Summations
-
Rapid and Sustained Antidepressant Effects: Psilocybin therapy has demonstrated rapid and substantial reductions in depressive symptoms, often within a single or few doses, with effects sustained for weeks to months.
-
5-HT2A Receptor and Neuroplasticity: The antidepressant effects of psilocybin are primarily mediated through 5-HT2A receptor activation, promoting synaptic plasticity and emotional processing, differentiating it from traditional antidepressants.
-
Controlled Use and Safety Profile: While generally well-tolerated, psilocybin therapy requires structured clinical settings with psychological support.
Considerations
-
Small Sample Sizes and Short Follow-Ups: Many studies involve small cohorts and short observation periods, limiting the generalisability of long-term effects and relapse rates.
-
Lack of Standardised Dosing and Protocols: Variability in psilocybin doses, session frequency, and psychological support structures complicates direct comparisons and clinical applicability.
-
Limited Diversity in Study Populations: Most studies focus on Western populations, with minimal representation of diverse ethnic, cultural, and socioeconomic backgrounds, affecting external validity.
Highlights
-
Psilocybin exhibits rapid and sustained antidepressant effects, often after one or two sessions, with improvements lasting weeks to months;
-
Its therapeutic mechanism primarily involves activation of the 5-HT2A receptor, which promotes neuroplasticity and emotional processing essential for alleviating depression;
-
Current research faces limitations such as small sample sizes, short follow-up periods, lack of standardised protocols, and limited diversity, underscoring the need for further comprehensive studies.
Introduction
Depression is a chronic mental disorder that affects millions globally, and according to the World Health Organization (WHO, 2019), over 350 million people worldwide suffer from this condition. Approximately 40.27 million individuals in Europe are affected, constituting 4.3% of the population. At the same time, in the United States, the National Institute of Mental Health (NIMH, 2023) reported that 8.4% of adults experienced at least one major depressive episode in 2021. Women are more likely than men to experience depression, and it is most commonly observed among young and elderly populations (Salk et al., Reference Salk, Hyde and Abramson2017; Kirkbride et al., Reference Kirkbride, Anglin, Colman, Dykxhoorn, Jones, Patalay, Pitman, Soneson, Steare, Wright and Griffiths2023; Wang et al., Reference Wang, Mathai, Gukasyan, Bowers, Carhart-Harris, Davis, DiBenedetto, Donnelly, Evans, Grob, Hoh, Johns, Johnson, Kalluri, Kettner, Kraynak, Kuypers, Lancelot, Lister, Litten, McMahon, Muench, Nutt, Pollan, Reifler, Rohan, Ross, Sachs, Shulman, Spriggs, Stroud, Tupper, Van der Waerden, Wilson, Wurzman and Zeno2024). Symptoms of depression can include persistent sadness, loss of interest in previously enjoyable activities, reduced attention, pessimism about the future, and feelings of guilt or unworthiness. Physical symptoms such as chronic pain, fatigue, sleep disturbance, appetite changes, and weight fluctuations are also common. Patients can be classified as having mild, moderate, or severe depression, with psychological therapy and pharmacotherapy being the primary treatment modalities (Fergusson et al., Reference Fergusson, Horwood, Ridder and Beautrais2005; Klein et al., Reference Klein, Shankman, Lewinsohn and Seeley2009; Kirkbride et al., Reference Kirkbride, Anglin, Colman, Dykxhoorn, Jones, Patalay, Pitman, Soneson, Steare, Wright and Griffiths2023; Bacigalupe et al., Reference Bacigalupe, Martín, Triolo, Sjöberg, Rydberg Sterner, Dekhtyar, Fratiglioni and Calderón-Larrañaga2024).
The most commonly prescribed medications for depression fall into five categories: serotonin reuptake inhibitors (SRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants, monoamine oxidase inhibitors (MAOIs), and atypical antidepressants (Boschloo et al., Reference Boschloo, Vogelzangs, Van den Brink, Smit, Beekman and Penninx2023). These medications primarily act as monoamine modulators and have demonstrated efficacy in mood disorder treatment. Medicinal chemistry and pharmacological data underscore the importance of serotonin (5-HT) and the 5-HT2A receptor in the pathophysiology of depression, with recent findings highlighting the rapid antidepressant effects of ligands targeting this receptor (Hieronymus et al., Reference Hieronymus, Emilsson, Nilsson and Eriksson2019; Kaiser et al., Reference Kaiser, Ting, Monteiro and Feng2021; Boschloo et al., Reference Boschloo, Vogelzangs, Van den Brink, Smit, Beekman and Penninx2023).
While traditional pharmacotherapy remains a cornerstone of treatment, alternative approaches, including psychedelic medicine, have garnered significant research interest. Psychedelics may influence depression through mechanisms tied to neuroplasticity and emotional processing. For instance, studies have shown that the intensity and emotional resonance of a psychedelic experience can positively affect depressive symptoms (Griffiths et al., Reference Griffiths, Richards, Johnson, McCann and Jesse2006; Carhart-Harris et al., Reference Carhart-Harris, Muthukumaraswamy, Roseman, Kaelen, Droog, Murphy, Tagliazucchi, Schenberg, Nest, Orban, Leech, Williams, Williams, Bolstridge, Sessa, McGonigle, Sereno, Nichols, Hellyer, Hobden, Evans, Singh, Wise, Curran, Feilding and Nutt2016; Carhart-Harris and Goodwin, Reference Carhart-Harris and Goodwin2017; Roseman et al., Reference Roseman, Nutt and Carhart-Harris2018).
These effects are thought to stem from the ability of psychedelics to engage specific receptors and induce structural and functional neuroplasticity. Advances in electrophysiological methods to study brain plasticity, such as those described by De Vos et al. Reference De Vos, Mason, Kuypers, Toennes, Reckweg and de la Torre(2022), have provided more profound insights into how psychedelics induce these neuroplastic changes, bridging the gap between preclinical and clinical research.
Amid these findings, the continued prevalence of depression emphasises the need for innovative treatment approaches. In 2019, WHO reported that approximately 970 million people worldwide, or one in eight, suffered from mental disorders, with depression and anxiety being the most common World Health Organization (WHO). These statistics underline the urgency for expanding therapeutic options and advancing our understanding of this complex condition, offering hope for better outcomes for sufferers worldwide.
The 5-HT2A receptor is a crucial mediator in the pharmacodynamics of psychedelic drugs, primarily facilitating their psychoactive effects through activation of serotonin 5-HT2A receptors predominantly located in the cortical regions of the brain. These receptors are densely expressed in the prefrontal cortex associated with higher-order cognitive functions. Their activation by psychedelics such as psilocybin, LSD, and DMT leads to profound alterations in perception, cognition, and emotion, which are hallmark features of the psychedelic experience (Cameron et al., Reference Cameron, Benetatos, Lewis, Bonniwell, Jaster, Moliner, Castrén, McCorvy, Palner and Aguilar-Valles2023; Pędzich et al., Reference Pędzich, Kogan and Bonafina2022; Nichols, Reference Nichols2016).
Upon activation of the 5-HT2A receptors, psychedelics trigger a cascade of intracellular signalling pathways, particularly involving the phospholipase C (PLC) pathway. This pathway subsequently increases the production of inositol trisphosphate (IP3) and diacylglycerol (DAG), leading to the release of calcium ions from intracellular stores (Raote and Bhattacharya, Reference Raote and Bhattacharya2020).
The activation of 5-HT2A receptors by psychedelics modulates the excitability of cortical neurons and influences synaptic plasticity. These mechanisms underpin the profound changes in consciousness and perception these substances induce (Preller and Vollenweider, Reference Preller and Vollenweider2019).
Moreover, the activation of 5-HT2A receptors influences glutamatergic neurotransmission by enhancing glutamate release in the prefrontal cortex. This glutamate release, in turn, affects the activity of other neurotransmitter systems, such as dopamine and GABA, contributing to the broad-spectrum effects on neural circuits that underpin the psychedelic state (Vollenweider and Kometer, Reference Vollenweider and Kometer2010). The intricate interplay between these neurotransmitter systems and the 5-HT2A receptor is still being explored to delineate the full scope of psychedelic-induced neuroplasticity and its potential therapeutic applications in treating psychiatric disorders (Wing et al., Reference Wing, Tapson and Geyer1990; Pędzich et al., Reference Pędzich, Jędrzejewski, Wróbel and Urbanik2022).
One of the most significant effects of the psychedelic experience triggered by the activation of the 5-HT2A receptors is the profound changes in perception and thought processes. This may include visual hallucinations, altered sense of time, and deep introspective or existential thoughts. These effects have been used traditionally in various cultures and studied more recently for the therapeutic potential for mental health conditions such as depression, anxiety, and PTSD (Hoskins et al., Reference Hoskins, Pearce, Bethell, Dankova, Barbui, Tol, van Ommeren, de Jong, Seedat, Chen and Bisson2015; Steenkamp et al., Reference Steenkamp, Litz, Hoge and Marmar2015).
To sum up, the 5-HT2A receptor’s role in psychedelics is critical to the psychotropic effects these substances produce, significantly impacting perception, cognition, and emotional states. Growing evidence suggests that psilocybin in low doses can help patients with depression symptoms (Gasser et al., Reference Gasser, Holstein, Michel, Doblin, Yazar-Klosinski, Passie and Brenneisen2014; Sessa, Reference Sessa2015; Carhart-Harris et al., Reference Carhart-Harris, Bolstridge, Day, Rucker, Watts, Erritzoe, Kaelen, Giribaldi, Bloomfield, Pilling, Rickard, Forbes, Feilding, Taylor, Curran and Nutt2018). The side effects are very low, and the systematic reviews have found that psychedelic drugs, when associated with safe use in a safe place, could help in the well-being of patients with depression or cancer terminal (De Vos et al., Reference De Vos, Mason, Kuypers, Toennes, Reckweg and de la Torre2022; Amsterdam et al., Reference Amsterdam, Panossian and Rhodin2022).
Existing reviews on psilocybin and depression often lack the methodological rigour needed to minimise bias and subjectivity. Furthermore, many do not adhere to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines or pre-registration protocols, which ensure transparency, reproducibility, and methodological consistency. This review seeks to address these gaps by employing a systematic approach guided by PRISMA and pre-registering the protocol in PROSPERO. By adhering to these standards, this review aims to provide a comprehensive and unbiased synthesis of the current evidence, critically evaluating the therapeutic potential of psilocybin in depression treatment.
