Introduction
Through their activity, animals modify their habitat and affect the availability of resources for other organisms. These modifications can create habitat heterogeneity, which plays a crucial role in shaping ecological processes and patterns (Mittelbach and McGill Reference Mittelbach and McGill2019). While clear examples of habitat modification are provided by ecosystem engineers (Briones Reference Briones2024), all species change their environment through their daily activities, though often in more subtle ways. While less dramatic, and consequently less studied, these effects are widespread and deserve more attention. Such is the case of the effects that animals can cause through their defecation (e.g., Ferraro and Lienau Reference Ferraro and Lienau2025).
The defecation behaviour of certain mammals, such as rhinoceroses, otters, and tapirs, is known to create accumulation of faeces due to the repeated use of specific sites for defecation, called latrines (e.g., Awasthi et al. Reference Awasthi, McConkey, Aluthwattha, Chen and Chen2024; Lautenschlager et al. Reference Lautenschlager, Souza, Villar, Galetti and Feeley2024). Some arboreal primates, such as howler monkeys (Alouatta Lacépède, 1799), use certain sleeping trees repeatedly over time (Bravo Reference Bravo2009; Julliot Reference Julliot1996). Because these primates tend to defecate upon waking up, their faeces may also be deposited repeatedly under sleeping trees (Arroyo-Rodríguez et al. Reference Arroyo-Rodríguez, Andresen, Bravo, Stevenson, Kowalewski, Garber, Cortes-Ortiz, Urbani and Youlatos2015). These sites are sometimes also referred to as latrines (Bravo and Cueto Reference Bravo and Cueto2020; Fuzessy et al. Reference Fuzessy, Sobral and Culot2022; Pouvelle et al. Reference Pouvelle, Jouard, Feer, Tully and Ponge2009). Studies have found that defecation sites under frequently used sleeping trees of howler monkeys can differ from control sites in several aspects, such as soil characteristics (Bravo and Cueto Reference Bravo and Cueto2020; dos Santos Neves et al. Reference dos Santos Neves, Feer, Salmon, Chateil and Ponge2010; Feeley Reference Feeley2005; Pouvelle et al. Reference Pouvelle, Feer and Ponge2008), seed and seedling assemblages (Anzures-Dadda et al. Reference Anzures-Dadda, Andresen, Martínez-Velázquez and Manson2011; Bravo Reference Bravo2012; Bravo and Cueto Reference Bravo and Cueto2020; Feer et al. Reference Feer, Ponge, Jouard and Gomez2013; Julliot Reference Julliot1997; Pouvelle et al. Reference Pouvelle, Jouard, Feer, Tully and Ponge2009), and seedling/sapling recruitment, survival, and growth (Bravo and Cueto Reference Bravo and Cueto2020; Fuzessy et al. Reference Fuzessy, Sobral and Culot2022; Zárate et al. Reference Zárate, Andresen and Santos-Heredia2019).
Two animal groups that are attracted to vertebrate faeces and that have been hypothesized to shape some of the characteristics observed in monkey defecation sites are dung beetles and rodents (Feer et al. Reference Feer, Ponge, Jouard and Gomez2013, Pouvelle et al. Reference Pouvelle, Jouard, Feer, Tully and Ponge2009, Russo Reference Russo2005). Dung beetles, through their dung-burying behaviour, perform many ecological functions (Nichols et al. Reference Nichols, Spector, Louzada, Larsen, Amezquita and Favila2008), some of which can affect soil and plants (e.g., soil bioturbation and secondary seed dispersal; Andresen and Urrea-Galeano Reference Andresen and Urrea-Galeano2022; deCastro-Arrazola et al. Reference deCastro-Arrazola, Hortal, Noriega and Sánchez-Piñero2020). On the other hand, rodents are seed predators, and they are believed to be responsible for the higher predation rates observed for primate-defecated seeds (e.g., Andresen Reference Andresen1999; Chapman Reference Chapman1989; McConkey Reference McConkey2005). However, to our knowledge, only three studies have directly quantified the activity of dung beetles attracted to monkey defecation sites under sleeping trees: two with howler monkeys (Feer et al. Reference Feer, Ponge, Jouard and Gomez2013; Fuzessy et al. Reference Fuzessy, Sobral and Culot2022) and one with spider monkeys (Whitworth et al. Reference Whitworth, Whittaker, Huarcaya, Flatt, Morales, Connor, Garrido Priego, Forsyth and Beirne2019). In the case of rodents, while several studies have assessed seed predation in defecation sites under monkey sleeping trees (e.g., Feer and Forget Reference Feer and Forget2002; Russo Reference Russo2005), only the above-mentioned study with spider monkeys has compared rodent visitation in defecation versus control sites (Whitworth et al. Reference Whitworth, Whittaker, Huarcaya, Flatt, Morales, Connor, Garrido Priego, Forsyth and Beirne2019). These three studies found that monkey defecation sites under frequently used sleeping trees created microhabitat heterogeneity.
