Introduction
Isopods (Peracarida: Isopoda) are morphologically and ecologically diverse crustaceans that inhabit a variety of freshwater, marine, and terrestrial environments (Schmidt Reference Schmidt2008; Wilson Reference Wilson, Balian, Lévêque, Segers and Martens2008; Poore and Bruce Reference Poore and Bruce2012). Terrestrial isopods belong to the suborder Oniscidea (Schmidt Reference Schmidt2008) and are also known as woodlice or sowbugs. With more than 3700 species, sowbugs form the largest isopod suborder (Schmalfuss Reference Schmalfuss2004; Sfenthourakis and Taiti Reference Sfenthourakis and Taiti2015) and are one of the few groups of non-hexapod crustaceans with fully terrestrial members (Richardson and Araujo Reference Richardson, Araujo, Thiel and Watling2015). Sowbugs are found worldwide in a range of different habitats, including caves, deserts, shorelines, ant nests, and urban areas (Hornung Reference Hornung2011; Szlavecz et al. Reference Szlavecz, Vilisics, Tóth and Hornung2018). The highest species diversity occurs in temperate climates such as that found in the Mediterranean region (Sfenthourakis and Hornung Reference Sfenthourakis and Hornung2018).
Sowbugs include many ecologically and economically important species. As sowbugs primarily feed on plant detritus, they are involved in the process of decomposition and can alter the structure of microbial communities in leaf litter (Zimmer Reference Zimmer2002; Des Marteaux et al. Reference Des Marteaux, Kullik, Habash and Schmidt2020). Some species also feed on seeds (Saska Reference Saska2008), dead animal matter (Kensley Reference Kensley1974), or live plants (Johnson et al. Reference Johnson, Alfaress, Whitworth and McCornack2012). The last behaviour may result in damage to crops in fields and greenhouses if sowbug populations reach large numbers (Messelink and Bloemhard Reference Messelink and Bloemhard2007; Souty-Grosset and Faberi Reference Souty-Grosset and Faberi2018). The role of sowbugs as agricultural pests was reviewed by Souty-Grosset and Faberi (Reference Souty-Grosset and Faberi2018): affected crops include canola, soybeans, sunflowers, and cereal crops, with records from Argentina, Australia, South Africa, and the midwestern United States of America. Sowbugs also act as intermediate hosts for parasites that have wild or domesticated vertebrates as final hosts. These parasites include the nematode genera Acuaria Bremser, 1811 and Dispharynx Railliet et al., 1912 (Rhabditida: Acuariidae), which infect birds (Carreno Reference Carreno, Atkinson, Thomas and Hunter2008), and the acanthocephalan Plagiorhynchus cylindraceus (Goeze, 1782) (Polymorphida: Plagiorhynchidae) (Dimitrova Reference Dimitrova2009), which infects a wide variety of birds and mammals (Llanos-Soto et al. Reference Llanos-Soto, Córdoba, Moreno, Kinsella, Mironov and Cicchino2019).
The first documented sowbug species in Canada was Porcellio scaber Latreille, 1804 (Isopoda: Porcellionidae), reported in 1859 from coastal British Columbia (GBIF.org 1859). The first review to include Canadian sowbug records listed six species from Canada (Richardson Reference Richardson1905). Subsequently, Johansen (Reference Johansen1926) listed 10 species from Canada, Van Name (Reference Van Name1936) listed 14 species, Hatch (Reference Hatch1947) listed 15 species, and Leistikow and Wägele (Reference Leistikow and Wägele1999) listed 16 species. The most recent review of sowbugs found north of Mexico lists 25 species from Canada (Jass and Klausmeier Reference Jass and Klausmeier2001). Many of these species are nonnative and originate from Europe (Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001). Except for the troglobitic Ligidium elrodii (Packard, 1873) (Isopoda: Ligiidae) (Schultz Reference Schultz1970), native sowbugs in northern North America are limited to coastal habitats that were not completely glaciated (Jass and Klausmeier Reference Jass and Klausmeier2000, Reference Jass and Klausmeier2001). However, the existence of a native cave-dwelling aquatic isopod in Alberta (Clifford and Bergstrom Reference Clifford and Bergstrom1976; Holsinger et al. Reference Holsinger, Mort and Recklies1983) suggests that troglobitic sowbugs could have survived glaciation.
Published records of sowbugs in Canada have been geographically limited to the eastern and western coasts and to localities near the Great Lakes, and records before 1980 are scarce. No published sowbug records exist from northern Canada (Yukon, Northwest Territories, Nunavut), and as of 2014, there were no records from any of the Prairie Provinces (Alberta, Saskatchewan, Manitoba; Snyder Reference Snyder, Cárcamo and Giberson2014). The first and only published record of a sowbug from the Prairie Provinces was Trachelipus rathkii (Brandt, 1833) (Isopoda: Trachelipodidae) from a single site in Edmonton, Alberta (Li Reference Li2022). However, sowbugs have been present in this area for at least two decades: community science websites such as iNaturalist (https://www.inaturalist.org) and BugGuide (https://bugguide.net/node/view/15740) include many unambiguous photographs of sowbugs from Alberta, Saskatchewan, and Manitoba (e.g., iNaturalist community 2024a, 2024b, 2024c; BugGuide 2024a, 2024b, 2024c), and there are also Global Biodiversity Information Facility (GBIF.org) records that are not reported from publications but that list sowbugs from this area (e.g., GBIF.org 1968, 2018b). Based on these sources, the earliest record of a sowbug from the Prairie Provinces is a specimen of Armadillidium vulgare (Latreille, 1804) (Isopoda: Armadillidiidae) in the Canadian Museum of Nature Crustacea Collection (Ottawa, Ontario, Canada) that was collected from Calgary, Alberta, in 1968 (GBIF.org 1968).
There are several possible reasons why few published records exist for sowbugs from the Prairie Provinces. First, this region has never been surveyed specifically for sowbugs (Floate et al. Reference Floate, Shorthouse, Giberson and Cárcamo2017). However, a variety of types of pitfall traps can effectively capture sowbugs (Siewers et al. Reference Siewers, Schirmel and Buchholz2014), and surveys designed to assess arthropod diversity in the Prairie Provinces using pitfall traps (Floate et al. Reference Floate, Shorthouse, Giberson and Cárcamo2017) would likely have captured sowbugs if they had been present: captured sowbugs may have gone unreported if they were not among the collectors’ taxa of interest. Second, arthropod surveys may have failed to capture sowbugs because arthropod surveys in the Prairie Provinces have typically focused on natural or seminatural habitats rather than on urban areas (Snyder Reference Snyder, Cárcamo and Giberson2014; Floate et al. Reference Floate, Shorthouse, Giberson and Cárcamo2017). Introduced species of sowbugs are common in urban areas (Cochard et al. Reference Cochard, Vilisics and Sechet2010; Szlavecz et al. Reference Szlavecz, Vilisics, Tóth and Hornung2018), particularly in climatically unfavourable regions where buildings provide warm refugia (Wright Reference Wright1997). Similarly, sowbugs in urban areas in eastern Russia are thought to have been present long before populations were first reported because soil arthropod surveys were focused on undisturbed areas (Gongalsky and Kuznetsova Reference Gongalsky and Kuznetsova2021). Finally, sowbugs may be a relatively recent introduction to the Prairie Provinces. Anecdotal evidence from long-time residents of Alberta suggests sowbugs were absent from the province until the late 1970s (H.C.P. and F.A.H.S., unpublished data; John Acorn, personal communication).
Initial introductions of sowbugs to North America may have occurred through transport of soil ballast on ships (Lindroth Reference Lindroth1957), and present-day introductions may take place through the sale of sowbugs as pet food (Cochard et al. Reference Cochard, Vilisics and Sechet2010), as pets themselves (e.g., Jungle Jewel Exotics 2023), through shipment of building materials (Slabber and Chown Reference Slabber and Chown2002) and gravel (McQueen Reference McQueen1976), or via shipment of soil, plants, and other gardening supplies to greenhouses and gardens (Hatch Reference Hatch1947; Jass and Klausmeier Reference Jass and Klausmeier2000; Vilisics and Hornung Reference Vilisics and Hornung2009; Cochard et al. Reference Cochard, Vilisics and Sechet2010; De Smedt et al. Reference De Smedt, Arijs, Segers and Boeraeve2017). These routes of introduction are also shared by other soil invertebrates such as earthworms (Bohlen Reference Bohlen, Scheu, Hale, McLean, Migge, Groffman and Parkinson2004), which, similarly to sowbugs, include multiple introduced species spread by human activity. The particular importance of greenhouses as routes of sowbug introduction is suggested by the abundance and species richness of sowbugs in greenhouses: sowbugs were present in 66 of 67 greenhouses examined in North America in the Pacific Northwest in the mid-20th century (Hatch Reference Hatch1947) and in all seven of the greenhouses surveyed in Britain more recently (Gregory and Lugg Reference Gregory and Lugg2020). Greenhouses also typically have high species richness (De Smedt et al. Reference De Smedt, Arijs, Segers and Boeraeve2017), even when compared to natural areas such as parks and forests (Korsós et al. Reference Korsós, Hornung, Szlávecz and Kontschán2002). Some species of sowbugs may be entirely restricted to greenhouses in colder parts of their nonnative range (Hatch Reference Hatch1947; Rapp Reference Rapp2001; Schmalfuss Reference Schmalfuss2004; Gregory Reference Gregory2014).
The present study is the first targeted survey of sowbugs in Alberta and includes new collections from four other Canadian provinces (British Columbia, Saskatchewan, Ontario, and Newfoundland and Labrador). These collections include the first Canadian record of Nagurus cristatus (Dollfus, 1899) (Isopoda: Trachelipodidae) (reported in Stormer and Proctor Reference Stormer and Proctor2022) and the first published record of a sowbug from Saskatchewan. We support our identifications with cytochrome c oxidase subunit 1 (CO1) barcode sequences for 12 sowbug species. We also provide an updated checklist of the sowbugs found in Canada, along with brief descriptions of the habitat each species occupies in Canada and the date of the first Canadian record. This study will help to guide researchers and naturalists in their identification of sowbugs currently found in Canada and to promote additional research on the routes of entry and ecological impact of sowbugs.
Methods
Canadian records of terrestrial isopods
To compile a list of sowbug species previously recorded from Canada, we performed literature searches in Google Scholar (https://scholar.google.ca/) and Web of Science (https://clarivate.com/academia-government/scientific-and-academic-research/research-discovery-and-referencing/web-of-science/) using combinations of keywords “isopod”, “terrestrial”, “sowbug”, “woodlice”, and “Canada”, with names of Canadian provinces and territories. All published records containing at least one species-level identification of a sowbug species from a Canadian locality were included in the dataset. We cite the original publication for each record whenever possible; if a publication listed a record without citing another source for the record, we assumed the record originated with that publication.
In addition to published literature, we compiled all records of sowbugs from Canada available on iNaturalist (https://www.inaturalist.org/) up to 20 October 2024 (see Supplementary material, File S3). We removed any records from our dataset that had Creative Commons Attribution, Share Alike (CC-BY-SA) or Creative Commons Attribution, No Derivatives (CC-BY-ND) licensing. Our decision to include iNaturalist data was based on the following points: (1) approximately 72% of the observations of terrestrial isopods from iNaturalist up to 20 October 2024 (Supplementary material, File S3) were identified by members of the American Isopod and Myriapod Group (AIMG) or the British Myriapod and Isopod Group (BMIG), including @hydrophilus, @americanisopodologist, @astrobirder, @aniedes, @bmig_steve, etc.; and (2) we assessed the reliability of iNaturalist sowbug records in general by examining all sowbug records from Alberta, Saskatchewan, Manitoba, Canada, and Montana and North Dakota, United States of America (n = 505 observations). Only 3% had been identified incorrectly. We also compiled sowbug records from BugGuide.net up to 20 October 2024 (see Supplementary material, File S3) and records from GBIF.org up to 18 January 2023 (GBIF.org 2023a, 2023b, 2023c, 2023d, 2023e, 2023f, 2023g, 2023h, 2023i, 2023j, 2023k, 2023l, 2023m) and from 18 January 2023 to 20 October 2024 (GBIF.org 2024). We obtained GBIF.org records up to 18 January 2023 by searching GBIF.org occurrences for each family in Oniscidea with the following filters applied: location = Canada and basis of record = material sample or preserved specimen. These filters exclude records from iNaturalist and limit records mostly to those originating from collections in museums and research institutions. We obtained GBIF.org records from 18 January 2023 to 20 October 2024 with the same filters applied using the R package, rgbif (Chamberlain et al. Reference Chamberlain, Barve, Mcglinn, Oldoni, Desmet, Geffert and Ram2024).
To illustrate species distributions, we used Google Maps (https://www.google.com/maps) to assign GPS coordinates to GBIF.org records and published records that lacked coordinates by using locality information listed in each source (e.g., descriptions, maps, or province). Coordinates were placed in the centre of the most precise locality possible given the information listed in the source. When the locality was identified only as a province, the coordinates were set to the centre of the province. We created a distribution map for each species using QGIS, version 3.14.16 (http://qgis.org).
