Introduction
Pertussis, commonly known as whooping cough, is an acute respiratory illness in humans caused by the Gram-negative bacterium Bordetella pertussis (B. pertussis) [Reference Cherry, Heininger, Feigin, Cherry, Demmler and Kaplan1]. The disease is characterized primarily by severe paroxysmal coughing, often accompanied by classic symptoms such as an inspiratory whoop and post-tussive vomiting [Reference Mikelova2]. Whooping cough is highly contagious, whose basic reproduction number (R0) is about 5.5 [Reference Kretzschmar, Teunis and Pebody3], nearly four times more than that of influenza (R0 = 1.28) [Reference Paules and Subbarao4]. In the pre-vaccine era, the vast majority of children were susceptible to pertussis. Although the incidence of pertussis declined dramatically following the widespread implementation of vaccination programmes in the mid-20th century [Reference Vitek5], the incidence of reported pertussis in many countries progressively increased in recent decades, which is called ‘recurrence of whooping cough’ [6, Reference Domenech de Cellès7]. At the same time, in recent years, several countries have reported the incidence of disease in adolescents and adults [Reference Skoff, Hadler and Hariri8, Reference Jõgi9]. Thus, the situation of whooping cough demands research attention.
The first isolation of B. pertussis was reported by Bordet and Gengou in 1906 [Reference Bordet10]. Since then, this microorganism has been widely studied by scholars. B. pertussis, a Gram-negative bacillus of the Bordetella genus, is a fastidious microorganism with specific and demanding nutritional requirements. In addition, B. pertussis grows slowly and often takes up to 5 to 7 days [Reference Young, Anderson and Mitchell11], limiting the timeliness for acute management and leading to difficulties in routine clinical testing. Antibiotics are believed to be most effective when administered early during the illness. Macrolides are the treatment of choice for B. pertussis [Reference Tiwari, Murphy and Moran12]. In the United States, erythromycin has been used for more than 65 years to treat patients [Reference Wilson13]. However, since the first report of erythromycin-resistant B. pertussis in Arizona in 1994 [14], more drug-resistant strains have been discovered worldwide, especially in China during recent years, which warrants attention. Currently, a few studies have summarized resistance data and minimum inhibitory concentration (MIC) on B. pertussis, and no associated meta-analysis was found. In this study, we aimed to review the data on the resistance of antimicrobials to B. pertussis and the MIC90 (90% minimum inhibitory concentration) of different kinds of antibiotics. These data serve as a guide for adjusting the antibiotic therapy for whooping cough.
Method
Search strategy and study selection
This systematic review and meta-analysis was conducted based on the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. The protocol of this systematic review has been registered in PROSPERO (ID: CRD42022325142). A comprehensive literature search was conducted across four electronic databases (PubMed, Embase, Cochrane Library, and Web of Science) from inception until December 2024, using the following search terms: (“Pertussis” OR “Pertusses” OR “Whooping Cough”) AND (“Antimicrobial-Drug Resistance” OR “drug resistance” OR “antibiotic resistance” OR “erythromycin” OR “beta-lactams” OR “Clarithromycin” OR “cephalosporins” OR “meropenem” OR “clindamycin” OR “tetracyclines” OR “quinolones” OR “macrolides” OR “carbapenems”) in all fields. No language restrictions were used in the search strategy. However, the criterion for inclusion was that studies must have an abstract available in English. All the records were merged and imported into EndNote X7 (Thomson Reuters, New York, NY, USA) to remove duplicates. Rayyan (https://www.rayyan.ai/) was used for screening and marking the records.
Data selection criteria and quality assessment
Study selection was performed independently by two reviewers (Keer Ma and Yao Chen). Studies were excluded for the following reasons: (1) absence or unclear reporting of antibiotic resistance data; (2) non-original research (e.g. reviews, meta-analyses, conference abstracts) or studies focussing on drug metabolism; (3) unavailability of full text; (4) sample size with fewer than five isolates or the use of non-human clinical strains; (5) duplication of previously reported data. Any inconsistencies with the selection of the article were resolved by a third author (Gujie Pan) based on the criteria above to reduce mistakes.
The adapted Newcastle–Ottawa Scale (Supplementary Table 1) was used by two independent authors (Keer Ma and Yao Chen) to assess the methodological quality of the included cross-sectional studies [Reference Modesti15, Reference Sholeh16]. Based on their scores across the domains of selection, comparability, and outcome assessment, studies were categorized as of high (≥5 points), moderate (3–4 points), or low (≤2 points) quality. A higher score indicated a higher study quality. Any cases with a disagreement were resolved through discussion. A third reviewer (Gujie Pan) adjudicated in any cases where there was a disagreement.
Data extraction
The following data were extracted from each included study: (1) first author; (2) publication year; (3) year of isolate collection; (4) study country; (5) total number of isolates; (6) antimicrobial susceptibility testing (AST) method; (7) interpretation of resistance; (8) number of resistant isolates; and (9) MIC90 values.
