Introduction
The Western Antarctic Peninsula (WAP) region is an area of particular concern in terms of both increasing levels of human activity (research/governmental operations, fisheries and tourism) and climate change impacts (Turner et al. Reference Turner, Bindschadler, Convey, Di Prisco, Fahrbach and Gutt2009, Reference Turner, Barrand, Bracegirdle, Convey, Hodgson and Jarvis2014, Chown et al. Reference Chown, Leihy, Naish, Brooks, Convey and Henley2022, Siegert et al. Reference Siegert, Bentley, Atkinson, Bracegirdle, Convey and Davies2023). These affect local terrestrial biodiversity both directly and indirectly (Convey & Peck Reference Convey and Peck2019, Lee et al. Reference Lee, Waterman, Shaw, Bergstrom, Lynch, Wall and Robinson2022). Furthermore, marine vertebrates, specifically their on-land breeding colonies or individual nest locations and haul-out/moult areas, are key drivers of terrestrial biodiversity as a result of their contributions to nutrient transfer, while changes in their distribution or size are predicted to lead to both increases and decreases in that biodiversity and/or biomass (Bokhorst et al. Reference Bokhorst, Convey and Aerts2019). Together, these factors emphasize the need for robust knowledge regarding the baseline biodiversity of this region and the development and application of effective conservation measures.
There is a clear recognition of the current inadequacy and lack of representativeness of the existing Antarctic protected area system (e.g. Shaw et al. Reference Shaw, Terauds, Riddle, Possingham and Chown2014, Hughes et al. Reference Hughes, Ireland, Convey and Fleming2015, Coetzee et al. Reference Coetzee, Convey and Chown2017), which serves to emphasize the case for proactive protection within this area. Under the established system for justifying area protection (Antarctic Specially Protected Areas; ASPAs) as defined in the Environmental Protocol to the Antarctic Treaty, one of the ‘values’ that is explicitly recognized as a justification is to protect the ‘type or only known locality of occurrence’ of a given species, while other values include protection of ‘representative’ and ‘outstanding’ ecosystems (Hughes & Convey Reference Hughes and Convey2010). This framework thus highlights the importance of identifying and documenting species with restricted distributions, the most extreme cases of which are rare species. To understand how such values manifest themselves at the regional level, it is necessary to have a clear understanding of existing botanical data. Mosses in the WAP, including in the Argentine Islands-Kyiv Peninsula (AI-KP) region, have been studied since the late nineteenth and early twentieth centuries (Smith & Corner Reference Smith and Corner1973, Ochyra et al. Reference Ochyra, Bednarek-Ochyra and Smith2008a,Reference Ochyra, Smith and Bednarek-Ochyrab, Parnikoza et al. Reference Parnikoza, Convey, Dykyy, Trokhymets, Milinevsky, Inozemtseva and Kozeretska2009, Reference Parnikoza, Berezkina, Moiseyenko, Malanchuk and Kunakh2018, Ivanets et al. Reference Ivanets, Wierzgoń, Yevchun and Parnikoza2023). Early botanical work in the Maritime Antarctic provided the first ecological descriptions and classifications of vegetation communities (de Gerlache Reference De Gerlache1904, Cardot & Hariot Reference Cardot and Hariot1907, Charcot Reference Charcot1908, Rymill Reference Rymill1938, Greene Reference Greene1967, Smith Reference Smith1972, Reference Smith and Laws1984, Longton Reference Longton1988). Local vegetation communities in the vicinity of the Argentine Islands were first comprehensively described by Smith & Corner (Reference Smith and Corner1973) and later by Parnikoza et al. (Reference Parnikoza, Berezkina, Moiseyenko, Malanchuk and Kunakh2018). In total, 66 moss species and one variety are currently known from the WAP, including its offshore islands (Ochyra et al. Reference Ochyra, Smith and Bednarek-Ochyra2008b), of which 51 have been reported from the Graham Coast (Ivanets et al. Reference Ivanets, Wierzgoń, Yevchun and Parnikoza2023) and the AI-KP region (Ochyra et al. Reference Ochyra, Smith and Bednarek-Ochyra2008b), highlighting the latter’s botanical importance within the WAP. However, although data on the moss community composition and distribution in the WAP have accumulated over more than a century and are now widely available through online resources such as the Global Biodiversity Information Facility (GBIF) and the Antarctic Plant Database (international herbarium code AAS) maintained by the British Antarctic Survey (BAS), biodiversity data availability and spatial coverage are still patchy and far from complete (Chown & Convey Reference Chown and Convey2007, Fretwell et al. Reference Fretwell, Convey, Fleming, Peat and Hughes2010, Pertierra et al. Reference Pertierra, Varliero, Barbosa, Biersma, Convey and Chown2024).