However, monkeys do not deposit all their defecations under frequently used sleeping trees. For howler monkeys, although little quantitative information exists, it appears that most sleeping trees are only used occasionally, at least in some study sites (Brividoro et al. Reference Brividoro, Kowalewski, Scarry and Oklander2019; Julliot Reference Julliot1996). Thus, studies that focus on defecation sites frequently used by monkeys only give us a partial view of the microhabitat heterogeneity created by howler monkey defecation. Furthermore, howler monkey species can differ in their group sizes and behaviours, and consequently in their use of space and defecation patterns (Andresen Reference Andresen1999; Arroyo-Rodríguez et al. Reference Arroyo-Rodríguez, Andresen, Bravo, Stevenson, Kowalewski, Garber, Cortes-Ortiz, Urbani and Youlatos2015). Most studies on the effects of howler monkey defecation sites have focused on species of the A. seniculus Group (including A. macconnelli Elliot 1910 and subspecies of A. seniculus (Linnaeus, 1766)), A. caraya (Humboldt, 1812), and A. guariba (Humboldt, 1812), while little information is available for other howler species. Additional research is thus needed to increase our knowledge about how different howler species may vary in the patterns of microhabitat heterogeneity they create through their defecations under sleeping trees in general, not only under frequently used sleeping trees.
Our goal was to determine if defecation sites under sleeping trees of the Mexican mantled howler monkey (A. palliata mexicana Merriam, 1902) can create microhabitat heterogeneity through differential dung beetle and rodent activity. Since monkey faeces attract beetles and rodents, we hypothesized that both groups of animals would have higher activity in defecation sites. Using a combination of field sampling and experiments, we tested the predictions that, compared to control sites, defecation sites would (1) attract more dung beetles and rodents, (2) have higher dung removal by beetles and duration of rodent visitation, and (3) have higher secondary seed dispersal by beetles and seed predation/removal by rodents. We also wanted to determine how long any observed difference might last after the defecation occurs. While most vertebrate dung in tropical forests is generally removed quickly from the soil surface, removal rates can vary greatly (Hanski and Cambefort Reference Hanski and Cambefort1991). Also, unless washed by rain, traces of dung and urine may remain, such that the odour of a howler monkey group defecation may remain perceivable for several days (EA, pers. obs.), attracting more dung beetles and rodents.
Materials and methods
Study area
The study was carried out at the Los Tuxtlas Biological Station (18°35′5″ N, 95°4′34″ W), in the Mexican state of Veracruz. This site encompasses 640 ha of conserved tropical rain forest connected to larger forest areas within the Los Tuxtlas Biosphere Reserve, which has a total area of ∼155,000 ha. Mean annual temperature is 24.1°C, and rainfall is 4,201 mm (Gutiérrez-García and Ricker Reference Gutiérrez-García and Ricker2011). Despite high levels of forest loss and fragmentation, the region still harbours much biodiversity, including approximately 50 species of Scarabaeinae dung beetles (Favila Reference Favila, Reynoso, Coates and Vázquez Cruz2017) and 18 species of rodents (González-Christen and Coates Reference González-Christen and Coates2019). Detailed information about the study area can be found elsewhere (Coates Reference Coates, Reynoso, Coates and Vázquez Cruz2017).
Defecation and control sites
At dawn, when monkeys vocalized, one of four groups whose home ranges were nearest to the Los Tuxtlas Biological Station’s headquarters was localized. Group sizes varied between four and ten individuals. Howler monkeys tend to have group defecations (i.e., most of the individuals of a group defecate within a short period) after prolonged resting periods (Arroyo-Rodríguez et al. Reference Arroyo-Rodríguez, Andresen, Bravo, Stevenson, Kowalewski, Garber, Cortes-Ortiz, Urbani and Youlatos2015). Defecation sites were areas of the forest floor, under sleeping trees, where we found fresh faeces after a group defecation. The location of each defecation site was based on a single observation, i.e., we did not assess how frequently monkeys used a site. Control sites were understory areas that had similar vegetation structure, but where monkeys were not observed defecating during the study period. For dung beetles, the distance between control and defecation sites was 100 m to avoid interference (da Silva and Hernández Reference da Silva and Hernández2015) between the dung we used in traps and experimental stations in both sites. For rodents, the distance between control and defecation sites was 20 m because we did not place any dung in control sites. Sites used for dung beetles were not the same as those used for rodents, as both parts of the study were conducted in different years (see below).
