Introduction
The escalating impacts of global warming have intensified the frequency and severity of extreme weather events, resulting in uneven rainfall distribution and widespread drought stress across agricultural landscapes (Auler et al., Reference Auler, Souza, da Silva Engela, do Amaral, Rossatto, da Silva and Braga2021). Drought, recognized as a major environmental challenge, profoundly affects plant survival, growth and productivity by disrupting both physiological processes and structural development (Abreha et al., Reference Abreha, Enyew, Carlsson, Vetukuri, Feyissa, Motlhaodi and Geleta2022; Bhusal et al., Reference Bhusal, Lee, Han, Han and Kim2020). Wheat, a staple crop critical to global food security, provides a significant portion of daily calorie and protein intake for billions of people worldwide (Guarin et al., Reference Guarin, Martre, Ewert, Webber, Dueri, Calderini, Reynolds, Molero, Miralles and Garcia2022). Its extensive cultivation across diverse agroecological zones underscores its importance in sustaining food systems globally (Ma et al., Reference Ma, Wanga, Jianchao and Beiming2021). As drought stress becomes more prevalent due to climate change, understanding wheat’s adaptive responses to water scarcity is essential for ensuring stable food production and safeguarding global food security.
Rainfed agriculture, which encompasses approximately 80 % of the world’s arable land, serves as a cornerstone of global crop production, contributing around 60 % of total yields (UNESCO, 2009). However, water scarcity remains the primary constraint in these systems, often leading to yield reductions of 40–60 % under drought conditions (Saha et al., Reference Saha, Choyal, Mishra, Dey, Bose and Prathibh2022). In wheat, drought stress disrupts critical developmental stages, including crown root formation, tiller development, booting, anthesis, grain filling, dry matter partitioning and harvest index, all of which impair normal plant growth and productivity (Zivcak et al., Reference Zivcak, Brestic, Sytar, Hossain, Wani, Bhattacharjee, Burritt and Tran2016). Research indicates that drought has a more pronounced effect on plant height, biomass accumulation and overall yield compared to traits like spike number and 1000-grain weight (Ahmad et al., Reference Ahmad, Waraich, Akhtar, Anjum, Ahmad, Mahboob, Abdul Hafeez, Tapera, Labuschagne and Rizwan2018). These adverse effects highlight the urgent need for strategies to mitigate drought-induced losses in rainfed wheat systems.
Supplementary irrigation has emerged as an effective approach to stabilize crop yields in arid and semi-arid regions, where water availability is a limiting factor. By providing targeted water applications during critical growth stages, such as flowering and grain filling, supplementary irrigation mitigates the negative impacts of water deficit stress (Oweis and Hachum, Reference Oweis and Hachum2004). For instance, Jafari et al. (Reference Jafari, Heidari and Khalesro2019) demonstrated that supplementary irrigation during the reproductive phases of wheat significantly increased plant dry weight, seeds per spike, 1000-seed weight and grain yield. Similarly, Liu et al. (Reference Liu, Lin, Wang, Zhang and Wang2022) reported that irrigation at the flowering stage in semi-arid regions enhanced grain yield by 30 % and water use efficiency by 25 % compared to rainfed conditions. These improvements were attributed to enhanced chlorophyll content and reduced production of reactive oxygen species (ROS), which supported reproductive growth and overall plant vigour under water-limited conditions.
Recent advancements in drought mitigation strategies have focused on enhancing wheat’s resilience through seed priming and foliar applications of biostimulants. Seed priming, particularly hydropriming, involves soaking seeds in water to initiate early metabolic processes, thereby improving germination rates, antioxidant defences and osmolyte accumulation (Malko et al., Reference Malko, Khanzada, Xiao, Samo, Li and Dong2022; Saha et al., Reference Saha, Choyal, Mishra, Dey, Bose and Prathibh2022). This technique enhances seedling establishment and drought tolerance, making it a cost-effective and practical solution for rainfed agriculture (Aboutalebian et al., Reference Aboutalebian, Zare Ekbatani and Sepehri2012; Murungu et al., Reference Murungu, Chiduza, Nyamugafata, Clark, Whalley and Finch-Savage2004). Hydropriming, often implemented as on-farm seed priming, involves soaking seeds overnight, surface drying them and sowing them immediately, a method easily adoptable by farmers in resource-constrained settings (Getachew and Jens, Reference Getachew and Jens2019). Studies have shown that seed priming enhances physiological processes such as photosynthesis, nutrient uptake, membrane integrity and osmotic adjustments, all of which contribute to improved drought resistance in wheat (Ceritoglu et al., 2024; Rehman et al., Reference Rehman, Farooq, Naveed, Nawaz and Shahzad2018). According to Manoj et al. (Reference Manoj, Gupta, Nissa, Uttamrao and Gupta2025), seed priming of red kidney bean with 0.25 % chitosan improved plant tolerance to severe drought stress by enhancing chlorophyll a and b contents and increasing leaf relative water content. They also stated that, particularly under drought stress, proline and malondialdehyde contents as well as catalase activity decreased in primed plants, whereas soluble sugar content and superoxide dismutase activity increased.
