Introduction
Patients with a history of excessive alcohol use are more likely to actively engage in smoking and illicit drug use, and are at risk for nonmedical opioid use (NMOU) (Dev et al. Reference Dev, Parsons and Palla2011), a condition which occurs in 20% of patients receiving opioids for cancer pain (Arthur et al. Reference Arthur, Tang and Lu2021; Carmichael et al. Reference Carmichael, Morgan and Del Fabbro2016, Yennurajalingam Reference Yennurajalingam, Arthur and Reddy2021). Several screening tools are used in clinical practice to detect alcohol-related issues including the Alcohol Use Disorders Identification Test, Michigan Alcohol Screening Test, Adolescent Drinking Index, and the Cut down, Annoyed, Guilty, and Eye opener (CAGE) questionnaire (Reid et al. Reference Reid, Fiellin and O’Connor1999). Of these, the CAGE questionnaire is the most widely used because of its concise nature which makes it more desirable among clinicians working in busy clinical settings (Allen et al. Reference Allen, Maisto and Connors1995). In a previous study, patients who screened positive for the CAGE questionnaire had worsening symptoms such as pain, sleep, dyspnea, well-being, and total symptom distress, as determined by the Edmonton Symptom Assessment System (ESAS) (Parsons et al. Reference Parsons, Delgado-Guay and El Osta2008). A variation of the CAGE questionnaire, the CAGE-Adapted to Include Drugs (CAGE-AID) questionnaire which substitutes “drink” with “drink or drugs,” has also been evaluated for the detection of substance misuse (Brown and Rounds Reference Brown and Rounds1995; Childers et al. Reference Childers, King and Arnold2014; Dev et al. Reference Dev, Parsons and Palla2011; Kwon et al. Reference Kwon, Tanco and Park2015). It is a well-validated patient self-administered screening tool with a high internal consistency reliability coefficient (r = 0.92) (Leonardson et al. Reference Leonardson, A. Koplin and Ness2005).
Inconsistent responses to the CAGE-AID questionnaire (CA) may reflect inaccurate patient self-report (Brown and Rounds Reference Brown and Rounds1995; Couwenbergh et al. Reference Couwenbergh, Van Der Gaag and Koeter2009; Katz and Fanciullo Reference Katz and Fanciullo2002) or irregular clinician administration of the questionnaire and has the potential to negatively impact effective patient care. The CA is theoretically considered a stable tool, but potential discrepancies in patient responses to the CA questionnaires in different clinical settings and visits have not been investigated. The main objective of this study was to evaluate the completion and positivity rate of the CA at the inpatient and outpatient supportive care center (SCC) visits among the same patient cohort. We also assessed changes in scoring of the questionnaire across the 2 clinical settings.
Method
Study participants, procedure, and data collection
Electronic records of 333 consecutive patients who were seen in the hospital and at the outpatient SCC between August 1, 2016 and March 31, 2017, were reviewed. Eligible patients were ≥18 years with a diagnosis of cancer who received an inpatient supportive care consultation, followed by an outpatient follow-up visit within 3 months of the initial consultation. The following demographic and clinical characteristics were collected at both visits: age, gender, race, and cancer diagnosis, ESAS, and the CA. Both CA and ESAS questionnaire are routinely administered during routine clinical care at the SCC. This study protocol was approved by the institutional review board at the University of Texas MD Anderson Cancer.
Instruments
The CA is a well-validated screening tool for excessive alcohol use and may suggest the possibility of an increased risk of maladaptive behavior when there is an exaggerated and inappropriate request for opioid medication. It comprises the following 4 question items: “have you ever felt that you should cut down on your drinking or drug use,” “have you ever been annoyed by people criticizing your drinking or drug use,” “have you ever felt bad or guilty about your drinking or drug use,” and “have you ever had a drink or used drugs first thing in the morning to get rid of a hangover (eye-opener).” Studies have shown that the CAGE-AID has a sensitivity of 88–91% and a specificity of 55–97% for the detection of current substance use disorder (Couwenbergh et al. Reference Couwenbergh, Van Der Gaag and Koeter2009; Dyson et al. Reference Dyson, Appleby and Altman1998). For our study, CAGE-AID positivity was defined as a total score ≥2 out of 4. A positive score raises concern for potential opioid misuse and chemical coping, which is an inappropriate and/or excessive use of opioids to cope with the various stressful events associated with the diagnosis and management of cancer (Childers et al. Reference Childers, King and Arnold2014; Dev et al. Reference Dev, Parsons and Palla2011; Kwon et al. Reference Kwon, Tanco and Park2015).
