Introduction
Antimicrobial resistance (AMR) poses a public health challenge with significant attributable global mortality in humans. Reference Murray, Ikuta and Sharara1 AMR is primarily driven by antimicrobial use (AMU); however, the use-resistance relationship is complex, poorly understood, and modified by factors such as transmission of resistant bacteria and antimicrobial effects beyond the target pathogen. Direct effects of AMU on selecting for AMR on recipients themselves are well understood. However, the total impact extends to those indirectly exposed, especially in settings with close contact with recipients such as patients, residents, or staff within healthcare facilities. Reference Hilty, Betsch and Bögli-Stuber2
Direct antimicrobial exposure increases the risk of AMR and collateral risks, such as Clostridioides difficile infection, because broad-spectrum antimicrobials affect not only targeted pathogens but also commensal bacteria (pathogenic and non-pathogenic) that comprise the human microbiome. Reference Sullivan, Edlund and Nord3 This selective pressure disrupts microbial balance while also enriching resistant strains and facilitating horizontal transfer of resistance mechanisms among non-target bystander bacteria. Reference Tedijanto, Olesen, Grad and Lipsitch4 The “bystander effect” has implications at the population-level as transmission of resistance organisms can occur even among individuals not receiving antimicrobials. Reference Lipsitch and Samore5
In the context of AMU, spillover is an indirect risk where people near the recipient of an antimicrobial are affected even if they do not receive the antimicrobial themselves. Reference Olesen, Lipsitch and Grad6 One example where spillover effects have been observed is mass azithromycin drug administration campaigns for infant child morbidity and mortality, where a secondary analysis showed a small spillover effect of resistance determinants among unexposed children in targeted communities. Reference Peterson, Arzika and Amza7
However, in the broader context of routine AMU the unintended population spillover consequences have had limited characterization. Institutional settings such as hospitals, rehabilitation facilities, complex continuing care centers, and nursing homes have high-intensity AMU and prevalence of antimicrobial-resistant organisms, with calls for antimicrobial stewardship initiatives to optimize AMU and reduce AMR and collateral risks. Reference Barlam, Cosgrove and Abbo8,Reference Crayton, Richardson and Fuller9
Understanding spillover is important for quantifying the combined effects of AMU. This knowledge is valuable for communicating the benefits and risks of AMU and informing the interpretation of stewardship interventions. Reference Olesen, Lipsitch and Grad6,Reference Langford, Daneman and Leung10 Given these considerations, we conducted a systematic review to synthesize and characterize the existing evidence regarding the direction, consistency, and magnitude of the association between institutional AMU and the collateral outcomes of AMR and C. difficile among individuals without antimicrobial exposure.
Methods
Search criteria and eligibility
We conducted a literature search in August 2024 using OVID Medline, Healthstar, and EMBASE databases. The search included no language or date restrictions. In consultation with a research librarian we developed four search concepts: spillover (including terms such as “indirect effects” and “secondary effects”), institution (including “long term care,” “NH,” and “hospital”), AMU (including “antibiotic prescribing” and “antibiotic utilization”), and AMR (including “antibiotic resistance” and “C difficile”). These concepts were operationalized using keywords and controlled vocabulary subject headings (see Appendix A). The term “spillover” is used infrequently in AMR research, where similar concepts are more commonly referred to as “indirect effects”. Reference Benjamin-Chung, Abedin and Berger11 To increase sensitivity of the search, we incorporated proximity search strategies which involved using adjacent terms within a specified number of words to capture the relevant search concept. These were informed by a previously published systematic review on spillover effects. Reference Benjamin-Chung, Abedin and Berger11 The search concepts were linked using a boolean “and” operator and refined using test citations. From identified sources, we supplemented our primary search by citation searches using forward and backward approaches of study reference lists.