Sowbug collection
We collected sowbugs in Alberta from 2021 to 2024, with smaller opportunistic collections from British Columbia and Ontario. Collecting efforts in Alberta focused on the greater Edmonton area. Sowbugs from the island of Newfoundland that were collected from 2011 to 2023 were contributed by D. Langor, L. Lafosse, D. Harrison, and A. Pretty, with collections from provincial parks conducted under permits to D. Langor (Survey of Insects of Newfoundland and Labrador, 2011–2013; Survey of Terrestrial Invertebrates of Newfoundland and Labrador, 2022–2024). Additional sowbugs were collected from Alberta, British Columbia, and Saskatchewan by colleagues (see Acknowledgements). Because the goal of these collections was to maximise the number of species encountered rather than to perform exhaustive surveys, many collection sites were chosen based on their similarity to published descriptions of sowbug habitat rather than on their geographic location. We used Google Maps to look for sites with suitable habitat (e.g., rock piles, mulch, and wooded areas with fallen logs). We searched for sowbugs in the spring, summer, and fall from a variety of urban areas, disturbed natural areas (e.g., nature trails in residential areas), and relatively undisturbed natural areas (e.g., trails in rural areas). In addition to outdoor collections, we searched for sowbugs in greenhouses in the Edmonton area, with permission from the greenhouse managers. Collection methods were similar across all types of collection sites: we hand-collected sowbugs by turning over gravel and mulch and by lifting items on the ground, and we obtained smaller sowbugs via Tullgren funnel extractions from leaf piles and soil. We also assessed the use of a UV flashlight and thermal imaging camera as aids to visually search for sowbugs. Although sowbugs placed on a white paper background were visible using both methods when placed on a flat table (fluorescing slightly under UV light or visible as a spot of differing temperature), neither method was sensitive enough to detect sowbugs in a more complex environment such as a leaf pile. With the exception of sowbug collections that were provided by colleagues and were already preserved in 70–95% ethanol, sowbugs were collected alive and temporarily housed in containers with a damp paper towel until they were identified. The sowbugs were then euthanised with carbon dioxide gas, photographed, and preserved in 95% ethanol for DNA extraction or in 70% ethanol as morphological vouchers. Euthanasia was conducted by placing sowbugs in a plastic tube with a fine mesh at one end, suspending the tube in the mouth of a glass vial with a mixture of baking soda and vinegar added to the bottom of the vial (ensuring the mixture did not contact the plastic tube), and corking the vial until the sowbugs stopped moving.
Our collections were augmented by specimens in the E.H. Strickland Entomological Museum teaching collections (University of Alberta, Edmonton) and by photos of sowbugs taken by the public via a community science initiative for the present study. In addition to publishing requests for photos and specimens in the Biological Survey of Canada newsletter (Stormer and Proctor Reference Stormer and Proctor2021, Reference Stormer and Proctor2022), we also posted instructions in 2022 on Facebook group pages belonging to nature and gardening clubs in Alberta, asking participants to submit photographs of sowbugs with locality and habitat information, but not the precise locality or any personal information. Features that allow sowbug identification to a particular species or genus are usually visible in photographs (Gregory Reference Gregory2022; H.G.S., unpublished data). Sowbugs collected by colleagues were obtained either by hand collection or with pitfall traps, and most were received after the sowbugs were preserved in 70% ethanol. Representatives of all species and most populations of sowbugs collected for the present study are deposited in the E.H. Strickland Entomological Museum (University of Alberta) under accession numbers UASM407001 to UASM407463.
To gather additional information pertaining to the entry of sowbugs into Canada and the species that may be present, we requested Canadian Food Inspection Agency import records for sowbugs imported from 2015 onwards (Canadian Food Inspection Agency 2022). Records before 2015 were not available electronically.
Sowbug identification
We identified all sowbug specimens and photographs morphologically using published keys and by comparison to species descriptions, from which we also derived our taxon concepts (Sars Reference Sars1898; Van Name Reference Van Name1936, Reference Van Name1940; Vandel Reference Vandel1960, Reference Vandel1962; Schultz Reference Schultz1982, Reference Schultz1984b; Ferrara and Schmalfuss Reference Ferrara and Schmalfuss1985; Garthwaite and Sassaman Reference Garthwaite and Sassaman1985; Hopkin Reference Hopkin1991; Bilton Reference Bilton1992, Reference Bilton1997; Garthwaite and Lawson Reference Garthwaite and Lawson1992; Schmidt Reference Schmidt1997, Reference Schmidt2003; Shultz Reference Shultz2018; Gregory Reference Gregory2019). For most morphologically identified species, we sequenced the CO1 barcode region from 3 to 5 individuals (Supplementary material, Table S1) and chose individuals to maximise intraspecific morphological variation and geographic distance among sampling sites. See Supplementary material, File S1 and Figs. S2, S3, S4, S5, S6, S7, and S8 for the methods and results of our molecular identification of sowbugs.
Results
Sowbug collection
We and our colleagues searched for sowbugs in 118 sites in Alberta (15 of which were greenhouses), 15 sites in British Columbia, 22 sites on the island of Newfoundland, two sites in Ontario, and one site in Saskatchewan (Fig. 1). In addition, we received 47 community science observations from Alberta and examined 17 Alberta-collected sowbugs from the E.H. Strickland Entomological Museum. Of the 118 surveyed Alberta sites, 79 had sowbugs present (66.95%), including 11 of 15 greenhouse sites (73.33%); sites where sowbugs were absent were noted only for Alberta. In total, we identified 2382 sowbugs to the species level, with 14 species and eight families represented in the examined material (Fig. 2; see Supplementary material, File S1, for the results of our molecular identification of sowbugs). Nine of the 14 species occur in Alberta and are in four families: Cylisticidae: Cylisticus convexus (De Geer, 1778); Trachelipodidae: Trachelipus rathkii (Brandt, 1833) and Nagurus cristatus (Dollfus, 1889); Armadillidiidae: Armadillidium nasatum (Budde-Lund, 1885) and Armadillidium vulgare (Latreille, 1804); Porcellionidae: Porcellionides pruinosus (Brandt, 1833), Porcellio spinicornis Say, 1818, Porcellio dilatatus Brandt, 1831, and Porcellio scaber Latreille, 1804. Species composition differed between Alberta, British Columbia, and Newfoundland and Labrador: sowbugs from Newfoundland and Labrador and British Columbia consisted primarily of Oniscus asellus Linnaeus, 1758 (Isopoda: Oniscidae) and Porcellio scaber, whereas Porcellio spinicornis and Trachelipus rathkii made up the majority of specimens from Alberta (Fig. 1).
Terrestrial isopod species composition for sites in British Columbia, Alberta, Saskatchewan, Ontario, and Newfoundland where sowbugs were collected for the present study. Each pie chart shows the species composition for a single collection event, with the size of the chart corresponding to the total number of sowbugs collected. Empty charts indicate localities where no sowbugs were found. The pie charts in the box show collections from greenhouses. The basemap is the ESRI street map (https://server.arcgisonline.com/arcgis/rest/services/World_Street_Map/MapServer).

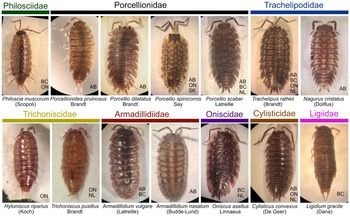
Terrestrial isopod species (Isopoda: Oniscidea) collected from Canada in the present study. Ligidium gracile (Ligiidae) is missing both exopodites. Species are not shown to scale relative to each other. Provinces where each species was collected are listed in the lower right of each image: BC, British Columbia; AB, Alberta; SK, Saskatchewan; ON, Ontario; NL, Newfoundland and Labrador.

Checklist of Canadian records of terrestrial isopods
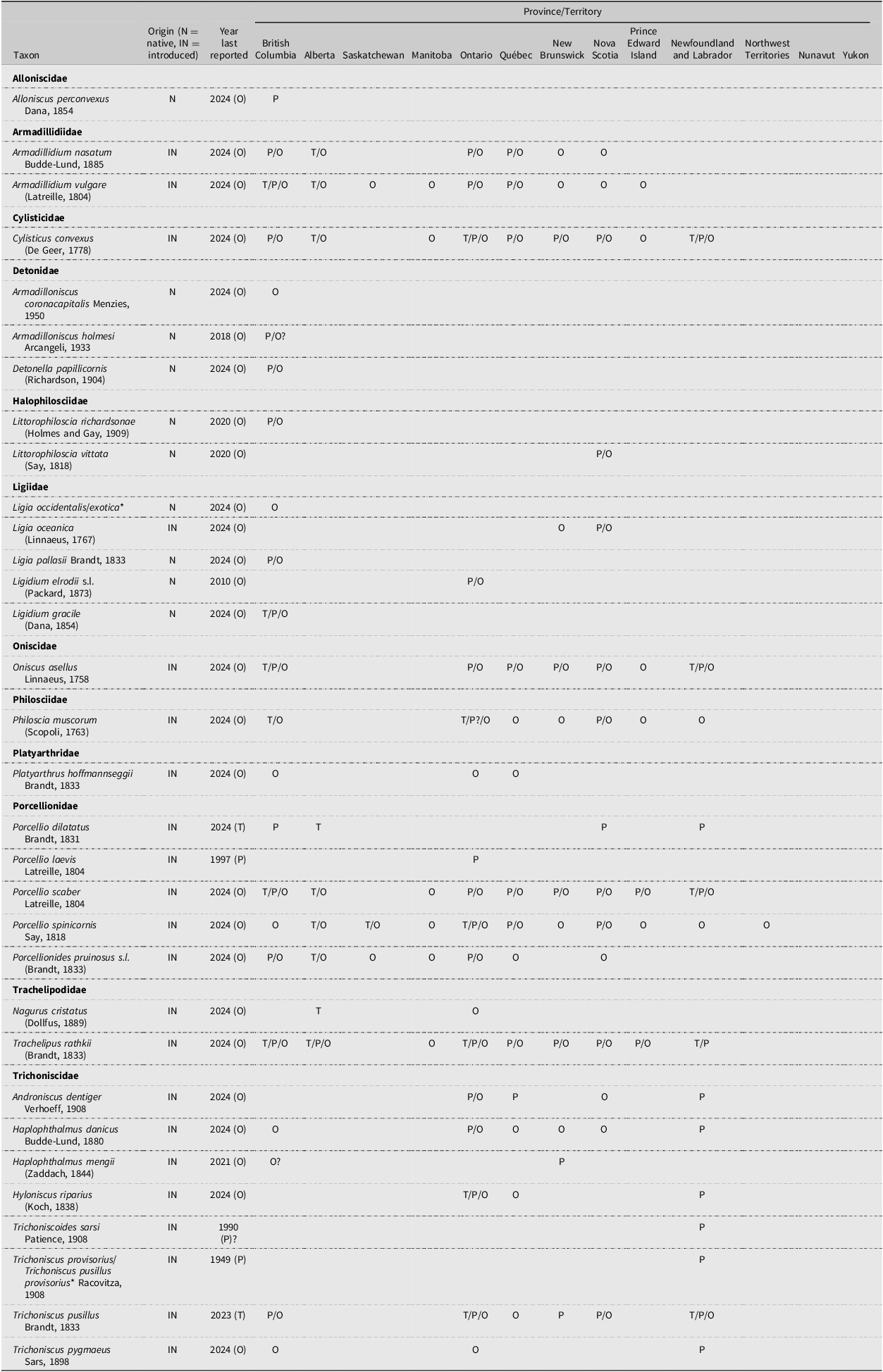
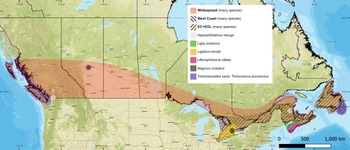
The terrestrial isopods (Isopoda: Oniscidea) included in this checklist have been reported from Canada. They comprise 32 well-documented species and four species with dubious records. Table 1 and Fig. 3 provide a summary of species recorded from each province and territory and generalised species distribution maps. These generalised species distributions represent the three most common distribution patterns that we observed among the species in Canada, which are as follows: west coast – coastal British Columbia; EC+EGL – east coast and the St. Lawrence River to the eastern Great Lakes (Lake Ontario and Lake Erie); and widespread – west coast and EC+EGL. Distribution maps depicting exact distributions are also available for each species (Supplementary material, Fig. S1). Most of the records listed in the present study were originally compiled as part of Stormer (Reference Stormer2024), and some were previously reported in Stormer and Proctor (Reference Stormer and Proctor2021, Reference Stormer and Proctor2022).
Summary of the species of terrestrial isopods (Isopoda: Oniscidea) reported from Canada. T, record from the present study; P, record from previous publications; O, other record (e.g., iNaturalist; BugGuide; GBIF.org record not associated with a publication); ?, record is uncertain, see details in Results; N, species is native to North America; IN, species is introduced to North America; *, the species identity associated with these records is unclear – see the main text for details. See Supplementary material, File S2, for the record source(s) for each species

Common distribution patterns of terrestrial isopod species (Isopoda: Oniscidea) found in Canada. West coast (only in British Columbia): Alloniscus perconvexus, Armadilloniscus holmesi, Armadilloniscus coronacapitalis, Detonella papillicornis, Ligia occidentalis, Ligia pallasii, Ligidium gracile, and Littorophiloscia richardsonae. EC+EGL (east coast and the St. Lawrence River to the eastern Great Lakes (Lake Ontario and Lake Erie)): Androniscus dentiger, Porcellio laevis, and Hyloniscus riparius. Widespread (west coast and EC+EGL): Armadillidium nasatum, Armadillidium vulgare, Cylisticus convexus, Haplophthalmus danicus, Oniscus asellus, Philoscia muscorum, Platyarthrus hoffmannseggii, Porcellio dilatatus, Porcellio scaber, Porcellio spinicornis, Porcellionides pruinosus, Trachelipus rathkii, Trichoniscus pusillus, and Trichoniscus pygmaeus. See Supplementary material, Fig. S1, for individual distribution maps for each species. See the main text for a specific description of the ranges for each species, and see Supplementary material, File S2, for a list of records for each species. Maps show only the Canadian distribution for each species; for distribution in other parts of North America, see Jass and Klausmeier (Reference Jass and Klausmeier2000, Reference Jass and Klausmeier2001). The basemap is the ESRI street map (https://server.arcgisonline.com/arcgis/rest/services/World_Street_Map/MapServer).

