Introduction
Cow mastitis, with a global incidence of up to 50% (Sharun et al. Reference Sharun, Dhama and Tiwari2021), is an inflammation of the mammary tissue triggered by pathogenic infection, mechanical injury, and environmental factors. Mastitis not only has the potential to harm the health of cows but can also affect the quality of milk, resulting in significant financial losses in the dairy sector (Li et al. Reference Li, Shao and Hou2021). Escherichia coli, a type of Gram-negative bacteria, is a prevalent cause of acute mastitis. Endotoxin lipopolysaccharide (LPS) of E. coli induces the secretion of inflammatory factors including TNF-α, IL-6, IL-1β from the breast gland (Ran et al. Reference Ran, Yan and Yang2020; Yin et al. Reference Yin, Xue and Dai2021). Although antibiotics can be used to treat mastitis, residues of antibiotics may compromise milk yield and quality and give rise to antibiotic resistance. Therefore, an alternative nutritional strategy is desired to reduce mastitis-induced inflammation in dairy cows.
Val-Pro-Pro (VPP) and Ile-Pro-Pro (IPP) are tripeptides obtained from milk that were originally recognized as inhibitors of angiotensin I-converting enzyme and exerted antihypertension activity (Nakumura et al. Reference Nakamura, Yamamoto and Sakai1995). VPP is located at fragments of 84-86 in β-casein, while IPP exists at fragments of 74–76 of β-casein and 108–110 of κ-casein (Li et al. Reference Li, Bu and Zheng2019). These two milk-derived tripeptides also exhibited anti-inflammation function both in vivo and in vitro (Sawada et al. Reference Sawada, Sakamoto and Toh2015; Chakrabarti and Wu, Reference Chakrabarti and Wu2015). For example, VPP and IPP were observed to prevent TNFα-induced loss of adiponectin through inhibiting the initiation of the nuclear factor kappa B (NF-κB) in 3T3-F442A murine pre-adipocytes. In vascular smooth muscle cells, VPP exerted anti-inflammatory activity via diminishing the initiation of extracellular signal-regulated kinases (ERK)1/2 to protect vascular function (Chakrabarti and Wu, Reference Chakrabarti and Wu2015; Chakrabarti et al. Reference Chakrabarti, Liao and Davidge2017). Additionally, VPP was found to decrease the activity of pro-inflammatory genes (i.e. TNFα and IL1β) in mice that were made obese by their diet (Sawada et al. Reference Sawada, Sakamoto and Toh2015). Although these findings support an anti-inflammatory effect of VPP and IPP, it is still unclear whether VPP and IPP can alleviate mastitis in dairy cows through their anti-inflammatory activity, and the specific mechanisms involved are yet to be determined. We hypothesize that VPP and IPP have the anti-inflammatory activity in mammary gland of dairy cow. Therefore, this study aimed to evaluate the role of VPP and IPP against LPS-induced inflammation in bovine mammary epithelial cells (BMECs) and a mouse model of mastitis. Our study will provide new insight in the application of milk-derived anti-inflammatory peptides in lactating cows.
Material and methods
Reagents
VPP and IPP were obtained from ChinaPeptides Co., Ltd (Shanghai, China). LPS (E.coli O111:B4), collagenase I and II were purchased from Sigma-Aldrich (St. Louis, MO, USA). Fetal bovine serum was obtained from Gibco (Grand Island, NY, USA). Enzyme-linked immunosorbent assay (ELISA) kits for TNF-α, IL-6, IL-1β, MCP-1, and IL-10 were purchased from Shanghai Yu Bo Biotech Co., Ltd (Shanghai, China). The primary antibodies β-tubulin were purchased from Beyotime Biotechnology (Shanghai, China). The primary antibodies ZO-1 and Occludin and the secondary antibodies used in this study including rabbit monoclonal antibodies and mouse monoclonal antibodies were purchased from Abcam (Cambridge, UK).
Cell culture and treatment
The procedures for the isolation and culture of primary BMECs were described as reported previously (Lin et al. Reference Lin, Li and Zou2018). In brief, the small pieces of mammary gland were incubated with 0.25% trypsin at 37℃ for 30 min in the dark, and then the digested sediments were incubated with 1 mg/mL of the mixture of collagenase I and II with the ratio of 1:1 at 37 ℃ for 4 h to separate BMECs and fibroblasts. The dispersed BMECs were cultured in α-Minimum Essential Medium (α-MEM, HyClone, Logan, UT) supplemented with 1% penicillin/streptomycin solution in a humidified incubator with 5% CO2 at 37℃.
