Introduction
Although symptoms of anxiety and depression are common among individuals with anorexia nervosa (AN), the small number of randomized placebo-controlled trials (RCTs) of antidepressants have provided little evidence of therapeutic benefit (Fornaro et al., Reference Fornaro, Mondin, Billeci, Fusco, De Prisco, Caiazza, Micanti, Calati, Carvalho and de Bartolomeis2023). Among these negative trials was an RCT from our group comparing the benefit of fluoxetine versus placebo in preventing relapse among women with AN in the 12 months following full weight restoration (Walsh et al., Reference Walsh, Kaplan, Attia, Olmsted, Parides, Carter, Pike, Devlin, Woodside, Roberto and Rockert2006). There was no significant difference between fluoxetine and placebo in time to relapse, the primary outcome measure. The only two significant differences were in secondary analyses of change in anxiety and in obsessionality over time favoring fluoxetine (uncorrected for multiple comparisons). No significant differences were found in similar analyses of nine other features, including depression, raising concerns about the validity of the finding regarding anxiety.
In light of the well-established efficacy of SSRIs in the treatment of depression and the surprising lack of efficacy of fluoxetine in the original study, we conducted a new analysis to examine whether the effect of fluoxetine versus placebo was moderated by depression. Specifically, we assessed whether fluoxetine was of greater benefit to the more depressed patients.
Methods
This study comprises secondary analyses of data from a two-site study (New York and Toronto) examining the effect of fluoxetine versus placebo in preventing relapse among 93 weight-restored women with anorexia nervosa, aged 16–45 years. Demographic information including race and ethnicity was obtained by self-report before entry into the study; information on socioeconomic status was not systematically collected. Full experimental details are provided in the original publication (Walsh et al., Reference Walsh, Kaplan, Attia, Olmsted, Parides, Carter, Pike, Devlin, Woodside, Roberto and Rockert2006). Briefly, patients were eligible to enter the study after they had successfully completed weight restoration treatment in an inpatient or day-hospital program during which their body mass index (BMI) reached at least 19 and remained at or above that level for 2 weeks. Patients were then randomly assigned to fluoxetine or placebo and the dose was raised to 60 mg/d or the equivalent dose of placebo over 1 week. Patients were discharged and followed for up to a year or until they relapsed. Patients also received outpatient cognitive-behavioral therapy (CBT) for up to 1 year. Patients were weighed at each vis/it. The study was approved by the appropriate institutional review boards at both sites and all adult patients provided written informed consent; patients less than 18 years of age provided assent and parents/guardians provided written informed consent. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
The primary aim of the original study was to determine whether fluoxetine extended the time to relapse compared to placebo; the primary outcome measure was time-to-relapse and was examined via survival analysis utilizing a Cox proportional hazards regression. Forty of the 93 patients completed the full study and therefore were considered not to have relapsed. The remaining 53 patients failed to complete the full study and were therefore considered to have relapsed. There were two reasons for considering failure to complete the study as relapse. First, it limited the role of investigators’ judgments in deciding who had relapsed and therefore protected the results from potential bias if the investigator suspected the patient was on fluoxetine or placebo. Second, it was essentially identical to the criterion utilized by Kaye et al. (Reference Kaye, Nagata, Weltzin, Hsu, Sokol, McConaha, Plotnicov, Weise and Deep2001), who conducted a prior, smaller RCT and reported that fluoxetine substantially reduced the rate of relapse. Several reasons contributed to the failure to complete the full study. Twenty patients met a priori criteria for severe deterioration (specifically, BMI ≤16.5 for two consecutive weeks, severe medical complications, imminent risk of suicide, or the development of another psychiatric disorder requiring treatment). These patients were withdrawn from the study to protect their well-being and were referred to a higher level of care. Six patients were withdrawn by the investigators because of non-compliance or possible side effects. In addition, 27 patients who voluntarily withdrew from treatment were classified as having relapsed.
