Dopaminergic and glutamatergic abnormalities are recognised as core components of the neurobiology of schizophrenia. Reference McCutcheon, Krystal and Howes1–Reference Coyle3 Evidence suggests that striatal dopaminergic hyperactivity may occur downstream of changes in the glutamatergic system – for example, induced by N-methyl-D-aspartate receptor antagonism. Reference Kokkinou, Irvine, Bonsall, Natesan, Wells and Smith4 Antipsychotic treatments primarily target dopamine D2 receptors: Reference Lobo, Whitehurst, Kaar and Howes5 by reducing presynaptic dopamine activity, antipsychotics may indirectly modulate glutamatergic signalling. Reference Egerton, Bhachu, Merritt, McQueen, Szulc and McGuire6 Specifically, blocking of D2 autoreceptors on dopaminergic neurons reduces the feedback inhibition of dopamine release, potentially affecting glutamatergic activity in subcortical structures. Reference Ford7,Reference Fasano, Kortleven and Trudeau8 Additionally, D2 heteroreceptors located on glutamatergic neurons, particularly in cortical regions, may play a role by altering the excitability of these neurons and influencing glutamate release. Reference Borroto-Escuela, Pintsuk, Schäfer, Friedland, Ferraro and Tanganelli9,Reference Borroto-Escuela, Tarakanov, Brito and Fuxe10 These mechanisms align with emerging evidence demonstrating that dopaminergic–glutamatergic interactions are critical for understanding both the therapeutic and side-effects of antipsychotic drugs.
Studies looking at acute effects in rodents have generally reported increases in glutamate levels following administration of D2 antagonists. For example, microdialysis studies have shown that a single dose of lurasidone increased glutamate efflux in rat medial prefrontal cortex and nucleus accumbens, while intraperitoneal administration of quetiapine increased medial prefrontal cortex glutamate release. Reference Yamamura, Ohoyama, Hamaguchi, Kashimoto, Nakagawa and Kanehara11,Reference Huang, Panos, Kwon, Oyamada, Rajagopal and Meltzer12 In contrast, chronic administration in rodents appears to reduce glutamatergic metabolites, possibly reflecting system adaptation to prolonged exposure. For instance, chronic oral administration of clozapine, haloperidol and olanzapine for 21 days reduced glutamatergic metabolites in the frontal cortex and hippocampus, as measured by proton nuclear magnetic resonance (1H-NMR) spectroscopy. Reference McLoughlin, Ma, Tsang, Jones, Cilia and Hill13 This was also seen using microdialysis, where chronic antipsychotic treatment reduced methamphetamine-induced increases in glutamate levels in mouse frontal cortex. Reference Abekawa, Ito, Nakagawa, Nakato and Koyama14 However, based on proton magnetic resonance spectroscopy (1H-MRS), 6 months of haloperidol treatment did not affect glutamatergic concentrations in multiple other brain regions. Reference Bustillo, Barrow, Paz, Tang, Seraji-Bozorgzad and Moore15 While most studies focus on the medial prefrontal cortex, the heterogeneity in findings could have been influenced by differences in methodologies such as the type of antipsychotic, duration of treatment, measurement technique (e.g. microdialysis versus MRS) and whether the experimental paradigm targeted baseline or pharmacologically stimulated glutamate levels. Further research, including studies on intermediate durations such as 1-week administration, could help resolve these discrepancies and clarify the mechanisms of adaptation.