Surveying the distribution of moss species at a defined and logistically practicable scale, particularly for those that can be considered under some definition of rarity, can provide an important baseline against which to assess future changes and any requirements for protective measures. This is particularly pertinent considering the combination of ongoing climate change and the increasing threat of the anthropogenic introduction of non-native species, as parts of the WAP region are among the most visited in Antarctica (Lee et al. Reference Lee, Waterman, Shaw, Bergstrom, Lynch, Wall and Robinson2022). Such an assessment has not yet been conducted for the AI-KP region, or indeed for comparable regional scales elsewhere in Antarctica. Previously, concepts of rarity have only been applied at the most general ‘whole-Antarctic’ scale by Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b). Research supporting a definition of moss rarity in Antarctica is, therefore, necessary to provide a vital underpinning for the ongoing development of effective conservation strategies within the Antarctic Treaty System.
As protection is typically granted only for specific types of rarity (IUCN 2012), and the concept of rarity in mosses encompasses not only the number of known occurrence locations but also aspects of geographical range, habitat specificity and consistently low population sizes (Longton & Hedderson Reference Longton and Hedderson2000), the Antarctic region presents a particular challenge. For instance, Anderson et al. (Reference Anderson, Chown and Leihy2024) noted that ‘the geographic range sizes and turnover of mosses in Antarctica may differ from the patterns seen in other continental systems generally, making generalities more difficult to apply to the continent’, which makes it difficult to apply generally used approaches to Antarctic mosses. In Antarctica, moss distributions are characterized by high species turnover over short distances, small and often disjunct geographical ranges and weak correspondences with broad-scale environmental gradients, largely due to the extreme fragmentation of ice-free habitats, limited dispersal and post-glacial history (Peat et al. Reference Peat, Clarke and Convey2007, Ochyra et al. Reference Ochyra, Smith and Bednarek-Ochyra2008b, Anderson et al. Reference Anderson, Chown and Leihy2024). This is further complicated by the fact that there are currently no formally adopted quantitative thresholds for Antarctic moss rarity. In this context, the term ‘rarity’ does not necessarily imply threat at the continental scale but reflects a combination of restricted distributions, fragmented habitats and limited occurrence data, all of which are directly relevant at regional scales. Very few studies have explicitly addressed the definition or spatial patterns of rarity in Antarctic mosses (Ochyra & Singh Reference Ochyra and Singh2008, Ochyra et al. Reference Ochyra, Smith and Bednarek-Ochyra2008b, Pertierra et al. Reference Pertierra, Lara, Benayas, Smith and Hughes2017). More generally, although the concept of rarity in mosses is well recognized in the global literature, definitions vary widely (Vellak et al. Reference Vellak, Vellak and Ingerpuu2006, Belland & Caners Reference Belland and Caners2021) and are often difficult to apply to Antarctic data. Even within the geologically linked South American region, such as the Cape Horn Biosphere Reserve within the extreme south of the continent, which is defined in part by its globally exceptional cryptogamic diversity (Rosenfeld et al. Reference Rosenfeld, Convey, Contador, Rendoll, Poulin, Maturana, Rozzi, Morello, Massardo and Aldunate2020, Villagrán Moraga Reference Villagrán Moraga2020), rarity in mosses has not been formally defined or assessed.