Definition of resistance
Neither the Clinical and Laboratory Standards Institute (CLSI) nor the European Committee on Antimicrobial Susceptibility Testing (EUCAST) has defined specific susceptibility and resistance breakpoints for B. pertussis. In accordance with relevant literature, an MIC of ≥32 mg/mL was used to define the resistance of erythromycin, azithromycin, clarithromycin, and clindamycin. Other antibiotics were evaluated for determining susceptibility by using the breakpoints specified by the 35th edition of the CLSI for Haemophilus influenzae.
Data analysis
The pooled resistance rate (PRR) for each antimicrobial was derived using a random-effects model to address anticipated heterogeneity. All statistical analyses were conducted with R software (version 4.0.3). Heterogeneity was quantified using the I2 statistic [Reference Machalek17]. The existence of publication bias was assessed by funnel plots and Egger’s test. We performed subgroup analyses to assess the influence of publication year, country, and AST method on the PRR. All statistical results are reported as point estimates with 95% confidence intervals (CIs).
Results
Search results
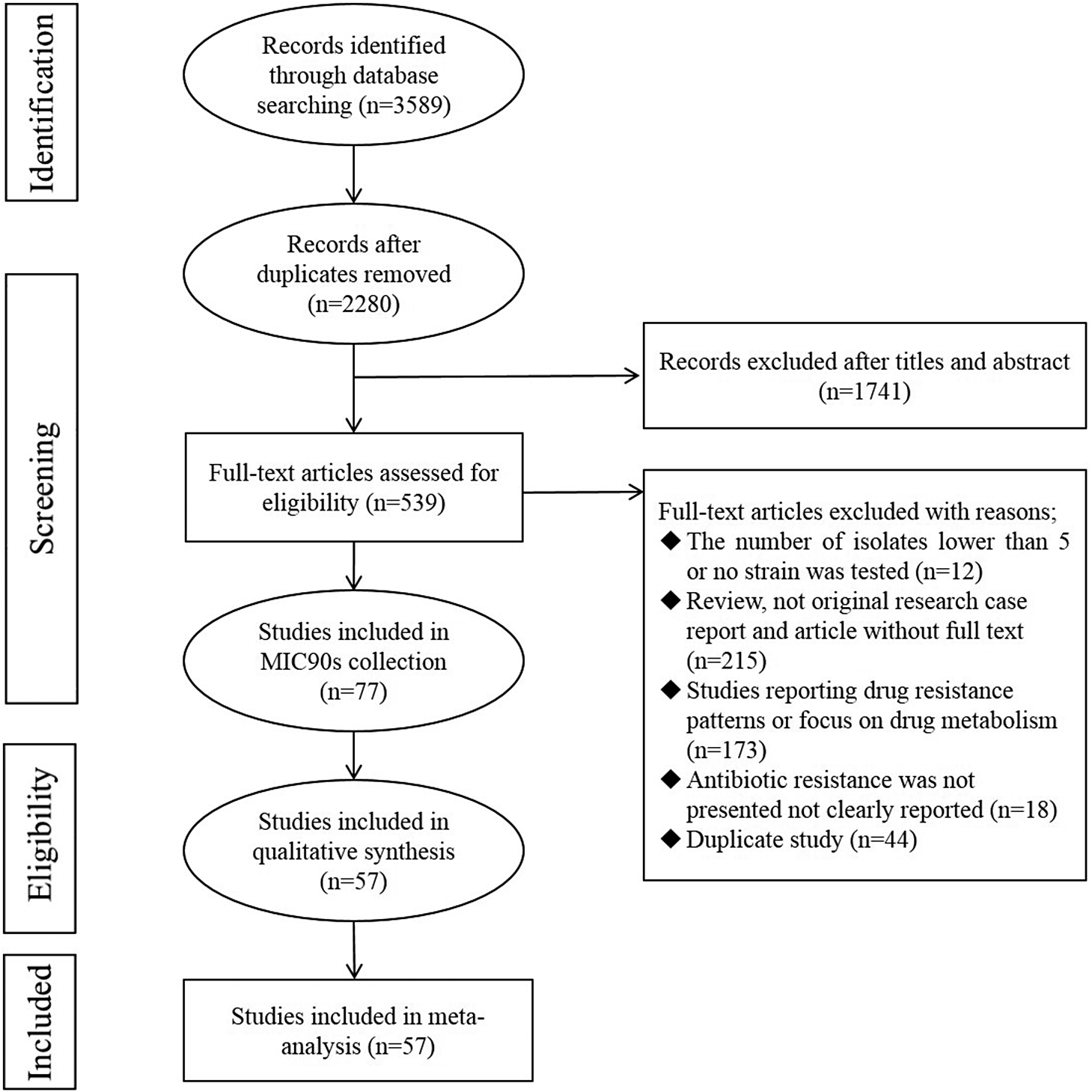
As shown in Figure 1, 3,589 articles were collected through four electronic databases published until December 2024. After removing duplication by using Endnote X7, 2,280 records remained. After an initial screening of the title and abstract, 1,741 articles were excluded due to their irrelevance, and the full texts of the remaining 539 articles were reviewed. At the end of the screening, 77 eligible articles were included in this review to extract MIC90 values, and 57 studies published from 1994 to 2024 were included in the meta-analysis according to the preset inclusion and exclusion criteria.
