Key messages
Chapter 3.1 draws out insights into the way primary care is paid. Primary care delivers first-contact access to health services and seeks to coordinate care. It is paid for in different ways in different settings: through salary, capitation (a fixed payment per enlisted patient), fee-for-service (FFS), pay for performance (P4P) or through blended payments that combine two or more of these methods. Key learning includes that:
The way health systems pay for primary care can incentivize treatment that supports wider health systems’ goals, such as:
○ reducing avoidable referrals to secondary care, improving efficiency and reducing waste; and
○ encouraging adherence to evidence-based clinical guidelines, enhancing quality.
P4P is often used to improve quality but the evidence on how effective it is, is mixed. If performance measures are not carefully designed to be context specific and adjusted for risk appropriately, they can create unintended barriers to and inequalities in access.
The design of primary care payment models needs to be “holistic”, to consider the goals of the health system and to underpin quality, access and efficiency.
Introduction
Payment models are increasingly being used by governments and health insurers to support and help drive efficient and equitable health care systems. This chapter examines the evidence on how payment methods in primary care can help solve global policy challenges faced by the primary health care sector.
The primary health care sector is usually the first point of contact for patients in need of health services. The sector is usually, though not exclusively, organized around primary care physicians (PCPs) (also referred to as general practitioners (GPs)), who can be supported by nurses, allied health professionals and other community health workers. However, there are also examples of other health professionals providing some primary care services, such as community pharmacists. Given the need for easily accessible health care by the population, primary care is usually geographically dispersed and organized into small community-based medical groups or health centres – primary care organizations (PCOs). In some countries, the population enrols with a PCP, PCO or health insurer, where continuity of care reflects a key defining element of primary care.
The organization of primary care across countries is varied. PCOs can be owned and run by local or national governments, or by vertically integrated health insurers, or can be privately owned by PCPs or private companies. In most health care systems with some form of health insurance, providers or their patients will receive payments and/or subsidies from insurers (including governments) for the care provided. Payments can be made to an individual PCP and/or to the PCO, who then distributes the payments to health professionals within that group. For example, the PCO could be paid on the basis of capitation, while the PCP is paid a fixed salary.
The aim of this chapter is to describe how different primary care payment models can be used to help solve two common global policy challenges relevant to the primary health care sector: (i) how to reduce avoidable secondary care; and (ii) how to improve the quality of primary care. These two policy challenges were selected because they contribute to achieving the overall goals of payers (defined as third-party insurers, such as health insurers and governments, as well as patients), they are relevant to primary care, and they are common issues faced by many health care systems. Primary care payment models provide financial incentives that can be used to solve these policy challenges. Different payment models may help solve different challenges, which explains why payment models are often blended.
Using payment models to help solve policy challenges assumes that income plays a role in motivating the clinical and labour market behaviour of PCPs. This assumption generally holds, but nonfinancial motives may also influence their behaviour, presumably to a greater extent if their target income is reached. To understand PCPs’ behaviour, the literature often uses the principal–agent framework (Scott, Reference Scott, Culyer and Newhouse2000). This framework assumes that PCPs (agents) seek to maximize their own utility, which depends on their income but also their altruistic concerns towards the patient (principal) and own leisure time (Ellis & McGuire, Reference Ellis and McGuire1986; Dionne & Contandriopoulos, Reference Dionne and Contandriopoulos1985). Some studies also include altruistic concerns towards a third-party insurer or society at large to capture PCPs’ double-agency role (e.g. Allen et al., Reference Allen2022; Blomqvist, Reference Blomqvist1991; Oxholm et al., Reference Oxholm2024). PCPs may also be motivated by other nonfinancial drivers such as their reputation among colleagues and patients and intrinsic motivation (enjoyment of tasks) (Pedersen et al., Reference Pedersen2018; Sicsic, Le Vaillant & Franc, Reference Sicsic, Le Vaillant and Franc2012; Pedersen et al., Reference Pedersen2021; Godager, Hennig-Schmidt & Iverson, Reference Godager, Hennig-Schmidt and Iversen2016; Bénabou & Tirole, Reference Bénabou and Tirole2006; Yordanov et al., Reference Yordanov2023). As PCPs may be motivated to different degrees by these factors, responses to payment models are thus often heterogeneous across providers (Hennig-Schmidt, Selten & Wiesen, Reference Hennig-Schmidt, Selten and Wiesen2011; Godager & Wiesen, Reference Godager and Wiesen2013).
PCPs may also respond differently to the same payment models depending on their context and setting. For example, any payment model can be accompanied by global budget mechanisms that provide overall caps to spending. PCPs could also receive income from many different payers (e.g. insurers) each with a different method of payment, such that the expected behavioural effects when one payer changes its payment model are muted. Availability of resources (Di Guida et al., Reference Di Guida, Gyrd‐Hansen and Oxholm2019; Oxholm et al., Reference Oxholm2019; Oxholm, Guida & Gyrd-Hansen, Reference Oxholm and Gyrd-Hansen2021) and the degree of competition among providers (Brosig‐Koch, Hehenkamp & Kokot, Reference Brosig‐Koch, Hehenkamp and Kokot2017; Iversen, Reference Iversen and Scheffler2016; Scott & Sivey, Reference Scott and Sivey2022) could also affect providers’ responses to changes in the level or type of payments. Payment methods could also influence the mix and characteristics of PCPs and PCOs through selection. This includes the entry and exit decisions of PCPs and PCOs, PCP hours of work, as well as the location decisions of PCPs and PCOs, such as practising in a rural or urban area or in an area with a high-need population (Li et al., Reference Li2014; Günther et al., Reference Günther2010; Pedersen & Gyrd-Hansen, Reference Pedersen and Gyrd-Hansen2014; Anell, Dackehag & Dietrichson, Reference Anell, Dackehag and Dietrichson2018).
The next section outlines how the basic types of primary care payment models affect PCPs’ behaviour and their ability to reach payers’ different goals. Afterwards, for each of the two policy challenges introduced above, we examine its context and then the extent to which primary care payment models could help solve the challenge.
Overview of primary care payment models and their effects
First, we summarize, from the payers’ perspective, the advantages and disadvantages of using the most common payment methods for PCPs. Any payment model involves an explicit or implicit contract between a payer and a provider. These contracts specify the unit of payment. The unit of payment has behavioural consequences as it defines the behaviour the payer is expecting in exchange for the payment. For example, salary payment is based on working a specified number of hours; capitation payment is paid per enrolled patient and so is related to the list size; FFS is related to the volume of services provided, usually defined in terms of consultations/visits, treatments or diagnostic tests; and P4P is based on a range of different quality measures that could be related to clinical guidelines or health outcomes (Quinn, Reference Quinn2015; Jia et al., Reference Jia2021; Stokes et al., Reference Stokes2018). These different payment models can also be blended together.