BMECs were treated with various concentrations of LPS (0, 0.1, 1, 10, 100 μg/mL) for 24 h to assess the impact of LPS-induced cells on viability and inflammatory response.
Based on the above results of LPS concentration (1 μg/mL), cells were further pretreated with various concentrations of VPP or IPP (25, 50, 100 µM) for 2 h and then exposed to 1 μg/mL LPS for 24 h to assess the protective effect of VPP and IPP on inflammatory response, cell death and β-casein expression.
Cell viability assay
Cell viability assay was performed using the Cell Counting Kit-8 (CCK-8) kit (Beyotime, Shanghai, China) by the instructions provided by the manufacturer. In summary, 96-well culture plates were seeded with 5000 BMECs and incubated in an incubator that was humidified with 5% CO2 at 37℃ for 24 h. Different concentrations of LPS were added for 24 h in a trial. Various concentrations of VPP or IPP were pretreated for 2 h and then activated with LPS (1 μg/mL) for 24 h in a separate experiment. Afterward, a volume of 10 µL of CCK-8 was introduced into each well and allowed to incubate for a duration of 2 h. Subsequently, the absorbance (optical density [OD]) was quantified at a wavelength of 450 nm using the BioTek SYNERGY HTX multi-mode microplate reader (BioTek, USA).
Cell apoptosis assay
The apoptosis cells were detected with Annexin V-FITC/PI apoptosis detection kit (BestBio, Nanjing, China). The cells were harvested using trypsin solution without EDTA and then subjected to centrifugation at a speed of 1500 revolutions per minute for 5 min at ambient temperature. After being rinsed twice with PBS, the cells were resuspended in 300 μL of 1× binding buffer. The cells were treated with 5 μL of Annexin V-FITC for 15 min and thereafter exposed to 10 μL of PI for another 15 min at room temperature in the absence of light. Ultimately, the levels of cell apoptosis were examined using flow cytometry (Becton Dickinson Company, NJ, USA).
Enzyme-linked immunosorbent assay
The protein levels of pro-inflammatory factors (TNF-α, IL-6, IL-1β), chemokine (MCP-1), anti-inflammatory factor (IL-10) in BMECs were determined using an ELISA kit following the instructions provided by the manufacturer (Shanghai Yu Bo Biotech Co., Ltd, Shanghai, China).
Western blot analysis
Proteins were recovered from BMECs utilizing a radioimmunoprecipitation assay lysis buffer including a proteinase inhibitor (PMSF, Beyotime). Protein concentration was determined by BCA Protein Assay Kit. Proteins, with a quantity of 20 μg, were isolated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then deposited onto the polyvinylidene difluoride (PVDF) membranes. The membranes were obstructed with blocking buffer for 2 h. Then, the PVDF membrane was exposed to the primary antibodies (β-casein or β-tubulin) at 4℃ overnight followed by exposure to the secondary antibody (goat anti-rabbit (1:5000) antibody) for 1 h at room temperature. The presence of immunopositive bands was detected using enhanced chemiluminescence (Bio-Rad, CA, USA).
Mice and treatments
BALB/c mice (8–9 weeks old, 20–25 g weight) were obtained from the Animal Experimental Center of Zhejiang Chinese Medical University (Hangzhou, China) and distributed in a small cage with two females and one male kept under a standard condition (12 h light-dark cycle, 22–23℃). Individual cages were provided for pregnant female mice, who were then exposed to various treatments. Forty-two nursing mice were allocated randomly into seven groups (n = 6 per group): Control, LPS (0.2 mg/mL, 50 µL), LPS + VPP (300, 600 µM/kg/d), LPS + IPP (300, 600 µM/kg/d), and LPS + dexamethasone (DEX) (5 mg/kg/d). The dosages of LPS, VPP or IPP were based on the previous study (Liu et al. Reference Liu, Yu and BU2023; Ran et al. Reference Ran, Liu and Fu2022). Prior to receiving LPS treatment on days 5–9 of lactation, the mice were gavaged VPP, IPP, and DEX for 5 days, whereas control and LPS-induced groups were gavaged with an equivalent amount of sterile saline. On day 9 of lactation, 1 h after gavage of VPP, IPP, DEX or saline, the mice were administered LPS by injecting it into their fourth mammary ducts using a microsyringe equipped with a 32-G needle. Twenty-four hours after the LPS injection, all of the mice were euthanized, and samples of mammary tissue were obtained for further studies.