We included studies looking at antimicrobial spillover effects on adults or children based on human AMU in institutional settings. Studies of selective digestive decontamination (SDD) in intensive care settings were excluded, as our focus was on describing spillover effects based on routine AMU. All studies reporting institutional level AMU were included, with no restriction on utilization metric. We considered all study reported resistant organisms, demonstrated phenotypically or through genotypic markers corresponding with antimicrobial susceptibility phenotypes. For C. difficile, studies reporting stool toxin using toxigenic culture, nucleic acid amplification tests, or enzyme immunoassay assay, were considered.
Both observational and randomized study designs were considered. A spillover effect was defined a priori as an association between institutional AMU and antimicrobial-resistant infection or C. difficile among individuals without direct antimicrobial exposure during a defined non-exposure window, with outcomes reported or extractable at the institutional level. The systematic review protocol was registered on PROSPERO Reference Raybardhan, Langford and Forrest12 and reporting was conducted using the PRISMA guidelines. Reference Page, McKenzie and Bossuyt13
Screening
Duplicate entries were identified using a citation manager (Zotero) and removed prior to importation to a systematic review software. All records were managed using Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia). Double screening of title and abstracts was completed independently by two authors (SR, BJL, RF) for eligibility and inclusion. Identified abstracts underwent full text screening independently by two authors (SR, BJL, RF) for eligibility and inclusion. Any discrepancies were adjudicated through discussion and consensus.
Data extraction
Data extraction was conducted using a standardized electronic form. Single author data (SR) abstraction was conducted with verification by a second author (BJL, RF) for included studies. Relevant information not readily available in the manuscript or supplemental material was requested from the corresponding author(s) or extracted from figures using digitized plotting techniques. Reference Kadic, Vucic, Dosenovic, Sapunar and Puljak14
Study characteristics
Data items extracted included study design (cohort with multilevel modeling, case-control with multilevel modeling, cluster-randomized controlled trial); country; time period of study; study setting; and any study-defined control for variables that might impact AMR/C. difficile transmission.
Antimicrobial measures
Data items extracted included measure of institutional AMU (eg days of therapy), the antimicrobial class evaluated (eg carbapenems), and the study-defined non-exposure window used to classify individuals as unexposed to antimicrobials. The type of antimicrobial exposure comparison was also categorized as either an incremental comparison per standard unit of AMU (eg per 10% relative increase in DOT/1,000 patient-days), categorical comparison per study defined metric (eg tertile-based groupings), or absolute comparisons by cumulative AMU (eg DOT). These distinctions were important for understanding how each study operationalized the exposure and how comparability across studies might be affected.
Outcome measures
The primary outcome was the rate of antimicrobial resistant or C. difficile infection/colonization among non-exposed individuals, quantified per institutional unit population over the study period. When available, overall effects of institutional AMU, irrespective of individual exposure, were also extracted.
Study quality
Study quality was assessed using a previously published modified GRADE approach that incorporates spillover assessment considerations based on underlying study methodology. Reference Benjamin-Chung, Abedin and Berger11 A quality score of 3 was given for cluster-randomized design estimating cluster-level spillover effects among unexposed individuals, observational studies estimating cluster-level spillovers received a score of 2, and purely ecological studies received a score of 1. Validity of spillover effects depends strongly on the chosen analytic approaches, including the ability to adequately control for confounding, address interference (individual’s outcome is affected by exposures of others), and isolate indirect transmission pathways. Reference Benjamin-Chung, Arnold and Berger15,Reference Halloran and Hudgens16 Scores were upgraded by 1 point if studies demonstrated control for confounding at both facility and patient levels, provided evidence of dose-response relationship between institutional AMU and the outcome regardless of the direction of association, or prespecified a spillover mechanism to reduce selective reporting. Reference Benjamin-Chung, Abedin and Berger11 Scores were reduced by 1 point if spillover effects were not explicitly stated in the manuscript, if evidence for spillover was indirect, or limitations in study design that would suggest a likelihood of bias. Because no cluster-randomized trials were identified, maximum observed scores ranged from 2 to 4, with scores of 4 being moderate-quality evidence, whereas scores 2–3 were classified as low-quality evidence.