Notes about the checklist
Records are listed in alphabetical order by family, then genus, and then species. Most of the families included in this list belong to the infraorder (a.k.a., “section”) Crinocheta (Dimitriou et al. Reference Dimitriou, Taiti and Sfenthourakis2019), except for Ligiidae (Diplocheta) and Trichoniscidae (Synocheta).
Recent molecular evidence suggests Ligiidae is polyphyletic, with Ligia more closely related to marine isopods (Dimitriou et al. Reference Dimitriou, Taiti and Sfenthourakis2019). This would exclude Ligia from Oniscidea, and Ligidium and other genera formerly in Ligiidae would be assigned to the new family “Ligidiidae” (Dimitriou et al. Reference Dimitriou, Taiti and Sfenthourakis2019). These changes would mean that Ligia is not an oniscidean isopod and therefore should not be included in this review and that Ligidium should be listed under the family Ligidiidae instead of Ligiidae. However, both of these changes are debated on the grounds that they do not match morphological evidence that supports the monophyly of Oniscidea (Tabacaru and Giurginca Reference Tabacaru and Giurginca2021), and recent transcriptomic and genomic evidence suggests Ligia nests within a monophyletic Oniscidea (Thorpe Reference Thorpe2024). For these reasons, and because Ligia has traditionally been considered part of Oniscidea, it is included in this list for continuity with older lists of sowbugs. The name “Ligiidae” is also used throughout this paper to include both Ligia and Ligidium.
The Canadian distribution for each species is described broadly to shorten the length of this section, and it includes both previous records and new records from the present study. See Supplementary material, File S2, for a detailed summary of previous Canadian records for each species and for the source of each record, and Supplementary material, File S3, for the complete dataset of all species records.
The term “new collections” refers to specimens collected and examined for the present study. The unaccessioned specimens from the E.H. Strickland Entomological Museum teaching collections (University of Alberta) that were examined and identified for the present study are also listed under “new collections” because we found no evidence that these records had been previously published. Records associated with specimens from the E.H. Strickland Entomological Museum teaching collections are referenced as “Strickland.”
The “First Canadian record” is the earliest collection date for a species in Canada, including all sources from which records were obtained for the present study (see Methods). The date of collection for published records was considered equivalent to the publication date unless otherwise specified in the publication (e.g., records without a collection date in a publication from 1929 were assumed to have been collected in 1929).
Similar habitat types are listed separately whenever they may not be identical (e.g., “sand” and “beach” are not combined because sand may occur in areas other than beaches).
Determining the specific native locality (or localities) for each species is outside the scope of this review, and the native range therefore is indicated broadly for most species (e.g., “Europe” instead of “southwestern Europe with the exclusion of the following areas…”).
Due to the journal’s design template, which features italicised third-level headings, species and genera names in the checklist headings are not italicised. This was done to distinguish the names from the headings’ background italics.
Alloniscidae
Alloniscus perconvexus Dana, 1854
First Canadian record. 1926, Vancouver Island, British Columbia (Hatch Reference Hatch1947).
Canadian distribution. West coast (Fig. 3), recorded only from Vancouver Island.
Canadian habitat. Marine littoral (Austin Reference Austin1985); creates burrows in wet beach sand (Hatch Reference Hatch1947; Schultz Reference Schultz1984a).
Origin. Native to the Pacific coast of North America (Hatch Reference Hatch1947; Schultz Reference Schultz1984a; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Armadillidiidae
Armadillidium nasatum Budde-Lund, 1885
First Canadian record. 1914, London, Ontario (Ross Reference Ross1914).
Canadian distribution. Widespread (Fig. 3), excluding Saskatchewan, Manitoba, Prince Edward Island, and Newfoundland and Labrador.
Canadian habitat. Common in greenhouses (Spencer Reference Spencer1942; Hatch Reference Hatch1947, Reference Hatch1949) but also found in garbage dumps and on grassy plains near creeks (Judd Reference Judd1965).
New collections. ALBERTA: Edmonton, Westerose.
New collections – habitat. ALBERTA: in greenhouses under rocks and concrete debris resting on damp soil, under pots sitting on rocks and gravel, and in damp concrete drainage channels; in the vicinity of greenhouses under cinder blocks, under boards and wooden debris on gravel and landscape fabric, under wooden pallets resting on grass (Poaceae), under pots on mulch, grass, and damp landscape fabric; under rocks near buildings in urban areas.
Origin. Native to Europe; introduced to North America (Hatch Reference Hatch1949; Lindroth Reference Lindroth1957; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2000; Schmalfuss Reference Schmalfuss2004). Canadian Food Inspection Agency import records from 2015 to 2022 list a shipment of 60 Armadillidium sp. from the United States of America to British Columbia in 2022 (Canadian Food Inspection Agency 2022).
Armadillidium vulgare (Latreille, 1804)
First Canadian record. 1914, London, Ontario (Ross Reference Ross1914).
Canadian distribution. Widespread (Fig. 3), excluding Newfoundland and Labrador.
Canadian habitat. Common in greenhouses throughout its distribution (Walker Reference Walker1927; Spencer Reference Spencer1942; Hatch Reference Hatch1947; Judd Reference Judd1965; Rafi and Thurston Reference Rafi and Thurston1982) but has also been found in garbage dumps, fields, and riverside wooded slopes (Judd Reference Judd1965); in dry conditions (Dexter et al. Reference Dexter, Hahnert, Beatty and Downhower1988); in leaf litter, under shrubs, and under boards (Taylor and Carefoot Reference Taylor and Carefoot1993); and associated with buildings (Walker Reference Walker1927; Taylor and Carefoot Reference Taylor and Carefoot1993; Orstan et al. Reference Orstan, Jass and Klausmeier2008). Austin (Reference Austin1985) lists this species as “supralittoral.”
New collections. BRITISH COLUMBIA: Parksville, Qualicum Beach, Lake Country; ALBERTA: Edmonton (Strickland; present study), Lethbridge, Coaldale, Drumheller.
New collections – habitat. BRITISH COLUMBIA: under rotting logs; in urban areas; ALBERTA: in urban yards; under rocks, mulch, gravel, and pots in urban areas; on moss in a greenhouse by an artificial waterfall; in riparian poplar forest in urban areas; in ornamental planters inside a large building; inside a school.
Origin. Native to the Mediterranean, introduced to North America (Lindroth Reference Lindroth1957; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). An individual observed in 1927 in Toronto, Ontario, was associated with imported vegetables, which were thought to be a potential route of sowbug introduction (Walker Reference Walker1927). Canadian Food Inspection Agency import records from 2015 to 2022 list a shipment of 60 Armadillidium sp. from the United States of America to British Columbia in 2022 (Canadian Food Inspection Agency 2022).
Cylisticidae
Cylisticus convexus (De Geer, 1778)
First Canadian record. 1876, Niagara, Ontario (Stuxberg Reference Stuxberg1876).
Canadian distribution. Widespread (Fig. 3), excluding Saskatchewan.
Canadian habitat. Under decaying bark and stones (Wallace Reference Wallace1919); under stones and vegetation associated with brooks, pastures, and buildings (Palmén Reference Palmén1951; Judd Reference Judd1965); in garbage dumps (Palmén Reference Palmén1951; Judd Reference Judd1965; Rafi and Thurston Reference Rafi and Thurston1982); under rocks in a field (GBIF.org 1926b); in grassland, by harbours (Palmén Reference Palmén1951); under debris on beaches and salt marshes (Bousfield Reference Bousfield1962); in wooded areas (Judd Reference Judd1963, Reference Judd1965; Rafi and Thurston Reference Rafi and Thurston1982); in damp areas (Dexter et al. Reference Dexter, Hahnert, Beatty and Downhower1988); on golfing greens and lawns, in marshes (Judd Reference Judd1965); in greenhouses (Judd Reference Judd1965; Rafi and Thurston Reference Rafi and Thurston1982); under boards (Wright Reference Wright1989).
New collections. ALBERTA: Edmonton; Calgary; ONTARIO: Port Hope; NEWFOUNDLAND AND LABRADOR: Fortune.
New collections – habitat. ALBERTA: in urban areas under damp places associated with cinderblocks, mulch, rocks, bricks, concrete rubble, compost bins, plastic raised-bed pieces, basketball hoop bases, leaves, and piles of soil; indoors in basements; in greenhouses under rocks, pots sitting on damp gravel and concrete, and in damp concrete drainage channels; in the vicinity of greenhouses under wooden pallets and in wet leaf litter; ONTARIO: under bricks bordering an urban garden bed; NEWFOUNDLAND AND LABRADOR: under rocks in a vacant lot.
Origin: Native to Europe and Asia, introduced to North America (Hatch Reference Hatch1947; Lindroth Reference Lindroth1957; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Note. This species typically exhibits a dark brown colouration, but a population in the Biological Sciences greenhouse (University of Alberta) consisted almost entirely of pale-coloured individuals (Fig. 4). Other species of Cylisticus (e.g., Cylisticus esterelanus Verhoeff, 1917) exhibit similar depigmentation (Gabiot et al. Reference Gabiot, Portalier, Bantwell and Ponel2020). However, the greenhouse population did not appear to be morphologically different from Cylisticus convexus, with typical colouration from other sites in Alberta, and CO1 from a depigmented individual was 100% identical to C. convexus from Germany (GenBank MN810826.1; https://www.ncbi.nlm.nih.gov/nucleotide/) and was more than 99% identical to CO1 from C. convexus collected for the present study from other sites in Alberta and from Ontario. When a few individuals were kept in a container with dead leaves (food) and soil, the colouration gradually darkened over several weeks until it resembled the typical colouration for this species, and offspring of these individuals exhibit the typical dark colouration for this species (H.G.S., unpublished data). There appear to be no published records of atypical colouration in C. convexus, although captive populations of a cylisticid resembling C. convexus include a “pied” form (e.g., Smug Bug Reference Bug2019).
Terrestrial isopods with unusual pigmentation collected from Canada in the present study: A, bright orange Oniscus asellus from Prince George, British Columbia; B, grey Oniscus asellus from Vancouver, British Columbia (left) with a dull orange individual from the same population (right); C, light brown, dark brown, and pale orange Cylisticus convexus from the Biological Sciences Greenhouse, University of Alberta; D, depigmented Trachelipus rathkii with red eyes from Edmonton, Alberta, with a pigmented individual from the same population; and E, bright orange Porcellio scaber from Bamfield, British Columbia (left), with a grey individual from the same population (right). Images have been edited to remove debris. Colour balance and brightness have been adjusted to reflect the natural appearance of the sowbugs.

Detonidae
Armadilloniscus coronacapitalis Menzies, 1950
This species has not been reported from Canada in previous publications.
First Canadian record. 2024, British Columbia (shawnb2 2024a, 2024b, 2024c).
Canadian distribution. West coast (Fig. 3), recorded only from Hesquiat, British Columbia.
Canadian habitat. Salt marsh, under driftwood (shawnb2 2024a). Specimens from California have been found under rocks on shorelines (Menzies Reference Menzies1950).
Origin. Native to the northwestern coast of North America (Jass and Klausmeier Reference Jass and Klausmeier2001). The coast of British Columbia represents the northernmost extent of the distribution of this species.
Armadilloniscus holmesi Arcangeli, 1933
Whether this species still occurs in Canada is doubtful, if indeed it ever did occur. Only two Canadian records of it are known, both from Calvert Island, British Columbia (Austin Reference Austin1985; GBIF.org 2018a). Austin (Reference Austin1985) does not provide a description or illustration of the specimen(s). A similar species, Armadilloniscus coronacapitalis (see Armadilloniscus coronacapitalis section, above), also occurs on the British Columbia coast. Although apparently lacking the flange on the penultimate article of the antennae flagella that would be present in A. coronacapitalis (Menzies Reference Menzies1950), the specimen collected in 2018 that is associated with the GBIF.org record displays antennal proportions consistent with A. coronacapitalis and contrary to those of A. holmesi (H.G.S., unpublished data; Shawn Brescia, personal communication; based on photos of the specimen in the University of Florida Invertebrate Zoology Collection, Gainesville, Florida, United States of America). Besides Austin (Reference Austin1985), only two published records of the species from further north than California, United States of America, currently exist (Hatch Reference Hatch1947; George and Stromberg Reference George and Strömberg1968); both were collected from Washington state, United States of America, more than 50 years ago. Armadilloniscus holmesi was not found in a search for the species on Dog Beach (Calvert Island) in 2025, although A. coronacapitalis was present (Shawn Brescia, personal communication). Past reports of A. holmesi from Canada may therefore have been misidentifications of A. coronacapitalis.
First Canadian record. 1985, British Columbia (Austin Reference Austin1985).
Canadian distribution. West coast (Fig. 3), recorded only from Calvert Island, British Columbia.
Canadian habitat. Marine littoral (Austin Reference Austin1985).
Origin. Found along the North American Pacific coast, including in Mexico and in the United States of America, and also in Japan (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Leistikow and Wägele Reference Leistikow and Wägele1999; Schmalfuss Reference Schmalfuss2004). The North American population is thought to be native (Jass and Klausmeier Reference Jass and Klausmeier2001). The British Columbia coast would therefore represent the northernmost edge of the distribution of this species in North America.
Detonella papillicornis (Richardson, 1904)
Only five records of this species have been reported from Canada (see Supplementary material, File S2).
First Canadian record. 1926, Hammond Bay, British Columbia (Fee Reference Fee1926).