Hematoxylin−eosin staining
The mammary gland tissues were immersed in a solution of 4% paraformaldehyde for a duration exceeding 24 h to fix them. The tissues were fixed in paraffin and then cut into pieces that were 5 μm thick. To assess histological alterations, the tissues were stained with haematoxylin–eosin (H&E) and examined using a microscope (NIKON ECLIPSE CI, Nikon, Tokyo Metropolis, Japan).
RNA extraction and real-time PCR analysis
Total RNA was extracted by the RNA Pure Kit (Accurate Biotechnology, Changsha, China) according to the manufacturer’s instructions. The cDNA was transcribed using the PrimeScript RT Reagent Kit (Accurate Biotechnology, Changsha, China). Quantitative real-time PCR was performed using SYBR ® Green Pro Taq HS (Accurate Biotechnology, Changsha, China) by a StepOnePlusTM real-time PCR detection system (Applied Biosystems, Waltham, MA, USA). Given the robust stability of β-actin (ACTB) gene expression and the absence of differential expression in the RNA-seq analysis, data normalization was performed using β-actin as an internal reference gene, and the relative gene expression values were determined using the 2–△△CT technique (Wan et al. Reference Wan, Zhang and Yang2021). The primer sequences employed in this investigation are enumerated in Table 1, and RNA quality was shown in Table 2.
Primers Used for qRT-PCR

RNA quality assay

Immunofluorescence analysis
The mammary gland samples were immersed in 4% paraformaldehyde for fixation and subsequently fixed in paraffin. The mammary gland that was embedded was sliced into paraffin sections that were 3 μm thick. The sections of the mammary gland were treated with xylene to remove the paraffin and prepare them for antigen retrieval. The sections of the mammary gland were treated with 5% donkey serum at room temperature for 1 h, followed by incubation with primary antibodies (ZO-1 and Occludin) at 4℃ overnight. Subsequently, slices of the mammary gland were stained using a goat anti-rabbit HRP-linked secondary antibody at room temperature for 1 h. Ultimately, sections of the mammary gland were stained with DAPI as a counterstain, and the resulting images were examined using a fluorescence microscope (NIKON ECLIPSE CI, Nikon, Tokyo Metropolis, Japan).
Transcriptome analysis
The total RNA of mammary gland samples was isolated using Trizol extraction methods. The purity and concentration of the extracted RNA samples were determined by a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). The library was constructed using the NEBNext® Ultra™ RNA Library Prep Kit for Illumina® (NEB, USA, Catalog #: E7530L) following the manufacturer’s instructions. The samples were sequenced on the illumina novaseq 6000 (illumina, CA, USA).
The clean data were obtained by eliminating the linker sequence and low-quality sequence from raw data, which was assessed for quality using FASTQC. Differential expression analysis was performed using DESeq2 (Bioconductor version 1.6.3), and gene expression with the threshold of Padj < 0.05 (followed an FDR correction) and the absolute value of |log2 fold change (FC)| ≥ 1 among two groups were regarded as the differentially expressed genes (DEGs). The selected DEGs were confirmed by RT-PCR.
Gene Ontology analysis encompassing biological process, molecular function, and cellular component was conducted by GOSeq (v1.34.1) with a significant Adj. p value < 0.05. KEGG (Kyoto Encyclopedia of Genes and Genomes) was implemented by clusterProfiler 3.8.1. The PPI networks of the DEGs were performed with STRING database (http://string-db.org/) with high confidence (0.7) and drawn with Cytoscape V3.9.1 software.
Molecular docking
The 3D structure of fibronectin was acquired from Uniprot (https://www.uniprot.org/), and the 3D structure of VPP and IPP were obtained from Pubchem (https://pubchem.ncbi.nlm.nih.gov/). The molecular docking of VPP or IPP with fibronectin was simulated using Autodock 4, and the docking time was 10. Finally, the results of protein-ligand complex were displayed using PyMOL Molecular Graphics System (Version 2.0 Schrodinger, LLC.)