Even though it may be the patient and/or insurer that pays for the care, the financial risk of treating patients may be shared between the payers and the providers. Who bears this risk depends on the unit of payment. For salary and capitation payments, payers can relatively easily predict total expenditures and so face little financial risk themselves. Instead, this risk is borne by the providers who are not compensated for changes in demand and workload, for example, increases in the numbers of patients with complex conditions. Capitation payment can partly account for financial risk through risk adjustment of payments that take into account the additional costs of treating more complex patients. For FFS and P4P, payers typically bear a higher financial risk as providers are compensated for additional workload. Providers’ financial risk can significantly influence the strength of their incentive to provide care themselves rather than refer to other health professionals. More specifically, a lower financial risk, such as under FFS and P4P, increases providers’ financial incentives to provide care themselves. These types of payments may also be used to incentivize providers to deliver different types of care through different price signals (Hsiao et al., Reference Hsiao1992).
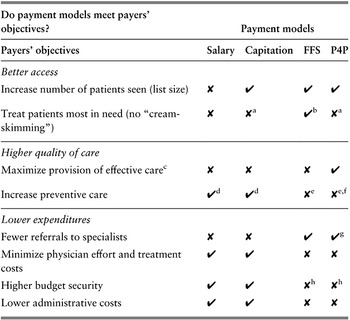
PCPs and PCOs may often receive remuneration from a blend of different payment methods to achieve the different goals of the payer. Table 3.1.1 shows whether these payment methods provide financial incentives to help achieve common high-priority payer goals. The following discussion of each payment method goes through the table in more detail. Box 3.1.2 briefly describes payment methods that are increasingly being used for community pharmacists as their role in the provision of first-level care expands to some primary care services in certain settings.

Table 3.1.1 Long description
The table has 2 main columns (with further subcolumns): Payers’ objectives and Payment models (Salary, Capitation, F F S, and P 4 P). It reads as follows. Under Better access. Increase number of patients seen (list size): cross; tick; tick; tick. Treat patients most in need (no cream skimming): cross; cross a; tick b; cross a.
Under Higher quality of care. Maximize provision of effective care c: cross; cross; cross; tick. Increase preventive care: tick d; tick d; cross e; cross e, f.
Under Lower expenditures. Fewer referrals to specialists: cross; cross; tick; tick g. Minimize physician effort and treatment costs: tick; tick; cross; cross. Higher budget security: tick; tick; cross h; cross h. Lower administrative costs: tick; tick; cross; cross.
FFS: fee-for-service; P4P: pay for performance.
a If payment is risk adjusted there will be a financial incentive to enlist high-need patients which also provides a signal from the payer to prioritize these patients’ treatment.
b Unless fees are paid to PCPs by patients (in whole or in part), which may reduce demand among those in most need, compared to fees being paid to PCPs directly by third-party insurers. It also assumes that fees are sufficiently differentiated to reflect the costs of treating different types of patients.
c Depends on adherence to clinical guidelines.
d Depends on the difference between PCPs’ costs of prevention and otherwise expected increase in PCPs’ future workload and costs of care.
e Unless payments are made for preventive activities such as immunization.
f Depends on whether prevention impacts reported performance.
g Depends on whether performance links to PCPs providing the services themselves or rewards “appropriate” referrals.
h Budget security may be higher if there is a cap on total payments and fees are set (regulated) by the payer.
Payment method 1: salary
Salaried providers receive a fixed revenue for working over a given time period. This payment method is typically used to pay providers who are employees of a PCO. Payers may find the fixed salary payment scheme attractive for several reasons. One is that a fixed salary ensures budget security for the payer because it shifts financial risk to providers. Another reason is that the payment method is easy to administer as it only requires information about the employed providers. A fixed salary, however, also has some unattractive features. As providers are not paid for their activities, they are (in the short run) not financially incentivized to exert more than the minimum required effort to avert being laid off. Generally, PCPs may provide less effort by, for example, avoiding costly patients, reducing the frequency of services, or referring patients to other providers. In the longer run, the prospect of receiving a promotion and pay rise as well as the fear of being laid off may financially incentivize them to increase effort beyond the minimum required level. The focus for the provider then becomes the criteria used by employers to enable progression up the career ladder. These criteria can create career incentives that are related to objectively or subjectively assessed performance (Prendergast, Reference Prendergast1999).
Payment method 2: capitation
Providers paid by capitation receive a fixed payment per enlisted patient for a given time period. The payment is independent of the amount of provided care. However, it may be risk adjusted such that patients with characteristics that indicate they are more costly to treat are linked to a higher payment. As the size of the served patient population is typically stable due to patient enrolment with a PCO, this payment method gives payers budget security by shifting financial risk to providers. Capitation payments financially incentivize providers to attract and keep patients on their lists when facing competition, thereby potentially increasing access to care and also improving those aspects of quality that patients can observe. However, if competition for patients is low, providers have less of a financial incentive to exert effort above the minimum requirement. An exception to this lack of financial incentive may be preventive care that reduces providers’ required effort in the longer run.
As with any fixed payment, capitation payments incentivize the providers to minimize their costs of providing care, which may lead them to provide less care than is necessary when quality is difficult to observe (Hennig-Schmidt, Selten & Wiesen, Reference Hennig-Schmidt, Selten and Wiesen2011; Brosig-Koch et al., Reference Brosig-Koch2016; Oxholm et al., Reference Oxholm2019). Capitation may also lead providers to only enlist patients whom they expect to be less costly to treat compared to the capitation payment itself (“cream-skimming” behaviour). This behaviour may increase inequality in patients’ access to care. Such selection issues may be alleviated by regulating patients’ access to providers’ lists or by risk-adjusting the payments based on patients’ expected treatment costs. Risk adjustment shifts some of the financial risk away from the provider to the payer and thereby weakens the providers’ incentives to reduce costs by exerting cream-skimming behaviour. The risk adjustment may also send a signal to providers about patient prioritization (Oxholm et al., Reference Oxholm2019), but there is no direct financial incentive to prioritize already enlisted high-need patients.