Canadian distribution. West coast (Fig. 3), south of Calvert Island, British Columbia.
Canadian habitat. Found in tide pools (Fee Reference Fee1926).
Origin. In addition to British Columbia records, this species has been recorded from Alaska and Washington, United States of America; from Japan; from Russia and the Kuril Islands (Hatch Reference Hatch1947; Leistikow and Wägele Reference Leistikow and Wägele1999; Schmidt Reference Schmidt2000, Reference Schmidt2002; Schmalfuss Reference Schmalfuss2004). The North American population of this species is thought to be native (Jass and Klausmeier Reference Jass and Klausmeier2001).
Halophilosciidae
Littorophiloscia richardsonae (Holmes and Gay, 1909)
Only two records of this species are reported from Canada, of which one consists of two females from Vancouver Island (Taiti and Ferrara Reference Taiti and Ferrara1986) and the other is a single individual depicted in two photographs on iNaturalist (thomasbarbin 2020). The iNaturalist observation is included in this list because it clearly shows many features that match with descriptions and figures of this species by Van Name (Reference Van Name1936) and Taiti and Ferrara (Reference Taiti and Ferrara1986), including reddish-brown pigmentation with three darker longitudinal lines, tiny spines on the body surface, the arched frontal margin of the head, the telson with deep concave sides, the second article of the flagellum shorter than the most proximal article (the terminal article is obscured in the iNaturalist image), and the uropod exopod 3× as long as endopod, etc.
First Canadian record. 1955, Vancouver Island, British Columbia (Taiti and Ferrara Reference Taiti and Ferrara1986).
Canadian distribution. West Coast (Fig. 3), recorded only from Vancouver Island.
Canadian habitat. Coastal regions (Taiti and Ferrara Reference Taiti and Ferrara1986).
Origin. Native to the Pacific coast of North America (Van Name Reference Van Name1936; Taiti and Ferrara Reference Taiti and Ferrara1986; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Littorophiloscia vittata (Say, 1818)
Only four records of this species are reported from Canada (Bousfield Reference Bousfield1962; Taiti and Ferrara Reference Taiti and Ferrara1986; adamssj 2018; ibycter 2020). The only recent observations both come from iNaturalist. One of these observations (adamssj 2018) depicts two individuals with the telson and many other features are obscured. However, the habitus appears consistent with L. vittata, and the visible features (narrowed pleon and colouration) do not correspond to any other species previously recorded from Canada. The other observation (ibycter 2020) depicts multiple individuals that match descriptions of the distinctive colouration and telson shape of Littorophiloscia vittata (Taiti and Ferrara Reference Taiti and Ferrara1986; Shultz Reference Shultz2018).
First Canadian record. 1958, Sandy Cove, Nova Scotia (Bousfield Reference Bousfield1962).
Canadian distribution. Recorded only from southwestern Nova Scotia (Fig. 3).
Canadian habitat. In a culvert near a pond (Taiti and Ferrara Reference Taiti and Ferrara1986); under Fucus (brown algae) (Fucaceae) and pebbles on a rocky beach (Bousfield Reference Bousfield1962).
Origin. Native to the Atlantic coast of North America (Van Name Reference Van Name1936; Taiti and Ferrara Reference Taiti and Ferrara1986; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Ligiidae
Ligia occidentalis/exotica
More than 570 iNaturalist observations from the south coast of British Columbia (Vancouver area) depict a species of Ligia that has been identified as Ligia occidentalis (iNaturalist community 2024de); this species has not been reported from Canada in previous publications. The distribution of L. occidentalis extends only as far north as California (Hurtado et al. Reference Hurtado, Mateos and Santamaria2010; Markow and Pfeiler Reference Markow and Pfeiler2010; Santamaria et al. Reference Santamaria, Mateos, DeWitt and Hurtado2016). If L. occidentalis occurs in the Vancouver area, this would leave a gap of approximately 700 km between the population in Vancouver and the population in California, assuming the gap is due to true absence and not to a lack of records. This gap could be explained by L. occidentalis having dispersed along the coast as far north as Vancouver and then disappearing from Washington and Oregon, United States of America, or by the introduction of L. occidentalis to the Vancouver area through human activity or drifting materials. However, because members of the genus Ligia are morphologically very similar (Van Name Reference Van Name1936), the unusual distribution casts doubt on whether the identification of Ligia occidentalis in Vancouver is correct. Two other Ligia species occur on the Pacific coast of the United States of America: L. pallasii Brandt, 1833 and L. exotica Roux, 1828 (Jass and Klausmeier Reference Jass and Klausmeier2000, Reference Jass and Klausmeier2001). Ligia pallasii has eyes positioned further apart than L. occidentalis and L. exotica do (gap between the eyes in L. pallasii is twice the horizontal width of the eye versus equal to the width of the eye in L. exotica) and has uropod bases with the length almost equal to the width versus greater than the width in L. occidentalis and L. exotica (Van Name Reference Van Name1936; Hiebert Reference Hiebert, Hiebert, Butler and Shanks2015). Observations from Vancouver clearly depict individuals with rectangular uropod bases and eyes positioned close together (e.g., pumakit 2020; lynnmoss 2022), which suggests the species is not L. pallasii. Distinguishing L. exotica from L. occidentalis is difficult: Van Name (Reference Van Name1936) noted the high similarity between the two species and distinguished them based on the blunt (“obtuse”) telson in L. occidentalis versus the pointed (“sharply angular”) telson in L. exotica. Jackson (Reference Jackson1922) separated the two based on antennal length in males, with “antennae shorter than [the] thorax” (= not reaching past the end of the pereon?) in L. occidentalis but longer than the thorax in L. exotica. Determining the sex of individuals reported in iNaturalist observations is difficult because the ventrally located male pleopods are frequently not visible, but most individuals in the Vancouver population appear to have antennae shorter than the pereon, which would be consistent with L. occidentalis (e.g., chloe_and_trevor 2020; comox 2020; marilynneb 2020b; saxifrages 2022; rebeccareaderlee 2023; slemtime 2023). However, many individuals from the same area also appear to have pointed telsons (chloe_and_trevor 2020; marilynneb 2020a, 2020b; doviende 2022; benkeen 2023), which would be consistent with L. exotica rather than L. occidentalis. More than one species of Ligia may be present, or antennae length and telson shape may not reliably distinguish between L. exotica and L. occidentalis. As is the case for L. occidentalis, L. exotica has been reported only as far north as California (Jass and Klausmeier Reference Jass and Klausmeier2001). It is therefore currently not possible to assign the species in Vancouver to L. occidentalis rather than L. exotica.
Members of the Vancouver Ligia population may be a separate species even if they appear to morphologically match L. occidentalis. Based on molecular genetic analyses, populations of L. “occidentalis” on the south Pacific coast of North America were found to represent a cryptic species complex whose members cannot be differentiated morphologically (Hurtado et al. Reference Hurtado, Mateos and Santamaria2010; Markow and Pfeiler Reference Markow and Pfeiler2010; Santamaria et al. Reference Santamaria, Mateos, DeWitt and Hurtado2016). The northern populations of L. “occidentalis” found in British Columbia may also be a morphologically cryptic undescribed species. Molecular and morphological comparison of Vancouver and southern populations of L. occidentalis would help resolve the identity of the Vancouver population.
First Canadian record. 2010, Vancouver, British Columbia (noammarkus 2010).
Canadian distribution. West coast (Fig. 3), recorded only south of Surge Narrows, British Columbia.
Canadian habitat. No records of Canadian habitat for this species are published. Van Name (Reference Van Name1936) reports this species from rocks along the Pacific coast.
Origin. Native to the southern Pacific coast of North America (Van Name Reference Van Name1936; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Ligia oceanica (Linnaeus, 1767)
First Canadian record. 1958, Sandy Cove, Nova Scotia (Bousfield Reference Bousfield1962).
Canadian distribution. Coast of Nova Scotia and New Brunswick around the Bay of Fundy (Fig. 3).
Canadian habitat. On rocky beach under Fucus (brown algae) and rocks (Bousfield Reference Bousfield1962).
Origin. Native to Europe, introduced to North America (Van Name Reference Van Name1936; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Ligia pallasii Brandt, 1833
First Canadian record. 1878, Victoria, British Columbia (Smith Reference Smith1879).
Canadian distribution. West coast (Fig. 3).
Canadian habitat. On rocky shore under flat rocks (Walker Reference Walker1927); on large rocks (Fee Reference Fee1926); in gaps in rocky cliffs (Carefoot Reference Carefoot1973); associated with seaweed (Carefoot Reference Carefoot1979); on cobble beaches (Zimmer et al. Reference Zimmer, Danko, Pennings, Danford, Ziegler, Uglow and Carefoot2001, Reference Zimmer, Danko, Pennings, Danford, Carefoot, Ziegler and Uglow2002); under boulders next to cliffs (Carefoot et al. Reference Carefoot, Wright, Pennings, Ziegler, Zimmer and Uglow2000; Wright et al. Reference Wright, Carefoot and Albers2003); on logs (GBIF.org 1905).
Origin. Native to the Pacific coast of North America (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Ligidium elrodii s.l. (Packard, 1873)
Populations of L. elrodii from the southern Appalachian Mountains were recently found to represent separate lineages (Recuero and Caterino Reference Recuero and Caterino2024, Reference Recuero and Caterino2025). Ontario populations were not included in Recuero and Caterino (Reference Recuero and Caterino2024, Reference Recuero and Caterino2025); however, because the lineages differ in male pleopod morphology (Recuero and Caterino Reference Recuero and Caterino2024, Reference Recuero and Caterino2025), morphological examination of Ontario L. elrodii populations may make it possible to assign the records in Ontario to one or more of the Appalachian lineages.
First Canadian record. 1876, Niagara, Ontario (Stuxberg Reference Stuxberg1876).
Canadian distribution. Ontario, south of Lake Simcoe (Fig. 3).
Canadian habitat. Under pebbles beside streams; on wet moss in a swamp; under dead leaves and humus in damp woods (Walker Reference Walker1927); on creek banks, in damp wooded areas (Walker Reference Walker1927; Judd Reference Judd1965).
Origin. Recorded only from northeastern North America (Leistikow and Wägele Reference Leistikow and Wägele1999; Schmalfuss Reference Schmalfuss2004). Jass and Klausmeier (Reference Jass and Klausmeier2001) list five entries for L. elrodii: L. elrodii is described as an “immigrant to Canada and the U.S.” but four subspecies of L. elrodii are listed as native to this area (Jass and Klausmeier Reference Jass and Klausmeier2001, p. 10). Jass and Klausmeier’s (Reference Jass and Klausmeier2001) statement that L. elrodii is an immigrant is likely an error because the absence of records of this species from any other part of the world suggests it is native to North America.
Ligidium gracile (Dana, 1854)
First Canadian record. 1913, Departure Bay, British Columbia (Walker Reference Walker1927).
Canadian distribution. West Coast (Fig. 3).
Canadian habitat. Under pebbles at brook edge (Walker Reference Walker1927); under dead leaves and logs in damp wooded areas (Walker Reference Walker1927; Fee Reference Fee1926); in woods in urban areas (Spencer Reference Spencer1942); along the coast (Johansen Reference Johansen1926).
New collections. BRITISH COLUMBIA: French Beach, Vancouver Island.
New collections – habitat. BRITISH COLUMBIA: under rocks near seaside cliffs.
Origin. Native to the Pacific coast of North America (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Oniscidae
Oniscus asellus Linnaeus, 1758
First Canadian record. 1871, Niagara, Ontario (Stuxberg Reference Stuxberg1873).
Canadian distribution. Widespread (Fig. 3) excluding Alberta, Saskatchewan, and Manitoba
Canadian habitat. Occupies a wide variety of environments: in urban areas, the species has been found in greenhouses (Ross Reference Ross1914; Spencer Reference Spencer1942; Hatch Reference Hatch1947; Judd Reference Judd1965), under stones, leaves, and boards around buildings (Walker Reference Walker1927; Palmén Reference Palmén1951; Rafi and Thurston Reference Rafi and Thurston1982; Wright Reference Wright1989; Taylor and Carefoot Reference Taylor and Carefoot1993), in gardens (Ross Reference Ross1914; Spencer Reference Spencer1942; Rafi and Thurston Reference Rafi and Thurston1982), in basements (Ross Reference Ross1914), on lawns and golfing greens (Judd Reference Judd1965), in dumps and garbage cans (Judd Reference Judd1965), in association with railway stations (Palmén Reference Palmén1951), and in woods near urban areas (Alikhan Reference Alikhan1995). Outside of urban areas, this species has been found in ravines and wooded areas (Ross Reference Ross1914; Palmén Reference Palmén1951; Judd Reference Judd1965; Dexter et al. Reference Dexter, Hahnert, Beatty and Downhower1988; Wright Reference Wright1995), by river banks (Judd Reference Judd1965) and lakes (Wright Reference Wright1995), along the coast (Palmén Reference Palmén1951), under debris on beaches (Bousfield Reference Bousfield1962), in fields and marshes (Judd Reference Judd1965), in moss and lichens (Wright Reference Wright1995), and under debris on shorelines (Bousfield Reference Bousfield1956b).
New collections. BRITISH COLUMBIA: Prince George, Vancouver, Parksville, Qualicum Beach, Bamfield; NEWFOUNDLAND AND LABRADOR: Channel–Port aux Basques, J.T. Cheeseman Provincial Park, Barachois Pond Provincial Park, Pasadena, Pynn’s Brook, Notre Dame Provincial Park, St. John’s, Butter Pot Provincial Park, La Manche Provincial Park, Mount Moriah.
