1 The Advent of a Technology
Where a technology is recognised as being transformative we are apt to attach equal significance to its discovery. The history of invention tells us this is not so, famously illustrated by the example of wheeled toys predating the use of wheels for transport in pre-colonial Meso-America. Nor did the wheel necessarily transform its host cultures, being discarded in the second and third centuries in the Near East in favour of the camel despite its invention many millennia prior. Faced with this paradox, Basalla conceded three points: ‘First, wheeled vehicles were not necessarily invented to facilitate the movement of goods; second, western civilization is a wheel-centered civilization that has carried rotary motion to a high state of development; and, third, the wheel is not a unique mechanical contrivance necessary, or useful, to all people at all times’ (Reference Basalla1988:11). These statements clearly advocate caution lest we make deterministic conclusions regarding technological change based on its consequences. Yet they also force us to question the circumstances of an invention if it was not necessary, or required, to fulfil a function it only later assumed. A technology might well be discovered long before the major expansion in its adoption and use, after intervening factors have contributed towards its viability in a new role. Until these factors are introduced a nascent technology may remain as a potential, persisting within a host tradition. Thus only once an invention transforms this tradition can this event be recognised as the advent of a technology.
A technology that conforms to this process might be expected to have roots in a parallel family of technologies. This is exemplified by the long association of the development of extractive methods to produce iron within preceding metallurgical traditions for the production of copper in the Near East during the 2nd millennium BCE. Early proponents drew this connection during the first half of the twentieth century after the prevailing theories of the previous century, the working of meteoritic iron or accidental discovery, were ruled out as viable routes for the establishment of technologies for the extraction of iron (Rickard Reference Rickard1939:86–90). At the same time, the discovery of a number of texts ascribed to the Hittite kingdoms of Central Anatolia dating from the seventeenth to fourteenth centuries BCE seemed to produce convincing evidence in descriptions of smelted iron among royal inventories far in advance of its widespread adoption in Europe nearly a thousand years later (Kempinski and Košak Reference Kempinski and Košak1977; Souckova-Siegelová Reference Souckova-Siegelová2001; Yalçin Reference Yalçın1999:184). These texts increasingly came to be corroborated with the recovery of a series of small iron artefacts at sites across the Hittite world, notably at the seventeenth-century BCE site of Alaca Höyük (Erb-Satullo Reference Erb-Satullo2019:564; Yalçin Reference Yalçın1999:180). This encouraged many broad syntheses to place the Hittite complex at the forefront of the history of early iron production and use in the Near East (Muhly et al. Reference Muhly, Maddin, Stech and Ozgen1985; Wertime and Muhly Reference Wertime and Muhly1980; Waldbaum Reference Waldbaum and Pigott1999). Yet with the failure to locate unambiguous evidence for iron smelting in the region before the eleventh-century BCE installation at Phokaia, this provenance was limited to culture-historical narratives (Yalçin and Özyiğit Reference Yalçin and Özyiğit2013:240–243). This begs the question of whether we are even capable of identifying acts of invention among extinct cultures.
Modern thought on how invention occurred in past societies has increasingly become rooted in perspectives of materiality or the way in which human cultures generate material culture through engagement with the physical world (Killick and Fenn Reference Killick and Fenn2012:564). Most often the human interactions with materials that lead to an invention have been construed as a sequence of stages involving observation, response and adoption. This is especially true of archaeological perspectives which rely on chronological sequences of related members in the material record (Erb-Satullo Reference Erb-Satullo2020:42; Schiffer Reference Schiffer2010; Shortland Reference Shortland, Bourriau and Phillips2004). This is also reflected in the breaking down of life histories of artefacts into a series of processes which in turn represent a specific set of activities and interactions (Schiffer Reference Schiffer2010:236). Though such perspectives have been criticised as leading to deterministic conclusions regarding processes of technological change, sometimes termed unilinear evolutionary models, sequential schemes remain indispensable in conceptualising invention (Shortland Reference Shortland, Bourriau and Phillips2004; Roberts and Radivojević Reference Roberts and Radivojević2015:299). Drawing on the concept of a sequence, the first stage of invention can be termed the ‘discovery’ or ‘inspiration’ stage which itself is preluded by a physical or cognitive phenomenon (Shortland Reference Shortland, Bourriau and Phillips2004:2). This initial position was most aptly described by Cyril Stanley Smith who related that: ‘A new thing of any kind whatsoever begins as a local anomaly, a region of misfit within the pre-existing structure’ (Reference Smith1977:144). As an anomaly, such events or phenomena represent a deviation from expected norms of the physical world or cognition subjective to an individual or group (local). An oft-cited if dubious example can be found in the description by Pliny of the setting of camp fires by sailors over soda blocks on a beach – the resultant reaction with sand leading to the production and discovery of glass (Pliny the Elder 1991, Book XXVI.191). An important caveat is that this anomaly occurs within but does not conform to a pre-existing structure. It is this characteristic of an anomaly that can allow it to persist within a structure while remaining non-actionable until favourable circumstances arise that permit its exploitation. Thus whether or not an anomaly instigates an invention depends upon existential factors. These may arise from intrinsic qualities, such as its non-conformity resulting in its dismissal as an undesirable component within a system, or from external or human factors stemming from a lack of proficiency or desire to exploit a new phenomenon before pre-requisite cognitive developments.
