Introduction
Attention-deficit/hyperactivity disorder (ADHD) affects between 2.5% and 6.8% of adults globally, with an estimated average prevalence of 3.3% in high-income countries (Fayyad et al., Reference Fayyad, Sampson, Hwang, Adamowski, Aguilar-Gaxiola and Al-Hamzawi2017; Simon et al., Reference Simon, Czobor, Bálint, Mészáros and Bitter2009; Song et al., Reference Song, Zha, Yang, Zhang, Li and Rudan2021). In Germany, however, estimates based on retrospective claims data from statutory health insurance are substantially lower, at just 0.7%, suggesting marked and longstanding underrecognition of adult ADHD (Libutzki et al., Reference Libutzki, Ludwig, May, Jacobsen, Reif and Hartman2019; Philipsen & Döpfner, Reference Philipsen and Döpfner2020; Schlander et al., Reference Schlander, Schwarz, Trott, Viapiano and Bonauer2007). This is particularly concerning given the high burden of the disorder: Adults with ADHD often face substantial difficulties affecting multiple domains of adult life, including academic and occupational functioning, management of everyday responsibilities such as finances, and interpersonal relationships (de Zwaan et al., Reference de Zwaan, Gruß, Müller, Graap, Martin, Glaesmer and Philipsen2012; Fuermaier et al., Reference Fuermaier, Tucha, Butzbach, Weisbrod, Aschenbrenner and Tucha2021; Koerts et al., Reference Koerts, Bangma, Fuermaier, Mette, Tucha and Tucha2021). These difficulties are often accompanied by a reduced quality of life extending well into older adulthood (Thorell, Holst, & Sjöwall, Reference Thorell, Holst and Sjöwall2019), as well as psychological comorbidities, such as depression and anxiety, further exacerbating personal and societal burden (Faraone et al., Reference Faraone, Bellgrove, Brikell, Cortese, Hartman, Hollis and Buitelaar2024; Fayyad et al., Reference Fayyad, Sampson, Hwang, Adamowski, Aguilar-Gaxiola and Al-Hamzawi2017; Libutzki et al., Reference Libutzki, Ludwig, May, Jacobsen, Reif and Hartman2019).
Current European clinical guidelines emphasize a multimodal approach to treating adult ADHD, combining pharmacological and psychosocial interventions to address both core symptoms as well as associated functional and psychosocial impairments (AWMF, 2017; Kooij et al., Reference Kooij, Bijlenga, Salerno, Jaeschke, Bitter and Balazs2019; NICE, 2018). The German evidence- and consensus-based guideline (AWMF, 2017) recommends psychosocial interventions, particularly in cases of insufficient response to pharmacotherapy, contraindications to medication, poor tolerability, or patient’s decision against pharmacological treatment. Additional indications include adults later in life needing support with diagnosis acceptance and treatment initiation, as well as cases of mild symptomatology where psychosocial treatment is considered sufficient to address low to moderate functional impairments (AWMF, 2017).
Among available psychosocial interventions, psychoeducation and cognitive behavioral therapy (CBT) have demonstrated effectiveness in reducing ADHD symptom severity and associated disorder burden (Hoxhaj et al., Reference Hoxhaj, Sadohara, Borel, D’Amelio, Sobanski, Müller and Philipsen2018; Knouse, Teller, & Brooks, Reference Knouse, Teller and Brooks2017; Liu, Hua, Lu, & Goh, Reference Liu, Hua, Lu and Goh2023; Nimmo-Smith et al., Reference Nimmo-Smith, Merwood, Hank, Brandling, Greenwood, Skinner and Rai2020; Philipsen et al., Reference Philipsen, Jans, Graf, Matthies, Borel and Colla2015; Young, Moghaddam, & Tickle, Reference Young, Moghaddam and Tickle2020). Accordingly, clinical guidelines endorse both approaches, with psychoeducation recommended as the basis of standard care (AWMF, 2017; Kooij et al., Reference Kooij, Bijlenga, Salerno, Jaeschke, Bitter and Balazs2019). However, access to qualified care in Germany remains limited: clinicians with expertise in adult ADHD are scarce (Schlander et al., Reference Schlander, Schwarz, Trott, Viapiano and Bonauer2007; Schneider et al., Reference Schneider, Schöttle, Hottenrott, Gallinat and Moritz2023), and waiting times for psychotherapy exceed several months (Kruse et al., Reference Kruse, Kampling, Bouami, Grobe, Hartmann, Jedamzik and Friederich2024). As a result, ~40% of adults with ADHD receive no psychotherapy within the first year after diagnosis, and among those who do, 30% receive only probatory sessions (Libutzki et al., Reference Libutzki, Ludwig, May, Jacobsen, Reif and Hartman2019).