Flow diagram showing the study selection process.

Antimicrobial resistance analysis
Characteristics of the included studies
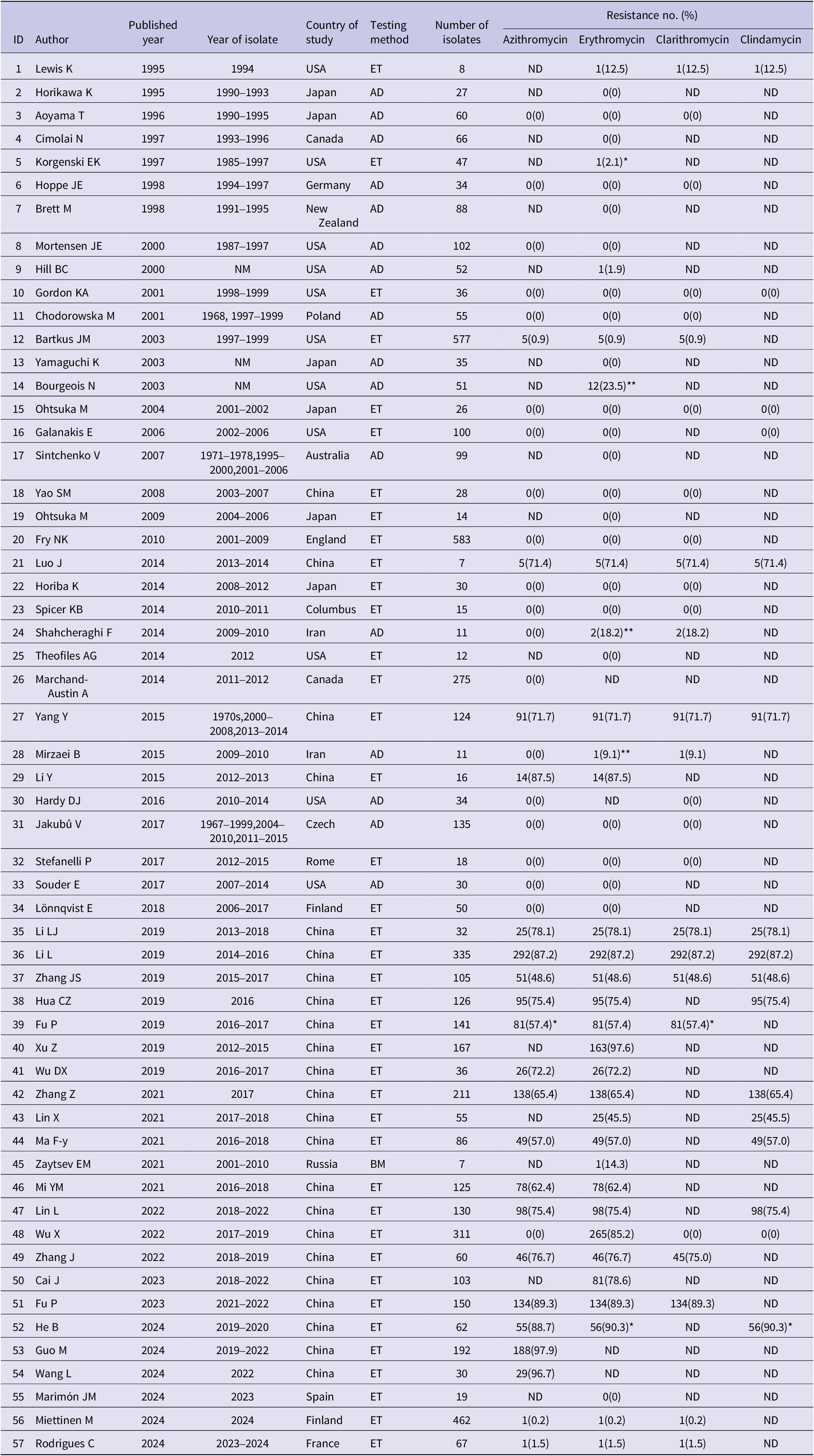
The 57 included studies describing the drug resistance of B. pertussis were selected from 16 countries covered 5,868 isolates. Most of the studies originated in China (n = 23), followed by the United States (n = 11). Epsilometer test (ET) (n = 40) was the most frequent AST method used, followed by agar dilution (AD, n = 16). All studies had a cross-sectional design, and the mean Newcastle–Ottawa score was 4.86. The quality was high in 36 (63.2%) studies and medium in 21 (36.8%) studies. All 57 studies in the meta-analysis reported resistance to macrolides (azithromycin, erythromycin, clarithromycin) and clindamycin (Table 1).