Dung beetles, dung removal, and secondary seed dispersal
Between April and May 2019, we established (i) 14 defecation sites where pitfall traps and experimental stations were deployed immediately after monkeys defecated (1d-defecation site), (ii) 14 sites where monkeys had defecated approximately one week before traps and stations were deployed (1wk-defecation site), and (iii) 14 control sites. One trap and one station were deployed in one site of each type on the same day. Since we used small dung piles in all three site types (to bait traps and to measure dung removal and seed dispersal; see below), we did not compare sites with dung versus sites without any dung (as in the case of rodents). Rather, we compared how the abundance and activity of dung beetles attracted to the small amounts of experimental faeces we placed differed when these faeces were deposited in sites where a group of monkeys had defecated (either the same day or a week before), and those where monkeys had not defecated.
To sample dung beetles, we deployed one pitfall trap per site. Traps consisted of 1 l plastic containers (opening of 15 cm), buried level with the soil and filled with soapy water up to two-thirds of their capacity. A plastic plate was placed above the trap to protect it from rain. The bait consisted of 15 g of fresh howler monkey dung placed in a tulle bag that was suspended from the plate. Traps were active for 24 h; all beetles were identified to the species level by one of us (AD-R) and counted.
To assess dung removal and secondary seed dispersal, we established one experimental station at each site, 5 m away from the pitfall trap. Stations consisted of small areas on the forest floor (∼10 cm diam), where we placed five piles of fresh howler monkey dung, each weighing ∼5 g (total of ∼25 g of dung per station). Inside each dung pile, we placed one artificial seed: a 4 mm spherical plastic bead with a 30 cm nylon thread attached to it. Artificial beads are commonly used to measure secondary seed dispersal by dung beetles (Andresen and Urrea-Galeano Reference Andresen and Urrea-Galeano2022). We chose a 4 mm bead because this is a size that is moved by dung beetles in the study area (Urrea-Galeano et al. Reference Urrea-Galeano, Andresen, Coates, Mora Ardila and Ibarra-Manríquez2019a), and a 30 cm thread is effective for finding seeds moved by dung beetles (Urrea-Galeano et al. Reference Urrea-Galeano, Andresen, Coates, Mora Ardila, Díaz Rojas and Ramos-Fernández2019b). After 24 h, we measured dung removal by counting the number of dung piles in a station that had been completely removed, and seed dispersal by recording whether beads had been dispersed horizontally and/or vertically. For dispersed beads, we measured dispersal distances to the nearest 1 cm with a ruler or a measuring tape.
Rodents, duration of rodent visitation, and seed predation/removal
We established 18 paired sample areas in July–August 2022. Each paired area consisted of a defecation site and a control site. Immediately after monkeys defecated, we deployed camera traps and seed stations. Both sites of a paired sample area were established on the same day, within an hour.
At each site, one camera trap (BROWNING BTC-5HD-850 or STEALTH CAM STC-G42NG) was fastened to a tree trunk at a height of 30–50 cm, pointing to at least one seed station (see below). Cameras were programmed to take three photographs every ten seconds; we chose this setting to obtain more information about animal activity while optimizing memory card storage capacity. In 14 of the 18 sampling areas, cameras were active continuously for 7 days. In the other 4 sampling areas, cameras were active only for 1 day.
We grouped three rodent species (paca, Cuniculus paca; agouti, Dasyprocta mexicana; squirrel, Sciurus aureogaster; see Table S2 for full scientific nomenclature) and two morphospecies (‘rodentia’ and ‘small mammal’) into the Rodent category. For each site and sampling day, we obtained the total number of independent rodent records. Contiguous photographs of the same species/morphospecies were considered independent when ≥40 minutes elapsed between them (a threshold of 30 min has been used in similar studies – e.g., Whitworth et al. Reference Whitworth, Whittaker, Huarcaya, Flatt, Morales, Connor, Garrido Priego, Forsyth and Beirne2019 – and we increased it by 10 min to be more conservative). To obtain the duration of rodent visitation, we used the number of minutes elapsed between the first and the last photo in the set of photos that constituted one independent record.
