. The spleen, liver, and adrenal glands were separately weighed, the reproductive system of females was examined for embryos to identify pregnant females and count the total number of embryos, and the GI tract was removed and kept frozen at –23°C until helminth survey. Once defrosted, GI tracts were individually placed in a Petri dish with a thin layer of tap water. The oesophagus was cut just before the stomach, and the rectum was cut near its distal end. Ligaments were cut and the small intestine was straightened by cutting the mesenteries. The small intestine, large intestine, and caecum were then separated. We processed every section separately, cutting it longitudinally (starting at the posterior end) and stirring carefully to spread the content. Then, we looked for helminths first with the naked eye and then screened them under stereo microscope at 40× magnification. Cestodes were placed in a separate Petri dish with a thin layer of tap water to allow them to flatten for identification. Specimens were preserved in 70% ethanol at –23°C afterwards. Helminths were identified based on morphological characters (Haukisalmi and Henttonen Reference Haukisalmi and Henttonen1993; Tkach et al. Reference Tkach, Hope and Greiman2019). A total of 380 common voles were processed for helminth infection.
Data analysis
To characterise the gastrointestinal helminth assemblage of common voles, we first described the composition of the helminth community across the entire sample. For each helminth taxon, we quantified the number of infected hosts and calculated its relative frequency as the proportion of individuals of that taxon relative to the total number of helminths recovered. In addition, we estimated standard parasitological descriptors, including prevalence (number of infected hosts divided by the number of hosts examined), mean abundance (average total number of parasites per host), and aggregation. Prevalence was estimated with 95% confidence intervals (CI; traditional Clopper-Pearson method) and mean abundance was expressed as mean ± standard error (SE). We quantified the level of aggregation using three complementary measures: the variance-to-mean ratio (VMR), the Discrepancy index (D) following Poulin (Reference Poulin1993), and the parameter k of the negative binomial distribution estimated by maximum likelihood from the distribution of parasite abundance among hosts. These descriptive statistics were obtained using the Quantitative Parasitology (QPweb) software version 1.0.15 (Reiczigel et al. Reference Reiczigel, Marozzi, Fábián and Rózsa2019).
Because the helminth community was overwhelmingly dominated by a single species, Syphacia sp., all subsequent inferential analyses focused exclusively on this nematode. To evaluate the associations between host and environmental variables and the prevalence and abundance of Syphacia sp., we used Generalized Linear Models (GLMs) that included host sex (male, female), season (spring, summer, autumn), and the phase of the host population cycle (increase, peak, or crash) as explanatory variables. The cycle phase was determined from time series of vole abundance (average number of voles trapped per 100 traps per 24 h, measured every 4 months; see Mougeot et al. Reference Mougeot, Lambin, Rodríguez-Pastor, Romairone and Luque-Larena2019 and Herrero-Cófreces et al. Reference Herrero-Cófreces, Mougeot, Lambin and Luque-Larena2021b). Prevalence was modelled using a binomial distribution, and abundance using a negative binomial distribution.
To examine relationships between vole condition and the prevalence of Syphacia sp., we used body mass and the weight of immune-related organs as indicators. We modelled body mass (analysing males and females separately) as the response variable and prevalence of Syphacia sp., season, phase, and body length as the explanatory variables. Due to the small sample size of infected individuals, we analysed the prevalence of Syphacia sp. rather than parasite abundance. Body length was included as a covariate to correct mass for individual size, thereby studying variation in a body condition index (mass relative to size). For females, the number of embryos was also included as a covariate to account for variation in body mass associated with pregnancy. To test for associations between the weight of spleen, liver, and adrenal glands and the prevalence of Syphacia sp., we included host body mass, host sex, and the prevalence of Syphacia sp. as the explanatory variables. Response variables were log-transformed.