In doing so, this review serves several objectives: (1) to consolidate existing evidence on the efficacy and safety of psilocybin for depression and the role of 5HT2a, (2) to identify limitations and inconsistencies in the current literature, and (3) to highlight areas requiring further investigation. By addressing these gaps, this review aims to enhance the understanding of psilocybin’s role in depression management and inform future research and clinical practice.
Materials and methods
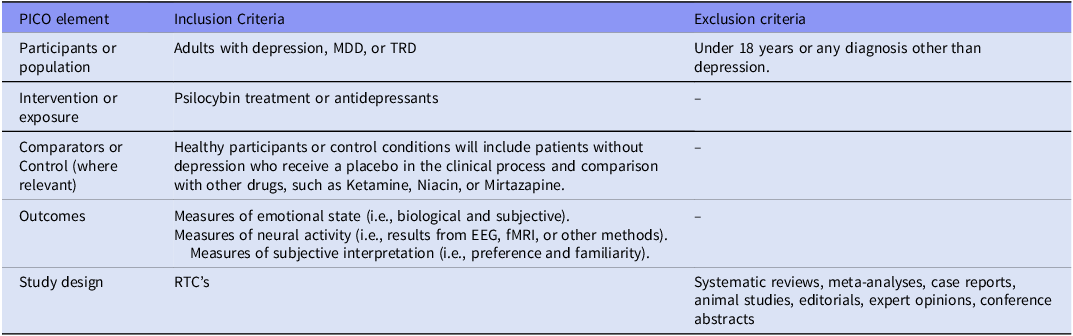
A systematic review was carried out according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement guidelines. The research question and search strategy were formulated using the PICO (Population, Intervention, Comparator, and Outcome) framework, which can be found in Table 3 of the supplementary material. The protocol was registered with PROSPERO under the CRD42023457537 registration number.
Search methods
Literature searches were conducted using five online databases: Embase, Scopus, PsycINFO, PubMed, and Web of Science to identify studies with relevant information. We searched for studies in any language between 2018 and 2023 along with an additional two studies from 2025, which refer to the research on the effects of psilocybin on the 5HT2a receptor in a depression model. The terms used were: [(psilocybin[MeSH Terms]) OR (psilocybin) OR (psilocybin) OR (psilocybin) OR (psiloc*)] AND [(depressive disorder [AND] (5HT2a)] psilocybin OR psilocybin OR psilocybine OR psilocybin OR psiloc* AND depressive AND disorder AND 5ht2a))’. No restrictions were applied on country, gender, or race/ethnicity.
Search strategy
Two authors (FRTA and GT) independently identified most of the double-blind RCTs published before 1 November 2023. In April 2025 (FRTA, GT, and RMMA), two more studies were added that examined the efficacy and safety of psilocybin in the treatment of MDD.
Inclusion and exclusion criteria
Original articles reporting the effects of psilocybin or other psychedelic drugs on depression were included. Studies focused on other mental health disorders, animal models, editorials, opinion pieces, conference abstracts, case reports, systematic reviews, meta-analyses, and articles not published in English were excluded. Also, studies involving healthy participants (Holze et al., Reference Holze, Ley, Müller, Becker, Straumann, Vizeli, Kuehne, Roder, Duthaler, Kolaczynska, Varghese, Eckert and Liechti2022; Madsen et al., Reference Madsen, Fisher, Burmester, Dyssegaard, Stenbæk, Kristiansen, Johansen, Lehel, Linnet, Svarer, Erritzoe, Ozenne and Knudsen2020; Barret et al., Reference Barrett, Carbonaro, Hurwitz, Johnson and Griffiths2018; Carbonaro et al., Reference Carbonaro, Johnson, Hurwitz and Griffiths2018; Ley et al, Reference Ley, Viechtbauer, Hohagen, Baune and Domschke2023) were included, with a justified rationale outlined below.
Including studies conducted with healthy participants in this systematic review was justified for several key reasons. First, these studies offer valuable mechanistic insights into how psilocybin modulates brain function, mainly through serotonergic pathways directly implicated in depression. Understanding these mechanisms is essential for interpreting psilocybin’s potential therapeutic effects. Second, they provide critical safety and tolerability data, which is a necessary foundation for designing and conducting clinical trials in vulnerable populations such as those with depression. Third, studies in healthy participants help determine the dose–response relationship and characterise subjective effects, such as altered states of consciousness, which are hypothesised to mediate therapeutic outcomes in clinical populations.
While studies with healthy participants do not directly measure antidepressant efficacy, they play a pivotal role in bridging preclinical research (e.g., animal models) and clinical trials. By providing data on pharmacokinetics, subjective experiences, and safety profiles, these studies contribute to developing appropriate therapeutic protocols. Therefore, including such studies broadens the scope and depth of this review by contextualising findings from clinical trials, enhancing its overall comprehensiveness.
Data extraction
Two authors (FRTA and GT) independently extracted the following data from each included RCT using a tailored form: author, year of publication, methods, instruments, country, clinical trials, aims, number of sessions, type of drugs, decreased depression, related side effects, sample, and conclusions. Any discrepancies were resolved by discussion and adjudication through a senior author. If the study data needed to be clarified, the first/corresponding authors were contacted by email to obtain further information.
Assessment of study quality
To ensure methodological rigour, three authors (FRTA, GT, and RMMA) assessed study quality independently using the Newcastle–Ottawa Scale (NOS) and the Jadad scale. The NOS evaluates non-randomised studies across three key domains: selection, comparability, and outcome assessment (Wells et al., Reference Wells, Shea, OConnell, Peterson, Welch, Losos and Tugwell2000). The Jadad scale was applied for randomised controlled trials, with a score of ≥ 3 indicating high-quality studies (Jadad et al., Reference Jadad, Moore, Carroll, Jenkinson, Reynolds, Gavaghan and McQuay1996). Additionally, the overall quality of primary and secondary outcomes was evaluated using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) system, providing a structured framework to rate the certainty of evidence. All assessments were performed independently by FRTA and RMMA, with discrepancies resolved through discussion.
Results
Search results
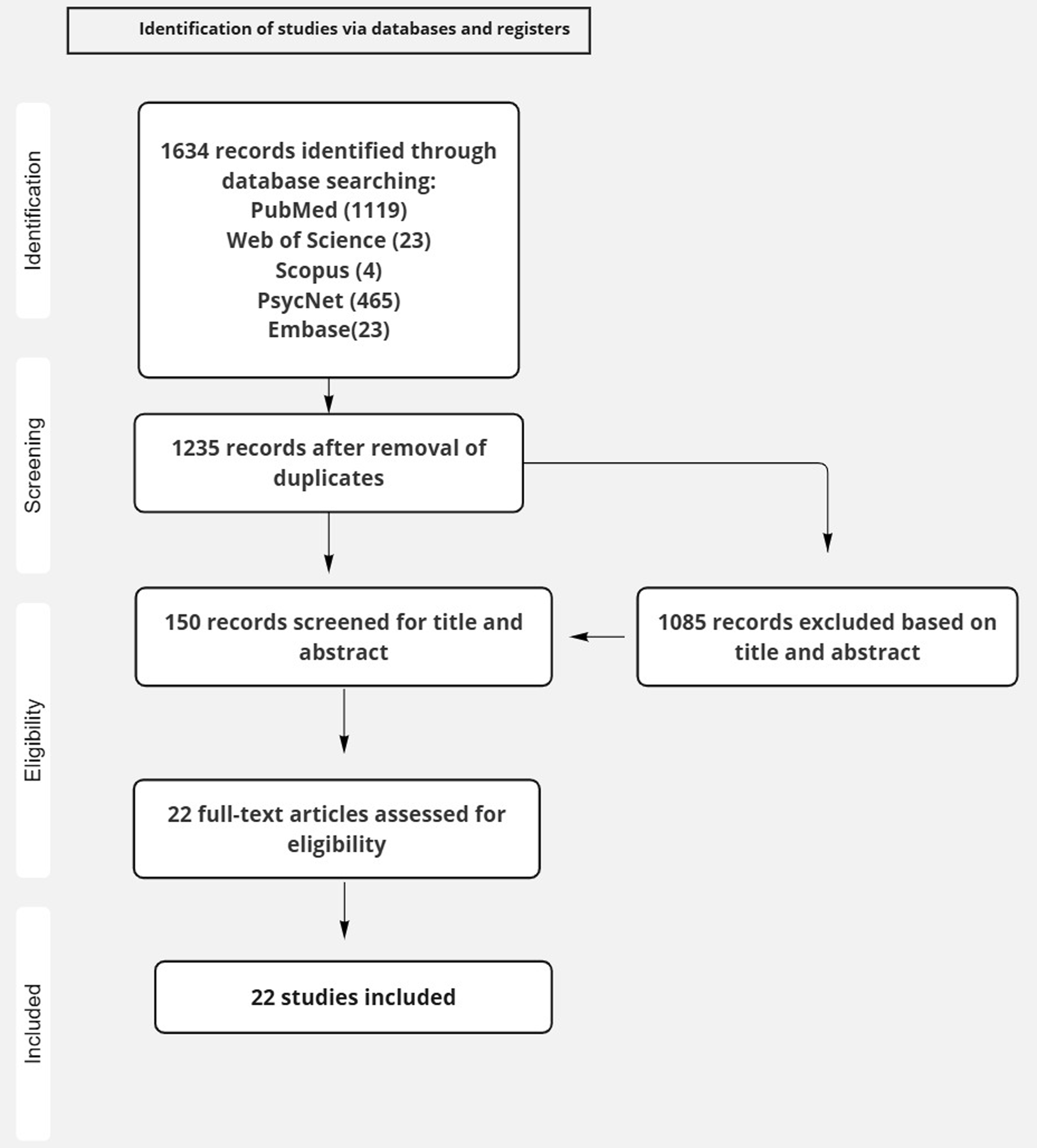
A comprehensive search identified 1,634 articles (Fig. 1), of which 105 studies met the inclusion criteria. 150 studies were excluded for not meeting the eligibility requirements. Exclusion criteria included review articles or meta-analyses, non-English language publications, studies using animal models, or investigations involving other psychiatric conditions such as addiction or bipolar disorder. Ultimately, 22 studies were included in the final analysis.
Flow char for selection of studies.