Synthesis methods
A meta-analysis using a random-effects model was planned for studies reporting standardized continuous antimicrobial exposure metrics and non-aggregate outcomes to enable interpretability and assessment of the direction of effect. A 95% prediction interval was calculated for the range of effects expected if similar studies were to be conducted in different institutional settings. Reference IntHout, Ioannidis, Rovers and Goeman17
Given the small number of eligible studies and the heterogeneity in outcome definitions and exposure metrics, we adopted a hybrid synthesis approach. A narrative synthesis was structured according to the Synthesis Without Meta-analysis reporting guideline. Reference Campbell, McKenzie and Sowden18 For the narrative synthesis, studies were grouped by primary outcome (e.g AMR vs C difficile), study setting, and antimicrobial class, prioritizing study-reported effect measures and using effect direction as a standardized summary metric. Reference Campbell, McKenzie and Sowden18 An effect direction plot enabled visual comparison of the direction and consistency of spillover effects across studies, despite differences in exposure comparison and outcome reporting. Reference Boon and Thomson19
Where a summary measure was not reported, we used digitized plot data extraction techniques to calculate the rate ratio with approximated person-time at risk, equally allocated between exposure groups. Reference Muller20 Estimated number of events were calculated by multiplying incidence rates by group-specific person-time and standard errors were derived accordingly. When the IRR was not reported, it was calculated based on the event rate and assuming equal study follow-up time, Reference Daneman, Bronskill and Gruneir21 or by refitting a log-linear model from digitized ecological data points. Reference Oliveira, Sampaio, Leite, Pereira and Fortaleza22 Where multiple analyses were presented within a study (eg ecological vs cohort), Reference Oliveira, Sampaio, Leite, Pereira and Fortaleza22 we extracted both estimates and prioritized those adjusted for individual or institutional confounding for pooled estimates. The complete extracted data and transformations can be found on an open data repository. Reference Raybardhan23
Results
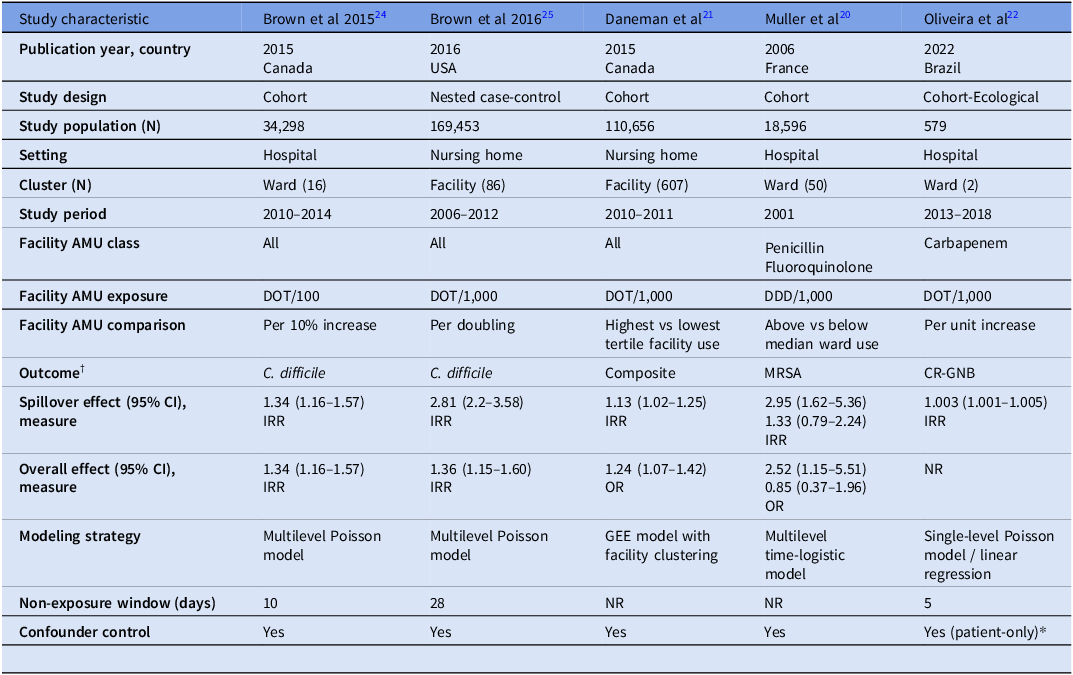
Of the 5,916 studies screened, 47 studies were retrieved for full-text review, with 5 studies meeting inclusion criteria (see Figure 1). All studies were observational, with three studies using a cohort design, one study using a mixed ecological-cohort design, and one study using a nested case-control design (see Table 1). Three of the studies were conducted across 68 hospital wards, measuring antimicrobial exposure at the ward-level, and two were conducted across 693 NH facilities, measuring exposure at the facility-level.