In parallel, foliar applications of nutrients and biostimulants, particularly amino acids, have gained attention for their role in alleviating abiotic stress in crops. Amino acids, such as methionine and lysine, are vital organic compounds that regulate plant physiological and biochemical processes, enhancing resilience to drought and other stresses (Ali et al., Reference Ali, Rizwan, Waqas, Hussain, Hussain, Liu, Alqarawi, Hashem and Abd_Allah2018; Henderson et al., Reference Henderson, Sanderson and Fowles2025; Maqsood et al., Reference Maqsood, Shahbaz, Kanwal, Kaleem, Shah, Luqman, Iftikhar, Zulfiqar, Tariq, Naveed, Inayat, Muhi Ud Din, Uzair, Khan and Farhat2022). Methionine, a sulphur-containing amino acid and precursor to cysteine and glutathione, plays a critical role in antioxidant defence by enhancing the activity of enzymes like catalase, peroxidase and superoxide dismutase (Merwad et al., Reference Merwad, Desoky and Rady2018; Sadak et al., Reference Sadak, Abd Elhamid and Mahmoud2017). Foliar application of methionine at concentrations of 50–100 mg/l has been shown to increase proline content, photosynthetic pigments and overall plant growth in crops like wheat and flax, thereby mitigating drought-induced damage (Bakry et al., Reference Bakry, El-Bassiouny, Sadak and Younis2018; Maqsood et al., Reference Maqsood, Shahbaz, Kanwal, Kaleem, Shah, Luqman, Iftikhar, Zulfiqar, Tariq, Naveed, Inayat, Muhi Ud Din, Uzair, Khan and Farhat2022). Similarly, lysine metabolism, mediated through the saccharopine pathway, contributes to stress tolerance by regulating osmotic balance and ROS scavenging (Arruda and Barreto, Reference Arruda and Barreto2020). For example, Baqir and Al-Naqeeb (2019) found that foliar spraying of 50 mg/ha L-lysine increased tiller number, seeds per spike and 1000-seed weight in wheat, while Han et al. (Reference Han, Sonmez, Qureshi, Guden, Gangurde and Yol2024) reported that a combination of 50 mg/l amino acids (including methionine and lysine) with zinc improved soybean branching and pod formation.
Foliar applications of amino acids have also been shown to enhance photosynthetic efficiency, leaf area, dry weight and protein content under both stress and non-stress conditions (Dromantienė et al., Reference Dromantienė, Pranckietienė, Šidlauskas and Smalstienė2019; Henderson et al., Reference Henderson, Sanderson and Fowles2025; Sadak et al., Reference Sadak, Sekara, Al-ashkar, Habib-ur-Rahman, Skalicky, Brestic, Kumar, Sabagh and Abdelhamid2022). For instance, Souza et al. (Reference Souza, da Cruz, Bonfá and Magalhães2018) noted that amino acid-based fertilizers stimulated various physiological processes, leading to improved crop yields. Similarly, Kandil and Marie (Reference Kandil and Marie2017) reported that amino acid foliar sprays promoted wheat growth by enhancing biochemical pathways, including antioxidant enzyme activity and proline accumulation. In Arabidopsis, high methionine levels triggered metabolic and transcriptomic responses linked to drought stress and mitochondrial energy metabolism, further underscoring its role in stress mitigation (Cohen et al., Reference Cohen, Israeli, Matityahu and Amir2014).
While seed priming and amino acid applications have been studied individually for their contributions to drought tolerance, their synergistic effects in rainfed wheat systems remain underexplored. Recent reviews suggest that combining seed priming with foliar nutrient applications can amplify biochemical responses, such as enhanced antioxidant enzyme activity, proline accumulation and yield stability under water-limited conditions (Malko et al., Reference Malko, Khanzada, Xiao, Samo, Li and Dong2022; Saha et al., Reference Saha, Choyal, Mishra, Dey, Bose and Prathibh2022). For instance, a study demonstrated that integrating hydropriming with foliar nutrient sprays significantly improved wheat growth indices, yield components and drought tolerance by modulating antioxidant defences and osmolyte levels (Malko et al., Reference Malko, Khanzada, Xiao, Samo, Li and Dong2022). However, specific data on the combined application of hydropriming and amino acid foliar sprays, particularly methionine and lysine, in wheat under rainfed conditions are limited. This gap is particularly relevant in semi-arid regions like Hamedan, Iran, where water scarcity poses a significant challenge to wheat production.
This study aims to address this knowledge gap by evaluating the combined effects of seed priming and amino acid foliar applications on the biochemical characteristics and grain yield of rainfed wheat under supplementary irrigation. By investigating these integrated strategies at various growth stages, the research seeks to provide insights into their potential to enhance drought tolerance and improve crop productivity in water-limited environments. Such findings could offer practical solutions for farmers in semi-arid regions, contributing to sustainable wheat production and global food security.
Materials and methods
Experimental site
The experiment was conducted during the 2020–2021 and 2021–2022 crop years at the Ekbatan Research Station in Hamedan Province. The latitude of the research site is 34°34’ N, its longitude is 48°32’ E and it has an altitude of 1730 m a.s.l. The experiment was conducted in a randomized complete block design with three replications, using a factorial arrangement.
Experimental Design and Treatments
Experimental factors included supplementary irrigation at four levels (sowing, flowering, grain filling and no irrigation) and seed hydropriming (on-farm seed priming) at two levels (primed and unprimed). Additionally, foliar sprays of the amino acids lysine and methionine were applied at the heading stage (Zadoks growth stage 50), while pure water was used as the control. Both amino acids were purchased from Sigma-Aldrich (USA) and used at a concentration of 50 mg/l. Plots measuring 1.2 × 6 m were established. Within each plot, six rows were marked out at 20 cm intervals for sowing. Soil samples were randomly collected from various locations within the field at a depth of 0–30 cm prior to the experiment. These samples were analysed to determine the physicochemical properties of the soil, with the results summarized in Table 1.
Table 1. Physical and chemical characteristics of soil experimental field

One day before each irrigation treatment, the soil moisture content was determined and irrigation was applied up to the field capacity of the experimental soil using Equation (1) (Alizade, Reference Alizade2001).
In Equation (1), d denotes the irrigation water depth (cm), Fc represents the soil moisture content at field capacity (13–15 %), P0 is the soil moisture content prior to irrigation, As is the soil bulk density (1.46 g/cm3) and D refers to the soil depth. For calculating the required irrigation volume, a soil depth of 10 cm was considered at the sowing stage, whereas a depth of 30 cm was used for subsequent growth stages. To convert the water depth to volume in m3/ha, d is multiplied by 100.
For seed hydropriming, seeds were soaked in tap water for 10 hours on the day prior to planting and then sown after surface drying. This procedure is commonly known as on-farm seed priming (Aboutalebian et al., Reference Aboutalebian, Zare Ekbatani and Sepehri2012; Carrillo-Reche et al., 2018; Harris, Reference Harris2006). In addition, for seed disinfection at the time of sowing, the fungicide Vitavax-200 was applied at a rate of 0.2 % (w/w). In this research, the rainfed wheat cultivar Baran was used, which was obtained from the Hamedan Agricultural and Natural Resources Research Center. This cultivar is recommended for cultivation in cold and temperate rainfed regions of Iran. Baran wheat is classified as a rainfed cultivar with a winter growth habit. It exhibits resistance to cold, lodging and grain shattering, and shows relative sensitivity to drought stress.