At the Antarctic continental scale, the most comprehensive synthesis of moss distributions to date remains that provided by Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b), who recognized 113 moss species and proposed the only explicit, quantitative approach to Antarctic moss rarity available. In this framework, species recorded from one or two locations were classified as very rare and those from three to five as rare, where a location was defined as an island or a distinct continental site. Recent research has increased the total number of recognized Antarctic moss species to 116 (Camara et al. Reference Camara, Carvalho-Silva and Stech2021), without altering the rarity framework itself. Using similar frequency-based reasoning, Pertierra et al. (Reference Pertierra, Lara, Benayas, Smith and Hughes2017) considered four of the 11 currently known Antarctic endemic moss species, each recorded from only one to three locations, as among the rarest mosses on the continent. At a regional scale, only Ivanets et al. (Reference Ivanets, Wierzgoń, Yevchun and Parnikoza2023) have explicitly addressed rarity in the AI-KP region, following Ochyra et al.’s (Reference Ochyra, Smith and Bednarek-Ochyra2008b) categorization.
Applying Ochyra et al.’s (Reference Ochyra, Smith and Bednarek-Ochyra2008b) rarity criteria at the continental scale, rare and very rare species account for 31 (27.4%) and 16 (14.2%) species, respectively, indicating that more than 40% of known Antarctic moss species fall into these categories. Although this pattern may partly reflect an uneven sampling effort across Antarctica, mosses are one of the best-documented groups of Antarctic terrestrial biota (Anderson et al. Reference Anderson, Chown and Leihy2024, Pertierra et al. Reference Pertierra, Varliero, Barbosa, Biersma, Convey and Chown2024).
Even though the frequency classes used by Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b) do not represent an endorsed standard but, rather, an expert-based approach, such categorization is consistent with long-standing floristic tradition in bryology and plant checklists. Within this tradition, species rarity is expressed through frequency classes based on the number of known occupied locations within a defined region (e.g. Rabinowitz’s (Reference Rabinowitz and Synge1981) classification of rarity, Perrin & Waldren’s (Reference Perrin and Waldren2020) vegetation rarity indices). Ochyra et al.’s (Reference Ochyra, Smith and Bednarek-Ochyra2008b) scheme can thus be interpreted as an adaptation of this commonly used floristic practice to the Antarctic context rather than a unique or formally established rarity system. In this study, we used all currently available literature and accessible databases with confirmed records based on herbarium specimens to assess the current status of rare moss species in the AI-KP region of the WAP. Thereby, we set out to illustrate the potential conservation value of a detailed within-region botanical survey that is still lacking for many parts of Antarctica. Our objectives were 1) to identify locally rare moss species and map their distribution by applying the only existing criteria of Antarctic moss rarity at a regional scale (Ochyra et al. Reference Ochyra, Smith and Bednarek-Ochyra2008b), 2) to place the distribution of these locally rare species in the context of their entire Antarctic distribution and 3) to identify patterns of rare moss diversity in the study area - a crucial step in identifying particularly valuable and/or vulnerable sites that may be appropriate for formal protection. We did not set out to develop or test new rarity criteria but rather to provide a first step in generating a comprehensive, pragmatic and up-to-date evaluation of the moss flora of the AI-KP region.
Methods
Study area
The AI-KP region comprises the northern part of the Graham Coast (Fig. 1) and stretches ~60 km from Booth Island (65°02′42″S, 63°56′01″W) to Lahille Island (65°33′26″S, 64°22′08″W). The region includes parts of the Wilhelm Archipelago, along with some islands south of the archipelago and several ice-free coastal areas on the Kyiv Peninsula. The study area lies towards the northern edge of the central zone of the WAP as described by Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b, p. 43), close to its boundary with the northern WAP zone. The area is therefore of interest for studies of edge-zone effects and species range limits (Humphries et al. Reference Humphries, Naveen, Schwaller, Che-Castaldo, McDowall, Schrimpf and Lynch2017, Wethington et al. Reference Wethington, Borowicz and Lynch2023). It also forms part of the region with the strongest climate change trends and predicted impacts in Antarctica since the mid-twentieth century (King & Harangozo Reference King and Harangozo1998, Turner et al. Reference Turner, Bindschadler, Convey, Di Prisco, Fahrbach and Gutt2009, Siegert et al. Reference Siegert, Atkinson, Banwell, Brandon, Convey and Davies2019).
Area of study: the Argentine Islands-Kyiv Peninsula region, Graham Coast, Western Antarctic Peninsula.

In this study, we follow the general biogeographical division of Antarctica into the Continental and the Maritime Antarctic (Smith Reference Smith and Laws1984, Ochyra et al. Reference Ochyra, Smith and Bednarek-Ochyra2008b, Convey & Biersma Reference Convey, Biersma and Scheiner2024), with the AI-KP region lying within the Maritime Antarctic.