Digital interventions have demonstrated robust effectiveness across various psychiatric conditions (Andersson, Reference Andersson2016; Karyotaki et al., Reference Karyotaki, Efthimiou, Miguel, F. M., Furukawa, Cuijpers and Gemmil2021; Pauley et al., Reference Pauley, Cuijpers, Papola, Miguel and Karyotaki2023) and are therefore considered a promising strategy to help bridge the treatment gap in adult ADHD. To date, five small-scale randomized controlled trials (RCTs) have evaluated the effectiveness of digital interventions in reducing core ADHD symptom severity, reporting effect sizes between d = 0.42 and 1.21 (Kenter, Gjestad, Lundervold, & Nordgreen, Reference Kenter, Gjestad, Lundervold and Nordgreen2023; Moëll et al., Reference Moëll, Kollberg, Nasri, Lindefors and Kaldo2015; Nasri et al., Reference Nasri, Cassel, Enhärje, Larsson, Hirvikoski, Ginsberg and Kaldo2023; Pettersson, Söderström, Edlund-Söderström, & Nilsson, Reference Pettersson, Söderström, Edlund-Söderström and Nilsson2017; Selaskowski et al., Reference Selaskowski, Steffens, Schulze, Lingen, Aslan, Rosen and Boll2022). Despite growing evidence for their effectiveness, digital interventions have not yet been integrated into the routine care of adult ADHD in Germany, and no program is currently approved for reimbursement by statutory health insurance under the Digital Health Application scheme.
Against this background, we developed attexis, a fully self-guided digital intervention grounded in CBT and mindfulness principles. Thus, attexis requires no professional oversight and is accessible online at the user’s convenience. The program provides personalized psychoeducation and therapeutic exercises tailored to individual needs and preferences. Features such as adaptive content delivery, automated reminders, and progress tracking are integrated to support user engagement and motivation.
The goal of the present pragmatic RCT was to evaluate the effectiveness of attexis when used as an adjunct to existing treatment pathways. To this end, we compared the addition of attexis to treatment as usual (TAU) to a TAU-only control group. TAU reflects the heterogeneous and unstandardized care typically received by adults with ADHD in routine practice in Germany, including pharmacological and nonpharmacological interventions, as well as no formal treatment. The primary endpoint was the severity of ADHD symptoms after 3 months. In addition, we assessed secondary endpoints reflecting functional and psychosocial outcomes relevant to patients, including functional impairment, depressive symptoms, self-esteem, and quality of life. We hypothesized that participants receiving attexis in addition to TAU would show greater improvements across these outcomes compared to those receiving TAU alone. Additional follow-up data were collected at 6 months to evaluate the durability of treatment effects.
Methods
Procedures
The study “Researching the Effectiveness of attexis, a Digital Health Application for Adults with Attention Deficit Hyperactivity Disorder – a Randomized Controlled Trial (READ-ADHD)” was approved by the Ethics Committee of the Medical Chamber Hamburg (reference number 2023–101052-BO-ff), and all procedures complied with the Declaration of Helsinki and General Data Protection Regulation.
Before recruitment, the study was registered in an international clinical trials registry (ClinicalTrials.gov ID: NCT06221930). German-speaking participants were recruited from March 2024 to June 2024 via an online advertising campaign using targeted Google Ads to reach adults with ADHD symptoms. Interested individuals were directed to a dedicated study website containing detailed study information and an option to register their interest.
After providing electronic informed consent, participants completed an initial online screening survey. Those who screened positively were invited to a structured diagnostic interview conducted by telephone. Trained psychological staff administered the German version of the DIVA-5 (Kooij, Francken, Bron, & Wynchank, Reference Kooij, Francken, Bron and Wynchank2019) to confirm an ADHD diagnosis and the Mini-DIPS (Margraf, Cwik, Pflug, & Schneider, Reference Margraf, Cwik, Pflug and Schneider2017) to assess relevant current psychiatric comorbidities. Ambiguous cases were reviewed in regular supervision meetings with GAJ. Final eligibility was determined by the study physician, SMJ, following a comprehensive review of inclusion and exclusion criteria.
Eligible participants were randomized in a 1:1 ratio to either the intervention group (attexis + TAU) or the control group (TAU only), using an automated and concealed allocation system using computer-generated random numbers (simulating a digital coin toss) to perform simple randomization. Allocation was triggered automatically by the study platform upon completion of eligibility verification. Due to the nature of the pragmatic design, participants were not blinded to group allocation. As all outcomes were collected via self-report, no outcome assessors were involved, and blinding was therefore not applicable. Participants in the intervention group received immediate access to attexis, while control group participants were offered access upon study completion. The study was designed as a pragmatic trial, aiming to approximate real-world conditions without restricting concurrent treatment options: TAU permitted participants in both groups to continue, modify, or discontinue any concurrent treatments, including psychotherapy and pharmacotherapy. Planned treatment changes in the upcoming 3 months were an exclusion criterion at baseline, in order to better attribute observed effects to attexis. Unplanned treatment changes that occurred during the course of the study were allowed and documented, reflecting the pragmatic nature of the trial.
Participants completed outcome assessments online at 3 months (T1, primary time point for evaluating effectiveness) and 6 months (T2, follow-up to assess the durability of effects over time). All study procedures were conducted fully remotely; questionnaires were administered online, and diagnostic interviews were conducted by telephone. Participants received a €10 gift voucher for every completed follow-up assessment.
The required sample size was determined a priori based on the primary outcome; see Supplementary Method 1 for details.
Inclusion and exclusion criteria
Participants of all sexes and genders were eligible for inclusion if they were between 18 and 65 years of age and had a diagnosis of ADHD confirmed via the structured diagnostic interview DIVA-5. To meet the symptom severity threshold, participants were required to score ≥ 17 on either the inattention or the hyperactivity/impulsivity subscale of the Adult ADHD Self-Report Scale v1.1 (ASRS), in line with thresholds used in prior research on digital interventions for ADHD (Kenter, Gjestad, Lundervold, & Nordgreen, Reference Kenter, Gjestad, Lundervold and Nordgreen2023; Moëll et al., Reference Moëll, Kollberg, Nasri, Lindefors and Kaldo2015). Additional inclusion criteria were stable treatment status (including psychotherapy, medication, or no treatment) for at least 30 days before enrollment, sufficient knowledge of the German language, and provision of informed consent.
