1. Introduction
Calcium ions (Ca2+) play multiple physiological and structural roles across prokaryote and eukaryote kingdoms. Eukaryotes maintain very low baseline cytosolic Ca2+ concentration [Ca2+ cyt] of around 100–200 nM since Ca2+ is essentially toxic at concentrations even in the low micromolar range due, at least in part, to the ability to bind inorganic phosphate (Clapham, Reference Clapham2007). A typical plant cell is faced with an approximately 1,000-fold inwardly directed concentration gradient across the plasma membrane (PM), so maintaining very low [Ca2+ cyt] requires highly efficient homeostatic mechanisms. In parallel to mechanisms for keeping [Ca2+ cyt] low, cells have evolved mechanisms to allow controlled entry of Ca2+ into the cytosol, giving rise to tightly regulated Ca2+ cyt elevations that can act to relay signals from the cell surface to downstream response elements in the cell interior. In plants, patterns of Ca2+ cyt elevations vary considerably in response to different stimuli and include single transient elevations, lasting a few seconds, repeated oscillatory elevations over longer time periods and more prolonged elevations (Edel et al., Reference Edel, Marchadier, Brownlee, Kudla and Heetherington2017; Lenzoni et al., Reference Lenzoni, Liu and Knight2018). Moreover, Ca2+ cyt elevations may occur uniformly across the cell or maybe highly localized to a particular region (Brownlee & Wheeler, Reference Brownlee and Wheeler2023). A further key feature of Ca2+ is its ability to bind reversibly to, and affect the activity of, a wide range of cellular regulatory proteins (Clapham, Reference Clapham2007; Edel et al., Reference Edel, Marchadier, Brownlee, Kudla and Heetherington2017) and different Ca2+ cyt elevation patterns represent signatures that can differentially activate a wide range of downstream elements (including calmodulin (CaM),calmodulin-like (CMLs), calcineurin B-like (CBLs), calcium-dependent protein kinases (CDPKs) and calcium/calmodulin kinases (CCamKs)) in stimulus- and cell-specific manners to bring about specific end responses (Demidchick et al., Reference Demidchick, Shabala, Isayenkov, Cuin and Pottosin2018; Edel et al., Reference Edel, Marchadier, Brownlee, Kudla and Heetherington2017; Lenzoni et al., Reference Lenzoni, Liu and Knight2018).
The generation of specific Ca2+ cyt signals involves the coordinate orchestration of channels, which allow passive movement of Ca2+ down its electrochemical potential gradient and active transporters that maintain resting [Ca2+ cyt] and return Ca2+ to baseline levels, shaping the Ca2+ cyt signature. This review provides an assessment of the key components of the Ca2+ homeostatic and signalling machinery. We consider some of the most significant recent advances and key questions still to be addressed. These include: What determines the set points for Ca2+ homeostasis? What are the relative capacities and roles of the different cellular Ca2+ buffering compartments? To what extent do these compartments act as releasable Ca2+ stores and how does this vary with different signalling processes?
Addressing these questions requires quantitative assessments of the contribution of individual and coordinated cellular compartments in the regulation of Ca2+ cyt. Until recently, measurement of concentrations and fluxes into and out of cellular compartments was primarily based on isolated organelles or microelectrode measurements of larger compartments, such as vacuoles. Recent years have witnessed a number of revolutionary advances, including improved electrophysiological approaches and the advent of targeted genetically encoded fluorescent Ca2+ reporters, that will provide new insights into the roles of different components of the Ca2+ homeostatic machinery and how its perturbation is finely controlled.
2. Components of the plant Ca2+ homeostat
2.1. Cytosolic buffers
A number of theoretical and experimental studies have shown that steady-state Ca2+ cyt levels in eukaryotic cells are determined primarily by the balance between influx and efflux mechanisms rather than passive cytosolic buffering (Eisner et al., Reference Eisner, Neher, Taschenberger and Smith2023; Rios, Reference Rios2010). However, cytosolic Ca2+ buffers do play an important role in modulating the rates of change of [Ca2+ cyt] as well as the amplitude of Ca2+ cyt elevations in response to changes in membrane Ca2+ fluxes (Eisner et al., Reference Eisner, Neher, Taschenberger and Smith2023). Consider a typical PM Ca2+ channel passing around 0.5 pA or ~106 Ca2+ ions/s in a single cuboid plant cell of volume approximately 10−14 m3 and a typical cytosolic volume of 10% total cellular volume. A simple calculation reveals that in the absence of any cytosolic buffering a single Ca2+ channel with an open probability of 0.5 could potentially raise the [Ca2+ cyt] at a rate of ~5 μM s−1. While there are no reliable estimates of Ca2+ channel density in the PM of plant or algal cells, single-channel patch clamp studies (e.g. Taylor et al., Reference Taylor, Manison, Fernandez, Wood and Brownlee1996; White et al., Reference White, Biskup, Elzenga, Homann, Thiel, Wissing and Maathius1999) suggest a conservative estimate of 0.5 channels μm−2, which would equate to a total potential channel complement in the thousands. Clearly, if all Ca2+ channels in the PM open simultaneously, then without buffering or active mechanisms to remove Ca2+, [Ca2+ cyt] could potentially elevate at a rate of several mM s−1 until equilibrium concentrations were achieved across the PM. A similar calculation based on a typical whole-cell plant Ca2+ current of around 100 pA gives a similar