The ESAS is a validated tool used to screen for the presence and intensity of symptoms in patients with advanced cancer. It comprises a numerical rating scale from 0 to 10 with 0 being the absence of symptoms and 10 being the worst symptom intensity imaginable. It assesses symptoms of pain, fatigue, nausea, depression, anxiety, drowsiness, appetite, and sensation of well-being, financial distress, and spiritual pain (Bruera et al. Reference Bruera, Kuehn and Miller1991; Chang et al. Reference Chang, Hwang and Feuerman2000; Philip et al. Reference Philip, Smith and Craft1998).
Statistical analysis
Descriptive statistics such as frequencies and percentages for categorical data and medians and ranges for continuous variables were provided to summarize the baseline demographic and clinical characteristics. CAGE-AID scores between the inpatient consult and outpatient follow-up visits were evaluated by McNemar’s test. Estimate of Kappa coefficient along with its 95% CI was provided to further investigate the agreement between the 2 visit scores. The rate of completion was calculated for individual CAGE-AID items at both visits. Association of demographics and symptom characteristics with CAGE-AID status at follow-up was assessed by Wilcoxon rank sum test for continuous and Chi square/Fisher’s exact test for categorical variables.
Results
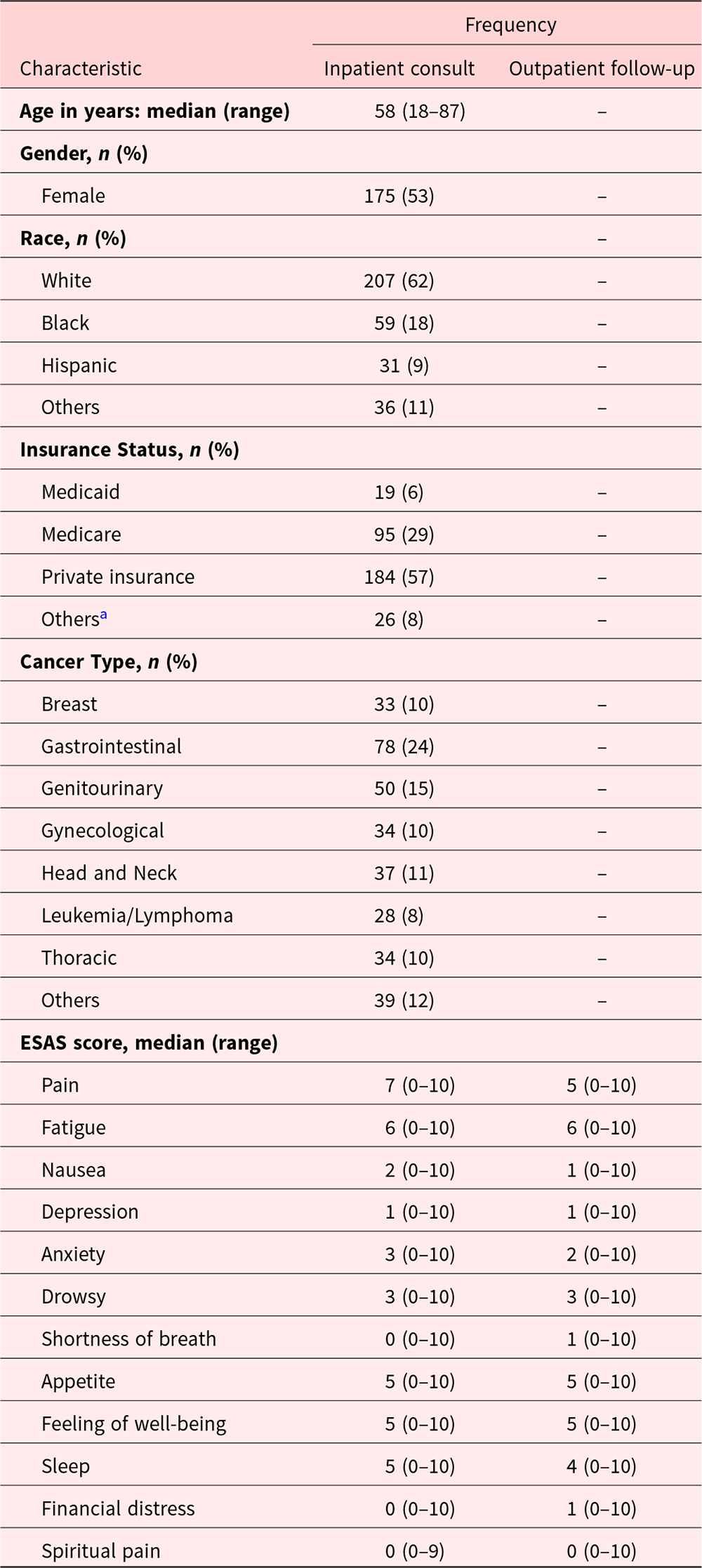