To assess seed predation/removal, we established seed stations with seeds of two tree species: Brosimum alicastrum Sw. and Poulsenia armata (Miq.) Standl. (hereafter referred to by their genus name), both in the Moraceae family. Both species are common in the Los Tuxtlas Biological Station; Brosimum seeds are large (9–13 × 16–20 mm, 0.91 g fresh weight) and those of Poulsenia are medium-sized (6–9 × 4–6 mm, 0.085 g; Ibarra-Manríquez et al. Reference Ibarra-Manríquez, Martínez-Morales and Cornejo-Tenorio2015). We used these seed species as models to assess rodent seed predation in general, and not specifically for seeds defecated by monkeys. For this reason, we did not cover seeds with faeces.
Seeds were obtained from fruits under fruiting trees; they were washed and kept in the shade until used in seed stations within a few days. One or two seed stations per species (depending on seed availability) were placed in each defecation and control site. Poulsenia seeds were used in the 18 sample areas, while Brosimum was only used in 14 sample areas. Each station contained 5 clean seeds of one of the species. We used a total of 580 seeds (330 Poulsenia seeds and 250 Brosimum seeds).
Each seed station consisted of a small circular area of forest floor (∼10 cm diam) delimited by a 10 cm high plastic strip that was buried 5 cm. Seed stations within a site were placed 1–3 m apart. We placed no dung inside the seed stations. Stations were checked once a week for four weeks and seeds were classified into one of four fate categories: (i) alive (seed with no apparent change or germinating), (ii) gnawed by rodents (with visible marks or husk parts), (iii) removed (seed not found), (iv) attacked by pathogens (seed had a different colour and texture). Seeds were also checked for insect predation, but none had this fate. For analysis, we pooled categories (ii) and (iii) into one category: seed predation/removal by rodents.
Data analyses
To compare the number of species and the abundance of dung beetles among site types, we fitted generalized linear mixed models (GLMM) with Poisson error distribution. To analyse the proportion of seeds dispersed by beetles, both vertically and horizontally, we fitted GLMMs with binomial error distribution. To analyse horizontal dispersal distance, we first obtained the mean horizontal distance for each seed station (we did not include the zero values). We then used a linear mixed model with normal error distribution to analyse those mean values (square-root transformed to achieve homogeneity of variance). In all these models, the fixed factor was site type (1d-defecation vs. 1wk-defecation vs. control) and the random factor was the day on which the trap and stations in one site of each type were deployed. To analyse vertical dispersal distance, we fitted a linear model with normal error on the log-transformed response variable. For this variable, we had many zeroes (i.e., stations in which none of the five seeds had been buried); thus, we used each buried seed as an independent sample, rather than the seed station. In this case, the model did not include random factors but only the fixed factor.
To assess the effect of site type on the relative abundance of rodents and the duration of rodent visitation, we analysed the cumulative number of records and the cumulative number of minutes, respectively, for two non-independent times: the first 24 hours of camera trapping (1d) and the first week of camera trapping (1wk). We did separate analyses because sample sizes were different for each time (after deleting sampling areas where no rodents were recorded, sample sizes were 16 paired areas for 1d, and 11 paired areas for 1wk). Data were analysed with GLMM’s with Poisson error distribution, with site type (defecation vs. control) as the fixed factor, and paired sampling area as the random factor.
To analyse seed predation/removal by rodents, we first tried fitting various survival models with mixed effects (Austin Reference Austin2017), but the assumptions of these models were not met satisfactorily. Thus, we opted to separately analyse seed predation/removal for two non-independent times: after the first week (1wk) and after four weeks (4wk). We then fitted GLMMs with binomial distribution to model the accumulated proportion of seeds preyed/removed during each time. Fixed factors were site type, seed species, and the interaction between them; the paired sampling area was the random factor, plus the station ID to account for overdispersion.
All data analyses were conducted in R 4.4.1. (R Core Team 2024). Models were fitted using the glmmTMB function in the homonymous package (Brooks et al. Reference Brooks, Kristensen, van Benthem, Magnusson, Berg, Nielsen, Skaug, Maechler and Bolker2017). Hypothesis testing was done using the Anova function in the ‘car’ package (Fox and Weisberg Reference Fox and Weisberg2019), which calculates Type-II analysis of variance F-tests and Wald chi-square tests to assess for fixed effects in LMs and (G)LMMs, respectively. For paired contrasts, we used the function pair in the ‘emmeans’ package using the default Tukey method to correct p-values (Lenth Reference Lenth2023). Model validation was done using a graphical approach (Zuur and Ieno Reference Zuur and Ieno2016) using the check_model function in the ‘performance’ package (Lüdecke et al. Reference Lüdecke, Ben-Shachar, Patil, Waggoner and Makowski2021). No relevant deviations from model assumptions were detected for any of the fitted models.