rate of [Ca2+ cyt] increase in the absence of buffering. Since most whole-cell stimulus-induced Ca2+ transients do not reach peak values higher than the low μM, there must exist highly efficient mechanisms for Ca2+ buffering or removal. Passive Ca2+ cyt buffers include Ca2+-binding proteins, polyvalent inorganic and organic anions anionic lipid heads, and carboxyl residues (Demidchick et al., Reference Demidchick, Shabala, Isayenkov, Cuin and Pottosin2018; Eisner et al., Reference Eisner, Neher, Taschenberger and Smith2023; Schwaller, Reference Schwaller2010). While the buffering capacity of plant cytosol has not been precisely determined and will vary with cell type, in a typical animal cell, the presence of a range of Ca2+ buffers with Kd values slightly higher than resting [Ca2+ cyt], suggests that passive buffering would become more effective as [Ca2+ cyt] began to rise above resting levels (Schwaller, Reference Schwaller2010). The affinities, concentrations, kinetics and mobilities of the Ca2+ buffers will subsequently determine the rate of [Ca2+ cyt] elevation and its amplitude (Eisner et al., Reference Eisner, Neher, Taschenberger and Smith2023; Neher, Reference Neher1998; Wagner & Keizer, Reference Wagner and Keizer1994), as well as the extent of Ca2+ cyt gradients resulting from localized PM Ca2+ fluxes in polarized plant cells, such as pollen tubes (e.g. Pierson et al., Reference Pierson, Miller, Callaham, Shipley, Rivers, Cresti and Hepler1994). The ratio of bound/free Ca2+ in plant cytosol has been estimated to be >90% (Demidchick et al., Reference Demidchick, Shabala, Isayenkov, Cuin and Pottosin2018; Schonknecht & Bethmann, Reference Schonknecht and Bethmann1998) and as little as 1% of total Ca2+ cyt is considered to be free in a typical animal cell (Eisner et al., Reference Eisner, Neher, Taschenberger and Smith2023), implying that an elevation of free [Ca2+ cyt] from 200 nM to 2 μM would require an influx sufficient to increase total Ca2+ cyt by >20 μM. The impact of strong cytosolic Ca2+ buffering is also evident from a wide range of animal studies that have shown highly localized Ca2+ elevations (sparks) at sites of Ca2+ entry through channels that only propagate further through coordinated Ca2+-dependent Ca2+ release from intracellular stores (Cheung & Lederer, Reference Cheung and Lederer2008). Non-equilibrium buffering of Ca2+ influx through a single channel is dependent on the rate of Ca2+ diffusion from the mouth of the channel and the probability that a Ca2+ ion will encounter the Ca2+ binding site of a buffer molecule, which is critically dependent on the buffer concentration (Stern, Reference Stern1992). The extent of [Ca2+ cyt] increase at the channel mouth will also depend on the rate of diffusion of Ca2+-buffer away from the channel as well as the buffer affinity, resulting in an exponential spatial [Ca2+ cyt] decay profile (Stern, Reference Stern1992). While there are no direct examples of such elemental Ca2+ elevations in vascular plants, these have been observed in rhizoid cells of the brown alga Fucus serratus in response to osmotically-induced channel activation on the PM and endomembranes (Goddard et al., Reference Goddard, Manison, Tomos and Brownlee2000) (Figure 1).
Highly localized Ca2+ elevations, visualized with the fluorescent Ca2+ indicator Calcium Green dextran, at the apex of a Fucus serratus rhizoid cell during the initiation of a Ca2+ cyt elevation in response to hypoosmotic treatment. Discrete Ca2+ elevations (red) are apparent and do not propagate >1 μm from the PM during putative channel opening (O) and disappear during channel closure (C). Adapted from Goddard et al. (Reference Goddard, Manison, Tomos and Brownlee2000).

2.2. Ca 2+ entry pathways
2.2.1. Apoplast-plasma membrane
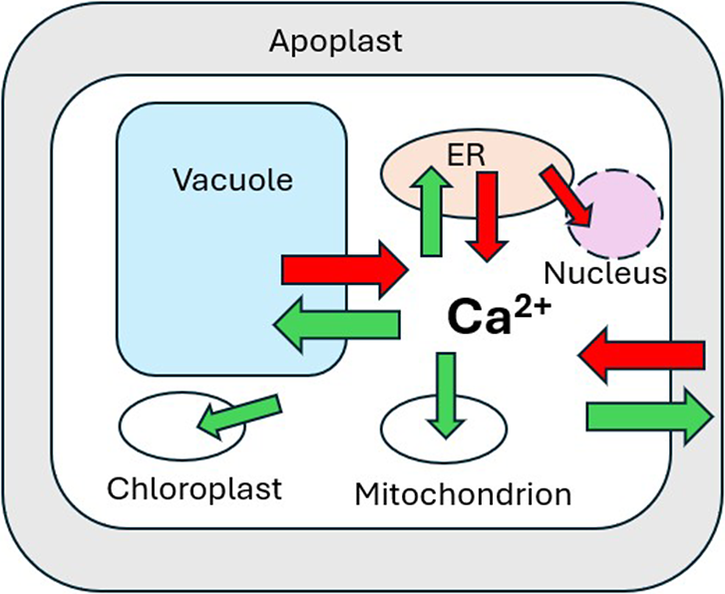
The apoplast represents the primary source of Ca2+ entering a plant cell with variable apoplastic [Ca2+] reported from a number of studies (Figure 2; e.g. Felle & Hanstein, Reference Felle, Hanstein, Sattlemacher and Horst2007; Conn et al., Reference Conn, Gilliham, Athman, Schreiber, Baumann, Moller, N-H, Stancombe, Hirschi, Webb, Burton, Kaiser, Tyerman and Leigh2011; Stael et al., Reference Stael, Wurzinger, Mair, Mehlmer, Vothnecht and Teige2012). This, coupled with the large negative PM membrane potential (Vm) produces a large inward-directed electrochemical potential gradient (ΔμCa2+). Ca2+ entry across the plasma membrane occurs primarily via Ca2+-permeable channels, including cyclic nucleotide-gated channels (CNGCs), glutamate receptors (GLRs), and mechanosensitive channels (MSLs, MCas and OSCAs) (Basu & Haswell, Reference Basu and Haswell2017; Brownlee & Wheeler, Reference Brownlee and Wheeler2023; Demidchick et al., Reference Demidchick, Shabala, Isayenkov, Cuin and Pottosin2018; Edel et al., Reference Edel, Marchadier, Brownlee, Kudla and Heetherington2017; Guichard et al., Reference Guichard, Thomine and Frachisse2022; Jiang & Ding, Reference Jiang and Ding2023; Tian et al., Reference Tian, Wang, Gao, Li and Luan2020; Yoshimura et al., Reference Yoshimura, Iida and Iida2021). Nucleotide-binding leucine-rich repeat receptors (NLRs) mediate immune responses and cell death in response to pathogens. Two plant NLRs (N REQUIREMENT GENE 1 (NRG1) and ZAR1) have also been shown to form Ca2+ channels in Arabidopsis involved in Ca2+-mediated resistance to pathogen attack (Bi et al., Reference Bi, Su, Li, Liang, Dang, Xu, Hu, Wang, Zou, Deng, Li, Huang, Li, Chai, He, Chen and Zhou2021; Jacob et al., Reference Jacob, Kim, Wu, El-Kasmi, Chi, Walton, Furzer, Lietzan, Sunil, Kempthorn, Redinbo, Pei, Wan and Dangl2021).