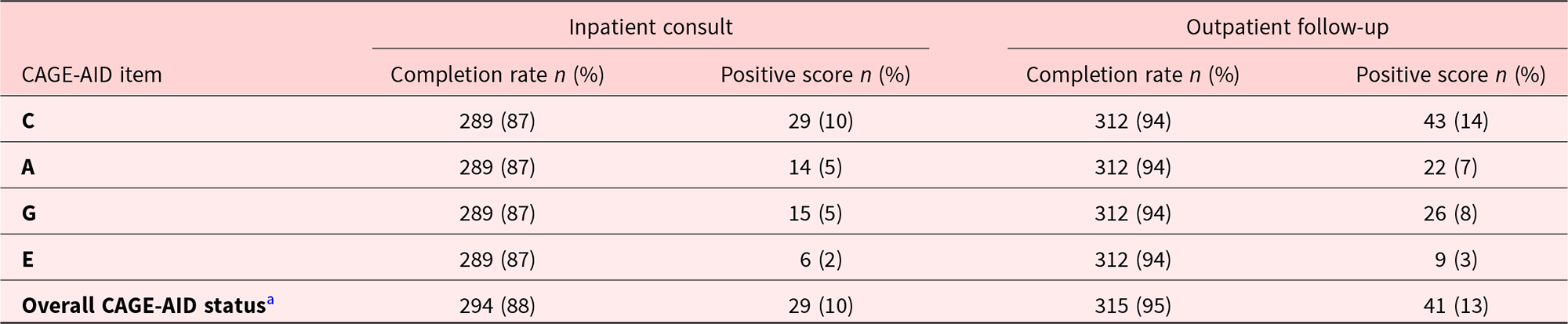
Table 1 provides information on the baseline demographic and clinical characteristics of eligible patients during the first inpatient consult (T1) visit. A total of 333 consecutive patients who also had an outpatient follow-up visit within 3 months were reviewed. Median age was 58 years (range, 18–87 years). Majority of the patients were female (53%), White (62%), and had gastrointestinal cancer (23%). Table 2 shows the completion rates and positive scores of CAGE-AID items at the T1 and first outpatient follow-up (T2) visits. Overall CAGE-AID status was documented for 294 (88%) and 315 (94%) patients at T1 and T2 visits, respectively. Of these, 10% and 13% were CAGE-AID positive, respectively. “Cut down” was the most frequent positive CAGE-AID item both in the inpatient (10%) and outpatient (14%) settings.
Baseline demographic and clinical characteristics of study participants (N = 333)

ESAS = Edmonton Symptom Assessment System, CAGE-AID = Cut down, Annoyed, Guilty, and Eye opener-Adapted to Include Drugs questionnaire, MEDD = Oral Morphine equivalent daily dose in milligrams per day.
a Self-pay, veterans, and other.
Completion rates and positive scores of CAGE-AID items at the inpatient consult and first outpatient follow-up visits (N = 333)

CAGE-AID = Cut down, Annoyed, Guilty, and Eye opener-Adapted to Include Drugs questionnaire.
