Significant Outcomes
-
• The distribution of the leptin (LEP) genotype differed significantly between schizophrenia (SCZ) patients and healthy controls, with the heterozygous LEP GA genotype being more prevalent in controls than in SCZ patients (p = 0.026).
-
• Among SCZ patients, the distribution of leptin receptor (LEPR) genotypes varied significantly according to suicide attempt history, with the heterozygous LEPR AG genotype being more common in non-attempted patients than in those with suicide attempts (p = 0.048).
-
• Logistic regression analysis indicated that LEPR gene polymorphism (p = 0.023), number of hospitalizations (p = 0.036), and Positive and Negative Syndrome Scale-psychopathology scores (p = 0.023) were significant independent predictors of suicide attempt history in SCZ patients.
Limitations
-
• Due to timing constraints, serum leptin levels could not be measured alongside the genetic analysis of LEP and LEPR polymorphisms.
-
• The relatively small sample size may have reduced the statistical power of the findings, limiting the generalizability of the results.
-
• The cross-sectional nature of the study may limit causal inferences about LEP and LEPR gene polymorphisms in SCZ.
Introduction
Schizophrenia (SCZ) is a chronic and severe psychiatric disorder associated with a markedly increased risk of suicide. Lifetime suicide rates among individuals with SCZ are estimated to range between 4% and 10%, while suicide attempts occur in up to half of affected patients (Hor & Taylor, Reference Hor and Taylor2010; Olfson et al., Reference Olfson, Stroup, Huang, Wall, Crystal and Gerhard2021; Correll et al., Reference Correll, Solmi, Croatto, Schneider, Rohani-Montez, Fairley, Smith, Bitter, Gorwood, Taipale and Tiihonen2022). This heightened vulnerability is driven by a complex interaction of demographic, clinical, psychosocial, and biological factors. Clinically, depressive symptoms, prior suicide attempts, substance misuse, hopelessness, poor treatment adherence, and recent hospital discharge have consistently been identified as robust predictors of suicidal behavior in SCZ. Demographic characteristics, including younger age, male sex, higher education, and good premorbid functioning, have also been linked to increased suicide risk (Hawton et al., Reference Hawton, Sutton, Haw, Sinclair and Deeks2005; Carlborg et al., Reference Carlborg, Winnerbäck, Jönsson, Jokinen and Nordström2010; Hor & Taylor, Reference Hor and Taylor2010; Sher & Kahn, Reference Sher and Kahn2019; Li et al., Reference Li, Chen, Zhang, Zhang, Zhang, Chen, Yang, Liang, Hu, Ni, Lu, Sun and Sun2025). In addition, psychosocial stressors such as social isolation, family history of psychiatric disorders or suicide, and childhood trauma, particularly sexual abuse and physical neglect, further increase vulnerability to suicidal behavior in individuals with SCZ. (Mohammadzadeh et al., Reference Mohammadzadeh, Azadi, King, Khosravani and Bastan2019). Beyond these established risk factors, emerging evidence underscores the contribution of genetic mechanisms – including specific single nucleotide polymorphisms (e.g., in SIRT1, MTHFR, ACP1, complement component C4, TNF-α, MBL2, and NR3C1) (Li et al., Reference Li, Yoshikawa and Meltzer2017; Aytac et al., Reference Aytac, Oyaci, Yazar, Erol and Pehlivan2020; Liu et al., Reference Liu, Zhu, Zheng, Tang, Gao, Trihn and Yang Zhang2020; Aytac et al., Reference Aytac, Ozdilli, Tuncel, Pehlivan and Pehlivan2022; Uçak et al., Reference Uçak, Aydin, Koçak and Altınbaş2022, Ebrahimi et al., Reference Ebrahimi, Teymouri, Chen, Mohiuddin, Pouget, Goncalves, Tiwari, Zai and Kennedy2024; Oyaci et al., Reference Oyaci, Yildirim, Aytac, Pehlivan and Aydin2024), altered gene expression in glial and immune-related genes (ALDH1L1, GS, CX3CR1, P2RY12) (Zhang et al., Reference Zhang, Verwer, Lucassen, Huitinga and Swaab2020) in the prefrontal cortex, and broader polygenic influences – in shaping suicide risk in SCZ. Together, evidence from demographic, clinical, psychosocial, and genetic studies highlights the multifactorial nature of suicidal behavior in individuals with SCZ and underscores the need for integrated clinical and genetic approaches to risk assessment and prevention (Pehlivan et al., Reference Pehlivan, Oyaci, Tuncel and Aytac2022; Goker et al., Reference Goker, Aytac and Guclu2023; Oyaci et al., Reference Oyaci, Aytac, Pehlivan and Pehlivan2023; Ozdilli et al., Reference Ozdilli, Aytac, Tuncel, Oyaci, Pehlivan and Pehlivan2024).
