Introduction
Nosocomial infections (NIs) are serious public health issues, causing patient suffering, increased hospitalisation costs, longer hospital stays, elevated mortality, and significant burdens on patients, families, and society [Reference Allegranzi1]. NIs vary significantly across regions and hospitals. A U.S. nationwide analysis revealed that NIs incur additional costs of US$7.2–14.9 billion annually [Reference Forrester, Maggio and Tennakoon2]. In Africa, NIs cause up to US$13 billion losses per year, representing approximately 1.14% of the gross domestic product (GDP) [Reference Hutton3]. In China, a study across 68 hospitals revealed that NIs increase medical costs and extend hospital stays [Reference Jia4].
Orthopaedic inpatients face increased risks due to limited mobility and prolonged bed rest [Reference Chen5]. Most patients undergo surgery, increasing the chance of infection and sepsis [Reference Rinke6]. Orthopaedic surgeries are intricate and time-consuming, prolonging the exposure of sterile tissues and increasing the likelihood of NIs [Reference Bayissa7]. Once infected, patients may need implant removal and multiple surgeries, which can result in serious consequences such as disability or death. Existing research has focused mainly on general patient populations [Reference Lv8] or specific groups such as surgical [Reference Xiong, Wang and Hanafi9], ICU [Reference Mergulhão10, Reference Osme11], and multidrug-resistant patients [Reference Puchter12]. However, studies on orthopaedic patients in China remain limited. Given their unique clinical profile, investigating the NI burden in this population is crucial to support prevention strategies, resource planning, and policy-making. This will help draw attention to the prevention and control of NIs in orthopaedic patients, provide an evidence-based foundation for hospital managers and policymakers, facilitate reasonable resource allocation, and enable precise prevention and control measures.
In China, hospital expenses are primarily covered by a multilayered health insurance system. The main component is national basic medical insurance (BMI), which includes urban employee basic medical insurance (UEBMI) for urban employees and retirees and urban–rural resident basic medical insurance (URRBMI) for children, students, older adults without formal employment, and other urban and rural residents who are not eligible for UEBMI. Contribution levels and benefit packages also differ between these schemes: UEBMI is funded by payroll contributions from employers and employees and generally provides higher reimbursement rates and more generous benefits, while URRBMI is financed mainly by government subsidies and flat-rate individual contributions and offers a more basic level of coverage [Reference Yip13]. The 2022 National Healthcare Security Administration statistical bulletin [14] stated that 1.34592 billion people (over 95% of the population) were enrolled in basic medical insurance. Of these, 362.43 million (26.9%) had UEBMI, and 983.49 million (73.1%) had URRBMI. Additionally, more residents are purchasing supplementary commercial health insurance for extra protection.
The use of diagnosis-related groups (DRGs) is a case classification technique based on disease type, treatment method, and individual characteristics. Cases with similar clinical processes and resource utilisation are grouped together, balancing the complexity and severity of diseases among patients from different departments or hospitals. Considering the close relationships between NIs and primary diseases, treatment methods, and underlying diseases, when the burden of NIs in orthopaedic patients is analysed, comparing the differences in hospitalisation duration and costs between nosocomial-infected and nonnosocomial-infected patients within the same DRG can effectively correct for potential biases. This allows for a more scientific quantification of the additional costs and hospitalisation days caused by NIs.
This study was conducted from 2022 to 2024 at the China National Orthopaedic Medical Center, a Grade III A hospital located in Beijing. The centre comprises eight orthopaedic subspecialties, including spine, hand, sports injury, general orthopaedics, paediatric orthopaedics, trauma, bone oncology, and foot and ankle surgery. The hospital has more than 2,000 beds and performs approximately 40,000 orthopaedic surgeries annually, providing a reliable data source for this study. From 2022 to 2024, the most common DRGs were IC29 (joint arthroplasty), IC39 (joint reconstruction), and IE39 (lower limb bone surgeries excluding femur), with per-episode operation costs of US$5919, US$5354, and US$4413, respectively, and a median hospitalisation duration of 7 days for each group (Supplementary Figure 1).
On the basis of this background, this study aims to systematically evaluate the economic and time burdens of NIs among orthopaedic patients, with a focus on identifying high-burden DRGs and infection sites to determine high-risk populations. Additionally, the aim of this study is to estimate the preliminary national economic burden of NIs among orthopaedic inpatients through scenario-based sensitivity analysis. The findings are expected to provide evidence-based support for promoting precise infection prevention and control strategies and optimising resource allocation in orthopaedic care.
Methods
Study design and setting
A nested case–control study was performed at a Grade III A hospital located in Beijing, China. This study was approved by the Ethics Committee of our hospital (Approval No. K2024381), and all methods were in accordance with the Declaration of Helsinki, including a waiver of informed consent.
Study participants
The study cohort consisted of inpatients admitted to any orthopaedic department of the study hospital between 1 January 2022 and 31 December 2024. Eligible patients were selected according to the inclusion and exclusion criteria.
The inclusion criteria were as follows: (1) admission to one of the following orthopaedic departments: spinal surgery, traumatic orthopaedics, general orthopaedics, hand surgery, bone oncology, sports medicine, paediatric orthopaedics, or foot and ankle surgery. (2) Having a DRG code assigned under the National Medical Security Diagnosis-related Groups Refinement (Version 2.0). (3) Admission date falling within the study window (1 Jan 2022–31 Dec 2024).