Molecular dynamics simulation
The stability of VPP–fibronectin and IPP–fibronectin complexes were assessed with Gromacs version 2022.2 software package with Amber99sb-ildn force filed. VPP–fibronectin and IPP–fibronectin complexes were inserted into a regularly shaped cubic container, and a TIP3P water model was included in the container to dissolve the complexes. To neutralize the charge, a sufficient amount of Na+ was introduced into the system. Subsequently, the simulation underwent energy minimization. The long-range interactions were evaluated using the Particle Mesh Ewald approach. A 12 Å cutoff was employed to handle van der Waals interactions. The Linear Constraint Solver (LINCS) method was employed to restrict all hydrogen-related bonds. The Canonical Ensemble (NVT) and Isothermal-Isobaric Ensemble (NPT) ensembles were used to achieve thermal equilibrium in the systems for 100 ps. Temperature and pressure coupling in Molecular Dynamics (MD) ensembles were accomplished by employing a V-rescale thermostat and a Parrinello–Rahman barostat, respectively.
Statistical analysis
All data are expressed as the mean ± SEM. Statistical significance was indicated by asterisks and hash symbols (*p < 0.05, ** p < 0.01; #p < 0.05, ##p < 0.01). For the cell trials, the differences in the cell viability, apoptosis, the expression of inflammatory cytokines and β-casein between the control group, LPS group, and different concentrations of VPP and IPP groups were compared. For the animal trials, the differences in the expression of inflammatory cytokines and tight junction proteins in the control group, LPS group, VPP and IPP groups with concentrations of 300 and 600 µM/kg were compared. In both cell and animal trials, when the data test was normally distributed, the distinctions among the various experimental groups were examined using a one-way ANOVA and Student–Newman–Keuls multiple comparison test in SPSS (version20, IBM Corp, Armonk, NY, USA). On the other hand, when the data were not normally distributed, the differences were examined using the Kruskal–Wallies test and Dunn’s multiple comparison test.
Results
Effects of VPP and IPP on alleviating inflammatory response in LPS-induced BMECs
To investigate the effect of LPS on BMECs, the cells were treated with different concentrations of LPS (0–100 μg/mL) for 24 h. A dose-dependent inhibition of cell survival by LPS was observed (Fig. 1A). The expression of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) and chemokine (MCP-1) increased after treatment with LPS in a dose-dependent manner, while the expression of anti-inflammatory cytokine (IL-10) was decreased when the treatment concentration of LPS was more than 1 μg/mL (Fig. 1B–F). Given the inflammatory response, 1 μg/mL of LPS was selected to assess the anti-inflammatory effects of VPP and IPP in BMECs.
Both VPP and IPP ameliorated inflammatory response in LPS-induced bmecs. BMECs were treated with different dosages of LPS (0, 0.1, 1, 10 and 100 µg/ml) for 24 h. (A) Cell viability was determined with a CCK-8 kit, and the concentrations of (B) TNF-α, (C) Il-1β, (D) IL-6, (E) MCP-1 and (F) IL-10 in the cell medium were estimated by ELISA. bmecs were pretreated with different concentrations of VPP and IPP (25, 50, 100 µm) for 2 h following by LPS (1 µg/ml) challenge for 24 h. (G) Effects of VPP and IPP on LPS-induced cell viability. the concentration of (H) TNF-α, (I) Il-1β, (J) IL-6, (K) MCP-1 and (L) IL-10 within LPS-stimulated MAC-T cells were analyzed by ELISA. Data are represented as mean ± SEM (n = 3). *p < 0.05 and **p < 0.01 compared with the untreated group; #p < 0.05 and ##p < 0.01 compared with the LPS group.

Supplementation of VPP or IPP at concentrations of 50 and 100 μg/mL significantly reversed the decrease in BMECs viability induced by LPS. Compared with LPS stimulation, pretreatment with both VPP and IPP significantly inhibited the expression of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) and chemokine (MCP-1), while enhancing the expression of anti-inflammatory cytokine (IL-10) (Fig. 1H–L).
Effects of VPP and IPP on death and β-casein expression in LPS-induced BMECs
As shown in Fig. 2A–B, VPP and IPP downregulated the increased cell apoptosis stimulated by LPS treatment at concentrations from 25 to 100 μM by 7.77% and 8.53%, 16.93% and 18.65%, 23% and 24.4%, respectively. Furthermore, western blot analysis revealed that VPP and IPP pretreatment at a concentration of 50 μM rescued the expression of β-casein in LPS-induced BMECs (Fig. 2C). The accumulating results suggest an anti-inflammatory effect of VPP and IPP on LPS-induced BMECs.