Clinical studies further suggest that antipsychotic treatment may influence brain glutamate levels in schizophrenia, although findings remain inconsistent. A mega-analysis of cross-sectional data reported that antipsychotic medication exposure was associated with lower medial frontal cortex glutamate levels, with glutamatergic metabolites negatively correlated with antipsychotic dose. Reference Merritt, McGuire, Egerton, Block, Bloemen and Borgan16 Similarly, a meta-analysis of longitudinal studies found that levels of frontal glutamate + glutamine (Glx) significantly decreased following antipsychotic treatment, with an effect size of −0.35 (P = 0.0003), but no changes were observed in thalamic glutamate levels. Reference Kubota, Moriguchi, Takahata, Nakajima and Horita17 De la Fuente-Sandoval et al Reference de la Fuente-Sandoval, León-Ortiz, Azcárraga, Stephano, Favila and Díaz-Galvis18 observed reduced glutamate levels in the striatum of antipsychotic-naive patients following 4 weeks of treatment, and Egerton et al Reference Egerton, Broberg, Van Haren, Merritt, Barker and Lythgoe19 reported a reduction in anterior cingulate cortex (ACC) glutamate levels in minimally treated patients during their first episode of non-affective psychosis following 4 weeks of antipsychotic treatment. In contrast to the meta-analytic finding of no change in thalamic glutamate, Bojesen et al Reference Bojesen, Ebdrup, Jessen, Sigvard, Tangmose and Edden20 reported a reduction in elevated thalamic glutamate levels following 6 weeks of aripiprazole treatment in antipsychotic-naive, first-episode psychosis patients; however, no change was observed in ACC glutamate levels. Similarly, other smaller longitudinal studies, such as those of Zahid et al Reference Zahid, McCutcheon, Borgan, Jauhar, Pepper and Nour21 and Kraguljac et al, Reference Kraguljac, Morgan, Reid, White, Jindal and Sivaraman22 found no significant changes in ACC or hippocampal glutamate or Glx levels following 4–6 weeks of antipsychotic treatment.
Notably, these clinical studies were largely observational, lacked placebo controls and were conducted in patient populations where the effects of illness progression, social factors and substance misuse are challenging to disentangle. To address these limitations, it is critical to investigate the direct pharmacological effects of antipsychotic drugs in a controlled setting using healthy volunteers. This approach allows us to disentangle drug-specific effects from the broader influence of schizophrenia pathology.
Preclinical and clinical findings suggest that antipsychotics may reduce glutamate metabolites, but it remains unclear how specific drug mechanisms influence this effect. For example, dopamine D2/3 antagonists such as amisulpride may decrease glutamate levels through downstream modulation of cortico-striatal circuits, while the partial agonism of dopamine D2/3 receptors combined with 5HT2A antagonism, as seen with aripiprazole, could produce either a similar or attenuated effect depending on the balance of agonistic versus antagonistic activity. Reference Ma, Raivio, Sabrià and Ortiz23 To date, these mechanistic distinctions have not been systematically examined in human studies.
The present study aims to address this gap by investigating the effects of 1 week of drug administration in a healthy volunteer group, focusing on amisulpride and aripiprazole. A 1-week regimen was chosen because this is sufficient to capture early pharmacological effects and potential system adaptations; previous meta-analytic work has shown that antipsychotic action starts early – within the first week of treatment – and is cumulative during the ensuing weeks, suggesting that this duration is suitable for observation of initial neurochemical changes. Reference Agid, Kapur, Arenovich and Zipursky24 Given the general trend of glutamate reduction with antipsychotic treatment observed in clinical studies, we hypothesised that both dopamine D2/3 antagonism and partial agonism would reduce glutamate metabolites, although the magnitude of change may differ due to the drugs’ distinct mechanisms of action.
Method
Study design and ethical approval
Ethical approval
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation, and with the Helsinki Declaration of 1975 as revised in 2013. All procedures involving human subjects/patients were approved by the London – West London and GTAC NHS Research Ethics Committee (no. 18/LO/1044). All participants provided informed written consent prior to participation.
Study design
This study used a single-centre, randomised, double-blind, placebo-controlled, cross-over design. In arm 1, participants were randomised to receive amisulpride or placebo first; in arm 2, participants were randomised to receive aripiprazole or placebo first. The order was counterbalanced to ensure approximately equal numbers receiving drug or placebo first. The amisulpride and placebo cross-over study (arm 1) and aripiprazole and placebo cross-over study (arm 2) were conducted sequentially at the same site (see Fig. 1).