Payment method 3: FFS
PCPs can receive a fee for each service they provide. This payment method requires providers to report and bill for the number of activities (e.g. consultations, visits, procedures, episodes). The advantage of FFS over salary and capitation is that providers have a financial incentive to deliver care themselves rather than referring patients to other providers (Hennig-Schmidt, Selten & Wiesen, Reference Hennig-Schmidt, Selten and Wiesen2011; Krasnik et al., Reference Krasnik1990; Brosig-Koch et al., Reference Brosig-Koch2016). A disadvantage of FFS is that providers may be reluctant to provide services that are not covered by the fee schedule, unless some services are bundled together. Another concern for the payer is that they bear most of the financial risks, especially when fees are unregulated. Consequently, FFS increases the risk of overtreatment and supplier-induced demand (Di Guida, Gyrd-Hansen & Oxholm, Reference Oxholm2019; Longden, Hall & Van Gool, Reference Longden, Hall and Van Gool2018), gaming of the system and budget deficits for payers. To minimize these risks some payers may choose to closely monitor service patterns through utilization reviews or to set caps on the payments to providers through controls on fees that can be charged (Wickizer & Lessler, Reference Wickizer and Lessler2002). The fees may be set by either the payers or the PCPs themselves or through negotiations. In cases where PCPs set the fees, they may choose to price discriminate, i.e. charge payers different fees based on patient characteristics. In cases where the payers are the patients themselves, the fee is an out-of-pocket (OOP) cost, and higher fees may reduce demand and thereby have implications for equity in access to care.
Payment method 4: P4P
P4P has become a popular way of remunerating PCPs (Cashin et al., Reference Cashin2014). This payment method links payments directly to performance measures. The performance measures may be based either on completion of processes (e.g. recording blood sugar levels) or on achieving specific intermediate health outcomes for patients (e.g. whether blood sugar levels are in accordance with clinical guidelines). The schemes require an administrative system that measures providers’ performance (which may rely on digital health infrastructure (see Box 3.1.1)). Depending on the performance measures in question, such administrative requirements may be costly. An advantage of P4P is, however, that providers receive feedback on their performance, which may improve the quality of care over and above the effect of the financial incentives (Kolstad, Reference Kolstad2013; Godager, Hennig-Schmidt & Iverson, Reference Godager, Hennig-Schmidt and Iversen2016).
P4P may be designed in different ways, which creates different incentives for the provider (as discussed later in the chapter). These design differences may explain why evidence on the impact of P4P on the provision of care is mixed. The mixed evidence, however, may also be a result of weak evaluation designs of existing studies (see, for example, reviews by Van Herck et al., Reference Van Herck2010; Jia et al., Reference Jia2021; Eijkenaar et al., Reference Eijkenaar2013; Diaconu et al., Reference Diaconu2021; Zaresani & Scott, Reference Zaresani2021; Scott, Liu & Yong, Reference Scott, Liu and Yong2018). Despite the different designs of P4P schemes, they also share several common features that make them attractive to payers. P4P schemes (as opposed to FFS schemes) incentivize providers to only deliver care defined as high quality. For example, by rewarding providers for only one blood sugar level measurement per diabetes patient per time period, as opposed to a FFS scheme rewarding providers for all such measurements independently of the number of previous measurements and the recipient. This focus on what constitutes high-quality care may also reduce heterogeneity in care across providers.
A disadvantage of P4P is that payers may find it difficult to predict providers’ performance and thereby risk facing overrun budgets. Payers may also end up paying for performance that is only weakly related to the individual provider’s efforts. Providers may also have financial incentives to treat patients who are close to meeting or have already met the performance requirements (cream-skimming behaviour), thus minimizing provider effort (Gravelle, Sutton & Ma, Reference Gravelle, Sutton and Ma2010). P4P may thereby increase inequality in patients’ access to care in some cases (Oxholm, Di Guida & Gyrd-Hansen, Reference Oxholm and Gyrd-Hansen2021). By risk-adjusting the performance requirements, payers could potentially alleviate such issues. Similar to the case of FFS, multitasking issues may also arise under P4P if focus is diverted towards more profitable care (Doran et al., Reference Doran2011). Under P4P, providers also have an incentive to game the system to secure higher payments (Gravelle, Sutton & Ma, Reference Gravelle, Sutton and Ma2010; Kalk, Paul & Grabosch, Reference Kalk, Paul and Grabosch2010; Wilding et al., Reference Wilding2022). There may also be spillovers of P4P to other non-incentivized areas of care. For example, resource-constrained providers may choose to focus on incentivized treatments at the expense of non-incentivized treatments (Singh et al., Reference Singh2020), whereas non-incentivized care which is complementary to incentivized care may be positively affected by P4P.
The choice and sophistication of payment models depend on the availability of information technology and digital health infrastructure. Electronic medical records can often support new payment models that require either extensive risk adjustment of payments or the automation of the collection of data on quality and activity to support P4P and other quality improvement policies. Small private PCOs may find it difficult to transition to digital technology without outside investment. In high-income countries (HICs) there has therefore been a trend towards larger sizes of PCOs that benefit from economies of scale to spread out the fixed costs of new infrastructure. Often payers provide separate payments or block grants to support information technology and digital health infrastructure, but such funding may be unavailable in some low- and middle-income countries (LMICs) with more severe resource constraints. In settings where PCOs are in the private sector or in vertically integrated insurance systems, it can also be difficult to coordinate a single information technology health platform that can be used to support system-wide payment models and the sharing of electronic health records.
Payment method 5: blended payments
To balance the incentives of the pure payment methods, payments are often blended in primary care (Robinson, Reference Robinson2001). Blended payments are attractive because they enable payers to both incentivize providers to deliver care (e.g. using FFS and P4P) while limiting their spending (e.g. using salary and capitation). Theoretically, it has been shown that mixed payment methods are superior to pure payment methods (Ellis & McGuire, Reference Ellis and McGuire1986). However, in practice, the complexity of these methods may impose high transaction costs and difficulties in predicting behaviour. Empirical evidence on providers’ response to blended payment methods is limited. The results of a laboratory experiment with medical students show that the introduction of a mix of FFS and capitation leads to greater health benefits from care compared to the pure payment methods (Brosig‐Koch et al., Reference Brosig‐Koch, Hehenkamp and Kokot2017). Other studies, using real-world data, show that a change in provider payments from pure capitation to a mix of capitation and FFS increases PCPs’ activity levels and reduces their referral rates (e.g. Krasnik et al., Reference Krasnik1990; Flierman & Groenewegen, Reference Flierman and Groenewegen1992). P4P schemes are also often blended onto other payment models, which accounts for the majority of funding. The blend that best achieves payers’ different goals is, however, difficult to determine in a real-world setting and is likely context dependent.
After nurses and physicians, pharmacists are the third largest group of professionals in the global health workforce. In many places, the role of pharmacists is expanding and patients are seeking their first point of contact with the health care system in community pharmacies rather than medical offices. For example, a study in the USA demonstrated that patients visit community pharmacists almost twice as often as their physicians and other qualified health care professionals (Cheema, Sutcliffe & Singer, Reference Cheema, Sutcliffe and Singer2014; Santschi et al., Reference Santschi2011; Newman et al., Reference Newman2020). Having roles in prescription counselling, medication therapy management, health screening and immunizations (generally referred to as either extended pharmacy services or cognitive pharmaceutical services), pharmacists actively provide education, including smoking cessation services and distributing health education leaflets that cover the issues related to contraception and the prevention of cardiovascular diseases (Urick & Meggs, Reference Urick and Meggs2019; Anderson, Reference Anderson2007). As countries face physician shortages and ageing populations, the evolving model of pharmacy care may offer additional routes to enhance access to some primary care services, improve disease-related health outcomes and reduce health care costs.