Ca2+ entry channels can be activated in highly specific manners by a very wide range of biotic and abiotic stimuli and can be subject to multiple forms of regulation, including voltage-dependent activation and inactivation (Hille, Reference Hille1978). For, example, in root hairs, CNGCs, including CNGC5, CNGC6, CNGC9 and CNGC14 play important roles in apically localised Ca2+ signalling (Tan et al., Reference Tan, Yang, Zhang, Fei, Gu, Sun, Xu, Wang, Liu and Wang2020; Tian et al., Reference Tian, Wang, Gao, Li and Luan2020). Notably, CNGC14 is inhibited by elevated [Ca2+ cyt] via calmodulin (Zeb et al., Reference Zeb, Wang, Hou, Zhang, Dong, Zhang, Zhang, Ren, Tian, Zhu, Li and Liu2020). In pollen tubes the Ca2+- permeable CNGC18/CNGC8 heterotetramer is preferentially localized at the growing tip and becomes active through interaction with calmodulin (CaM2) at low [Ca2+ cyt], leading to increased Ca2+ influx (Frietsch et al., Reference Frietsch, Wang, Sladek, Poulsen, Romanowsky, Schroeder and Harper2007; Gao et al., Reference Gao, Fei, Dong, Gu and Wang2014; Pan et al., 2019; Tian et al., Reference Tian, Wang, Gao, Li and Luan2020). GLRs also contribute significantly to the shaping of the pollen tube Ca2+ cyt gradient (Michard et al., Reference Michard, Simon, Tavares, Wudick and Feijo2017; Tian et al., Reference Tian, Wang, Gao, Li and Luan2020) and regulation of their activity and localization involves CORNICHON homologues (AtCNIHs) (Wudick et al., 2018). In stomatal guard cells, three classes of channels are involved in the closure response to external stimuli and are differentially regulated in response to external cues. The kinase BIK1 activates OSCA1.3 in response to bacterial flagellin (flg22) (Thor et al., Reference Thor, Jiang, Michard, George, Scherzer, Huang, Dindas, Derbyshire, Leitão, DeFalco, Köster, Hunter, Kimura, Gronnier, Stransfeld, Kadota, Bücherl, Charpentier, Wrzaczek, MacLean, Oldroyd, Menke, Roelfsema, Hedrich, Feijo and Zipfel2020). Abscisic acid (ABA) was recently shown to activate CNGC channels via the Ca2+-independent kinase OST1 (Yang et al., Reference Yang, Tan, Wang, Li, Du, Zhu, Wang and Wang2024). GLR channels, activated by external L-methionine were also shown to be involved in stomatal Ca2+ signalling, involving further Ca2+ channel activation via reactive oxygen (ROS) production (Kong et al., Reference Kong, Hu, Okuma, Lee, Lee, Munemasa, Che, Ju, Pedoeim, Rodriquez, Wang, Im, Murata, Pei and Kwak2016). There are also numerous reports of depolarization- and hyperpolarization-activated Ca2+ channels in plants for which detailed electrophysiological information is available (Demidchick et al., Reference Demidchick, Shabala, Isayenkov, Cuin and Pottosin2018). Genes encoding depolarization-activated Ca2+ channels have only been identified in animals and those encoding hyperpolarization currents in guard cells or root hairs remain unidentified.
Ca2+ concentrations, gradients and membrane potentials in a typical plant cell. Blue triangles represent the magnitude and direction of the electrochemical potential gradient (ΔμCa2+).

While Ca2+ entry across the PM may initiate elevation of [Ca2+ cyt] during signalling events, there is good evidence that in many cases Ca2+ release from intracellular stores may account for the bulk of Ca2+ entering the cytosol:
2.2.2. Channel-mediated Vacuolar Ca2+ release
The vacuole may occupy more than 90% of the total cell volume in a typical plant cell. While vacuolar free [Ca2+] ([Ca2+ vac]) can vary substantially (Pottosin & Schonknecht, Reference Pottosin and Schonknecht2007), microelectrode measurements indicate [Ca2+ vac] in the low mM range (Felle, Reference Felle1988). This, coupled with a cytosol-negative membrane potential (Dindas et al., Reference Dindas, Dreyer, Huang, Hedrich and Roelfesma2021) generates a large (ΔμCa2+) directed into the cytosol (Figure 2). Despite the obvious potential to represent the largest releasable Ca2+ store, the role of vacuolar Ca2+ release during signalling remains unclear. Direct involvement of the Ca2+-permeable slow vacuolar (SV) channel TPC1 (Peiter et al., Reference Peiter, Maathuis, Mills, Knight, Pelloux, Hetherington and Sanders2005) as a major pathway for vacuolar Ca2+ release into the cytosol (Hedrich & Neher, Reference Hedrich and Neher1987; Ward & Schroeder, Reference Ward and Schroeder1994) was questioned following the finding that Arabidopsis Attpc1 mutants did not show any differences from wild type in stomatal [Ca2+ cyt] elevations in response to external ABA or methyl jasmonate (Islam et al., Reference Islam, Munemasa, Hossain, Nakamura, Mori and Murata2010). Attpc1 mutants were able to close their stomata normally in response to ABA but showed either no response (Peiter et al., Reference Peiter, Maathuis, Mills, Knight, Pelloux, Hetherington and Sanders2005) or a reduced response to increased external Ca2+ (Islam et al., Reference Islam, Munemasa, Hossain, Nakamura, Mori and Murata2010). Moreover, long-distance salinity- or wounding-induced root-shoot Ca2+ waves were respectively significantly slower or abolished in Attpc1 mutants (Choi et al., Reference Choi, Toyota, Kim and Gilroy2014; Kiep et al., Reference Kiep, Vadassery, Lattke, Maass, Boland, Peiter and Mithofer2015).