Participant eligibility
Participants were recruited through public advertisement and research volunteer circulars. The exclusion criteria were: a history of psychiatric illness (including alcohol/substance dependence or abuse, other than caffeine or nicotine) as determined by self-report and the Mini International Neuropsychiatric Interview; Reference Sheehan, Lecrubier, Sheehan, Amorim, Janavs and Weiller25 current use of illicit substances, as determined by urine drug testing and self-report; pregnancy, as determined by urine pregnancy testing and self-report; breastfeeding; self-report of a first-degree relative with a psychotic disorder; contraindications to dopamine antagonists/partial agonists or magnetic resonance imaging (MRI) scanning; significant previous use of psychotropic or dopamine-modulating drugs; participation in a study of unlicensed medicines within the previous 30 days; and the presence of significant central nervous system disorders (e.g. head trauma, epilepsy). Additional exclusions included medical disorders or clinically relevant abnormal findings at screening assessment, as determined by the principal investigator, and any condition that would pose a significant risk to the participant’s safety or invalidate their participation in the study.
Study allocation
The allocation of the order of drug administration for participants was determined independently of the study team using a generalised Latin square, which accounts for order and first-order carry-over effects. Both study clinicians and participants were blinded to the treatment order, with participant identities allocated by an independent researcher.
Medication
The study medication was manufactured by the Pharmacy Manufacturing Unit, Guy’s and St Thomas’ NHS Foundation Trust, London, UK. Lactose powder was used to produce placebo capsules that were identical in shape, size and colour to the capsules containing the active compounds. The study medication was packaged and labelled by the Pharmacy Manufacturing Unit, Guy’s and St Thomas’ NHS Foundation Trust in accordance with The Rules Governing Medicinal Products in the European Union, Volume 4: Good Manufacturing Practice (GMP). Following manufacture, the study medication was released to the Maudsley Hospital Pharmacy, South London and Maudsley NHS Foundation Trust for dispensing.
Pharmacokinetic assessments
Plasma amisulpride or aripiprazole + dehydro-aripiprazole levels were measured following each treatment week, and aripiprazole + dehydro-aripiprazole levels were also measured following the washout period to detect and exclude slow metabolisers of aripiprazole. Participants were asked to record the time of day of the last dose taken before the blood draw, which was scheduled to occur as close as possible to the MRI scanning, with a maximum interval of 2.5 h.
Imaging
Participants attended on four occasions: baseline visit (at which they received the first compound, either the drug or placebo), follow-up one, dosing visit (when the second compound, either the drug or placebo, was administered) and follow-up two. Prior to the administration of the initial study, drug participants underwent a baseline MRI. MRI scans were conducted the day following the final dose of both drug and placebo.
1H-MRS acquisition
Scans were acquired on a 3 T Siemens MAGNETOM Prisma scanner (Siemens Healthcare, Erlangen, Germany) using a 64-channel head coil. Single-voxel spectroscopy data were collected using a point-resolved spectroscopic (PRESS) sequence with the following parameters: echo time 30 ms, repetition time 3000 ms, 96 averages, bandwidth 2500 Hz). Spectral resolution was improved using high-order shimming, and manual shimming was performed to optimise B0 field homogeneity across each voxel, with an unsuppressed water signal line width of <20 Hz considered satisfactory.