Cultural practices
Land preparation involved shallow ploughing, disking and application of urea and triple superphosphate fertilizers based on soil analysis results. Drip irrigation was applied using drip tapes. Planting was conducted on November 2 in the first year and November 11 in the second year, with a seeding density of 300 viable seeds/m2.
Data collection
The biochemical traits were assessed using the flag leaf of 10 plants per plot when the crop reached Zadoks growth stage 75 (mid-grain filling stage). Harvests occurred on June 16 in the first year and July 1 in the second year. Grain yield was determined by sampling 3 m2 quadrats from each plot, accounting for border effects. Table 2 displays the weather conditions during the two growing seasons.
Table 2. Weather characteristics during two growing seasons

Statistical analysis
The data were analysed using a combined analysis of variance (ANOVA) in SAS (version 9.4). The F-test was employed to assess statistical significance, and means were compared using Duncan’s multiple range test at a 5 % significance level. Prior to ANOVA, the normality of residuals was confirmed by the Shapiro-Wilk test, and the homogeneity of variances across the two years of the study was verified using Bartlett’s test. Graphs were plotted using Excel.
Evaluation of leaf antioxidant enzymes and proline status
To prepare the enzyme extract for assays, 0.4 g of wet plant tissue from aerial parts (5:1 w/w ratio) was finely ground in a porcelain mortar with 3 ml of 50 mM phosphate buffer (pH 6.8) in an ice bath for 30 minutes per sample. The homogenate was transferred to sterile 2 ml microtubes and left to stand for 10 minutes to ensure complete protein solubilization. The microtubes were then centrifuged at 13 000 rpm for 20 minutes at 4 °C in a refrigerated centrifuge. The supernatants were collected in small sterile vials and stored at −20 °C for subsequent enzyme assays (Gomes and Torres, Reference Gomes and Torres2016).
Catalase (CAT) activity was determined following the method of Alici and Arabaci (Reference Alici and Arabaci2016) by monitoring the consumption of hydrogen peroxide (H2O2). A reaction mixture containing 10 μl of 15 mM hydrogen peroxide and 50 μl of enzyme extract was prepared in 3 ml of 50 mM potassium phosphate buffer (pH 7). The decrease in H2O2 concentration was measured spectrophotometrically at 240 nm for 1 minute following the addition of enzyme extract. The control for the CAT assay was a blank reaction mixture containing 3 ml of 50 mM potassium phosphate buffer (pH 7) and 10 µL of 15 mM H2O2, without the enzyme extract.
To determine the activity of superoxide dismutase (SOD), 1.0 g of fresh leaf samples was homogenized in 3 ml of 50 mM potassium phosphate buffer (pH 7.4) containing 2 mM ethylenediaminetetraacetic acid (EDTA) and 1 % (w/v) polyvinylpyrrolidone. The homogenate was centrifuged at 15,000 rpm for 10 minutes at 4°C (Coban and Baydar, Reference Coban and Baydar2016). The assay mixture for SOD (final volume 1.0 ml) consisted of 901 µl of 50 mM sodium phosphate buffer (pH 7.8), 33 µl of 0.75 mM NBT, 33 µl of 0.12 mM riboflavin and 33 µl of enzyme extract. The SOD assay mixture was then incubated under fluorescent light for 10 minutes at room temperature prior to measuring absorbance at 560 nm using a VIS 7200 spectrophotometer. One unit of enzyme activity was defined as the amount of protein required to inhibit NBT reduction by 50 %, as monitored by absorbance at 560 nm. The control for the SOD assay was a reaction mixture without enzyme extract, containing 934 µl of 50 mM sodium phosphate buffer (pH 7.8), 33 µl of 0.75 mM NBT and 33 µl of 0.12 mM riboflavin (final volume 1.0 ml), exposed to the 10 minutes under fluorescent light as the test samples (Alici and Arabaci, Reference Alici and Arabaci2016).
Proline was quantified using the method described by Bates et al. (Reference Bates, Waldren and Teare1973). Samples (0.5 g) were homogenized in 10 ml of 3 % (w/v) sulfosalicylic acid solution. A 2 ml sample of this homogenate was reacted with 2 ml of glacial acetic acid and 2 ml of fresh acid ninhydrin solution in a test tube for 1 hour at 100 °C. The reaction was terminated by placing the test tube in an ice bath. After vortexing, the homogenate was extracted with 4 ml of toluene and the upper phase was analysed spectrophotometrically at 520 nm. The control for the proline estimation assay was a blank reaction mixture containing 2 ml of distilled water instead of the plant extract, combined with 2 ml of glacial acetic acid and 2 ml of acid ninhydrin reagent. This blank was subjected to the same treatment as the test samples, including incubation in a boiling water bath for 1 hour, cooling in an ice bath and extraction with 4 ml of toluene.
Leaf soluble protein and sugar content analysis
The extraction of soluble proteins was performed as part of the enzyme extract preparation described earlier in section Experimental site. The protein content in the supernatant was measured using the Bradford method (1976), which involves mixing the supernatant with Coomassie Brilliant Blue G-250 dye and measuring absorbance at 595 nm with a spectrophotometer. A standard curve prepared with bovine serum albumin (BSA) was used to calculate protein concentrations.
Soluble sugars (sucrose, fructose and glucose) were extracted from 0.5 g of fresh wheat leaf tissue homogenized in 5 ml of 80 % (v/v) ethanol at room temperature. The homogenate was incubated at 80 °C for 30 minutes and centrifuged at 5 000 rpm for 10 minutes. The pellet was re-extracted with 5 ml of 80 % ethanol, and the combined supernatants were evaporated to dryness at 40 °C under nitrogen. The residue was dissolved in 2 ml of distilled water and filtered through a 0.45 µm membrane. Sugar concentrations were quantified spectrophotometrically at 340 nm using enzymatic assays as described by Wu et al. (Reference Wu, Srivastava and Li2015). The assay was performed in a 1 ml reaction mixture containing 100 mM imidazole-HCl buffer (pH 6.9), 5 mM MgCl2, 0.5 mM NADP+, 1 mM ATP and 50 µl of sugar extract.