New collections – habitat. BRITISH COLUMBIA: in yards and under wet decaying logs in urban areas; under planters and pots associated with buildings; climbing up the outside walls of cabins at night; NEWFOUNDLAND AND LABRADOR: under rocks in disturbed open areas; in vegetation at the edges of forests and roadsides; in gravel, sand, and moss on river banks; in mixed and conifer forests.
Origin. Native to Western Europe, introduced to North America (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Lindroth Reference Lindroth1957; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
The presence of this species only at railway stations in the northern part of Newfoundland in 1949 suggests that it could be dispersed by train (Palmén Reference Palmén1951). This species and others are thought to have been introduced to Kejimkujik National Park, Nova Scotia, through farming activity (Wright Reference Wright1995). Walker (Reference Walker1927) noted that this species was rare in Toronto, Ontario, in 1915 but had become common by 1927. He noted that it was found only in urban environments and concluded that the species must have arrived in Toronto not long before. Similarly, Spencer (Reference Spencer1942) noted that the species was uncommon in Vancouver, British Columbia, in 1929 but had become abundant by 1942.
Note. This species typically exhibits a dark grey pigmentation, but a single orange individual was present in each of two populations in British Columbia in collections made for the present study (Fig. 4). Orange-coloured individuals of species with typically dark colouration have been documented in several families of terrestrial isopods, including Armadillidae, Armadillidiidae, Detonidae, Philosciidae, Porcellionidae, Platyarthridae, and Trachelipodidae (Sutton Reference Sutton1972; Johnson Reference Johnson1976; Sassaman and Garthwaite Reference Sassaman and Garthwaite1980; George Reference George1986; Garthwaite Reference Garthwaite1988; Negishi et al. Reference Negishi, Hasegawa, Martin, Juchault and Katakura1994). There appear to be no published records of an orange form of Oniscus asellus or other Oniscidae. However, orange O. asellus have been documented by terrestrial isopod enthusiasts and are kept as pets, with captive-bred populations thought to have originated from British Columbia (Smug Bug Reference Bug2021).
Philosciidae
Philoscia muscorum (Scopoli, 1763)
McAlpine and Oldham (Reference McAlpine and Oldham2011) state that this species has been recorded from Ontario by Rafi and Thurston (Reference Rafi and Thurston1982), but we were unable to find mention of this species or any synonyms in this source.
First Canadian record. 1958, Grosses Coques, Nova Scotia (Bousfield Reference Bousfield1962).
Canadian distribution. Widespread (Fig. 3) excluding Alberta, Saskatchewan, and Manitoba.
Canadian habitat. On gravel beach under beach drift (Bousfield Reference Bousfield1962), under rocks and logs in wooded areas (Wright Reference Wright1995).
New collections. BRITISH COLUMBIA: Qualicum Beach, Parksville; ONTARIO: Port Hope.
New collections – habitat. BRITISH COLUMBIA: from an urban yard; ONTARIO: under logs and bark by disturbed creekside trail.
Origin. Native to Europe, introduced to North America (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Lindroth Reference Lindroth1957; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). Van Name (Reference Van Name1936) thought this species could have been introduced to North America via imported plants. This species and others are thought to have been introduced to Kejimkujik National Park, Nova Scotia, via farming activity (Wright Reference Wright1995).
Platyarthridae
Platyarthrus hoffmannseggii Brandt, 1833
This species has not been reported from Canada in previous publications. We did not encounter this species in our collections; however, Platyarthrus hoffmannseggii and P. aiasensis Legrand, 1954 are the only Platyarthrus found in North America (Schmalfuss Reference Schmalfuss2004), and the latter species has clear ridges on the pereon that P. hoffmannseggii lacks (Bakhshi and Sadeghi Reference Bakhshi and Sadeghi2019; Abidi and Hamaied Reference Abidi and Hamaied2024). The numerous photographs of a smooth-bodied Platyarthrus from Canada on iNaturalist and BugGuide therefore are assigned to P. hoffmannseggii.
First Canadian record. 2016, Chateauguay, Quebec (Ulrich Reference Ulrich2016).
Canadian distribution. Widespread (Fig. 3) but recorded only from Vancouver, British Columbia; southern Ontario; and along the St. Lawrence River in Quebec and Ontario.
Canadian habitat. This species is found in association with ants (Harding and Sutton Reference Harding and Sutton1985). There are no published records of Canadian habitat for this species.
Origin. Introduced to North America, native to Europe, Africa, and Asia (Lindroth Reference Lindroth1957; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). Lindroth (Reference Lindroth1957) proposed that this species was introduced to North America via soil ballast in ships.
Porcellionidae
Porcellio dilatatus Brandt, 1831
Only five records of this species are reported from Canada, two of which (our own collections from greenhouses in Alberta) are recent records. This species, therefore, may be currently limited to greenhouses in its Canadian range.
First Canadian record. 1946, Burnaby, British Columbia (Hatch Reference Hatch1947).
Canadian distribution. Widespread (Fig. 3) but rare; recorded only from Burnaby, British Columbia; Edmonton, Alberta; Sable Island, Nova Scotia; and the island of Newfoundland.
Canadian habitat. In greenhouses (Hatch Reference Hatch1947; Palmén Reference Palmén1951), near railway stations (Palmén Reference Palmén1951).
New collections: ALBERTA: St. Albert, Edmonton.
New collections – habitat. ALBERTA: in a greenhouse under boards and buckets resting on a mix of damp soil and gravel; in a greenhouse in gravel under pots.
Origin. Native to Europe, introduced to North America (Hatch Reference Hatch1947; Lindroth Reference Lindroth1957; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Porcellio laevis Latreille, 1804
Only four records of this species are reported from Canada, of which most are questionable, and the species has not been reported from Canada in almost 30 years. Thompson (Reference Thompson1932) identified a species from Ontario as “probably” P. laevis, but no illustrations or additional notes are provided to help confirm the identification. It is not possible to tell what characteristics were used to identify the P. laevis that Dexter et al. (Reference Dexter, Hahnert, Beatty and Downhower1988) recorded from Ontario because the cited key (Hatchett Reference Hatchett1947) does not include P. laevis, and additional literature used to identify collected sowbugs is not cited. The authors state that the P. laevis specimens were missing at the time the article was written. The GBIF.org record of P. laevis from Nova Scotia does not include a photograph of the specimen (GBIF.org 1927), and we were unable to verify its identity. However, the collections by Belaoussoff et al. (Reference Belaoussoff, Guenther, Kevan, Murphy and Swanton1998) from Ontario likely do represent P. laevis, because the authors created a key that mentions the characteristically smooth dorsal surface of this species.
This species appears to be declining in parts of its European range (Harding Reference Harding2016; Boeraeve et al. Reference Boeraeve, Arijs, Segers and De Smedt2021).
First Canadian record. 1927, Nova Scotia (GBIF.org 1927), but see the note, above, for this species.
Canadian distribution. EC+EGL (Fig. 3) but recorded only from Ontario south of Kitchener and possibly also from Nova Scotia (see the note, above, for this species).
Canadian habitat. In corn, soy, and wheat fields (Belaoussoff et al. Reference Belaoussoff, Guenther, Kevan, Murphy and Swanton1998). Thompson (Reference Thompson1932) documented an enormous population of sowbugs in Ontario that was associated with a garbage dump and basements; however, the identification of these species as P. laevis is questionable (see the note, above, for this species). The GBIF.org record from Nova Scotia (GBIF.org 1927) is recorded on an accession card from the Smithsonian National Museum of Natural History that says the specimen was found “in soil about roots of plants,” as a transfer associated with the “Fed. Hort. Bd” (presumably referring to the Federal Horticulture Board). The card also includes the note “Boston #3655.” Possibly, the specimen was recovered from a shipment of plants between Boston and Nova Scotia, but which location represents the receiving end is unclear.
Origin. Native to Europe and Northern Africa, introduced to North America (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Lindroth Reference Lindroth1957; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). Human-mediated dispersal to new areas via plant shipment has been documented (see note for this species under “Canadian habitat,” above).
Porcellio scaber Latreille, 1804
First Canadian record. 1859, Gulf of Georgia, British Columbia (GBIF.org 1859).
Canadian distribution. Widespread (Fig. 3), including Labrador but excluding Saskatchewan.
Canadian habitat. Found in a wide variety of urban and natural habitats. This species has been recorded from greenhouses (Spencer Reference Spencer1942; Hatch Reference Hatch1947; Judd Reference Judd1965); in gardens and yards (Judd Reference Judd1965; Rafi and Thurston Reference Rafi and Thurston1982); associated with buildings (Spencer Reference Spencer1942; Palmén Reference Palmén1951; Judd Reference Judd1965; Taylor and Carefoot Reference Taylor and Carefoot1993); in garbage dumps (Judd Reference Judd1965); in compost heaps (Bhella et al. Reference Bhella, Fung, Harrison, Ing, Larsen and Selby2006); under boards (Judd Reference Judd1965; Wright Reference Wright1989; Taylor and Carefoot Reference Taylor and Carefoot1993); on shorelines (Richardson Reference Richardson1905); under driftwood (Wallace Reference Wallace1919; Walker Reference Walker1927), stones, and dried seaweed (Bousfield Reference Bousfield1956b); in decayed wood and under logs (Johansen Reference Johansen1924; Wright Reference Wright1995); under stones (Packard Reference Packard1867; Johansen Reference Johansen1924; Wright Reference Wright1995); among decayed leaves (Fee Reference Fee1926; Rafi and Thurston Reference Rafi and Thurston1982; Taylor and Carefoot Reference Taylor and Carefoot1993); by marshes (Walker Reference Walker1927) and lakes (Wright Reference Wright1995); in woods (Palmén Reference Palmén1951; Wright Reference Wright1995); beside streams (Palmén Reference Palmén1951); in fields of wheat and soy (Belaoussoff et al. Reference Belaoussoff, Guenther, Kevan, Murphy and Swanton1998).
New collections. BRITISH COLUMBIA: Prince George, Vancouver, Sechelt, Parksville, Qualicum Beach, Bamfield, Port Clements, Masset (Strickland; present study); ALBERTA: Edmonton; NEWFOUNDLAND AND LABRADOR: Channel–Port aux Basques, Blomidon Nature Trail, Notre Dame Provincial Park, Traytown, Lockston Path Provincial Park, Trinity, Fortune, Burgeo, St John’s, Corner Brook, Lumsden.
New collections – habitat. BRITISH COLUMBIA: in urban areas in yards and under rotting logs, planters, and pots; under a wooden board on mulch in a small backyard greenhouse; ALBERTA: in urban areas under rocks, plastic containers, bricks, cinderblocks, wood debris, and concrete rubble, and in mulch; under cinderblocks in the vicinity of greenhouses; inside greenhouses under rocks on soil and pots on gravel, and on moss by an artificial waterfall; in a bathroom sink inside a building; NEWFOUNDLAND AND LABRADOR: under rocks in a disturbed open area, by roadsides, along trails, in fields, beside forests, in conifer and mixed forests, and under detritus on sandy coastal beach.
Origin. Usually considered to be of European origin and introduced to North America (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Lindroth Reference Lindroth1957; Vandel Reference Vandel1962; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). Porcellio scaber has been introduced to new localities via shipments of building supplies (Slabber and Chown Reference Slabber and Chown2002). This species and others are thought to have been introduced to Kejimkujik National Park, Nova Scotia, through farming activity (Wright Reference Wright1995).
Some have argued that a subspecies of Porcellio scaber is native to North America. After noting morphological differences between European and North American west coast populations, Arcangeli (Reference Arcangeli1932) named the North American population P. scaber americanus and suggested it was native to North America because the specimens had been collected far from human habitation. Hatch (Reference Hatch1947) agreed with this assessment, noted that the name P. scaber niger Say, 1818 had priority over P. scaber americanus, and added additional records that extended its distribution to the east coast of North America. Hatch (Reference Hatch1947) did not disagree with the designation of a separate North American subspecies but noted that drawings of P. scaber niger from California (Miller Reference Miller1936) differed from those in Arcangeli (Reference Arcangeli1932) and that substantial variation existed in the characteristics used to differentiate the North American and European populations. Palmén (Reference Palmén1951) came to a different conclusion: after examining P. scaber from Newfoundland and Europe and noting the variability in the characteristics separating P. scaber niger from European populations, he suggested that the differences between the two had been exaggerated and recommended further study. Vandel (Reference Vandel1962) stated that P. scaber niger (= P. scaber americanus) was not a valid subspecies because the characters used to differentiate it represented normal variation within P. scaber, and he thought that the North American populations of P. scaber were nonnative. Most recently, Song (Reference Song2020) noted that morphological differences between six subspecies of P. scaber, including P. scaber americanus, represent differences in age rather than subspecific differences. Song (Reference Song2020) concluded that all subspecies of P. scaber are synonymous after failing to find CO1 differences among South Korean populations of P. scaber (including individuals with differing morphology similar to that seen in P. scaber subspecies) and noting that CO1 sequences from P. scaber collected worldwide did not form geographically distinct clades. Comparison of the genomic data of North American and European populations of P. scaber would provide additional insight into the question of whether a cryptic native species or subspecies of P. scaber exists in North America.
Note. Porcellio scaber typically exhibits dark grey, reddish, or light brown pigmentation, but three bright orange individuals (two males, one female) were present in a population in British Columbia in collections made for the present study (Fig. 4). Orange individuals of P. scaber have been documented by George (Reference George1986) and Bhella et al. (Reference Bhella, Fung, Harrison, Ing, Larsen and Selby2006) and are also kept as pets (Smug Bug Reference Bug2021).
Porcellio spinicornis Say, 1818
First Canadian record. 1876, Niagara, Ontario (Stuxberg Reference Stuxberg1876).