Mechanosensitive tonoplast-localized Ca2+-permeable (PIEZO) channels have been shown to be involved in the transduction of mechanical signals in Arabidopsis root columnellar cells (Mousavi et al., Reference Mousavi, Dubin, Zeng, Coombs, Do, Ghadiri, Keenan, Ge, Zhao and Patapoutian2021). Chimaeras of AtPIEZO with mouse mPIEZO generated non-selective mechanosensitive currents in HEK cells. PIEZO channels are therefore potentially involved in mechanosensitive Ca2+ release from the vacuole. PIEZO channels were also shown to contribute to Ca2+ cyt oscillations in the moss Physcomitrium and to be an important factor regulating vacuolar morphology (Radin et al., Reference Radin, Richardson, Coomey, Weiner, Bascom, Li, Bezanilla and Haswell2021).
Examples of identified endomembrane transporters with demonstrated roles in modulation of Ca2+ cyt or organellar Ca2+.

2.2.3. Endoplasmic reticulum (ER)
While the Vm across the ER membrane has not been directly measured in plants, in animals ER Vm is clamped to near zero by high K+ conductance and equimolar [K+] on both sides of the ER membrane (Lam & Galione, Reference Lam and Galione2013). However, an ER free [Ca2+] ([Ca2+ ER]) >100 μM (Daverkausen-Fischer & Pröls, Reference Daverkausen-Fischer and Pröls2022) establishes a large cytosol−directed ΔμCa2 (Figure 2). While most studies of [Ca2+ ER] changes in response to various stimuli do not report calibrated values in plants, lower affinity variants of GECI reporters (e.g. ER-GCAMP6) have been used successfully to report [Ca2+ ER] changes (Resentini et al., Reference Resentini, Grenzi, Ancora, Cademartori, Luoni, Franco, Bassi, Bonza and Costa2021a). In animals, the Ca2+ release pathway from the ER is very well studied with both Inositol 1,4,5 trisphosphate (InsP3) and ryanodine receptors acting as the major pathways for Ca2+ release (Katona et al., Reference Katona, Bartók, Nichtova, Csordás, Berezhnaya, Weaver, Ghosh, Várnai, Yule and Hajnóczky2022; Lam & Galione, Reference Lam and Galione2013). However, while there are reports of InsP3-induced Ca2+ release in plant cells (e.g. Manzoor et al., Reference Manzoor, Chiltz, Madani, Vatsa, Schoefs, Pugin and Garcia-Brugger2012; Muir & Sanders, Reference Muir and Sanders1996), vascular plants do not possess canonical InsP3 or ryanodine receptors (Edel et al., Reference Edel, Marchadier, Brownlee, Kudla and Heetherington2017; Verret et al., Reference Verret, Wheeler, Taylor, Farnham and Brownlee2010) and there is no characterized mechanism for facilitating Ca2+ release directly from the ER into the cytosol in plants. A clear exception, however, comes from work with root nodulation and mycorrhizal symbioses responses (Lam et al., Reference Lam, Cooke, Wright, Lawson and Charpentier2024). Distinct from plasma membrane channel-mediated increases in [Ca2+ cyt], mycorrhizal (Myc) factors from arbuscular mycorrhizal fungi and nodulation (Nod) factors from rhizobia can give rise to Ca2+ elevations localized to the nucleus (Charpentier et al., Reference Charpentier, Sun, Martins, Radhakrishnan, Findlay, Soumpourou, Thouin, Very, Sanders, Morris and Oldroyd2016; Lam et al., Reference Lam, Cooke, Wright, Lawson and Charpentier2024; Oldroyd, Reference Oldroyd2013). In the legumes of Medicago trunculata or Lotus japonicus, Nod factor perception by LysM-type plasma membrane receptors is conveyed to the ER-derived nuclear envelope through a cytosolic mevalonate pathway (Venkateshwaran et al., Reference Venkateshwaran, Jayaraman, Chabaud and Ane2015). Interaction between CNGC15 and the CASTOR/POLLUX/DMI1 channels on the inner nuclear envelope membrane underlies the release of Ca2+ into the nucleoplasm. While DMI1 has been proposed to behave as a K+ channel, more recent evidence suggests that both CNGC15 and DMI1 are Ca2+ channels (Kim et al., Reference Kim, Zeng, Bernard, Liao, Venkatechwaran, Ane and Jiang2019).
While molecular evidence for specific pathways underlying ER-mediated Ca2+ release into the cytosol is lacking, there is clear physiological evidence for a role for ER Ca2+ release in Ca2+ cyt signalling. The mechanisms of trap closure by the Venus flytrap involve an initial depolarization of the trap lobe cell PM following mechanical stimulation of trap lobe hair cells. This involves an initial influx of Ca2+, most likely through GLR3.6 channels (Scherzer et al., Reference Scherzer, Bohm, Huang, Iosip, Kreuzer, Becker, Heckmann, Al-Rasheid, Dreyer and Hedrich2022). Based on the effects of the ER Ca2+-ATPase inhibitor cyclopiazonic acid (CPA), which increased resting [Ca2+ cyt] but decreased the amplitude of the transient Ca2+ elevation required for the trap closure response, Scherzer et al. (Reference Scherzer, Bohm, Huang, Iosip, Kreuzer, Becker, Heckmann, Al-Rasheid, Dreyer and Hedrich2022) deduced that an initial [Ca2+ cyt] elevation associated with Ca2+ influx through GLR channels was augmented substantially by Ca2+ release from the ER. A similar role for ER Ca2+ release in elevation of [Ca2+ cyt] has recently been indicated by Huang et al. (Reference Huang, Shen, Roelfsema, Becker and Hedrich2023), who monitored Ca2+ cyt in Arabidopsis guard cells expressing a light-activated H+-permeable channelrhodopsin (HcKCR2) to generate H+-induced [Ca2+ cyt] elevations. Pharmacological treatment with cyclopiazonic acid (CPA) or low apoplastic Ca2+ (EGTA) indicated a role in ER Ca2+ release. Moreover, repetitive stimulation led to a stepwise reduction of the Ca2+ cyt signals, indicating that ER Ca2+ stores could be depleted if Ca2+ release exceeded the recharging ability of ER-localized ATPases (see below). More recently, Huang et al. (Reference Huang, Roelfsema, Gilliham, Hetherington and Hedrich2024) used the Ca2+-permeable, blue light-activated optogenetic probe channelrhodopsin ChR2-XXM2.0 to elicit pulses of Ca2+ influx across the PM of stomatal guard cells. This study showed that Ca2+ influx gave rise to Ca2+ cyt elevations, which were also associated with incremental closure of the stomata. CPA prevented the ChR-induced transient Ca2+ cyt elevation, presumably by inhibiting the loading of Ca2+ cyt into ER stores and provided evidence for ER store depletion with repetitive (every 6 min) Ca2+ release events.