Glucose was measured by adding 2 units of hexokinase (HK) and 2 units of glucose-6-phosphate dehydrogenase (G6PDH), monitoring NADPH formation for 5 minutes. Fructose was quantified by adding 2 units of phosphoglucose isomerase (PGI) to the same mixture, and sucrose was measured by adding 10 units of invertase, with absorbance changes recorded at 340 nm for 5 minutes per step at 25 °C. A blank reaction mixture without sugar extract was used for each step. Concentrations were calculated using standard curves for glucose, fructose and sucrose (0–100 µM) and expressed as mg/g fresh weight.
Evaluation of photosynthetic pigments
Photosynthetic pigments were extracted from 0.5 g fresh wheat leaf slices using a 1:1 mixture of 95.5 % acetone and absolute ethanol. The extraction was conducted in darkness for 48 hours. Chlorophyll a and b contents were determined spectrophotometrically (Cary 60 UV spectrophotometer) by measuring absorbance at 663 and 645 nm, respectively, following the method of Arnon (Reference Arnon1949). Carotenoid content was estimated using the method of Wellburn (Reference Wellburn1994) by measuring absorbance at 470 nm. Equations (2) through (5) provide the calculations for chlorophyll a, b, total chlorophyll and carotenoids (Awachare et al., Reference Awachare, Kurian, Upreti and Laxman2018). The pigment concentrations were expressed as mg/g fresh weight.
Where V = Extract volume (ml), W = Fresh weight of leaf tissue (g, and A = Absorbance.
Results
Soluble protein
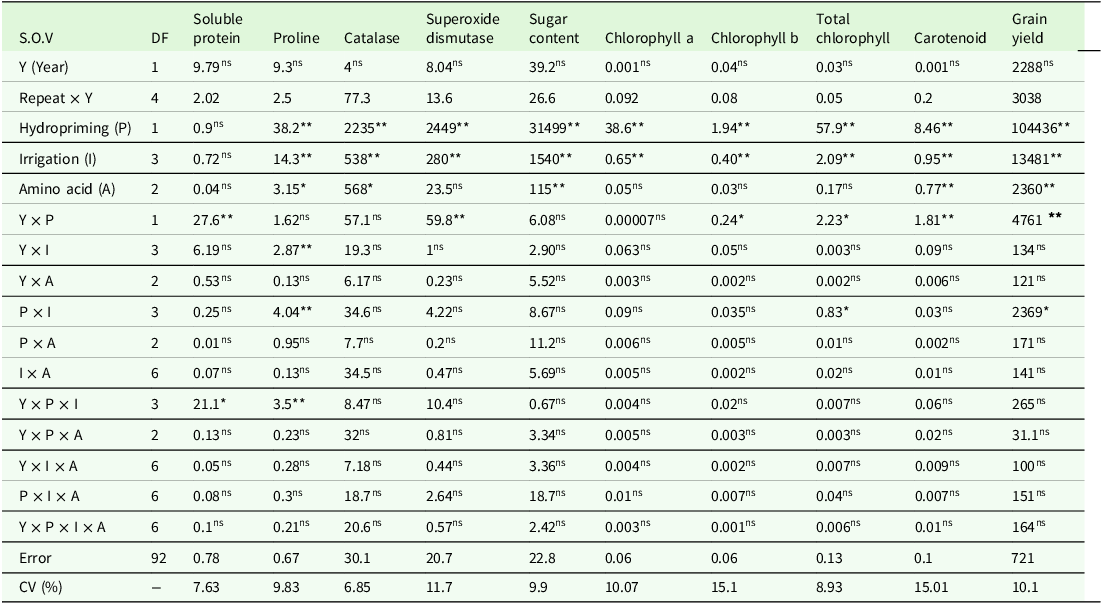
The results of the variance analysis showed that leaf soluble protein was affected by the year × hydropriming interaction and the year × hydropriming × supplementary irrigation triple interaction (Table 3). In both experimental years, the highest leaf soluble protein was observed under non-supplementary irrigation conditions. The lowest leaf soluble protein was also observed with supplementary irrigation at the flowering and grain filling stages. In the second year of the experiment, characterized by an 18 % decrease in rainfall and colder January and February temperatures compared to the first year (Table 2), seed hydropriming resulted in a 5.3 % increase in soluble leaf protein under supplementary irrigation during the grain-filling stage compared with unprimed plants (Figure 1).
Table 3. Analysis of variance of biochemical traits associated with wheat leaf under the influence of investigated factors during two cropping years (2020–2022)

ns,* and** are non-significant, significant at P ≤ 0.05 and P ≤ 0.01, respectively.

Figure 1. Means comparison for year × seed hydropriming × supplementary irrigation on the soluble protein of wheat leaves. Error bars represent standard error. Significant differences between treatments are indicated by different letters (α = 0.05).
Proline
According to the results of the analysis of variance (Table 3), the effects of all three factors (seed hydropriming, supplementary irrigation and foliar application of amino acid) as well as the two-way interactions of year × supplementary irrigation, seed hydropriming × supplementary irrigation and the three-way interaction of year × seed hydropriming × supplementary irrigation had significant effects on the level of proline in wheat leaves.