Figure 1. PRISMA flowchart of study selection for the systematic review.
Table 1. Characteristics of included studies assessing spillover effects of antimicrobial use

Note. AMU, antimicrobial use; DOT, days of therapy; DDD, defined daily dose; MRSA, methicillin-resistant Staphylococcus aureus; CR-GNB, carbapenem resistant gram negative bacilli GEE, generalized estimating equation; NR, Not reported.
† Composite outcome = Antimicrobial resistant organisms including methicillin-resistant Staphylococcus aureus, vancomycin-resistant Enterococcus, extended-spectrum β-lactamase producing Enterobacterales; C difficile; and adverse drug event.
* No facility-level confounder control reported.
Four studies measured antimicrobial utilization using days of therapy (DOT) and one study reported the exposure in defined daily doses. Exposure classifications varied between studies, with three studies reporting an incremental comparison (eg per DOT or DDD/ 1,000 patient-days) and two studies reporting a categorical comparison (eg highest vs lowest tertile use).
One study Reference Oliveira, Sampaio, Leite, Pereira and Fortaleza22 presented two measures of antimicrobial utilization: a cohort-level cumulative DOTs during each patient’s time at risk and an ecological measure standardized as DOT per 1,000 resident-days. For study characteristics (see Table 1), we included the ecological measure (DOT per 1,000 resident-days) to facilitate comparability with other studies and used the adjusted cumulative measure for pooled estimates. Three of the studies looked at overall antimicrobial utilization, while two studies reported class-specific antimicrobial utilization. The median non-exposure window reported for three studies was 10 days (IQR 5–28), with four studies reporting facility and patient-level confounder control, and one study reporting patient-level confounder control among unexposed individuals. Study reported outcomes included C difficile, Reference Brown, Valenta, Fisman, Simor and Daneman24,Reference Brown, Jones and Daneman25 methicillin-resistant S. aureus, Reference Muller20 carbapenem-resistant Enterobacterales, Reference Oliveira, Sampaio, Leite, Pereira and Fortaleza22 and aggregate of adverse drug events, AMR, and C difficile. Reference Daneman, Bronskill and Gruneir21
Study quality
Two studies were of moderate quality and three studies were of low quality (see Table 2). Studies with higher quality ratings used multilevel models that accounted for clustering and reported both facility and patient-level confounding control. One study used a mixed ecological-cohort design, because the primary AMU exposure was an aggregated DOT/1,000 patient-day metric, the study was assigned the ecological baseline score (1 point) and was upgraded because a consistent dose-response gradient was demonstrated in both the ecological time-series and the patient-level cohort studies.