The exclusion criteria were as follows: (1) Length of stay ≤48 h. (2) The principal diagnosis was not associated with surgical procedures, as determined by the International Classification of Diseases, 10th Revision (ICD-10) coding system. (3) Incomplete core data, defined as any missing values such as medical record number, age, sex, principal diagnosis, DRG code, admission and discharge dates, and hospitalisation expenses.
Selection of cases and controls
NIs were defined as infections acquired in hospitals, which included infections acquired during hospitalisation and those contracted in the hospital but manifesting after discharge, such as some surgical site infections (SSIs). The follow-up period for SSIs was 30 days for those without implants and 90 days for those with implants.
We defined a case as any inpatient who acquired infections during hospitalisation and follow-up. All reported NI cases are reviewed and confirmed by nosocomial infection management staff on the basis of diagnostic criteria established by the Ministry of Health of the People’s Republic of China [15]. The remaining cohort of inpatients who did not acquire NIs was used as controls.
To more accurately assess the economic and time burden of NIs, 1:3 case–control matching was conducted on the basis of age (±5 years), sex (male or female), DRG code, payment method, and length of hospital stay prior to infection (LOSprior), as previous studies have indicated that these factors influence hospitalisation and costs [Reference Ali16, Reference Heister, Kaier and Wolkewitz17].
The LOSprior refers to the period when inpatients are at risk of acquiring an infection but have not yet acquired it. Considering the time-dependent nature of hospital-acquired infections, avoiding the bias of the LOSprior was necessary. In the case group, the LOSprior was the period from admission to NI occurrence, whereas in the control group, it was the period from admission to discharge.
Data source and collection
DRG information, NI information, and medical records of all orthopaedic inpatients between 2022 and 2024 were collected from the Beijing Inpatient Medical Service Evaluation Platform, the Beijing Hospital Infection Monitoring and Management System, and the Hospital Information System (HIS) respectively. The three databases were matched using the patient’s medical record number and discharge date.
The medical records from the HIS included medical record number, age, sex, principal diagnosis, admission and discharge dates, and hospitalisation expenses. These expenses were further categorised into medical, nursing, technology, administrative, consumable, and medical costs.
Cost calculation and national economic burden analysis
We focused on the direct medical costs (prior to insurance reimbursements or patient copayments) associated with NIs from the perspective of the healthcare system, excluding indirect costs.
With a nested case–control study design, we calculated the direct economic burden of NIs at the China National Orthopaedic Medical Centre. We subsequently conducted a national economic burden analysis under various scenarios.
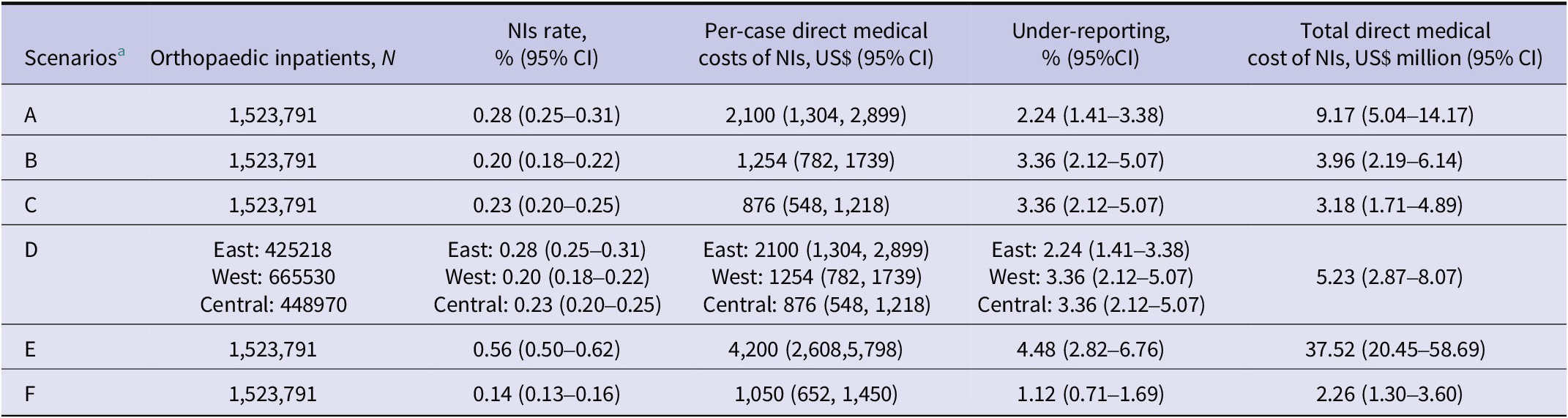
Considering the impact of regional disparities on economic burden, this study estimated the national economic burden of NIs among orthopaedic inpatients through scenario-based sensitivity analysis. The principal approach entailed utilising the direct economic cost per case and the incidence of NIs identified in this study as the foundational baseline. These figures were subsequently adjusted using ratios of economic burdens associated with NIs as reported in diverse regional studies within the literature. In conjunction with nationally reported inpatient statistics from orthopaedic hospitals [18] and documented underreporting rates of NIs [Reference Cao, Wang and Liu19], an analysis of the national direct economic burden of NIs was conducted under various scenarios. The detailed calculation method is provided in the Supplementary Material.