Considerations regarding included studies
Some included studies (Carhart-Harris et al., Reference Carhart-Harris, Giribaldi, Watts, Baker-Jones, Murphy-Beiner, Murphy, Martell, Blemings, Erritzoe and Nutt2021; Barba et al., Reference Barba, Buehler, Kettner, Radu, Cunha, Nutt, Erritzoe, Roseman and Carhart-Harris2022; Murphy et al., Reference Murphy, Jones, Alonso, Calvert and Lyon2022; Goodwin et al., Reference Goodwin, Aaronson, Alvarez, Arden, Baker, Bennett, Bird, Blom, Brennan, Brusch, Burke, Campbell-Coker, Carhart-Harris, Cattell, Daniel, DeBattista, Dunlop, Eisen, Feifel, Forbes, Haumann, Hellerstein, Hoppe, Husain, Jelen, Kamphuis, Kawasaki, Kelly, Key, Kishon, Knatz Peck, Knight, Koolen, Lean, Licht, Maples-Keller, Mars, Marwood, McElhiney, Miller, Mirow, Mistry, Mletzko-Crowe, Modlin, Nielsen, Nielson, Offerhaus, O’Keane, Páleníček, Printz, Rademaker, van Reemst, Reinholdt, Repantis, Rucker, Rudow, Ruffell, Rush, Schoevers, Seynaeve, Shao, Soares, Somers, Stansfield, Sterling, Strockis, Tsai, Visser, Wahba, Williams, Young, Ywema, Zisook and Malievskaia2022) were derived from the same randomised clinical trials, sharing participant samples and protocols. However, these studies report on different outcomes, sub-analyses, or follow-up measures. In accordance with our predefined criteria, these studies were retained because each contributes unique and complementary insights into psilocybin’s therapeutic profile.
Characteristics of the included studies
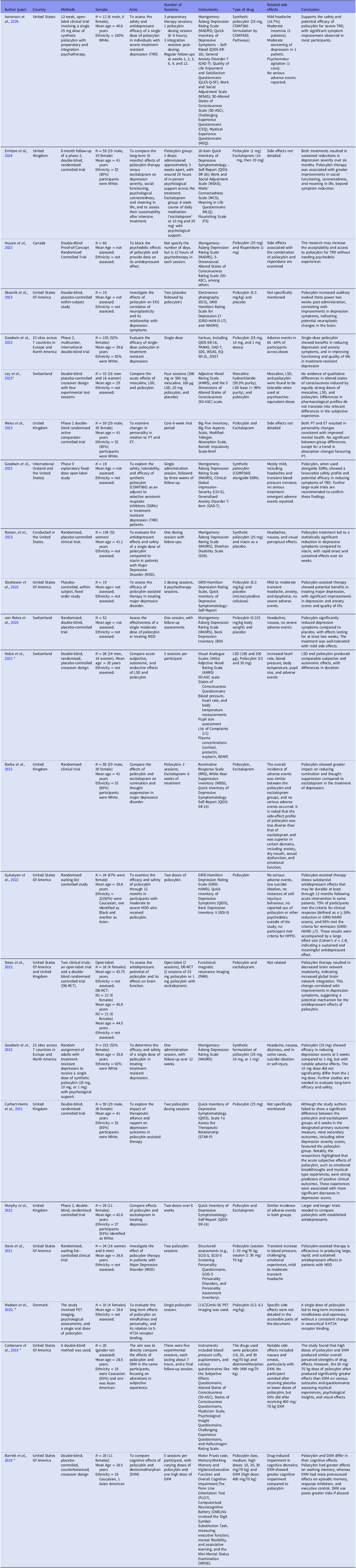
The 22 included studies primarily focused on psilocybin’s efficacy in treating major depressive disorder (MDD) and treatment-resistant depression (TRD). These studies employed robust methodologies, particularly randomised clinical trials (RCTs), to examine psilocybin’s therapeutic potential. Sample characteristics for each study are provided in Table 1, and additional methodological details are summarised in Tables 2–3 (Supplementary Material).
Examples include Skosnik et al. Reference Skosnik, Cortes-Briones and De Lisi(2023), who used a double-blind, placebo-controlled, within-subject design to assess EEG correlates of neural plasticity in MDD, and Ley et al. Reference Ley, Viechtbauer, Hohagen, Baune and Domschke(2023), who employed a double-blind, placebo-controlled crossover design. Phase II trials, such as Goodwin et al. Reference Goodwin, Aaronson, Alvarez, Atli, Bennett, Croal, DeBattista, Dunlop, Feifel, Hellerstein, Husain, Kelly, Lennard-Jones, Licht, Marwood, Mistry, Páleníček, Redjep, Repantis, Schoevers, Septimus, Simmons, Soares, Somers, Stansfield, Stuart, Tadley, Thiara, Tsai, Wahba, Williams, Winzer, Young, Young, Zisook and Malievskaia(2023), further evaluated safety and efficacy.
Selected Studies.
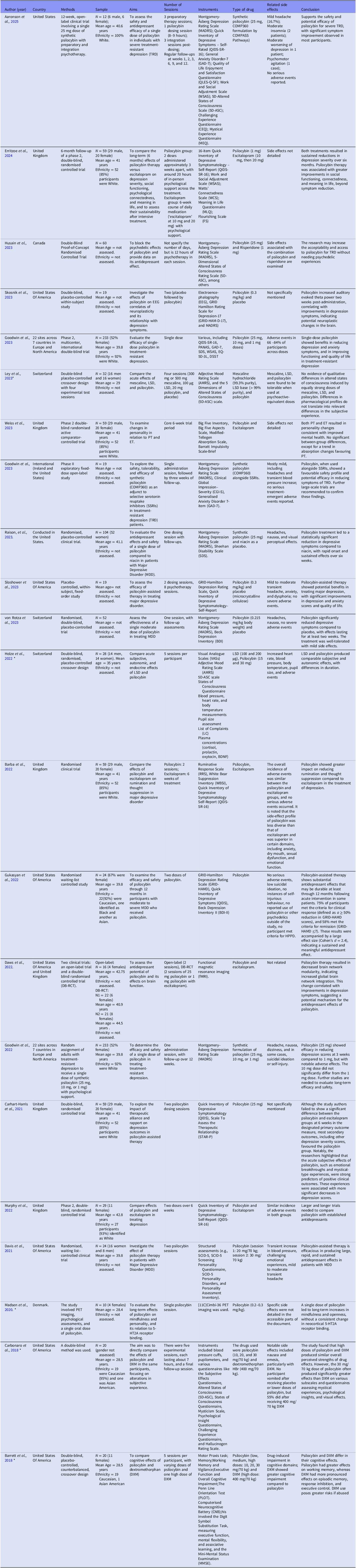
Note: The Montgomery–Åsberg Depression Rating Scale (MADRS) is a clinician-administered tool to assess depressive symptom severity. It focuses on core symptoms such as sadness, tension, reduced sleep, appetite, concentration, and inability to feel. Scores range from 0 to 6 per item, with higher scores indicating greater severity (Montgomery & Åsberg, Reference Montgomery and Åsberg1979).
The GRID-Hamilton Depression Rating Scale (GRID-HAMD) is a standardised clinician-administered tool for assessing depression severity across mood, sleep, appetite, and physical symptoms. Higher scores reflect more severe depression, while lower scores suggest improvement (Williams, Reference Williams1988; Williams et al., Reference Williams, Kobak, Bech, Engelhardt, Evans, Lipsitz, Olin, Pearson and Kalali2008).
The Quick Inventory of Depressive Symptomatology–Self-Report (QIDS-SR-16) evaluates depressive symptoms such as mood, sleep, weight, and energy through patient self-report. Scores indicate depression severity (Rush et al., Reference Rush, Trivedi, Ibrahim, Carmody, Arnow, Klein, Markowitz, Ninan, Kornstein, Manber, Thase, Kocsis and Keller2003).
The Beck Depression Inventory (BDI) is a self-report tool assessing cognitive, emotional, and physical depression symptoms, with higher scores reflecting greater severity (Beck et al., Reference Beck, Ward, Mendelson, Mock and Erbaugh1961).
The Sheehan Disability Scale (SDS) measures functional impairment across work, social, and family domains, offering insights into how depressive symptoms interfere with daily life (Sheehan, Reference Sheehan1983).
The Five-Dimensional Altered States of Consciousness Rating Scale (5D-ASC) assesses subjective altered states during psychedelic experiences. It evaluates dimensions such as oceanic boundlessness, visual restructuralization, and ego dissolution (Dittrich, Reference Dittrich1998).
The Positive and Negative Affect Schedule (PANAS) measures mood states by assessing positive and negative affect dimensions, offering insights into emotional responses during interventions (Watson et al., Reference Watson, Clark and Tellegen1988).
The Generalised Anxiety Disorder Scale (GAD-7) is a widely used self-report tool that evaluates the severity of generalised anxiety symptoms, with higher scores indicating greater severity (Spitzer et al., Reference Spitzer, Kroenke, Williams and Löwe2006).
The Work and Social Adjustment Scale (WSAS) measures functional impairment in work, social, and personal domains, providing an understanding of the impact of mental health conditions on daily life (Mundt et al., Reference Mundt, Marks, Shear and Greist2002).
The EQ-5D-3L is a standardised instrument for measuring health-related quality of life. It assesses mobility, self-care, usual activities, pain/discomfort, and anxiety/depression (EuroQol Group, 1990).
The Digit Symbol Substitution Test (DSST) measures cognitive functioning, including attention, processing speed, and executive function, often used in clinical studies involving neurological or psychological conditions (Wechsler, Reference Wechsler1955).
The Adjective Mood Rating Scale (AMRS) evaluates mood changes, including calmness, energy levels, and happiness, and is often used in studies involving psychoactive substances (Janke & Debus, Reference Janke and Debus1978).
The Mystical Experience Questionnaire (MEQ) measures mystical or spiritual experiences during interventions like psilocybin. It focuses on feelings of unity, transcendence, and sacredness (Barrett et al., Reference Barrett, Johnson and Griffiths2015).
The Emotional Breakthrough Inventory (EBI) assesses emotional release and transformation, capturing moments of resolving struggles and gaining new perspectives during therapy (Roseman et al., Reference Roseman, Haijen, Idialu-Ikato, Kaelen, Watts and Carhart-Harris2019).