1H-MRS voxels were positioned in the ACC (20 × 20 × 20 mm3; see Supplementary Fig. 1a available at https://doi.org/10.1192/bjp.2025.10319), the right striatum (20 × 20 × 20 mm3; see Supplementary Fig. 1b) and the left thalamus (20 × 15 × 20 mm3; see Supplementary Fig. 1c). Non-water-suppressed spectra were obtained for quantification (see Supplementary Figs 2, 3 and 4 for sample spectra). An anatomical T1 magnetization prepared rapid-gradient echo image was acquired for each participant to aid placement of voxels, and for co-registration. These images were also used for tissue segmentation to correct for the grey matter, white matter and cerebrospinal fluid (CSF) content of 1H-MRS voxels. 1H-MRS details are provided in the minimum reporting standards for the in vivo magnetic resonance spectroscopy checklist (see Supplementary Tables 1 and 7), according to consensus recommendations. Reference Lin, Andronesi, Bogner, Choi, Coello and Cudalbu26
1H-MRS quantification
Spectra were analysed using LCModel version 6.3-0I (http://s-provencher.com/lcm-manual.shtml), and raw metabolite concentrations were estimated through a standard basis set of 16 metabolites (L-alanine, aspartate, creatine, phosphocreatine, GABA, glucose, glutamine, glutamate, glycerophosphocholine, glycine, myo-inositol, L-lactate, N-acetylaspartate, N-acetylaspartylglutamate, phosphocholine and taurine). Metabolite analyses were restricted to spectra with line width (full-width at half-maximum, FWHM) ≤0.1 ppm, Cramér–Rao lower bounds (CRLB) for glutamate ≤20% and signal:noise ratio ≥5. Water-scaled glutamate, Glx and glutamine levels obtained from LCModel were corrected for voxel tissue content using the formula below. M corr and M are corrected and uncorrected metabolite concentrations, respectively, and wm, gm and CSF indicate the fractions of white and grey matter and CSF, respectively, per voxel: Reference Gasparovic, Chen and Mullins27
Dosing
Dosing of study medication was carried out by a study clinician who also monitored participants for approximately 40 min following dosing, for reasons of safety. Participants were given the oral medication to take home for the next 7 days. For enhanced tolerability, the doses were gradually raised and the titration regimes were as follows: for amisulpride, 1st day 200 mg, 2nd day 300 mg and 3rd to 7th day 400 mg; and for aripiprazole, 1st day 5 mg, 2nd day 7.5 mg and 3rd to 7th day 10 mg.
Statistical analysis
Data analysis was conducted in Python using open-source packages: pandas for data processing, Reference McKinney28 NumPy for descriptive statistics, Reference Harris, Millman, van der Walt, Gommers, Virtanen and Cournapeau29 statsmodels for implementing linear mixed models and mixed-effects analysis of variance (ANOVA), Reference Seabold and Perktold30 as well as matplotlib Reference Hunter31 and seaborn Reference Waskom32 for data visualisation.
Linear mixed-effects models were used to examine the effect of drug versus placebo on Glx levels. To account for within-subject correlations among repeated measurements collected from the same individual, a random intercepts-only model was used. The dependent variable in the model was Glx levels, which were measured as a combined value across three brain regions: ACC, striatum and thalamus. Brain region was included as an independent variable to assess its effect on Glx levels and potential interactions with treatment. The primary predictor of interest was the fixed effect of treatment condition (drug versus placebo). Additional predictors included baseline metabolite levels, treatment order, age and gender. Models were fit by maximum likelihood estimation (MLE). The results of glutamate and glutamine levels are also available in Supplementary Tables 2 and 3 and Supplementary Figs 5 and 6. Summary statistics for Glx, Glu and Gln levels aross conditions and brain regions are reported in Supplementary Tables 4–6.
After fitting the primary model, we explored the interaction between brain region (ACC, striatum thalamus) and treatment condition (drug versus placebo). This analysis was conducted to determine whether the effect of the antipsychotic treatment on glutamate levels varied across the different regions.
In addition to the primary analysis, a sensitivity analysis was conducted using a mixed-effects ANOVA to examine the combined effects of amisulpride and aripiprazole in one model. This allowed us to assess the relative impact of each treatment on Glx levels, providing a more comprehensive understanding of how the two drugs compare when considered simultaneously. By including both treatments in the same model, we could evaluate whether their effects differed significantly and how they influenced the outcome in comparison with each other.
Results
Demographic details of participants are provided in Table 1. Twenty-five healthy participants (15 females, 10 males; mean age 26.5 ± 7.74 years) completed baseline and follow-up visits for amisulpride, while a further 25 healthy participants (14 females, 11 males; mean age 26.68 ± 8.74 years) completed baseline and follow-up visits for aripiprazole.
Sociodemographic and clinical characteristics of participants
Aripiprazole
Aripiprazole demonstrated a statistically significant effect on Glx levels (z = 2.71, 95% CI [0.15, 0.94], P = 0.007, Cohen’s d = 0.31; see Table 2). The mean Glx level was 8.16 institutional units (s.d. = 2.40) at the end of the aripiprazole intervention, compared with 7.61 institutional units (s.d. = 2.36) at the end of the placebo condition. This indicates a significant increase in Glx levels in the aripiprazole condition compared with placebo. A post hoc analysis was conducted to explore the interaction between brain region and treatment condition: this analysis revealed no significant interaction, indicating that the observed effect of aripiprazole on Glx levels did not differ significantly across regions. After adjusting for plasma drug levels and the active metabolite dehydro-aripiprazole, the effect remained significant.