Statistical methods
Statistical analysis of the data was performed using SPSS (version 22.0) and R (version 4.5.0). Descriptive statistics were calculated using the means (standard deviations, SDs) or medians (interquartile ranges, IQR) for continuous variables and frequency counts (percentage, %) for categorical variables, as appropriate.
To determine the significance of differences, t tests were used for normally distributed data, the Wilcoxon rank-sum test was used for nonnormally distributed data, and the χ2 test was used for categorical variables. Median differences with 95% confidence intervals were calculated using the Hodges–Lehmann method. The growth rate and development rate were calculated using geometric mean methods. Statistical significance was set at P < 0.05.
Costs were recorded in Renminbi (RMB) and converted to U.S. dollars using the average RMB per US$ exchange rate for each year (2022: 6.7290; 2023: 7.0809; 2024: 7.1957; USD = RMB ÷ rate). To express all amounts in constant 2022 U.S. dollars, costs from 2023 and 2024 were discounted at 3% per year (i.e., divided by 1.03 and 1.032 respectively).
Results
Patient characteristics of the cohort and matching
From January 2022 to December 2024, a total of 145,232 orthopaedic inpatients were admitted to the hospital. In accordance with the inclusion and exclusion criteria, 120,764 inpatients were included in the study cohort. On the basis of predefined matching criteria, 321 patients were successfully matched at a 1:3 ratio with 916 controls (Supplementary Figure 2).
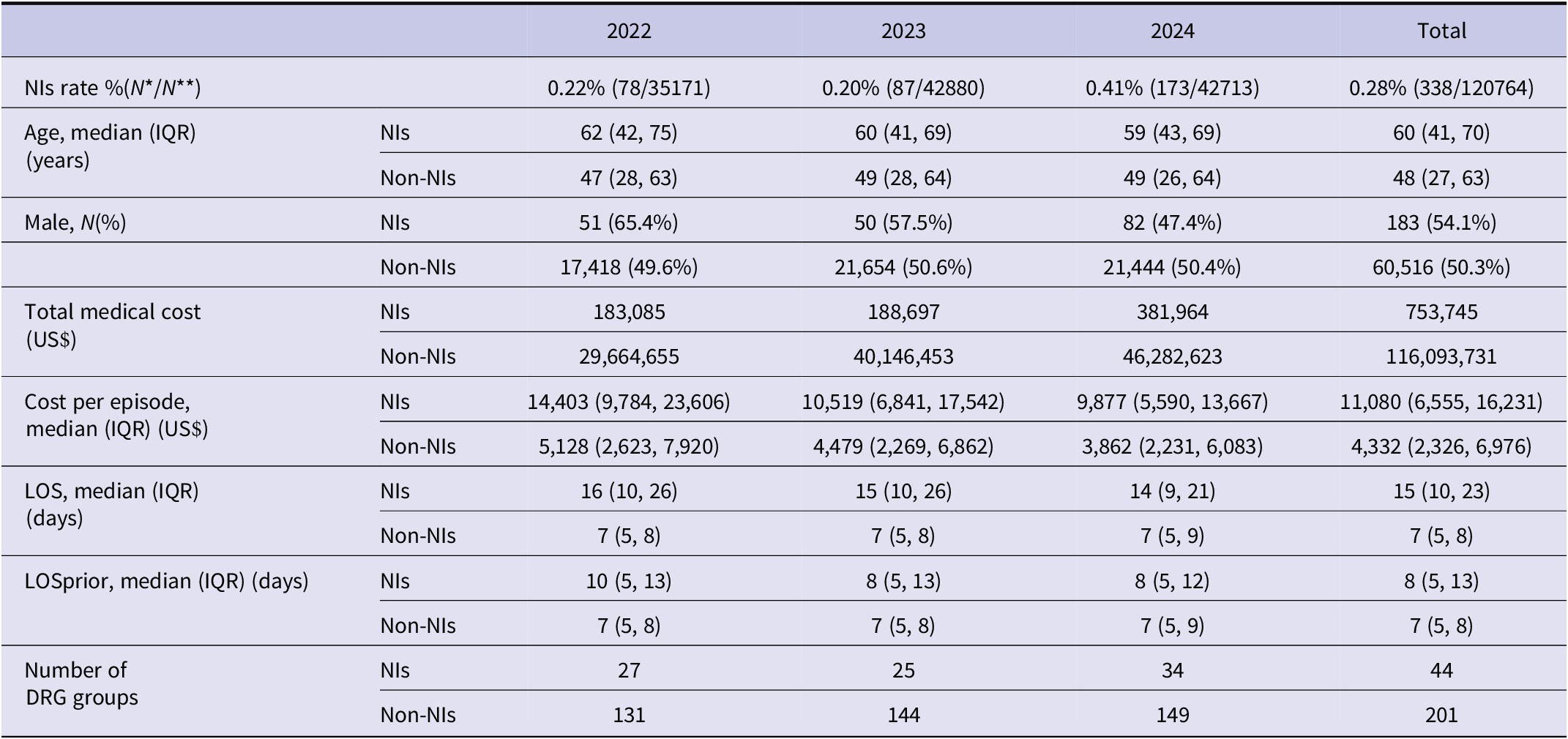
The median age of the cohort was 48 years, with 60,699 males (50.26%) and 60,065 females (49.74%). A total of 338 patients in the cohort developed NIs, corresponding to an overall NI rate of 0.28%, with an increase noted (0.22% in 2022 to 0.41% in 2024). The overall medical cost of NIs in the study population was >US$750,000, with an average cost of US$11,080 per episode. The cohort included a total of 201 DRGs, and the patients with NIs were concentrated across 21.89% of the DRGs (44/201) (Table 1).