Table 2. Quality assessment of included studies evaluating spillover effects

Risk of bias was driven by limited reporting of confounder adjustment for unexposed individuals and the non-exposure window. Precision was influenced by the rarity of the outcome and the varying facility-level exposure comparisons. Consistency of effect was supported as all studies reported a positive association between facility-level exposure and the outcome among unexposed individuals. Of the four studies that also reported the overall effect of facility-level exposure (irrespective of individual-level exposure) the magnitude of the spillover estimate was similar or slightly attenuated compared to the overall effect. With only five studies identified, a formal meta-bias assessment was not completed. 26
Evidence synthesis
Five studies met inclusion criteria and were included in the narrative synthesis. The five studies reported IRRs greater than 1.0 (range 1.003 to 2.95), demonstrating a consistent positive direction of association between higher institutional AMU and increased AMR or C difficile among unexposed individuals (see Figure 2), with the strength of the association driven primarily by moderate-quality observational evidence (see Table 2). For C. difficile, in nursing-home settings a doubling in DOT/1,000 resident-days corresponded to an IRR 2.81 (95% CI 2.20–3.58), whereas in hospital settings a 10% relative increase in DOT/100 patient-days yielded an IRR 1.34 (95% CI 1.16–1.57). Among AMR studies, effect size varied by drug–pathogen pair: penicillin use and MRSA yielded the largest increase (IRR 2.95 [1.62–5.36]), fluoroquinolone use and MRSA showed a smaller, statistically non-significant rise (IRR 1.33 [0.79–2.24], and carbapenem use and carbapenem-resistant Enterobacterales exhibited a modest elevation (IRR 1.003 [1.001–1.005]). Despite heterogeneity in exposure metrics, all estimates favored a positive direction of association with higher facility use and AMR or C. difficile among the unexposed.

Figure 2. Effect-direction plot of spillover effects of institutional AMU among unexposed individuals. AMR, antimicrobial resistance; CDI, Clostridioides difficile; IRR, incidence rate ratio.
Meta-analysis
Three studies reported continuous measures of institutional AMU and non-aggregate outcomes. We standardized the outcome to a common unit, specifically the estimated incidence rate ratio per 100 DOT increase in antimicrobial utilization per 1,000 patient-days. This increment was selected for interpretability and to minimize transformations required for standardization across studies (see Appendix B).
The meta-analysis, utilizing a random effects model, revealed a pooled incidence rate ratio of 1.54 (95% CI: 0.85–2.80, p-value .15), which did not reach statistical significance (see Figure 3). The corresponding prediction interval ranged from 0.12 to 20.51, indicating a true effect in similar settings could plausibly range from an 88% reduction to a twenty-fold increase. Significant heterogeneity was observed across the included studies, with an I 2 of 97.6% (p value < .001).

Figure 3. Forest plot of meta-analysis of spillover effects per 100 DOT increase in institutional AMU (per 1,000 patient-days). DOT, days of therapy.
Discussion
To our knowledge this is the first systematic review to examine the effects of institutional AMU among unexposed individuals. Despite a comprehensive search strategy including adjacent terms for spillover and indirect effects, we identified only five studies. Inconsistent terminology, evolving definitions, and inadequate indexing of spillover concepts within routine AMU literature impeded identification. Citation searches of identified studies did not yield any additional studies and relevant studies may have been missed. While a meta-bias assessment could not be undertaken, accurate spillover estimation depends on prespecifying hypothesized mechanisms, yet only two studies explicitly did so. This lack of prespecification can lead to missed spillover measurement or under-reporting of spillover findings. Beyond publication bias, spillover estimates may be affected by selection bias within studies due to exposure misclassification or differential outcome detection correlated with institutional AMU, potentially attenuating or inflating associations. Reference Raybardhan, Langford and Forrest12,Reference Benjamin-Chung, Arnold and Berger15 Although several studies adjusted for individual- and institutional-level confounders, such adjustment may not fully address selection processes, contributing to variability in spillover estimates across studies. Recently, a checklist for reporting of spillover effects in health interventions has been advocated Reference Benjamin-Chung, Abedin and Berger11 ; however, this tool postdated all included studies and none reported use of an equivalent structured reporting framework. Adoption of standardized reporting tools can enhance methodological rigor and comparability of spillover effects with routine AMU.
Our review suggests that higher facility-level AMU is associated with increased AMR or C difficile among individuals not recently exposed to antimicrobials. A subset meta-analysis of three studies reporting continuous institutional AMU supported a positive direction of effect ( IRR 1.54). However, the wide 95% prediction interval (0.12–20.51) reflects significant heterogeneity (I 2 = 97.6 %) and a small number of studies; therefore findings should be interpreted as hypothesis-generating rather than definitive evidence of spillover effects.