Extracted information from eligible studies included in the meta-analysis during 1994–2024

Note: For studies involving resistant strains, the breakpoint was set at 256 mg/L (unless specially noted).
*, breakpoint was 32 mg/L; **, breakpoint was 128 mg/L.
BM, broth dilution method; ND, not determined; AD, agar dilution; ET, Epsilometer test. Resistant No (%): number of resistant isolates (number of resistant isolates/number of isolates*100%).
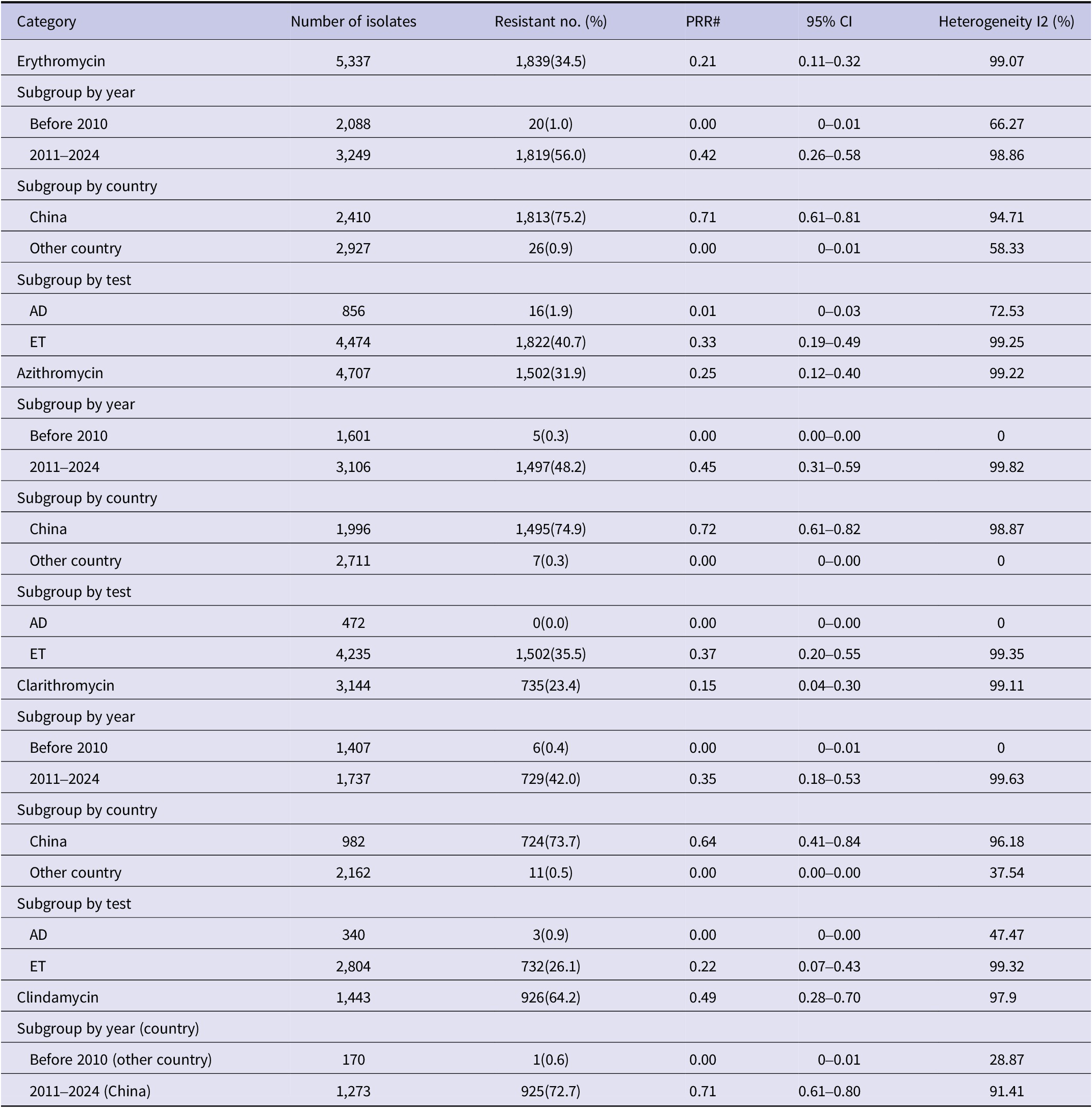
Based on the integrated data regarding B. pertussis drug resistance, the PRR for four antimicrobial agents are summarized in Table 2. Subgroup analyses based on year, country, and AST method are also provided. Forest plots illustrating resistance to each antimicrobial agent and subgroup comparisons are available in Supplementary Table 2.
Pooled resistance rates for azithromycin, erythromycin, clarithromycin, and clindamycin

Note: The resistance breakpoint was set at 32 mg/L for all four antibiotics.
