Introduction
CHD are the most common congenital anomaly, affecting about 9.4 in 1000 live births globally.Reference Liu, Chen and Zuhlke1,Reference Gelb, Nakanishi, Markwald, Baldwin, Keller, Srivastava and Yamagishi2,Reference Wilsdon3 Fetal echocardiography is typically performed between 18 and 22 weeks’ gestation and is the standard prenatal screening tool for pregnancies in which either parent has known CHD.Reference Haxel, Johnson and Hintz4,Reference Moon-Grady, Donofrio and Gelehrter5 Reported sensitivity rates vary, but fetal echocardiography is approximately 75% sensitive for the prenatal detection of CHD.Reference Liu, Zhou and Feng6 Postnatal echocardiography is performed when clinical concerns arise, such as in the presence of an abnormal fetal echocardiogram, abnormal neonatal cardiac examination, strong family history, or haemodynamic instability.Reference Wasserman, Shea and Cassidy7,Reference Mertens, Seri and Marek8
Most CHDs are isolated and thought to result from a complex interplay of genetic and environmental factors.Reference Boyd, McMullen, Beqaj and Kalfa9 However, about 20% of cases are associated with inherited syndromes or known chromosomal anomalies, including Down Syndrome and 22q11.2 Deletion Syndrome.Reference Blue, Kirk, Sholler, Harvey and Winlaw10 Advances in medical and surgical management of CHDs have significantly improved outcomes, and most individuals with CHD now survive to adulthood, and many have children.Reference Downing, Nembhard and Rose11 Genetic counselling can help assess the risk of CHD in children, using family history and, when needed, genetic testing. As genetics research has advanced, CHD gene panels have grown from 30 genes in 2012 to over 50 today.Reference Blue, Kirk, Sholler, Harvey and Winlaw10,12
Given its lifelong impact, individuals with CHD often seek counselling about the potential risk of CHD in their offspring. Large population-based studies have shown that children of mothers with CHD have significantly higher risk—one study in Denmark reported a relative risk of 5.39, and a Canadian study found an adjusted odds ratio of 9.92.Reference Liu, Joseph and Lisonkova13,Reference Yokouchi-Konishi, Yoshimatsu and Sawada14 Despite the relatively high prevalence of CHD and the increasing number of individuals with CHD reaching reproductive age, contemporary data on the incidence of CHD in offspring of affected mothers in the United States remains limited. This study aims to evaluate the incidence of CHD among offspring of mothers with CHD at a large academic medical centre in the southeastern United States.
Materials and methods
We conducted an observational study of pregnant patients who received prenatal care from the Cardio-Obstetrics Program at a large academic medical centre in the southeastern United States between 2015 and 2023. As part of routine clinical practice, all patients with CHD seen in this clinic are recommended to undergo fetal echocardiography, performed in an ultrasound unit certified by the American Institute of Ultrasound in Medicine. Postnatal echocardiography is recommended for patients with prenatally diagnosed fetal CHD, incomplete fetal imaging (e.g., due to poor acoustic windows or maternal obesity), lesions with low prenatal detection rates (e.g., pulmonary stenosis), or a history of a prior child with a CHD missed on fetal echocardiography.
Patients were eligible for inclusion if they carried a diagnosis of CHD and delivered at our institution or had outside hospital delivery data available to us. Exclusion criteria included acquired maternal heart disease, CHD associated with inherited disorders or genetic syndromes such as Marfan Syndrome, referrals for other reasons such as congenital arrhythmias, and deliveries at outside hospitals with missing records in our system. The study was approved by the Institutional Review Board at the University of Alabama at Birmingham.