The climate of the AI-KP region is relatively mild and humid, typical of the Maritime Antarctic, with mean annual air temperatures ranging between −2.4°C and −5.4°C (Parnikoza et al. Reference Parnikoza, Berezkina, Moiseyenko, Malanchuk and Kunakh2018) and an average annual precipitation of 433 mm (Martazinova et al. Reference Martazinova, Tymofeev and Ivanova2010). At the Ukrainian Vernadsky Station (formerly the UK’s Faraday Base) on Galindez Island, located in the central part of the region (65°14′44′ ′S, 64°15′25′ ′W), the average annual temperature increased by 2°C between 1947 and 2007 (Martazinova et al. Reference Martazinova, Tymofeev and Ivanova2010), highlighting that this region is an important area for studies of climatic changes and their impacts on Antarctic ecosystems.
Accessible moss occurrence data
The study is based on occurrence data of Antarctic mosses compiled, verified and updated from three primary sources, with only records meeting the following criteria included: sampling location within the study region, availability of coordinates or a clear location description and the presence of a herbarium specimen suitable for taxonomic verification. Data sources comprised 1) the monograph The Moss Flora of Antarctica (Ochyra et al. Reference Ochyra, Smith and Bednarek-Ochyra2008b), based on herbarium collections worldwide, 2) records from GBIF, including datasets contributed by the BAS (Andrino Reference Andrino2025, British Antarctic Survey 2025, Terauds Reference Terauds2025), and 3) unpublished and newly verified specimen records from the KRAM herbarium (W. Szafer Institute of Botany, Polish Academy of Sciences), including collections from 2019 to 2022 that have not yet been digitized or formally published. All eligible records were standardized, verified and merged into a single dataset, and the Web of Science database was additionally searched for moss records, satisfying the same inclusion criteria retained.
The spatial coordinates of all records were checked and, where necessary, adjusted for map representation, with each record cross-verified against its herbarium reference. Clearly erroneous coordinates (e.g. located in the ocean or on ice-covered areas lacking terrestrial habitat) were manually corrected. Where only location names were available, approximate coordinates were assigned based on known moss distribution at the respective sites. Coordinates are reported as provided in the original sources; however, Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b) do not report geographical coordinates and only indicate locations on small-scale maps. Herbarium specimens cited by Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b) were additionally verified via the GBIF database.
Maps were created using QGIS software, version 3.28.6-Firenze, and the freely available base layer ‘High resolution vector contours for Antarctica (7.3)’, available from the Scientific Committee on Antarctic Research (SCAR) Antarctic Digital Database (Gerrish et al. Reference Gerrish, Fretwell and Cooper2020).
The only continental-scale rarity classification of Antarctic mosses currently available is that of Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b). This considered a species to be very rare if the total number of unique occurrence locations was 1–2, rare for 3–5, occasional for 6–10, frequent for 11–20 and common for > 21 locations. Applying this classification at the scale of the study area defined here, our study focuses only on species classified as very rare or rare, with 1–2 or 3–5 unique occurrences, respectively. We recognize that any such definition of rarity will be influenced by the wide differences in survey effort and data availability across the region, and that classifications will inevitably change with further survey work, but pragmatically this provides the only dataset presently available.
Taxonomic check
All identified records were checked for synonyms and were included in this study under their current names; overall systematics follow Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b) and correspond to valid names under the International Code of Nomenclature for algae, fungi and plants. Each record used in this study provides information on an individual specimen identified to the species level and with a unique catalogue number. However, samples collected from the same location and contributing more than one specimen in the original databases are often listed as a single record. In such cases, as noted earlier, each species was reported here as an individual record. For other species identification notes, see Supplemental Material S3.