Two other definitions of relapse, based on different ways of classifying dropouts, were used to examine the sensitivity of results to relapse definition: (1) Dropouts were classified as having relapsed based on clinical criteria; specifically, they were classified as having relapsed only if, at the time they chose to terminate participation, their BMI was <17.5, they were binge eating and/or purging two or more times per week for the prior 4 weeks, or they exhibited depressive or anxiety symptoms sufficient to interfere with functioning (e.g. serious suicidal ideation). (2) In a third method, only patients who were withdrawn for severe deterioration as defined above were considered to have relapsed at the time they ended participation. None of the patients who dropped out or were withdrawn for other reasons were considered to have relapsed; that is, their data were censored. In none of the analyses based on these definitions of relapse was a significant difference detected between the fluoxetine- and placebo-treated groups in time to relapse (Walsh et al., Reference Walsh, Kaplan, Attia, Olmsted, Parides, Carter, Pike, Devlin, Woodside, Roberto and Rockert2006).
The current analysis extended the original analyses to examine the effect of fluoxetine versus placebo on time to relapse among more depressed versus less depressed patients. The current study included all individuals from the original trial who completed the Beck Depression Inventory (BDI) (Beck et al. Reference Beck, Ward, Mendelson, Mock and Erbaugh1961) prior to randomization, resulting in a sample of 92 patients (43 in the placebo group; 49 in the fluoxetine group). One individual in the placebo group was not included due to missing BDI data.
Patients were categorized as having low (BDI ≤ 20) or high (BDI > 20) depression severity immediately prior to randomization based on their total score. This cut-off was chosen on the basis of recommendations for the identification of depressed individuals using the BDI (Kendall et al., Reference Kendall, Hollon, Beck, Hammen and Ingram1987).
Data analysis
Time-to-relapse analysis
In the main analyses, we used the primary definition of relapse in the original study (i.e. drop-out for any reason was considered a relapse); analyses with the two alternate definitions are presented in the Supplementary Material.
To assess whether there was a significant difference between fluoxetine versus placebo in time to relapse among patients with high depression versus low depression severity, we used Cox proportional hazards models that included drug status (i.e. fluoxetine or placebo), depression severity status (i.e. high or low), and their interaction. In post-hoc analyses, we examined the effect of drug on relapse status for high and low depression severity groups separately. These analyses were Bonferroni-corrected for multiple comparisons. All models were adjusted for site and subtype as in the original analyses.
Change in symptoms and weight over time
We also examined the effect of depression on the impact of fluoxetine versus placebo on the rate of change of self-reported symptoms over time using mixed effects modeling. The symptoms assessed and the rating instrument used were: anxiety, Beck Anxiety Inventory (BAI) (Beck et al. Reference Beck, Epstein, Brown and Steer1988); depression, BDI (Beck et al., Reference Beck, Ward, Mendelson, Mock and Erbaugh1961); obsessions, Yale-Brown-Cornell Eating Disorder Scale (YBC-EDS) (Mazure et al., Reference Mazure, Halmi, Sunday, Romano and Einhorn1994); drive for thinness, Eating Disorders Inventory (Garner et al., Reference Garner, Olmsted and Polivy1983) Drive for Thinness scale (EDI-DT); bulimia, Eating Disorders Inventory Bulimia scale (EDI-BN); Eating Disorders Inventory Body Dissatisfaction scale (EDI-BD), Eating Disorders Inventory Perfectionism scale (EDI-Perf); self-esteem, Rosenberg Self-Esteem Scale (RSE) (Rosenberg, Reference Rosenberg1965); and quality of life, Quality of Life Enjoyment and Satisfaction Questionnaire (QlesQ) (Endicott et al., Reference Endicott, Nee, Harrison and Blumenthal1993). These measures were obtained on three occasions: immediately before randomization, at 6 months, and at study termination.
First, we examined whether fluoxetine-by-time effects were significant in the high and low depression groups. When these analyses indicated a significant effect of time on symptoms in either group, we assessed whether the effects of drug differed between low and high depression severity groups. In these analyses, we included all individuals; a drug-by-depression-severity-by-time interaction term was used to examine the differential effect of drug on groups varying in depression severity status. In all statistical models, we specified a random intercept for each participant.