Results
Dung beetles, dung removal, and secondary seed dispersal
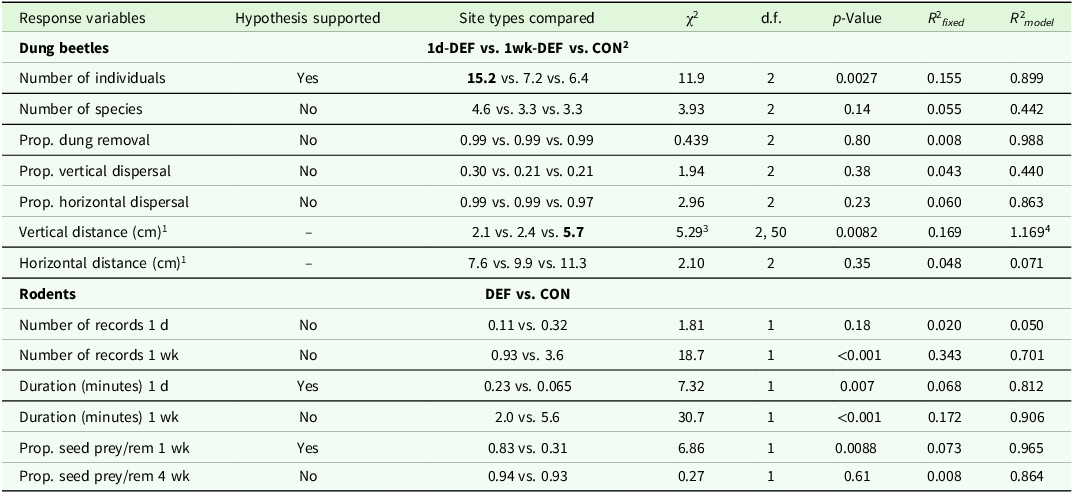
We captured a total of 496 dung beetles belonging to 22 species: 256 individuals and 17 species in 1-day defecation sites (1d-DEF), 121 beetles and 18 species in 1-week defecation sites (1wk-DEF), and 119 individuals and 18 species in control sites (CON). Of the 15 species with ≥5 individuals captured, 13 occurred in all three site types (Table S1). On average, beetle abundance per trap was significantly higher in 1d-DEF sites (15.2) compared to 1wk-DEF sites (7.2) and control sites (6.4), but there was no statistically significant difference in the number of species among site types (4.6 vs. 3.3 vs. 3.3; here and elsewhere the statistical values corresponding to all comparisons mentioned in the text can be found in Table 1).
Results of testing our hypothesis that howler monkey defecation sites would have higher dung beetle and rodent activity than control sites. The second column indicates whether the hypothesis was supported. Dung beetle variables: number of individuals and species, proportions of dung removed and seeds dispersed, and dispersal distances. Rodent variables: number of independent photo records, duration of rodent visitation, and proportion of seeds preyed/removed. For dung beetles, we compared three site types: 1d-DEF, where monkeys defecated on the day of measurements; 1wk-DEF, where monkeys defecated a week before measurements, and CON, control sites. For rodents, we compared defecation (DEF) and control sites (CON) separately at different times: one day (1 d), one week (1 wk) and four weeks (4 wk). The last two columns indicate the amount of variability explained by fixed factors and the whole model, respectively; the difference between these two values can be attributed to random factors (see data analyses)

1 We had no specific hypothesis and prediction regarding these variables
2 Value shown in bold font is statistically different from the other two
3 F-statistic
4 This model did not include random factors; thus, the value in the last two columns is the same
Dung removal after 24 h was almost complete (0.99) in the three site types (Table 1). The probabilities of seed dispersal also did not differ among site types, both for vertical dispersal (1d-DEF = 0.30, 1wk-DEF = 0.21, CON = 0.21) and horizontal dispersal (1d-DEF = 0.99, 1wk-DEF = 0.99, CON = 0.97; Table 1). Similarly, horizontal dispersal distances were not statistically different among site types (1d-DEF = 7.7 cm, 1wk-DEF = 9.9 cm, CON = 11.3 cm). Seed burial depth was statistically similar in both types of defecation sites (1d-DEF = 2.1 cm, 1wk-DEF = 2.4 cm), but deeper in control sites (5.7 cm; Table 1). The amount of variability in response variables that was explained by the random factor (the day in which traps and seed stations were deployed) was high (Table 1).