#PRR, pooled resistance rate; CI, confidence interval; Resistant No. (%): number of resistant isolates (number of resistant isolates/number of isolates*100%).
Resistance to erythromycin
A total of 53 studies involving 5,337 B. pertussis isolates assessed erythromycin susceptibility. Among these, 1,839 isolates (34.5%) were reported as resistant. The PRR was 21% (95% CI 11–32%) with substantial heterogeneity (I2 = 99.07%), and Egger’s test indicated no publication bias (p = 0.95).
The subgroup analysis that compared the data from China (PRR 71%; 95% CI 61–81%) and other countries (PRR 0%; 95% CI 0–1%) indicated that macrolide-resistant B. pertussis strains are highly prevalent in China but rarely observed in other regions. This difference was statistically significant (p < 0.01). The groups differed in AST (p < 0.01), with higher rates in ET (33%, 95% CI 19–49%) and lower in AD (1%, 95% CI 0–3%). By analysing the data of 1994–2010 (PRR 0%; 95% CI 0–1%) and 2011–2024 (PRR 42%; 95% CI 26–58%), we found a significant increase in the resistance rate (p < 0.01).
Resistance to azithromycin
Susceptibility to azithromycin was determined in 40 studies where 4,707 B. pertussis isolates were tested. According to the statistics, 1,502 (31.9%) of tested isolates were considered resistant, and the PRR was 25% (95% CI 12–40%) with substantial heterogeneity (I2 = 99.22%). Egger’s test indicated no publication bias (p = 0.33).
The subgroup analysis based on the AST studies showed that the resistance rates differ between the groups (p < 0.01); the PRR was 37% (95% CI 20–55%) by ET and 0 (95% CI 0–0%) by AD. Interestingly, almost all studies from China reported azithromycin-resistant isolates, except one study [Reference Yao18]. The raw resistance rate was analysed, showing China with a PRR at 72% (95% CI 61–82%) and other countries with a PRR 0% (95% CI 0–0%). This difference was obviously statistically significant (p < 0.01). By analysing the data of 1994–2010 (PRR 0%; 95% CI 0–0%) and 2011–2024 (PRR 45%; 95% CI 31–59%), we found a significant increase in the resistance rate (p < 0.01).
Resistance to clarithromycin
Susceptibility of B. pertussis to clarithromycin was evaluated across 26 studies, encompassing a total of 3,144 isolates. Among these, 735 isolates (23.4%) exhibited resistance. The overall PRR was 15% (95% CI 4–30%), with a substantial heterogeneity (I2 = 99.11%). Egger’s test indicated no publication bias (p = 0.73).
The subgroup analysis found a significant difference (p < 0.01) between China (PRR 64%; 95% CI 41–84%) and other countries (PRR 0%; 95% CI 0–0%). Based on the AST of studies, the resistance rates also differed between groups (p < 0.01), exhibiting a higher PRR in the ET group (22%; 95% CI 7–43%) and a very low PRR in the AD group (0%; 95% CI 0–0%). A subgroup analysis, a comparison of 1994–2010 and 2011–2024 data, showed a significant difference (p < 0.01), 0% (95% CI 0–1%) versus 35% (95% CI 18–53%).
Resistance to clindamycin
Meanwhile, susceptibility to clindamycin was investigated in 15 studies. A total of 1,443 B. pertussis isolates were surveyed, and 926 isolates were reported as resistant (64.2%); the total PRR was 49% (95%CI 28–70%), with high heterogeneity (I2 = 97.9%). Egger’s test indicated publication bias (p = 0.04), which means long-term data monitoring was necessary. All studies employed the ET method. Interestingly, all the studies conducted in China were reported during 2011–2024 (PRR 71%; 95% CI 61–80%). Meanwhile, research from other countries was published between 1994 and 2010 (PRR 0%; 95% CI 0–1%).
Resistance to quinolone
As we know, there was only one study that reported the resistance to quinolone. Six B. pertussis strains isolated from 2004 to 2006 in Japan showed high-level resistance to nalidixic acid (NAL; MIC >256 mg/L).