Among the biological systems proposed to link metabolic regulation with neuropsychiatric outcomes, the leptin signalling pathway has received increasing attention. Leptin is a pleiotropic hormone involved not only in energy homeostasis but also in neurodevelopment, synaptic plasticity, stress responsivity, and emotional regulation (Park & Ahima, Reference Park and Ahima2015). The leptin gene (LEP), composed of three exons, is located on human chromosome 7q32.1 (Isse et al., Reference Isse, Ogawa, Tamura, Masuzaki, Mori, Okazaki, Satoh, Shigemoto, Yoshimasa and Nishi1995). Recent large-scale genetic studies have identified a locus near 7q32.1 on chromosome 7 as significantly associated with suicide attempts. This link appears to be independent of psychiatric diagnoses, implying a direct genetic contribution to suicide risk (Fiori et al., Reference Fiori, Chawla, Nathan, Maitra, Nagy and Turecki2025). LEP is highly polymorphic, with numerous single nucleotide polymorphisms (SNPs) found throughout its promoter, exon, and intron regions. A notable variant, (-2548G>A) (rs7799039), involves a guanine-to-adenine substitution at position -2,548 upstream of the ATG start codon in the LEP promoter (Mammès et al., Reference Mammès, Betoulle, Aubert, Giraud, Tuzet, Petiet, Colas-Linhart and Fumeron1998).
Leptin exerts its biological effects through binding to its receptor, which activates intracellular signalling pathways that increase energy expenditure and reduce nutrient intake. Leptin receptor belongs to the cytokine receptor superfamily and is expressed in various tissues, including the placenta. The leptin receptor gene (LEPR) is situated on chromosome 1 at cytogenetic band 1p31.3 (Marroqui et al., Reference Marroqui, Gonzalez, Neco, Caballero-Garrido, Vieira, Ripoll, Nadal and Quesada2012; Aytac et al., Reference Aytac, Oyaci, Aytac, Pehlivan, Alptekin and Pehlivan2025). Large-scale genetic analyses have also implicated a locus on chromosome 1 in suicide attempt risk, though results are somewhat inconsistent, establishing chromosome 1 as a key region in the genetic architecture of suicide (Kimbrel et al., Reference Kimbrel, Ashley-Koch, Qin, Lindquist, Garrett, Dennis, Hair, Huffman, Jacobson, Madduri, Trafton, Coon, Docherty, Kang, Mullins, Ruderfer, Harvey, Mcmahon, Oslin, Hauser, Hauser and Beckham2022). SNPs within LEPR have been extensively studied and may impair receptor signalling by favoring the production of the truncated Ob-Ra isoform over the full-length Ob-Rb, or by decreasing receptor expression on the cell surface, thereby limiting leptin receptor interactions. One important SNP, rs1137101 (LEPR c.668A>G), has been shown to affect leptin receptor structure and function (Saad et al., Reference Saad, Adam, Elzaki, Awooda and Hamdan2020).
Leptin has also been extensively investigated in SCZ through genetic, epigenetic, and clinical perspectives. Several LEP gene polymorphisms (e.g., rs3828942 and c.-2548G/A) have been associated with an increased risk of metabolic syndrome, although their direct link to SCZ remains uncertain (Boiko et al., Reference Boiko, Pozhidaev, Paderina, Mednova, Goncharova, Fedorenko, Kornetova, Semke, Bokhan and Loonen2022). Epigenetic studies show increased DNA methylation in the LEP promoter and reduced leptin expression in patients, with methylation at specific CpG sites inversely correlated with positive symptom severity (Song et al., Reference Song, Chen, Zhao, Li, Li, Chen, Yu, Fu and Chen2022). Clinically, serum leptin levels are generally elevated in SCZ patients even after adjusting for body mass index (BMI) and medication use, correlating with metabolic syndrome; however, some subgroups exhibit lower leptin levels, indicating biological heterogeneity (Akan et al., Reference Akan, Ozgen, Colak, Acar, Oncu, Yesilbursa, Turkcan and Atalar2013; Stubbs et al., Reference Stubbs, Wang, Vancampfort and Miller2016; Petrikis et al., Reference Petrikis, Karampas, Leondaritis, Markozannes, Archimandriti, Spyrou, Georgiou, Skapinakis and Voulgari2022). Mendelian randomisation studies do not support a causal role for leptin levels in SCZ risk, but leptin/adiponectin ratios and correlations with metabolic markers highlight its biomarker potential (Chen et al., Reference Chen, Chen, Chiu, Lin, Li and Lu2018b, Reference Chen, Wang, Xue, Liu and Yu2021). Lower leptin levels have been linked to greater severity of suicidal behavior in SCZ spectrum disorders, and leptin receptor polymorphisms (e.g., rs1171276) have been associated with increased suicide risk in other psychiatric populations, such as depression, potentially through mechanisms involving leptin resistance and impulsivity (Acikel et al., Reference Acikel, Eroglu, Ugras Dikmen and Kurar2020b). While leptin and its receptor have been implicated in both SCZ and suicidal behavior through altered circulating levels, gene expression, and genetic variants, evidence on their specific role in suicide risk among SCZ patients is limited. To date, no studies have specifically examined leptin gene polymorphisms in relation to suicide attempts in SCZ.
