Refine listing
Actions for selected content:
Volume 73 - Issue 1 - February 2009
Contents
Research Article
The central Kenya peralkaline province: a unique assemblage of magmatic systems
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1-16
-
- Article
- Export citation
Preferred ion diffusion pathways and activation energies for Ag in the crystal structure of stephanite, Ag5SbS4
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 17-26
-
- Article
- Export citation
The mineralogy of efflorescence on As calciner buildings in SW England
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 27-42
-
- Article
- Export citation
Mavlyanovite, Mn5Si3: a new mineral species from a lamproite diatreme, Chatkal Ridge, Uzbekistan
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 43-50
-
- Article
- Export citation
Eldfellite, NaFe(SO4)2, a new fumarolic mineral from Eldfell volcano, Iceland
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 51-57
-
- Article
- Export citation
The role of fractional crystallization and late-stage peralkaline melt segregation in the mineralogical evolution of Cenozoic nephelinites/phonolites from Saghro (SE Morocco)
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 59-82
-
- Article
- Export citation
Aschamalmite (Pb6Bi2S9): crystal structure and ordering scheme for Pb and Bi atoms)
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 83-94
-
- Article
- Export citation
The thermal equation of state of (Fe0.86Mg0.07Mn0.07)3Al2Si3O12 almandine
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 95-102
-
- Article
- Export citation
The crystal structure and chemistry of mereheadite
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 103-117
-
- Article
- Export citation
Cation ordering and phase transitions in feldspars along the join CaAl2Si2O8-SrAl2Si2O8: a TEM, IR and XRD investigation
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 119-130
-
- Article
- Export citation
Description and crystal structure of a new mineral – plimerite, ZnFe3+ 4(PO4)3(OH)5 – the Zn-analogue of rockbridgeite and frondelite, from Broken Hill, New South Wales, Australia
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 131-148
-
- Article
- Export citation
Chevkinite-group minerals from granulite-facies metamorphic rocks and associated pegmatites of East Antarctica and South India
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 149-164
-
- Article
- Export citation
Correction
Erratum
-
- Published online by Cambridge University Press:
- 05 July 2018, p. 165
-
- Article
- Export citation
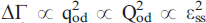 . The evolution of the order parameter with time can be modelled as
. The evolution of the order parameter with time can be modelled as 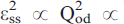 lnt, as predicted in order-disorder processes at intermediate degrees of order. It is shown that even after the longest annealing runs in An
lnt, as predicted in order-disorder processes at intermediate degrees of order. It is shown that even after the longest annealing runs in An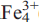 (PO
(PO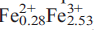 Al
Al