MIC 90 of B. pertussis
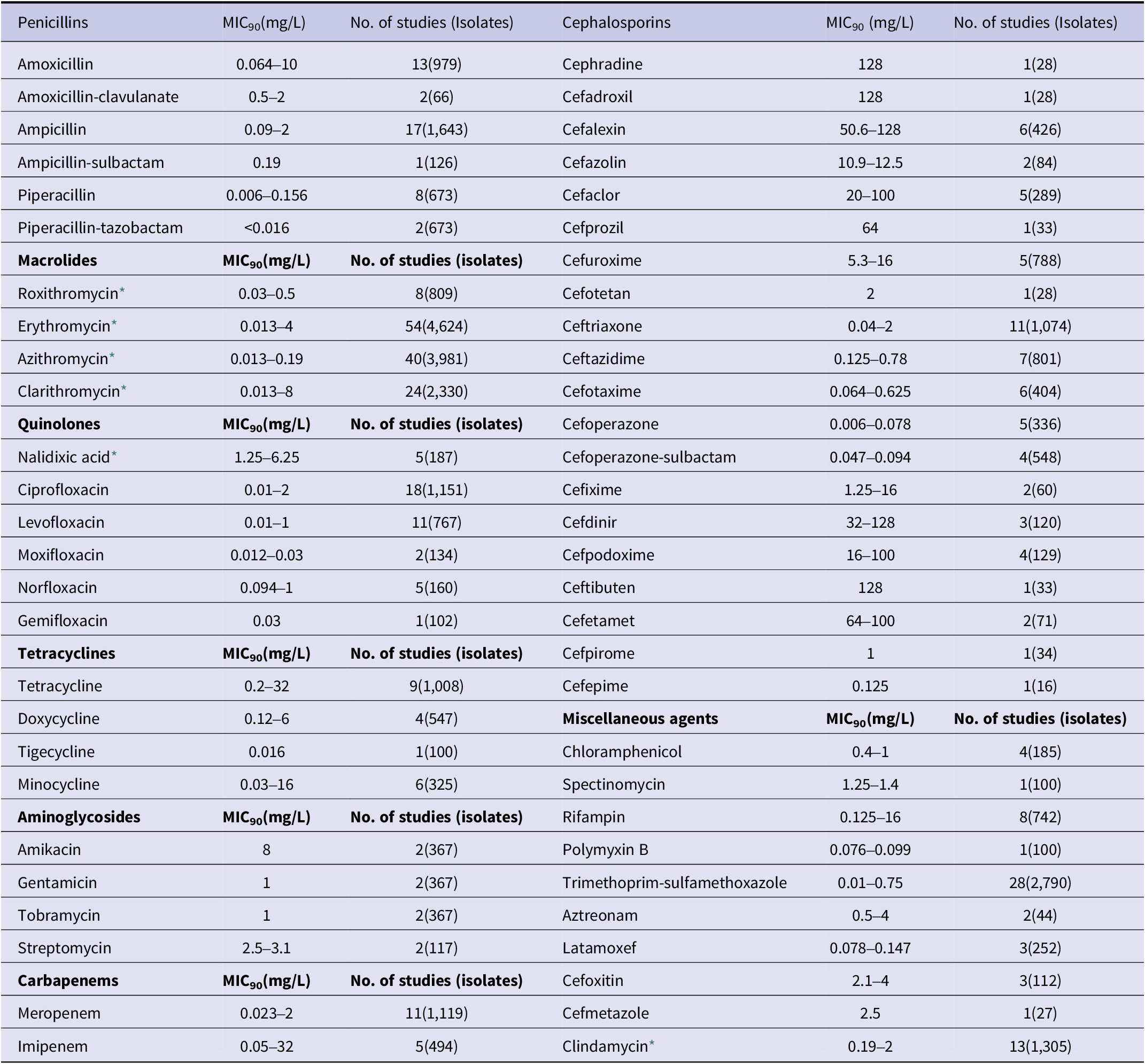
MIC90 values were collected from all 77 included studies [Reference Yao18–Reference Wang94]. Among these, 40 studies reported the MIC90 of azithromycin. With the exception of 18 studies that indicated an MIC90 of 256 mg/L, the remaining studies reported MIC90s ranging from 0.013 mg/L to 0.19 mg/L. For erythromycin, 54 studies provided MIC90 data; 17 of these reported an MIC90 of 256 mg/L, while the majority indicated a range of 0.013 mg/L to 4 mg/L. Twenty-four studies reported MIC90 values for clarithromycin, which ranged from 0.013 mg/L to 8 mg/L, and for clindamycin, which ranged from 0.19 mg/L to 2 mg/L, excluding reports involving resistant strains. The MIC90 values of several common antimicrobial agents – including penicillins, cephalosporins, quinolones, tetracyclines, aminoglycosides, and carbapenems – against B. pertussis are summarized in Table 3. The number of studies and the number of isolates for each antibiotic are also included.
Reported ranges of the minimum inhibitory concentration required to inhibit 90% of isolates (MIC90) of common antimicrobial agents against Bordetella pertussis

* In addition to the MIC90 ranges listed, values as high as 256 mg/L were reported in some studies due to a high prevalence of resistant isolates.
Discussion
The primary objective of this meta-analysis was to evaluate macrolide resistance rates in B. pertussis to assess the current therapeutic utility of this drug class. Due to its shared mechanism of action and significant cross-resistance with macrolides, clindamycin was also included in the analysis. To enhance the reliability of resistance data, case reports and studies involving fewer than five isolates were excluded, as smaller sample sizes are prone to increased bias in resistance rate estimation [Reference Sholeh16]. Due to the fastidious growth requirements of B. pertussis, AST is performed only in a limited number of tertiary hospitals, primarily for research purposes rather than routine diagnosis [Reference Zhang58, Reference Lin63]. During the actual statistical assessment, it was observed that the number of studies reporting resistant strains was limited, with only 57 studies meeting the inclusion criteria. Among these, a considerable proportion reported a resistance rate of 0%. Therefore, this meta-analysis includes high heterogeneity to some extent. The inclusion of negative results suggests a low risk of publication bias, a conclusion further supported by the results of Egger’s test (p > 0.05 in 3 out of 4 analyses).