In most places, pharmacy remuneration is concentrated on dispensing (a margin, maximum reimbursement price, or dispensing fee) (WHO Regional Office for Europe, 2020). However, some countries have started transitioning towards payment models (primarily FFS, capitation and blended payments of the two) to align funding with the expansion of cognitive pharmaceutical services; and some are opting for different modes of reimbursement for different pharmaceutical services. For example, Australia, Belgium, Canada, Germany, Japan, the Republic of Korea, Switzerland and some USA states are employing FFS schemes; Denmark, England, Ghana, Indonesia, the Netherlands, Portugal, Thailand, and some places in the USA (including Medicare and Medicaid) are using capitation; and Canada, China, and New Zealand are utilizing blended payments (mostly FFS integrated into capitation models).
These payment models for community pharmacists can have similar effects as they do in the primary care setting. FFS can encourage pharmacists to actively provide services (Jackson & Urick, Reference Jackson and Urick2019), but the fee does not account for the quality of the service provided and risks encouraging overprescription and overcounselling (Standridge, Fisher & Tsai, Reference Standridge, Fisher and Tsai1983). In capitation systems, pharmacists are reimbursed based on the number of patients on their lists, which can incentivize improved service quality and reductions in the costs of prescriptions (Yesalis et al., Reference Yesalis1980). This type of payment system can also allow for better financial planning, as pharmacies can better predict their revenue flows. However, this system gives less financial incentive for pharmacists to provide extended pharmacy services or cognitive pharmaceutical services because it does not change the individual payment received for services. Blended payments are often used to address the shortcomings of the two models when they are used alone (Jackson & Urick, Reference Jackson and Urick2019; Freeman et al., Reference Freeman2016).
As the role of community pharmacists continues to evolve, information sharing across the health care system (e.g. primary care, hospitals, long-term care) is becoming increasingly important to not only ensure the provision of safe, efficient and coordinated care delivery, but also to develop appropriate remuneration models for different health care providers. After all, there is an opportunity for community pharmacists to fill growing gaps in other parts of the health workforce, and appropriate payment models will be important to fully scale up the services provided in the community pharmacist setting.
Policy challenge 1: Can primary care payment models reduce avoidable secondary care?
Background to policy challenge
Many policy-makers believe that a strong primary care system can help reduce the growth or level of health care expenditures of more specialized hospital care. There exists a large literature showing strong associations between the strength (e.g. number of PCPs) of primary care and patients’ health outcomes (Starfield, Shi & Macinko, Reference Starfield, Shi and Macinko2005). Some of this literature also finds associations between a strong primary care system and lower total health expenditures (Baicker & Chandra, Reference Baicker and Chandra2004; Starfield & Shi, Reference Starfield and Shi2002) and fewer inpatient and emergency hospitalizations (Chen et al., Reference Chen2010; Jacobs et al., Reference Jacobs2020; Fortney et al., Reference Fortney2005; Lippi Bruni, Mammi & Ugolini, Reference Lippi Bruni, Mammi and Ugolini2016; Whittaker et al., Reference Whittaker2016; Morciano et al., Reference Morciano2020; Dolton & Pathania, Reference Dolton and Pathania2016). However, if primary care is expanding and the number of specialists and hospitals remain the same, then total health care expenditures will increase overall at the system level (Lau et al., Reference Lau2021; Miller et al., Reference Miller1999).
PCPs can potentially play an important role in improving efficiency by acting as gatekeepers to more expensive specialized care and by providing preventive care. Primary care can also be a substitute for or complement to more specialized care (Lau et al., Reference Lau2021). Primary care can act as a direct substitute when the same tasks can be provided with equal effectiveness by PCPs and specialists (Van Hoof et al., Reference Van Hoof2019). This substitution can, for example, include general health checks, skin checks, minor surgical procedures, antenatal care, care for less severe mental health conditions, child health (in the USA paediatricians act as PCPs for children whereas GPs take on this role in other countries), and monitoring patients with chronic diseases. In cases of substitution, the policy challenge is to ensure that the same quality of care is provided by the least costly health professional, which often is the PCP.
PCPs are usually paid less (Cheng et al., 2012; Leigh et al., Reference Leigh2010) and face lower fixed costs of providing care than specialists. From the patient’s perspective, it may also be less costly to access primary care in terms of travel time and time off work to visit a PCP (or a community pharmacist (see Box 3.1.2)) closer to home than to receive more specialized care in a more distant hospital. Lower costs of accessing care are especially relevant in rural areas where the distance to health care providers is a key issue. However, lower unit costs in primary care do not always translate into lower overall health care expenditure. As expenditure is a function of both unit costs as well as the volume of care, effects on expenditure also depend on the volume of care provided, which could increase in primary care if PCPs, for example, spend more time with patients or see more patients. A systematic review comparing consultations by PCPs to other specialists found that though outcomes were similar and waiting times were lower, costs were generally higher when treating patients in primary care compared to secondary care (Van Hoof et al., Reference Van Hoof2019). Despite the possibility of higher costs of treating patients in primary care, these may be partly offset by lower volume of referrals if the number of patients treated in primary care increases. The most cost-effective setting will, however, differ depending on the health condition and context.
In cases where primary care acts as a complement to specialized care, the policy challenge is to ensure the optimal mix of and collaboration between PCPs and other specialists for each patient. This challenge is related to integrated care for patients with multimorbidities and complex conditions (see Chapter 3.5) and is also related to referrals between PCPs and specialists. More generally, complementarity is about how PCPs and other health professionals with different skill sets can work to achieve the most cost-effective care for each patient. There will be much variation in the most cost-effective mix of PCPs and other specialists across different health conditions, with patients with multimorbidities and chronic diseases more likely to require care from providers across a range of specialties. In these cases, the impact of primary care on reducing the costs of specialized care is much less obvious, though it is believed that generalists, such as PCPs, will have a more holistic view of the patient’s health and circumstances.