Though finding success in revealing invention among past cultures may appear improbable, many expectations regarding the perceptibility of inventive activities in the material record may no longer apply if invention is conceptualised as a process. To do this, it is necessary to place invention within the wider contexts of the entire corpus of human activity (Frieman Reference Frieman2021). These activities can then be investigated through a series of methodological steps to reveal this process in action among past peoples. Beginning with the ‘discovery’ stage of invention, existing anomalies can be identified and related to specific activities and formation processes with a much greater degree of confidence especially with the application of archaeomaterials techniques (Killick and Fenn Reference Killick and Fenn2012; Roberts and Radivojevic Reference Roberts and Radivojević2015:301). As any anomaly represents an outlier within an existing structure, if the nature of the structure can be defined the anomaly can be placed within the much broader technological contexts of related practices. This opens up a far more substantial body of evidence that can be drawn upon from the archaeological record while any conclusions will better reflect the actual circumstances under which an invention was discovered rather than the later more visible processes of innovation and adoption that followed.
As part of efforts to conceptualise invention in a behavioural context or in other words, as a process, Schiffer proposed three steps for revealing invention in the archaeological record: ‘First, search for those archaeological deposits that might retain high-resolution evidence of inventive activities … Second, develop models by employing research strategies such as ethnoarchaeology, computer simulation, experimental archaeology, modern material-culture studies, and historical research … Third, formulate research questions in behavioural-not archaeological context-terms’ (2010:236). Throughout the rest of this section I evaluate each of these steps, beginning by asking to what extent we can reveal invention among primary production contexts before moving on to the use of analogy to inform attempts at modelling inventive processes. Here I argue for the use of experiment to inductively investigate these processes through the reproduction of assemblages and the use of analytical techniques to form a basis for comparison to the material record. The third step, while specifying the behavioural theory presented by Schiffer and colleagues, can be viewed as a call to relate the findings of the previous steps back to theory. The section ends with an overview of three theoretical frameworks for inventive processes that have been applied to past cultures among recent studies. Embracing this three-pronged approach, this element first assesses the evidence available from the material record for the anomalous production of iron during copper smelting practices, using the conclusions to inform a series of experiments. The resulting assemblages are compared directly to those recovered from the archaeological record of the region before being incorporated into the three theoretical frameworks discussed later in this section. In summary, this element argues that the extractive traditions of the Near East continue to offer the most promising avenue of research into the advent of iron in the region during the 2nd millennium BCE. At its core, this approach accepts that questions of technological change are existential, arising as a direct consequence of human behaviour and engagement with the material world.