Participants were excluded if they had a lifetime diagnosis of a severe psychiatric disorder, including severe affective disorder, psychotic disorder, autism spectrum disorder, borderline or antisocial personality disorder, substance use disorder, or suicidality. Individuals were also excluded if they planned to change their current treatment regimen (e.g., psychotherapy or medication) within the 3 months following enrollment.
Intervention
attexis is a self-guided online intervention based on cognitive-behavioral and mindfulness principles, developed to support adults with ADHD. attexis combines psychoeducation, therapeutic exercises, and personalized techniques tailored to the user’s reported needs and preferences (see Table 1). A central feature is its use of “simulated dialogues,” in which users interact with brief text passages by selecting response options that guide the flow of information and tailor the experience (see Supplementary Figure for example screenshots). In this context, attexis specifically addresses the interpersonal needs of patients with ADHD. For instance, depending on the individual’s attentional state, it may offer to repeat certain topics or suggest humorous or unexpected exercises to help sustain attention. This format supports personalization and sustained engagement. Thus, the interaction itself is highly flexible and individualized. However, the main topics covered by the program are set in a fixed order. The program begins with psychoeducation, for example, about the diagnosis and the concept of neurodiversity, followed by techniques to improve attention. Only then are topics such as self-image addressed. This is consistent with the approach used in manualized psychotherapies. The program also includes simple homework tasks, flexible pacing, and automated reminders to encourage continued use. In addition to its core dialogues, attexis provides Supplementary Materials such as guided audio exercises, PDF worksheets, and summary sheets. Users may also opt to receive motivational messages via SMS or email, and self-monitoring tools are included to support behavioral tracking and progress monitoring over time.
Content of attexis

Measures
Baseline data, as well as follow-up data at 3 months (T1) and 6 months (T2) post-randomization, were collected via a secure and encrypted online survey platform (LimeSurvey). All measures were based on self-report. Participants were invited to complete each assessment via email, and nonresponders received up to three reminders. For follow-up assessments, additional contact methods for reminders, including SMS and telephone calls, were employed to maximize response rates.
Primary endpoint
The primary endpoint was the ASRS total score, a widely used 18-item patient-reported outcome measure assessing core ADHD symptoms (Kessler et al., Reference Kessler, Adler, Ames, Demler, Faraone, Hiripi and Walters2005), evaluated at T1 (3 months post-randomization). The validated German version of the ASRS has demonstrated good psychometric properties in adults with ADHD (Mörstedt, Corbisiero, & Stieglitz, Reference Mörstedt, Corbisiero and Stieglitz2016).
Secondary endpoints
Secondary outcomes included: (1) functional impairment, assessed using the Work and Social Adjustment Scale (Heissel et al., Reference Heissel, Bollmann, Kangas, Abdulla, Rapp and Sanchez2021; Lundqvist et al., Reference Lundqvist, Lindberg, Brattmyr, Havnen, Hjemdal and Solem2024; Mundt, Marks, Shear, & Greist, Reference Mundt, Marks, Shear and Greist2002); (2) depressive symptoms, measured by the Patient Health Questionnaire-9 (Hennig et al., Reference Hennig, Koglin, Schmidt, Petermann and Brähler2017; Kliem et al., Reference Kliem, Sachser, Lohmann, Baier, Braehler, Gündel and Fegert2024; Martin, Rief, Klaiberg, & Braehler, Reference Martin, Rief, Klaiberg and Braehler2006); (3) self-esteem, assessed with the Rosenberg Self-Esteem Scale (Masuch et al., Reference Masuch, Bea, Alm, Deibler and Sobanski2019; Rosenberg, Reference Rosenberg1965; Roth, Decker, Herzberg, & Brähler, Reference Roth, Decker, Herzberg and Brähler2008; von Collani & Herzberg, Reference von Collani and Herzberg2003); and (4) quality of life, measured by the Assessment of Quality of Life - 8 Dimensions (Richardson, Iezzi, Khan, & Maxwell, Reference Richardson, Iezzi, Khan and Maxwell2014; Richardson, Khan, Iezzi, & Maxwell, Reference Richardson, Khan, Iezzi and Maxwell2012).
Comorbidities and user satisfaction
Relevant sections from the Mini-DIPS were used to assess current psychiatric comorbidities in the sample (Margraf, Cwik, Pflug, & Schneider, Reference Margraf, Cwik, Pflug and Schneider2017). To assess satisfaction with attexis, participants were asked to rate how likely they were to recommend attexis to a friend or colleague on a numeric rating scale from 0 (not likely at all) to 10 (extremely likely). Moreover, subjective improvement of ADHD symptoms as well as subjective improvement in the impact of ADHD on daily activities in the last 3 months (from T0 to T1 and from T1 to T2) was evaluated with the Patient Global Impression of Change scale (PGIC; Guy, Reference Guy1976).