Accurate spillover effects estimation is crucial for understanding how institutional AMU impacts unexposed individuals, yet methodological challenges persist. Observational studies employing cluster-level designs can address these challenges by accounting for facility- and patient-level confounding through multilevel analytic strategies or cluster-level propensity scores. Reference Benjamin-Chung, Arnold and Berger15,Reference Halloran and Hudgens16,Reference Halloran and Struchiner27–Reference Tchetgen and VanderWeele29 These methods enhance validity by explicitly considering how cluster-level antimicrobial exposure (eg wards, facilities) influences outcomes among unexposed individuals. While four of five studies in this review used multilevel techniques, one lacked explicit reporting of confounder control for spillover. Future studies should explicitly report adjustment strategies, including exploring propensity-score methods to handle interference, where one individual’s exposure impacts another individual’s outcome.
Population-level spillover effects of AMU have been reported in mass-drug-administration trials, household studies, and community settings. However, comparable evidence for routine institutional use is limited. The ecological impact of SDD in institutional intensive care unit settings has been assessed in a prospective cluster randomized trial, however did not specifically look at unexposed individuals and did not have enough power to detect changes in AMR given the low prevalence AMR in the study settings. Reference Boschert and Broadfield30,Reference Davis and Cheng31 We did not include SDD studies in our systematic review as these interventions did not constitute routine AMU in institutional settings.
Peterson et al reported a 3.4 % (95 % CI –4.1 to 10.8) increase in macrolide-resistance genes among unexposed children in a secondary analysis of the cluster-randomized trial of azithromycin mass administration across 30 communities in Niger. Reference Peterson, Arzika and Amza7 At the household level, Gottesman et al. found maternal fluoroquinolone use was associated with a 50 % increase in odds of resistant gram-negative bacteriuria among unexposed children. Reference Gottesman, Low, Almog and Chowers32 In a population-based study, Low et al. showed women living in neighborhoods with the highest quintile of fluoroquinolone use had 1.47-fold (95 % CI 1.33–1.65) higher odds of resistant E. coli urinary isolates despite no personal antibiotic exposure. Reference Low, Neuberger and Hooton33 Together, these data confirm that antimicrobial pressure can extend beyond treated individuals; however, none of these studies quantify indirect effects within healthcare institutions–a setting characterized by concentrated antibiotic use and high transmission potential.
Several important limitations affect the interpretation of our systematic review, including considerable heterogeneity in study design, exposure definitions, and confounder adjustment, as well as challenges in identifying relevant studies and the potential for publication bias limit the certainty of these findings. These issues likely obscure mechanistic reasons for differences seen in the magnitude of the spillover effect between antimicrobial pathogen pairs that could be driven by differential impact of bystander selection, Reference Tedijanto, Olesen, Grad and Lipsitch4 cross-class AMR, Reference Gbaguidi-Haore, Dumartin and L’Hériteau34,Reference Cherny, Nevo and Baraz35 and environmental persistence. Reference Kelly, Bekele and Loughrey36,Reference Freedberg, Salmasian, Cohen, Abrams and Larson37 Improved standardization of terminology, explicit prespecification of spillover mechanisms, and adoption of robust observational designs can advance research of spillover effects with routine AMU.
Acknowledgements
We would like to thank Kaitlyn Merriman at Gerstein Science Information Centre at the University of Toronto Libraries for her valuable assistance.
Financial support
None reported.
Competing interests
All authors report no conflicts of interest relevant to this article.
Appendix A. Search strategy
(spillover* adj5 effect*) or (spill over adj5 effect*) or (externalit* adj5 effect*) or (contamination adj5 effect*) or (indirect adj5 effect*) or (indirect adj5 $receipt*) or (indirect adj5 risk*) or (herd adj5 effect*) or (seconda* adj5 effect*) or (unexpected adj5 effect*) or (diffusion adj5 effect*) or (ecolog* adj5 effect*) or (independent adj5 effect*) or (independent adj5 risk*) or (independent adj5 association*) or (without adj5 exposure*) or nonreceipt or (prior adj3 use*) or (prior adj3 antibiotic*) or (previous adj3 use*) or (previous adj3 antibiotic*) or multilevel* or multi-level* or cluster*).tw,kf.