Effects of VPP and IPP on death and β-casein expression in LPS-induced BMECs. BMECs were pretreated with different concentrations of VPP and IPP (25, 50, 100 µm) for 2 h, following by LPS (1 µg/ml) challenge for 24 h. (A) Flow cytometry analysis of annexin V/FITC/PI staining cells. (B) Quantification of the total cell apoptotic rates. (C) The protein levels of β-casein were measured by western blot and quantified by comparison with β-tubulin. Data are represented as mean ± SEM (n = 3). *p < 0.05 and **p < 0.01 compared with the control group; #p < 0.05 and ##p < 0.01 compared with the LPS group.

Effects of VPP and IPP on alleviating LPS-induced mammary inflammatory response in mice
As shown in Fig. 3B, H&E staining results showed hyperemia oedema and a high level of neutrophil infiltration in the mammary tissue acini in the LPS group compared to the control. Nevertheless, the gavage of VPP, IPP and the positive control (DEX) considerably diminished the degenerative alterations of these mammary gland tissues in the mice (Fig. 3B). Moreover, VPP, IPP, and DEX significantly inhibited the expression of pro-inflammatory cytokines (TNFα, IL1β, IL6) induced by LPS (Fig. 3C–E).
VPP and IPP alleviate LPS-induced mammary inflammatory response in mice. (A) Flow chart of mice experiment. (B) Representative images of mammary gland stained with H&E in mice (bar = 50 μm). (C–E) Qrt-pcr analysis of mammary gland tissue from 3 mice for each group to determine expression of inflammatory factors. data are represented as mean ± SEM (n = 3). *p < 0.05 and **p < 0.01 compared with the control group; #p < 0.05 and ##p < 0.01 compared with the LPS group.

Effects of VPP and IPP on maintaining the integrity of the blood–milk barrier
As immunofluorescence results shown in Fig. 4A, VPP and IPP significantly reversed the reduced expression of occludin and ZO-1, as well as alleviated the disorder of tissue distribution stimulated by LPS in mice. These phenomena were also validated by the mRNA expression of occludin and ZO1 treated with VPP and IPP (Fig. 4B–C), suggesting that VPP and IPP could maintain the integrity of the junction complex by promoting the expression of tight junction proteins.
VPP and IPP maintain the integrity of the blood–milk barrier. (A) Immunofluorescence staining of ZO-1 and occludin (bar = 50 μm). (B) qRT-PCR analysis of mammary gland tissue from 3 mice for each group to determine expression of ZO1 and occludin. Data are represented as mean ± SEM (n = 3). *p < 0.05 and **p < 0.01 compared with the control group; #p < 0.05 and ##p < 0.01 compared with the LPS group.

Transcriptome analysis of DEGs modified by VPP or IPP in the mammary gland of mice
RNA-seq analysis showed that the mammary transcriptome profiles of the control, LPS, LPS with addition of VPP (600 µM/kg) or IPP (600 µM/kg) groups were distinct from different treatments and highly reproducible among each groups (Figure 5A and 5C, 6A and 6C). A total of 741 DEGs including 156 upregulated genes and 585 downregulated genes were identified in the VPP groups, while 797 DEGs involving in 280 upregulated genes and 517 downregulated genes were identified in the IPP groups compared to the LPS group (Figs 5B and 6B). The DEGs in the VPP group were mainly enriched in the inflammation-related pathways including PI3K-AKT and MAPK pathways. These inflammation-related pathways might be responsible for the protective effect of VPP in this study (Fig. 5D). The KEGG enriched in the IPP group was similar to the VPP vs. LPS group (Fig. 6D). In addition, the qPCR results indicated the changes in the expression of selected DEGs related to PI3K-AKT signaling pathways (Fig. 5E–F and Fig. 6E–F).
Transcriptome analysis of overlapping genes modified by VPP in the mammary gland of mice. (A) Principal component analysis (PCA) plot. (B) Volcano diagram. (C) Heatmap (top 30 genes with significant differences). (D) KEGG enrichment analysis. (E) DEGs enriched in PI3K/AKT signaling pathway. (F) qRT-PCR analysis. Abbreviation: CK, control.

Transcriptome analysis of overlapping genes modified by IPP in the mammary gland of mice. (A) Principal component analysis (PCA) plot. (B) Volcano diagram. (C) Heatmap (top 30 genes with significant differences). (D) KEGG enrichment analysis. (E) DEGs enriched in PI3K/AKT signaling pathway. (F) qRT-PCR analysis. Abbreviation: CK, control.