Adverse events
Given that attexis is a Conformité Européenne (CE)-marked Class I medical device, it is inherently associated with a very low risk profile. Nonetheless, adverse events were monitored throughout the study and operationalized as any unplanned or emergency outpatient or inpatient treatment reported for the preceding 3 months. Participants were asked to report such events at each follow-up assessment (T1 and T2). In addition, any events potentially related to the use of the intervention were documented as adverse device effects. All reported events were reviewed for potential causal association with attexis.
Statistical analyses
All analyses followed a prespecified analysis plan and were conducted using R, version 4.4.1 (R Core Team, 2024). The primary analyses applied the intent-to-treat (ITT) principle under a missing at random assumption, using analysis of covariance adjusted for baseline values. Results are reported as baseline-adjusted mean differences with 95% confidence intervals (CIs) and standardized effect sizes (Cohen’s d based on estimated marginal means). Missing data were handled via bootstrapped multiple imputation (von Hippel & Bartlett, Reference von Hippel and Bartlett2021). As a sensitivity analysis, we applied jump-to-reference (J2R) imputation, a conservative method assuming intervention dropouts follow the control trajectory (Bartlett, Reference Bartlett2023). Per-protocol (PP) analyses, defined as including only intervention participants who had registered to use attexis, alongside all participants in the control group, applied the same statistical procedures as the ITT analyses. All analyses were repeated at T2 to assess the durability of effects. Clinical relevance was evaluated using responder analyses. For the primary endpoint, responders were defined as participants who showed a ≥30% reduction in the ASRS total score from baseline to T1 (Buitelaar, Montgomery, & van Zwieten-Boot, Reference Buitelaar, Montgomery and van Zwieten-Boot2003). For secondary endpoints, responder criteria were based on predefined minimal clinically important differences (MCIDs), or on the reliable change index (RCI) where no MCID was available. Group differences in responder rates were analyzed using χ 2 tests and odds ratios (ORs). As a safety analysis, we assessed symptom worsening, defined as any increase in the ASRS total score from baseline to T1. Subgroup analyses assessed potential treatment moderators (sex, psychotherapy, psychotropic medication, and treatment changes). Statistical tests were considered significant at p < .05 (two-sided). A gatekeeping testing strategy was applied to control for multiplicity. Full details of the statistical analysis are provided in Supplementary Method 2.
Results
Recruitment and retention overview
A total of 2,058 individuals were screened for eligibility, of whom 337 met all inclusion criteria and were randomized to the intervention group (n = 164) or the control group (n = 173) (see Figure 1). The slight imbalance in group sizes is consistent with expected variation under simple randomization. Attrition rates at 3 months were 9.1% in the intervention group and 3.5% in the control group; at 6 months, attrition increased slightly to 13.4% and 8.1%, respectively. Following the predefined criteria for inclusion in the PP analyses, 163 out of 164 participants (99.4%) in the intervention group had activated the voucher to use attexis. The resulting PP dataset comprised 336 participants: 163 in the intervention group and all 173 in the control group.
Flow of participants through the study. Note: ASRS, ‘Adult ADHD Self-Report Scale’; TAU, ‘treatment as usual’.

Baseline characteristics
Table 2 presents baseline characteristics of the study sample. The mean age was ~38 years, with 71% reporting female sex. Most participants were in a relationship and had a higher level of education; over half held a university degree. Employment was common, with 42% working full-time and 33% part-time. The majority identified as White (93%), with smaller proportions reporting Middle Eastern (4.5%), Latin American (2.1%), or Black (1.2%) ethnicity. Current comorbid psychiatric conditions were frequent: 27% met diagnostic criteria for major depressive disorder, and 12% for social anxiety disorder. About 24% were currently in psychotherapy, and 36% were taking psychotropic medication, most commonly psychostimulants (28%).
Subject demographics and clinical characteristics at baseline. Values represent mean (SD) unless stated otherwise

Note: AQoL-8D, Assessment of Quality of Life - 8 Dimensions; ASRS, Adult ADHD Self-Report Scale; PHQ-9, Patient Health Questionnaire-9; RSES, Rosenberg Self-Esteem Scale; WSAS, Work and Social Adjustment Scale.
a Calculated across all participants, including those with zero sessions.
b Including medications classified under the Anatomical Therapeutic Chemical (ATC) system as N05 (psycholeptics) and N06 (psychoanaleptics).
Missingness
Group differences in baseline characteristics between participants who completed the primary endpoint assessment at T1 (N = 316) and those who dropped out for any reason (N = 21) are reported in Supplementary Table 1. Compared to completers, dropouts were significantly younger, less likely to identify as White, more likely to be treated by an ADHD specialist, and more likely to be taking psychotropic medication. They also reported higher baseline levels of ADHD symptoms and functional impairment.