1.1 Can We Reveal Invention?
Demonstrating acts of invention within the archaeological record has often been approached with reluctance and scepticism (Roberts and Radivojević Reference Roberts and Radivojević2015:300; Schiffer Reference Schiffer2009). Doubt has been expressed along both practical (Schiffer Reference Schiffer2009:239–241; Shortland Reference Shortland, Bourriau and Phillips2004) and theoretical (Fitzhugh and Trusler Reference Fitzhugh, Trusler and Shennan2009; O’Brien and Shennan Reference O’Brien and Shennan2010:3–5; Roberts and Radivojević Reference Roberts and Radivojević2015:299, 303) lines of reasoning regarding the capacity of archaeological methodologies to reveal invention among past cultures. The former largely stem from the limitations of archaeology to perceive evidence of invention among a fragmentary and incomplete material record reliant on chance discovery. Misgivings regarding the objectivity of interpretations also highlight the risk of projecting presentist concepts onto a sparse material record or falling foul of a misattribution of terms when lacking specificity (Killick and Fenn Reference Killick and Fenn2012:566; O’Brien and Shennan Reference O’Brien and Shennan2010:3–5; Roberts and Radivojević Reference Roberts and Radivojević2015:299, 303). This is due in no small part to the ambiguity of the nature of invention itself; if it is conceived of as an ‘idea’ or ‘recognition’ of the advantageous it must pre-exist any engagement with the physical world (Schiffer Reference Schiffer2010:236; Shortland Reference Shortland, Bourriau and Phillips2004:1). Yet as will be attempted in Section 2, it may be plausible to reconstruct the circumstances of invention if no direct evidence is available or recognisable.
Inventions that have their origins in a singular and likely unprolific or transient event are unlikely to be discovered among the deposits of established practices. This may lead to mature technologies becoming confounded with acts of invention simply as a result of appearing earliest in the archaeological record. This applies particularly to the uncovering of evidence for iron production among the primary production contexts of copper smelting traditions due to the greatly increased rates of corrosion for iron artefacts in soils compared to copper. In a study by Neff et al. iron was shown to corrode at an average rate of 4 µm retreat of the surface per year (Reference Neff, Dillmann, Bellot-Gurlet and Béranger2005). This rate of corrosion would, outside of anoxic conditions, total up to 16 mm in penetration and loss of the surface for iron deposited from the end of the 2nd millennium BCE. By contrast, copper corrodes at a rate of just 0.45 mm every 1,000 years or just over 1 mm for copper deposited during the same period (Tylecote Reference Tylecote1979:367). As a result, only those iron artefacts that exceeded some kilograms in size could potentially survive into the material record, likely the products of mature technologies that developed after the earlier and smaller scale occurrences that led to their invention. The diminutive quantities of iron that might be expected may also be vulnerable to further exploitation especially at many candidate sites that saw intensive activity, re-occupation and exploitation of the same local ore deposits over multiple centuries.
Chance discovery of direct evidence for inventive activities should not be ruled out with the possibility of finding one of a kind items that fall outside of existing typologies among particularly complete and well-studied deposits demanding a case by case approach (Schiffer Reference Schiffer2010:240). Similarly, unique objects may also exist in the form of ‘wasters’ or anomalous objects which Shortland describes as ‘hopeful monsters’; technological dead ends whose use may be revived if a new purpose is discovered for them, leading to new innovations (Reference Shortland, Bourriau and Phillips2004:7). In order to identify the types of deposits described above that contain rare instances of direct evidence for invention, it is crucial that we understand the wider technological contexts of these deposits. Not only does this approach allow us to recognise their significance for later developments among those contexts; in the absence of direct evidence it also provides an insight into the processes of invention that might have occurred. In Section 2, I undertake the first step in the scheme proposed by Schiffer (Reference Schiffer2010) and attempt to define the technological contexts of copper production among the traditions of the Near East during the 2nd millennium BCE.
1.2 Through Analogy
The available evidence from the archaeological record may not always be sufficient to reveal the nature and extent of an anomaly, bringing us to Schiffer’s second step. For this step, Schiffer highlights a number of research strategies for which the: ‘ Confounding effects of many formation processes are lessened’ (Reference Schiffer2010:236). Here Schiffer recognises that the majority of artefacts exist in the terminal stage of their life histories. This makes it difficult to reconstruct the circumstances of their production. Through the use of analogous sources or historical accounts it is possible to overcome this disconnect between artefacts and formation processes (Bell Reference Bell, Chapman and Wiley2014:43). Of course these sources may also provide other insights not directly related to the manufacture of an artefact including social organisation, recipes and any other behavioural question relating to the interaction of people with objects (Schiffer Reference Schiffer and Schiffer2013:43,54). In this Element I employ analogy in a third use, the replication of formation processes to reveal the potential for an anomaly to occur within a specific technological context. In the following paragraphs, I discuss the theory and approaches to experimental archaeology before arguing for the application of an experimental methodology to reveal the extent of the anomalous production of iron among the extractive traditions of the 2nd millennium BCE in the Near East.