Based on the existing evidence, we hypothesised that LEP (G-2548A) and LEPR (668A>G) polymorphisms may be involved in suicide attempt risk among patients with SCZ. Accordingly, the present study aimed to compare LEP and LEPR genotype distributions between SCZ patients and healthy controls, and to investigate the relationship between these polymorphisms and suicide attempts within the SCZ group.
Method
Patient selection
This case-control study included 120 patients diagnosed with SCZ who were consecutively recruited over a three-month period from the outpatient psychiatry clinic of Malazgirt State Hospital, along with 130 healthy control participants matched for age, sex, BMI, and ethnicity.
Inclusion criteria
For the patient group, inclusion criteria were a DSM-IV diagnosis of SCZ confirmed by the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I), age between 18 and 65 years, literacy, and absence of intellectual disability or neurodevelopmental disorders.
Exclusion criteria
Exclusion criteria for the patient group included refusal to participate, the presence of any Axis I psychiatric disorder other than SCZ according to SCID-I, SCZ secondary to substance use disorders or general medical conditions, and the presence of neurological or systemic diseases that could impair cognitive functioning.
The healthy control group consisted of volunteers recruited from hospital staff working at the same institution and within the same geographic region as the patient group. All control participants underwent a detailed clinical interview conducted by a psychiatrist to exclude current or lifetime psychiatric disorders according to DSM-IV criteria. Individuals with a personal history of suicide attempts, non-suicidal self-injury, substance use disorders, neurodevelopmental disorders, or major neurological or systemic medical conditions were excluded from the control group. In addition, participants with a first-degree family history of SCZ or other psychotic disorders were not included. These procedures were implemented to ensure that the control group represented a psychiatrically healthy population and to minimise potential confounding factors in genetic comparisons.
Diagnostic tools and scales
Participants were informed about the study’s aim, materials, and methods, and written informed consent was obtained. Sociodemographic data, clinical parameters related to SCZ, and information regarding suicide attempts, methods of suicide, type of suicide, and family history of suicide were collected. In addition, a detailed clinical information interview form prepared by the researchers was administered. Subsequently, the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) was conducted to confirm the patients’ diagnoses (First et al., Reference First, Gibbon, Spitzer and Williams1997, Çorapçıoğlu et al., Reference Çorapçıoğlu, Aydemir, Yıldız, Esen and Köroğlu Iv1999). For participants with SCZ, the Positive and Negative Syndrome Scale (PANSS) (Kay et al., Reference Kay, Fiszbein and Opler1987, Kostakoğlu et al., Reference Kostakoğlu, Batur, Tiryaki and Göğüş1999) and the Clinical Global Impression Scale (CGI) were used to assess symptom severity (Guy, Reference Guy1976).
DNA analyses
Genotyping of LEP -2548G/A (rs7799039) and LEPR 668A/G (rs1137101) single nucleotide polymorphisms (SNPs) was performed using the polymerase chain reaction–restriction fragment length polymorphism (PCR–RFLP) method. PCR amplification was carried out with the following primer sequences: forward 5’-TTTCCTGTAATTTTCCCGTGAG-3’ and reverse 5’-AAAGCAAAAGACAGGCATAAAAA-3’ for LEP -2548G/A, and forward 5’-GCCTAATCCAGTATTTTATATCTG-3’ and reverse 5’-GCCACTCTTAATACCCCCCAGTAC-3’ for LEPR 668A/G.
For the LEP -2548G>A polymorphism, genotyping was performed using the HhaI restriction enzyme, while the LEPR 668A>G polymorphism was analyzed with the MspI enzyme. Following agarose gel electrophoresis, the samples were visualised under UV light, and genotypes were determined. The fragment patterns were as follows: for LEP -2548G>A, GG: 242 bp; GA: 242, 181, and 61 bp; AA: 181 and 61 bp. For LEPR 668A>G, AA: 416 bp; AG: 416, 229, and 187 bp; GG: 229 and 187 bp (Aytac et al., Reference Aytac, Oyaci, Aytac, Pehlivan, Alptekin and Pehlivan2025).
Statistical analyses
Statistical analyses were performed using IBM SPSS Statistics for Windows, version 21.0. Descriptive data were presented as means ± standard deviations for continuous variables and as frequencies and percentages for categorical variables. Continuous variables were assessed for normality using visual inspection of histograms and Q–Q plots together with the Shapiro–Wilk test, and homogeneity of variances was evaluated using Levene’s test. Normally distributed continuous variables are presented as mean ± standard deviation and were compared between groups using independent samples t-tests; Welch’s t-test was applied when the assumption of homogeneity of variances was violated. Non-normally distributed continuous variables are presented as median (interquartile range) and were compared using the Mann–Whitney U test. Group comparisons for categorical variables were conducted using Pearson’s chi-square or Fisher’s exact test.