Rodents, duration of rodent visitation, and seed predation/removal
We photo-captured a total of 16 species/morphospecies of vertebrates (11 mammals, 4 birds, 1 unknown), 13 in defecation sites and 13 in control sites. Overall, we had 175 independent photo records and 442 minutes of vertebrate visitation. For the pooled rodent category, we had 78 independent records and 133 minutes of visitation (Table S2).
The mean number of rodent records per site during the first day of camera trapping did not differ statistically between defecation and control sites (0.11 vs. 0.32; Table 1). However, after one week, significantly fewer rodent records accumulated at defecation sites than at control sites (0.93 vs. 3.6; Table 1). During the first day, the average number of minutes of rodent visitation was significantly higher in defecation sites than in control sites (0.23 vs. 0.065 min/site; Table 1). However, after one week, the duration of rodent visitation was significantly lower in defecation versus control sites (2.0 vs. 5.6 minutes/site; Table 1).
Regarding seed fate, we found that after 4 weeks, 94% of all seeds had suffered predation/removal by rodents, 2% had been attacked by pathogens, and 4% remained apparently viable. After one week, the probability of seed predation/removal was significantly higher in defecation sites than in control sites (0.83 vs. 0.31; Table 1) but did not differ after four weeks (0.94 vs. 0.93; Table 1). The effects of seed species and of the interaction between seed species and site type were not statistically significant. As with dung beetles, the amount of variability in the response variables that was explained by the random factor (paired sample area) was high (Table 1).
Discussion
Our goal was to test whether defecation sites under the sleeping trees of the Mexican mantled howler monkey can create microhabitat heterogeneity. We hypothesized that dung beetle and rodent activity would be higher in defecation sites. Some of our results supported this hypothesis, but others did not. One day after monkey defecation, dung beetles were more abundant and rodent visitation duration was longer in defecation sites compared to control sites, as expected; these effects were not detected after one week. Similarly, seed predation/removal by rodents was higher in defecation sites after one week, but not after four weeks. Contrary to our expectations, we found that rodents were recorded more often in control sites than defecation sites. Our study shows that monkey defecation sites can create short-term spatial heterogeneity in the understory, although not necessarily in the expected direction. Habitat heterogeneity, even at small spatial and temporal scales, can facilitate species coexistence and thus shape diversity patterns (Mittelbach and McGill Reference Mittelbach and McGill2019). However, our knowledge on the types and scales of subtle habitat changes that animals can create through daily behaviours, such as defecation, is still scant. Our study contributes towards filling this gap.
We found that defecation sites had twice the abundance of dung beetles than control sites, when assessed during the day when monkeys defecated, although the number of species did not differ. Since we placed pitfall traps immediately after a monkey group defecated, the amount of faeces (and urine) was undoubtedly higher in defecation than control sites, creating a stronger and longer-lasting sensory cue. After one week, the effect of the defecation was not detected anymore, and the number of beetles and species captured were the same as in control sites. This last result is comparable to, and coincides with, that for howler monkeys (A. guariba) in Brazil, where dung beetles were not sampled on days when monkeys defecated (Fuzessy et al. Reference Fuzessy, Sobral and Culot2022). Instead, for spider monkeys (Ateles geoffroyii Kuhl, 1820) in Costa Rica, even though sampling was also done on days when monkeys were not observed defecating, dung beetle abundance was higher under frequently used sleeping trees than control sites (Whitworth et al. Reference Whitworth, Whittaker, Huarcaya, Flatt, Morales, Connor, Garrido Priego, Forsyth and Beirne2019). Thus, while some defecation sites may only attract more dung beetles while faeces are present, in others, the effect of defecation sites may be more prolonged. Such differences could be due to differences in the frequency of use of defecation sites or in the beetle communities.