Analysis of distribution patterns
Using RStudio software, version 2023.12.0, sites were grouped into descriptive richness categories based on the number of locally rare and very rare species present (1–2, 3–5 and ≥ 6 species), providing a framework for comparing patterns of species overlap without applying formal similarity indices. Quantitative similarity indices such as Sørensen, although commonly used to compare species assemblages, were not appropriate in this case because the analysis focused exclusively on locally rare and very rare species rather than the complete regional moss species pool, which could lead to misleading estimates of assemblage similarity or failure to detect strict inclusion relationships. Overlap in species composition was assessed using a presence-absence matrix for locally rare and very rare moss species to identify hierarchical relationships among sites. Sites were then compared to identify core assemblages, subset assemblages among sites and site-unique occurrences. Strict subset relationships were determined by testing whether the complete species set of one site was fully contained within another site. Sites not forming subsets were defined as core assemblages.
Results
Rare species
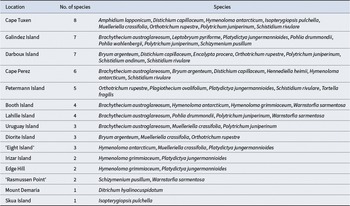
The GBIF database included 16 682 moss records for the entire Antarctic Peninsula. After filtering and verifying and processing data from Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b), 23 moss species and 78 records matching the rarity criteria required were obtained from the AI-KP region (Table S1, data source ‘gbif’, ‘both’ and ‘Ochyra’, see the explanation in the table). The additional Web of Science search provided five new records of three very rare and rare moss species associated with yet-to-be-registered herbarium specimens. Other records of five species already listed in this study, uncatalogued in GBIF, were added from the KRAM herbarium, giving a total of 91 unique records of 23 rare and very rare moss species for the AI-KP region. Of these 91 records, 18 (20%) were given only in the GBIF database, 20 (22%) were given only in Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b), 40 (44%) were listed in both sources and 13 (14%) were uncatalogued records from KRAM herbarium, three of which have been recently published (Blockeel et al. Reference Blockeel, Bednarek-Ochyra, Ochyra, Hájková, Hájek and Kuĉera2006, Ellis et al. Reference Ellis, Asthana, Srivastava, Omar, Rawat and Sahu2016, Reference Ellis, Afonina, Czernyadjeva, Konoreva, Potemkin and Kotkova2020, Ivanets et al. Reference Ivanets, Wierzgoń, Yevchun and Parnikoza2023).
Of the 23 identified species, 12 (52.2%) were defined as ‘very rare’ and 11 (47.8%) as ‘rare’ for the AI-KP region. Among these, however, only four (17.4%) were categorized as rare or very rare for Antarctica as a whole. The two very rare species are Amphidium lapponicum (Hedw.) Schimp. and Leptobryum pyriforme (Hedw.) Wilson, and the rare species are Schizymenium pusillum (Hook. & Wilson) A.J. Shaw and Pohlia wahlenbergii (F. Weber & D. Mohr) A.L.Andrews. Of the remaining 19 locally rare species, three (13.0%) are occasional, eight (34.8%) are frequent and eight (34.8%) are common across Antarctica (Table I).
Rarity on the general Antarctic scale of the 23 moss species identified to be locally rare or very rare in the Argentine Islands-Kyiv Peninsula region, and their local distributions. Following Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b), rarity is assessed as: VR = very rare (1–2 unique locations); R = rare (3–5 unique locations); O = occasional (6–10 unique locations); F = frequent (11–25 unique locations); C = common (> 25 unique locations).

Mapping rare moss species biodiversity
For clarity of presentation, the distributions of the 23 locally rare species in the AI-KP region are presented separately for locally very rare moss species (Fig. 2) and for locally rare moss species (Fig. 3).
Distribution of moss species considered locally very rare in the Argentine Islands-Kyiv Peninsula (AI-KP) region, Graham Coast, Western Antarctic Peninsula. Circle colours indicate the general Antarctic rarity of each species following Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b). Numbers correspond to species listed in the legend at the bottom of the figure.

Distribution of moss species considered locally rare in the Argentine Islands-Kyiv Peninsula (AI-KP) region, Graham Coast, Western Antarctic Peninsula. Circle colours indicate the general Antarctic rarity of each species following Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b). Numbers correspond to species listed in the legend at the bottom of the figure.

Spatial distribution and patterns of rare moss diversity
The spatial distributions of the 23 locally very rare and rare moss species in the AI-KP region are summarized in Table II and represented in the form of a presence-absence matrix across individual locations (Table S2). The total numbers of rare and very rare species at any given location ranged between one and eight.