The Big Five Inventory (BFI) and the Big Five Aspects Scale assess personality traits across five dimensions: openness, conscientiousness, extraversion, agreeableness, and neuroticism (John & Srivastava, Reference John and Srivastava1999).
The Modified-Tellegen Absorption Scale measures individuals’ openness to immersive and transformative experiences, often in psychedelic contexts (Tellegen & Atkinson, Reference Tellegen and Atkinson1974).
The Barratt Impulsivity Scale-Brief evaluates impulsivity across cognitive, motor, and non-planning domains, commonly used in psychological studies (Patton et al., Reference Patton, Stanford and Barratt1995).
The Ruminative Response Scale (RRS) measures the extent to which individuals focus on negative thoughts and emotions, often used in depression studies (Nolen-Hoeksema, Reference Nolen-Hoeksema1991).
The White Bear Suppression Inventory (WBSI) assesses thought suppression tendencies, which are associated with mental health outcomes, including depression and anxiety (Wegner & Zanakos, Reference Wegner and Zanakos1994).
The Visual Analogue Scale (VAS) is a simple tool for measuring subjective experiences, such as mood or pain intensity, by having participants mark a point on a line representing their experience (Huskisson, Reference Huskisson1974).
The QLES-Q-SF measures the degree of enjoyment and satisfaction individuals experience across various areas of daily life, including physical health, mood, work, and social relationships. It is widely used in psychiatric research and clinical trials to assess quality of life, particularly in depression and related disorders. (Endicott, et al., Reference Endicott, Nee, Harrison and Blumenthal1993 ).
Geographical and demographic distribution
The majority of studies were conducted in North America (n = 12) and Europe (n = 10), with three studies involving participants from both regions. Across all studies, the combined sample included 852 participants, with a mean age of 39.1 years (range: 28.5 – 43.3). Due to inconsistent reporting, a standard error of the mean (SEM) could not be uniformly calculated.
Methodological insights
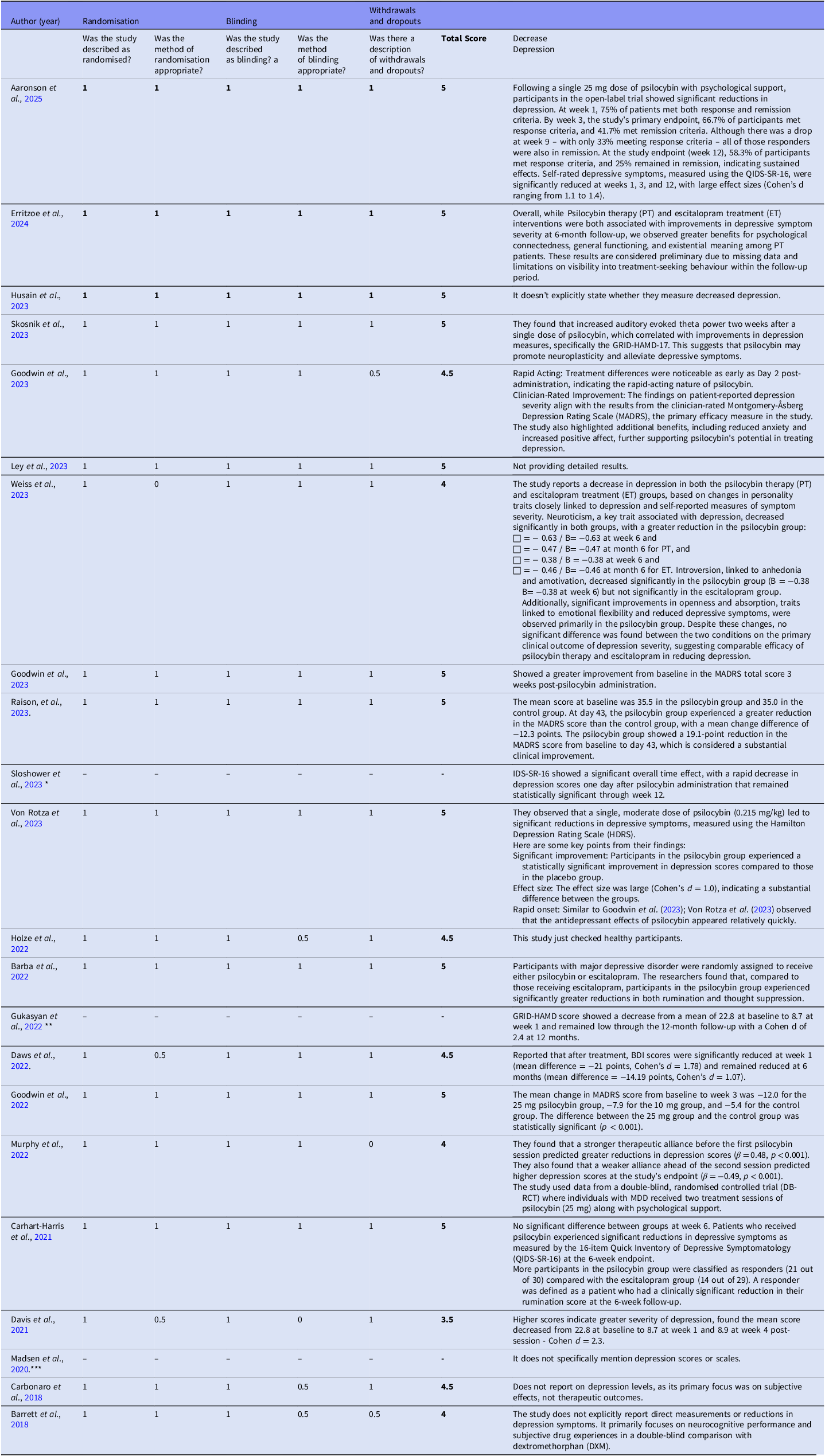
Randomisation methods varied across studies and included computer-generated randomisation, block randomisation, and stratified randomisation. Blinding procedures, essential for minimising bias, were inconsistently reported. All studies were rated as 3–5 points (high-quality studies with low risk of bias) on the Jadad scale (Table 2). In Table 2, we evaluated three variables related to bias level: randomisation, blinding, and withdrawals/dropouts. Additionally, we indicated whether there was a reduction in depression.
Methodological quality and risk of bias (Jadad Scale).

* This exploratory, placebo-controlled, fixed-order trial investigated the effects of psilocybin-assisted therapy for major depressive disorder. While the trial was placebo-controlled, it utilised a fixed-order design, meaning that all participants received both psilocybin and placebo in a predetermined sequence, potentially introducing order effects.
** Not specified like a RTCs.
*** Do not explicitly state the study’s design. They lack crucial information about the methodology, including whether there was a control group or randomisation.
PICO framework
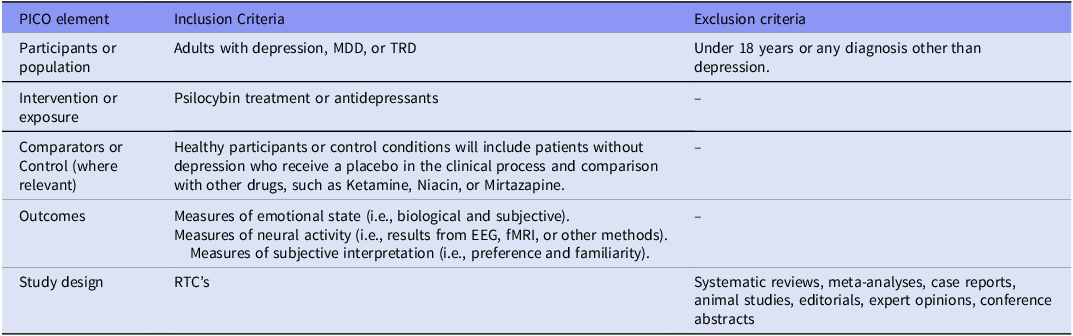
Abbreviations:.
MDD: Major Depressive Disorder; PICO-P- Participants, I- Intervention, C- Comparators or control, O-Outcomes; TRD:Treatment Resistant Depression.
EEG – Electroencephalography; fMRI – Functional Magnetic Resonance Imaging..