The role of primary care payment models in reducing avoidable secondary care
Primary care payment models influence avoidable secondary care through their effects on PCP referrals to secondary, more specialized care (Forrest, Reference Forrest2003). Gatekeeping is a system where a referral from a PCP is required before a patient can visit a specialist. If gatekeeping does not exist then patients can access specialists directly, thereby reducing the impact of primary care payment models. Patients’ direct access could result in specialists seeing patients who do not “need” specialized care, such that specialists are not using their more costly time and training to its maximum benefit. In the longer term there would then be a higher demand for specialists and a misallocation of resources between primary and secondary care, which may also lead to higher health care expenditures overall. If gatekeeping does exist, then different primary care payment models can be used to influence the probability of a referral. More specifically, payment models can provide financial incentives to provide additional care (such as under FFS and P4P) and so reduce referrals, or share the financial risk of care with secondary care providers in vertically integrated systems which have pooled primary and secondary care budgets.
Capitation payment is one payment method which may influence referrals to more specialized care. However, its effect on referrals depends crucially on whether the capitated budget is pooled (also known as a “global” budget) across primary and secondary care, or only covers primary care (“single” budget). Single budget capitation may be paid to the PCO or directly to the PCP based on the number of enrolled patients and covers only the care provided by the PCPs themselves. In this situation, PCPs have an incentive to minimize their own treatment costs, which can be achieved by increasing referrals (Iversen & Lurås, Reference Iversen and Lurås2000). They may also minimize their own costs by choosing to spend less time with each patient and be less focused on coordination of care compared to under FFS and P4P payments. These financial incentives are similar to those present under salaried payment (see Table 3.1.1).
Empirical studies comparing single budget capitation payment to other payment models such as FFS and P4P have confirmed these hypotheses. With FFS payments, PCPs are paid on the basis of volume. PCPs therefore have a financial incentive to do more themselves, such as more follow-up visits. Referrals to specialists may therefore be delayed and be less likely under FFS compared to under (single budget) capitation or salary. Krasnik and colleagues (Reference Krasnik1990) found in Denmark that introducing FFS on top of single budget capitation substantially reduced GPs’ referrals to specialists and hospitals, while these GPs increased the number of face-to-face consultations and the number of diagnostic services undertaken themselves. The net effect on costs and health outcomes was not examined. Flierman and Groenewegen (Reference Flierman and Groenewegen1992) also reported increased activity under FFS mixed with capitation compared to capitation alone. The introduction of capitation in addition to FFS in Norway resulted in more referrals (Iversen & Lurås, Reference Iversen and Lurås2000), but in Canada there was no impact on hospital utilization when physicians switched from FFS to capitation payments plus an incentive to reduce hospitalization (Hutchison et al., Reference Hutchison1996).
Similar to FFS, P4P may encourage PCPs to provide more care themselves than under a fixed payment scheme such as capitation and salary. This increased activity may reduce referrals to hospital. Several studies have examined the spillover effects of P4P on secondary care in the context of the Quality and Outcomes Framework (QOF) (see Box 3.1.3), which is a large-scale P4P scheme added to an existing capitation payment model for PCPs in the United Kingdom (Doran et al., Reference Doran2006). Grigoroglou and colleagues (Reference Grigoroglou2020) found a very small reduction in avoidable hospitalizations for incentivized health conditions, while Harrison and colleagues (Reference Harrison2014) found a larger reduction in avoidable hospitalizations. Gunn et al. (Reference Gunn2021) found a substantial reduction in all-cause, diabetes and cardiovascular disease hospital admissions and emergency admissions for patients with diabetes if QOF targets were attained.
For some treatments, however, the Framework led to an increase in referrals. Gutacker and colleagues (Reference Gutacker2015) found that it led to an increase in psychiatric hospital admissions for people with serious mental illness. This finding suggests that in some circumstances P4P may lead to the identification of unmet need or incentivize more appropriate care. P4P can also directly reward more referrals if this is thought to improve health outcomes (Mcmanus et al., Reference McManus2021). A local P4P scheme in the United Kingdom found that emergency hospital admissions fell when GPs were directly rewarded for reducing them, but health outcomes were not examined (Khedmati Morasae et al., Reference Khedmati Morasae2021).
Even though there may be exceptions as described above, capitation under a single budget is generally found to increase the referral rate compared to FFS and P4P schemes. This financial incentive to refer patients under a single budget capitation-based system may be strengthened further if there is competition for patients. Iversen and Ma (Reference Iversen and Ma2011) show that incentives to refer under capitation payments are higher for PCPs facing more competition, because they may be more likely to respond to patients’ requests to be referred to prevent the patient from moving to another PCP.
The second main form of capitation is global capitation payment, which is where there is a “global” or “pooled” budget that includes total spending on primary and secondary care (and potentially other services). This budget may be held by a practice or a medical group or within a vertically integrated insurance system. Global capitation payments thereby provide a total health care budget based on the number of patients in the population or enrolled with an insurer, which is typically risk adjusted to account for patient complexity. An example is GP Fundholding in the United Kingdom or the use of capitated budgets in Health Maintenance Organizations and Accountable Care Organizations (ACOs) in the USA. In these cases, as under the first type of capitation payment (single budget), there is a financial incentive to minimize costs as providers can keep the savings. But in cases of a global (pooled) budget, this cost minimization includes keeping the savings that would have otherwise been used to pay for hospital care, which is why this payment method is also known as a shared-savings model. Several studies have shown that referrals by PCPs are lower with this type of global capitation payment (Davidson et al., Reference Davidson1992; Lurie et al., Reference Lurie1992; Dusheiko et al., Reference Dusheiko2006).
In ACOs, the global budget has also been combined with P4P. In these models, medical groups can share in any savings made from the global budget as long as they meet a minimum quality threshold (Chernew et al., Reference Chernew2011). Systematic reviews find positive evidence including reductions in spending and no deterioration of outcomes (Cattel & Eijkenaar, Reference Cattel and Eijkenaar2020; Kaufman et al., Reference Kaufman2017). Global budgets may also involve pooling of budgets across different sectors of care. For example, this approach has been tried for health and social care budgets in the United Kingdom, though it was found to have no impact on the utilization of hospitals, but there was some evidence of increased utilization for the most complex patients (Stokes et al., Reference Stokes2019). In this way global budgets that merge primary, secondary care, and social care budgets aim to help support integrated care across different settings (see Chapter 3.5).
A derivative of capitation payments are bundled payments. Bundled payments have been discussed as a way to incentivize integrated care across different sectors and settings for specific groups of patients defined by disease or diagnosis (Tsiachristas et al., Reference Tsiachristas2013; Stokes et al., Reference Stokes2018). Instead of the fixed capitation payment covering all care for each enrolled patient, fixed bundled payments are disease specific. They are designed as a single payment for all care required by a patient in the same episode or multiple services provided across different providers or sectors. Often, multiple FFS payments across a hospital episode may be “bundled” into a single payment (e.g. a diagnosis-related group). Alternatively, a single payment may be used to cover the care of patients in hospital and after they are discharged if they have a chronic disease. Similar to capitation payment, bundled payments provide incentives to reduce costs through providers being able to retain savings.