Canadian distribution. Widespread (Fig. 3), including northern Alberta, northern Manitoba, and one record from the Northwest Territories (Mongoose 2011). Sowbugs have not been reported from the Northwest Territories in previous publications; the BugGuide observation reports a single individual found near the mouth of the Hay River at Great Slave Lake.
Canadian habitat. Associated with buildings, yards, and lawns (Walker Reference Walker1927; Judd Reference Judd1965; McQueen Reference McQueen1976; Rafi and Thurston Reference Rafi and Thurston1982; Orstan et al. Reference Orstan, Jass and Klausmeier2008); in city gardens and in greenhouses (Rafi and Thurston Reference Rafi and Thurston1982); in woods near urban areas (Alikhan Reference Alikhan1995); under rocks and bricks (Walker Reference Walker1927; Rafi and Thurston Reference Rafi and Thurston1982); under boards (McQueen Reference McQueen1976); under leaves (Rafi and Thurston Reference Rafi and Thurston1982); in garbage dumps and on golfing greens; by marshes (Judd Reference Judd1965); in wooded areas and river banks (Judd Reference Judd1965); under rocks on lake shores (Wright Reference Wright1995); in dry areas (Dexter et al. Reference Dexter, Hahnert, Beatty and Downhower1988).
New collections. ALBERTA: Edmonton, Sherwood Park, St. Albert, Drumheller, Taber, Calgary, Pigeon Lake, Canmore, Claresholm, Crowsnest, Lethbridge, Coaldale (Strickland, present study); SASKATCHEWAN: Regina; ONTARIO: Port Hope.
New collections – habitat. ALBERTA: in urban areas in mulch, gravel, compost piles, leaf piles, and soil piles, under boards, pots, rocks, paving stones, concrete steps, metal parking signs, logs, bricks, and concrete rubble, and by ponds; under rocks on soil and on coniferous needles in urban gardens; in urban yards; crawling on sidewalks, brick walkways, and wooden fences in urban areas; on floors inside buildings (including upper floors of condominiums and public buildings); in sinks; in damp places in basements; in heated garages, by walls outside buildings; under pots on an upper-floor apartment deck; in greenhouses under pots sitting on damp gravel, concrete, and landscape fabric, in gravel, and under concrete on mulch; in gravel in the vicinity of greenhouses; under large rocks in landscaping around a cottage; SASKATCHEWAN: on floor inside building in urban area; ONTARIO: under bricks bordering urban garden bed; under logs and bark by disturbed creekside trail.
Origin. Native to Europe, introduced to North America (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Lindroth Reference Lindroth1957; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). One population in Toronto, Ontario, is assumed to have arrived in a shipment of gravel (McQueen Reference McQueen1976). This species and others are thought to have been introduced to Kejimkujik National Park, Nova Scotia, through farming activity (Wright Reference Wright1995).
Porcellionides pruinosus s.l. (Brandt, 1833)
Porcellionides “pruinosus” represents a complex of several morphologically indistinguishable species (Lefebvre and Marcadé Reference Lefebvre and Marcadé2005; Delhoumi et al. Reference Delhoumi, Zaabar, Bouslama, Zayani and Achouri2019). Porcellionides floria is another species of Porcellionides that occurs in North America, including in the southern United States of America and in Mexico (Garthwaite and Sassaman Reference Garthwaite and Sassaman1985), and is morphologically very similar to P. pruinosus. Characteristics that may differentiate it from P. pruinosus without use of an electron microscope include a smoother dorsal surface, flagellar articles that are more equal in length (although the ratio changes with age), and less distinct white bands on the antennae, but 10% of Porcellionides specimens examined were unidentifiable with these characteristics (Garthwaite and Sassaman Reference Garthwaite and Sassaman1985). Because the northernmost published distribution of P. floria is southern California (Garthwaite and Sassaman Reference Garthwaite and Sassaman1985), all records of Porcellionides from Canada are likely to be a species other than P. floria. Therefore, iNaturalist records of sowbugs from Canada identified as Porcellionides sp. (iNaturalist community 2024df) are included here as Porcellionides pruinosus s.l.
First Canadian record. 1924, De Grassi Point, Ontario (Walker Reference Walker1927).
Canadian distribution. Widespread (Fig. 3), excluding New Brunswick, Prince Edward Island, and Newfoundland and Labrador
Canadian habitat. In manure (Walker Reference Walker1927); in garbage dumps; in greenhouses; in fields; on river banks; in woods (Judd Reference Judd1965); associated with buildings; under leaves; under stones; in gardens (Rafi and Thurston Reference Rafi and Thurston1982); on shorelines (Austin Reference Austin1985); in fields of corn, soy, and wheat (Belaoussoff et al. Reference Belaoussoff, Guenther, Kevan, Murphy and Swanton1998); in woods near urban areas (Alikhan Reference Alikhan1995).
New collections. ALBERTA: Drumheller, Edmonton
New collections – habitat. ALBERTA: in a residential area; in gravel under pots in a greenhouse.
Origin. Native to the Mediterranean region, introduced to North America (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Lindroth Reference Lindroth1957; Vandel Reference Vandel1962; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004), but see the note, above, for this species.
Trachelipodidae
Nagurus cristatus (Dollfus, 1889)
Previous checklists have not reported this species from Canada, and it has, to date, been recorded only twice from Canada. Both records are associated with greenhouses (see note for this species, below, under “Origin”); it is therefore likely that this species is limited to greenhouses in its Canadian range.
First Canadian record. 2022, Edmonton, Alberta (Stormer and Proctor Reference Stormer and Proctor2022).
Canadian distribution. Recorded only from Edmonton, Alberta, and Toronto, Ontario (Fig. 3).
Canadian habitat. In a greenhouse under pots sitting on damp gravel.
Origin. Native to Southeast Asia (Leistikow and Wägele Reference Leistikow and Wägele1999) but found worldwide, including in the southern United States of America (Johnson Reference Johnson1986; Jass and Klausmeier Reference Jass and Klausmeier2000; iNaturalist community 2024dg). Multiple records of this species are noted from greenhouses in Europe (Shaw Reference Shaw1948; Holthuis Reference Holthuis1949; Soesbergen Reference Soesbergen2003; Gregory Reference Gregory2014; De Smedt et al. Reference De Smedt, Arijs, Segers and Boeraeve2017; Gregory and Lugg Reference Gregory and Lugg2018). The iNaturalist record of this species from Toronto specifies that it was found in association with a purchased tropical plant (phylojenie 2024). Its presence in a greenhouse in Edmonton can probably be attributed to introduction via potted plants or other gardening supplies.
Trachelipus rathkii (Brandt, 1833)
First Canadian record. 1876, Niagara, Ontario (Stuxberg Reference Stuxberg1876).
Canadian distribution. Widespread (Fig. 3), excluding Saskatchewan.
Canadian habitat. In city gardens (Johansen Reference Johansen1929); in greenhouses; on lawns; on golfing greens (Judd Reference Judd1965); associated with buildings (Palmén Reference Palmén1951) and inside buildings (Spencer Reference Spencer1942); in garbage dumps (Palmén Reference Palmén1951; Judd Reference Judd1965; Rafi and Thurston Reference Rafi and Thurston1982); in water reservoirs (Johansen Reference Johansen1929); under boards (Palmén Reference Palmén1951; Bousfield Reference Bousfield1970; Wright Reference Wright1989); under stones (Palmén Reference Palmén1951; Rafi and Thurston Reference Rafi and Thurston1982; Wright Reference Wright1995); under logs (Rafi and Thurston Reference Rafi and Thurston1982; Wright Reference Wright1989, Reference Wright1995; Li Reference Li2022); in leaf litter (Rafi and Thurston Reference Rafi and Thurston1982; Wright Reference Wright1995); in moss and lichens (Wright Reference Wright1995); in fields (Palmén Reference Palmén1951; Judd Reference Judd1965; Bousfield Reference Bousfield1970; Rafi and Thurston Reference Rafi and Thurston1982); in sand (Wallace Reference Wallace1919); on beaches (Bousfield Reference Bousfield1956a); in woods (Wallace Reference Wallace1919; Judd Reference Judd1963, Reference Judd1965; Rafi and Thurston Reference Rafi and Thurston1982; Rapp Reference Rapp1988; Wright Reference Wright1995); in skunk cabbage (Judd Reference Judd1961); on lake shores (Walker Reference Walker1927; Judd Reference Judd1965; Wright Reference Wright1995); on river banks; in marshes (Judd Reference Judd1965); in bogs (Judd Reference Judd1963, Reference Judd1965); in corn, soy, and wheat fields (Belaoussoff et al. Reference Belaoussoff, Guenther, Kevan, Murphy and Swanton1998). Rapp (Reference Rapp1988) noted that this species was present in oak woodlands in southern Ontario but was not found in coniferous woods.
New collections. BRITISH COLUMBIA: Prince George; ALBERTA: Edmonton, Westerose, Pigeon Lake, Calgary, Taber, Lethbridge, Coaldale, intersection of Highways 5 and 6; ONTARIO: Port Hope; NEWFOUNDLAND AND LABRADOR: Pasadena.
New collections – habitat. BRITISH COLUMBIA: under damp rotting logs; ALBERTA: In urban areas under mulch, concrete debris, rocks, pots, logs, bags of soil, and boards sitting on damp grass and soil; in urban yards; under logs in mixed forest trails associated with urban areas; on river banks and on paths next to lake shores; in piles of dirt and sand; under logs on wet leaves; in gravel and mulch next to buildings; by dried pond on prairie; in Salix and Populus groves on prairie; in the vicinity of greenhouses under pots and wooden debris sitting on grass and in leaf litter; in greenhouses under plastic flower pot holders; in a roadside ditch under fibreglass detritus; ONTARIO: under logs and bark by disturbed creekside trail; NEWFOUNDLAND AND LABRADOR: in gravel and sand on river bank.
Origin. Native to Europe, introduced to North America (Van Name Reference Van Name1936; Hatch Reference Hatch1947; Lindroth Reference Lindroth1957; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2000; Schmalfuss Reference Schmalfuss2004). This species and others are thought to have been introduced to Kejimkujik National Park, Nova Scotia, through farming activity (Wright Reference Wright1995).
Note. This species typically exhibits a dark grey or orange – brown pigmentation with dark eyes, but a single depigmented individual with red eyes was present in a population in Edmonton, Alberta, in collections made for the present study (Fig. 4). Albinism in typically pigmented species has been documented in the terrestrial isopod families Agnaridae, Armadillidae, Armadillidiidae, Philosciidae, and Porcellionidae (Johnson Reference Johnson1980; Roder and Linsenmair Reference Roder and Linsenmair1998; Bhella et al. Reference Bhella, Fung, Harrison, Ing, Larsen and Selby2006; Achouri and Charfi-Cheikhrouha Reference Achouri and Charfi-Cheikhrouha2009). There appear to be no published records of albinism in Trachelipodidae.
Trichoniscidae
Androniscus dentiger Verhoeff, 1908
First Canadian record. 1935, London, Ontario (Medcof Reference Medcof1939).
Canadian distribution. EC+EGL (Fig. 3), excluding New Brunswick and Prince Edward Island.
Canadian habitat. In greenhouses (Medcof Reference Medcof1939; Palmén Reference Palmén1951) and basements (Medcof Reference Medcof1939). More recent observations of this species include specimens found outdoors in urban areas (oridgen10 2020; minty-gnat 2024).
Origin. Native to Europe and North Africa, introduced to North America (Lindroth Reference Lindroth1957; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). Medcof (Reference Medcof1939) proposed that this species arrived in North America via transport of greenhouse plants from Europe.
Haplophthalmus danicus Budde-Lund, 1880
First Canadian record. 1927, Humber Valley, Ontario (Walker Reference Walker1928)
Canadian distribution. Widespread (Fig. 3), excluding Alberta, Saskatchewan, Manitoba, and Prince Edward Island
Canadian habitat. In greenhouses (Van Name Reference Van Name1936); associated with urban areas and gardens (Palmén Reference Palmén1951); near watercourses (Walker Reference Walker1928; Palmén Reference Palmén1951); under stones in ravines (Walker Reference Walker1928); in wet leaf litter (Palmén Reference Palmén1951; Judd Reference Judd1965); in rotting wood (Dexter et al. Reference Dexter, Hahnert, Beatty and Downhower1988).
Origin. Native to Europe, introduced to North America (Van Name Reference Van Name1936; Lindroth Reference Lindroth1957; Vandel Reference Vandel1960; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). Lohmander (Reference Lohmander1927) records this species being transported to North America in soil associated with the “roots of asparagus from Germany.”
Haplophthalmus mengii (Zaddach, 1844)
An iNaturalist record from southern British Columbia (Chilliwack River Provincial Park) shows a species of Haplophthalmus that clearly has large tubercles on the third pleon segment (beeworldly 2021). This feature rules out Haplophthalmus danicus, which lacks these tubercles, but would be consistent with Haplophthalmus mengii (Hopkin Reference Hopkin1991). However, H. montivagus Verhoeff, 1941, also has large tubercles and is indistinguishable from H. danicus without close examination of the pereiopods (Hopkin Reference Hopkin1991). Collection of specimens from this site is required to determine which species of Haplophthalmus occurs in this area.
First Canadian record. 2012, Miramichi, New Brunswick (McAlpine and Fairweather Reference McAlpine and Fairweather2016).
Canadian distribution. Recorded only from Miramichi, New Brunswick (Fig. 3) and possibly also from Chilliwack River Provincial Park, British Columbia (see the note, above, for this species).
Canadian habitat. On decayed wood buried in soil (McAlpine and Fairweather Reference McAlpine and Fairweather2016).