For each pregnancy, data were collected on demographic characteristics, including maternal age, race, and select comorbidities, including pre-gestational diabetes mellitus, chronic hypertension, obesity (defined as a body mass index > 30 at the first prenatal care visit), thyroid disease, and current or former tobacco use. Maternal cardiac diagnosis based on the echocardiogram interpreted by an adult congenital cardiologist was also recorded. Pregnancy-related characteristics and delivery data, including gravidity, number of living children, and complications such as gestational diabetes mellitus, gestational hypertension, preeclampsia, and eclampsia; mode of delivery (vaginal or caesarean); gestational age at delivery; and APGAR scores, were abstracted from the medical record.
The primary outcome was defined as the presence or absence of CHD in the infant, as diagnosed by postnatal echocardiogram. In cases where the CHD lesion was identified prenatally by fetal echocardiogram, the diagnosis had to be confirmed by postnatal imaging. The specific type of defect was recorded. Secondary outcomes included neonatal intensive care unit admission, early in-hospital cardiac surgery, and in-hospital infant mortality. For this analysis, small patent ductus arteriosus findings on postnatal echocardiograms were not classified as defects since they are considered physiologic in the neonatal period and were deemed normal by the interpreting cardiologist.
For classification purposes, patients with Tetralogy of Fallot or Shone complex were each counted as having a single cardiac lesion. However, in mothers with multiple cardiac defects not part of a known cardiac syndrome, each defect was counted separately. Continuous variables are presented as mean ± standard deviation and categorical variables as counts and percentages. Baseline and pregnancy characteristics are reported as proportions of total pregnancies rather than total maternal count, given interval changes in maternal characteristics across pregnancies. Infant outcomes are reported as a proportion of total infants, which exceeds the number of pregnancies. Data analysis was performed using Microsoft Excel (Redmond, WA).
Results
From a cohort of 400 patients seen at our institution’s Cardio-Obstetrics Clinic between 2015 and 2023, 186 mothers with 195 pregnancies met inclusion criteria. A total of 151 pregnancies were excluded due to the mother having an acquired heart condition, 41 due to the mother having an inherited or syndromic CHD, 9 pregnancies were excluded because delivery occurred at an outside hospital and postnatal records were unavailable to definitively confirm fetal cardiac status, and 4 were excluded due to referrals for other non-CHD reasons (Figure 1). Due to twin gestations, the 195 pregnancies resulted in a total of 198 infants.