Depression measurement tools and quantitative insights
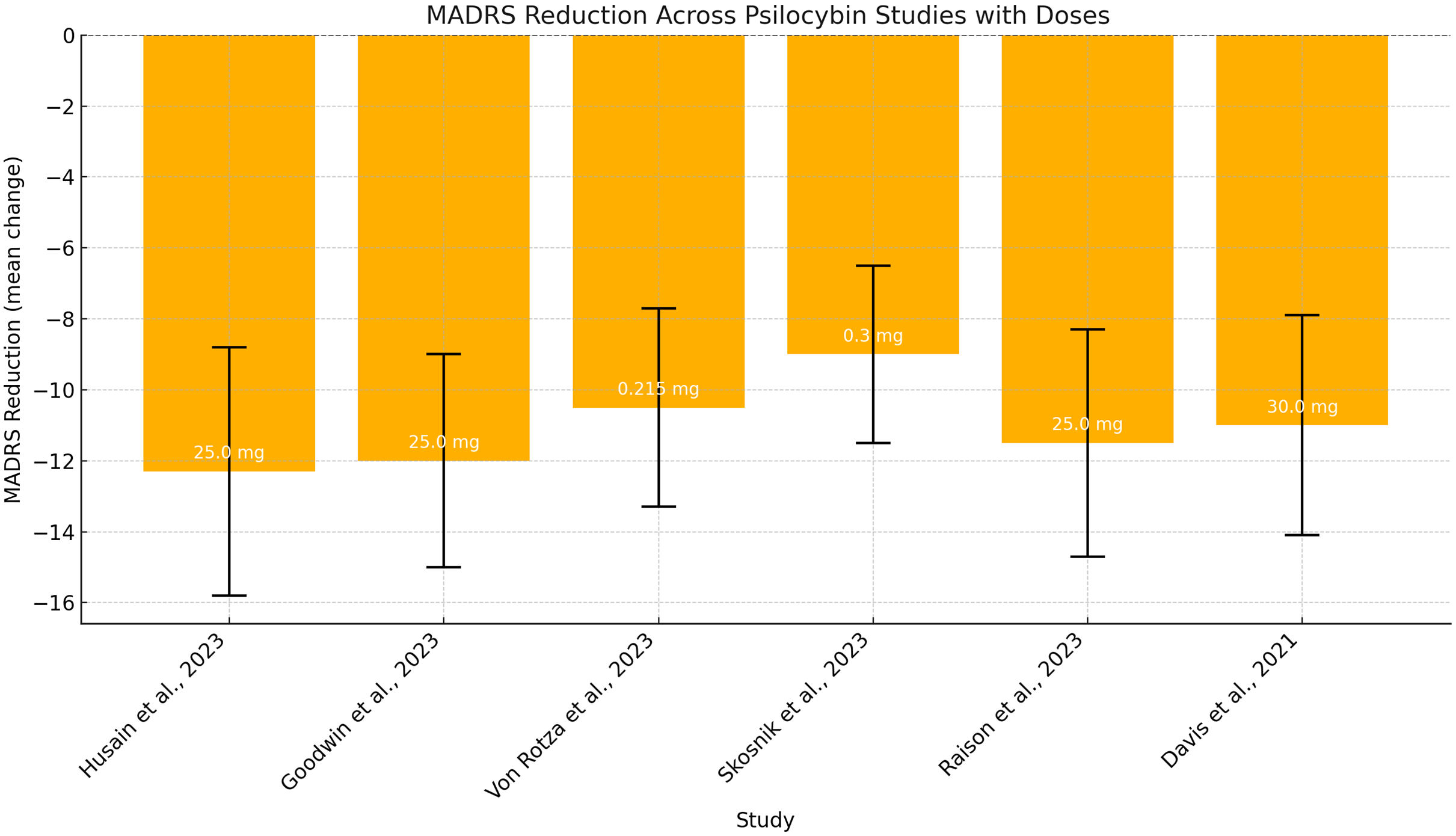
The Montgomery–Åsberg Depression Rating Scale (MADRS) was the most commonly employed tool for evaluating psilocybin’s efficacy, as reported in studies by Goodwin et al., (Reference Goodwin, Aaronson, Alvarez, Arden, Baker, Bennett, Bird, Blom, Brennan, Brusch, Burke, Campbell-Coker, Carhart-Harris, Cattell, Daniel, DeBattista, Dunlop, Eisen, Feifel, Forbes, Haumann, Hellerstein, Hoppe, Husain, Jelen, Kamphuis, Kawasaki, Kelly, Key, Kishon, Knatz Peck, Knight, Koolen, Lean, Licht, Maples-Keller, Mars, Marwood, McElhiney, Miller, Mirow, Mistry, Mletzko-Crowe, Modlin, Nielsen, Nielson, Offerhaus, O’Keane, Páleníček, Printz, Rademaker, van Reemst, Reinholdt, Repantis, Rucker, Rudow, Ruffell, Rush, Schoevers, Seynaeve, Shao, Soares, Somers, Stansfield, Sterling, Strockis, Tsai, Visser, Wahba, Williams, Young, Ywema, Zisook and Malievskaia2022, 2023), Raison et al. (Reference Raison, Sanacora, Woolley, Heinzerling, Dunlop, Brown, Kakar, Hassman, Trivedi, Robison, Gukasyan, Nayak, Hu, O’Donnell, Kelmendi, Sloshower, Penn, Bradley, Kelly, Mletzko, Nicholas, Hutson, Tarpley, Utzinger, Lenoch, Warchol, Gapasin, Davis, Nelson-Douthit, Wilson, Brown, Linton, Ross and Griffiths2023); Skosnik et al. (Reference Skosnik, Cortes-Briones and De Lisi2023); Von Rotza et al. (Reference von Rotza, Endres, Rosser and Grimm2023); Weiss et al. (Reference Weiss, Erritzoe, Giribaldi, Nutt and Carhart-Harris2023); and Husain et al. (Reference Husain, Rehman, Hodsoll and Young2023) (See Fig. 2). Other depression scales used included the GRID-Hamilton Depression Rating Scale (GRID-HAMD) in studies by Davis et al. (Reference Davis, Barrett, May, Cosimano, Sepeda, Johnson, Finan and Griffiths2021) and Gukasyan et al. (Reference Gukasyan, Davis, Barrett, Cosimano, Sepeda, Johnson and Griffiths2022) and the Beck Depression Inventory (BDI) in studies by Gukasyan et al. (Reference Gukasyan, Davis, Barrett, Cosimano, Sepeda, Johnson and Griffiths2022) and von Rotza et al. (Reference von Rotza, Endres, Rosser and Grimm2023).
MADRS score changes following psilocybin administration at different doses (mg/kg) across diferent studies.

Additional measures, such as the Mystical Experience Questionnaire (MEQ) and Emotional Breakthrough Inventory (EBI), provided insights into psilocybin’s psychological mechanisms, highlighting emotional release and transformative experiences as influencing outcomes.
Efficacy of psilocybin in treating depression
Major depressive disorder (MDD)
Two RCTs demonstrated that psilocybin-assisted therapy produced substantial and lasting antidepressant effects in MDD, with large effect sizes observed when comparing high vs. low doses (Davis et al., Reference Davis, Barrett, May, Cosimano, Sepeda, Johnson, Finan and Griffiths2021; Gukasyan et al., Reference Gukasyan, Davis, Barrett, Cosimano, Sepeda, Johnson and Griffiths2022). Studies also showed significant reductions in GRID-HAMD scores over time (Barrett et al., Reference Barrett, Johnson and Griffiths2015; Carbonaro et al., Reference Carbonaro, Johnson and Griffiths2020; Davis et al., Reference Davis, Barrett, May, Cosimano, Sepeda, Johnson, Finan and Griffiths2021).
A trial comparing psilocybin to an active placebo (niacin) reported significantly greater MADRS score reductions in the psilocybin group (−12.3 to day 43; −12.0 to day 8), along with improved functional impairment, though remission rates were comparable (Raison et al., Reference Raison, Sanacora, Woolley, Heinzerling, Dunlop, Brown, Kakar, Hassman, Trivedi, Robison, Gukasyan, Nayak, Hu, O’Donnell, Kelmendi, Sloshower, Penn, Bradley, Kelly, Mletzko, Nicholas, Hutson, Tarpley, Utzinger, Lenoch, Warchol, Gapasin, Davis, Nelson-Douthit, Wilson, Brown, Linton, Ross and Griffiths2023).
A smaller RCT using a 0.215 mg/kg dose reported 58% of participants responding on MADRS and 54% on BDI by day 14, compared to 16% and 12% in the placebo group (von Rotza et al., Reference von Rotza, Endres, Rosser and Grimm2023). However, another RCT comparing psilocybin to escitalopram showed no significant difference in QIDS-SR-16 scores at week 6, although secondary outcomes favoured psilocybin (Carhart-Harris et al., Reference Carhart-Harris, Giribaldi, Watts, Baker-Jones, Murphy-Beiner, Murphy, Martell, Blemings, Erritzoe and Nutt2021).
Treatment-resistant depression (TRD)
One RCT reported a −14.9 point MADRS reduction in TRD patients already on SRIs at week 3, with 42.1% of participants achieving both response and remission. Effects were observed as early as day 2 and sustained over time (Goodwin et al., Reference Goodwin, Aaronson, Alvarez, Arden, Baker, Bennett, Bird, Blom, Brennan, Brusch, Burke, Campbell-Coker, Carhart-Harris, Cattell, Daniel, DeBattista, Dunlop, Eisen, Feifel, Forbes, Haumann, Hellerstein, Hoppe, Husain, Jelen, Kamphuis, Kawasaki, Kelly, Key, Kishon, Knatz Peck, Knight, Koolen, Lean, Licht, Maples-Keller, Mars, Marwood, McElhiney, Miller, Mirow, Mistry, Mletzko-Crowe, Modlin, Nielsen, Nielson, Offerhaus, O’Keane, Páleníček, Printz, Rademaker, van Reemst, Reinholdt, Repantis, Rucker, Rudow, Ruffell, Rush, Schoevers, Seynaeve, Shao, Soares, Somers, Stansfield, Sterling, Strockis, Tsai, Visser, Wahba, Williams, Young, Ywema, Zisook and Malievskaia2022; Goodwin et al., Reference Goodwin, Aaronson, Alvarez, Atli, Bennett, Croal, DeBattista, Dunlop, Feifel, Hellerstein, Husain, Kelly, Lennard-Jones, Licht, Marwood, Mistry, Páleníček, Redjep, Repantis, Schoevers, Septimus, Simmons, Soares, Somers, Stansfield, Stuart, Tadley, Thiara, Tsai, Wahba, Williams, Winzer, Young, Young, Zisook and Malievskaia2023).
Another study found that a 25-mg dose produced the most significant effect (−12.0 at week 3), compared to −5.4 in controls. The 10-mg dose showed a moderate effect (−7.9), which was not statistically significant (Goodwin et al., Reference Goodwin, Aaronson, Alvarez, Arden, Baker, Bennett, Bird, Blom, Brennan, Brusch, Burke, Campbell-Coker, Carhart-Harris, Cattell, Daniel, DeBattista, Dunlop, Eisen, Feifel, Forbes, Haumann, Hellerstein, Hoppe, Husain, Jelen, Kamphuis, Kawasaki, Kelly, Key, Kishon, Knatz Peck, Knight, Koolen, Lean, Licht, Maples-Keller, Mars, Marwood, McElhiney, Miller, Mirow, Mistry, Mletzko-Crowe, Modlin, Nielsen, Nielson, Offerhaus, O’Keane, Páleníček, Printz, Rademaker, van Reemst, Reinholdt, Repantis, Rucker, Rudow, Ruffell, Rush, Schoevers, Seynaeve, Shao, Soares, Somers, Stansfield, Sterling, Strockis, Tsai, Visser, Wahba, Williams, Young, Ywema, Zisook and Malievskaia2022).