Baseline characteristics of the study population (2022–2024)

IQR, interquartile range; LOS, length of stay; LOSprior, length of hospital stay prior to infection; N*, number of nosocomial infections; N**, number of inpatients included that year; NIs, nosocomial infections.
Furthermore, to scientifically evaluate the economic and time burden of NIs, matching was conducted to reduce the impact of confounding factors on the results. After matching, the distribution of characteristics that were significantly different between the patients with NIs and those without NIs was well balanced (Supplementary Table 1). The standardised mean differences of the covariates became more balanced after matching (Figure 1).
Covariate balance assessed by standardized mean differences. Categorical variables before matching are presented only for the categories retained after matching. Sex_1: Male; Sex_2: Female. Payment methods were recoded as follows: (1) Off-site Urban Resident’s Basic Medical Insurance; (2) Full Self-Payment; (3) Off-site Urban Employee’s Basic Medical Insurance; (4) Other Social Insurances; (5) Local Urban Employee’s Basic Medical Insurance; (6) Local Urban Resident’s Basic Medical Insurance. LOSprior: length of hospital stay prior to infection.

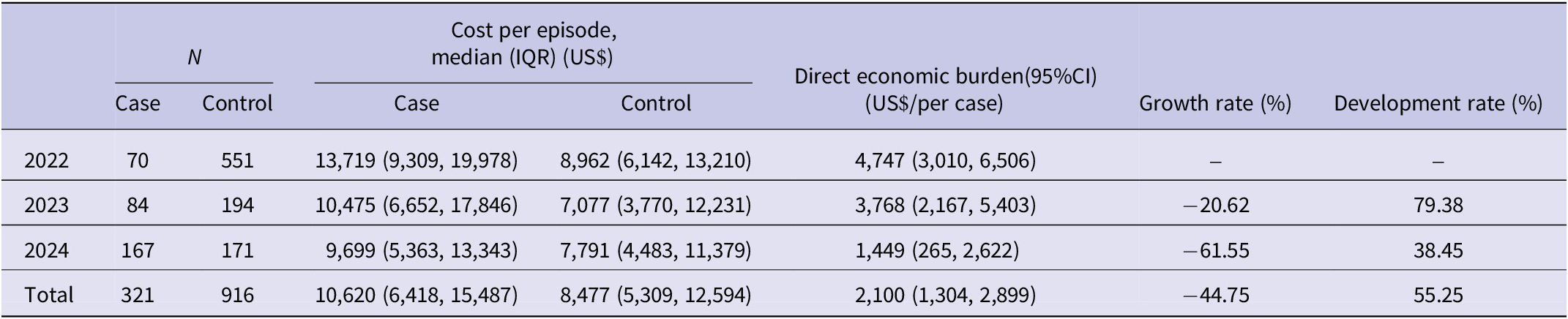
Direct economic burden of NIs and its dynamic changes
After matching, the direct economic burden attributable to NIs was US$2,100 per episode (95% CI: 1,304–2,899). It decreased by US$3298 overall, with a reduction rate of 69.48% and an average growth rate of −44.75%, from 2022 to 2024. The annual development rate still showed sustained cost pressure, and the overall average development rate was 55.25% (Table 2).
Annual direct economic burden of NIs (2022–2024)

The total value of absolute growth (year by year) is calculated as the arithmetic mean of the annual absolute changes. The total values for growth rate (YoY) and development rate (YoY) are calculated using the geometric mean method, also known as the average development rate. The geometric mean formula is as follows:
![]() $ \overline{x}=\sqrt[n]{\left({x}_1\;{x}_2\;{x}_3\cdots {x}_n\right)}=\sqrt[n]{\frac{y_1}{y_0}\;\frac{y_2}{y_1}\cdots \frac{y_n}{n-1}}. $
$ \overline{x}=\sqrt[n]{\left({x}_1\;{x}_2\;{x}_3\cdots {x}_n\right)}=\sqrt[n]{\frac{y_1}{y_0}\;\frac{y_2}{y_1}\cdots \frac{y_n}{n-1}}. $
Where:
![]() $ \overline{x} $
: average development rate;
$ \overline{x} $
: average development rate;
![]() $ {x}_i $
: development rate in year i;
$ {x}_i $
: development rate in year i;
![]() $ {y}_i $
: development level in year i; then, average growth rate =
$ {y}_i $
: development level in year i; then, average growth rate =
![]() $ \overline{x}-1 $
.