The use of experiment in archaeology has been a subject of some controversy since its potential in addressing broader theoretical questions was first made explicit with the rise of processualist thought under Binford during the 1960s (Bell Reference Bell, Chapman and Wiley2014:41). While attitudes towards incorporating experimental approaches have once again become more favourable with recognition of its advantages in providing useful archaeometric data and promotion of cross-disciplinary discourse, it is worth addressing the major points of controversy (Bell Reference Bell, Chapman and Wiley2014:42). Much of the criticism has centred on the objectivity of experiment in making inferences about the past when it is embedded in modern theoretical, social and cultural perspectives (Shanks and Hodder Reference Shanks, Hodder, Alexandri, Buchli, Carman, Hodder, Last, 71Lucas and Shanks2013:3). Bell highlights the first of these stumbling blocks faced in experiment with the philosophical conundrum that the ‘Gathering of data is not independent of the theory to be tested’ (Reference Bell, Chapman and Wiley2014:43). The selection of phenomena to test, the variables considered, the design of the methodology and the building of hypotheses are all made according to a preconceived theoretical basis. The objectivity of observation and interpretation of reconstructed past technologies and processes have also been called into question given the modern scientific and academic perspectives of participants (Outram Reference Outram2008: Reynolds Reference Reynolds1999).
With the growth of experimental archaeology into an ever-more distinct discipline over the past few decades, the validity of many of these criticisms have been reassessed in light of developments in experimental practice (Bell Reference Bell, Chapman and Wiley2014:43; Deady et al. Reference Deady, Fregni and Stewart2015; Schenck Reference Schenck2011). One fundamental change has been to reconsider the role of experimental methods as a means of analogy. With the use of analogy requiring critical evaluation to make accurate comparison to the archaeological record, Bell highlights that it is important to define the types of analogy that experimental methods are best suited to provide (Reference Bell, Chapman and Wiley2014:43). He suggests that experiment is more suited to explore relational analogies i.e. the causal relationships between two variables, as opposed to formal analogies which involve cross-cultural and temporal comparisons (Bell Reference Bell, Chapman and Wiley2014:43; Hodder Reference Hodder2012:56). By avoiding formal analogy, modern scientific and cultural bias is less likely to influence any conclusions and the causal relationships between factors can be investigated through a hypothetico-deductive lens. Another development has been the acknowledgement of the advantages of actualistic versus controlled experiments (Schenck Reference Schenck2011:87). This relates to the degree of control maintained over variables within an experiment, with actualistic experiments exercising less control over many variables and controlled experiments maintaining tight control over a few (Schenck Reference Schenck2011:88). Actualistic experiments are often termed ‘field’ experiments as they are conducted at a scale and authenticity of materials and processes that reflect the actual contexts of the technology or activity under investigation. The advantage of introducing so many variables is to more accurately recreate a process which may reveal unanticipated consequences and thus increase current knowledge (Schenck Reference Schenck2011:90). However, with less control of variables it is more difficult to apply the results to the hypothesis and communicate the findings in a scientific or replicable context (Ferguson Reference Ferguson2010:4; Schenck Reference Schenck2011:88). The extent to which past materials, circumstances and environments are replicable presents another challenge that actualistic methodologies must negotiate or risk introducing false variables which may produce misleading data that is not representative. Advocates for control during experimentation stress that the data obtained is measurable and communicable while remaining explicit to the hypothesis (Reynolds Reference Reynolds1999:387–388; Schenck Reference Schenck2011:89–90). Though the two approaches have sometimes been presented as a dichotomy, Schenck points out that in both types of experiment focus should be placed on what control of the variable is meant to accomplish in terms of generating knowledge (Reference Schenck2011:89). Turning aside from the strict definition of an experiment given by Reynolds (Reference Reynolds1999), a final development is the framing of an experiment inductively (Schenck Reference Schenck2011). This is more commonly associated with actualistic experiment where less control of variables is expected resulting in unanticipated consequences. Following inductive lines of reasoning, this method consists of recreating experimental conditions to inform a conclusion (Schenck Reference Schenck2011:90). While inductive experiments may not be fully independent of theory, upon which the methodology is designed, the conclusions drawn from the results are.