a A positive CAGE-AID score is ≥2/4
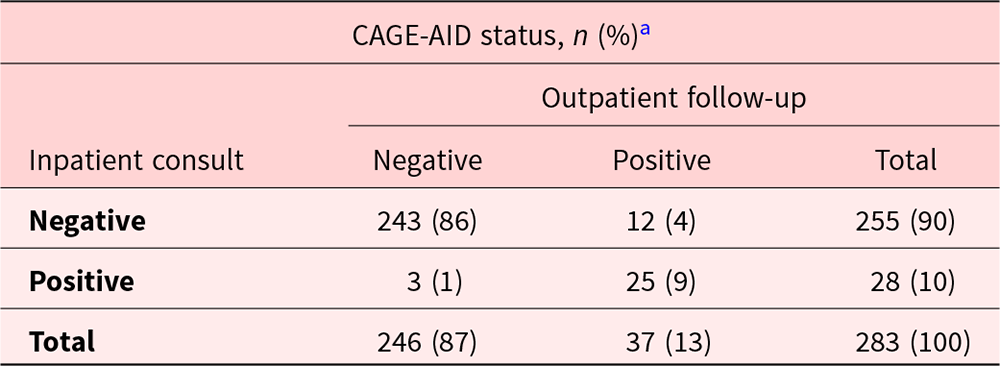
Factors associated with CAGE-AID positivity were also evaluated. Hispanics (23%) were more likely to be CAGE-AID positive as compared with Caucasians (15%) and African Americans (11%), p = 0.02. Table 3 illustrates the level of agreement of completed CAGE-AID scores between the 2 time periods. The Kappa coefficient for agreement was 0.74 (95% CI: 0.62–0.86, p = 0.02). CAGE-AID status changed from negative to positive in 4% and from positive to negative in 1% of patients between the 2 time periods.
Level of agreement of completed CAGE-AID scores between inpatient consult and outpatient follow-up visits (N = 283)

CAGE-AID, Cut down, Annoyed, Guilty, and Eye opener-Adapted to Include Drugs questionnaire.
McNemar’s test p-value = 0.02; Kappa coefficient = 0.74 (95% CI: 0.62–0.86).
a A positive score is ≥2/4 in men or≥1/4 in women.
Discussion
In this pilot study, most patients had CA information, but the questionnaire was not universally completed for all patients seen in the inpatient and the outpatient settings. CA is a vital screening tool that helps identify patients who are at high risk for NMOU (Lau et al. Reference Lau, Mazzotta and Fazelzad2021). It may assist prescribers to arrive at the right diagnosis, develop appropriate treatment plans, and enhance their therapeutic decision-making process for patients receiving opioids. While a positive CAGE-AID score does not necessarily mean a patient is engaging in NMOU behaviors, it heightens provider vigilance and allows them to access further assistance from an interdisciplinary team if needed (Arthur et al. Reference Arthur, Edwards and Reddy2018). Deficiencies in CAGE-AID completion can impair clinicians’ ability to identify at-risk patients and may further affect subsequent opioid therapy. In this era of the opioid crisis, best practices require clinicians to adopt safe and rational opioid prescribing practices. Utilization of validated risk assessment tools such as CA is a key component of such measures. One reason that could potentially decrease its utilization is inadequate time in an already busy clinical setting (Pereira et al. Reference Pereira, Chasen and Molloy2016). Efforts should be made to improve adherence and increase universal utilization of this tool during opioid therapy. These may include ongoing educational sessions, email reminders, documentation checklists, reminder cards, and prompts in electronic health records (Innis and Berta Reference Innis and Berta2016). Since the questionnaire is self-administered by patients themselves, electronic health records can be designed to make the questionnaire accessible for patients to complete before their visit with the clinician.
Of these patients, 10% and 13% were CAGE-AID positive which is an indicator of elevated risk for NMOU. In a similar study by Parsons et al., 17% of patients with advanced cancer seen at an ambulatory SCC were CAGE positive. The patients had more symptom expression and were taking opioids more frequently than patients who were CAGE negative (Parsons et al. Reference Parsons, Delgado-Guay and El Osta2008). The reason why the CAGE-AID positivity rate in this current study was relatively lower than studies that used the CAGE questionnaire (Arthur et al. Reference Arthur, Tang and Lu2021; Bruera et al. Reference Bruera, Moyano and Seifert1995; Dev et al. Reference Dev, Kim and Reddy2019, Reference Dev, Parsons and Palla2011; Hui et al. Reference Hui, Abdelghani and Chen2020; Parsons et al. Reference Parsons, Delgado-Guay and El Osta2008; Yennurajalingam et al. Reference Yennurajalingam, Edwards and Arthur2018) is unclear, but it is possible that the inclusion of “drugs” in the CA version could have deterred some participants from accurately reporting on their substance use. Future research is needed to compare the epidemiological performance of CAGE with CA.