$ \overline{x}-1 $
.
Both Growth rate and Development rate represent year-over-year (YoY) sequential changes. CI, confidence interval; IQR, interquartile range; NIs, nosocomial infections.
Direct economic burden of NIs across different categories of costs
A detailed breakdown of cost components revealed that compared with controls, cases had significantly higher expenses in major categories, including medical, nursing, technical, administrative, and medication costs.
Among them, the difference in medical service expenses between the two groups was the greatest (US$449, P < 0.001). Although consumables accounted for the greatest proportion of overall costs for both the case and control groups, no significant difference was observed between the two groups (P = 0.077) (Table 3). In addition, compared with controls, cases had a longer length of hospital stay, with a median difference of 5 days (95% CI: 5–6) (Table 4).
Hospitalisation costs of patients with and without NIs

CI, confidence interval; IQR, interquartile range; NIs, nosocomial infections.
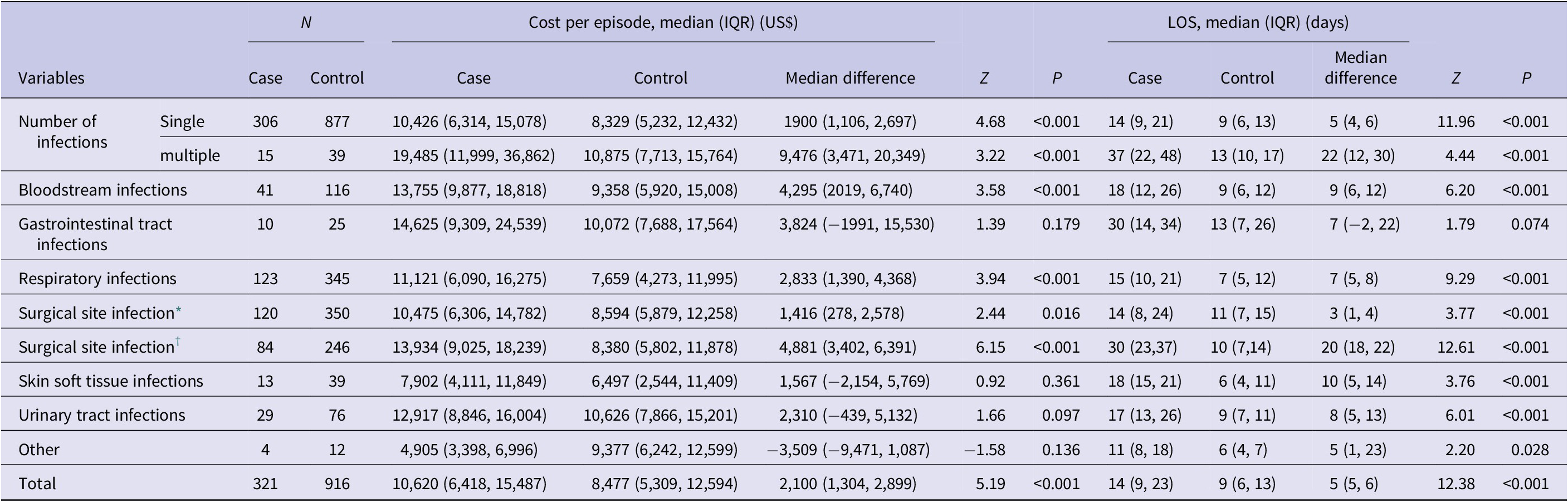
Comparison of cost per episode and length of stay by infection site and number of infections

* : the first admissions.
† : the first and readmissions; IQR, interquartile range; LOS, length of stay.
Economic and time burdens across different infection sites
Cases with multiple-site infections were associated with significantly greater increases in hospitalisation costs (US$9,476 vs. US$1,900) and length of stay (22 vs. 5 days) than those with single-site infections (P < 0.05) (Table 4).
In terms of hospitalisation costs, patients with NIs in the haematologic, respiratory, and surgical site systems had significantly higher hospitalisation costs than controls did (P < 0.05). Haematologic infections led to the greatest cost increase, averaging an additional US$4295 per case. With respect to the length of hospital stay, patients with NIs in the haematologic, respiratory, surgical site, skin and soft tissue, and urinary systems experienced significantly longer hospital stays than controls did (P < 0.05). Skin and soft tissue infections led to the greatest increase, extending hospital stays by an average of 10 days per case.
In addition, the burden analysis of SSI included not only the costs and LOS during the initial hospitalisation but also the costs and duration of readmission after discharge due to the occurrence of SSI. Compared with patients with non-NIs, those with SSIs incurred significantly higher costs (US$10,475 vs. US$8,594; P < 0.001) and had a longer median LOS (14 vs. 11 days; P < 0.001). When considering both initial hospitalisation and readmissions, patients with SSI experienced significantly higher total hospitalisation costs (US$13,934 vs. US$8,380; P < 0.001) and a markedly longer cumulative LOS (30 vs. 10 days; P < 0.001).