Treatment effects
Primary endpoint: ADHD symptom severity
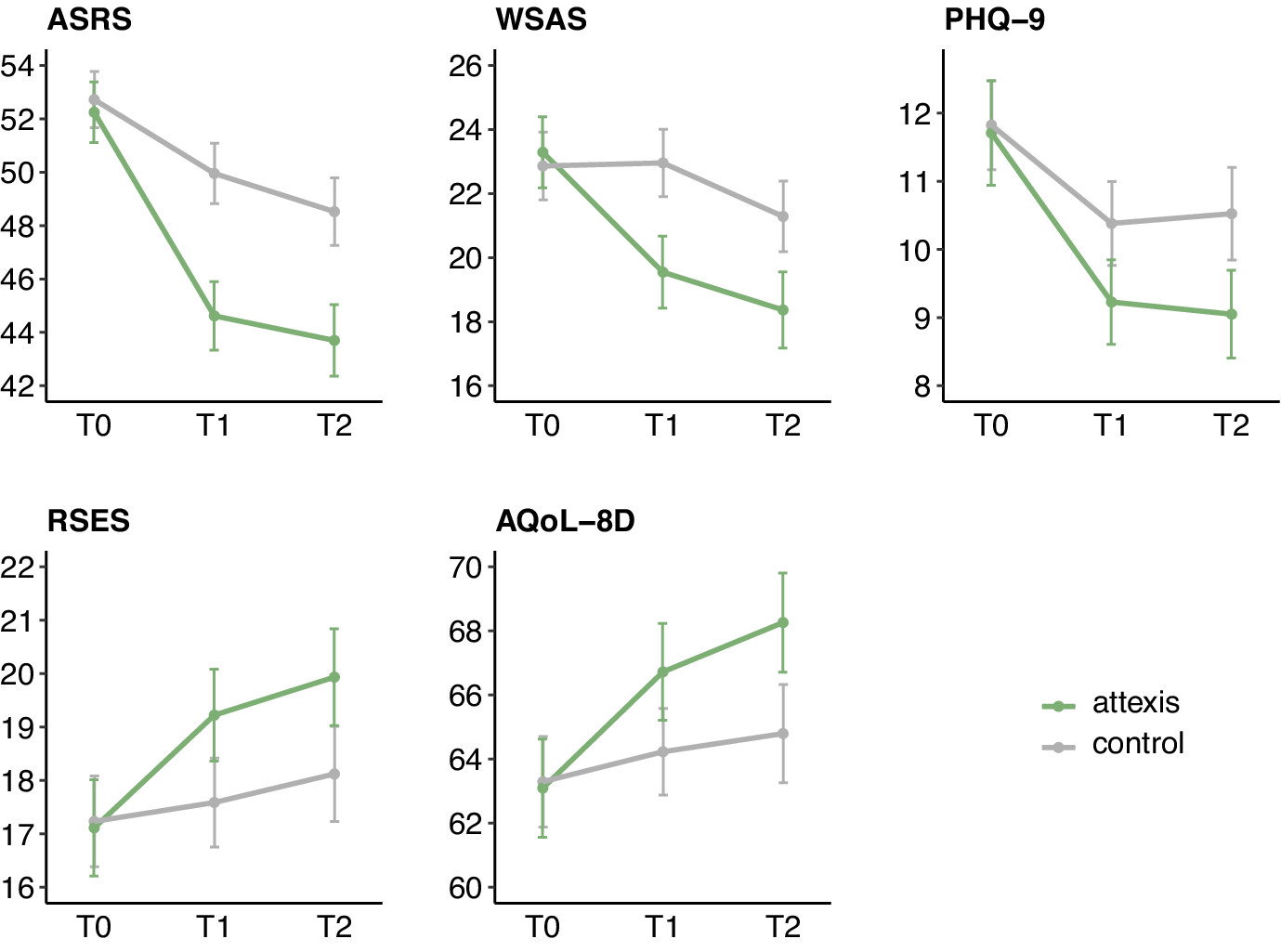
After 3 months, participants in the attexis + TAU group showed significantly lower ADHD symptom severity compared to the TAU-only group (see Table 3 and Figure 2). The baseline-adjusted mean difference on the ASRS total score was −5.0 points (95% CI = [−6.4, −3.6], p < .001; d = 0.85). Comparable effects were found in the J2R sensitivity analysis (−4.4 points, 95% CI [−5.7, −3.2], p < .001; d = 0.75) and the PP analysis (−5.0 points, 95% CI = [−6.4, −3.6], p < .001; d = 0.86). After 6 months, the ITT analysis confirmed a continued intervention effect (−4.5 points, 95% CI = [−6.2, −2.9], p < .001; d = 0.61). The J2R and PP analyses yielded comparable results (see Supplementary Tables 2 and 3), confirming the robustness of the treatment effect in relation to different assumptions concerning missing outcome data.
Results of primary and secondary endpoints for ITT analyses

Note: AQoL-8D, Assessment of Quality of Life - 8 Dimensions; ASRS, Adult ADHD Self-Report Scale; PHQ-9, Patient Health Questionnaire-9; RSES, Rosenberg Self-Esteem Scale; WSAS, Work and Social Adjustment Scale.
a Between-group difference on the original scale at 3 months (T1) and 6 months (T2), adjusted for baseline scores.
b Based on baseline-adjusted means; positive values show effects in favor of the intervention group.
Symptom course on primary and secondary outcomes (means ±95% confidence interval) using intent-to-treat analyses with multiple imputation for missing data. Plotted values represent total scores. Note: ASRS, ‘Adult ADHD Self-Report Scale’; AQoL-8D, ‘Assessment of Quality of Life - 8 Dimensions’; PHQ-9, ‘Patient Health Questionnaire-9’; RSES, ‘Rosenberg Self-Esteem Scale’; WSAS, ‘Work and Social Adjustment Scale’.