2.3. Transporters maintaining Ca 2+ cyt homeostasis
A combination of empirical and modelling approaches has provided evidence for the recovery phase of Ca2+ signals in determining the nature of downstream signalling responses (Lenzoni et al., Reference Lenzoni, Liu and Knight2018). The rate of recovery of Ca2+ cyt following elevation, and the set point for resting [Ca2+ cyt] is largely determined by the affinity and regulated activity of Ca2+ extrusion systems on the PM and endomembranes (Table1). Two main classes of Ca2+-ATPases remove Ca2+ from the cytosol: Type PIIA (ER-type, ECA) located mainly, but not restricted to endomembranes, unlike animal ECA ATPases, and Type IIB, autoinhibited ATPases (ACA), which can be located at both PM and endomembranes (vacuole, ER, Golgi) (see Costa et al., Reference Costa, Resentini, Buratti and Bonza2023 for a recent review). ATPases exchange 1 Ca2+ for 1 or 2 H+, therefore utilising both the energy of ATP hydrolysis and the H+ electrochemical potential gradient. Arabidopsis AtACA8 comprises 10 transmembrane domains and a cytoplasmic head with nucleotide binding and CaM binding domains. Other regulatory mechanisms include phosphorylation (e.g. via CPK1/16, CIPK9/14, CBL1, CaM, CML36) and differences in ACA regulatory sequences likely reflect differential regulation (Costa et al., Reference Costa, Resentini, Buratti and Bonza2023). To date, only one ECA regulatory protein (MIZ1) has been identified (Yamazaki et al., Reference Yamazaki, Miyazawa, Kobayashi, Moriwaki, Fujii and Takahashi2012).
ATPases show diverse patterns of localization that are both cell-type and species-dependent (Costa et al., Reference Costa, Resentini, Buratti and Bonza2023). For example, Arabidopsis possesses 4 Type IIA ECAs and 10 Type IIB ACAs, 5 of which are located at the PM. ACA9 is expressed in pollen tubes while ACA8 and ACA10 are expressed in vegetative cells. ACA12 and ACA13 13 are preferentially expressed under biotic or abiotic stress. Both type IIA ECA and IIB ACA are found in the ER membrane. Two type II ACAs (ACA4 and ACA11) are localized to the vacuole. ATPases and other transporters potentially serve two roles. Firstly, to restore and maintain resting [Ca2+ cyt] and secondly to charge up Ca2+ stores involved in Ca2+ release during signalling.
2.3.1. Plasma membrane transporters
The roles of different Ca2+-ATPases have been inferred from both genetic and inhibitor studies (Costa et al., Reference Costa, Resentini, Buratti and Bonza2023; Demidchick et al., Reference Demidchick, Shabala, Isayenkov, Cuin and Pottosin2018). Mutant studies have revealed significant redundancy and have indicated important roles for localization in determining function (Costa et al., Reference Costa, Resentini, Buratti and Bonza2023; Resentini et al., Reference Resentini, Grenzi, Ancora, Cademartori, Luoni, Franco, Bassi, Bonza and Costa2021a; Resentini et al., Reference Resentini, Ruberti, Grenzi, Bonza and Costa2021b). Examples include impaired pathogen defence responses and attenuated Ca2+ cyt signals of PM-localized Ataca8/10 double knockout mutants in response to flg22 (Frei dit Frey et al., Reference Frei dit Frey, Mbengue, Kwaaitaal, Nitsch, Altenbach, Häweker, Lozano-Duran, Njo, Beeckman, Huettel, Borst, Panstruga and Robatzek2012). Behera et al. (Reference Behera, Xu, Luoni, Bonza, Doccula, De Michelis, Morris, Schwarzländer and Costa2018) showed that resting Ca2+ cyt was unchanged in aca8/10 double mutants, which also showed decreased Ca2+ cyt signal amplitude and delayed recovery in response to external ATP compared with wild-type plants. This response was considered to reflect a degree of acclimation via modified expression of other transporters. By monitoring both Ca2+ cyt and pH these workers also demonstrated that Ca2+ and pH fluxes were tightly linked.
2.3.2. Tonoplast transporters
In contrast to mutants with disabled PM ATPases Ataca4/11 double knockout mutants of vacuolar Ca2+-ATPases have elevated baseline [Ca2+ cyt], elevated Ca2+ cyt response to CO2 and enhanced defence responses (Hilleary et al., Reference Hilleary, Paez-Valencia, Vens, Toyota, Palmgren and Gilroy2020). By imaging [Ca2+ cyt] with a YC-Nano65 sensor at both whole organ and sub-cellular scales, the flg22 response was found to be homogeneous across cells. Mis-localization of PM ACA8 suppressed the aca4/11 phenotype, despite not having the same regulatory elements as ACA4/11. Perhaps surprisingly, while aca4/11 mutants showed a significantly higher [Ca2+ cyt] signal in response to flg22, [Ca2+ cyt] returned to basal levels in a similar time frame as wild-type plants, suggesting the involvement of other efflux systems in re-establishing baseline [Ca2+ cyt]. In contrast, knockout of the Physcomitrium tonoplast Ca2+-ATPase PCA1 gave higher Ca2+ cyt transients in response to high NaCl, which were of longer duration than wild type (Qudeimat et al., Reference Qudeimat, Faltusz, Wheeler, Lang, Holtorf, Brownlee, Reski and Frank2008). These studies also indicated that vacuolar Ca2+-ATPases act very quickly to modulate the amplitude of the Ca2+ cyt signal.