DRG-specific burden of NIs
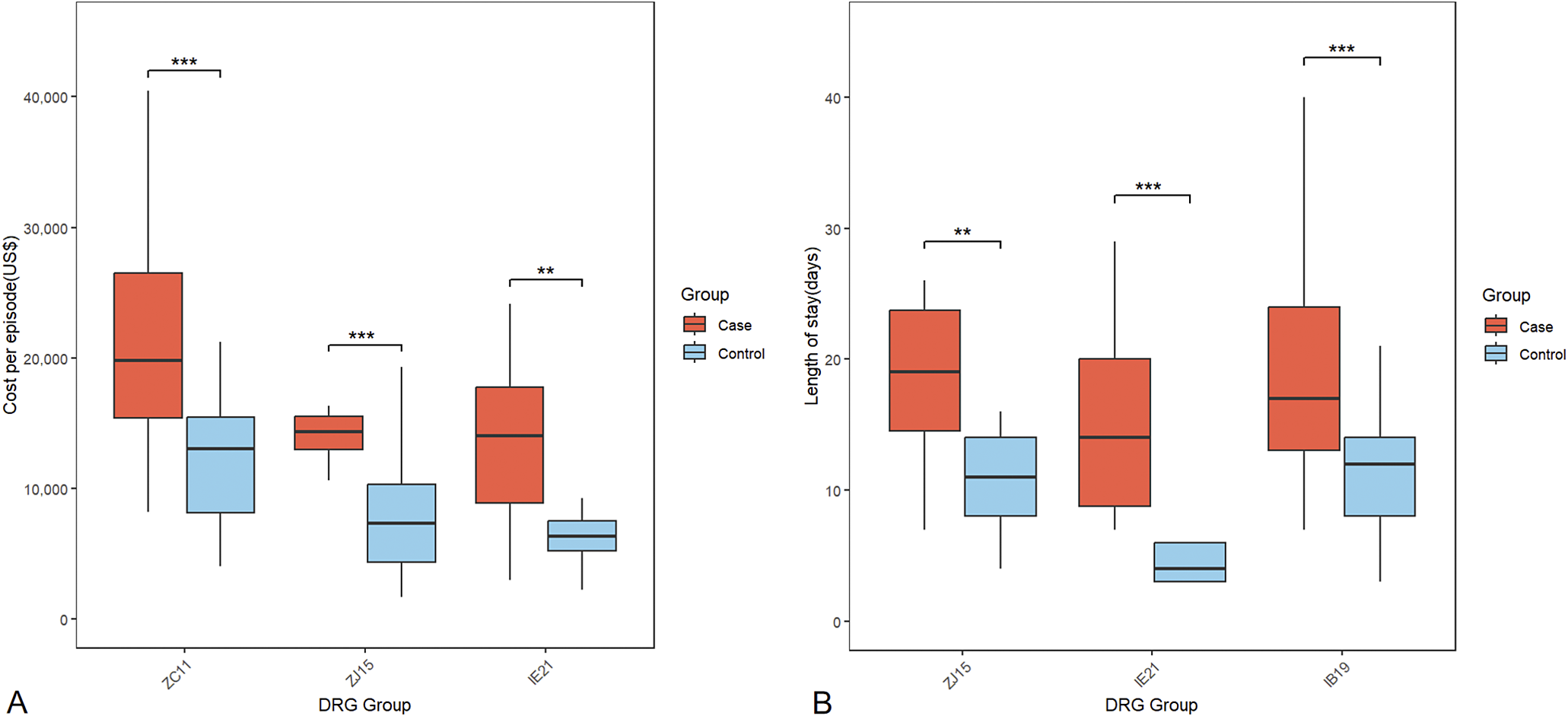
Subgroup analysis of the orthopaedic DRGs revealed substantial differences in the weights of NIs across the different DRGs (Supplementary Table 2). Patients in the IB19, ZC11, IE21, and ZJ15 groups who developed NIs experienced significant increases in hospitalisation costs (P < 0.05). In the majority of the DRGs, NIs led to a significant increase in the length of stay (P < 0.05). The three DRGs with the top economic and time burdens were ZC11 (US$9,410), ZJ15 (US$7,215), and IE21 (US$6,552) for costs, and ZJ15 (9 days), IE21 (8 days), and IB19 (7 days) for duration (Figure 2). These include surgeries for multiple severe traumas with serious complications (ZC11), multiple traumas without serious complications (ZJ15), femoral procedures with major complications (IE21), and complex spinal fusions or revision surgery (IB19).
The top 3 DRG groups of economic and temporal burden. A (Economic burden): The x-axis indicates DRG groups, and the y-axis shows median cost per episode (US$);B (Temporal burden): The x-axis represents Diagnosis Related Group (DRG) codes, and the y-axis indicates the length of stay (days). ** indicates P < 0.01; *** indicates P<0.001.

National economic burden analysis of direct NI medical costs
The analysis of the national economic burden under different scenarios revealed that the annual total economic burden caused by NIs ranged from US$2.26 million to US$37.52 million. Specifically, in the mixed scenario of the eastern, western, and central regions, the annual total economic burden was estimated at US$5.23 million (95% CI: 2.87–8.07) (Table 5). If the NI rate is reduced by 20% through comprehensive prevention and control, US$1.05 million (95% CI: 0.57–1.61 million) can be saved in the current year.