Hardy–Weinberg equilibrium (HWE) was tested using an online calculator (https://gene-calc.pl/hardy-weinberg-page), and all polymorphisms conformed to HWE (χ 2 test, p > 0.05) in both SCZ and control groups. Post hoc power analysis was conducted using G*Power version 3.0.5 (http://www.psycho.uni-duesseldorf.de/abteilungen/aap/gpower3/) with a goodness-of-fit χ 2 test, assuming an alpha error probability of 0.05. Potential population stratification bias was evaluated according to the method described by Lee and Wang. At the time of study design and sample collection, the minimum required sample size was calculated as 200 participants (100 per group) to achieve at least 80% statistical power at α = 0.05.
Demographic, clinical, and genetic variables associated with suicide attempt history were first examined using univariate logistic regression analyses, and variables with a p value <0.10 were subsequently entered into a multivariate logistic regression model to identify independent predictors, with results expressed as adjusted odds ratios and 95% confidence intervals. Model adequacy was assessed using standard goodness-of-fit indices, and multicollinearity was evaluated using variance inflation factors. All statistical tests were two-tailed, and a p value <0.05 was considered statistically significant.
Results
LEP and LEPR genotyping
A total of 120 patients with SCZ (33 females, 87 males) and 130 age- and sex-matched healthy controls were evaluated in terms of their clinical characteristics and psychometric scale scores (Tables 1 and 2). There was no significant difference in LEPR genotype distribution between patients with SCZ and healthy controls. In contrast, the LEP genotype distribution differed significantly between the two groups. Specifically, the frequency of the heterozygous GA genotype was significantly higher in the healthy control group compared to the SCZ group (OR: 1.767; 95% Cl: 1.068–2.925; p = 0.026; GA vs. AA + GG) (Table 3). Interaction analysis of LEP and LEPR gene genotypes revealed that none were associated with a significantly increased risk of SCZ (Table 4).
Clinical scale scores of patients with SCZ

SCZ, schizophrenia; PANSS, positive and negative syndrome scale; CGI-S, clinical global impression-severity; CGI-I, the clinical global impression-improvement; pos, positive; neg, negative; psycho, psychopathology.
Sociodemographic and clinical characteristics of patients with SCZ

Comparison of LEP (-2548G>A) and LEPR (668A>G) polymorphism frequencies between patients with SCZ and healthy controls

*; Pearson chi-square, &; Fisher’s Exact Test, SCZ; schizophrenia, OR; odds ratio, CI; confidence interval.
LEP (-2548G>A): Recessive, AA vs. GA+GG; Over-dominant, GA vs. AA + GG; Dominant, GG vs. AA+GA.
LEPR (668A>G): Dominant, AA vs. AG+GG; Over-dominant, AG vs. AA + GG; Recessive, GG vs. AA+AG.
Interaction analysis of LEP (-2548G>A) and LEPR (668A>G) polymorphisms in SCZ risk

SCZ; schizophrenia, OR; odd ratio, CI; confident interval, *; Pearson chi-square, &; Fisher’s exact test.
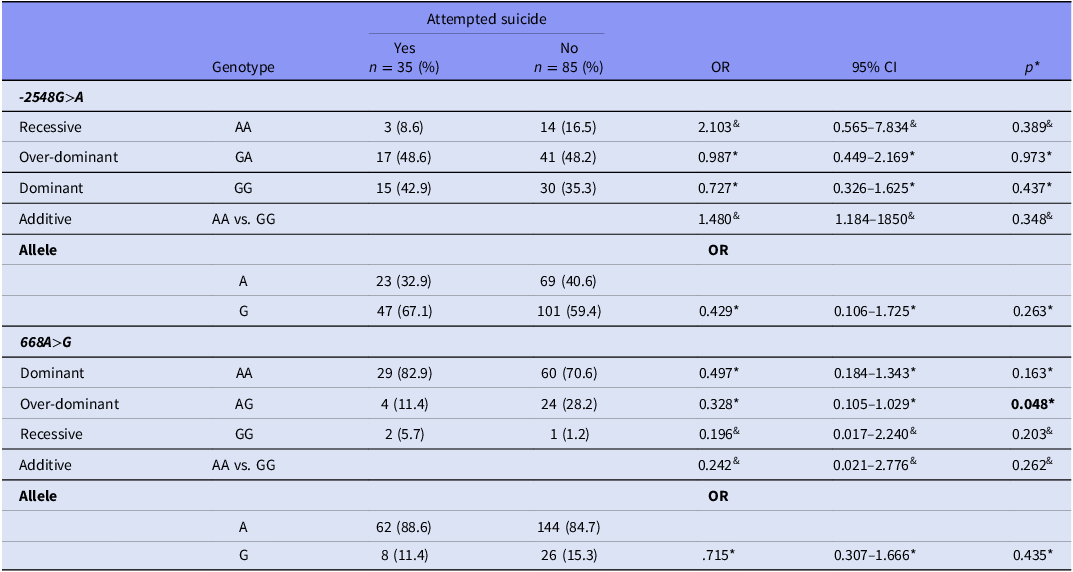
Comparison of LEP and LEPR genotype and allele frequency distributions in SCZ patients with and without a history of suicide attempt
Analysis of LEPR genotype distributions in SCZ patients, based on the presence or absence of a suicide attempt history, revealed significant differences between those with and without a history of suicide attempts. Specifically, the AG genotype frequencies were notably higher in the group without a history of suicide attempt (OR: 0.328; 95% CI: 0.105–1.029; p = 0.048; AG vs. AA + GG). However, LEP genotype and allele frequency distributions did not show statistically significant differences between those with and without a history of suicide attempt (p > 0.005) (Table 5).