Finally, to investigate associations between vole reproduction and the prevalence of Syphacia sp., we fitted the number of embryos per female (including zero for non-pregnant individuals) as a response variable and host body length, season, phase of the host population cycle, and the prevalence of Syphacia sp. as explanatory variables. All statistical analyses were carried out using the “lme4” (Bates et al. Reference Bates, Mächler, Bolker and Walker2015) and “R2admb” (Bolker et al. Reference Bolker, Skaug and Maintainer2017) packages, and the R software version 3.6.1 (R Core Team 2021). Initial models included all biologically meaningful two-way interactions among explanatory variables. Model selection followed a backwards simplification procedure based on likelihood ratio tests, sequentially removing non-significant interactions and main-effect terms to obtain the minimal adequate model. Model diagnostics were performed by visual inspection of residuals to assess goodness-of-fit and overdispersion. We tested differences between levels of significant variables using post hoc Tukey tests.
Results
Helminth community composition in common voles
From the 380 screened common voles, we collected 641 helminth individuals belonging to eight different taxa (Fig. 1, Table 1), namely: Anoplocephaloides dentata, Heligmosomoides laevis, Heligmosomoides sp., Heligmosomum sp., Paranoplocephala gracilis, Paranoplocephala omphaloides, Syphacia sp., and Trichuris sp. Twelve specimens (1.8% of the total) could not be identified. The commonest helminth was Syphacia sp., accounting for 82.5% of all the identified helminths collected and present in 49% of the parasitized voles (47 out of 95).

Figure 1. Frequency of occurrence of gastrointestinal helminth species collected from wild common voles in NW Spain (2010–2015). Nematodes are shown in blue colours and cestodes in green colours. The yellow colour indicates unidentified helminths.
Table 1. Parasitological parameters of the gastrointestinal helminth community of common voles in NW Spain (2010–2015)

Common voles screened = 380. Total helminths collected = 641 (12 unidentified)
a ST, stomach; SI, small intestine; C, caecum;
b 95% confidence interval by Clopper-Pearson;
c Standard error;
d 95% confidence interval by bootstrap method.
The prevalence of Syphacia sp. was 12.4% (95% CI: 9.2–16.1), and mean abundance was 1.39 ± 0.54 parasites per host. The distribution of Syphacia sp. among hosts was highly aggregated, as indicated by a high variance-to-mean ratio (79.30), a discrepancy index of 0.96 (95% CI: 0.94–0.98), and a very low negative binomial aggregation parameter (k = 0.03). These metrics consistently indicate that most parasites were concentrated in a small fraction of infected individuals, while the majority of hosts harboured few or no worms. Parameters for the other identified helminths are provided in Table 1 for descriptive purposes. All were found at low prevalence, abundance, and aggregation levels.
Variation in the prevalence and abundance of Syphacia sp., according to host sex, season, and host population phase
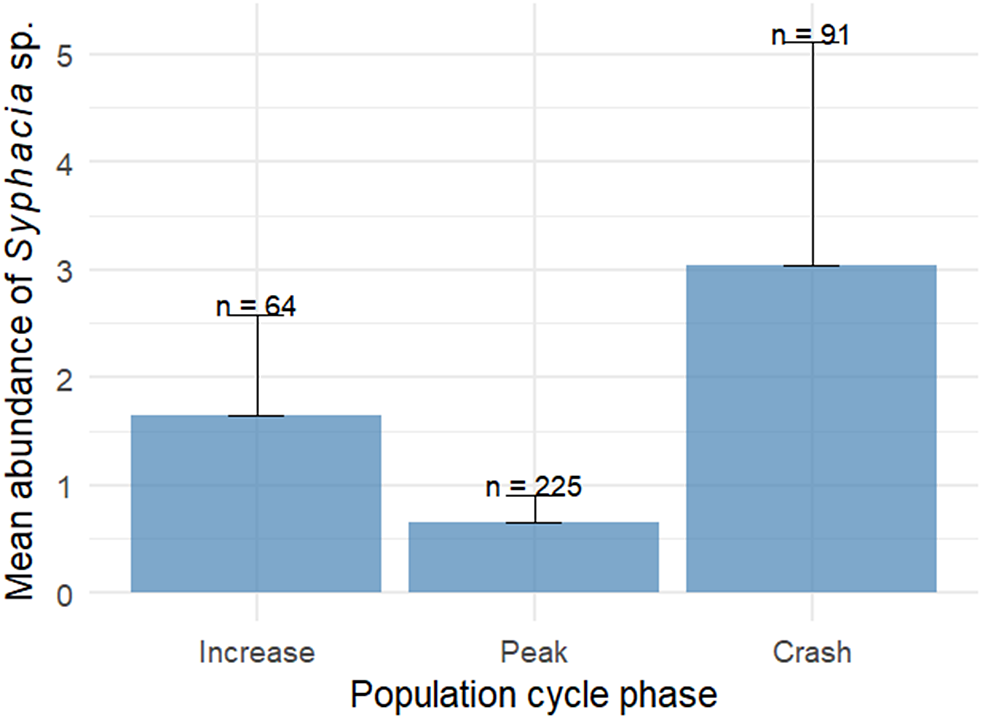
The prevalence of Syphacia sp. tended to be higher in males than in females, a difference that did not reach statistical significance (χ2 = 3.37, df = 1, p = 0.066). Prevalence did not differ between seasons (χ2 = 0.322, df = 2, p = 0.851) or population cycle phases (χ2 = 0.327, df = 2, p = 0.849). The abundance of Syphacia sp., however, varied according to the phase of the host population cycle (χ2 = 6.69, df = 2, p = 0.035), with no significant additional associations with host sex (χ2 = 0.615, df = 1, p = 0.433) or season (χ2 = 1.120, df = 2, p = 0.549). Post hoc comparisons indicated that abundance was higher during the crash phase than during the peak phase (Tukey-adjusted p = 0.041; Fig. 2), whereas no significant differences were detected between the increase and peak phases or between the increase and crash phases.