Distribution of 23 locally very rare and rare moss species at different locations in the Argentine Islands-Kyiv Peninsula region, Western Antarctic Peninsula.

Categorizing the 15 distinct record sites in terms of rare and very rare moss species diversity (Fig. 4) demonstrated that only five specific locations within the AI-KP region hosted a low diversity of very rare and rare species (one to two species). These included some of the Argentine Islands (Skua and Irizar islands), ‘Rasmussen Point’ (locally used name;) and two mainland sites (Edge Hill, Mount Demaria). Almost half of the sites (six) hosted moderate diversity (three to five very rare and rare species), including Uruguay, ‘Eight’ (Yevchun et al. Reference Yevchun, Fedchuk, Drohushevska, Pnyovska, Chernyshenko and Parnikoza2021b), Petermann, Diorite (informally referred to as ‘Ukraine Island’ in earlier Ukrainian Antarctic studies; Yevchun et al. Reference Yevchun, Fedchuk, Drohushevska, Pnyovska, Chernyshenko and Parnikoza2021b), Lahille and Booth islands. Cape Tuxen and Cape Pérez (two Antarctic Peninsula mainland sites) as well as Darboux Island and Galindez Island had the highest very rare and rare moss species diversity, with eight, six, seven and seven species, respectively.
Distribution of 23 locally rare and very rare moss species across the 15 sites of the Argentine Islands-Kyiv Peninsula region (Graham Coast, Western Antarctic Peninsula), showing the number of species recorded at each site.

Although some sites hosted similar numbers of locally rare and very rare moss species, their species compositions were not identical. Comparison of presence-absence data (Table S2) showed incomplete overlap among sites, with some locations representing subsets of richer core assemblages, while others contributed locally unique occurrences to the regional species pool.
The four most species-rich sites (Cape Tuxen, Cape Pérez, Galindez Island, Darboux Island) shared a substantial proportion of rare species and together formed a core assemblage within the AI-KP region. Overlap among the four most species-rich sites was partial: no single species occurred at all four locations, but several taxa were shared across three sites, and multiple species were shared pairwise. Pairwise similarity was highest between Cape Tuxen and Darboux Island (four shared species) and lowest between Cape Tuxen and Galindez Island (one shared species). Overall, Galindez Island was the most distinct of the group, sharing only one species with each of the other three sites.
The moderately diverse sites with three to five species shared species with the core assemblage but differed in the degree of overlap. Some locations (Uruguay Island, Diorite Island, ‘Eight Island’) represented a full species pool of the four most species-rich sites, whereas others (Booth Island, Lahille Island, Petermann Island) combined shared taxa with additional species not recorded at the four most species-rich sites. However, none of them individually represents a subset of any one of those richest sites; the correspondence only becomes apparent when their species lists are considered together.
Sites with low numbers of locally rare species (one or two taxa) also differed in their relationship to the core assemblage. Skua Island, Edge Hill and Irizar Island contained rare species recorded at the species-rich core sites, with the latter two locations being identical, and therefore they represent strict subsets of the combined species pool of the four richest locations. In contrast, Mount Demaria hosted a single locally rare species (Ditrichum hyalinocuspidatum) that was not recorded at any other site within the AI-KP region, and Cape Tuxen hosted an Antarctica-wide (continental and offshore islands) single occurrence of A. lapponicum (its only other Antarctic occurrence is in the South Shetland Islands according to GBIF).
Species-level distribution and ranges
In the following, we provide descriptions of distributional patterns for each species. Detailed information for each specimen is given in Table S1, including the original specimen number, location coordinates, conservation significance and other features. For newly reported findings not available in GBIF or AAS but voucher-verified the in KRAM collection, we also provide the available details of the collection site in Table S1 (growing conditions, altitude, habitat, etc.).
For the four species that are rare or very rare at the entire Antarctic scale, several of our records represent both the only known mainland Antarctic and its offshore islands’ occurrences and their southernmost distribution limit, or only their southernmost distribution limit. A. lapponicum has its only known Antarctic mainland occurrence at Cape Tuxen, despite its wide global distribution. The three other species (L. pyriforme, S. pusillum, P. wahlenbergii) reach their southernmost limits in the AI-KP region (see Table S1), marking a southern latitudinal boundary at ~65°15′S.