Comparison of LEP (−2548G>A) and LEPR (668A>G) polymorphism frequencies between SCZ patients with and without a history of suicide attempt

*; Pearson chi-square, &; Fisher’s Exact Test, OR; odds ratio, CI; confidence interval.
LEP (-2548G>A): Recessive, AA vs. GA+GG; Over-dominant, GA vs. AA + GG; Dominant, GG vs. AA+GA.
LEPR (668A>G): Dominant, AA vs. AG+GG; Over-dominant, AG vs. AA + GG; Recessive, GG vs. AA+AG.
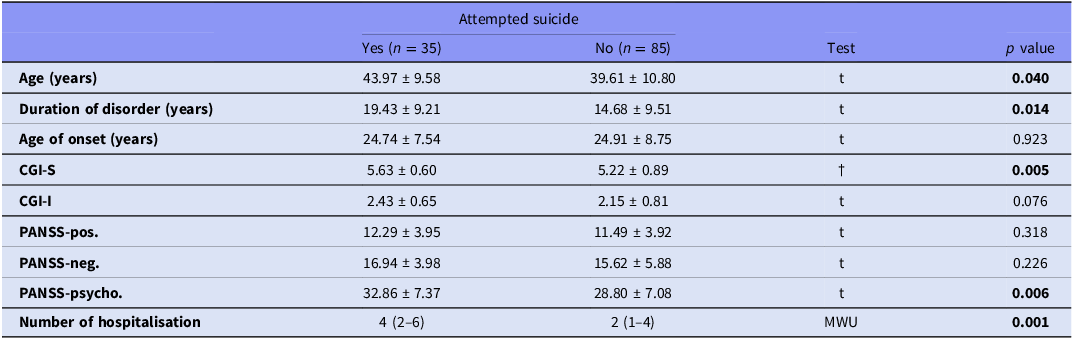
Comparison of demographic and clinical variables according to suicide attempt history
Demographic and clinical characteristics of patients with SCZ were compared according to the presence or absence of a history of suicide attempt (Table 6). Patients with a history of suicide attempt were significantly older than those without such a history (p = 0.040) and had a significantly longer duration of disorder (p = 0.014). No significant difference was observed between the groups with respect to age at illness onset (p = 0.923).
Comparison of demographic and clinical variables according to suicide attempt history

t, Independent samples t-test; MWU, Mann–Whitney U test; †, Welch’s t-test (t-test with unequal variances). PANSS, positive and negative syndrome scale; CGI-S, clinical global impression-severity; CGI-I, the clinical global impression-improvement; pos, positive; neg, negative; psyho, psychopathology.
Regarding clinical severity, patients with a history of suicide attempt exhibited significantly higher CGI–Severity scores compared with patients without a suicide attempt history (p = 0.005). However, CGI–Improvement scores did not differ significantly between the two groups (p = 0.076). In terms of psychopathology, PANSS-psychopathology scores were significantly higher in patients with a history of suicide attempt (p = 0.006). By contrast, PANSS positive (p = 0.318) and negative (p = 0.226) subscale scores did not differ significantly between the groups. Finally, patients with a history of suicide attempt had a significantly higher number of hospitalisations compared with those without a suicide attempt history (p = 0.001).