Figure 2. Mean abundance of Syphacia sp. according to the phase of the common vole cycle in NW Spain (2010–2015). Error bars represent standard error; sample sizes (n) are shown above bars.
Associations between common vole condition and the prevalence of Syphacia sp
We tested for associations between body mass and the prevalence of Syphacia sp. in males and females separately. No significant association with Syphacia sp. was detected in either males or females (Table 2). For males, significant interactions were found between season and body length and between season and phase (Table 2a). For females, the interaction between season and phase was significant, and both body length and the number of embryos were positively associated with body mass (Table 2b).
Table 2. Predictors of body mass in male (a) and female (b) common voles in NW Spain (2010–2015) based on generalized linear models

Significant p-values are highlighted in bold.
When testing for relationships between the weight of immune-related organs (spleen, liver, and adrenal glands) and the prevalence of Syphacia sp. we found no significant associations (Table 3). In all three organs, the interaction between sex and body mass was significant (Table 3): the relationship between body mass and organ size differed between males and females, i.e., as body mass increases, organ size increases more steeply in females than in males (Table 3).
Table 3. Generalized linear models testing the associations between the immune organ weights in the common vole ((a) spleen, (b) liver, and (c) adrenal glands) and body mass, sex, and Syphacia sp.

Significant p-values are highlighted in bold.
Association between common vole litter size and the prevalence of Syphacia sp
Regarding female fecundity, we found that larger females produced bigger litters (Table 4). We also found an association between litter size and the prevalence of Syphacia sp., depending on season (significant interaction between the prevalence of Syphacia sp. and season (Table 4). Mean litter size was lower in infected females in spring (3.09 vs 2.17 embryos) and summer (2.28 vs 0 embryos), whereas it did not vary between infected and uninfected females in autumn (1.13 vs 1.29 embryos; Fig. 3).
Table 4. Results of general linear models examining associations with litter size of female common voles in NW Spain (2010–2015). Significant p-values are highlighted in bold.

Significant p-values are highlighted in bold.

Figure 3. Season-dependent associations between common vole litter size and the prevalence of Syphacia sp. Points indicate mean litter size, error bars represent standard error, and sample sizes (n) are shown above bars.
Discussion
Overview and main findings
The main aims of this study were i) to describe the gastrointestinal helminth infections of common voles and ii) to evaluate whether infection by the dominant helminth, Syphacia sp., shows patterns consistent with theoretical conditions associated with parasite-mediated population regulation in a cyclic rodent system. Our results show that (i) the helminth community was species-poor and overwhelmingly dominated by Syphacia sp.; (ii) infections by this nematode were highly aggregated among hosts; (iii) the abundance of Syphacia sp. varied across phases of the population cycle, being higher during crash phases; and (iv) litter size was smaller in females infected by Syphacia sp. during spring and summer.