Among the mosses classified as rare or very rare within the AI-KP region, several species that are frequent or occasional in the entire Antarctic also reach their southernmost or near-southern distributional limits within the study region (Muelleriella crassifolia, Orthotrichum rupestre, Pohlia drummondii, Schistidium rivulare, Warnstorfia sarmentosa). Of these five species, P. drummondii and W. sarmentosa are rare also on a broader scale across the Antarctic Peninsula.
Within the AI-KP region, and in many cases for the entire Graham Coast, several species that are occasional, frequent or common elsewhere in Antarctica are represented at only one or two locations. Examples include D. hyalinocuspidatum (Mount Demaria, which also constitutes its only mainland Antarctic Peninsula site and highest known elevation of 600 m above sea level), Hennediella heimii (very rare on the Graham Coast, only recorded on Cape Pérez), Isopterygiopsis pulchella (Cape Tuxen, Skua Island), Plagiothecium ovalifolium and Tortella fragilis (both restricted within the Graham Coast only to Petermann Island), P. drummondii (Lahille Island) and Schistidium andinum (Darboux Island, which is also its only occurrence on the Graham Coast) and Encalypta procera, which has only one Graham Coast occurrence (Darboux Island), even though it is considered common in Antarctica, with two occurrences on the South Shetland Islands, one in the South Orkney Islands, several on the eastern Antarctic Peninsula and one occurrence each for the Danco Coast, the Graham Coast and the Loubet Coast.
Finally, several taxa (Brachythecium austroglareosum, Bryum argenteum, Distichium capillaceum, Hymenoloma antarcticum, Hymenoloma grimmiaceum, Platydictya jungermannioides, Polytrichum juniperinum) are widespread across the Maritime Antarctic but occur in the AI-KP region only in small, spatially isolated populations and are rare on the Graham Coast. Also see the Bryum spp. taxonomic notes in Notes S3, based on Cannone et al. (Reference Cannone, Vanetti, Convey, Sancho and Zaccara2024).
Two moss species present in the AI-KP region show additional distinctive features. First, several local populations of H. antarcticum are fertile (Ochyra et al. Reference Ochyra, Smith and Bednarek-Ochyra2008b), whereas the many populations in other Antarctic regions consist of sexually sterile plants (that do not produce sporophytes), including the three southernmost populations on the eastern Antarctic Peninsula and on Stork Ridge. Second, P. juniperinum appears to show a significant spatial separation between its southernmost known occurrence in Marguerite Bay and its presence in the AI-KP region on Lahille Island (Ochyra et al. Reference Ochyra, Smith and Bednarek-Ochyra2008b, GBIF).
Discussion
Rare moss species distribution
Using the same numerical classification as proposed by Ochyra et al. (Reference Ochyra, Smith and Bednarek-Ochyra2008b) but applied at the scale of our specific study region, those species whose rarity classifications at the continental and local levels coincide can be identified. These represent species, based on the best available information, those whose effective protection in Antarctica is a particular priority. These species and their sites of occurrence in the AI-KP region are A. lapponicum (Cape Tuxen, the species’ only known mainland Antarctic and its offshore islands’ location), L. pyriforme (only known mainland Antarctic and its offshore islands’ location), P. wahlenbergii (previously known only from the South Shetland Islands, but recently found on Galindez Island; Ivanets et al. Reference Ivanets, Wierzgoń, Yevchun and Parnikoza2023) and S. pusillum (Galindez Island and ‘Rasmussen Point’).
At the local scale of the AI-KP region, all 23 species identified in the current study are classified as either ‘very rare’ or ‘rare’. Eight of these species are also rare or very rare at the scale of the WAP, while the classifications of two of these species matched those of their general Antarctic classifications. For two species, the AI-KP region provides their only known locations in Antarctica (A. lapponicum, L. pyriforme) and for eight species (A. lapponicum, D. hyalinocuspidatum, E. procera, L. pyriforme, P. ovalifolium, P. drummondii, S. andinum, T. fragilis) the AI-KP region provides their only known locations in the Graham Coast and its associated offshore islands. Eight species (A. lapponicum, L. pyriforme, M. crassifolia, O. rupestre, P. drummondii, P. wahlenbergii, S. rivulare, S. pusillum) reach their southern distributional limits in the AI-KP region, and one species shows a very sporadic distribution, with records from only two other very remote regions in Antarctica (W. sarmentosa). Finally, the local population of H. antarcticum, although not regionally rare, is the southernmost fertile population in Antarctica, and the local population of D. hyalinocuspidatum is the only mainland Antarctic Peninsula population, as well as it being found here at its highest known elevation in Antarctica.