Origin. Native to Europe (Vandel Reference Vandel1960; Harding and Sutton Reference Harding and Sutton1985; Schmalfuss Reference Schmalfuss2004), introduced to North America. McAlpine and Fairweather (Reference McAlpine and Fairweather2016) hypothesised that this species was brought to North America in the 1800s in ship ballast.
Hyloniscus riparius (Koch, 1838)
Hyloniscus riparius is superficially similar to Trichoniscus pusillus, but the two species can be distinguished by the number of antennae flagella segments (four or five in T. pusillus, six in H. riparius) and the number of ocelli (three in T. pusillus, one in H. riparius), and H. riparius is bigger and darker (British Myriapod and Isopod Group 2024; Shultz Reference Shultz2018). iNaturalist records of H. riparius include observations from Nova Scotia and New Brunswick (e.g., ipat 2021; john_walsworth 2024); however, none of these observations clearly show the ocelli or antennae. We discarded these and other iNaturalist observations of H. riparius from our dataset when neither of these features was visible.
First Canadian record. 1949, St. John’s, Newfoundland and Labrador (Palmén Reference Palmén1951).
Canadian distribution. EC+EGL (Fig. 3), excluding New Brunswick, Nova Scotia, and Prince Edward Island.
Canadian habitat. On shoreline (Dexter et al. Reference Dexter, Hahnert, Beatty and Downhower1988); on rocky plain (McAlpine and Oldham Reference McAlpine and Oldham2011); in city gardens and in greenhouses (Palmén Reference Palmén1951).
New collections. ONTARIO: Port Hope.
New collections – habitat. ONTARIO: under logs and bark by disturbed creekside trail.
Origin. Native to Europe, introduced to North America (Lindroth Reference Lindroth1957; Vandel Reference Vandel1960; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Trichoniscoides sarsi Patience, 1908
Only two records of this species are reported from Canada. The most recent is Muchmore (Reference Muchmore and Dindal1990), which likely refers to an earlier observation by Palmén (Reference Palmén1951) rather than representing a new collection. No recent records of this species are reported from north of Mexico (Jass and Klausmeier Reference Jass and Klausmeier2001). Whether this species still occurs in Canada is therefore doubtful.
First Canadian record. 1949, St John’s, Newfoundland and Labrador (Palmén Reference Palmén1951).
Canadian distribution. Recorded only from the Avalon Peninsula, Newfoundland (Fig. 3).
Canadian habitat. Under rocks associated with urban areas (Palmén Reference Palmén1951; Muchmore Reference Muchmore and Dindal1990).
Origin. Native to Europe, introduced to North America (Lindroth Reference Lindroth1957; Vandel Reference Vandel1960; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Trichoniscus provisorius Racovitza, 1908
Only Palmén (Reference Palmén1951) mentions this species occurring in Canada. The lack of records may be due in part to confusion around the taxonomic status of “T. provisorius” (see next paragraph). Recent collections from Newfoundland (2011–2023) that included Trichoniscus species were examined by H.G.S. The three largest samples (each from a single site) contained 255, 10, and 9 individuals, respectively. All individuals were female, which strongly suggests T. pusillus rather than T. provisorius (see next paragraph). The apparent absence of T. provisorius from Canada in recent years is therefore not entirely attributable to the species being overlooked.
In the original description of the species, Racovitza (Reference Racovitza1908) noted that T. provisorius might be identical to T. pusillus Brandt, 1833, but he was unable to determine if this was the case due to the vague original description of T. pusillus. Populations of T. pusillus are parthenogenetic and produce males rarely, whereas T. provisorius populations contain roughly equal numbers of males and females (Frankel et al. Reference Frankel, Sutton and Fussey1981). Palmén (Reference Palmén1951) noted similarity between males of the two species but was unsure whether T. provisorius should be considered a subspecies of T. pusillus. Upon discovering that T. pusillus was triploid and T. provisorius was diploid, Legrand et al. (Reference Legrand, Strouhal and Vandel1950) proposed T. provisorius was a subspecies of T. pusillus. However, some preferred to use the term “form” rather than “subspecies” (Frankel et al. Reference Frankel, Sutton and Fussey1981; Harding and Sutton Reference Harding and Sutton1985), and others continued to use the name “T. provisorius” without listing it as a form or subspecies (Leistikow and Wägele Reference Leistikow and Wägele1999; Mattern Reference Mattern, Sfenthourakis, Araujo, Hornung, Schmalfuss, Taiti and Szlávecz2003). In a global list of sowbug species, Schmalfuss (Reference Schmalfuss2004) elevated T. pusillus provisorius and three other subspecies of T. pusillus to the species level. The basis for this taxonomic change is not clear but may have been influenced by the discovery of high genetic divergences between subspecies of T. pusillus (Cobolli Sbordoni et al. Reference Cobolli Sbordoni, Ketmaier, de Matthaeis and Taiti1997). Regardless, the change appears to have been accepted, as “T. provisorius” is now typically used instead of “T. pusillus provisorius” (Vilisics et al. Reference Vilisics, Sólymos, Nagy, Farkas, Kemencei and Hornung2011; De Smedt et al. Reference De Smedt, Arijs, Boeraeve and Proesmans2016; Vittori and Štrus Reference Vittori and Štrus2023). For the purposes of the present checklist, we are considering T. provisorius and T. pusillus to be separate species.
First Canadian record. 1949, St. John’s, Newfoundland and Labrador (Palmén Reference Palmén1951).
Canadian distribution. Recorded only from the Avalon Peninsula, Newfoundland (Fig. 3).
Canadian habitat. Associated with urban areas, including on wet soil, in leaf litter, and by streams (Palmén Reference Palmén1951).
Origin. Native to Europe, parts of the Middle East and Northern Africa, and introduced to North America (Lindroth Reference Lindroth1957; Vandel Reference Vandel1960; Harding and Sutton Reference Harding and Sutton1985; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004).
Trichoniscus pusillus Brandt, 1833
Species in the genus Trichoniscus can be distinguished by male pleopod morphology. It was initially impossible to tell if North American populations of Trichoniscus were identical to European T. pusillus because no male specimens had been found, and so the name Trichoniscus demivirgo Blake, 1931 was used for North American specimens (Lohmander Reference Lohmander1927; Hatch Reference Hatch1947; Palmén Reference Palmén1951). Later, Palmén (Reference Palmén1951) found male Trichoniscus in Newfoundland. Upon morphological comparison with male T. pusillus from Europe, he concluded that the two were identical and that T. demivirgo was likely a synonym of T. pusillus. Because comparison to European Trichoniscus has taken place only for Newfoundland specimens, it is possible that “T. demivirgo” from other parts of North America may differ from T. pusillus. However, for the purposes of the present checklist, all records of T. demivirgo from Canada are considered T. pusillus rather than T. demivirgo.
iNaturalist observations include many records of a Trichoniscus species that is likely to be T. pusillus, given the rarity of T. provisorius in Canada (see note for T. provisorius , above); however, confirmation of the identity is not possible without examination of the sex ratios of large sampling sizes or examination of male pleopods (see note for T. provisorius , above). iNaturalist observations of Trichonisus pusillus have therefore been excluded from our dataset.
First Canadian record. 1876, Niagara, Ontario (Stuxberg Reference Stuxberg1876).
Canadian distribution. Widespread (Fig. 3), excluding Alberta, Saskatchewan, Manitoba, and Prince Edward Island.
Canadian habitat. In greenhouses (Van Name Reference Van Name1936; Judd Reference Judd1965; Rafi and Thurston Reference Rafi and Thurston1982) and outside greenhouses (Hatch Reference Hatch1947); in gardens; associated with buildings and railway stations; in graveyards (Palmén Reference Palmén1951); in garbage dumps; on golfing greens (Judd Reference Judd1965); under rocks (Walker Reference Walker1927; Palmén Reference Palmén1951; Bousfield Reference Bousfield1956b); among leaves (Walker Reference Walker1927; Palmén Reference Palmén1951; Rafi and Thurston Reference Rafi and Thurston1982; Wright Reference Wright1995); in moss (Palmén Reference Palmén1951); under logs (Walker Reference Walker1927; Palmén Reference Palmén1951); associated with streams (Walker Reference Walker1927; Palmén Reference Palmén1951; Judd Reference Judd1965), springs (Palmén Reference Palmén1951), creeks (Walker Reference Walker1927; Judd Reference Judd1965), ponds, rivers (Palmén Reference Palmén1951; Judd Reference Judd1965), and lakes (Walker Reference Walker1927; Judd Reference Judd1965; Wright Reference Wright1995); in woods (Walker Reference Walker1927; Palmén Reference Palmén1951; Judd Reference Judd1965; Rafi and Thurston Reference Rafi and Thurston1982; Wright Reference Wright1995); under boards (Bousfield Reference Bousfield1956b); in marshes; on wet soil; in wet fields (Palmén Reference Palmén1951; Judd Reference Judd1965); on shorelines (Palmén Reference Palmén1951; Bousfield Reference Bousfield1956b).
New collections. ONTARIO: Port Hope; NEWFOUNDLAND AND LABRADOR: J.T. Cheeseman Provincial Park, Lockston Path Provincial Park, Butter Pot Provincial Park, La Manche Provincial Park.
New collections – habitat. ONTARIO: under logs and bark by disturbed creekside trail; NEWFOUNDLAND AND LABRADOR: in mixed and conifer forests.
Origin. Native to Europe, introduced to North America (Lindroth Reference Lindroth1957; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). Lohmander (Reference Lohmander1927) mentions this species being transported to North America in a plant shipment.
Trichoniscus pygmaeus Sars, 1898
First Canadian record. 1949, St. John’s, Newfoundland and Labrador (Palmén Reference Palmén1951).
Canadian distribution. Widespread (Fig. 3), but recorded only from Greater Vancouver and Vancouver Island, British Columbia; southern Ontario; and Newfoundland. There are no recent records of this species from Newfoundland.
Canadian habitat. Associated with urban areas, including in gardens, in wet soil, and under boards (Palmén Reference Palmén1951).
Origin. Native to Europe, introduced to North America (Van Name Reference Van Name1936; Lindroth Reference Lindroth1957; Vandel Reference Vandel1960; Harding and Sutton Reference Harding and Sutton1985; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). Lohmander (Reference Lohmander1927) mentions transport of this species to North America among “lily bulbs” from Scotland.
Other records of interest
The following records lack strong support for the existence of a stable population in Canada or are not identified to species level.
Alloniscidae
Alloniscus mirabilis (Stuxberg, 1875)
This species is native to North America and is found along the southern west coast (Schultz Reference Schultz1984a; Leistikow and Wägele Reference Leistikow and Wägele1999; Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). One GBIF.org record lists this species from Vancouver Island, British Columbia (GBIF.org 1992c), but no other records of this species currently exist north of California. The GBIF.org record may represent Alloniscus perconvexus, which is found in British Columbia (see “ Alloniscus perconvexus” section, above).
Armadillidae
Armadillo sp
A single GBIF.org record lists an “Armadillo” species recorded from British Columbia (GBIF.org [date unknown]a). No specific locality is listed. All true Armadillo are found in tropical regions (Schmalfuss Reference Schmalfuss2004). One “Armadillo” species, Armadillo affinis (Dana, 1854), has been recorded from California, but it does not actually belong to the genus Armadillo, and the status of the name is unclear (Schmalfuss Reference Schmalfuss2004). The GBIF.org record may be an error: the species is identified as “Oniscidea” in the original source for this record, and the genus listed in GBIF.org is noted as being “inferred.” Possibly, the transcriber intended to record this record as Armadillidium rather than Armadillo, as two Armadillidium species occur in British Columbia (see “ Armadillidium vulgare ” and “ Armadillidium nasatum ” sections, above).
Armadillidium maculatum (Risso, 1816)
A video from Edmonton, Alberta, submitted via the community science initiative for the present study depicts a dark-coloured sowbug with a convex body shape, truncated uropods, a smooth dorsal surface, and a thin bright white stripe running along the dorsal posterior edges of the pereon and pleon segments. This appearance is consistent with Armadillidium maculatum (Vandel, 1962), which is a popular pet species (Jungle Jewel Exotics 2023). According to the participant who submitted the video, the sowbug was discovered next to a building under a rock, and only a single individual was found. Armadillidium maculatum is found along the Mediterranean coast of France and Italy (Vandel Reference Vandel1962; Schmalfuss Reference Schmalfuss2004), and the only evidence of a wild population of this species in North America is an iNaturalist observation from Texas, United States of America, that might depict an individual in an outdoor environment (jonathanash37 2023). The record from Edmonton likely represents a captive individual that had escaped or was released. There is currently no evidence to suggest that an established wild population of A. maculatum is present in Edmonton.
Platyarthridae
Trichorhina sp
There are two GBIF.org records of this genus from 1992 in Montréal, Quebec (GBIF.org 1992a, 1992b). Most Trichorhina species are found close to the equator (Schmalfuss Reference Schmalfuss2004), but four are found in North America: T. donaldsoni Schultz, 1963, T. heterophthalma Lemos de Castro, 1964, T. triocis Mulaik and Mulaik, 1943, and T. tomentosa (Budde-Lund, 1893) (Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). Of these four, all can be found in the southern United States of America, and all except T. tomentosa are thought to be native to North America (Jass and Klausmeier Reference Jass and Klausmeier2001; Schmalfuss Reference Schmalfuss2004). The species represented by the GBIF.org record would most likely be T. tomentosa because this species has been widely spread and can be found in greenhouses in many parts of the world (Schmalfuss Reference Schmalfuss2004; Gregory Reference Gregory2014). Because no images are associated with the GBIF.org record, this identification cannot be confirmed.