The same approach was used to examine changes in weight. In these analyses, a random intercept and slope were specified for each participant, as there were more assessments per individual since weight was measured weekly.
Analyses were completed using Rstudio (version 4.1.2; R Core Team). Survival analyses were conducted using the ‘survival’ package (DOI: 10.32614/CRAN.package.survival).
Results
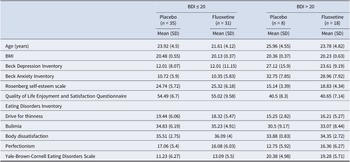
At the time of study entry, the mean age of the patients was 23.3 ± 4.6(SD) years, and the mean BMI was 20.3 ± 0.5. The mean duration of illness was 4.5 ± 3.6 years. Seventy-eight (83.9%) of the patients were White, 8 (8.6%) were Asian, 2 (2.2%) were Hispanic, 1 (1.1%) was Black, and 4 (4.3%) reported more than one race. Table 1 describes patient demographic and clinical characteristics by drug and depression severity group.
Demographic and clinical characteristics of patients at study initiation

Time to relapse
The analysis of time to relapse, using the primary definition of relapse in which failure to complete the study for any reason was considered a relapse, found a significant interaction between drug (fluoxetine vs placebo) and level of depression (high vs low): hazard ratio = 0.46, 95% CI [−0.25, 0.85], p = 0.01) (Figure 1).
Fractions of patients receiving placebo and fluoxetine remaining in treatment vs number of days after treatment initiation by level of BDI at initiation. Solid lines indicate placebo group; dashed lines indicate fluoxetine group. Light gray lines indicate time course for all patients; blue lines for the more depressed group; red lines for the less depressed group.

In post-hoc analyses, among the more depressed patients, the time to relapse was significantly longer for those treated with fluoxetine compared to those treated with placebo hazard ratio = 0.46, 95% CI:[0.24, 0.89], p corrected = 0.04). The difference in time to relapse between the fluoxetine and placebo groups among the less depressed patients was not significant (hazard ratio = 1.15, 95%CI: [0.82, 1.61], p corrected = 0.82).
Sensitivity analyses using alternative definitions of relapse produced results that were qualitatively consistent with those of the primary analysis (see Supplementary Material).
Change in symptoms over time
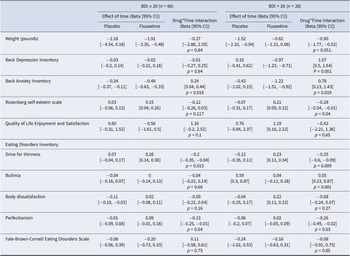
Table 2 shows the results of the mixed effects models examining the effect of drug in both low and high depression severity groups. Outcomes of these analyses indicated a significant effect of the drug on BDI, BAI and RSE (greater improvement in symptoms), and BN symptoms (reduced deterioration) in the high depression severity group; the effect on weight approached but did not reach significance. Fluoxetine versus placebo was associated with a worsening in drive for thinness and perfectionism among the high depression severity group.
Changes in symptoms over time by drug and depression severity group

Note: Beta and CI for effect of time on symptoms were estimated in linear mixed effects models including a random intercept for participant; for the weight outcome models also included a random slope for participant.
Analyses including all individuals and a drug-by-depression-severity-group-by-time interaction indicated greater (protective) effects of the drug on BDI (β = −0.27, 95% CI: [−0.42, −0.12], p = 0.001) and EDI-BN over time (β = −0.15, 95% CI: [−0.25, −0.05], p = 0.004) for high versus low depression severity groups (Table 2 and Figure 2).
Moderation of drug effect on weight and psychological symptoms by depression severity group. In each plot, the left panel depicts the symptom change for the group with lower levels of depression (BDI < 20), while the right panel depicts symptom change for the more depressed group (BDI ≥ 20). *denotes a statistically significant effect of the drug (within the depression severity group). Note that these are raw estimates rather than model-predicted values. EDI = Eating Disorders Inventory; Quality of Life and Satisfaction = Quality of Life Satisfaction and Enjoyment Questionnaire; YBC=Yale-Brown-Cornell.