Analysis of the national economic burden of NIs in different scenarios

a A, Eastern region as the average; B, Western region as the average; C, Central region as the average; D, Mixed scenario of eastern, western, and central regions; E, High burden and cost (upper bound); F, Low burden and cost (lower bound). Estimates rounded to two decimal places.
Discussion
NI rates vary significantly across countries and regions [Reference Allegranzi1]. Our study revealed a 0.28% NI rate among orthopaedic inpatients from 2022 to 2024, which is lower than the general inpatient rate in Chinese hospitals [Reference Yang20]. This may reflect the generally better health and immunity of orthopaedic patients, who mainly undergo surgery for fractures or joint diseases, unlike ICU or oncology patients. A review of 400 studies from 2000 to 2021 revealed a global NI incidence of 0.14%, with 0.09% of orthopaedic patients having NIs, which is lower than that observed in our study [Reference Raoofi21]. This difference may be due to variations in patient populations and the follow-up of patients who were readmitted for SSIs.
In this study, the median direct economic burden attributable to NIs was US$2,100 per episode after matching, representing 15% of China’s per capita GDP in 2024 [22]. This is slightly lower but comparable to the national estimate of ¥24,881 (US$3,470) reported by a 2022 systematic review [Reference Liu23] and falls within the range of estimates from different regions in China: US$2,930 (western) [Reference Lv24], US$2,047 (central) [Reference Li25], and US$4,908 (eastern) [Reference Xiong, Wang and Hanafi9]. These variations likely reflect differences in healthcare resource allocation, infection management strategies, and hospital capabilities across regions. The lower burden observed at our centre may further imply the implementation of more effective infection control measures and the optimisation of healthcare delivery systems.
Further analysis of the cost structure revealed that medical service expenses ($449; 95% CI: 307–604) were the primary source of the economic burden of NIs, followed by medication costs ($311; 95% CI: 230–407). A study in a Chinese tertiary hospital reported significantly higher Western medicine costs for NI patients than for non-NI patients (US$1,931.12 vs. US$838.56; P < 0.001), indicating that drug costs were the primary burden in 2017 [Reference Lv8]. Similarly, another study in China revealed that the burden of treatment costs ($422) attributable to NIs was comparable to our medical service expenses but still notably lower than medication costs ($1,381) [Reference Xiong, Wang and Hanafi9]. These discrepancies may reflect healthcare reforms in China in recent years, including the removal of drug price markups, centralised procurement, and increased medical service fees to better reflect labour value. This shift likely contributed to the changing cost structure we observed.
In terms of the time burden, our study revealed that NIs extended hospital stays by a median of 5 days, which is shorter than the durations reported by Liu et al. (13.89 days) [Reference Liu23] and Xiong et al. (13 days) [Reference Xiong, Wang and Hanafi9] and the 10.4 day-extension reported across 68 hospitals [Reference Jia4]. These discrepancies may partly stem from earlier studies not adjusting for the preinfection LOS, which could lead to time-dependent bias and an overestimation of the LOS. Manoukian et al. [Reference Manoukian26] conducted a matched cohort study in the United Kingdom, matching controls to cases by preinfection time to minimise time-dependent bias. They estimated an excess LOS of 7.8 days (95% CI: 5.7–9.9), which is more conservative and aligns closely with our findings.
The burden of NIs varies significantly depending on the infection site, with bloodstream infections often associated with the highest economic costs across multiple studies [Reference Manoukian26, Reference Hasan27]. In our study, bloodstream infections had the highest median direct cost (US$4,295) and the second-longest hospital stay extension (9 days). Studies from China [Reference Li25], Italy [Reference Orlando28], and the United Kingdom [Reference Manoukian26] have also consistently identified NIs of bloodstream infections as imposing a substantial economic burden, with estimated costs of $4,734, €7,146, and £5,917 respectively. Jia et al. [Reference Jia4] reported that bloodstream infections led to the longest hospital stay extension (12.8 days). The differences in NI burdens across infection sites emphasise the need for targeted prevention and control strategies. In particular, bloodstream infections should be prioritised in prevention and surveillance efforts to minimise hospital costs, hospital stays, and adverse outcomes.
SSIs, especially in orthopaedic surgery, are among the most common postoperative complications [Reference Forrester, Maggio and Tennakoon2, [Reference Al-Mulhim29], Reference Liu30]. Since SSIs can occur after discharge and lead to readmission, we assessed both the burden during initial hospitalisation and that from subsequent admissions due to SSIs. In our study, SSIs extended the initial hospital stay by a median of 3 days and increased costs by US$1,416. When readmissions were included, the cumulative burden increased to 20 days and US$4,881. Li et al. reported a similar increase of US$1,825 in hospitalisation costs associated with SSIs during initial admission [Reference Li25]. Shambhu et al. [Reference Shambhu31] reported that in the United States, SSIs prolong the LOS by 3.99 days during index admission, which is comparable to our findings. For the cumulative burden, including the index hospitalisation and the subsequent follow-up period, a Dutch study on hip replacements reported a 12-month adjusted cost difference of €21,569 for deep SSI [Reference Koek32], and another U.S. cohort study reported cost increases of US$29,414 during the initial hospital stay and US$51,621 at 12 months following discharge [Reference Shambhu31]. These higher costs were likely due to differences in the national economic levels of the study population and the variety of surgical procedures. Notably, they all highlight that SSIs continue to impose a considerable financial burden on patients even after discharge. These results underscore the long-term clinical and economic burden of SSIs beyond initial hospitalisation and support the necessity of including both initial and readmission-related resource utilisation in cost assessments.