Genus-level records of interest
The following two genus-level records are included here because they represent unusual collection localities or include other information of interest. Both genera contain multiple species that have been recorded from Canada.
Porcellionidae
Porcellio sp
One unpublished GBIF.org record lists three individuals collected from the “Beaufort Sea,” Northwest Territories (GBIF.org [date unknown]b). If the locality is correct, this would be the northernmost sowbug record in North America. Whether this record represents an established population or a temporary introduction (e.g., a few individuals introduced in the summer and discovered the same season) is unclear.
Trichoniscidae
Trichoniscus sp
One unpublished GBIF.org record lists a collection of this genus from 1926 in what is now Nunavut, on the border with Manitoba (GBIF.org 1926a). The record refers to a single specimen found “in soil used for Dahlia (Asteracecae) tubes packing,” as a transfer associated with the “Fed. Hort. Bd” (presumably referring to the Federal Horticulture Board). The card also includes the note “Chicago #371.” Possibly, this refers to specimens recovered from a shipment of plants between Chicago and Nunavut, but which location represents the destination is not specified. If the locality is correct, this record would represent the only sowbug recorded from Nunavut.
Discussion
Based on new collections in the present study, postings on BugGuide and iNaturalist, GBIF.org records, reports from the community science initiative for the present study, and our literature survey, at least 32 species and 12 families of sowbugs now occur in Canada. Some are limited to the west coast (Fig. 3): these include Alloniscus perconvexus, all Detonidae, Littorophiloscia richardsonae, and all Ligiidae except Ligia oceanica and Ligidium elrodii. All species sharing this distribution pattern are native to North America. Other species are found only along the east coast and the St. Lawrence River to Lake Erie and Lake Ontario (EC+EGL, Fig. 3): these include Androniscus dentiger, Hyloniscus riparius, Ligia oceanica, Ligidium elrodii, Littorophiloscia vittata, Porcellio laevis, Trichoniscus provisorius, Trichoniscoides sarsi, and possibly Haplophthalmus mengii. Of this group, only Ligidium elrodii and Littorophiloscia vittata are native to North America. The remaining species, all of which are introduced, fall into the “widespread” distribution pattern (i.e., they can be found in both eastern and western Canada; Fig. 3). Of these species, only Porcellio spinicornis appears to be common in Alberta, Saskatchewan, and Manitoba (Supplementary material, Fig. S1, Porcellio spinicornis).
Although the apparent difference in species composition in our own collections between sowbugs on the coasts versus in the Prairie Provinces is likely due in part to the relative lack of sampling in urban areas in the island of Newfoundland, a similar pattern is also present in the iNaturalist records, where sowbug species that are abundant throughout the rest of Canada are seemingly absent or uncommon in the Prairie Provinces (e.g., Oniscus asellus, Porcellio scaber, Armadillidium vulgare, and Cylisticus convexus). This pattern has also been noted for nonnative Staphylinidae in Canada, where species diversity is lowest in the Prairie Provinces and highest in British Columbia and eastern Canada (Klimaszewski and Brunke Reference Klimaszewski, Brunke, Betz, Irmler and Klimaszewski2018). However, it is difficult to draw conclusions about the apparently limited distribution of sowbugs in the Prairie Provinces for the following reasons: (1) many records in this area are from community science observations (e.g., iNaturalist and BugGuide), which may favour large, conspicuous species (Caley et al. Reference Caley, Welvaert and Barry2020); (2) most Canadian sowbug distribution data is based only on presence (i.e., researchers rarely report searching for a particular species and not finding it, and many records originate from presence-only data generated by community scientists); (3) focused investigations of sowbugs in this part of Canada are sparse – aside from the present study, the Prairie Provinces have never been surveyed for sowbugs. Additional surveys of this area, particularly targeting Saskatchewan and Manitoba, would help confirm which species are truly absent.
Of the 32 sowbug species in Canada, 10 are native to North America. The Canadian distribution of these 10 species is limited to either the eastern side of Canada (Littorophiloscia vittata and Ligidium elrodii) or the west coast. Of the 22 nonnative species, at least 14 are found on both coasts. This is consistent with Jass and Klausmeier’s (Reference Jass and Klausmeier2000) observation that native North American sowbugs tend to occur on a single coast and nonnative species are found on both coasts. The other eight nonnative species are found only on the eastern side of Canada, apart from Nagurus cristatus, which has been reported only from Alberta and Ontario (present study). The greater diversity of introduced sowbugs in eastern Canada may reflect the longer history of European settlement and trade in that region. All but one of the 22 nonnative sowbugs that occur in Canada have a native range that includes Europe. Lindroth (Reference Lindroth1957) hypothesised that the thousands of tonnes of soil ballast brought from Europe to North America in the 1600s–1800s played a major role in introducing new soil-associated species, and he noted that ballast consisted not only of soil but also of stones and bricks from demolitions in urban areas and of gravel and sand from beaches. Ballast sources therefore included a wide range of sowbug habitats and could have been the source of introduction for a variety of European species. The exception to the European origin of nonnative Canadian sowbugs is Nagurus cristatus, which is native to Southeast Asia (Leistikow and Wägele Reference Leistikow and Wägele1999). It is also found in many places worldwide, including the southern United States of America (Johnson Reference Johnson1986; Jass and Klausmeier Reference Jass and Klausmeier2000; iNaturalist community 2024dg). Whether introduction of this species to Canada occurred secondarily from another introduced population or directly from Southeastern Asia is unknown.
Direct introduction of sowbugs via ship ballast would not have been possible in the landlocked provinces of Alberta and Saskatchewan. Because sowbugs are generally assumed to be poor dispersers (Hornung Reference Hornung2011; Szlavecz et al. Reference Szlavecz, Vilisics, Tóth and Hornung2018; Dimitriou et al. Reference Dimitriou, Antoniou, Alexiou, Poulakakis, Parmakelis and Sfenthourakis2022), it is unlikely that sowbugs in this area arrived via active dispersal from coastal populations into Canada’s interior. Instead, these populations may be the result of human-mediated introduction from the eastern, western, or both coasts, due to primary introductions directly from Europe, or both scenarios. Sowbug transport could have been accidental – for example, via track ballast used for railway lines, in shipments of landscaping and gardening materials, as stowaways in household belongings – or deliberate, if they were imported as pets or as live pet food. Canadian Food Inspection Agency import records from 2015 to 2022 list only a single sowbug shipment consisting of 60 Armadillidium sp. sent from the United States of America to British Columbia in 2022 (Canadian Food Inspection Agency 2022). The actual frequency of imports is likely higher because these records list only shipments that had Canadian Food Inspection Agency import authorisation. Evidence of nonlisted imports includes the fact that Alberta residents have kept “fancy” sowbugs as pets prior to 2022, including selectively bred forms and species not yet found as free-living populations in Canada (H.C.P. and H.G.S., unpublished data). Molecular comparisons of European, coastal Canadian, and Prairie Province populations could resolve questions regarding the origins of sowbugs currently found in the Prairie Provinces.
The presence of sowbugs of European origin at high latitudes in the Prairie Provinces (Supplementary material, Fig. S1, Porcellio spinicornis) raises the question of how these species can survive winter. According to Wright (Reference Wright1997), sowbugs can live in regions with winter temperatures up to 40 °C colder than their lethal limit by burrowing deeply into substrate or remaining next to heated buildings. If sowbugs in Canada survive winters by overwintering next to heated buildings or even inside buildings, their distribution should be limited to urban areas. A sowbug survey with more extensive coverage of natural areas would determine whether this is the case. It is also possible that nonnative sowbugs in Canada have undergone climatic adaptation. Further research investigating the current cold tolerance of sowbugs in Canada and a comparison of this data to similar studies on European populations of the same species may provide evidence of climate-associated selection and adaptation.
There appears to be a correlation between the extent of the Canadian distribution of a sowbug species and the date it was first recorded in Canada (Fig. 5). Of the 11 introduced species with the widest Canadian distribution, 10 have Canadian records dating from before 1930: Haplophthalmus danicus, Oniscus asellus, Cylisticus convexus, Porcellio spinicornis, Porcellio scaber, Porcellionides pruinosus, Trachelipus rathkii, Trichoniscus pusillus, Armadillidium nasatum, and Armadillidium vulgare. The exception is Philoscia muscorum, for which the first Canadian record is 1958. All but T. pusillus and H. danicus are also among the 10 most common species in North America (Jass and Klausmeier Reference Jass and Klausmeier2000). This correlation may be the result of species with the earliest introductions having had more time to disperse passively, actively, or both, which may have made them more common and more likely to be noticed and recorded. If this is the case, we would expect to see similar range expansion in the future of the more recently introduced species. Alternatively, this pattern may simply be due to species that are larger-bodied or common (due to having experienced rapid range expansion or population growth, or both) being noticed sooner regardless of date of introduction. Most trichoniscids, which have small bodies, were not reported until after 1925 (Fig. 5). Finally, the pattern may result from the fact that species better able to disperse (passively, actively, or both) may have been more likely to be introduced frequently and earlier and may also have spread to more provinces once introduced. More recently introduced species may therefore represent those with limited dispersal ability and so may not be expected to experience significant future range expansion in Canada.
Simple linear regression between the number of provinces occupied and date of first record for all species of introduced sowbugs in Canada. “Provinces occupied” indicates the number of provinces with at least one record of a species regardless of the recency of the record. There is a significant negative correlation between the two variables (species with more recent dates of first record tend to occupy fewer provinces). Species abbreviations are as follows: Ar na, Armadillidium nasatum; Ar vu, Armadillidium vulgare; Cy co, Cylisticus convexus; Li oc, Ligia oceanica; On as, Oniscus asellus; Ph mu. Philoscia muscorum; Pl ho, Platyarthrus hoffmannseggii; Po di, Porcellio dilatatus; Po la, Porcellio laevis; Po sc, Porcellio scaber; Po sp, Porcellio spinicornis; Po pr, Porcellionides pruinosus; Na cr, Nagurus cristatus; Tr ra, Trachelipus rathkii; An de, Androniscus dentiger; Ha da, Haplophthalmus danicus; Ha me, Haplophthalmus mengii; Hy ri, Hyloniscus riparius; Tr sa, Trichoniscoides sarsi; Tr pr, Trichoniscus provisorius; Tr pu, Trichoniscus pusillus; Tr py, Trichoniscus pygmaeus.

Although only a single species of sowbug from one site in the Prairie Provinces was reported in a published, peer-reviewed venue (Li Reference Li2022), we found that records from community scientists provided a more comprehensive picture, highlighting the value of community science initiatives in documenting and monitoring sowbug diversity. As of 20 October 2024, seven of the nine species recorded from Alberta in the present study had also been documented from the province at least once on iNaturalist (iNaturalist community 2024a), BugGuide (2024a), or both, with sowbug records originating from many different parts of Alberta. Because many sowbugs are identifiable to species based on photos alone (Gregory Reference Gregory2022; H.G.S., unpublished data), using community science to monitor introduced populations of sowbugs would be an excellent way to track range expansion and to document the arrival of additional species. For many smaller species (e.g., Trichoniscidae), identification to particular species may not be possible with only photos, but observations of this group would still be useful in detecting morphotypes that do not match any known species in an area and in identifying sowbug populations that could be sampled in the future for identification purposes or for research. However, whereas most of the sowbug species identifications for observations on iNaturalist and BugGuide were a good match for the associated image(s), some identifications are doubtful or clearly inaccurate. Critical evaluation of records obtained from community science initiatives is important to ensure identifications are reasonable, particularly for observations that record species in unusual localities. Species identifications should also be assessed by detailed morphological examination and molecular data from collected specimens whenever possible, both to confirm the identification (particularly for newly documented species) and to provide material for future studies.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.4039/tce.2025.10037.
Acknowledgements
The authors thank the following: D. Langor, L. Lafosse, R. Palmer, D. Harrison, B. Tonn, C. Paszkowski, H. Carcamo, B. Grappone, A. Pretty, H. Beck, C. Goater, K. Floate, L. Pollett, P. Perry, B. Wingert, K. Kent, C. Crooks, G. Pohl, C. Davis, D. Edwards, K. Hudson, H. Kastelic, G. and L. Jackson, C. Kolt, J. Sperling, E. Viluand, K. Van Slyke, L. Lumley, and the participants in the “Sowbugs of Alberta” community science project for collecting sowbugs for the present study; the members of the American Isopod and Myriapod Group (AIMG) and British Myriapod and Isopod Group (BMIG) for adding identifications to Canadian sowbug observations on iNaturalist; the many iNaturalist and BugGuide users who posted sowbug observations from Canada; Ariana Lane for photos of GBIF.org 2018a (Armadilloniscus sp.); Shawn Brescia for input on the identification of this specimen; the Newfoundland and Labrador Department of Tourism, Culture, Arts and Recreation for providing permits for the collection of invertebrates from provincial parks; Marlee Kohlman for assisting with fieldwork; Shawn Abraham, Leah Jackson, Grace Ren, and other members of the Sperling Lab (University of Alberta) for providing molecular methods training and advice; John Acorn for sharing his observations and UV flashlight; and greenhouse owners for allowing collection of sowbugs from their facilities. Data analysis was enabled in part by resources provided by the Digital Research Alliance of Canada (alliancecan.ca). This project was supported by a Natural Sciences and Engineering Research Council (NSERC) Discovery Grant to H.C.P. (RGPIN-2019-04427), an Alberta Conservation Association (ACA) Grant in Biodiversity to H.G.S., and an NSERC Canada Graduate Scholarship – Master’s (CGS – M) to H.G.S.
Competing interests
The authors declare that they have no competing interests.