We found no difference in dung removal or secondary seed dispersal in defecation versus control sites. This result might seem paradoxical, since we found twice the abundance of beetles in defecation sites. However, since dung removal was 99% in defecation and control sites, one can deduce that in both sites the number of beetles attracted was enough to bury all dung in 24 h. Mirroring dung removal, horizontal seed dispersal was also observed for almost all artificial seeds, since at least small movements (2–10 cm) are a natural consequence of dung relocation by beetles (Urrea-Galeano et al. Reference Urrea-Galeano, Andresen, Coates, Mora Ardila, Díaz Rojas and Ramos-Fernández2019b). On the other hand, the occurrence of vertical seed dispersal often depends on the size of the seed relative to the size of the beetle handling it, since seeds are ‘dung contaminants’ form a beetle’s perspective (Andresen Reference Andresen2002a). Thus, since beetles removed all dung, and the same beetle species occurred in both site types (Table S1), our result of similar seed burial rates in defecation and control sites is understandable. Interestingly, the study with A. guariba in Brazil, where beetle assemblages had the same abundance in defecation sites and under fruiting trees, found that dung removal and seed burial were higher in defecation sites (Fuzessy et al. Reference Fuzessy, Sobral and Culot2022). A possible explanation for the differences in results can be related to how quickly dung is removed. Both studies used similar amounts of dung to measure dung removal (25 g in our study, 30 g in the Brazilian study). However, while in our study almost all dung had been removed after 24 h, in the Brazilian study, dung removal was slower (77% and 71% removal in defecation and control sites, respectively; Fuzessy et al. Reference Fuzessy, Sobral and Culot2022). Thus, it could be possible that differences in dung removal do exist in our study site between defecation and control sites, but to detect them, we would need to measure dung removal after a shorter time (e.g., 12 h). The same logic can apply to rates of secondary seed dispersal.
Regarding rodents, we found that the duration of rodent visitation was higher in defecation sites than in control sites, when measured after one day. However, after one week, the number of records and the duration of rodent activity were unexpectedly higher in control sites. Several studies have shown that vertebrates, including rodents, are attracted to and forage in mammal latrines (e.g., tapir latrines, Lautenschlager et al. Reference Lautenschlager, Souza, Villar, Galetti and Feeley2024; Srbek-Araujo et al. Reference Srbek-Araujo, Da Cunha and Roper2017; raccoon latrines, Page et al. Reference Page, Swihart and Kazacos2001; Weinstein et al. Reference Weinstein, Moura, Mendez and Lafferty2018). In the only study we could find that has compared vertebrate visitation in monkey defecation sites versus control sites, a large rodent (the paca) was found at a rate 427.2% higher in spider monkey defecation sites (Whitworth et al. Reference Whitworth, Whittaker, Huarcaya, Flatt, Morales, Connor, Garrido Priego, Forsyth and Beirne2019). So, why did we observe the contrary pattern after one week? On the one hand, our result was mostly driven by two paired areas (out of 11), both of which had no rodent records in the defecation site after seven days, but 14–15 records in the control site. On the other hand, it is possible that some rodent species might avoid defecation sites as a means of preventing predation or infection with parasites, as reported for raccoon latrines (Weinstein et al. Reference Weinstein, Moura, Mendez and Lafferty2018). Interestingly, the five visits of coyote (Canis latrans Say, 1823) that we registered occurred in defecation sites (Table S2), which would support the idea of predator avoidance. Overall, given that the variability among sampling areas was very high (i.e., the random factor; Table 1), we recommend additional studies with larger sample sizes.
In terms of seed fate, we found that after one week, seed predation/removal by rodents was higher in defecation sites (83%) than in control sites (31%), as expected, which might be explained by the higher duration of rodent visitation during the first day. However, after four weeks, seed predation/removal was almost complete (93–94%) and did not differ between sites. Our results are consistent with previous findings showing that the presence of dung increases seed predation/removal rates (McConkey Reference McConkey2005; Wehncke et al. Reference Wehncke, Valdez and Domínguez2004), but that this effect diminishes or is not detectable after a short period (1–3 weeks; Andresen Reference Andresen1999; Chapman Reference Chapman1989; Feer and Forget Reference Feer and Forget2002). In contrast to these studies, our experimental seeds were not within monkey faeces, nor did we place any dung inside the seed stations. Thus, our results show that in howler monkey defecation sites, predation pressure can be higher (at least during a short time following the defecation) for any seeds present in those sites, not just for defecated seeds.