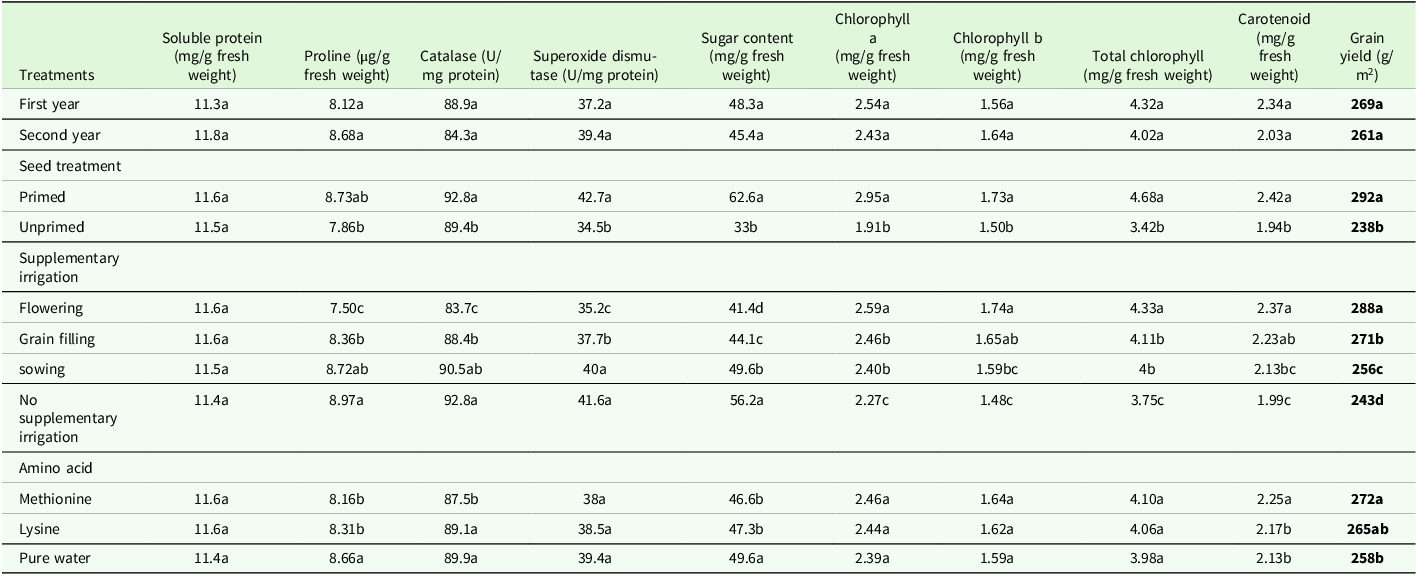
The results of mean comparison indicate that the highest amount of proline was produced from water spray (8.66 µg/g fresh weight), while spraying methionine solution resulted in the lowest proline level (8.16 µg/g fresh weight), which was statistically comparable to lysine spray (Table 4). In the comparison of the mean effects of the three-way interactions of year × seed hydropriming × supplemental irrigation on leaf proline.
Table 4. Means comparison results of the main effects of seed hydropriming, supplementary irrigation and foliar application of amino acid on wheat leaves biochemical characteristics

In each trait, significant differences between treatments are indicated by different letters (α = 0.05).
The highest proline content in both years was observed in unprimed plants under conditions without supplemental irrigation, although it did not differ significantly from treatments with supplemental irrigation at the sowing and grain-filling stages. The lowest proline level also occurred in the second year under conditions of seed hydropriming and irrigation at the flowering stage (Figure 2). Additionally, among the treatments in the first year, at all three irrigation levels during sowing, flowering and grain filling under seed hydropriming conditions, the lowest proline amount was observed (Figure 2).

Figure 2. Means comparison for year × seed hydropriming × supplementary irrigation on the proline wheat leaves content Error bars represent standard error. Significant differences between treatments are indicated by different letters (α = 0.05).
Antioxidant enzymes
Catalase (CAT)
CAT activity was significantly influenced by all three experimental factors (Table 3). Seed hydropriming enhanced catalase activity by 3.8 % relative to unprimed seeds. Irrigation at the flowering and grain-filling stages led to reductions in catalase activity of 9.7 and 4.6 %, respectively, compared to the non-supplementary irrigation treatment (Table 4). Regarding foliar spraying, the highest catalase activity (89.9 U/mg protein) was associated with water spraying, while the lowest activity (87.5 U/mg protein) was observed with methionine spraying (Table 4). Methionine foliar spraying resulted in reductions in catalase activity of 2.6 and 1.7 % compared to pure water and lysine application, respectively (Table 4).
Superoxide dismutase (SOD)
Based on the variance analysis results presented in Table 3, the main effects of seed hydropriming irrigation and the two-way interactions of year × seed hydropriming on the activity of SOD were significant at the 1 % probability level. The results of comparing the main effect of irrigation showed that the highest level of superoxide dismutase enzyme activity was related to no irrigation and sowing irrigation while the lowest enzyme activity was observed in the flowering irrigation treatment (Table 4).
Seed hydropriming significantly enhanced SOD activity by 28 % in the second year, as revealed by the mean comparison between primed and unprimed seeds. This effect was not observed in the first year (Figure 3).

Figure 3. Means comparison for year × seed hydropriming on superoxide dismutase enzyme activity. Error bars represent standard error. Significant differences between treatments are indicated by different letters (α = 0.05).
Sugars content
The results presented in Table 3 indicate that the main effects of seed hydropriming, supplementary irrigation and amino acid foliar spraying on soluble sugar content were significant at the 1 % probability level, while no significant interactions were observed for this trait. Primed seed plants exhibited approximately 89 % higher sugar content compared to unprimed seeds. Conversely, foliar application of amino acids resulted in an average 5.2 % reduction in leaf sugar content. Supplementary irrigation during the flowering, seed-filling and sowing stages led to reductions in soluble sugar content compared to the non-supplementary irrigation treatment, with decreases of 26.3, 21.4 and 11.6 %, respectively (Table 4).
Plant pigments
Chlorophylls
Analysis of variance indicated that seed hydropriming and supplemental irrigation had a significant effect (p < 0.01) on the concentrations of chlorophyll a, chlorophyll b and total chlorophyll. For chlorophyll b and total chlorophyll content, the year × hydropriming interaction was statistically significant at the 5 % probability level. Additionally, for total chlorophyll content, the hydropriming × supplementary irrigation interaction was also statistically significant at the 5 % probability level (Table 3).