A large trial involving 233 TRD patients further supported these findings: the 25-mg group showed statistically significant improvement across measures of depression, anxiety, affect, functioning, and quality of life (Carhart-Harris et al., Reference Carhart-Harris, Muthukumaraswamy, Roseman, Kaelen, Droog, Murphy, Tagliazucchi, Schenberg, Nest, Orban, Leech, Williams, Williams, Bolstridge, Sessa, McGonigle, Sereno, Nichols, Hellyer, Hobden, Evans, Singh, Wise, Curran, Feilding and Nutt2016, 2021; Goodwin et al., Reference Goodwin, Aaronson, Alvarez, Arden, Baker, Bennett, Bird, Blom, Brennan, Brusch, Burke, Campbell-Coker, Carhart-Harris, Cattell, Daniel, DeBattista, Dunlop, Eisen, Feifel, Forbes, Haumann, Hellerstein, Hoppe, Husain, Jelen, Kamphuis, Kawasaki, Kelly, Key, Kishon, Knatz Peck, Knight, Koolen, Lean, Licht, Maples-Keller, Mars, Marwood, McElhiney, Miller, Mirow, Mistry, Mletzko-Crowe, Modlin, Nielsen, Nielson, Offerhaus, O’Keane, Páleníček, Printz, Rademaker, van Reemst, Reinholdt, Repantis, Rucker, Rudow, Ruffell, Rush, Schoevers, Seynaeve, Shao, Soares, Somers, Stansfield, Sterling, Strockis, Tsai, Visser, Wahba, Williams, Young, Ywema, Zisook and Malievskaia2022; Davis et al., Reference Davis, Barrett, May, Cosimano, Sepeda, Johnson, Finan and Griffiths2021; Grob et al., Reference Grob, Danforth, Chopra, Hagerty, McKay, Halberstadt and Greer2011; Griffiths et al., Reference Griffiths, Johnson, Carducci, Umbricht, Richards, Richards, Cosimano and Klinedinst2016; Ross et al., Reference Ross, Bossis, Guss, Agin-Liebes, Malone, Cohen, Mennenga, Belser, Kalliontzi, Babb, Su, Corby and Schmidt2016).
Role of psychotherapy and set/Setting
Multiple studies (Davis et al., Reference Davis, Barrett, May, Cosimano, Sepeda, Johnson, Finan and Griffiths2021; Goodwin et al., Reference Goodwin, Aaronson, Alvarez, Arden, Baker, Bennett, Bird, Blom, Brennan, Brusch, Burke, Campbell-Coker, Carhart-Harris, Cattell, Daniel, DeBattista, Dunlop, Eisen, Feifel, Forbes, Haumann, Hellerstein, Hoppe, Husain, Jelen, Kamphuis, Kawasaki, Kelly, Key, Kishon, Knatz Peck, Knight, Koolen, Lean, Licht, Maples-Keller, Mars, Marwood, McElhiney, Miller, Mirow, Mistry, Mletzko-Crowe, Modlin, Nielsen, Nielson, Offerhaus, O’Keane, Páleníček, Printz, Rademaker, van Reemst, Reinholdt, Repantis, Rucker, Rudow, Ruffell, Rush, Schoevers, Seynaeve, Shao, Soares, Somers, Stansfield, Sterling, Strockis, Tsai, Visser, Wahba, Williams, Young, Ywema, Zisook and Malievskaia2022; Raison et al., Reference Raison, Sanacora, Woolley, Heinzerling, Dunlop, Brown, Kakar, Hassman, Trivedi, Robison, Gukasyan, Nayak, Hu, O’Donnell, Kelmendi, Sloshower, Penn, Bradley, Kelly, Mletzko, Nicholas, Hutson, Tarpley, Utzinger, Lenoch, Warchol, Gapasin, Davis, Nelson-Douthit, Wilson, Brown, Linton, Ross and Griffiths2023; von Rotza et al., Reference von Rotza, Endres, Rosser and Grimm2023; Aaronson et al., Reference Aaronson, van der Vaart, Miller, LaPratt, Swartz, Shoultz, Lauterbach, Suppes and Sackeim2025) emphasise that preparatory and integration therapy enhance both the safety and efficacy of psilocybin treatment. These components typically include structured preparation and post-session integration, which help participants process their experiences and mitigate adverse effects (Swieczkowski et al., Reference Swieczkowski, Kwaśny, Pruc, Gaca, Szarpak and Cubała2025).
Griffiths et al. (Reference Griffiths, Johnson, Carducci, Umbricht, Richards, Richards, Cosimano and Klinedinst2016) and Carhart-Harris et al. (Reference Carhart-Harris, Giribaldi, Watts, Baker-Jones, Murphy-Beiner, Murphy, Martell, Blemings, Erritzoe and Nutt2021) noted that psychological support appears especially important for managing high-dose effects. However, participants receiving active placebos (e.g., low-dose psilocybin or niacin) may still experience physiological sensations that amplify expectancy effects, complicating blinding and possibly inflating placebo responses (Reference Aday, Heifets, Pratscher, Bradley, Rosen and WoolleyAday et al., 2022; Barstowe, A. and Kajonius, P.J. Reference Barstowe and Kajonius2024 ‘Masking influences: a systematic review of placebo control and masking in psychedelic studies’, Journal of Psychoactive Drugs, published online 6 November, pp. 1–11. https://doi.org/10.1080/02791072.2024.2424272 &, ).
Although evidence supports these psychotherapeutic elements, the optimal format, timing, and intensity remain undefined. This highlights the need for standardised interventions within the “set and setting” framework.
Dose–Response variability across diagnoses
Effective psilocybin dosing across patient groups variably complicates interpretation. Meta-analyses (Li et al., Reference Li, Hu, Chen and Zhang2022; Perez et al., Reference Perez, Langlest, Mallet, De Pieri, Sentissi, Thorens, Seragnoli, Zullino, Kirschner, Kaiser, Solmi and Sabé2023) suggest lower doses may alleviate depressive symptoms in secondary depression, especially with comorbid anxiety. In contrast, TRD patients often require higher doses, likely due to pharmacoresistance and neurobiological factors. This dose heterogeneity emphasises the need for individualised treatment protocols and further studies that clarify dose–response relationships by diagnosis.
Involvement of the 5HT2A receptor in psilocybin’s antidepressant effects
The 5-hydroxytryptamine 2A (5-HT2A) receptor is widely recognised as the principal pharmacological target of psilocybin via its active metabolite, psilocin. Psilocin acts as a partial agonist at 5-HT2A receptors, which are abundantly expressed in cortical regions involved in mood regulation, including the prefrontal and posterior cingulate cortices (Nichols, Reference Nichols2016; Ray, Reference Ray2010). Activation of this receptor is considered essential for both the acute psychedelic effects and the therapeutic outcomes observed with psilocybin administration (Vollenweider & Kometer, Reference Vollenweider and Kometer2010).
Direct evidence of 5-HT2A receptor involvement comes from human PET imaging studies. Madsen et al. (Reference Madsen, Fisher, Burmester, Dyssegaard, Stenbæk, Kristiansen, Johansen, Lehel, Linnet, Svarer, Erritzoe, Ozenne and Knudsen2019) demonstrated that oral psilocybin leads to up to 72% 5-HT2A receptor occupancy, with occupancy levels closely correlating with plasma psilocin concentrations and the subjective intensity of the psychedelic experience. Similarly, Vollenweider et al. (Reference Vollenweider, Vollenweider-Scherpenhuyzen, Bäbler, Vogel and Hell1998) showed that pretreatment with ketanserin, a selective 5-HT2A antagonist, blocks the subjective and neurophysiological effects of psilocybin, reinforcing the receptor’s central role.
Although much of the evidence for neuroplasticity-related pathways such as TrkB and mTOR comes from broader psychedelic research, Vargas et al., (Reference Vargas, Dunlap, Dong, Carter, Tombari, Jami, Cameron, Patel, Hennessey, Saeger, McCorvy, Gray, Tian and Olson2023) demonstrated that psilocin, but not serotonin, can activate intracellular 5-HT2A receptors, particularly those located in the Golgi apparatus, leading to structural plasticity. These intracellular receptor populations may play a distinct role in the cellular mechanisms underlying psilocybin’s therapeutic potential.
These findings support the conclusion that activation of 5-HT2A receptors, both membrane-bound and possibly intracellular, is a key mediator of psilocybin’s antidepressant effects. While direct receptor binding was not assessed in most clinical trials reviewed here, the observed clinical efficacy and sustained psychological changes are consistent with known 5-HT2A receptor pharmacology. This aligns with a growing body of evidence suggesting that psilocybin’s therapeutic action arises primarily from 5-HT2A receptor activation and the downstream modulation of neural circuits involved in affect regulation.
Discussion
The therapeutic potential of psilocybin in treating depression has been extensively studied, with more than 10 investigations evaluating its effects on depressive symptoms at multiple time points post-administration. Clinical trials consistently demonstrate that psilocybin induces rapid and significant reductions in depressive symptoms, often following just one or two treatment sessions. For instance, Carhart-Harris et al. (Reference Carhart-Harris, Giribaldi, Watts, Baker-Jones, Murphy-Beiner, Murphy, Martell, Blemings, Erritzoe and Nutt2021) compared psilocybin with escitalopram using the QIDS-SR-16 scale and found that while no statistically significant differences were observed between groups at six weeks, secondary outcomes favoured psilocybin. Notably, 57% of psilocybin-treated patients achieved remission compared to 28% in the escitalopram group. Similarly, Davis et al. (Reference Davis, Barrett, May, Cosimano, Sepeda, Johnson, Finan and Griffiths2021) reported substantial reductions in GRID-HAMD scores, with a mean score decrease from 22.9 at baseline to 8.0 at one week post-treatment, corresponding to a large effect size (Cohen’s d = 2.5).
Psilocybin, a serotonergic compound found in certain mushrooms, has reemerged in psychiatric research over the past decade. From a pharmacokinetic standpoint, psilocybin acts as a prodrug, undergoing rapid dephosphorylation in the gut and liver to produce psilocin, the active metabolite responsible for its central nervous system effects (Hasler et al., Reference Hasler, Grimberg, Benz, Huber and Vollenweider2004; Meshkat et al., Reference Meshkat, Al-Shamali, Perivolaris, Tullu, Zeifman, Zhang and Bhat2025). Psilocin crosses the blood–brain barrier efficiently, reaching peak plasma concentrations within 1.5 to 2 hours after oral ingestion, with a half-life ranging from approximately 1.8 to 4.8 hours depending on dose and study population (Madsen et al., Reference Madsen, Fisher, Burmester, Dyssegaard, Stenbæk, Kristiansen, Johansen, Lehel, Linnet, Svarer, Erritzoe, Ozenne and Knudsen2019; Meshkat et al., Reference Meshkat, Al-Shamali, Perivolaris, Tullu, Zeifman, Zhang and Bhat2025). Psilocin primarily acts as a partial agonist at the 5-HT2A receptor, but also engages 5-HT1A and 5-HT2C receptors, contributing to its complex psychoactive and therapeutic profile (Ray, Reference Ray2010; Nichols, Reference Nichols2016).