Multiple logistic regression analysis of factors associated with suicide attempt history in patients with SCZ
In the univariate logistic regression analysis, a history of suicide attempt was used as the dependent variable. All demographic, clinical, and genetic variables were initially evaluated in univariate analyses. Variables with a p value less than 0.1 in the univariate analysis were included in the multivariate logistic regression model: age (OR: 1.040; 95% CI: 1.001–1.081; p = 0.044), LEPR gene polymorphism (OR: 3.049; 95% CI: 0.972–9.566; p = 0.056), number of hospitalisations (OR: 1.120; 95% CI: 1.023–1.225; p = 0.014), duration of disorder (OR: 1.053; 95% CI: 1.009–1.098; p = 0.017), PANSS-psychopathology score (OR: 1.078; 95% CI: 1.019–1.139; p = 0.009), CGI-S score (OR: 1.898; 95% CI: 1.116–3.227; p = 0.018), and CGI-I score (OR: 1.595; 95% CI: 0.947–2.687; p = 0.079). In the multivariate logistic regression analysis, LEPR gene polymorphism (OR: 4.597; 95% CI: 1.232–17.152; p = 0.023), number of hospitalisations (OR: 1.105; 95% CI: 1.006–1.213; p = 0.036), and PANSS-psychopathology score (OR: 1.097; 95% CI: 1.013–1.188; p = 0.023) were identified as independent predictors of suicide attempt history. Results are presented in Table 7.
Univariate and multivariate logistic regression analyses for suicide attempt history in patients with SCZ

Initial –2 Log Likelihood: 144.874; Final –2 Log Likelihood: 120.332; Omnibus Test: χ 2 = 24.541, p = 0.001; Nagelkerke R 2 = 0.264; Hosmer–Lemeshow Test: χ2 = 4.498, p = 0.810. LEP, leptin gene; LEPR, leptin receptor gene; PANSS, positive and negative syndrome scale; CGI-S, clinical global impression-severity; CGI-I, the clinical global impression-improvement; pos, positive; neg, negative; psycho, psychopathology. * Variables with a p value < 0.10 in univariate analyses were included in the multivariate logistic regression model.
Discussion
Our findings revealed that the LEP genotype distribution differed significantly between SCZ patients and healthy individuals. Although the overall LEPR genotype distribution did not differ significantly between the SCZ and control groups, subgroup analyses based on suicide attempt history within the patient group demonstrated significant differences in LEPR genotype distribution. Although the relevant literature indicates that LEP and LEPR gene polymorphisms have been extensively investigated in SCZ – mainly in relation to their association with metabolic side effects rather than as direct risk factors for the disorder – there are very few studies that directly examine whether the LEP or LEPR polymorphisms are genetically implicated in patients diagnosed with SCZ. While Chen et al., found that drug-naïve SCZ patients exhibited some incidence of metabolic disorders, polymorphisms in the LEP (-2548G/A) and 5-HTR2C (-759C/T) genes were not associated with SCZ or metabolic disorders (Chen et al., Reference Chen, Tan, Long, Wang, Hu and Yang2018a). Akan et al., reported that the frequency of genotypes carrying the A allele of the LEP -2548G/A polymorphism (GA + AA), was two-fold lower in SCZ patients compared to controls, and no significant difference was observed in LEPR p.Q223R genotypes (Akan et al., Reference Akan, Ozgen, Colak, Acar, Oncu, Yesilbursa, Turkcan and Atalar2013). In our study, consistent with the findings of Akan et al., the frequency of the heterozygous GA genotype was significantly higher in the healthy control group compared to the SCZ group. We speculate that heterozygosity in the LEPR gene may confer a protective advantage against the development of SCZ.
A large meta-analysis reported that leptin levels are modestly to moderately elevated in individuals with SCZ compared to controls, particularly among females and those with multiple episodes of illness. This elevation remains significant even after adjusting for BMI and antipsychotic medication use, indicating that illness-related factors and lifestyle may also contribute (Stubbs et al., Reference Stubbs, Wang, Vancampfort and Miller2016). Numerous studies have confirmed increased leptin levels in both early and chronic phases of SCZ, as well as in patients treated with second-generation antipsychotics (Potvin et al., Reference Potvin, Zhornitsky and Stip2015; Martorell et al., Reference Martorell, Muntané, Porta-López, Moreno, Ortega, Montalvo, Sanchez-Gistau, Monseny, Labad and Vilella2019). However, some research has found no significant differences or even reduced fasting leptin levels in certain subgroups, reflecting variability likely due to differences in sample characteristics and study design (Nurjono et al., Reference Nurjono, Neelamekam and Lee2014; Erzin et al., Reference Erzin, Topcuoglu, Kotan, Bayram and Fountoulakis2018; Martorell et al., Reference Martorell, Muntané, Porta-López, Moreno, Ortega, Montalvo, Sanchez-Gistau, Monseny, Labad and Vilella2019). Considering that LEP and LEPR gene polymorphisms can influence leptin expression and receptor functionality, the higher frequency of the heterozygous GA genotype observed in healthy controls compared to patients with SCZ may reflect a genetic profile associated with relatively balanced leptin signalling. Such heterozygosity could plausibly help maintain leptin activity within an optimal physiological range, thereby avoiding both insufficient and excessive leptin signalling, which have been implicated in metabolic dysregulation, altered stress responsivity, and neurobiological vulnerability in SCZ.