Orthopaedic patients in different DRGs demonstrated variations in hospitalisation costs and lengths of stay. Notably, there were significant differences in the burden of NIs across different DRGs. NIs significantly increase the economic and time burden for patients in the multiple severe trauma with complications (ZC11), multiple trauma without complications (ZJ15), femoral procedures with major complications (IE21), and complex spinal fusion or revision (IB19) groups. These burdens were likely driven by two factors: the patients’ compromised baseline status and the complexity of the procedures. This pattern aligns with prior studies [Reference Missios and Bekelis33] identifying high costs and resource use following complex spine surgeries. The heterogeneity observed in clinical trajectories and resource demands across DRGs highlights the need for targeted infection control strategies based on DRG classifications to achieve more precise resource allocation and risk management.
This study is the first to analyse the direct economic burden of NIs in specialised orthopaedic hospitals in China from a healthcare system perspective, offering evidence for resource allocation in prevention and control. Considering the impact of regional disparities on economic burden, this study utilised a scenario-based sensitivity analysis to preliminarily estimate the annual national economic burden of NIs among orthopaedic inpatients. Owing to the lack of data on orthopaedic inpatients in general hospitals, the current economic burden is only calculated for specialised orthopaedic hospitals. Under a mixed scenario encompassing the eastern, western, and central regions, the annual economic burden was estimated to be US$5.23 million (95% CI: 2.87–8.07), accounting for 0.15‱ of the government health expenditure [34]. Although we conducted a sensitivity analysis based on regional differences in parameters, the analysis was a preliminary estimate of the direct economic burden at the national level, and there may be some inaccuracies in the estimation. Future research should use national multicentre data to achieve more precise estimations. In this context of the mixed scenario, a 20% reduction in the NI rate through comprehensive prevention and control measures could save an estimated US$1.83 million (95% CI: 1.01–$2.83) in direct medical costs within the year. Savings could be maximised by prioritising high-burden sites (e.g., bloodstream and SSIs including readmissions) and high-impact DRGs (ZC11, ZJ15, and IE21). These results highlight the need for improved infection prevention and management strategies in hospitals.
This study has several strengths. First, to more accurately assess the economic and time burdens of NIs, we matched cases and controls not only by sex, age, and DRG but also by the LOSprior, since NIs are time-dependent exposures. Comparing total LOS without this adjustment can lead to overestimation, as only postinfection stays should be attributed to NIs [Reference Heister, Kaier and Wolkewitz17]. Second, we included SSIs that occurred during hospitalisation and postdischarge follow-up and assessed both readmission time and cost, thus capturing the longitudinal burden of SSIs more comprehensively. Third, by analysing NI burden in orthopaedic patients, we identified high-burden DRGs and infection sites, providing an evidence-based foundation for precise prevention and control.
This study has several limitations. First, we analysed the dynamic changes in the economic burden of NIs from 2022 to 2024, which is insufficient for reflecting long-term trends. Future studies should extend the observation period. Second, as a single-centre nested case–control study, caution is needed when the results are extrapolated to a wider population. However, our national orthopaedic medical centre admits more than 40,000 inpatients annually, with 55% from other provinces in China, partly mitigating regional limitations. Third, our findings quantify the direct economic burden of NIs among orthopaedic inpatients from the healthcare system’s perspective. They do not encompass the indirect economic burden, social burden, medical insurance reimbursement rates, or individual out-of-pocket expenses. It is advisable for future research to undertake a more comprehensive health economic evaluation from the perspectives of payers, patients, and society, which could provide evidence-based support for broader health policy decisions.
In conclusion, this study systematically quantified the economic and temporal burdens of NIs in orthopaedic patients. We accurately identified the DRGs and infection types with a high burden of NIs, thereby providing an evidence-based foundation for the next steps in targeted prevention and control and evaluating the cost-effectiveness of prevention and control measures.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0950268826101125.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author (chenhuijst@outlook.com). The data are not publicly available due to their containing information that could compromise the privacy of patients.
Author contribution
Conceptualization: L.Y.; Data curation: R.M., L.L.; Formal analysis: Y.R.; Supervision: Y.T., H.L.; Investigation: H.W.; Funding acquisition: H.C.
Funding statement
This work was supported by the funding for the discipline construction of burn science in 2025(grant 3–1–992-926-29, to Hui Chen); The Beijing Municipal Administration of Hospitals Incubating Program (grant PG2024008, to Lin Yang), and the Capital’s Funds for Health Improvement and Research (Beijing Municipal Health Commission; grant 2024-1G-2071, to Hui Chen)
Competing interests
The authors have no conflicts of interest or financial ties to disclose.
Ethical standard
This study was approved by the Ethics Committee of our hospital (Approval No. K2024381), and all methods were in accordance with the Declaration of Helsinki, with a waiver of informed consent.