Importantly, a distinction must be made between the acute and long-term effects of psilocybin. Acutely, users experience altered perception, time distortion, and emotional shifts – effects that are temporally linked to psilocin’s peak brain activity (Carhart-Harris et al., Reference Carhart-Harris, Leech, Williams, Erritzoe, Abbasi, Bargiotas and Nutt2012). However, Griffiths et al. (Reference Griffiths, Johnson, Carducci, Umbricht, Richards, Richards, Cosimano and Klinedinst2016) and Davis et al. (Reference Davis, Barrett, May, Cosimano, Sepeda, Johnson, Finan and Griffiths2021) report that a single or few doses can lead to long-term improvements in mood, anxiety, and existential distress, persisting for weeks to months. These enduring effects are hypothesised to result from downstream neurobiological mechanisms, including enhanced emotional plasticity, altered default mode network activity, and increased synaptogenesis (Ly et al., Reference Ly, Greb, Cameron, Wong, Barragan, Wilson, Burbach, Zarandi, Sood, Paddy, Duim, Dennis, McAllister, Ori-McKenney, Gray and Olson2018; Carhart-Harris & Friston, Reference Carhart-Harris and Friston2019).
Pharmacodynamically, psilocin’s agonism at the 5-HT2A receptor has been linked to increased cortical glutamate release and enhanced synaptic plasticity, which may underlie the rapid and durable antidepressant effects observed in clinical settings (Vollenweider & Preller, Reference Vollenweider and Preller2020). Recent research has emphasised the relevance of biased agonism, whereby ligands selectively activate specific intracellular signalling pathways at the same receptor. Evidence suggests that psilocin and other psychedelics may preferentially activate β-arrestin-mediated pathways over G protein-dependent signalling at the 5-HT2A receptor, potentially contributing to their unique therapeutic profile (Wacker et al., Reference Wacker, Wang, McCorvy, Betz, Venkatakrishnan, Levit, Lansu, Schools, Che, Nichols, Shoichet, Dror and Roth2017; Wallach et al., Reference Wallach, Cao, Calkins, Heim, Lanham, Bonniwell, Hennessey, Bock, Anderson, Sherwood, Morris, de Klein, Klein, Cuccurazzu, Gamrat, Fannana, Zauhar, Halberstadt and McCorvy2023). This functional selectivity has prompted interest in developing non-hallucinogenic 5-HT2A modulators, such as lisuride or newer analogues, that may retain antidepressant properties without eliciting psychedelic effects (Nichols, Reference Nichols2016; Wulff et al., Reference Wulff, Nichols and Thompson2023).
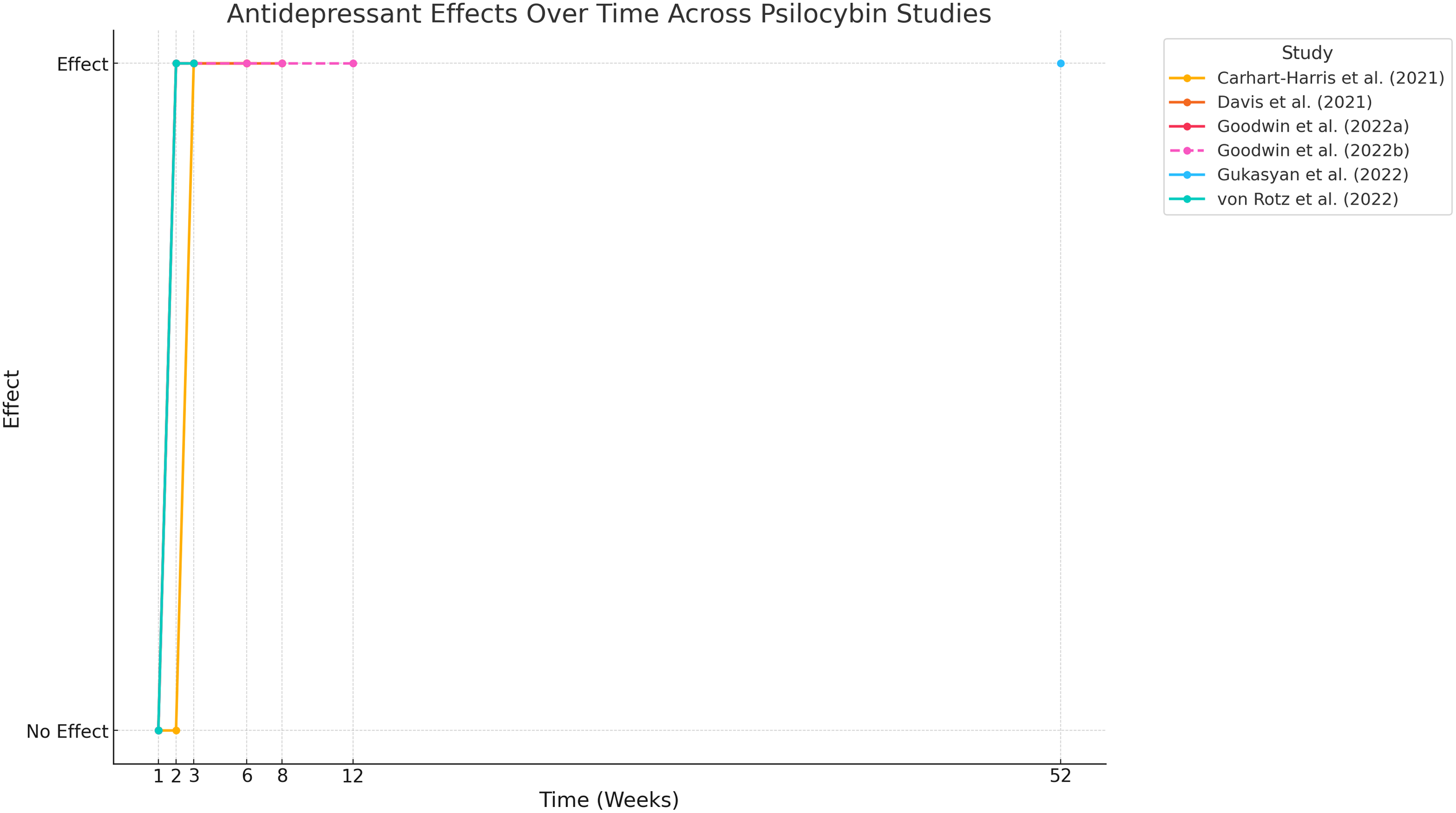
Research also highlights the durability of psilocybin’s effects. Goodwin et al., (Reference Goodwin, Aaronson, Alvarez, Arden, Baker, Bennett, Bird, Blom, Brennan, Brusch, Burke, Campbell-Coker, Carhart-Harris, Cattell, Daniel, DeBattista, Dunlop, Eisen, Feifel, Forbes, Haumann, Hellerstein, Hoppe, Husain, Jelen, Kamphuis, Kawasaki, Kelly, Key, Kishon, Knatz Peck, Knight, Koolen, Lean, Licht, Maples-Keller, Mars, Marwood, McElhiney, Miller, Mirow, Mistry, Mletzko-Crowe, Modlin, Nielsen, Nielson, Offerhaus, O’Keane, Páleníček, Printz, Rademaker, van Reemst, Reinholdt, Repantis, Rucker, Rudow, Ruffell, Rush, Schoevers, Seynaeve, Shao, Soares, Somers, Stansfield, Sterling, Strockis, Tsai, Visser, Wahba, Williams, Young, Ywema, Zisook and Malievskaia2022, 2023) showed significant reductions in MADRS and QIDS-SR-16 scores as early as day two post-treatment, with these effects sustained through weeks three and twelve. Gukasyan et al. (Reference Gukasyan, Davis, Barrett, Cosimano, Sepeda, Johnson and Griffiths2022) extended these findings, reporting large effect sizes (Cohen’s d = 2.4) and high response and remission rates up to 12 months (52 weeks) post-treatment (see Fig. 3).
Time Course of psilocybin antidepressant (follow-up up to 52 weeks).

Furthermore, Raison et al. (Reference Raison, Sanacora, Woolley, Heinzerling, Dunlop, Brown, Kakar, Hassman, Trivedi, Robison, Gukasyan, Nayak, Hu, O’Donnell, Kelmendi, Sloshower, Penn, Bradley, Kelly, Mletzko, Nicholas, Hutson, Tarpley, Utzinger, Lenoch, Warchol, Gapasin, Davis, Nelson-Douthit, Wilson, Brown, Linton, Ross and Griffiths2023) and von Rotz et al., (2022) observed rapid reductions in MADRS scores within 8–14 days and significant improvements compared to placebo.
Beyond depression, psilocybin therapy has shown broader mental health benefits, including reductions in anxiety (Grob et al., Reference Grob, Danforth, Chopra, Hagerty, McKay, Halberstadt and Greer2011; Ross et al., Reference Ross, Bossis, Guss, Agin-Liebes, Malone, Cohen, Mennenga, Belser, Kalliontzi, Babb, Su, Corby and Schmidt2016), improved mindfulness (Madsen et al., Reference Madsen, Fisher, Burmester, Dyssegaard, Stenbæk, Kristiansen, Johansen, Lehel, Linnet, Svarer, Erritzoe, Ozenne and Knudsen2020), and changes in personality traits such as increased Openness and decreased Neuroticism (Weiss et al., Reference Weiss, Magnesa, Gambini, Gurrieri, Annuzzi, Elefante, Perugi and Marazziti2025). These psychological outcomes may play a critical role in sustaining therapeutic gains.
Another topic of interest is the interaction between psilocybin and serotonin reuptake inhibitors (SRIs). Barbut-Siva et al. (Reference Barbut-Siva, Barba, Kettner, Kuc, Nutt, Carhart-Harris and Erritzoe2024) found no significant differences in subjective psychedelic experiences or therapeutic outcomes between individuals on SRIs and those without, suggesting that concurrent SRI use does not negate psilocybin’s benefits. These findings support the potential feasibility of inclusive treatment protocols.