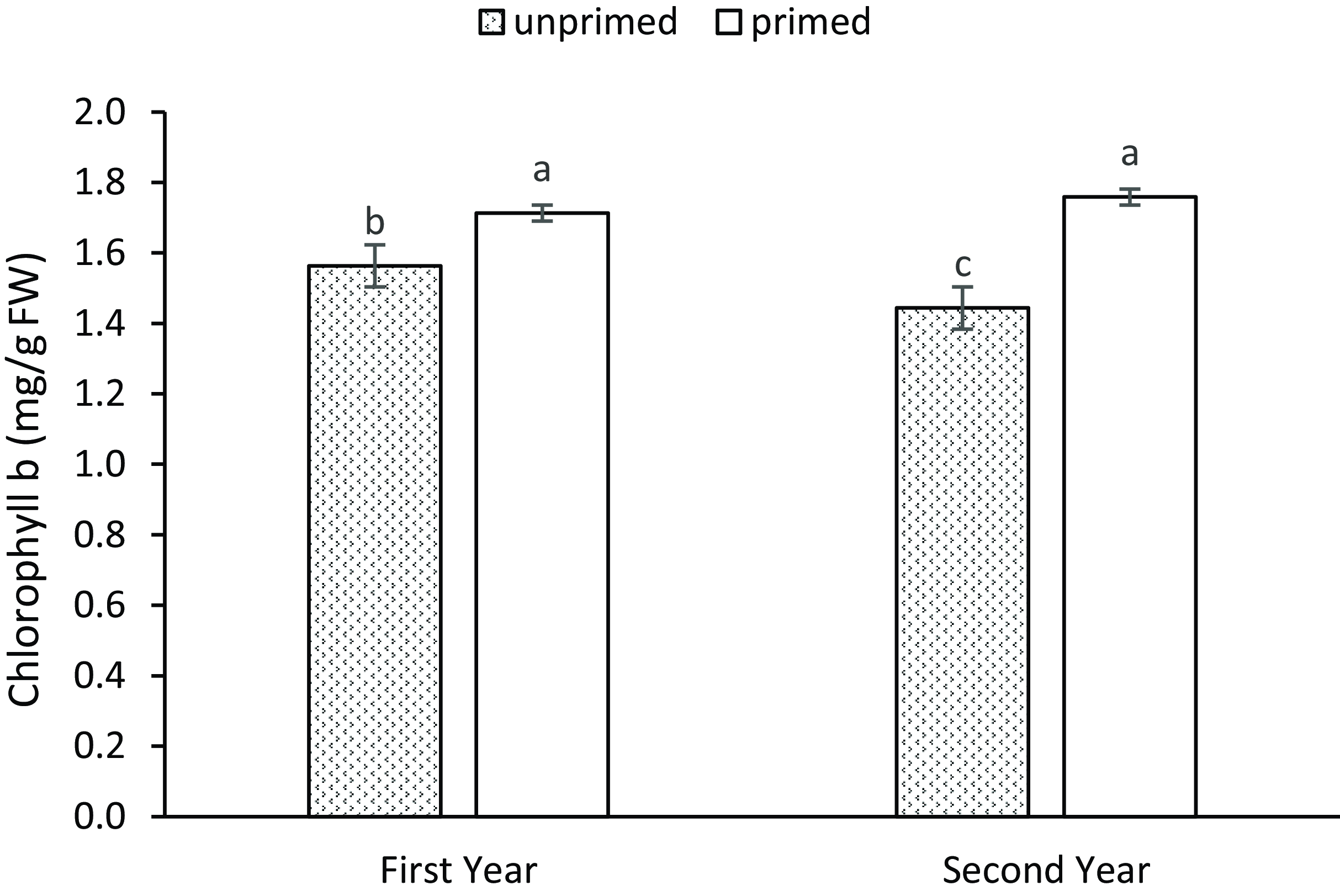
Hydropriming of seeds significantly increased chlorophyll a and b content by 54.4 and 15.3 %, respectively, compared to unprimed seeds. Supplementary irrigation applied during the flowering stage increased chlorophyll a content by 14 % and chlorophyll b content by 17.5 % compared to the non-irrigated control (Table 4). Across both years of the experiment, seed hydropriming significantly enhanced chlorophyll b content, with a more pronounced effect in the second year. However, in the absence of seed hydropriming, chlorophyll b content was higher in the first year (Figure 4).

Figure 4. Means comparison for year × seed hydropriming on leaf chlorophyll b. Error bars represent standard error. Significant differences between treatments are indicated by different letters (α = 0.05).
Based on the experimental results, seed hydropriming significantly enhanced total chlorophyll content across all supplementary irrigation treatments. Notably, the most substantial increase was observed in the supplementary irrigation treatment applied during the flowering stage (39.1 %) (Figure 5).

Figure 5. Means comparison for supplementary irrigation × seed hydropriming on total leaf chlorophyll. Error bars represent standard error. Significant differences between treatments are indicated by different letters (α = 0.05).
Carotenoid
The results of analysis of variance showed that among the investigated factors, the main effects of seed hydropriming, supplementary irrigation and foliar spraying and the interaction effect of year × seed hydropriming were significant (Table 3).
The results of comparing the main effect of foliar spraying showed that the highest amount of carotenoids with an average of 2.25 mg/g of fresh weight was related to methionine foliar spraying and the lowest amount was related to water foliar spraying with an average of 2.13 mg/g of fresh weight (Table 4). The highest carotenoid content was observed in irrigation treatments during flowering and grain filling, with carotenoid levels during flowering irrigation being 19 % higher than non-supplemented irrigation treatments (Table 4).