In the USA’s Medicare, bundled payments have been aimed at hospitals which become responsible for the care of patients up to 90 days after discharge. Though the role of primary care in these schemes is unclear, these schemes have some effects on reducing Medicare spending (Joynt Maddox et al., Reference Joynt Maddox2018; Barnett et al., Reference Barnett2019). In the Netherlands, bundled payments in primary care increased total health care expenditures, especially for those with multimorbidities. Medication and spending on medical specialists increased, while spending on primary care did not. The Dutch PCPs may have increased referrals because they were unable to keep savings from the reduction in use of specialists or because unmet needs were identified. In this context the “bundle” may have been too narrow by only including primary care, such that PCPs were incentivized to reduce only their own costs (Karimi et al., Reference Karimi2021; Stokes et al., Reference Stokes2018).
Until now, our focus has been on whether capitation (single or pooled budgets) may reduce avoidable hospitalizations, also in comparison to other payment models such as P4P and FFS. Adding P4P to FFS may also impact patients’ use of the secondary care sector. The effect on referrals depends on the precise performance measure used in P4P. If for a specific condition referral is a part of improving health outcomes, then PCPs’ referrals to specialists may increase. Several studies have examined the impact of adding other types of P4P to FFS. For example, Chen and colleagues (Reference Chen2017) found no impact of P4P on hospitalizations for hepatitis in Taiwan, where this payment was added onto FFS payment. In Canada, Lavergne and colleagues (Reference Lavergne2018) examined the introduction of an annual payment for guideline-based care plans on top of existing FFS for patients with diabetes, hypertension or chronic obstructive pulmonary disease (COPD). There were no changes in total hospitalizations, though for hypertension hospitalizations for stroke and acute myocardial infarction fell. The addition of P4P to existing FFS payment was also examined in the USA by Rosenthal et al. (Reference Rosenthal2016). They found that inpatient admissions fell in two out of the three examined states. Chen and colleagues (Reference Chen2010) found that after three years of P4P added to FFS payment, patients with diabetes were less likely to be hospitalized. There also exists a specific form of P4P directly incentivizing better coordination of care, called pay for coordination (P4C) (Stokes et al., Reference Stokes2018; Tsiachristas et al., Reference Tsiachristas2013). Under P4C additional payments are made to improve integration with other providers through information sharing. Less evidence exists on the effect of these types of payments on referrals and the use of secondary care.
In summary, there is evidence that primary care payment models can provide financial incentives that influence referrals to more specialized care depending on payment design. Single budget capitation models that only cover PCPs’ care can increase referrals. Generally, FFS and P4P encourage PCPs to undertake more care themselves and so are likely to reduce the use of secondary care compared to other forms of payment, though for P4P this depends on whether referrals are appropriate and lead to improved health outcomes. Global budget capitation payments that pool primary and secondary care funding can also reduce hospitalizations and encourage integration of care across settings. Though we have not examined the quality of the above evidence, many studies across a range of settings and countries had consistent findings. We could find little evidence, however, on how increases or reductions in referrals influence health outcomes and so this question remains an important avenue for further research.
Policy challenge 2: Can primary care payment models improve the quality of primary care?
Background to the policy challenge
A recent focus on quality of care and avoiding low-value care has increased the interest in ensuring that primary care payment models support the provision of high-value health care. In most health care systems, there is a difference between the actual care delivered by PCPs and the care recommended in evidence-based clinical guidelines. This difference is particularly present in LMICs (Das & Hammer, Reference Das2014). If patients do not receive high-quality primary care, it may reduce their quality of life and increase their need for more acute and specialized treatment, which is expensive for payers. As many PCPs face an ageing patient population with more complex care needs, following clinical guidelines is becoming even more important. A key policy challenge is therefore to improve the quality of primary care. This challenge is a part of a larger policy agenda on creating value-based health care systems (Smith et al., Reference Smith2021; Porter, Reference Porter2010). Policy-makers may use different interventions to try to improve the quality of primary care. One of these interventions is paying PCPs for their performance.
How can primary care payment models be used to improve quality?
The key payment method used to improve quality of care is P4P. Such schemes are characterized by linking PCPs’ payments directly to their attainment of quality standards. Even though P4P schemes are based on a quite simple concept, they can be designed and implemented in many different ways and settings and can become complex. These design and implementation choices require careful consideration as they may affect PCPs’ provision of care (Mehrotra, Sorbero & Damberg, Reference Mehrotra, Sorbero and Damberg2010; Eijkenaar, Reference Eijkenaar2013). In the following text, we highlight some of the key features to consider for P4P schemes that aim to improve the quality of primary care.
One key feature is the chosen performance indicators. The indicators should ideally reflect patients’ outcomes from care. However, patients’ outcomes may be difficult to measure and depend on factors unrelated to PCPs’ care. Consequently, the schemes instead often include structure and process indicators (Donabedian, Reference Donabedian1988). An example of a structure indicator could be “whether PCPs have a register of their patients’ smoking status”, whereas an example of a process indicator could be “whether patients with COPD were given a spirometry test”. The schemes may also include indicators related to changes in clinical measures that are more closely related to health outcomes, such as the reduction of blood pressure or blood sugar for diabetes. The different types of indicators to a different degree affect PCPs’ ability to control their own performance, potentially affecting their response to the payment scheme (Oxholm, Kristensen & Sutton, Reference Oxholm, Kristensen and Sutton2018).
As all aspects of quality of care cannot be captured by a single performance indicator, payers therefore need to consider the number of indicators to incentivize. On one hand, incentivizing only a few indicators could introduce multitasking issues and myopia because providers may choose to focus only on certain aspects of care (Eggleston, Reference Eggleston2005; Holmstrom & Milgrom, Reference Holmstrom and Milgrom1991). On the other hand, an increase in the number of incentivized indicators may increase the administrative burden and the complexity of the scheme (e.g. the United Kingdom QOF initially listed 146 performance indicators (see Box 3.1.3)). PCPs who operate in small PCOs may find the administrative burden of P4P schemes, including information technology infrastructure, particularly challenging (see Box 3.1.1). Therefore, P4P schemes should strike a balance between these two conflicting concerns.
Providers’ responses to P4P may depend on the basis for their reward – more specifically, the performance measure and the payment function that trigger the payment (Eijkenaar, Reference Eijkenaar2013). The performance measure may be based on their absolute performance (one time point), their improved performance (two time points), or their relative performance (comparison to other providers at one point in time or over two time points). An example of an absolute performance measure could be “the share of patients with COPD who received a spirometry test in a given year”, whereas an improvement in performance could be “the change in the share of patients with COPD who received a spirometry test in a given year compared to one year ago”. An example of a relative performance could be “the share of patients with COPD who received a spirometry test in a given year compared to the share provided across all PCPs”.