Discussion
The current analysis suggests that the impact of fluoxetine on relapse following acute treatment of AN was moderated by the level of depression and that fluoxetine reduced the risk of relapse over the following year among patients who were moderately or more severely depressed following weight restoration. In addition, among these patients, fluoxetine hastened the rate of decline of symptoms of anxiety and depression and increased the rate of improvement in self-esteem. Although based on secondary analyses of a small number of patients, the current findings are derived from an RCT, are based on a priori criteria for relapse, and used statistical methods identical to those utilized in the original analysis, increasing the likelihood that they are not due to chance. In addition, they are consistent with the well-established utility of antidepressant medication in the treatment of depressed individuals (Cipriani et al., Reference Cipriani, Furukawa, Salanti, Chaimani, Atkinson, Ogawa, Leucht, Ruhe, Turner, Higgins, Egger, Takeshima, Hayasaka, Imai, Shinohara, Tajika, Ioannidis and Geddes2018).
Among the more depressed patients, two measures of the specific psychopathology of AN (the EDI drive for thinness and perfectionism scales) increased over time among patients on fluoxetine but fell among those on placebo. On the other hand, the EDI-bulimia subscale rose more rapidly on placebo than on fluoxetine. Interpretation of these results is difficult. First, it should be noted that all the changes were small in magnitude and therefore of uncertain clinical significance. Second, the findings are based on a small number of patients (8 on placebo and 18 on fluoxetine) assessed on only three occasions (pre-randomization, after 6 months, and at study termination) reducing confidence in the findings. One possible interpretation is that the extension of time to relapse and continued weight maintenance contributed to greater concern about shape and weight among patients with higher levels of depression randomized to fluoxetine, but this hypothesis requires direct testing.
The present results should be viewed in light of major limitations. Of greatest importance, the original study was not intended to test the hypothesis that the effect of fluoxetine versus placebo varied with the level of depression at the time of randomization. Therefore, the current analysis must be viewed as exploratory in nature and is in need of replication. Since support for a large RCT is unlikely in the present funding environment, it might be informative to examine, via analysis of existing data (e.g. electronic health records) or in a prospective observational study, the course of patients who were depressed following acute treatment and who did or did not receive antidepressant treatment. In the current study, the sole criterion for categorizing individuals as more depressed was a score > 20 on the BDI, a self-report instrument; while this is reasonable, there was no interview-based assessment of the level of depression. In addition, the sample was comprised almost entirely of white/non-Hispanic women, potentially limiting generalizability to more diverse populations and to samples with different sociodemographic characteristics.
Despite these limitations, the data were obtained from a rigorously controlled two-site RCT, the largest study of an antidepressant in AN yet conducted. The primary outcome measure, time to relapse, was evaluated using a priori criteria intended to reduce bias, and results were robust to alternate definitions of relapse. Although the current findings are in need of replication, the results of this secondary analysis suggest that clinicians should consider the use of an antidepressant for individuals with AN who remain significantly depressed following full weight restoration.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S003329172510127X.
Acknowledgments
We gratefully acknowledge the major contributions of Allan S. Kaplan, MD, University of Toronto, to the original study. These results were presented, in part, at the 30th annual meeting of the Eating Disorders Research Society, Sitges, Spain, September 26–28, 2024.
Funding statement
This study was supported in part by NIH grants MH060336, MH060271, and MH117458.
Competing interests
Dr. Walsh reports receiving royalties and honoraria from Guilford Publications, McGraw-Hill, Oxford University Press, Wolters Kluwer, and Wiley. Dr. Attia reports receiving royalties from Wolters Kluwer and Oxford University Press and holds equity from Equip Health, Inc. Dr. Griffen is employed by Boehringer Ingelheim Pharmaceuticals, Inc. Drs. Lloyd and Wang report no disclosures.