CAX transporters belong to the multigene family of cation/H+ exchangers (Demidchick et al., Reference Demidchick, Shabala, Isayenkov, Cuin and Pottosin2018, Pittman & Hirschi, Reference Pittman and Hirschi2016). Plant CAX transporters have a lower affinity for Ca2+ than Ca2+-ATPases and transport H+ and Ca2+ in a 3:1 ratio (Demidchick et al., Reference Demidchick, Shabala, Isayenkov, Cuin and Pottosin2018; Dindas et al., Reference Dindas, Dreyer, Huang, Hedrich and Roelfesma2021). Arabidopsis possesses 6 CAX genes (AtCAX1–6) and 5 further Ca2+/cation antiporters that behave as K+-dependent Na+/Ca2+ exchangers (Manohar et al., Reference Manohar, Shigaki and Hirschi2011; Maser et al., Reference Maser, Thomine, Schroeder, Ward, Hirschi, Sze, Talke, Amtmann, Maathuis, Sanders, Harper Tchieu, Gribskov, Persans, Salt, Kim and Guerinot2001, Shigaki et al., Reference Shigaki, Rees, Nakhleh and Hirshi2006). CAX transporters possess an N-terminal autoinhibitory domain and transport specificity is controlled by a 9 amino acid region between TM1 and TM2. Activity depends on ΔμH+ across the tonoplast, the degree of phosphorylation and interaction of regulatory proteins with the N-terminal region (Demidchick et al., Reference Demidchick, Shabala, Isayenkov, Cuin and Pottosin2018; Matthew et al., Reference Matthew, Rhein, Yang, Gradogna, Carpaneto, Guo, Tappero, Scholze-Starke, Barkla, Hirchi and Punshon2024; Pittman & Hirchi, 2016; Wang et al., Reference Wang, Tang, Kuo, Xu, Lu, Rauscher, Voelker and Luan2024).
Despite their lower affinity, physiological and molecular studies indicate that CAX transporters play a pivotal role in the regulation of Ca2+ cyt dynamics. They also play important roles in the regulation of cytosolic and apoplast pH (Cho et al., Reference Cho, Villiers, Kroniewicz, Lee, Seo, Hirschi, Leonhardt and Kwak2012). Arabidopsis det3 V-type H+-ATPase mutants (Allen et al., Reference Allen, Chu, Schumaker, Shimazaki, Vafeados, Kemper, Hawke, Tallman, Tsien, Harper, Chory and Schroeder2000) had altered Ca2+ dynamics in response to increased apoplastic Ca2+ and ROS, displaying sustained stomatal guard cell Ca2+ cyt elevations rather than oscillations normally associated with these stimuli, likely reflecting defective Ca2+/H+ regulation via Ca2+/H+ transporters. However, det3 mutants did show a normal pattern of Ca2+ cyt oscillations in response to ABA. Electrophysiological manipulation of tonoplast Vm combined with Ca2+ cyt imaging in Arabidopsis root hairs has provided further evidence for tonoplast Ca2+/H+ exchange in the regulation of [Ca2+ cyt] (Dindas et al., Reference Dindas, Dreyer, Huang, Hedrich and Roelfesma2021). Depolarizing the tonoplast (i.e. rendering the cytosolic side more positive) elevated [Ca2+ cyt] and reduced [H+]cyt. This can be interpreted as reduced ΔμH+ leading to reduced Ca2+ uptake into the vacuole in exchange for H+. Hyperpolarizing the tonoplast produced the opposite effect. Two recent reports provide further direct evidence for the roles of CAX in the maintenance of Ca2+ cyt homeostasis. Bakshi et al. (Reference Bakshi, Choi, Kim and Gilroy2023) showed transcripts of CAX2 and ACA1 were rapidly upregulated in Arabidopsis plants subject to flooding or hypoxia. Moreover, cax2 knockout mutants showed larger and more sustained Ca2+ cyt signals and enhanced survival in response to flooding. In a separate study (Conn et al., Reference Conn, Gilliham, Athman, Schreiber, Baumann, Moller, N-H, Stancombe, Hirschi, Webb, Burton, Kaiser, Tyerman and Leigh2011) Arabidopsis cax1/3 double mutants had reduced overall mesophyll Ca2+ contents and apolastic free Ca2+ that was 3-fold higher than wild-type plants. This was indicative of compensatory increased PM ATPase activity in response to reduced tonoplast Ca2+ transport.
2.3.3. ER transporters
There are numerous reports of disruption of ER Ca2+-ATPases leading to altered Ca2+ cyt signalling and downstream responses (Costa et al., Reference Costa, Resentini, Buratti and Bonza2023). Analysis of Ca2+ ER dynamics in pollen tubes expressing ER-localized yellow cameleon 3.6 Ca2+ sensor showed that CPA triggered growth arrest and a decrease in [Ca2+ ER] (Iwano et al., Reference Iwano, Entani, Shiba, Kakita, Nagai, Mizuno, Miyawaki, Shoji, Kubo, Isogai and Takayama2009). CPA also reduced the tip-focused Ca2+ cyt oscillations in the growing pollen tube tip and caused [Ca2+ cyt] to elevate in sub-tip regions indicating a key role for ER Ca2+-ATPase in regulating the tip-focused [Ca2+ cyt] gradient. More specifically, Arabidopsis triple mutants aca1/2/7 of ER-localized Ca2+-ATPase show higher Ca2+ cyt response to flg22 or blue light, higher resting [Ca2+ cyt] and associated changes in downstream responses (Ishka et al., Reference Ishka, Brown, Rosenberg, Romanowsky, Davis, Choi and Harper2021). Triple aca1/2/7 mutants also had slower recovery of [Ca2+ cyt] to resting levels following repeated cycles of elevated CO2 as well as altered stomatal conductance (Jezek et al., Reference Jezek, Silva-Alvim, Hills, Donald, Ishka, Shadbolt, He, Lawson, Harper, Wang, Lew and Blatt2021). Increases in [Ca2+ cyt] were also progressively reduced in mutants in response to successive CO2 cycles, indicating that ACA-mediated recovery of the ER Ca2+ store was required for response to repeated stimuli. A further role for Ca2+-ATPase in shaping Ca2+ signatures comes from studies of nod factor signalling in Medicago. Silencing the nuclear envelope-localized Ca2+-ATPase MCA8 blocked nod factor-induced nuclear Ca2+ oscillations (Capoen et al., Reference Capoen, Sun, Wysham, Otegui, Venkateshwaran, Hirsch, Miwa, Downie, Morris, Ané and Oldroyd2011).
A role for an ER-localised cation/Ca2+ exchanger (CCX) in the regulation of both [Ca2+ ER] and [Ca2+ cyt] in Arabidopsis has also been demonstrated (Corso et al., Reference Corso, Doccula, de Melo, Costa and Verbruggen2018). Surprisingly, knockout of AtCCX resulted in decreased [Ca2+ cyt] and increased [Ca2+ ER] under salt and osmotic stress conditions. The underlying mechanism and role in cytosol-ER Ca2+ exchange have yet to be fully elucidated.