Flowchart of inclusion criteria for pregnancies included in the analysis. This figure illustrates the inclusion criteria for pregnancies analysed in the study. From 400 pregnancies seen at the UAB Cardio-Obstetrics Program between 2015 and 2023, exclusions were made for acquired heart disease, inherited or genetic syndromes, deliveries at outside hospitals with missing data, and other referrals for non-congenital heart defect indications. This final cohort included 195 pregnancies in mothers with CHD. The incidence of infant CHD is listed at the bottom of the diagram.

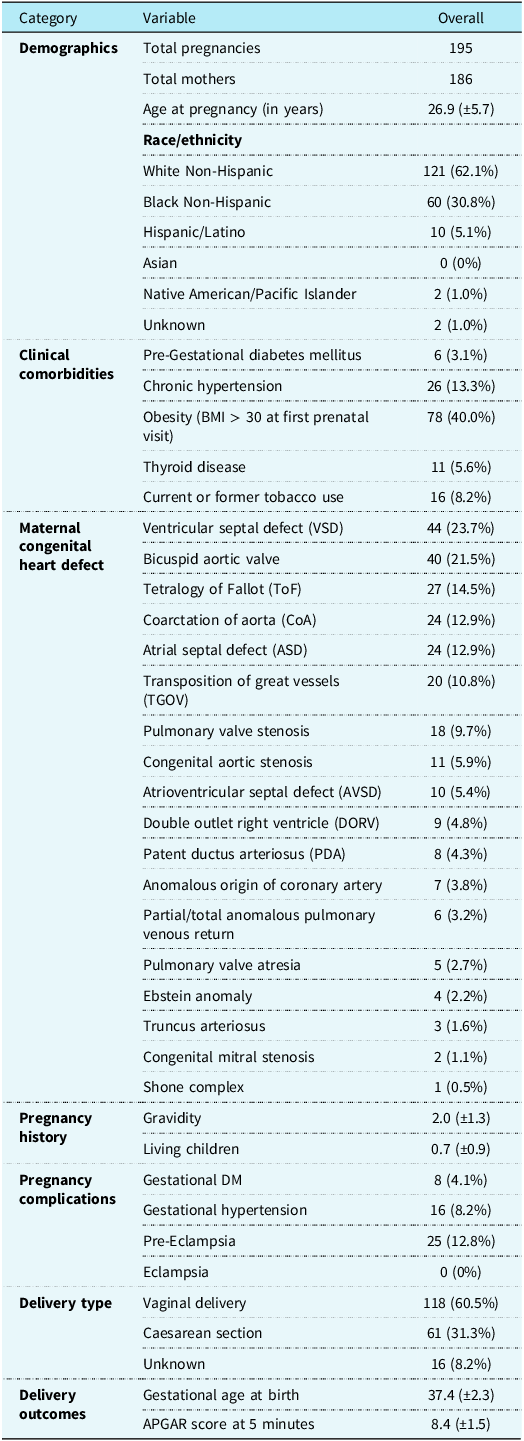
Mean maternal age was 26.9 years (SD ± 5.7). Most of the cohort were White Non-Hispanic/Latino (62.1%), followed by Black Non-Hispanic/Latino (30.8%) and Hispanic/Latino (5.1%). The most prevalent comorbidities in the cohort were obesity (40.0%), chronic hypertension (13.3%), and current/former tobacco use (8.2%) (Table 1). The most prevalent maternal cardiac defects were ventricular septal defect (23.7%), bicuspid aortic valve (21.5%), Tetralogy of Fallot (14.5%), coarctation of the aorta (12.9%), and atrial septal defect (12.9%).
Demographics, clinical characteristics, congenital heart defect types, and pregnancy outcomes in study cohort

This table presents a comprehensive summary of cardio-obstetric patients included in our study. The mean age was 26.9 years (SD ± 5.7), and the most prevalent congenital heart defects were ventricular septal defect (23.7%) and bicuspid aortic valve (21.5%). Obesity was the most prevalent comorbidity (40.0%), while pre-eclampsia was the most frequent pregnancy complication (12.8%). Vaginal delivery occurred in 60.5% of cases, and the mean gestational age at birth was 37.4 weeks with a mean 5-minute APGAR score of 8.4.
Data are mean (standard deviation) or no. (%), unless otherwise specified.
The mean gravidity of the study cohort was 2.0 (±1.3), with an average of 0.7 (±0.9) living children. Regarding pregnancy complications, 12.8% were diagnosed with pre-eclampsia, 8.2% with gestational hypertension, and 4.1% with gestational diabetes mellitus. No patients in the cohort were diagnosed with eclampsia. As for delivery method, most patients had a vaginal delivery (60.5%), followed by caesarean section (31.3%). The average gestational age at birth was 37.4 weeks (±2.3), and the mean 5-minute APGAR score was 8.4 (±1.5) (Table 1).