Mean (s.d.) of glutamate + glutamine (Glx) levels at baseline, following amisulpride, aripiprazole or placebo

a Comparison of treatment effect with placebo is quantified by the corresponding β-coefficient, representing changes in metabolite levels; for example, aripiprazole resulted in an increase of 0.55 institutional units in Glx levels.
Amisulpride
Amisulpride did not show a detectable effect on Glx levels compared with the placebo condition (z = 0.14, 95% CI [−0.40, 0.45], P = 0.891, Cohen’s d = 0.02; see Table 2). The mean Glx level was 8.09 institutional units (s.d. = 2.62) at the end of the amisulpride intervention, compared with 8.06 institutional units (s.d. = 2.61) at the end of the placebo condition. Adjusting for plasma drug levels did not alter these findings.
Sensitivity analyses
In order to explore potential class-wide effects of antipsychotic treatment on Glx levels we conducted a sensitivity analyses, examining the effect of amisulpride and aripiprazole combined versus placebo. We found that the antipsychotic treatment appeared to increase Glx levels compared with placebo, but this was not formally significant (β = 0.30, P = 0.051).
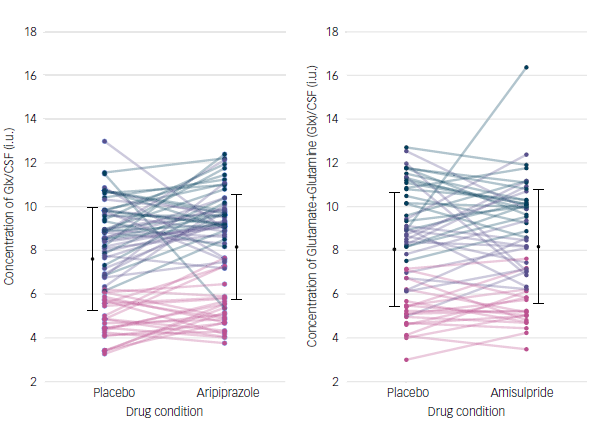
As evident from Fig. 2, outlier values were present in both the amisulpride and aripiprazole groups. To account for this, we conducted a sensitivity analysis excluding observations identified as outliers (defined as data points more than three standard deviations from the mean); one data point was removed from each drug group. The results indicated a statistically significant effect for aripiprazole (z = 2.52, 95% CI [−1.10, −0.14], P = 0.012), while no significant effect was observed for amisulpride (z = −0.27, 95% CI [−0.52, 0.39], P = 0.791).
Diagram illustrating the within-subject, placebo-controlled, cross-over study design. 1H-MRS was carried out at baseline, follow-up one and follow-up two visits. The study drug in each arm was administered at the end of the baseline visit, and again at the dosing visit following the washout period. Medications were titrated for tolerability: amisulpride (day 1, 200 mg; day 2, 300 mg; days 3–7, 400 mg) and aripiprazole (day 1, 5 mg; day 2, 7.5 mg; days 3–7, 10 mg). The order of treatments was randomised and counterbalanced to ensure approximately equal numbers of subjects receiving drug or placebo first. Participants were blinded to treatment allocation. The washout period was a minimum 10 and 28 days for amisulpride and aripiprazole, respectively. 1H-MRS, magnetic resonance spectroscopy.

Individual change in glutamine + glutamine (Glx) levels (left) from placebo to aripiprazole condition, with data plotted for three regions: anterior cingulate cortex (ACC), striatum and thalamus (represented in green, purple and pink, respectively). Group mean (s.d.) Glx levels for both placebo and drug conditions (black circles and error bars represent s.d.). Individual change in Glx levels (right) from placebo to amisulpride condition, with data plotted for three regions: ACC, striatum and thalamus (represented by green, purple and pink respectively). Group mean (s.d.) Glx levels for both placebo and drug conditions (black circles and error bars represent s.d.). CSF, cerebrospinal fluid; i.u., institutional units.