Each of the different types of performance measure comes with both advantages and disadvantages from the payer’s perspective. While rewards based on absolute performance may be perceived as transparent, this approach rewards previously achieved performance. In contrast, payments based on improvements reward only performance that has not previously been achieved. The issue with the measure of improvement is that it gives PCPs a financial incentive to lower performance in one year to be able to improve performance in the subsequent year. Paying based on relative performance (such as tournament-based payments) ensures that the reward adapts to general time trends in performance across PCPs. PCPs may, however, then be rewarded without having changed their behaviour, simply by performing better than others. The relative reward may therefore discourage collaborations between PCPs, unless it is paid at a “team” level such as a medical group.
The payment function determines the level of performance that triggers a reward. The function is often linear (flat rate) or non-linear based on thresholds. Flat-rate payments reward PCPs the same amount for each unit increase in their performance measure. For example, PCPs are rewarded for “each COPD patient that has received a spirometry test in a given year”. This payment function is simple and incentivizes continuous increases in the performance measure. However, it does not provide a clear overall performance target for the PCPs to reach. An alternative is to use threshold-based payment functions, which are characterized by lower or upper limits on the performance payments. One example is target-based payments, where a payment is made only if a certain performance target is met. For example, PCPs are only rewarded if “90% of patients with COPD receive a spirometry test in a given year”. Target-based payment schemes set a clear goal for the PCPs to reach, but only focus on the top end of the performance distribution and may thereby fail to incentivize PCPs who either cannot reach the target or have already reached it. Setting multiple targets may solve this issue by setting obtainable goals for those at the lower end of the performance distribution (Eijkenaar, Reference Eijkenaar2013; Mehrotra, Sorbero & Damberg, Reference Mehrotra, Sorbero and Damberg2010).
The basis of reward may also affect patients’ access to care. Providers may choose to prioritize patients where the costs of triggering a performance payment is low, to the detriment of patients where the cost of improving performance are higher, such as those with more complex conditions (Oxholm, Di Guida & Gyrd-Hansen, Reference Oxholm and Gyrd-Hansen2021). A solution to this unintended consequence of P4P could be to risk-adjust the performance requirements, similar to capitation payment. Another factor that may affect patients’ access to care is whether P4P affects patients’ OOP spending. In many LMICs patients pay OOP for their care. P4P may influence these payments if, for example, providers are incentivized to stimulate patients’ service use to improve their performance (Anselmi, Binyaruka & Borghi, Reference Anselmi, Binyaruka and Borghi2017). In such cases, P4P may make care more accessible to lower socioeconomic patient groups.
The size of the payments may also affect PCPs’ responses to the scheme (Eijkenaar, Reference Eijkenaar2013; Mehrotra, Sorbero & Damberg, Reference Mehrotra, Sorbero and Damberg2010; Zaresani & Scott, Reference Zaresani2021). The payment should be sufficient to cover the costs of changing behaviour, but not of a magnitude that creates budget deficits or removes focus from non-incentivized tasks. The costs of achieving a certain level of performance differ across indicators, PCOs and populations, which must be taken into account when setting payment rates.
The salience of the payment also influences the effectiveness of the scheme. A separate bonus may have more of an impact than if the same payment were delivered as part of usual revenues. The time lag between PCPs’ effort and payment may also affect their response to the scheme, with long lags being demotivating for PCPs (e.g. McMahon, Muula & De Allegri, Reference McMahon, Muula and De Allegri2018). Such delays may in some cases be due to auditing of the performance data. Auditing is an important task for reducing the risk of gaming under P4P, which has been found to be present in both HICs and LMICs (e.g. Gravelle, Sutton & Ma, Reference Gravelle, Sutton and Ma2010; Kalk, Paul & Grabosch, Reference Kalk, Paul and Grabosch2010; Wilding et al., Reference Wilding2022). The payers should therefore try to strike a balance between reducing payment delays and ensuring data accuracy.
Payments made to the medical group rather than the individual PCP may decrease the saliency of the scheme as the medical group may decide not to use the payment to increase the personal income of PCPs but for some other purpose. Earmarking the reward for specific quality-enhancing initiatives instead of to PCPs’ personal income could, however, also put focus on continuous quality improvement. In cases where the reward is given as personal income to the PCPs, the payers may choose to allocate payments in a way that reduces the strength of the incentives, such as sharing the payments equally rather than according to relative performance. This approach incentivizes collaboration among PCPs, but may also create so-called free-riding problems (Prendergast, Reference Prendergast1999), where some PCPs receiving the bonus do not contribute their fair share to the joint achievement.
The saliency of the scheme is also affected by feedback to PCPs on their performance. Some electronic health record systems allow for real-time feedback to PCPs or may automatically prompt PCPs to undertake certain actions based on clinical guidelines, which may improve the quality of their care beyond the financial incentives. The feedback on performance may also be made publicly available. As reputation among colleagues and patients may be important to the PCPs, public access to this information could potentially incentivize PCPs to improve their performance further (Prang et al., Reference Prang2021).
The United Kingdom’s QOF is one of the most comprehensive P4P schemes within a primary care setting. The scheme was introduced in 2004 with the aim of improving GPs’ job satisfaction, pay and quality of care (Doran & Roland, Reference Doran and Roland2010). Participation in the scheme was voluntary, but almost all general practices signed up (Campbell et al., Reference Campbell2009). General practices in the United Kingdom were also remunerated based on capitation, which gave them a financial incentive to keep costs down. The performance payment was added to this remuneration, thereby incentivizing general practices to improve quality of care. GPs’ personal income increased around 30% the first year of the QOF (Allen, Mason & Whittaker, Reference Allen and Whittaker2014).
Initially the scheme included 146 performance indicators, which were linked to clinical care, patient experience, organizational standards and additional services. A barrier for implementation of the QOF was the continuous measurement of practices’ performances on these indicators, which required automatic extraction of data from electronic records. A large investment in health information technology systems was therefore made prior to the introduction of the scheme. The practices were fully reimbursed for these information technology-related costs (Doran & Roland, Reference Doran and Roland2010).
For most performance indicators practices received payments determined by a threshold-based payment function, by which additional absolute performance below a lower (25%) and above an upper (50–90%) threshold was not financially rewarded. The payments were adjusted for disease prevalence and list size of practices. Practices could also exclude patients from the performance measurement if they were untreatable or not willing to be treated. Practices’ performance on each quality indicator was made publicly available (Doran & Roland, Reference Doran and Roland2010). Since the introduction of the scheme, many changes have been made both in terms of the basis for reward and the incentivized indicators.