3. Mitochondrial and chloroplast Ca2+ transport
In animal cells, mitochondrial Ca2+ uptake is critical for control of energy metabolism and mitochondria play a fundamental role in shaping spatio-temporal Ca2+ cyt increases. This occurs primarily via InsP3-induced release of Ca2+ from ER stores during Ca2+ cyt wave propagation and reuptake of Ca2+ by mitochondrial Ca2+ uniporters (MCUs) at specific locations where mitochondria make close contact with the ER (ER-mitochondrial contacts, ERMCs) (Katona et al., Reference Katona, Bartók, Nichtova, Csordás, Berezhnaya, Weaver, Ghosh, Várnai, Yule and Hajnóczky2022; Lee et al., Reference Lee, Huh, Lee, Wu, Kim, Kamg and Lu2018). Mitochondria maintain a large inside-negative Vm across the inner mitochondrial membrane (Zorova et al., Reference Zorova, Popkova, Plotnikova, Silacheva, Pevznera, Jankauskasa, Babenkoa, Zorovc, Balakirevad, Juhaszovae, Sollotte and Zorov2018) resulting in an inwardly-directed ΔμCa2+, given mitochondrial matrix Ca2+ [Ca2+ mit] of 100–200 nM (Finkel et al., Reference Finkel, Menazza, Holmstrom, Parks, Liu, Sun, Pan and Murphy2015) (Figure 2). While there is little or no evidence for functional ERMCs in plant cells, recent work has shown that MCU proteins play a key role in Ca2+ uptake. Arabidopsis possesses 6 MCU homologues (Teardo et al., Reference Teardo, Carraretto, Wagner, Formentin, Behera, De Bortoli, Larosa, Fuchs, Lo Schiavo, Raffaello, Rizzuto, Costa, Schzrzlander and Szvabo2017) and Ruberti et al. (Reference Ruberti, Feitosa-Araujo, Xu, Wagner, Grenzi, Darwish, Lichtenauer, Fuchs, Parmagnani, Balcerowicz, Schoenaers, de la Torre, Mekkaoui, Nunes-Nesi, Wirtz, Vissenberg, Van Aken, Hause, Costa and Schwarzlander2022) demonstrated in vitro Ca2+ transport activity by Arabidopsis MCU and defective mitochondrial Ca2+ uptake in a mcu1/2/3 triple knockout mutant. However, this study also showed that Ca2+ cyt dynamics were unaffected in the triple mutant, indicating that mitochondria played a minimal role in Ca2+ cyt homeostasis. A homologue of the animal regulatory MCU-associated MICU proteins has been implicated in the regulation of MCU-mediated Ca2+ uptake (Wagner et al., Reference Wagner, Behera, De Bortoli, Logan, Fuchs, Carraretto, Teardo, Cendron, Nietzel, Füßl, Doccula, Navazio, Fricker, Van Aken, Finkemeier, Meyer, Szabò, Costa and Schwarzländer2015). Arabidopsis mice mutants showed higher and faster mitochondrial (Ca2+ mit) elevations in response to auxin and ATP while Ca2+ cyt remained unchanged. Thus, similar to animal cells, by modulating plant mitochondrial Ca2+ uptake MICU shapes mitochondrial Ca2+ signatures and helps to maintain mitochondrial Ca2+ homeostasis.
Similar to mitochondria, chloroplasts have a large inside negative membrane potential (Svabo & Spetea, Reference Svabo and Spetea2017) and a stromal [Ca2+] ([Ca2+ strom]) of 50–200 nM (Frank et al., Reference Frank, Happeck, Meier, Hoang, Stribny, Hause, Ding, Morsomme, Baginsky and Peiter2019; Hochmal et al., Reference Hochmal, Schulze, Trompelt and Hippler2015), maintained by active transporters such as the thylakoid membrane-localized Ca2+/H+ exchanger CCHA1 (Wang et al., Reference Wang, Xu, Jin, Zhang, Lai, Zhou, Zhang, Liu, Duan, Wang, Peng and Yang2016). A chloroplast-localized homologue of mitochondrial MCU transporters (cMCU) has also been identified and shown to mediate chloroplast Ca2+ uptake (Teardo et al., Reference Teardo, Carraretto, Moscatiello, Cortese, Vicario, Festa, Maso, Bortoli, de Bortoli, Cali, Vothknecht, Formentin, Cendron, Navazio and Szabo2019). By using targeted aequorin reporters they showed that cMCU was required for stress-specific Ca2+ strom signatures. The dynamic nature of chloroplast Ca2+ signalling is further illustrated by responses of Ca2+ strom to high light and temperature. By targeting YC3.6 to the cytosol, chloroplast and mitochondria in the green alga Chlamydomonas, Pivato et al. (Reference Pivato, Grenzi, Costa and Ballottari2023) monitored elevated Ca2+ specific to the chloroplast, which correlated with H2O2 production and was dependent on functional cryptochrome. Similarly, Flori et al. (Reference Flori, Dickenson, Gaikward, Cole, Smirnoff, Helliwell, Brownlee and Wheeler2024) demonstrated sustained Ca2+ strom elevations in response to high light or H2O2 treatments that were independent of changes in [Ca2+ cyt] and accompanied chloroplast H2O2 accumulation in high light. A further class of chloroplast Ca2+ transporter, BICAT proteins, are involved in the elevation of Ca2+ strom on transfer from light to dark (Frank et al., Reference Frank, Happeck, Meier, Hoang, Stribny, Hause, Ding, Morsomme, Baginsky and Peiter2019). Knockout mutations of BICAT1, which is located on the chloroplast envelope reduced the dark-induced Ca2+ strom signal, monitored with chloroplast-targeted aequorin. In contrast, knockout mutation of BICAT2, which transports Ca2+ into the thylakoid lumen increased the light-dark Ca2+ strom signal and produced severe defects in chloroplast morphology.