Both VPP and IPP alleviated inflammation by interacting with fibronectin in PI3K-AKT signaling pathway
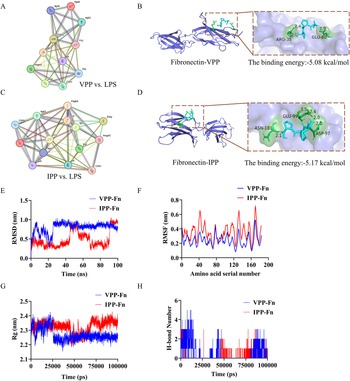
The genes enriched in PI3K-AKT signaling pathway were analyzed by the PPI network. By accessing the degree centrality analysis of the PPI network, 11 hub genes were obtained in the VPP vs LPS group, 14 hub genes were obtained in the IPP vs LPS group (degree ≥ 10)(Fig. 7A, C). Of these, fibronectin ranked highest in both VPP and IPP compared to LPS groups. Molecular docking predicted that hydrogen bonds existed between VPP and fibronectin at the ARG-35 and GLU-80 amino acid residues with a total energy of –5.08 kcal/mol; and hydrogen bonds existed between IPP and fibronectin at the ASP-35, ASN-1810 and GLU-99 amino acid residues with a total energy of −5.17 kcal/mol (Fig. 7B, D). Furthermore, the stability of VPP–fibronectin and IPP–fibronectin complexes was evaluated to predict the interaction between VPP/IPP and fibronectin. As shown in Fig. 7E, the root–mean–square deviation (RMSD) of VPP–fibronectin reached equilibrium after 30 ns, while IPP–fibronectin maintained equilibrium at 70 ns. The average root–mean–square fluctuation (RMSF) values of VPP−fibronectin and IPP–fibronectin complexes were 0.24 ± 0.07 and 0.35 ± 0.11 nm, respectively (Fig. 7F). The radius of gyration (Rg) values of VPP–fibronectin (2.27 ± 0.04 nm) was lower than IPP–fibronectin (2.32 ± 0.04 nm) (Fig. 7G). As shown in Fig. 7H, the number of hydrogen bonds formed in VPP–bronectin and IPP–fibronectin complexes were 0–4 and 0–3, respectively. The results of molecular dynamics simulations suggested that both VPP–fibronectin and IPP–fibronectin complexes have stable global structure and tight binding.
PPI network and molecular docking combined with molecular dynamics simulation in the VPP–fibronectin and IPP–fibronectin complexes. (A),(C) PPI network of the DEGs with a confidence score >0.7 and degree >9 in VPP and IPP compared to LPS. (B),(D) The molecular docking simulation of the binding pattern of VPP with fibronectin and IPP with fibronectin. (E) RMSD change profiles of the VPP–fibronectin and IPP–fibronectin complexes systems over time. (F) RMSF values of binding site residues in the VPP–fibronectin and IPP–fibronectin complex systems. (G) Gyration radius of the VPP–fibronectin and IPP–fibronectin complexes systems. (H) Changes in hydrogen bond number over time in the VPP–fibronectin and IPP–fibronectin complexes systems.