The observation that many species reach their southernmost distributional limits within the study region highlights the transitional biogeographical position of the AI-KP region. Within the AI-KP region, several species that are frequent or occasional elsewhere in Antarctica are represented at single or very few locations, indicating strong regional fragmentation. These patterns collectively indicate that the AI-KP region functions as both a southern range margin and as a distributional bottleneck for multiple Antarctic moss species.
Implications for conservation
Based on the results obtained and the analysis of the distribution of rare species in the AI-KP region, the locations with the highest numbers (six to eight) of locally rare species were Cape Tuxen, Cape Pérez, Darboux Island and Galindez Island. However, the importance of protecting areas with lower numbers of rare species must also be considered. Some of these may represent either very valuable populations for scientific research (e.g. species at the edge of their distribution range) or the only location of a given species’ occurrence in Antarctica as a whole or in the Antarctic Peninsula region (e.g. W. sarmentosa, P. wahlenbergii). Simultaneously, although several islands host species that are present in the combined pool of the four most species-rich sites, this overlap does not diminish their other conservation values. Some of these sites harbour biogeographically critical features, including the southernmost Antarctic populations of particular species, whereas others contribute additional species not recorded at the four richest sites, underscoring their irreplaceability within the regional conservation network. We also note that multiple ‘values’ can be used when considering the necessity of area protection in Antarctica, including the occurrence of individual species, recognitions of both representative and outstanding community composition and locations that are not specifically rich in rare species but that include important biological features such as southern distributional limits.
Future measures for Antarctic vegetation protection
Building on the outcomes of our analyses of the distributions of rare moss species in the AI-KP region, further comprehensive research is required on the distribution of the more common moss species present. Furthermore, ongoing mapping and monitoring of rare moss species distributions in this region will confirm whether the significant locations identified here will remain relevant in the face of ongoing and predicted environmental changes in this part of Antarctica (Chown et al. Reference Chown, Clarke, Fraser, Cary, Moon and Mcgeoch2015, Lee et al. Reference Lee, Raymond, Bracegirdle, Chadès, Fuller, Shaw and Terauds2017, Reference Lee, Waterman, Shaw, Bergstrom, Lynch, Wall and Robinson2022, Hughes et al. Reference Hughes, Convey and Turner2021). However, our initial analysis of rare species occurrences already supports the identification of specific locations requiring urgent protection through the mechanisms of inclusion in Visitor Site Guidelines (Yevchun et al. Reference Yevchun, Dykyi, Kozeretska, Fedchuk, Karamushka and Parnikoza2021a, ATCM 2022a,b) and the establishment of appropriate and representative ASPAs (Hughes et al. Reference Hughes, Ireland, Convey and Fleming2015, Coetzee et al. Reference Coetzee, Convey and Chown2017, ATCM 2023, 2024).
Supplemental material
To view supplemental material for this article, please visit http://doi.org/10.1017/ans.2026.10065.
Acknowledgements
HY, IP and ED thank the Armed Forces of Ukraine for defending Ukraine and thus enabling the authors to continue their research on behalf of Ukraine, as well as the countries and people of Poland and the UK for their support of the authors. We thank Prof. R. Ochyra and Dr M. Wierzgoń for consultation and access to the specimens of the KRAM herbarium, Dr R. Mackenzie for advice on the Chilean bryophyte literature and the editor and reviewers for their critical and constructive comments.
Financial support
PC is supported by NERC core funding to the BAS ‘Biodiversity, Evolution and Adaptation’ Team.
Competing interests
The authors declare none.
Author contributions
HY and IP conceived the study. HY gathered and interpreted the data, including the analysis in QGIS. HY prepared the figures and tables and drafted the initial manuscript, with input and additions from IP, PC and ED. JS participated in the material processing. All authors contributed to the drafts and gave final approval for the manuscript.