After 3 months, 11.6% of participants in the intervention group (19/164) met the predefined responder criterion (≥30% reduction in the ASRS total score), compared to 1.2% in the control group (2/173). The difference between groups was statistically significant (χ 2 = 15.67, p < .001), corresponding to an OR of 11.2 (95% CI = [2.6, 48.9]). The safety analysis further indicated that symptom worsening (i.e. any increase in ASRS total score from baseline to T1) was observed in 10.7% of participants in the intervention group (16/149) and 24% in the control group (40/167). The difference was statistically significant (χ 2 = 9.43, p = .002; OR = 2.6, 95% CI = [1.4, 5.0]), indicating that the use of attexis was associated with a lower likelihood of symptom worsening compared to TAU alone.
Secondary endpoints
After 3 months, ITT analyses showed significant improvements in the intervention group across all secondary outcomes (see Table 3 and Figure 2), including work and social functioning, depression, self-esteem, and quality of life. Effect sizes ranged from d = 0.32 to 0.61. J2R and PP analyses yielded consistent results (see Supplementary Tables 2 and 3).
Responder analyses confirmed the clinical relevance of these effects: significantly more participants in the intervention group showed meaningful improvements in work and social functioning (based on an MCID of 8 points [Zahra et al., Reference Zahra, Qureshi, Henley, Taylor, Quinn, Pooler and Byng2014]; 26.2% vs. 11.6%; χ 2 = 11.9, p < .001; OR = 2.7, 95% CI = [1.5, 4.9]), depression levels (based on an MCID of 5 points [Löwe et al., Reference Löwe, Unützer, Callahan, Perkins and Kroenke2004]; 31.1% vs. 19.7%; χ 2 = 5.85, p = .016; OR = 1.8, 95% CI = [1.1, 3.0]), self-esteem (based on the RCI; 20.1% vs. 9.8%; χ 2 = 7.06, p = .008; OR = 2.3, 95% CI = [1.2, 4.3]), and quality of life (based on the RCI; 21.3% vs. 9.8%; χ 2 = 8.55, p = .003; OR = 2.5, 95% CI = [1.3, 4.6]).
At 6 months, intervention effects continued to be observable across all secondary outcomes (see Table 3 and Figure 2). Effect sizes ranged from d = 0.36 to 0.47. These results were consistent in both PP and J2R analyses (see Supplementary Tables 2 and 3).
Subgroup analyses
Subgroup analyses showed consistent effectiveness of attexis across sex, psychotherapy status at baseline, psychotropic medication use, and treatment changes between T0 and T1 (see Supplementary Tables 4–7).
User satisfaction
Mean ratings for the likelihood to recommend the program to a friend or colleague were 6.1 (standard deviation [SD] = 2.9) at T1 and 5.9 (SD = 3.3) at T2, indicating a generally positive evaluation of attexis.
Subjective ADHD symptom improvement (assessed via the PGIC) was significantly greater in the intervention group at both T1 and T2 (T1: d = 0.56; T2: d = 0.55; both ps < .001). Similar effects were observed for perceived improvements in daily functioning (T1: d = 0.66; T2: d = 0.63; both ps < .001). In binary ratings, 36.5% of participants in the intervention group reported meaningful perceived functional improvement at T1 versus 9.6% in the control group (χ 2 = 32.9, p < .001); at T2, this was 40.1% versus 17.6% (χ 2 = 18.8, p < .001).
Adverse events
After 3 months, unplanned or emergency medical treatments were reported by 7.8% of participants in the control group (13/167) and 5.4% in the intervention group (8/147), with no significant group difference (χ 2 = 0.69, p = .407). A similar pattern was observed after 6 months (control: 6.3%, 10/159; intervention: 4.2%, 6/142; χ 2 = 0.63, p = .426). No adverse events were linked to the use of attexis, and no adverse device effects were reported.
Usage
Registered patients spent an average of 10.9 h (SD = 6.5) in the program up to T1 and 13.3 h (SD = 6.9) up to T2, across an average of 10 (SD = 5.1) and 11 different use days (SD = 6.2), respectively. Changes in ADHD symptom severity from baseline to T1 were not significantly correlated with total hours spent in the program up to T1 (r = 0.08, p = .292), and showed only a negligible correlation with the number of active days up to T1 (r = 0.17, p = .044).
Discussion
This pragmatic RCT evaluated the effectiveness of attexis, a fully self-guided digital intervention based on cognitive-behavioral and mindfulness principles for adults with ADHD. Results show that attexis, when added to TAU, significantly reduced ADHD symptom severity and improved a range of patient-relevant functional and psychosocial secondary outcomes compared to TAU-only. Effects were robust under conservative assumptions regarding missing outcome data and sustained at the 6-month follow-up, highlighting the long-term potential of the intervention. Responder analyses confirmed the clinical relevance of the intervention effects across all outcomes. Participants reported good satisfaction with attexis, along with perceived improvements in ADHD symptoms and their impact on daily life. Importantly, no adverse events were attributed to the use of attexis.