Discussion
This study is the first to investigate the effects of subchronic administration of amisulpride and aripiprazole in healthy participants. We aimed to answer a key question: whether repeated doses of these drugs lead to changes in Glx levels. Understanding these effects is crucial, because glutamatergic dysregulation has been implicated in psychosis and antipsychotic treatments may modulate this system.
Our study combined glutamate levels from three key regions: the ACC, thalamus and striatum, which allowed us to examine the overall impact of the drugs across these areas. The composite analysis revealed that both aripiprazole and amisulpride increased Glx levels compared with placebo; however, while the effects for both drugs were similar, only that for aripiprazole reached statistical significance. Importantly, this effect remained significant after accounting for the region–treatment condition interaction, suggesting that it is a relatively brain-wide, as opposed to localised, effect. This contrasts with clinical studies where localised effects have been observed, with reductions in glutamate levels noted specifically in the ACC, Reference Egerton, Broberg, Van Haren, Merritt, Barker and Lythgoe19 striatum Reference de la Fuente-Sandoval, León-Ortiz, Azcárraga, Stephano, Favila and Díaz-Galvis18 and thalamus Reference Bojesen, Ebdrup, Jessen, Sigvard, Tangmose and Edden20 following antipsychotic treatment. Such discrepancy could reflect alterations in glutamatergic circuits in people with psychosis relative to healthy controls. Reference Howes, Dawkins, Lobo, Kaar and Beck33,Reference Jauhar, McCutcheon, Borgan, Veronese, Nour and Pepper34 It is clinically relevant because it adds to other evidence that glutamatergic circuits respond differently to some challenges in psychosis, Reference Pillinger, Rogdaki, McCutcheon, Hathway, Egerton and Howes35 indicating that these circuits are perturbed. It also highlights the value of studies in healthy volunteers in providing normative data for comparison, without which it is not possible to know whether alterations in patients are a direct drug effect or may reflect an interaction with the underlying pathophysiology.
The location of the prescribed voxel within the ACC may also have contributed to the observed findings; our voxel primarily covered the ventral portion of the medial prefrontal cortex, consistent with previous functional parcellation studies. Reference de la Vega, Chang, Banich, Wager and Yarkoni36 This distinction is important, because emerging evidence suggests region-specific glutamatergic profiles along the dorsal–ventral axis of the ACC. For example, in unmedicated individuals with schizophrenia, elevated glutamatergic metabolites have been reported in ventral ACC regions whereas dorsal regions have shown no significant differences or even reductions. Reference Simmonite, Steeby and Taylor37 Recent data from healthy populations further support this heterogeneity, showing higher glutamatergic metabolite concentrations in ventral compared with dorsal ACC regions. Reference Choles, Archibald, Ortiz, MacMillan, Zölch and Kramer38 Thus, our voxel’s ventral positioning may have captured an area that is more sensitive to pharmacological modulation, particularly in the context of dopaminergic–glutamatergic interactions. These findings highlight the importance of careful voxel placement and anatomical referencing in spectroscopy studies, and may help in contextualisation of our results in light of inconsistencies in the broader literature.
Preclinical studies offer further support for an interaction between dopamine and glutamate systems. For example, chronic oral administration of clozapine, haloperidol and olanzapine reduced glutamatergic metabolites in the frontal cortex and hippocampus of rats. Reference McLoughlin, Ma, Tsang, Jones, Cilia and Hill13 Similarly, microdialysis studies demonstrate that some antipsychotics, such as lurasidone, increase glutamate efflux in the medial prefrontal cortex, Reference Huang, Panos, Kwon, Oyamada, Rajagopal and Meltzer12 which suggests that specific pharmacological profiles may elicit divergent effects on the glutamatergic system. These findings align with some human imaging studies showing an inverse relationship between cortical glutamate concentrations and striatal dopamine synthesis capacity in psychosis, Reference Jauhar, McCutcheon, Borgan, Veronese, Nour and Pepper34 although this was not replicated by another study. Reference Bojesen, Ambrosen, Sigvard, Nielsen, Gjedde and Kumakura39 Nevertheless, these findings suggest that glutamate modulation could be a shared mechanism of these drugs, independent of specific pharmacological profiles, providing important insights into their broader neurochemical impact.