We discuss these findings considering key theoretical prerequisites identified in host, parasite population models (e.g. aggregation and delayed density dependence), as necessary conditions for parasite-mediated regulation (Anderson and May Reference Anderson and May1978; May and Anderson Reference May and Anderson1978; Scott and Dobson Reference Scott and Dobson1989).
Helminth community structure and dominance of Syphacia sp
We identified eight different helminth species infecting common voles, yet practically all individuals belonged to a single species from the genus Syphacia sp., present in 49% of screened voles and accounting for more than 80% of recovered individual helminths. We identified up to five nematode species and three species of cestodes. Compared to other European studies on common vole, helminth species richness in our study area was relatively low (Feliu et al. Reference Feliu, Renaud, Catzeflis, Hugot, Durand and Morand1997; Gubányi et al. Reference Gubányi, Mészáros, Murai and Soltész1992; Kisielewska and Zubczewska Reference Kisielewska and Zubczewska1973; Tenora et al. Reference Tenora, Murai and Mészáros1973). In north-western Spain, the common vole is virtually the only vole species present in agricultural landscapes (Herrero-Cófreces et al. Reference Herrero-Cófreces, Flechoso, Rodríguez-Pastor, Luque-Larena and Mougeot2021a; Rodríguez-Pastor et al. Reference Rodríguez-Pastor, Luque-Larena, Lambin and Mougeot2016) which may partly explain the reduced helminth richness and diversity patterns found. Host community composition is known to influence parasite assemblage structure, and lower diversity of closely related hosts may constrain opportunities for parasite exchange and persistence at the regional scale (Morand et al. Reference Morand, Krasnov and Poulin2006; Poulin Reference Poulin2007). Our results also show that Syphacia sp. was the predominant helminth species, a pattern also reported in other European populations of common vole (Tenora et al. Reference Tenora, Murai and Mészáros1973). Unfortunately, we could not identify the Syphacia species involved in this system, due to limitations in terms of specimen preservation, and this should be clarified in future studies, for instance, using molecular techniques.
Aggregation of Syphacia sp. infections
Our analyses revealed that Syphacia sp. infections were highly aggregated among hosts, as indicated by a very low negative binomial aggregation parameter (k), a high variance-to-mean ratio (VMR), and elevated discrepancy index (D) values. Aggregation of macroparasites is a widespread phenomenon in wildlife populations and has been extensively documented across host–parasite systems (Shaw and Dobson Reference Shaw and Dobson1995). Theoretical models of host–parasite dynamics incorporate aggregation through the negative binomial parameter k, showing that strong overdispersion is a necessary condition for parasites to exert substantial regulatory effects at the population level (Anderson and May Reference Anderson and May1978). When parasites are highly aggregated (low k values), a small fraction of hosts carries much of the parasite population, concentrating potential fitness costs in heavily infected individuals.
While k is often emphasized, complementary indices such as VMR and the discrepancy index D provide additional perspectives on overdispersion, with VMR reflecting the variance relative to the mean and D quantifying the unevenness of parasite distribution across hosts (Poulin Reference Poulin1993; Shaw and Dobson Reference Shaw and Dobson1995). This pattern may have implications for host population dynamics, as aggregated infections concentrate potential fitness effects in a subset of hosts rather than distributing them evenly across the population, a key condition for parasite-mediated regulation (Anderson and May Reference Anderson and May1978).
Parasite abundance across population phases
We observed higher abundances of Syphacia sp. during crash phases of the vole cycle compared to peak phases. This pattern is consistent with the idea of delayed density dependence, in which parasite burden may increase after host density has already declined, as predicted by theoretical host-parasite models (May and Anderson Reference May and Anderson1978; Scott and Dobson Reference Scott and Dobson1989). Importantly, our phase-based comparisons do not allow explicit testing of time-lagged density dependence, as we did not model parasite abundance as a function of host density with varying temporal delays. Therefore, although the observed pattern aligns with theoretical expectations, it cannot be interpreted as direct evidence of delayed regulatory feedback.