The rate of CAGE-AID completion was lower in the inpatient setting as compared with that in the outpatient setting. The reason for this observed discrepancy is unclear, but this might be reflective of the nature of the inpatient setting where contact of the clinical team with the patient is usually episodic, with a predominant focus on the management of acute issues. Decreased reporting and documentation of the CA is therefore more likely to occur in the inpatient setting as compared with the outpatient setting. The prevalence of NMOU in the inpatient setting is presumably comparable to the outpatient oncologic settings since it is the same cancer patient population who are eventually hospitalized for management of acute issues. Moreover, patients are more likely to display NMOU behaviors in the inpatient setting due to access to parenteral opioids which may increase the peak serum concentration in the brain more rapidly, leading to heightened reward (Arthur et al. Reference Arthur, Reddy and Smith2019). It is therefore critical that inpatient clinical teams utilize risk mitigation strategies to the same degree as in the outpatient settings (Chou Reference Chou2009; Dowell et al. Reference Dowell, Haegerich and Chou2016).
The level of agreement in CAGE-AID scoring was significantly high in the 2 different clinical settings and time periods, but the scoring changed unexpectedly from positive to negative for a minority of patients within the 3-month period. Theoretically, the CA information should remain stable over time since the anchor for timeline is based on alcohol or illicit drug use in their entire life. A prior history of problematic alcohol use as determined by CA therefore carries a lifetime risk for NMOU. The observed discrepancy may be due to errors in patient reporting. Notably, CAGE-AID is based on patient self-report which limits their efficacy (Cook et al. Reference Cook, Bernstein and Arrington1995; Fishbain et al. Reference Fishbain, Cutler and Rosomoff1999). Information from this questionnaire should therefore be combined with other tools such as urine drug testing, pill counts, and prescription monitoring programs when assessing or monitoring for NMOU behaviors. The observed variation could also suggest errors in clinician documentation rather than a real change in the patient’s alcohol behavior. This will require further provider education and awareness to rectify such potential errors. To our knowledge, this is the first study to test the stability and variation in CAGE-AID scoring between 2 clinical settings within the same patient cohort.
The study found that Hispanics were more likely to be CAGE-AID positive compared with other racial groups. Future studies may be necessary to further investigate this finding since NMOU is not known to be relatively higher in the Hispanic population as compared to others. Epidemiological data indicate that Native Americans have the highest prevalence (12.1%) of heavy drinking, followed by Caucasians (8.3%) before Hispanics (6.1%). The prevalence of alcohol use in adults is highest for Whites (59.8%), similar for Native Americans (47.8%), Hispanics (46.3%), and Blacks (43.8%), and lowest for Asian Americans (38.0%) (Vaeth et al. Reference Vaeth, Wang-Schweig and Caetano2017). Risk for NMOU is not well understood among the Hispanic population. One study suggested that regular opioid use might be less common in the Hispanic population compared with other demographic groups (Kelly et al. Reference Kelly, Cook and Kaufman2008). The Hispanic community represents a vital growing segment of the American population with increasing access to health care and opioid analgesics. Hence, a better understanding of their level of risk for NMOU based on the CAGE-AID risk assessment tool will allow clinicians to proactively address any potential opioid-related complications in this population.
One limitation of our study was its retrospective design. Additionally, the study was conducted in a single tertiary cancer center, and the results may not be generally applicable to patients in other clinical settings. Future studies may need to be conducted prospectively at multiple centers to ensure its generalizability and improve its methodological rigor. Another potential limitation is that responses were based on patient self-report. It is possible that patients inaccurately reported on the severity of their alcohol intake. Regrettably, this is an inherent limitation of many NMOU research.
Conclusion
In this study, 10% to 13% of patients with advanced cancer were CAGE-AID positive which is an indicator of elevated NMOU risk. The rate of completion and consistency of the CA was high, but it was not universally completed for all patients seen in both the inpatient and the outpatient settings. Further studies are needed to evaluate ways to ensure more consistency in the completion of the questionnaire and enhance its utilization among patients receiving opioids for cancer pain in routine clinical practice.
Acknowledgments
We would like to thank Ms. Leela Kuriakose for assisting with the acquisition of data for this study.
Author contributions
Tonya Edwards and Joseph Arthur equally contributed to the manuscript.
Competing interests
None.