How generalizable are the results of our study? We believe that effects of howler monkey defecation sites may be highly context dependent because different species, and even populations, may differ in their defecation pattern. For some howler monkeys, it has been observed that members of a group often defecate from the same or close-by branches, and that they move to lower branches to defecate, resulting in large accumulations of dung in relatively small areas of the forest floor (A. macconnelli, Andresen Reference Andresen2002b; A. caraya, Bravo Reference Bravo2009, Brividoro et al. Reference Brividoro, Kowalewski, Scarry and Oklander2019; A. palliata, Wehncke et al. Reference Wehncke, Valdez and Domínguez2004). In our study, we did not observe these behaviours, and we found that only small portions of dung (∼5–10 g) reached the forest floor, widely dispersed in large areas (∼20 × 20 m). Also, howler monkey species and populations differ in their group sizes (Kowalwski et al. Reference Arroyo-Rodríguez, Andresen, Bravo, Stevenson, Kowalewski, Garber, Cortes-Ortiz, Urbani and Youlatos2015), which affects the amount of dung deposited during group defecations under sleeping trees. Similarly, howler populations living in different forests may differ in their use of space. For example, howlers living in fragments or small islands may use the same sleeping trees more frequently, with a higher recurrence of defecations in the same sites (Arroyo-Rodríguez et al. Reference Arroyo-Rodríguez, Andresen, Bravo, Stevenson, Kowalewski, Garber, Cortes-Ortiz, Urbani and Youlatos2015). Indeed, several studies reporting significant effects of defecation sites have been carried out in islands (A. caraya, Bravo Reference Bravo2012; A. seniculus, Feeley Reference Feeley2005) and forest fragments (A. palliata, Anzures-Dadda et al. Reference Anzures-Dadda, Andresen, Martínez-Velázquez and Manson2011, Reference Anzures-Dadda, Manson, Andresen and Martínez2016).
The frequency of use of sleeping trees, and thus of defecations being deposited under those trees, has most likely a large influence on the effects of a defecation site (Feer et al. Reference Feer, Ponge, Jouard and Gomez2013). As shown by long-term studies, howler monkey groups use different sleeping trees with varying frequencies (Bravo Reference Bravo2009; Julliot Reference Julliot1997). Microhabitat differences associated with defecation sites can be expected to occur more often or be stronger when defecation sites are located under more frequently used sleeping trees. To our knowledge, this has been tested, and confirmed, only once for howler monkeys (Feer et al. Reference Feer, Ponge, Jouard and Gomez2013) and once for spider monkeys (Whitworth et al. Reference Whitworth, Whittaker, Huarcaya, Flatt, Morales, Connor, Garrido Priego, Forsyth and Beirne2019). Nevertheless, these and other studies have generally focused on defecation sites under sleeping trees that were known to be frequently used by monkeys (e.g., Bravo Reference Bravo2009; Fuzzessy et al. Reference Fuzessy, Sobral and Culot2022; Julliot Reference Julliot1997; Whitworth et al. Reference Whitworth, Whittaker, Huarcaya, Flatt, Morales, Connor, Garrido Priego, Forsyth and Beirne2019). As mentioned above, this focus on frequently used defecation sites gives us a partial picture of the effects that howler monkeys may have through their defecations, since some studies report that most sleeping trees are only used occasionally (Brividoro et al. Reference Brividoro, Kowalewski, Scarry and Oklander2019; Julliot Reference Julliot1996). In our study, we lacked information on the frequency of use of defecation sites. As such, the sites we used represented a random sample of all possible defecation sites under sleeping trees, which may give us a more general idea of the effects of howler monkey defecations. Nonetheless, the frequency of use of defecation sites is an important variable that future studies will need to take into account, to gain a better understanding of the spatial and temporal scales of habitat heterogeneity that howler monkeys create within their home ranges.
In conclusion, our study shows that the defecation sites of the Mexican mantled howler monkey can create short-term microhabitat heterogeneity in terms of dung beetle and rodent activity. It confirms that animals, other than ecosystem engineers, can introduce subtle changes in their habitats through their behaviours. A recent review has shown that animals introduce heterogeneity in their habitat at a variety of spatial scales by depositing chemical elements unevenly in the soil through their wastes, which can shape patterns of biodiversity and species interactions (Ferraro and Lienau Reference Ferraro and Lienau2025 and references therein). We believe that our study adds to the growing evidence that deposition of animal faeces can create microhabitat heterogeneity through other mechanisms as well, such as the attraction or repulsion of other organisms.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/S0266467426100480
Acknowledgements
We thank Rosamond Coates for logistical support at the Los Tuxtlas Biological Station. We thank Fernando Ocampo Saure and Nury Alfaro for their help and guidance on camera trapping and photo processing. We are grateful to Sr. Carlos Santos Landa Ferman for sharing his knowledge of the forest and the monkeys and assisting in all the fieldwork. We also thank Carmen Vázquez Ribera for helping with fieldwork.
Financial support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
The authors declare none.
Ethical standards
This study complies with ethical standards. Dung beetle sampling was approved by the Mexican government secretariat in charge of the environment (Secretaría de Medio Ambiente y Recursos Naturales; permit FAUT-0018).