Consequently, the macrolide resistance rate of B. pertussis in China was considerably higher than that reported in other countries, a difference that may be attributable to the extensive use of macrolides in China. Furthermore, macrolide-resistant B. pertussis has also been documented in several other countries, including the United States, Russia [Reference Xu80], Iran, France [Reference Guillot95], Japan [Reference Yamaguchi96], Vietnam [Reference Kamachi97], and Finland. The true prevalence of macrolide-resistant B. pertussis is likely underestimated, largely due to limitations in diagnostic practices. In China, bacterial culture remains a primary diagnostic method. Although culture is time-consuming and may yield false-negative results, it provides viable isolates essential for AST. In contrast, most other countries rely on polymerase chain reaction (PCR) or serology as gold-standard diagnostic techniques due to their high sensitivity and rapid turnaround time [Reference Feng98]. However, a significant drawback of these methods is their inability to provide antibiotic susceptibility profiles, greatly increasing the risk of undetected resistant strains. It is therefore reasonable to suggest – at least theoretically – that high macrolide resistance rates in B. pertussis may be overlooked outside of China due to these diagnostic limitations.
Overall, our results indicate that ET was the most commonly used method for AST, followed by AD. The AD method may be better suited for testing large numbers of isolates, given that fresh plates must be prepared for each experiment. In contrast, commercially available ET strips allow for individual testing of each isolate. It is worth noting that China, which accounts for the majority of reported macrolide-resistant B. pertussis strains, demonstrates a clear preference for the use of ET over AD in research settings. This preference may explain the statistically significant differences observed between the AD and ET groups in the subgroup analysis. The earliest study included in this review was published in 1967. However, the first documented case of erythromycin-resistant B. pertussis was reported in Arizona in 1994 [14], while macrolide-resistant strains were not identified in China until 2013 [Reference Zhang99]. To better understand this temporal trend, we performed a subgroup analysis by categorizing publication dates into periods before and after 2010. The results clearly indicate a marked increase in macrolide-resistant B. pertussis strains reported within the past decade.
In 2003, Bartkus and colleagues [Reference Bartkus19] identified the A2047G mutation in the 23S rRNA gene as a mechanism conferring erythromycin resistance in B. pertussis. Since then, the vast majority of erythromycin-resistant B. pertussis strains have been reported to carry either the A2047G or A2058G point mutation [Reference Yang34, Reference Li62–[Reference Lönnqvist64, Reference Xu80, Reference Kamachi97, Reference Zhang99]–Reference Wang104], with only a limited number of isolates lacking these mutations [Reference Yang34, Reference Mirzaei69]. However, the presence of a small number of isolates lacking these mutations suggests that additional, yet unidentified, resistance mechanisms may also exist. This phenomenon indicates that although homologous resistance is currently predominant in B. pertussis, continuous monitoring for the emergence of heteroresistance remains crucial, and in vitro AST of B. pertussis strains is still necessary. In 2009, Ohtsuka and colleagues [Reference Ohtsuka72] reported quinolone resistance in B. pertussis. Six clinical isolates from Japan exhibited high-level resistance to nalidixic acid (NAL; MIC>256 mg/L), which was attributed to a mutation in the quinolone resistance-determining region (QRDR) of the gyrA gene. To the best of our knowledge, no other quinolone-resistant strains have been reported elsewhere. Nevertheless, continued surveillance is essential to monitor for the emergence of additional mutations that may confer further antibiotic resistance.
The CLSI and the EUCAST have not yet established official breakpoints for AST of B. pertussis. Therefore, to evaluate the exposure of B. pertussis to various antibiotics, we have summarized the MIC90 values of commonly tested antibiotics reported in the 77 included studies. This compilation provides a valuable reference for future research.
Since B. pertussis is difficult to culture and testing for the bacteria by any strategy must be specifically requested, treatment for serious respiratory illness is empirical. Macrolide antibiotics, particularly azithromycin, are frequently endorsed for respiratory tract infections and behave actively against B. pertussis, according to previous experiments in the 20th century. Azithromycin remains one of the recommended drugs for the treatment and prophylaxis of pertussis, yet a substitute medication is required, especially in China, due to the high resistance rate. In fact, macrolide-resistant isolates have been shown to have a worse clearance rate at 7 and 14 days post-treatment [Reference Mi68]. For paediatric patients who have failed macrolide therapy, some studies recommend oral sulfamethoxazole (STX) [Reference Fu45, Reference Lin63]. STX, a second-line agent for pertussis treatment, demonstrated low MIC90 values in this review. No resistant B. pertussis isolates have been reported to date. However, STX is not recommended for neonates or infants under 2 months of age.