Methodologically, studies have adopted diverse designs and objectives. Husain et al. (Reference Husain, Rehman, Hodsoll and Young2023) examined the combination of psilocybin and risperidone for treatment-resistant depression. Skosnik et al. (Reference Skosnik, Cortes-Briones and De Lisi2023) investigated psilocybin’s effects on neuroplasticity via EEG, while Goodwin et al. (Reference Goodwin, Aaronson, Alvarez, Atli, Bennett, Croal, DeBattista, Dunlop, Feifel, Hellerstein, Husain, Kelly, Lennard-Jones, Licht, Marwood, Mistry, Páleníček, Redjep, Repantis, Schoevers, Septimus, Simmons, Soares, Somers, Stansfield, Stuart, Tadley, Thiara, Tsai, Wahba, Williams, Winzer, Young, Young, Zisook and Malievskaia2023) assessed dosing efficacy. Ley et al. (Reference Ley, Viechtbauer, Hohagen, Baune and Domschke2023) compared subjective effects across several psychedelics under controlled settings.
Finally, the necessity of the psychedelic experience itself for therapeutic benefit remains debated. Studies report a positive correlation between mystical-type experiences and clinical outcomes (Roseman et al., Reference Roseman, Nutt and Carhart-Harris2018; Romeo et al., Reference Romeo, Kervadec, Fauvel, Strika-Bruneau, Amirouche, Bezo, Piolino and Benyamina2025; Menon et al., Reference Menon, Ramamurthy, Venu and Andrade2025 , raising questions about whether these subjective effects are essential or incidental. This has led to increasing exploration of biased signalling mechanisms, as described above, and the development of novel compounds that may uncouple therapeutic effects from hallucinogenic states (Urban et al., Reference Urban, Clarke, Von Zastrow, Nichols, Kobilka, Weinstein, Javitch, Roth, Christopoulos, Sexton, Miller, Spedding and Mailman2007; Cameron et al., Reference Cameron, Tombari, Lu, Pell, Hurley, Ehinger, Vargas, McCarroll, Taylor, Myers-Turnbull, Liu, Yaghoobi, Laskowski, Anderson, Zhang, Viswanathan, Brown, Tjia, Dunlap, Rabow, Fiehn, Wulff, McCorvy, Lein, Kokel, Ron, Peters, Zuo and Olson2021).
Due to the considerable heterogeneity in study designs, populations, interventions, and outcome measures, a meta-analytic approach was not feasible. Instead, this narrative synthesis aimed to capture the variability and evolving trends in psilocybin research. This heterogeneity reflects both the emerging nature of this field and the diverse methodologies employed across trials.
Overall, these findings contribute to a growing evidence base supporting psilocybin’s therapeutic utility. By integrating pharmacokinetic insights, neurobiological mechanisms, and clinical outcomes, this review emphasises the importance of optimising treatment protocols and understanding individual variability. Future directions include identifying optimal dosing strategies, clarifying the role of subjective experiences, and advancing the development of non-hallucinogenic analogues, each of which may further establish psilocybin as a transformative agent in psychiatric care. Importantly, these insights converge on the central role of 5-HT2A receptor activation as a key mechanism underlying psilocybin’s antidepressant effects, reinforcing its relevance as both a therapeutic target and a focal point for future research.
Safety concern
While the therapeutic benefits of psilocybin are increasingly supported by clinical trials, its safety profile warrants careful scrutiny. Although most studies report only mild and transient adverse effects, such as nausea, anxiety, or headaches, there is emerging evidence of more serious risks in some individuals. Notably, Goodwin et al. (Reference Goodwin, Aaronson, Alvarez, Arden, Baker, Bennett, Bird, Blom, Brennan, Brusch, Burke, Campbell-Coker, Carhart-Harris, Cattell, Daniel, DeBattista, Dunlop, Eisen, Feifel, Forbes, Haumann, Hellerstein, Hoppe, Husain, Jelen, Kamphuis, Kawasaki, Kelly, Key, Kishon, Knatz Peck, Knight, Koolen, Lean, Licht, Maples-Keller, Mars, Marwood, McElhiney, Miller, Mirow, Mistry, Mletzko-Crowe, Modlin, Nielsen, Nielson, Offerhaus, O’Keane, Páleníček, Printz, Rademaker, van Reemst, Reinholdt, Repantis, Rucker, Rudow, Ruffell, Rush, Schoevers, Seynaeve, Shao, Soares, Somers, Stansfield, Sterling, Strockis, Tsai, Visser, Wahba, Williams, Young, Ywema, Zisook and Malievskaia2022) documented transient increases in suicidal ideation in a subset of participants, underscoring the need for comprehensive psychiatric screening and continuous monitoring. Moreover, the intensity and emotional depth of the psychedelic experience may pose psychological risks if administered outside of a controlled and supportive setting. Therefore, safe administration requires more than appropriate dosing; it depends critically on trained psychological support, preparation, and integration practices that reduce the likelihood of adverse reactions and enhance therapeutic outcomes.
Challenges and strategies in dose standardisation
Standardising psilocybin dosing presents a complex challenge due to significant interindividual variability in pharmacokinetics. Psilocybin is rapidly converted into psilocin, whose absorption and metabolism are influenced by cytochrome P450 enzymes, particularly CYP2D6 and CYP3A4 (Thomann et al., Reference Thomann, Kolaczynska, Stoeckmann, Rudin, Vizeli, Hoener, Pryce, Vollenweider, Liechti and Duthaler2024; Meshkat et al., Reference Meshkat, Al-Shamali, Perivolaris, Tullu, Zeifman, Zhang and Bhat2025). Peak plasma concentrations occur between 1.5 and 4 hours post-ingestion, with elimination half-lives extending up to 4.8 hours depending on the dose and individual metabolic profiles (Madsen et al., Reference Madsen, Fisher, Burmester, Dyssegaard, Stenbæk, Kristiansen, Johansen, Lehel, Linnet, Svarer, Erritzoe, Ozenne and Knudsen2019). This variability implies that fixed-dose regimens, such as the commonly used 25 mg oral dose, may yield uneven effects across individuals. Pharmacokinetic modelling and identifying predictive biomarkers may help tailor dosing more precisely, optimising safety and efficacy.
Toward safer and more inclusive dosing protocol
Establishing robust and inclusive treatment protocols also involves addressing the concurrent use of antidepressants. Recent findings suggest that selective serotonin reuptake inhibitors (SRIs) do not significantly diminish the subjective or therapeutic effects of psilocybin (Barbut-Siva et al., Reference Barbut-Siva, Barba, Kettner, Kuc, Nutt, Carhart-Harris and Erritzoe2024), supporting the feasibility of inclusive approaches that do not require antidepressant discontinuation. This is particularly important for patients with severe or unstable depression, where withdrawal from SRIs could pose additional risks. Furthermore, research into varied dosing paradigms, from single high-dose sessions to repeated moderate dosing, offers promising avenues for individualised care (Goodwin et al., Reference Goodwin, Aaronson, Alvarez, Atli, Bennett, Croal, DeBattista, Dunlop, Feifel, Hellerstein, Husain, Kelly, Lennard-Jones, Licht, Marwood, Mistry, Páleníček, Redjep, Repantis, Schoevers, Septimus, Simmons, Soares, Somers, Stansfield, Stuart, Tadley, Thiara, Tsai, Wahba, Williams, Winzer, Young, Young, Zisook and Malievskaia2023; Ley et al., Reference Ley, Viechtbauer, Hohagen, Baune and Domschke2023). These emerging strategies, along with ongoing efforts to develop non-hallucinogenic analogues, mark critical steps toward integrating psilocybin into psychiatric practice with greater safety, flexibility, and therapeutic reach.
Conclusion
The data collected demonstrated that there is evidence that indicates promising therapeutic potentials of psilocybin for depression. It also underscores the necessity for further studies. Additional research is needed to fully understand psilocybin’s long-term effects, optimal dosing, safety in diverse populations, and mechanisms of action, especially regarding its interaction with the 5-HT2A receptors. This will enable a more comprehensive assessment of its efficacy and integration into clinical practice for treating depression and other mental health disorders. This body of research marks a significant step towards understanding and potentially revolutionising the treatment of depression, especially in cases where traditional therapies have been ineffective.
Limitations of this study
The study has limitations, as the included studies have small sample sizes, short follow-up periods, and a lack of diversity among the studied populations. These factors restrict the generalisability of the findings across different ethnic, cultural, and age groups. Expanding participant recruitment to include a broader range of populations is crucial for understanding potential variations in the efficacy and safety of psilocybin.
While some studies report sustained effects of psilocybin treatment lasting up to 12 months, most only extend follow-up beyond a few weeks or months. The durability of psilocybin’s antidepressant effects and the potential for relapse have not been fully explored. Future research should prioritise long-term follow-up to assess outcomes and safety over several years.
There is also considerable variability in dosing regimens, session frequency, and adjunctive therapies across studies, complicating direct comparisons. Standardising protocols, including dose ranges and session structures, is necessary to establish consistent findings and optimise clinical applications.
More extensive, diverse, and rigorously designed trials must confirm and expand upon existing findings. The scope of future research should involve a variety of methodologies, a wider range of populations, and long-term outcome assessments to thoroughly evaluate the efficacy and safety of psilocybin in treating depression.
Future directions
Exploring the individual differences in response to psilocybin, focusing on the influence of personality traits, 5-HT2AR binding, and other relevant factors. Studies (Carhart-Harris et al., Reference Carhart-Harris, Muthukumaraswamy, Roseman, Kaelen, Droog, Murphy, Tagliazucchi, Schenberg, Nest, Orban, Leech, Williams, Williams, Bolstridge, Sessa, McGonigle, Sereno, Nichols, Hellyer, Hobden, Evans, Singh, Wise, Curran, Feilding and Nutt2016; Barrett et al., Reference Barrett, Johnson and Griffiths2017b; MacLean et al., Reference MacLean, Johnson and Griffiths2011; Griffiths et al., Reference Griffiths, Johnson, Richards, Richards, Jesse, MacLean, Barrett, Cosimano and Klinedinst2018; Madsen et al., Reference Madsen, Fisher, Burmester, Dyssegaard, Stenbæk, Kristiansen, Johansen, Lehel, Linnet, Svarer, Erritzoe, Ozenne and Knudsen2020) indicate variability in how individuals respond to psilocybin. Future research should investigate the role of personality traits, such as openness, and changes in 5-HT2AR levels as potential moderators of treatment effects. Additionally, it is essential to examine factors like symptom severity, depression subtypes, comorbidities, and prior 5-HT2AR stimulation.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/neu.2025.10039.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.