4. Insights from modelling studies
A number of modelling studies have provided insights into the interactions between signalling and homeostatic components of the plant Ca2+ signalling machinery. Bose et al. (Reference Bose, Pottosin, Shabala, Plamgren and Shabala2011) used a 4-component model, comprising 2 Ca2+-permeable channels on the PM and endomembrane, together with 2 efflux systems – a PM Ca2+-ATPase and an endomembrane Ca2+/H+ exchanger. They also factored in the ROS sensitivity of the endomembrane Ca2+ channel and the buffering capacity of the cytosol. The model predicted that specific Ca2+ signatures could be achieved by modifying the activities of ATPase and Ca2+/H+ exchangers. Dindas et al. (Reference Dindas, Dreyer, Huang, Hedrich and Roelfesma2021) combined electrophysiological manipulation of vacuolar Vm, Ca2+ cyt monitoring and modelling to demonstrate the role of a voltage-dependent vacoular Ca2+ homeostat involving tonoplast Ca2+/H+ exchange and vacuolar electrical excitability providing a clear demonstration that Ca2+ fluxes across the tonoplast are important in regulating Ca2+ cyt.
The “On guard” model (Jezek et al., Reference Jezek, Silva-Alvim, Hills, Donald, Ishka, Shadbolt, He, Lawson, Harper, Wang, Lew and Blatt2021) presents arguably the most comprehensive analysis of transport and essential metabolism in predicting stomatal signalling patterns and behaviour. The model considers Ca2+- and H+-ATPases, along with cation and anion channel activities shown to be associated with stomatal responses to ABA or CO2. The model accurately simulated elevated [Ca2+ cyt] and oscillations resulting from cyclic Ca2+ influx across the PM, promoting much larger Ca2+-induced Ca2+ release (CICR) from endomembrane stores. Modelling also predicted, and experiments verified, a delay in Ca2+ cycling that was enhanced in ER and tonoplast Ca2+-ATPase mutants, identifying both endomembrane Ca2+-ATPases and Ca2+ channels as important targets for the stomatal closure response to high CO2.
5. Outlook
There has been substantial progress in understanding the essential components of the plant Ca2+ homeostatic machinery and how these contribute to the shaping of Ca2+ signals in response to a wide range of stimuli in different plant, tissue and cell types. This progress has been facilitated largely by advances in molecular characterization of key transporters, along with the development of targeted genetically encoded indicators with differing affinities suited to imaging Ca2+ in the cytosol and other cellular compartments, along with increasingly refined modelling simulations. Technological advances in microscopy from sub-cellular to whole plant imaging have further enabled spatio-temporal Ca2+ signalling patterns to be analysed in far greater depth than previously possible. Discoveries enabled through studies of a wider range of organisms, such as the recently described MID1-COMPLEMENTING ACTIVITY (MCA) PM Ca2+-permeable channel with a role in the regulation of cell proliferation in the liverwort Marchantia polymorpha (Iwano et al., Reference Iwano, Suetsugu, Nishihama, Ishida, Horie, Costa, Katsuno, Kimura, Iida, Nagai and Kohchi2025) will provide further insights into the roles and evolution and of plant Ca2+ signalling components.
Despite these advances, there is still a need to further define the roles of endomembrane stores as sources and/or sinks for Ca2+. There has been substantial progress in understanding how both PM and endomembrane Ca2+-ATPases and CAX transporters can shape different Ca2+ signatures, though with still much to discover. While CNGCs have been identified as the major Ca2+ release pathway from the nuclear envelope underlying nuclear Ca2+ oscillations during symbiotic signalling, our more general understanding of the roles of endomembrane Ca2+ release mechanisms is less advanced. How is an initial Ca2+ influx across the PM augmented and amplified by the release of Ca2+ from internal stores? Of particular importance is the ongoing need to identify ER Ca2+ release mechanisms in the absence of molecular homologues of InsP3 receptors that are widespread in animal cells and play a central role in Ca2+ signalling. There is also a need to further understand the interactions between different endomembrane compartments. In animals, interactions between ER and mitochondria are pivotal in defining spatiotemporal Ca2+ signalling. To date, no similar evidence exists for plant cells. Are there similar direct interactions between the ER and mitochondria or chloroplasts in plants? The vacuole represents the largest potential Ca2+ stored in most plant cells. However, while tonoplast Ca2+ transporters have been shown to play a role in Ca2+ cyt homeostasis, apart from the recently discovered tonoplast PIEZO channels (Mousavi et al., Reference Mousavi, Dubin, Zeng, Coombs, Do, Ghadiri, Keenan, Ge, Zhao and Patapoutian2021; Radin et al., Reference Radin, Richardson, Coomey, Weiner, Bascom, Li, Bezanilla and Haswell2021) there is no fully characterized channel-mediated mechanism for vacuolar Ca2+ release associated with specific Ca2+ signalling events.
Finally, in animals, store-operated Ca2+ entry (SOCE) is an important mechanism for recharging ER stores during repetitive Ca2+ signalling and is a key component in shaping Ca2+ signatures. However, the molecular machinery for SOCE – STIM proteins that sense [Ca2+ ER] and ORAI channels in the PM that allow Ca2+ entry in close proximity to ER-PM contact points (Lunz et al., Reference Lunz, Romanin and Frischauf2019; Wang et al., Reference Wang, Wang, Zhou, Hendron, Mancarella, Andrake, Rothberg, Soboloff and Gill2014) are absent in embryophytes, though ORAI proteins are present in the green lineage as far as gymnosperms (Edel et al., Reference Edel, Marchadier, Brownlee, Kudla and Heetherington2017). This begs the question of whether SOCE exists, at least in embryophytes.
Acknowledgements
C.B. is grateful to the Marine Biological Association for its continued support in the preparation of this manuscript.
Competing interest
The authors declare none.
Data availability statement
This review article does not rely on original data or resources.
Author contributions
C.B. conceived the study. C.B. and G.L.W. wrote the article.
Funding statement
This work received no specific grant from any funding agency, commercial or not-for-profit sectors.
Open peer review
To view the open peer review materials for this article, please visit http://doi.org/10.1017/qpb.2025.2.





Comments
Dear Editor
I am submitting a review manuscript “Cellular calcium homeostasis in plants and regulation of its dynamic perturbation” to be considered for publication in Quantitative Plant Biology. This is an invited review for the special edition on Plant Ion Homoestasis.
The manuscript provides an overview of plant cellular calcium homeostatic mechanisms that have been characterized at molecular and physiological levels. It outlines the roles of different cellular compartments and transporters in contributing both to cytosolic calcium homeostasis and in shaping calcium signatures, pointing to gaps in knowledge and opportunities for future research.
Kind regards
Colin Brownlee
Glen Wheeler