Yet perhaps the most significant development in experimental practice in recent years has been to define the relationship of an experiment to theory (Bell Reference Bell, Chapman and Wiley2014; Ferguson Reference Ferguson2010:3). A major breakthrough has been the placing of experiment into three categories based on how they relate to theory: crucial, corroborative and new phenomena experiments (Ferguson Reference Ferguson2010:3). Experiments in the crucial category support an existing theory over other explanations, those in corroborative provide evidence for a single theory and new phenomena experiments produce unexpected results that do not correlate with pre-existing theories and may form the basis of new theories (Ferguson Reference Ferguson2010:3). By relating an experiment to theory it is possible to assess how the experimental methodology addresses the theory in question rather than pre-determined expectations of outcomes. As the field of experimental archaeology has matured it is clear that archaeologists have become more proficient in utilising experiment while navigating the issue of objectivity, both through experimental design and by incorporating it into wider theoretical discussions and cross-disciplinary research. This element hopes to provide an example of the valuable contribution experimental archaeology can make in elucidating a key theoretical question regarding the development of iron metallurgy in the Late Bronze Age Near East.
As discussed earlier, it is important to relate the experimental design to theory to establish what knowledge the experiment is intended to generate. For an investigation into the potential for an anomaly to occur as well as its nature and extent within a specific technological context, a good experimental methodology will have the following objectives. Firstly, the experiment should aim to provide a relational analogy with the technology under investigation, as an anomaly is contingent upon causal factors that must be identified and, if possible, quantified to reveal the magnitude of any resulting anomaly. In order to identify these factors, the methodology should be designed to incorporate every variable present during the formation processes in question or in other words attempt to reproduce actualistic conditions. Thus many variables will be uncontrolled with the possible exception of a variable that is preselected as a determining factor for the existence of an anomaly. As the consequences of the reproduction of these conditions and the control of the variable cannot be known before obtaining the results of the experiment, a working hypothesis cannot be constructed and so it is necessary to frame the experiment inductively. Any hypothesis will therefore be built based on observation of the experimental results. While theory may be used to inform the experimental methodology it does not form the basis of the hypothesis in inductive experimentation. As an experiment using an inductive methodology cannot correlate with existing theories and the results are likely to support the creation of new theories as new hypotheses are made, it can be defined as a new phenomena experiment. The experimental methodology presented in Section 3 of this Element was designed to reflect these objectives as an example of how the relating of experimental practice to both analogy and theory can produce valuable data that more directly addresses questions of technological change in past cultures.
1.3 Through Theory
While experiment and other sources of analogy can yield useful information, without being related to theory its application is reduced to providing empirical generalisations, experimental laws and recipes or the technological and technical components of a technology (Bell Reference Bell, Chapman and Wiley2014:43; Ferguson Reference Ferguson2010; Schiffer Reference Schiffer and Schiffer2013:43). It is only through the combination of both analogy and theory that we can begin to understand technology in the social and cultural contexts of past traditions. The final step, which Schiffer describes as the framing of research questions in behavioural terms, emphasises that focus should be placed on the activities that took place during the life history of an artefact (Reference Ferguson2010:236). This is critical to the understanding of invention as a process or series of interactions in which humans participated with the outcome of creating a novel technology. If this series of interactions is broadly understood under the concepts of observation, response and adoption there are three existing theories that may contribute towards our understanding of each stage. The end of this section provides a brief outline of each of these theories, beginning with analogical reasoning drawn from cognitive psychology, which may have the greatest implications for the observation stage at the level of the individual. Moving on to response, the behavioural theory proposed by Schiffer and colleagues provides a mechanism for the creation of new technologies via a process of selection of variants. For the final stage of adoption, evolutionary theories that take inspiration from biological processes may explain how the most successful variants are selected and transmitted from among competing variants resulting in mature technologies.