Analysis of LEPR genotype distributions among SCZ patients, stratified by suicide attempt history, revealed significant differences between those with and without such a history. Furthermore, multivariate logistic regression identified LEPR gene polymorphism as a significant predictor of suicide attempts. Notably, the frequency of the AG genotype was significantly higher in SCZ patients without a history of suicide attempts. Currently, no studies directly investigate LEP or LEPR gene polymorphisms in SCZ patients with a history of suicide attempts. While one study reported that the LEPR rs1171276 polymorphism is associated with higher suicide probability scores in depressed adolescents, suggesting a potential link between leptin signalling and suicidal behavior in psychiatric populations, this association has not been specifically examined in SCZ or with the more commonly studied LEPR variants (Acikel et al., Reference Acikel, Eroglu, Dikmen and Kurar2020a). Research on suicide risk in SCZ has primarily focused on other genes, such as NR3C1 (glucocorticoid receptor), 5-HTTLPR (serotonin transporter), HTR2A, MIF, and MBL2, some of which have shown associations with suicide attempts (de Medeiros Alves et al., Reference De Medeiros Alves, Goncalves Bezerra, Gomes De Andrade, Leao De Melo Neto and E. Nardi2015; Bani-Fatemi et al., Reference Bani-Fatemi, Howe, Matmari, Koga, Zai, Strauss and De Luca2016; Aytac et al., Reference Aytac, Oyaci, Yazar, Erol and Pehlivan2020; Oyaci et al., Reference Oyaci, Yildirim, Aytac, Pehlivan and Aydin2024). These findings underscore the complex genetic landscape underlying suicide risk in SCZ.
A 2020 meta-analysis of nine case-control studies reported significantly lower serum leptin levels in individuals exhibiting suicidal behavior compared to controls, suggesting a potential association between low leptin and heightened suicide risk (González-Castro et al., Reference González-Castro, De Los Angeles Almeida De La, Tovilla-Zárate, López-Narváez, Mendoza, Juárez-Rojop, Pérez-Hernández and Rodríguez-Pérez2020). Several investigations involving psychiatric patients – including those with SCZ, major depressive disorder, and bipolar disorder – have found that suicide attempters exhibit lower serum or cerebrospinal fluid (CSF) leptin levels relative to healthy controls (Atmaca et al., Reference Atmaca, Kuloğlu, Tezcan, Ustundag, Geçici and Firidin2002, Reference Atmaca, Kuloglu, Tezcan and Ustundag2003, Reference Atmaca, Kuloglu, Tezcan and Ustundag2008). Notably, women with major depressive disorder who attempted suicide showed reduced CSF leptin compared to women with other diagnoses, implying a possible gender-specific effect (Westling et al., Reference Westling, Ahrén, Träskman-Bendz and Westrin2004). Additionally, decreased leptin levels were more pronounced in violent suicide attempters compared to non-violent attempters and controls (Atmaca et al., Reference Atmaca, Kuloglu, Tezcan and Ustundag2008). Conversely, some studies reported no significant differences in leptin levels between suicide attempters and non-attempters after adjusting for confounders such as BMI, suggesting that other metabolic or clinical factors may modulate this relationship (da Graça Cantarelli et al., Reference Da Graça Cantarelli, Nardin, Buffon, Eidt, Godoy, Fernandes and Gonçalves2015, Papadopoulou et al., Reference Papadopoulou, Douzenis, Christodoulou, Gournellis, Papageorgiou and Markianos2017).
The leptin receptor plays a crucial role in regulating energy balance and appetite through leptin signalling. The 668A>G polymorphism influences receptor functionality, potentially affecting leptin binding or signal transduction. Specifically, the G allele (coding for arginine) is linked to reduced receptor sensitivity or altered signalling, which may result in elevated circulating leptin levels due to compensatory responses to diminished receptor activity (Hryachkova et al., Reference Hryachkova, Khutornaya, Sinitskaya, Sinitskiy and Kashtalap2024). For example, in women with preeclampsia or pregnancy-induced hypertension, carriers of the G668 allele (either AG or GG genotypes) exhibit significantly higher leptin levels compared to normotensive controls, suggesting that this polymorphism can increase leptin in certain pathological conditions (Tennekoon et al., Reference Tennekoon, Indika, Sugathadasa, Karunanayake, Kumarasiri and Wijesundera2012). In our study, we observed a higher prevalence of the heterozygous AG genotype among non-suicidal SCZ patients, implying that this genotype may be less common in those with a history of suicide attempts. This pattern suggests that the homozygous AA or GG genotypes could be more frequent in suicidal patients. The heterozygous AG state might represent a balanced leptin signalling profile, avoiding the extremes of receptor dysfunction seen in GG-associated leptin resistance or AA-associated receptor overactivity, thereby potentially contributing to mood stabilisation or reduced impulsivity in SCZ patients.