In terms of the primary outcome, i.e. ADHD symptom severity after 3 months, attexis demonstrated a large effect size (d = 0.85). This finding aligns with previous research on digital interventions for adult ADHD, which reported effect sizes ranging from d = 0.42 to 1.21 for this outcome (Kenter, Gjestad, Lundervold, & Nordgreen, Reference Kenter, Gjestad, Lundervold and Nordgreen2023; Moëll et al., Reference Moëll, Kollberg, Nasri, Lindefors and Kaldo2015; Nasri et al., Reference Nasri, Cassel, Enhärje, Larsson, Hirvikoski, Ginsberg and Kaldo2023; Pettersson, Söderström, Edlund-Söderström, & Nilsson, Reference Pettersson, Söderström, Edlund-Söderström and Nilsson2017; Selaskowski et al., Reference Selaskowski, Steffens, Schulze, Lingen, Aslan, Rosen and Boll2022). However, all previous studies featured small sample sizes, a common limitation in research on nonpharmacological treatments for adult ADHD (Nimmo-Smith et al., Reference Nimmo-Smith, Merwood, Hank, Brandling, Greenwood, Skinner and Rai2020). In contrast, the present study was adequately powered and, to our knowledge, represents the largest RCT to date evaluating a digital intervention for adults with ADHD. Therefore, our study strengthens the evidence base for digital interventions in this population. Notably, the magnitude of the intervention effect falls within the range reported in meta-analyses of face-to-face CBT for adult ADHD (d = 0.71–0.98) (Liu, Hua, Lu, & Goh, Reference Liu, Hua, Lu and Goh2023; Lopez et al., Reference Lopez, Torrente, Ciapponi, Lischinsky, Cetkovich-Bakmas, Rojas and Manes2018; Young, Moghaddam, & Tickle, Reference Young, Moghaddam and Tickle2020). Thus, our results mirror previous meta-analytic findings suggesting that therapeutic content may matter more than delivery format in the therapy of adult ADHD (Knouse, Teller, & Brooks, Reference Knouse, Teller and Brooks2017).
Regarding secondary outcomes, use of attexis was linked to consistent benefits in a wide range of functional and psychosocial domains, specifically, occupational and social functioning, depressive symptoms, self-esteem, and quality of life. These outcomes are highly relevant from a patient perspective, as they reflect the real-world burden of ADHD and are closely linked to long-term functioning and well-being (Coghill et al., Reference Coghill, Banaschewski, Soutullo, Cottingham and Zuddas2017; Cook, Knight, Hume, & Qureshi, Reference Cook, Knight, Hume and Qureshi2014; D’Amelio, Retz, Philipsen, & Rösler, Reference D’Amelio, Retz, Philipsen and Rösler2009). Despite their clinical relevance and inclusion in guideline recommendations, they remain insufficiently addressed in both routine care and research (AWMF, 2017; Kooij et al., Reference Kooij, Bijlenga, Salerno, Jaeschke, Bitter and Balazs2019; López-Pinar et al., Reference López-Pinar, Martínez-Sanchís, Carbonell-Vayá, Sánchez-Meca and Fenollar-Cortés2020). This may, in part, be due to the fact that pharmacotherapy, currently the predominant treatment approach for adult ADHD, has shown only limited and inconsistent effects on these broader psychosocial outcome domains (Bellato et al., Reference Bellato, Perrott, Marzulli, Parlatini, Coghill and Cortese2024; Castells, Blanco-Silvente, & Cunill, Reference Castells, Blanco-Silvente and Cunill2018; Coghill et al., Reference Coghill, Banaschewski, Soutullo, Cottingham and Zuddas2017; Lenzi, Cortese, Harris, & Masi, Reference Lenzi, Cortese, Harris and Masi2018).
Over the 6-month follow-up, we observed relevant dynamics in the evaluated outcomes: While the intervention effects of attexis on core ADHD symptoms and functional impairment were maintained at 6 months, improvements in depressive symptoms and quality of life increased further over time. This pattern suggests that as core symptoms decrease, individuals begin to engage more with their environment and implement adaptive coping strategies in daily life. According to CBT models of adult ADHD, such behavioral engagement and cognitive restructuring can, over time, reduce comorbid internalizing symptoms and the broader real-world burden associated with ADHD (López-Pinar et al., Reference López-Pinar, Martínez-Sanchís, Carbonell-Vayá, Sánchez-Meca and Fenollar-Cortés2020; Safren, Sprich, Chulvick, & Otto, Reference Safren, Sprich, Chulvick and Otto2004).
Overall, our findings address a relevant gap in the current treatment for adults with ADHD. Psychosocial interventions, particularly CBT, are recommended in the treatment of adult ADHD (AWMF, 2017; Kooij et al., Reference Kooij, Bijlenga, Salerno, Jaeschke, Bitter and Balazs2019), but access remains limited, with long waiting times and a lack of trained professionals representing well-documented barriers in Germany (Kruse et al., Reference Kruse, Kampling, Bouami, Grobe, Hartmann, Jedamzik and Friederich2024; Libutzki et al., Reference Libutzki, Ludwig, May, Jacobsen, Reif and Hartman2019; Schneider et al., Reference Schneider, Schöttle, Hottenrott, Gallinat and Moritz2023). This is reflected in our sample, where only 3.6% of participants received care by an ADHD specialist at baseline. Importantly, many adults with ADHD report that pharmacological treatment alone is inadequate and express a desire for psychoeducation and psychosocial support (AWMF, 2017; Matheson et al., Reference Matheson, Asherson, Wong, Hodgkins, Setyawan, Sasane and Clifford2013). Reflecting this, meta-analytic evidence indicates that combining pharmacotherapy with CBT may lead to improved treatment outcomes in adult ADHD compared to pharmacotherapy alone (Lopez et al., Reference Lopez, Torrente, Ciapponi, Lischinsky, Cetkovich-Bakmas, Rojas and Manes2018). Easily accessible digital interventions, such as attexis, may represent a valuable addition to the treatment of adult ADHD across different clinical contexts, including, as evidenced by subgroup analyses, both as a stand-alone tool and in combination with pharmacotherapy.