The triggering of a response by an anomalous occurrence within a structure may be caused by a cognitive mechanism which has been likened to analogical reasoning among cognitive psychologists (de Beaune Reference de Beaune2004, Reference de Beaune, Coolidge and Wynn2009). This aspect of our cognition involves the transferral of knowledge from one domain, our anterior experience stored in the long-term memory, to a similar yet non-identical domain of our present experience (de Beaune Reference de Beaune2004:150). This process requires a number of cognitive tools, from the retrieval of relevant information perhaps with the selection of recognisable structures in the memory, to generalisation and abstraction in order to apply this data outside of its immediate context (de Beaune Reference de Beaune2004:150). Furthermore to produce a new response as a result of analogical reasoning, a mental projection of the objective of the transfer is necessary. As a ‘conditioning reversed in time’, the faculties of creative imagination and forethought likely play a role in this projection from the virtual to reality (de Beaune Reference de Beaune2004:150). After the transferral of an analogical situation has been made to the new situation, subsequent experience may be stored as a new reference in the memory, resulting in the variation of behaviours in response to an anomaly and an increase in knowledge of it.
Once an anomaly is recognised as a misfit within the existing structure, it is this very non-conformity that drives subsequent interest and investment into it until the structure itself is transformed into the new. The process by which this technological change occurs has attracted a great deal of attention within archaeology over the past half century, with the most influential theories operating around a principle of selection of variants controlled by numerous social, behavioural and cultural factors (O’Brien and Bentley Reference O’Brien and Bentley2011:311; Skibo and Schiffer Reference Skibo and Schiffer2008:67). In the behavioural theory developed by Michael Schiffer and colleagues, variants are selected on the basis of ‘performance characteristics’ (Schiffer and Skibo Reference Schiffer and Skibo1997:30). These are the innate characteristics of a material or technology that allow it to fulfil an interaction, such as the resistance to thermal shock that clay possesses and that relate to its formal properties, which in turn are influenced by technical choices, such as the decision to use clay for the production of cooking vessels (Schiffer Reference Schiffer2010:236; Schiffer and Skibo Reference Schiffer and Skibo1997:31). Through their performance characteristics, variants are then subjected to immediate and extended selective contexts (O’Brien and Bentley Reference O’Brien and Bentley2011:312). The immediate selective context includes all activities and formation processes in the life history of an artefact, while the extended selective context consists of all other components that contribute towards its manufacture ranging from activities to flows of energy and mechanisms. Both contexts act as selective pressures, allowing only those variants with favourable performance characteristics to persist thus reducing variation where inventive activity increases it (O’Brien and Bentley Reference O’Brien and Bentley2011:312). Returning to inventive processes that are instigated via an anomaly, this model is faced with a dilemma. If an anomaly represents a region of misfit and possibly disruption to the existing structure, how could it possess a positive performance characteristic and become selected for? The answer is for a change in the selective contexts to occur: a process that Schiffer referred to as stimulated variation (O’Brien and Bentley Reference O’Brien and Bentley2011:312–313; Schiffer Reference Schiffer1996). This change may be brought about through technological, cultural or social developments which reorient the priorities of producers, leading to bursts of inventive activity and the increase of variants. From here, selection may proceed in a highly patterned sequence that Schiffer terms a cascade (O’Brien and Bentley Reference O’Brien and Bentley2011:314; Schiffer Reference Schiffer2005). Cascades begin where a performance problem or non-favourable performance characteristic is recognised, leading to successive inventive spurts until the performance problem is eliminated (O’Brien and Bentley Reference O’Brien and Bentley2011:314). Once a solution to this initial problem is found the cycle begins anew as emergent performance problems are discovered or as selective contexts change; progressing in a series of rapid expansions and contractions of variation that typify cascades.