Delayed density dependence describes negative feedback operating with a time lag, a mechanism invoked to explain cyclical population dynamics in a range of taxa, including host-parasite systems (May and Anderson Reference May and Anderson1978; Scott and Dobson Reference Scott and Dobson1989). Similar patterns of elevated parasite loads during low host density periods have been reported in other systems (Cerqueira et al. Reference Cerqueira, Delattre, De Sousa, Gabrion, Morand, Quere and Quéré2006; Redpath et al. Reference Redpath, Mougeot, Leckie, Elston and Hudson2006; Winternitz et al. Reference Winternitz, Yabsley and Altizer2012), where collapse periods coincide with higher parasite intensity in the surviving hosts.
Alternative explanations for a higher parasite abundance during low-density phases should also be considered. For example, survival bias could result in more resilient or heavily infected individuals persisting through population declines. Similarly, age structure changes may contribute if older animals accumulate infections over time (Barnard et al. Reference Barnard, Behnke, Bajer, Bray, Race, Frake, Osmond, Dinmore and Sinski2002; Behnke et al. Reference Behnke, Lewis, Zain and Gilbert1999; Cowan et al. Reference Cowan, Shutler, Herman and Stewart2009; Haukisalmi et al. Reference Haukisalmi, Henttonen and Batzli1995; Rossin et al. Reference Rossin, Malizia, Timi and Poulin2010). Because host age was not explicitly incorporated into our models, we cannot exclude the possibility that variation in parasite abundance partly reflects age-related accumulation rather than density-dependent processes per se.
Additionally, gastrointestinal helminths may impose energetic costs on hosts, competing for nutrient absorption and potentially requiring trade-offs between immune response and other biological processes such as metabolism, feeding, or reproduction (Morand et al. Reference Morand, Krasnov and Poulin2006). Such mechanisms could hypothetically increase physiological stress and affect host performance, but causal links cannot be established from our observational data.
Associations between the prevalence of Syphacia sp. and vole condition
We found no significant association between the prevalence of Syphacia sp. and indicators of host vole condition (body mass or immune organ mass), suggesting that infection by this oxyurid helminth does not substantially compromise these aspects of physiological state under natural conditions. Pinworms (Syphacia spp.) are intestinal nematodes that inhabit the lumen of the gastrointestinal tract and are generally not associated with marked tissue pathology in rodents, particularly at low to moderate infection intensities (Behnke et al. Reference Behnke, Lewis, Zain and Gilbert1999; Morand et al. Reference Morand, Krasnov and Poulin2006; Shaw and Dobson Reference Shaw and Dobson1995). Although parasite-induced reductions in host fitness constitute a key requirement for parasite-mediated population regulation (Anderson and May Reference Anderson and May1978), our results indicate that, at the levels of infection observed here, Syphacia sp. does not strongly impair host somatic condition. A minimum infection threshold may be necessary before measurable physiological costs arise, as hosts can tolerate low to moderate helminth burdens (Cowan et al. Reference Cowan, Shutler, Herman and Stewart2009; Haukisalmi et al. Reference Haukisalmi, Henttonen and Pietiäinen1994). This is consistent with broader empirical evidence showing that many helminths exert measurable fitness costs primarily at high burdens or under additional environmental stress (Shaw and Dobson Reference Shaw and Dobson1995).
Associations between the prevalence of Syphacia sp. and female fecundity
We detected season-specific associations between female litter size and the prevalence of Syphacia sp. Interestingly, in spring, infected females produced fewer offspring than uninfected females (3.09 vs. 2.17 embryos), whereas in summer infected females had no embryos (2.28 vs. 0), suggesting a potential reproductive cost associated with infection. In autumn, mean litter size was similar between infected and uninfected females (1.13 vs. 1.29; Fig. 3), potentially reflecting lower reproductive investment and energy allocation during this period. In NW Spain, common vole populations typically grow mostly between spring and summer (Mougeot et al. Reference Mougeot, Lambin, Rodríguez-Pastor, Romairone and Luque-Larena2019), when vole reproductive rates are highest, so a potential negative influence of Syphacia sp. infection then may be relevant for population growth.