Discussion
Gram-negative pathogens such as Escherichia coli are the main cause of mastitis in dairy cows (Yang et al. Reference Yang, Yue and Li2020). LPS, a component of Gram-negative bacteria’s cell wall, causes an inflammatory response during bacterial infection (Fang et al. Reference Fang, Zou and Yang2021). Following an LPS challenge, the proinflammatory mediators (i.e. TNF-α, IL-6, IL-1β) are released (Huang et al. Reference Huang, Teng and Liu2022; Ashraf and Imran, Reference Ashraf and Imran2020). We propose that bioactive tripeptides VPP and IPP originating from milk casein may act as an efficient anti-inflammatory agent to prevent mastitis. In this study, BMECs and a mastitis mouse model were applied to investigate the effect of milk-derived tripeptides VPP and IPP on LPS-stimulated inflammation. First, an inflammatory model via LPS-stimulated BMECs was established as an in vitro model of cow mastitis (Jiang et al. Reference Jiang, Lv and Huang2022). LPS could decrease cell viability and the generation of anti-inflammatory cytokine IL-10, while increasing the generation of the pro-inflammatory mediators TNF-α, IL-6, IL-1β and the chemokine MCP-1. Both VPP and IPP reversed the negative impact on cell viability and inflammatory cytokines stimulated by LPS. Besides, VPP and IPP also reduced expression of proinflammatory cytokines TNFα, IL1β, and IL6 in LPS-stimulated mice, which additionally confirms their anti-inflammatory effect in mastitis. Similar results were found by Song et al. (Reference Song, Lv and Zhang2020a, Reference Song, M. and Sun2020b), demonstrating that VPP and IPP reduced the protein contents of proinflammatory mediators (i.e. IL-6, IL-1β) and chemokine MCP-1 to modulate the endothelial dysfunction caused by extracellular vesicles (EV). The anti-inflammatory function of VPP and IPP was thought to be attributed to their functional amino acid composition, such as Val and Ile. Val has been shown to exert anti-inflammatory properties via decreasing the secretion of inflammatory molecules (i.e. IL1β, IL6, ICAM1, and TNFα) in the adipose tissues and liver (Zhou et al. Reference Zhou, Zhang and Shen2023b). Ile has been found to exert an inhibitory effect on inflammation by modulation IL-8 generation in Caco-2 cells when induced by LPS (Garcia et al. Reference Garcia, Makiyama and Sampaio2024). Val and Ile released from VPP and IPP digestion may contribute to anti-inflammatory effect (Gart et al. Reference Gart, van Duyvenvoorde W. and Caspers2022). The inflammation in mammary gland caused by LPS also could induce neutrophil infiltration in mice (Yu et al. Reference Yu, Liu and Yu2020). The decreased neutrophil infiltration of the mammary tissue in VPP and IPP group is another reason for their anti-inflammatory effects.
The β-casein is one of major components of lactogenic protein synthesized in the udder in dairy cows (Lv et al. Reference Lv, Liu and Yang2020; Rehan et al. Reference Rehan, Ahemad and Gupta2019). Previous studies have demonstrated that the β-casein secretion was reduced in LPS-stimulated cow mastitis (Silanikove et al. Reference Silanikove, Rauch-Cohen and Shapiro2012). Consistent with previous studies, our findings demonstrated that LPS decreased expression of β-casein in BMECs, while VPP and IPP supplementation reversed this phenomenon. These results revealed that VPP and IPP protected the milk β-casein synthesis against the LPS-induced inflammation in mammary gland of BMECs. Moreover, the blood–milk barrier is a natural shield against external pathogens during lactation (Tsugami et al. Reference Tsugami, Matsunaga and Suzuki2017), in which key components of mammary epithelial cells are the tight junction proteins, including occludin and ZO-1 (Wellnitz et al. Reference Wellnitz, Wall and Saudenova2014). When stimulated by external pathogens such as LPS, the completeness of blood–milk barrier was disrupted in reaction to inflammation (Kobayashi et al. Reference Kobayashi, Oyama and Numata2013). This disruption may allow more pathogens to penetrate the breast tissue from the blood to further exacerbate inflammation (Wang et al. Reference Wang, Wei and Zhang2017). In this study, VPP and IPP could reverse the reduced level of tight junction proteins occludin and ZO1 induced by LPS, suggesting that these two peptides can maintain the integrity of blood–milk barrier and subsequently control the exacerbation of mastitis (Guo et al. Reference Guo, Liu and Yin2019).
To investigate the potential molecular mechanism, RNA-seq transcriptome was employed to screen the key pathway involved in the positive effect of VPP and IPP on mammary inflammation. We found that both VPP and IPP could exert protective effects by regulating PI3K/AKT and MAPK signaling pathways in mice stimulated by LPS. The pathway’s upstream target, PI3K/AKT, is essential for inflammation (Manning and Toker, Reference Manning and Toker2017). Different external signals (such as TLRs, cytokines, and growth factors), activate the PI3K/AKT pathway, which subsequently specifically causes the release of inflammatory mediators (i.e. TNF-α, IL-1β, IL-6, etc.) and eventually enhances inflammation (Li et al. Reference Li, Ran and Zeng2023, Reference Li, Xi and Wang2018). Hence, PI3K/AKT might be an important route in the development of mastitis. By stimulating the transcription of pro-inflammatory genes and boosting the phosphorylation of AKT, LPS stimulation can activate the PI3K/AKT signaling pathway (Chen et al. Reference Chen, Zheng and Zhang2018). MAPK signaling pathway, one of the principal pathways regulating inflammation, is composed of extracellular signal-regulated kinases (ERK1/2), c-JUN N-terminal kinases (JNK1/2) and p38. These components in MAPK pathway also can be phosphorylated by LPS stimulation and stimulate the transcription of a variety of inflammatory mediators including c-JUN, AP-1, etc (Ronkina and Gaestel, Reference Ronkina and Gaestel2022). In this study, both VPP and IPP inhibited expression of key genes of PI3K-ATK and MAPK pathways, which might play a critical part in alleviating mastitis.