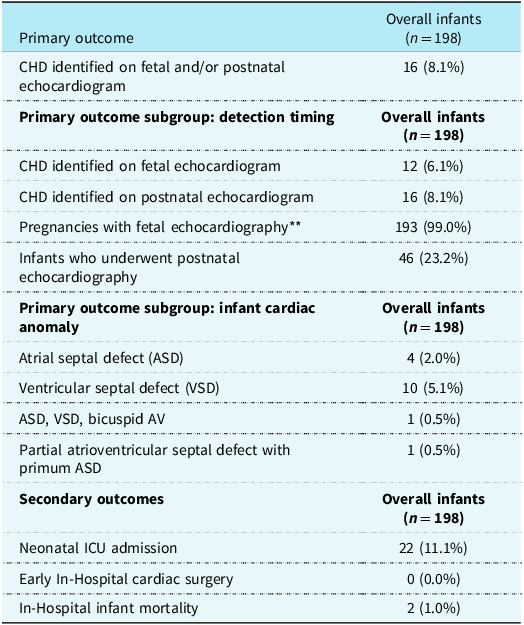
Fetal echocardiography was performed in 193 pregnancies (99.0%), and 46 (23.2%) infants underwent postnatal echocardiography. A total of 16 infants (8.1%) had abnormal postnatal echocardiogram findings (Table 2). Among the 16 infants with abnormal findings, 10 were diagnosed with isolated ventricular septal defect, four with isolated atrial septal defect, one infant with a constellation of atrial septal defect, ventricular septal defect, and bicuspid aortic valve, and one with partial atrioventricular septal defect (Table 3).
Primary and secondary infant outcomes of study cohort

This table presents primary and secondary outcomes related to congenital heart disease (CHD) among infants born to the study cohort. Of the cohort, 99.0% of mothers underwent fetal echocardiography, and 23.2% of infants underwent postnatal echocardiography. CHD was identified on fetal and/or postnatal echocardiograms in 8.1% of cases, with ventricular septal defect being the most common anomaly (5.1%). Secondary outcomes included neonatal ICU admission in 11.3% of cases, no instances of early in-hospital cardiac surgery, and two cases of in-hospital infant mortality (1.0%).
Data are mean (standard deviation) or no. (%), unless otherwise specified.
** Number of pregnancies = 195, number of infants = 198.
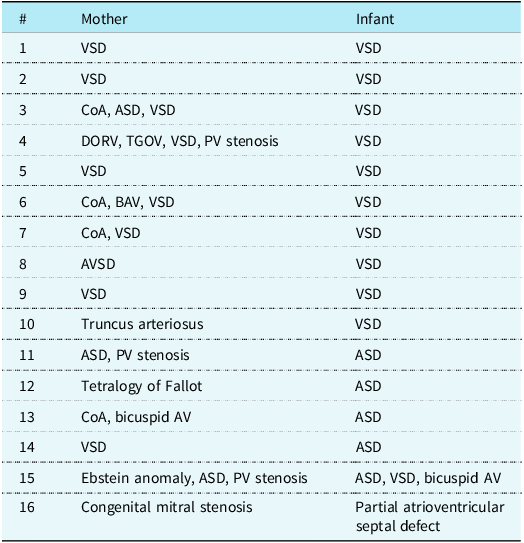
Mother and infant diagnostic parings of congenital heart defect

This table summarises the congenital heart defect diagnoses identified in affected mothers and their corresponding infants. Ventricular septal defect (VSD) was the most frequently observed lesion in both groups. Several mothers demonstrated complex or combined defects; however, their infants most commonly presented with isolated VSD or ASD.
ASD = atrial septal defect; CoA = coarctation of aorta; DORV = double outlet right ventricle; PV = pulmonary valve; TGOV = transposition of great vessels; VSD = ventricular septal defect.
Overall, 11.1% of infants required admission to the neonatal intensive care unit, primarily due to prematurity. Two infants experienced in-hospital mortality, including one infant with a ventricular septal defect who also had severe extracardiac abnormalities concerning for an underlying genetic syndrome. No infants in the cohort underwent early in-hospital cardiac surgery.
Discussion
This retrospective cohort study demonstrates an 8.1% incidence of CHD among offspring of mothers with CHD, with ventricular septal defect (both membranous and muscular) being the most common. None of the affected infants required early cardiac surgery, and one experienced in-hospital mortality related to severe extracardiac abnormalities. Although the incidence of CHD in this cohort was approximately eight times higher than in the general population, most cases were non-critical, and affected infants are expected to have a normal life expectancy. These findings provide meaningful insight into the incidence of CHD in offspring of affected mothers and may help inform prenatal counselling and shared decision making in pregnancy.