In our study, the number of hospitalisations and the PANSS-psychopathology scores were identified as independent predictors of suicide attempt history, alongside LEPR gene polymorphism, in multivariate logistic regression analysis. This finding aligns with extensive existing research showing that both the frequency of psychiatric hospitalisations and elevated PANSS scores serve as independent predictors of suicide attempts in individuals with SCZ. Multiple large-scale meta-analyses and cohort studies have demonstrated that a higher number of hospitalisations reflects greater illness severity, treatment resistance, or recurrent psychiatric crises, all of which increase suicide risk (Cassidy et al., Reference Cassidy, Yang, Kapczinski and Passos2018; Zaheer et al., Reference Zaheer, Olfson, Mallia, Lam, De Oliveira, Rudoler, Carvalho, Jacob, Juda and Kurdyak2020; Mamtani et al., Reference Mamtani, Pathak, Sakhardande, Gowda, Muliyala, Philip, Moirangthem, Reddi and Varambally2024). Similarly, elevated PANSS general psychopathology scores–which encompass symptoms such as depression, anxiety, and cognitive impairment—have been consistently associated with an increased likelihood of suicide attempts (Cassidy et al., Reference Cassidy, Yang, Kapczinski and Passos2018; Pascariu et al., Reference Pascariu, Bosun, Kalinovic, Vlad and Enatescu2022, Fernández et al., Reference Fernández, Martínez-Cao, Couce-Sánchez, González-Blanco, Sáiz and García-Portilla2024). Taken together, our findings reinforce the critical role of clinical severity markers, alongside genetic factors like LEPR polymorphisms, in shaping suicide risk profiles in SCZ patients.
One of the key strengths of our study is its novel focus on investigating LEP (-2548G>A) and LEPR (668A>G) gene polymorphisms specifically in SCZ patients with a history of suicide attempts, comparing them to both SCZ patients without suicide attempts and healthy controls. This comprehensive comparison enhances the clinical relevance of our findings. Additionally, recruiting all participants from the same ethnic group and geographic region helped minimise environmental and population stratification biases, thereby strengthening the internal validity of the results. Although our study did not include epigenetic analyses, this homogeneity in sampling provides a robust genetic investigation framework. Furthermore, our use of peripheral blood samples facilitates less invasive and more feasible genetic testing, which is practical for clinical research.
Despite these strengths, there are important limitations to consider. The relatively small sample size may have limited the statistical power to detect subtle genetic associations, reducing the generalizability of the findings. The cross-sectional design restricts our ability to infer causality between LEP and LEPR polymorphisms and SCZ or suicide risk. Moreover, the lack of serum leptin measurements prevents direct correlation of genotypes with leptin levels, limiting functional interpretation. Another limitation is that our genetic analyses were conducted on peripheral blood samples rather than central nervous system tissues or CSF, which might more directly reflect neurobiological mechanisms underlying SCZ and suicidal behavior.
In summary, our findings suggest that the LEP gene polymorphism may contribute to susceptibility to SCZ in the Turkish population, whereas the LEPR gene polymorphism appears to be associated with suicide attempts among individuals with SCZ. Moreover, logistic regression analysis indicated that the LEPR polymorphism, number of hospitalisations, and PANSS-psychopathology score were significant predictors of suicide attempt history in patients with SCZ. From a clinical perspective, these results highlight the potential relevance of leptin pathway–related genetic variation for understanding biological heterogeneity in SCZ and for informing future suicide risk stratification approaches when integrated with established clinical indicators. Although these findings do not have immediate diagnostic or therapeutic implications, they provide valuable preliminary insights into the genetic factors underlying suicide risk in SCZ. Importantly, future research should aim to replicate these findings in larger and more diverse samples using longitudinal designs, and to integrate genetic data with biochemical, epigenetic, and central nervous system–derived measures, which will be essential for deepening our understanding and validating the observed associations.
Data availability statement
Data is available from the corresponding author upon reasonable request.
Acknowledgements
We want to thank all patients and healthy controls for their willingness to participate in this study.
Author contributions
HMA, EA, FBA, and SP were responsible for developing research goals and aims. HMA, SP, and HSB conceived and designed the study. SP, MP, and YO were responsible for obtaining laboratory samples. HMA, MP, HSB, FBA, and SP acquired, analyzed, and interpreted all data. EA and HMA prepared the manuscript. MP and SP supervised the work.
Funding statement
The authors did not accept any funding for this work.
Competing interests
All authors report that they have no conflicts of interest that might be interpreted as affecting the manuscript’s content.
Ethical standards
The authors affirm that all procedures contributing to this work comply with the ethical standards of the appropriate national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2013 (Association, Reference Association2013).