Synergies between parasitism and other stressors have been suggested to explain such seasonal variation in host fitness, including reproductive performance (Keymer and Dobson Reference Keymer and Dobson1987; Marcogliese and Pietrock Reference Marcogliese and Pietrock2011; Pedersen and Greives Reference Pedersen and Greives2008). In our study, the aggregative distribution of helminths and the increase in overall abundance during the crash phase may further concentrate effects on the few heavily infected females, potentially intensifying reproductive costs at a critical stage of the population cycle. Our results are also consistent with a reduced reproduction of most heavily infected females during spring and summer. However, we must stress out that our results are correlative and do not establish causality and should be taken with caution given the small number of infected females. Future experiments (manipulating parasite loads) are needed to clarify the effects of Syphacia sp. on common vole reproduction and a possible negative influence on female fecundity.
Implications for parasite-mediated regulation
Parasite-mediated regulation requires two conditions: measurable negative effects on host demographic parameters and reciprocal feedback between host density and parasite transmission (Anderson and May Reference Anderson and May1978; Hudson et al. Reference Hudson, Dobson and Newborn1998; Tompkins et al. Reference Tompkins, Dobson, Arneberg, Begon, Cattadori, Greenman, Heerterbeek, Hudson, Newborn, Pugliese, Hudson, Rizzoli, Grenfell, Heesterbeek and Dobson2002). In this study, we found evidence consistent with high parasite aggregation, phase-related variation in the abundance of Syphacia sp., and seasonal associations with female fecundity (infected females producing smaller litters in spring and summer). However, we did not directly measure survival, transmission rates, or time-lagged density dependence.
Thus, while some necessary conditions for parasite-induced regulation appear to be present in our system, the available evidence does not demonstrate regulatory feedback. Instead, our results suggest that Syphacia sp. may contribute modestly to variation in reproductive output and parasite burden across cycle phases, potentially interacting with other ecological stressors, such as co-infections or environmental challenges (Ezenwa Reference Ezenwa2016; Turner et al. Reference Turner, Beldomenico, Bown, Burthe, Jackson, Lambin and Begon2014).
Although Syphacia sp. is relatively low-pathogenic and largely gut-restricted, its aggregated distribution and seasonal association with fecundity indicate it could exert fitness effects that become ecologically meaningful under certain demographic or environmental conditions. Future studies manipulating parasite loads are necessary to clarify the potential for these effects to influence vole population dynamics and contribute to population regulation.
Conclusions
In cyclic common vole populations of NW Spain, gastrointestinal helminth diversity was low and dominated by the directly transmitted nematode Syphacia sp. Infections were highly aggregated, parasite abundance varied across population phases, and seasonal associations with female fecundity were detected. These patterns are compatible with several assumptions of parasite-regulation theory but remain correlative.
Rather than demonstrating parasite-induced population regulation, our findings provide a quantitative assessment of theoretical expectations in a natural vole-helminth system and highlight the need for experimental or longitudinal approaches to disentangle causality, demographic structure, and parasite effects. Even parasites traditionally considered of low pathogenicity may have subtle demographic consequences under particular ecological contexts, warranting further investigation.
Acknowledgements
We thank Voitto Haukisalmi for helping with helminth identification. This work was funded by the projects ECOTULA (CGL2015-66962-C2-1-R), BOOMRAT (PID2019-109327RB-I00), and RATALERT (PID2022-136850NB-I00) by the Government of Spain. Silvia Herrero-Cófreces was supported by a PhD studentship from Junta de Castilla-y-León (co-funded by European Social Fund, Orden 10/11/2016).
Competing interests
The authors declare none.
Ethics approval and permits
All the trapping, transport, and euthanasia methods applied to animals in this study were approved by our institution’s ethics committee (CEEBA, Universidad de Valladolid; authorization code: 4801646), and we counted with the official capture permits from DGMN (Junta de Castilla-y-León) as well as compulsory national certificates (B and C categories) to manipulate living animals for research.