Contemporary data on the incidence of CHD in offspring of affected mothers are limited. While the incidence in the general population is approximately 1%, a large population-based study in Denmark of over 2.3 million births (1977–2011) reported a relative risk of 5.39 in offspring of mothers with CHD, compared to 3.04 for fathers, with higher risk for conotruncal and ventricular septal defects.Reference Oyen, Boyd, Carstensen, Sondergaard, Wohlfahrt and Melbye15 A population-based study in Canada of 2.2 million individuals (2002–2010) reported an adjusted odds ratio of 9.92 for CHD in offspring of mothers with CHD.Reference Liu, Joseph and Lisonkova13 Similarly, a single-centre study in Japan (1982–2016) found a threefold increased risk in children of mothers with CHD.Reference Yokouchi-Konishi, Yoshimatsu and Sawada14 We are not aware of any contemporary studies investigating the incidence of CHD in the offspring of mothers with CHD in the U.S.
The pathophysiology underlying CHD remains incompletely understood. Approximately 15% of cases are attributed to chromosomal aneuploidies (e.g., Down syndrome, trisomy 13, Turner syndrome, and 22q11.2 deletion syndrome), and fewer than 5% to monogenic disorders (e.g., Holt-Oram syndrome and Noonan syndrome).Reference Bouma and Mulder16,Reference Kerstjens-Frederiske and Mulder17 In non-syndromic cases, as in our cohort, the aetiology is less clear, though maternal factors such as obesity, diabetes, alcohol, and certain medications have been implicated.Reference Hedermann, Hedley and Thagaard18 Additionally, as genetic knowledge advances, it is possible that conditions currently classified as sporadic may become classified as genetic. In select cases, genetic testing may be considered through shared decision making. However, for most non-syndromic CHD, current testing yield remains low, and decisions should be individualised based on phenotype and family history.Reference Wilde, Semsarian and Marquez19,Reference Sumathi, Kristen, Ina and Ying20,Reference Cowan and Ware21
Our findings have important clinical relevance, as patients with CHD often seek guidance regarding the risk of CHD in their offspring. This study offers valuable data to guide risk-benefit discussions between clinicians and patients who are pregnant or planning pregnancy, particularly regarding genetic counselling and prenatal screening. While the incidence of CHD in infants of affected mothers is higher than in the general population, our findings may provide reassurance by demonstrating that most observed lesions portend favourable outcomes. As advances in the management of CHD continue to enhance long-term survival and quality of life, more women with CHD are reaching reproductive age, making it increasingly important to understand the risk of recurrence for reproductive planning. Larger, multicentre studies are warranted to better define estimates and assess additional genetic and environmental factors.
This study has several limitations. Firstly, CHD was identified primarily through fetal echocardiography, which has an estimated sensitivity of 77%, and not all neonates underwent a postnatal echocardiogram, potentially underestimating incidence.Reference Liu, Zhou and Feng6 However, our specialised maternal-fetal medicine group has previously demonstrated high sensitivity when conducting fetal echocardiograms, with critical CHDs missed rarely at a rate of 1.3%.Reference Cawyer, Kuper, Ausbeck, Sinkey and Owen22 Secondly, this study was conducted at a single academic centre, which may limit the generalisability of our findings to broader populations. Another potential limitation is misclassification of seemingly sporadic or isolated lesions, which may represent undetected syndromic or inherited conditions. Additionally, although we excluded patients with acquired heart disease, some congenital lesions diagnosed later in adulthood may present like acquired pathology and could have been inadvertently excluded. Finally, routine fetal echocardiography in our Cardio-Obstetrics Clinic may increase detection compared to the general population, introducing potential sampling bias.
Acknowledgements
We gratefully acknowledge the University of Alabama at Birmingham’s multidisciplinary Cardio-Obstetrics Program for their support in this study.
Financial support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
None.