Fibronectin encoded by the Fn1 gene is a part of the extracellular matrix and also is critical in signal transduction between the extracellular matrix and cell surface proteins (Liu et al. Reference Liu, Hong and Ma2024). In the current study, fibronectin is enriched in the PI3K/AKT signaling pathway which could affect proliferation of cell, inflammation, etc (Han et al. Reference Han, Khuri and Roman2006). Molecular docking along with the results of molecular dynamics simulations suggested that both VPP and IPP may form stable complexes with fibronectin, providing further insight into the possible function of fibronectin in the VPP or IPP. Molecular docking showed that the binding energy of VPP–fibronectin and IPP–fibronectin were lower than −5 kcal/mol, suggesting well-being binding activity (Sohretoglu, Reference Sohretoglu, Sari and Soral2018). The RMSD, RMSF, Rg, and the number of hydrogen bonds of VPP–fibronectin and IPP–fibronectin complexes were the key indicators to evaluate the stability between VPP/IPP and fibronectin (Jia et al. Reference Jia, Yang and Liao2024). During simulations, RMSD can reflect the stability of peptides relevant to the protein (Sariyer et al. Reference Sariyer, Kocer and Danis2021). Global structure is more stable when the RMSD is lower. The degree of variation in the amino acid residues is shown by RMSF values (Zhou et al. Reference Zhou, Bie and Li2023a). Rg may describe the density of protein structure. A lower Rg value means a more stable protein structure (Chen et al. Reference Chen, Wu and Zhang2020). Hydrogen bonding is an important non-covalent structural force that affects the stability of protein structures (Sohretoglu et al. Reference Sohretoglu, Sari and Soral2018). Molecular docking along with the results of molecular dynamics simulations showed that VPP–fibronectin and IPP–fibronectin complexes had good binding activity. Furthermore, transcriptome results showed that VPP and IPP downregulated the expression of fibronectin which was confirmed in the qPCR results, suggesting VPP and IPP may inhibit PI3K/AKT signaling pathway through regulating fibronectin. It is reported that fibronectin activates IL-17A-mediated inflammatory pathway in EV recipient cells, promoting the secretion of proinflammatory cytokines such as IL-6 and TNF-α (Sriwastva et al. Reference Sriwastva, Teng and Mu2023). Fibronectin can stimulate the phosphorylation of AKT, the expression of which positively correlates with the activation or suppression of the PI3K/AKT pathway, which then regulates inflammation (Yang et al. Reference Yang, Shen and Zhu2024). In this study, fibronectin may be a potential target for VPP and IPP to reduce mammary inflammation.
Conclusion
In summary, our study suggested that milk-derived bioactive peptides VPP and IPP had a protective role against LPS-induced inflammation in BMECs and a mastitis mouse model. This anti-inflammatory activity was achieved by regulating PI3K/AKT and fibronectin may be the key target of both VPP and IPP. Furthermore, VPP and IPP also ameliorated inflammation through enhancing the blood–milk barrier in mastitis mice. These results provide innovative insights into the deeper mechanisms underlying protective effect of VPP and IPP on mastitis inflammation.
Author Contributions
Meifei Zhu and Jingyan Li contributed to the experimental design and project management; Meifei Zhu, Ruike Wei, Jingyan Li, Jiayi Bao, Lin Wei, and Xinyu Yu contributed to sample collection; Meifei Zhu and Ruike Wei analyzed data; Meifei Zhu wrote the paper; Meifei Zhu, Shanshan Li, Zheng Zhou, and Fuliang Hu revised the paper. All authors have read and approved the final manuscript.
Financial Support
This study was financially supported by National Natural Science Foundation of China (31972588), the earmarked fund for Modern Agroindustry Technology Research System from the Ministry of Agriculture of China (CARS-44), and Michigan Alliance for Animal Agriculture (AA-22-0073).
Conflicts of Interest
The authors declare no competing financial interest.
Ethical Standards
The Animal Care and Use Committee of Zhejiang Chinese Medical University (No. 86469) approved all mouse studies conducted in this work.