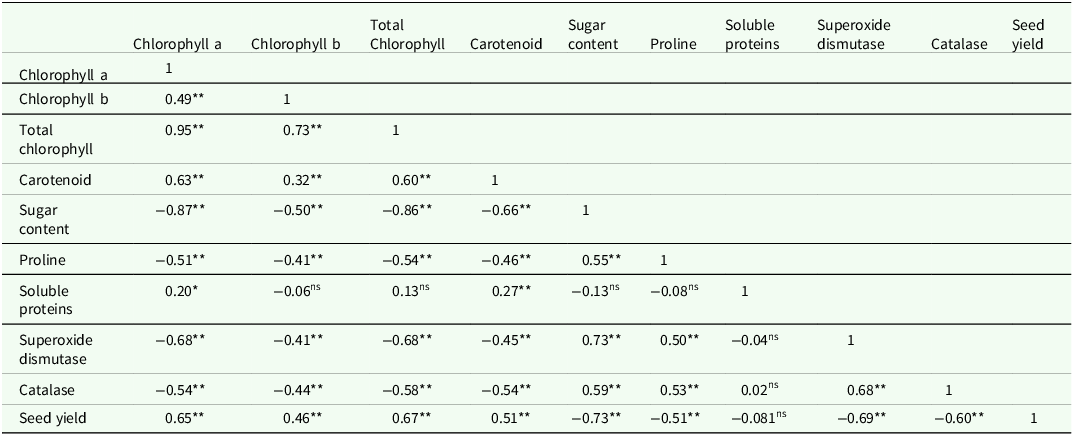
According to (Figure 6), in the second year under seed hydropriming conditions, the highest carotenoid content was observed. However, in the same year, unprimed seeds resulted in a 17.3 % reduction in carotenoid content, whereas this reduction was only 4.9 % in the first year (Figure 6). The table Pearson correlation coefficients shows that the performance has a linear relationship with all the investigated indicators (Table 5).

Figure 6. Means comparison for year × seed hydropriming on leaf carotenoids. Error bars represent standard error. Significant differences between treatments are indicated by different letters (α = 0.05).
Table 5. Pearson correlation coefficients between measured traits

ns,* and** are non-significant, significant at P ≤ 0.05 and P ≤ 0.01, respectively.
Grain yield
Analysis of variance showed that grain yield was significantly affected by all main effects and the two-way interactions of year × seed hydropriming and seed hydropriming × supplementary irrigation at the1 and 5 % probability level (Table 3).
The application of methionine increased grain yield by 5.4 % compared to water spray (Table 4). Seed hydropriming led to a significant 28.6 % increase in grain yield in the second year of the experiment, as opposed to no significant difference found in the first year (Figure 7). Seed hydropriming significantly enhanced grain yield under all supplementary irrigation regimes, with the highest yield of 324.7 g/m² achieved when irrigation was applied at flowering.

Figure 7. Means comparison for year × seed hydropriming on grain yield. Error bars represent standard error. Significant differences between treatments are indicated by different letters (α = 0.05).
Seed hydropriming significantly enhanced grain yield by 28.5, 17.9, 21 and 22.7 % when applied at the flowering, grain filling, sowing and no supplementary irrigation stages, respectively (Figure 8). Primed seeds appear to germinate faster and root better, thus being more successful in absorbing water and nutrients. Priming activates drought resistance mechanisms (such as proline accumulation and antioxidant enzymes). It seems that supplemental irrigation and seed hydropriming ultimately increase yield through their effects on yield components.

Figure 8. Means comparison for supplementary irrigation × seed hydropriming on grain yield. Error bars represent standard error. Significant differences between treatments are indicated by different letters (α = 0.05).
Correlation among measured traits
All measured traits of sugar content, proline, superoxide dismutase and catalase demonstrated significant negative correlations with chlorophyll a, b, total chlorophyll and carotenoid contents. Furthermore, grain yield was negatively correlated with sugar content, proline, superoxide dismutase and catalase levels. However, positive correlations were generally observed among other measured traits (Table 5).
Discussion
The present study provides evidence that seed hydropriming, supplementary irrigation and foliar methionine application can substantially improve the physiological and agronomic performance of rainfed wheat under semi-arid conditions. While each treatment individually contributed to drought mitigation, their combined use produced the strongest positive effects on photosynthetic pigments, antioxidant responses, osmotic regulation and ultimately grain yield. These results highlight the potential of integrating simple and low cost practices to stabilize wheat productivity in water-limited environments.
Seed hydropriming was consistently effective in enhancing drought resilience. Primed seeds showed greater soluble sugar accumulation, higher chlorophyll a and b, increased carotenoid content and improved antioxidant enzyme activity, particularly superoxide dismutase (SOD). The beneficial effects were more pronounced in the second year, when rainfall was reduced by 18 % and winter temperatures were colder, suggesting that priming benefits are amplified under harsher conditions. Similar findings have been reported in previous studies, where hydropriming improved osmotic adjustment, accelerated seedling emergence and increased activity of antioxidant enzymes in cereals (Hussain et al., 2021; Marthandan et al., Reference Marthandan, Geetha, Kumutha, Renganathan, Karthikeyan and Ramalingam2020; Saha et al., Reference Saha, Choyal, Mishra, Dey, Bose and Prathibh2022). Enhanced chlorophyll accumulation in primed plants in this study is in line with reports that priming improves nutrient uptake, particularly phosphorus, which is essential for porphyrin and chlorophyll biosynthesis (Manoj et al., Reference Manoj, Gupta, Nissa, Uttamrao and Gupta2025; Tariq et al., Reference Tariq, Pan, Olatunji, Graciano, Li, Li, Song, Sun, Wu, Dakhil, Sun and Zhang2019). Recent metabolomic analyses also indicate that hydropriming alters the expression of hundreds of metabolites, influencing osmolyte production and antioxidant pathways (Zhang et al., Reference Zhang, Zhu, Wang, Sun, Tian, Chen, Mou, Zhang, Wei, Fang, Ravenscroft, O’Connor, Chang and Yan2023). Thus, the present findings support the idea that priming provides a metabolic ‘head start’ that strengthens physiological responses to subsequent drought stress.