Beds/ or Hospitalization/ or Intensive Care Units/ or Hospitals/
Inpatients/ or Homes for the Aged/ or Nursing Homes/ or Residential Facilities/
Skilled Nursing Facilities/
exp Health Facilities/
(bed* or hospitali?ation or intensive care unit or ICU or hospital* or inpatient*).tw,kf.
(homes for the aged or senior home* or old folks home* or retirement home or nursing home* or residential facilities or complex continuing care or rehabilitation facil*).tw,kf.
or/2-7 [Institutional]
Anti-Bacterial Agents/ or Practice Patterns, Physicians’/ or Drug Prescriptions/ or exp Beta-lactamase inhibitors/ or Quinolones/ or exp Fluoroquinolones/ or exp beta-Lactams/ or azithromycin/ or clarithromycin/ or erythromycin/ or trimethoprim/ or exp trimethoprim, sulfamethoxazole drug combination/ or nitrofurantoin/ or anti-infective agents, urinary/
(antibiotic or antibiotic utili?ation or antibiotic usage or antibiotic prescrib* or antibacterial* or anti-bacterial* or quinolone* or fluoroquinolone* or beta-lactam* or azithromycin or clarithromycin or erythromycin or trimethoprim or sulfamethoxazole or trimethoprim-sulfamethoxazole or urinary tract anti-infective*).tw,kf.
or/9-10 [antibiotic usage ]
Cross Infection/ or Community-Acquired Infections/ or Clostridioides difficile/ or Clostridium Infections/ or exp Drug Resistance, bacterial/ or exp beta-lactam resistance/ or vancomycin-resistant enterococci/ or carbapenem-resistant enterobacteriaceae/
(cross infection* or community acquired infection or drug resistan* or C difficile or Clostridioides difficile or colonization or bacterial resistance* or antibacterial resistance* or Extended Spectrum Beta-Lactamase* or ESBL).tw,kf.
or/12-13 [Antibacterial resistance]
1 and 8 and 11 and 14
Appendix B. Outcome standardization
For studies reporting a one DOT increase in exposure (Oliviera 2022) we assumed a log-linear model. The beta coefficient was back-calculated from the reported IRR using the study reported median duration of exposure and scaled for the corresponding 100 DOT/1000 patient-days. Brown (2016) originally reported IRR per doubling, which corresponds approximately to a 100 DOT increase. Thus, the reported IRR (2.81; 95% CI, 2.20–3.58) was directly used without transformation to reflect an increase per 100 DOT per 1000 patient-days. For studies reporting an IRR for a 10% relative increase DOT/100 patient-days (Brown 2015) we used the study-reported IRR, as the proportional change is equivalent to 100 DOT/1000 patient-days.
Oliveira et al
-
1. Log-linear Poisson Model equation:
 $${\rm{ln(E[Yi])}} = {\beta _0} + \;{\beta _{1\;}}{X_{i1}}$$
$${\rm{ln(E[Yi])}} = {\beta _0} + \;{\beta _{1\;}}{X_{i1}}$$
Solving for
 $$\;{\beta _1}$$
(per DOT exposure)
$$\;{\beta _1}$$
(per DOT exposure)1.009 =
 ${e^{{\beta _1}}}$
${e^{{\beta _1}}}$
 $${\beta _1} = 0.00896$$
$${\beta _1} = 0.00896$$
-
2. Convert to DOT/Patient-day based on study reported median time at risk (5 days)
 $$\beta_{1}^{\prime}$$
= 0.00896 x 5 = 0.0448
$$\beta_{1}^{\prime}$$
= 0.00896 x 5 = 0.0448 -
3. Calculate standardized IRR for 100 DOT per 1000 patient-days
100 DOT per 1000 patient-days = 0.1 DOT per patient-day
IRR =
 ${e^{0.0448*0.1}}$
= 1.004 (95% CI 1.000, 1.009)
${e^{0.0448*0.1}}$
= 1.004 (95% CI 1.000, 1.009)