Evidence of the consequences of P4P for primary care
The majority of the many literature reviews on the effectiveness of P4P schemes in health care conclude that the effect of P4P on quality of care is mixed and limited (see, for example, the reviews by Van Herck et al., Reference Van Herck2010; Eijkenaar et al., Reference Eijkenaar2013; Scott et al., Reference Scott2011). This conclusion also holds when only considering schemes implemented in LMICs (Diaconu et al., Reference Diaconu2021; Singh et al., Reference Singh2020). Whether care is provided in the primary or secondary health care sector may, however, affect the effectiveness of P4P. Some reviews state that the effect of P4P on the quality of care tend to be more positive when targeted at primary care rather than hospitals (Eijkenaar et al., Reference Eijkenaar2013). A recent systematic review of studies based only on outpatient health care finds that P4P slightly improves the quality of incentivized care (Jia et al., Reference Jia2021).
When assessing the consequences of P4P for primary care, most evidence has come from one of the most comprehensive schemes, the United Kingdom’s QOF (see Box 3.1.3). Studies conclude that in the first years of the QOF the performance on several incentivized indicators; for example, related to diabetes and asthma, improved at a faster rate than before the scheme (Gillam, Siriwardena & Steel, Reference Gillam and Siriwardena2012; Roland & Guthrie, Reference Roland and Guthrie2016; Campbell et al., Reference Campbell2009). In fact, many practices received maximum performance payment in the first year of the scheme (Roland & Guthrie, Reference Roland and Guthrie2016). In the longer run the GPs’ performance on the incentivized indicators generally remained at a high level but without further improvements (Guthrie & Tang, Reference Guthrie and Tang2016). Studies also find indications of the scheme leading to relative reductions in some emergency hospital admissions (Harrison et al., Reference Harrison2014), but no effects on mortality rates (Ryan et al., Reference Ryan2016; Kontopantelis et al., Reference Kontopantelis2015).
P4P’s moderate impact on quality of care could be explained by poor evaluation designs, but also by the schemes being designed and implemented in many different ways and settings. For example, small sizes of payments have been highlighted as an explanation for providers’ lack of response. However, a recent meta-regression analysis finds only weak evidence of a positive association between payment size and the effectiveness of P4P schemes (Zaresani & Scott, Reference Zaresani2021). In fact, the analysis finds only a few design features to be associated with the effectiveness of P4P. This conclusion should, however, be interpreted with caution as P4P studies often lack a detailed description of the design features of the interventions. More evidence is therefore needed on the impact of different designs of P4P on the quality of primary care.
P4P may not only impact quality of care but also access to care. The evidence on how P4P affects access to care is limited. Access issues are especially present in LMICs (Diaconu et al., Reference Diaconu2021). Some studies based on LMICs suggest that P4P may improve access to care (Singh et al., Reference Singh2020). This finding may partly be explained by providers doing more outreach work under P4P. For example, a study evaluating a P4P scheme in Rwanda suggests that community outreach may play a role in improving quality of child and maternal care services under P4P (Basinga et al., Reference Basinga2011). Several studies based on LMICs also mention the importance of availability of skilled personnel, drugs and equipment to improve patients’ access to care under P4P (Singh et al., Reference Singh2020). P4P may, however, in itself also have a positive effect on the availability of these resources and thereby improve patients’ access to care. Another factor that may affect patients’ access to care is OOP payments. A recent review of P4P based on LMICs, however, finds no clear changes in user fees under P4P (Diaconu et al., Reference Diaconu2021).
Conclusions
Payment models for primary care can influence broader health system objectives. This chapter, after summarizing the basic methods of payment, explored the extent to which two global policy challenges can be met through primary care payment methods. The evidence suggests primary care payment methods can be used to reduce avoidable secondary care and can influence the quality of primary health care. Though we have not provided a critical review of the quality of the evidence, previous studies suggest that these effects are context dependent and their measurement may vary with the quality of study designs. Furthermore, most studies focus on a subset of outcomes and rarely fully examine cost–effectiveness or effects on health outcomes of patients.
Strengthening primary care to keep patients out of hospital is an important solution to keep health care costs down, but it might not keep costs down if primary care is expanded while secondary care is not reduced. This solution therefore ignores the much more relevant policy question of the optimal mix between primary and secondary care. Existing empirical evidence suggests that payment models can help achieve an appropriate balance between primary and secondary care. This is possible with FFS which encourages PCPs to provide more care themselves, or with pooled capitation budgets, which can exist in vertically integrated health systems. However, expenditures may increase for single budget capitation, where the capitated budget covers only primary care. There also exists some evidence that P4P can encourage more appropriate referrals if performance is measured in terms of health outcomes or following evidence-based guidelines.
Evidence on whether payment models, in the form of P4P, can improve quality of care has mixed results. However, studies based on the primary care setting generally suggest that P4P slightly improves quality of care. The moderate response to P4P could be due to poor evaluation designs, but also due to the schemes being implemented in different contexts and with different payment designs. These design- and context-specific elements therefore require careful consideration. For example, if performance measures are not appropriately risk adjusted, P4P could lead to inequalities in access to primary care. Context-specific factors, such as availability of health care resources and OOP payments, could also impact the effectiveness of P4P. This might especially be the case in LMICs where many patients pay OOP for their care and access to skilled staff, equipment and medicine may be limited. Meanwhile, P4P may in some cases also be used as an instrument to improve the availability of these resources in such settings.
Payment methods in primary care are often blended to create a balanced mix of incentives. For example, salary and capitation payments provide an incentive to minimize costs, whereas P4P and FFS incentivize the PCP to increase activity. The precise blend of these different payment schemes required for payers to reach their goals is difficult to determine as it may depend on the health care setting and context, such as availability of resources or competition among providers. More knowledge is therefore needed on how to blend payments to achieve payers’ goals.
Primary care payment models are of course only one way to help meet health care payers’ goals. These payment models need to be carefully considered within the context of each health care system and its level of development and funding. For example, there are clear differences between HICs and LMICs, where in the latter primary care is usually publicly funded and the last part of the health care system to develop after more specialized hospital care in metropolitan areas. In addition, LMICs generally have fewer resources and so are less likely to have the digital infrastructure that is needed to underpin some payment models such as P4P and the extensive risk adjustment necessary in capitation models (see Box 3.1.1). Payers therefore often face a trade-off between complexity and effectiveness when designing a primary care payment model.
We have examined two important global policy challenges that primary care payment models may impact, i.e. reducing avoidable secondary care and increasing quality of care. There are also many other aspects of health care that primary care payment models can potentially influence, including access to health care such as PCPs’ labour supply, geographical distribution and specialty choice. Reforms to primary care payment models therefore need to be considered holistically in terms of their effects on payers’ goals.Footnote 1