In clinical populations, studying the impact of antipsychotics on glutamate levels is challenging due to various confounding factors, including the heterogeneity of psychotic disorders and the lack of placebo control. These factors complicate the interpretation of changes in metabolite levels post-treatment. Thus, our study highlights the value of healthy participants as a model in isolating the direct effects of antipsychotic medications without the influence of underlying pathology or other variables, providing normative data to aid the interpretation of clinical findings. Despite the insights thereby gained, it is important to acknowledge the limitations of studying healthy individuals. While the binding profiles of these drugs are unlikely to differ between healthy and clinical populations, the effects of antipsychotics may interact with the underlying pathology in individuals, leading to different outcomes. For example, the presence of psychosis or chronic illness could modulate the observed effects on glutamate metabolites. Another limitation of our study is that participants were not scanned at the same time point, which could have influenced glutamate levels due to potential diurnal or other time-dependent variations. Future studies should control for timing to reduce this source of variability. Relatedly, while we observed significantly higher Glx levels during aripiprazole compared with placebo, the data suggest that this effect is driven more by a decrease in Glx under the placebo condition than by an absolute increase during aripiprazole administration. This finding, while statistically robust in the context of our randomised, cross-over design, highlights the need for caution in interpreting absolute changes over time. Indeed, the stability of Glx levels across repeated measures is not well established, and previous research has reported both moderate reliability and sensitivity to various physiological Reference Di and Tasker40 and contextual factors. Reference John, Kodama and Siegel41,Reference Sailasuta, Ernst and Chang42 As such, the observed pattern may reflect natural fluctuation rather than a true pharmacological increase. We therefore emphasise that the increase in Glx associated with aripiprazole should be interpreted as a relative effect when compared with placebo, and future studies should further investigate the temporal dynamics and test–retest reliability of Glx measures in similar populations.
We also note that our study employed a 1-week treatment duration. Previous research, including work by Emsley et al Reference Emsley, Rabinowitz and Medori43 and Agid et al, Reference Agid, Kapur, Arenovich and Zipursky24 shows that this time frame is sufficient to observe the clinical effects of antipsychotics. However, it is possible that there may be longer-term adaptive changes in the glutamatergic system with sustained antipsychotic treatment that lead to glutamate reductions. Future studies could explore these possibilities in healthy volunteers with extended treatment durations to better understand the trajectory of changes over time.
This study’s design has several strengths. Using a cross-over approach, each participant served as their own control, reducing the influence of confounding variables. Additionally, plasma levels were assessed to confirm adherence to the medication regimen, ensuring reliability in interpreting the results. Furthermore, the careful titration of amisulpride and aripiprazole minimised the risk of side effects.
Implications
The implications of our findings include identification of glutamatergic effects that could contribute to the mechanism of antipsychotic action, although it remains to be determined whether this underlies clinical efficacy for psychosis. These findings are also useful in helping to interpret the effects of antipsychotics on glutamatergic measures in clinical studies where other factors complicate interpretation. Our results also underline the necessity of accounting for antipsychotic treatment when interpreting comparisons of glutamatergic measures between those taking antipsychotics and untreated individuals or healthy controls, because the drugs themselves may influence metabolite levels.
Summary and future directions
One week of aripiprazole administration in healthy participants altered brain Glx levels as compared with placebo administration. Our findings provide novel insights into the relationship between antipsychotic treatment and brain metabolites in a healthy participant cohort. While longitudinal studies have explored these effects in clinical populations, further research – including longer-term studies in healthy individuals – is warranted to deepen our understanding of the mechanisms underlying these effects.
Supplementary material
The supplementary material is available online at https://doi.org/10.1192/bjp.2025.10319
Data availability
The data that support the findings of this study are available from the corresponding author, U.Z., upon reasonable request.
Acknowledgements
The authors thank those individuals who participated in the study. We also extend our thanks to Angela Stewart Park for designing Fig. 1.