The role of supplementary irrigation was also clearly demonstrated. Irrigation at flowering produced the greatest benefits, increasing total chlorophyll by 39 % and achieving the maximum grain yield of 324.7 g/m² in combination with primed seeds. These results are consistent with the understanding that the reproductive stage is highly sensitive to drought and water supply during anthesis ensures successful pollination, assimilate translocation and grain filling (Lin et al., Reference Lin, Wang, Gu, White, Han, Zhou and Jin2016; Mohammadi, Reference Mohammadi2024). Earlier work by Jafari et al. (Reference Jafari, Heidari and Khalesro2019) similarly showed that supplementary irrigation at reproductive phases significantly enhanced wheat yield components. In the present study, irrigation also reduced leaf proline and soluble sugar levels, indicating a lower degree of osmotic stress. This agrees with findings that supplemental irrigation mitigates the need for osmolyte accumulation under drought (Bhusal et al., Reference Bhusal, Lee, Han, Han and Kim2020; Zivcak et al., Reference Zivcak, Brestic, Sytar, Hossain, Wani, Bhattacharjee, Burritt and Tran2016). Moreover, improvements in pollen viability and reproductive efficiency under flowering irrigation, as reported in wheat and other cereals, may partly explain the observed yield gains (Arathi and Smith, Reference Arathi and Smith2023; Dong et al., Reference Dong, Zheng, Liu, Able, Yang, Zhao, Zhang, Qiao, Wang and Liu2017). Together, these results confirm that flowering is the critical stage at which supplementary irrigation maximizes returns on limited water resources.
Foliar application of methionine further contributed to drought mitigation, mainly by enhancing carotenoid levels and increasing grain yield by 5.4 % compared to the control. Although catalase activity was slightly reduced, this appears to reflect a shift from enzymatic to non-enzymatic antioxidant mechanisms. Methionine, being a sulphur containing amino acid, serves as a precursor for cysteine and glutathione, both of which are central to redox regulation (Maqsood et al., Reference Maqsood, Shahbaz, Kanwal, Kaleem, Shah, Luqman, Iftikhar, Zulfiqar, Tariq, Naveed, Inayat, Muhi Ud Din, Uzair, Khan and Farhat2022; Sadak et al., Reference Sadak, Abd Elhamid and Mahmoud2017). The increased carotenoid content observed in this study aligns with reports that methionine stimulates metabolic pathways leading to higher antioxidant and photoprotective pigments, which help to maintain photosystem stability under stress (Bakry et al., Reference Bakry, El-Bassiouny, Sadak and Younis2018; Perez-Galvez et al., Reference Perez-Galvez, Viera and Roca2020; Rosenfeld et al., Reference Rosenfeld, Yurina and Vasilyeva2023). The positive correlation between carotenoid content and grain yield in the present study further supports the notion that enhanced photoprotection contributes directly to productivity. Conversely, the negative correlation between catalase activity and yield suggests that plants relying more heavily on non-enzymatic antioxidants, such as carotenoids, may achieve higher efficiency under methionine application (Shahid et al., Reference Shahid, Kausar, Zahra, Hafeez, Raza and Ashraf2023). These findings highlight methionine as a useful foliar treatment for improving metabolic balance and yield stability in rainfed wheat.
The interactions among hydropriming, irrigation and methionine spraying revealed a clear synergistic effect. Hydropriming strengthened the baseline physiological capacity of plants, supplementary irrigation at flowering alleviated stress during the most sensitive reproductive stage and methionine enhanced photoprotection and antioxidant balance. Together, these treatments maximized chlorophyll and carotenoid contents and delivered the highest yields. Such synergies confirm previous observations that combining seed priming with foliar biostimulants can amplify biochemical and physiological responses under drought (Malko et al., Reference Malko, Khanzada, Xiao, Samo, Li and Dong2022; Saha et al., Reference Saha, Choyal, Mishra, Dey, Bose and Prathibh2022). Manoj et al. (Reference Manoj, Gupta, Nissa, Uttamrao and Gupta2025) reported that increasing drought stress led to higher levels of free amino acids, including lysine and methionine, in red bean seeds primed with 0.25 % chitosan, which exhibited greater drought tolerance. This finding may be consistent with the synergistic effects of seed priming and amino acid foliar application observed in the present study. By integrating these simple techniques, farmers can exploit complementary mechanisms that collectively buffer yield against environmental variability.
Year dependent differences observed in this study further highlight the adaptive value of these treatments. Hydropriming, for example, had a stronger effect on SOD activity and yield in the second year, while the impact of supplementary irrigation on reducing proline was also more evident under drier conditions. These findings align with the concept of stress memory, whereby plants previously exposed to priming or stress conditions display heightened responses to subsequent stress events (Auler et al., Reference Auler, Souza, da Silva Engela, do Amaral, Rossatto, da Silva and Braga2021). This suggests that the integrative approach tested here is robust across different climatic scenarios and particularly valuable in years of higher stress intensity.
Overall, the findings of this study demonstrate that hydropriming, supplementary irrigation and methionine foliar spraying each contribute unique benefits to drought stress mitigation in wheat and their combined application results in the most effective improvement of growth, biochemical function and yield. By enhancing antioxidant defences, maintaining osmotic balance, preserving photosynthetic pigments and securing reproductive success, these strategies provide an integrated framework for stabilizing wheat productivity under rainfed conditions.
Conclusion
This study demonstrated that seed hydropriming, supplementary irrigation and foliar application of methionine significantly enhanced physiological traits, antioxidant responses and yield stability of wheat under rainfed conditions. Hydropriming improved osmotic adjustment and antioxidant activity, particularly under severe drought, while supplementary irrigation at flowering proved to be the most effective stage for securing reproductive success and grain filling. Foliar methionine application increased carotenoid accumulation, improved photoprotection and contributed to yield enhancement. The combined application of these practices showed synergistic effects, maximizing chlorophyll retention, oxidative balance and productivity. These findings suggest that integrating simple and cost-effective strategies can sustainably improve wheat performance in semi-arid rainfed regions.
Author contributions
N. Hojati Fahim and M. A. Aboutalebian planned and designed the research, analysed data through consultation with M. Chaichi. All authors reviewed and edited the final manuscript. Supervision of the research was done by M. A. Aboutalebian.
Funding statement
This work was funded by the Bu-Ali Sina University as a part of Core Institutional Grant.
Competing interests
The authors declare no competing interests.
Ethical standards and consent to participate
This study did not involve any human participants or animals.