In addition, penicillins and cephalosporins are good alternatives for children. However, it should be noted that B. pertussis, as a Gram-negative bacterium, exhibits inherently low susceptibility to first- and second-generation cephalosporins. Actually, cephalexin was used as the inhibitory agent in charcoal horse blood agar. The data on the in vitro activities of the new oral cephalosporins must be interpreted as comparable to the concentrations achievable in respiratory secretions. As shown in Table 3, cephalosporins, including cefixime, cefdinir, cefpodoxime, ceftibuten, and cefetamet, all exhibited high MIC values against B. pertussis, suggesting that oral cephalosporins may have limited efficacy in the treatment of pertussis. Based on the breakpoints specified by the 35th edition of the CLSI for H. influenzae, the MIC90 values of penicillins (including amoxicillin-clavulanate, ampicillin-sulbactam, piperacillin, and piperacillin-tazobactam) and intravenous third-generation cephalosporins (such as ceftriaxone, ceftazidime, and cefotaxime) described in Table 3 suggest potential susceptibility of B. pertussis to these agents.
In recent years, the incidence of pertussis in adults has also become a notable concern. Therefore, it is necessary to investigate the susceptibility of B. pertussis to a broader range of antibiotics in order to guide clinical treatment. Quinolones are currently recommended for the empirical treatment of respiratory tract infections in adult patients [Reference Hoppe, Rahimi-Galougahi and Seibert28]. Multiple studies have indicated that certain quinolones exhibit low MIC values against B. pertussis, and their concentrations in respiratory secretions are sufficient to exceed the MIC values for the pathogen [Reference Hoppe, Dalhoff and Pfründer25]. Nakamori and colleagues reported that the sputum concentration of levofloxacin reaches 1.27–4.36 mg/L [Reference Nakamori105], suggesting its potential in vivo efficacy against pertussis. Theoretically, quinolones with enhanced antibacterial activity may play an important role in the treatment of pertussis in adults.
The treatment of pertussis in adults appears to be relatively straightforward, as indicated by the low MIC values of numerous antimicrobial agents shown in Table 3. Given that pertussis infection in adults is generally mild, quinolones, SXT, and certain β-lactam antibiotics are considered sufficient for treatment. Other agents were not analysed in this study.
Conclusion
In summary, there has been a significant increase in macrolide-resistant B. pertussis in recent years. China has been particularly affected by this trend, underscoring the need to reevaluate the clinical utility of macrolides in this region and implement enhanced antimicrobial resistance surveillance. In contrast, macrolide resistance remains rare in B. pertussis isolates from other countries; however, this may reflect underreporting rather than true susceptibility, emphasizing the continued importance of routine monitoring globally. To date, only six B. pertussis strains exhibiting high-level resistance to nalidixic acid have been documented. Nevertheless, ongoing vigilance is essential to detect emerging mutations that may confer additional drug resistance.
Additionally, based on the aggregated data, STX, penicillins (including amoxicillin-clavulanate, ampicillin-sulbactam, piperacillin, and piperacillin-tazobactam), and intravenous third-generation cephalosporins (such as ceftriaxone, ceftazidime, cefotaxime, and cefoperazone-sulbactam) demonstrate strong in vitro activity against B. pertussis, suggesting their potential as alternative treatments for pertussis in children. For adults, quinolones currently remain effective.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0950268826101010.
Data availability statement
This systematic review and meta-analysis is based on previously published studies; therefore, no new primary data were generated. All data supporting the findings of this meta-analysis are available within the article and its Supplementary Information files. Specifically:
-
• Supplementary Table 1 lists all original studies included in the meta-analysis as well as the quality assessment tables.
-
• Supplementary Table 2 contains the forest plots illustrating resistance to each antimicrobial agent and the subgroup comparisons.
-
• The study protocol is available on PROSPERO (ID: CRD42022325142).
No original code was used beyond the standard functions of the R software (version 4.0.3).
Acknowledgements
We are grateful for the useful comments and suggestions from the anonymous referees. We also thank Huali Zuo from the Fourth Academy of Zhejiang University for her assistance in polishing the language of the manuscript.
During the preparation of this work, the authors used the AI tool DeepSeek (developed by Deep Seek Company) solely for the purpose of linguistic refinement and sentence polishing. After using this tool, the authors reviewed and edited all content critically and assume full responsibility for the entire publication.
Author contribution
Conceptualization: K.M.; Data Curation: W.S., Y.X., C.Y.; Formal Analysis: K.M., C.Y.; Investigation: K.M., W.S., C.Y.; Methodology: K.M.; Project Administration: G.P.; Software: K.M.; Supervision: G.P.; Validation: W.S.; Visualization: W.S., S.Y.; Writing – Original Draft: K.M.; Writing – Review and Editing: Y.X., S.Y., C.Y.
Funding statement
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
The authors declare none.