Author contributions
U.Z.: methodology, data analysis, data curation, visualisation, writing (original draft, review and editing), investigation, project administration, data collection. M.O.: methodology, data analysis, data collection, writing (review and editing), investigation, project administration. P.S.: methodology, data analysis, writing (review and editing), investigation, project administration, data collection. D.J.L.: methodology, data analysis, writing (review and editing). C.F.: methodology, data curation, visualisation, writing (review and editing). K.D.: methodology, writing (review and editing). A.K.: visualisation, writing (review and editing). M.B.W.: data analysis, writing (review and editing). T.W.: data collection, writing (review and editing). E.C.O.: data collection, writing (review and editing). B.S.: investigation, data collection, writing (review and editing). A.B.: data collection, writing (review and editing). M.D.: data curation, data analysis, writing (review and editing). R.L.: data curation, visualisation, writing (review and editing). R.A.M.: investigation, writing (review and editing). R.M.: supervision, resources, writing (review and editing). T.R.M.: supervision, writing (review and editing), project administration, conceptualisation. M.A.M.: data analysis, writing (review and editing), conceptualisation, supervision. O.D.H.: methodology, data analysis, data curation, visualisation, writing (review and editing), validation, supervision, resources, project administration, funding acquisition, investigation, conceptualisation.
Funding
U.Z. was supported by funding from The Lord Leverhulme Charitable Trust. O.D.H. is funded by the UK Medical Research Council (grant no. MC_ U120097115), the Maudsley Charity (grant no. 666), the Wellcome Trust (grant no. 094849/Z/10/Z) and the National Institute for Health Research Biomedical Research Centre (NIHR-BRC) at South London and Maudsley (SLaM) National Health Service Foundation Trust and King’s College London. R.A.M.’s work is funded by a Wellcome Trust Clinical Research Career Development Fellowship (no. 224625/Z/21/Z). P.S. has been supported by a PhD studentship jointly funded by NIHR-BRC at SLaM and the Department of Neuroimaging, King’s College London, and is currently supported by the European Union’s Horizon 2020 research and innovation programme under grant agreement no. 964874 (REALMENT). M.O. was supported by an NIHR Academic Foundation post and an NIHR Academic Clinical Fellowship, and acknowledges support from NIHR-BRC. E.C.O. acknowledges funding from NIHR. M.D. was supported by EU-AIMS (European Autism Interventions)/EU AIMS-2-TRIALS, an Innovative Medicines Initiative Joint Undertaking under grant agreement no. 777394. Infrastructure support was provided by NIHR-BRC and the NIHR Imperial Clinical Research Facility.
Declaration of interest
O.D.H. has received investigator-initiated research funding from and/or participated in advisory/speaker meetings organised by Angellini, Autifony, Biogen, Boehringer-Ingelheim, Eli Lilly, Elysium, Heptares, Global Medical Education, Invicro, Jansenn, Karuna, Lundbeck, Merck, Neurocrine, Ontrack/Pangea, Otsuka, Sunovion, Recordati, Roche, Rovi and Viatris/ Mylan. He was previously a part-time employee of Lundbeck A/v. Neither O.D.H. nor his family have holdings or a financial stake in any pharmaceutical company. He has a patent for the use of dopaminergic imaging. R.A.M. has received speaker/consultancy fees from Boehringer Ingelheim, Janssen, Karuna, Lundbeck, Newron, Otsuka and Viatris, and co-directs a company that designs digital resources supporting the treatment of mental ill health. M.A.M. has received speaker/consultation fees from Takeda, Neurocrine, Boehringer Ingelheim, Lundbeck and Nxera, and has acted as an advisor for Quolet Pharmaceuticals. M.B.W.’s main employer is Perceptive Inc., a contract research organisation that provides services to the pharmaceutical and biotechnology industries, and he has also received support (speaking fees, travel) from Compass Pathways. T.R.M. is an employee of Pasithea Therapeutics, in which he owns shares and stock options. He has received speaker/consultation fees from Viatris, Lundbeck and Pfizer. All the other authors declare no conflicts of interest.







eLetters
No eLetters have been published for this article.