Limitations
Several limitations need to be considered when interpreting our results. First, while ADHD diagnoses were confirmed before inclusion by trained clinicians using structured interviews, all outcome measures were based on self-report, which may be subject to recall or social desirability bias. In line with patient-centered care, however, self-report captures patients’ subjective experience, and the use of validated instruments and objective responder criteria supports the reliability of the findings. Moreover, existing evidence does not suggest that self-reports systematically overestimate treatment effects; for example, a recent meta-analysis comparing self- and clinician-rated outcomes of psychotherapies for depression found that both masked and unmasked clinician ratings yielded larger effects than self-ratings (Miguel et al., Reference Miguel, Harrer, Karyotaki, Plessen, Ciharova, Furukawa and Cuijpers2025). Second, participants were not blinded to group allocation, which, combined with longer treatment time delivered through attexis compared to TAU, may have exaggerated intervention effects, for example, via expectancy effects. That said, blinding is generally difficult in psychotherapy research, as the development of credible but ineffective sham interventions poses significant conceptual challenges (Kirsch, Wampold, & Kelley, Reference Kirsch, Wampold and Kelley2016). Moreover, TAU is considered an appropriate comparator when the primary objective is to establish an intervention’s effectiveness in the context of existing clinical practice, which was also the case in our trial (Freedland et al., Reference Freedland, King, Ambrosius, Mayo-Wilson, Mohr and Czajkowski2019). Third, satisfaction ratings suggest that experiences with the program varied. Future qualitative research should examine more in-depth which components were perceived as helpful or lacking by patients. Fourth, attrition was overall low compared to the literature (Linardon & Fuller-Tyszkiewicz, Reference Linardon and Fuller-Tyszkiewicz2020), but slightly higher in the intervention group. While this pattern is common (Assmann et al., Reference Assmann, Jacob, Schaich, Berger, Zindler, Betz and Klein2025; Betz et al., Reference Betz, Jacob, Knitza, Koehm and Behrens2024; Specht et al., Reference Specht, Betz, Riepenhausen, Jauch-Chara, Jacob, Riemann and Göder2024) and was addressed analytically, underlying reasons for dropout warrant further investigation. Fifth, in absolute terms, the proportion of patients in the intervention group who reached the predefined responder criterion for the primary endpoint was relatively low, albeit significantly higher than in the control group. The corresponding effect size was considerable (OR = 11.2), supporting the clinical relevance of the finding. Finally, generalizability may be limited to digitally savvy individuals willing to engage with a digital solution for the treatment of ADHD symptoms. However, this population likely mirrors those who are most likely to use and benefit from prescribed digital interventions in real-world care settings (Betz et al., Reference Betz, Jacob, Knitza, Koehm and Behrens2024).
Conclusion
In conclusion, this study adds important evidence to the growing field of digital interventions for adult ADHD. attexis, a fully self-guided digital intervention based on BT and mindfulness principles, was associated with significant and clinically relevant reductions in ADHD symptom severity and improvements in a broad range of patient-relevant secondary outcomes, including functional impairment, depressive symptoms, self-esteem, and quality of life. The intervention showed consistent effects across diverse subgroups and was well accepted by users. While further research is needed to replicate and extend these findings, particularly in routine care settings, digital interventions like attexis may offer a promising treatment option for adults with ADHD, especially in light of limited access to evidence-based psychosocial interventions.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0033291726103390.
Acknowledgments
The authors would like to thank all patients who took part in the READ-ADHD RCT. Part of these findings were presented by LTB at the 2024 Annual Congress of the German Association for Psychiatry, Psychotherapy, and Psychosomatics (DGPPN) in Berlin, Germany.
Author contribution
Conception and Design of the Study: R.D., L.T.B., J.P.K., G.A.J., and P.R.J.; Critical Revision of the Manuscript for Important Intellectual Content: R.D., L.T.B., S.M.J., W.R., A.P., J.P.K., E.F., G.A.J., and P.R.J.; Data Analysis and Interpretation: L.T.B., R.D., P.R.J., and G.A.J.; Data Collection: L.T.B., S.M.J., and G.A.J.; Drafting of the Manuscript: L.T.B., R.D., G.A.J., and P.R.J.; Final Approval of the Version to be Published: R.D., L.T.B., S.M.J., W.R., A.P., J.P.K., E.F., G.A.J., and P.R.J.
Funding statement
This study was funded by GAIA, the developer, owner, and manufacturer of attexis.
Competing interests
LTB and GAJ are employees of GAIA, the developer, owner, and manufacturer of attexis.
AP declares that she served on advisory boards, gave lectures, performed phase-3 studies, and received travel grants within the last 3 years from MEDICE Arzneimittel, Pütter GmbH and Co KG, Takeda, and Boehringer, and received royalties from books published by Elsevier, Hogrefe, Schattauer, Kohlhammer, Karger, Oxford Press, Thieme, Springer, and Schattauer.
EF declares that she received royalties from Beltz Verlag and Elsevier Book; personal fees for supervision, workshops, and presentations on CBT, personality disorders, and posttraumatic stress disorder and depression; is co-chair of the Deutsche Fachverband für Verhaltenstherapie e.V. (unpaid); and received payments from GAIA for consulting activities for designing digital therapeutics for patients for BPD as well as for presentations on schema therapy.
Ethical standard
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.