Selection of variants is also a concept that is core to the application of evolutionary biology to questions of technological change. Rather than see variation necessarily as a consequence of behaviour, evolutionary archaeologists emphasise the role that the biased transmission of knowledge can play in technological divergence (Eerkens and Lipo Reference Eerkens and Lipo2005; O’Brien and Shennan Reference O’Brien and Shennan2010:9). These biases relate to the contexts of learning new knowledge, be it social through transmission from others or individual as a result of trial and error or fortuitous observation (Boyd and Richerson Reference Boyd and Richerson1985; O’Brien and Bentley Reference O’Brien and Bentley2011:315–317). For social learning, the choice of whose actions to imitate and why to imitate them reflects the bias of the recipient, who may in turn imitate these actions incorrectly and introduce ‘copy errors’ which generate new variants (Eerkens and Lipo Reference Eerkens, Lipo, Stark, Bowser and Horne2008:67; O’Brien and Bentley Reference O’Brien and Bentley2011:315–317). The impact of these errors may be immediate or delayed depending on the capacity of the practitioner or wider tradition to perceive their consequences. Often the material or conceptual outcomes of such errors may be discarded as ‘wasters’ until a new purpose is found for them, termed a ‘hopeful monster’ (Shortland Reference Shortland, Bourriau and Phillips2004:7). Alternately in individual or environmental learning new variants are generated only through experimentation or the modification of existing activities which is likely stimulated through observation of an anomaly or thing of interest. This is without the influence of others so is said to be unbiased. The individual responsible may then transmit the new knowledge to those around them, who in turn may modify these practices, still further resulting in the directed alteration of activities across generations in a process described as guided variation (Boyd and Richerson Reference Boyd and Richerson1985; O’Brien and Bentley Reference O’Brien and Bentley2011:317). Of these two, the mode of learning that is best suited to describe the triggering of invention from an anomaly is that of individual learning via a process of guided variation. Within this category mechanisms for both the observation and recognition of an anomaly as well as the experimentation required to exploit it are present. That is not to ignore the destabilising effect that the introduction of copy errors through social learning may have upon the magnitude of an anomaly, potentially increasing its perceptibility among individuals. The guided variation model may also be insufficient to explain how an anomaly may cause rapid technological change with only the typically slow and localised forces of individual trial to drive it. In Section 5 of this Element I discuss the implications that these theoretical models may have for our understanding of the development of iron metallurgy among the extractive traditions of the Near East during the 2nd millennium BCE.
2 Iron in Context
Defining a technological context requires the total reconstruction of a technology. This includes the practices, constructs, materials and processes utilised within a branch of related technologies as well as socially-embedded aspects such as the transmission of knowledge, organisation and economy. This has been likened to a recipe, which Schiffer and Skibo list as ‘(1) A list of raw materials … (2) a list of tools and facilities used … (3) a description of the specific actions undertaken in the technological process, and (4) the contingent rules used to solve problems that arise’ (1987:597). A further step is to categorise each technological component as an ‘ingredient’, those placed within categories (1) and (2), or a ‘rule’ belonging in (3) and (4) (O’Brien and Shennan Reference O’Brien and Shennan2010:8). The contribution that archaeomaterials can make towards understanding processes of technological change has increasingly come to be recognised in recent years (Erb-Satullo Reference Erb-Satullo2020; Killick and Fenn Reference Killick and Fenn2012; Schiffer Reference Schiffer2010). Proponents cite a range of applications from discovery of evidence for inventive activity among archaeological deposits (Killick and Fenn Reference Killick and Fenn2012:566; Schiffer Reference Schiffer2010:236) to reconstruction of the chaîne opératoire or life histories behind an artefact (Erb-Satullo Reference Erb-Satullo2020:36). Here I will suggest a third application: the reconstruction of technological contexts. In this capacity archaeomaterials, borrowing techniques from numerous disciplines such as earth sciences, material science and analytical chemistry, can furnish a wealth of data regarding past technologies allowing for their reconstruction (Erb-Satullo Reference Erb-Satullo2020:36; Killick and Fenn Reference Killick and Fenn2012). From this basis we can begin to infer how past technologies operated, from the physical and chemical parameters required for production to technical management and overall production strategies employed.
Reconstructing the practices employed among the copper producing traditions of the Late Bronze Age Near East presents a major challenge. At the intraregional level these practices were far from homogeneous; representing localised technological developments and adaptation to ore resources while displaying a high degree of technical complexity. Nevertheless these traditions shared the same objective: to produce copper metal at a scale that was economically viable. This required certain physical and chemical conditions to be met, achieved via various production strategies, before the copper could be extracted from an ore. These strategies can further be broken down into stages of production which as a whole can be described under the term chaîne opératoire (Torres Reference Torres2002). This concept will provide the basis for reconstructing the technological contexts of copper production in this section with the objective of identifying how each stage might have contributed towards the anomalous production of iron among the Late Bronze Age smelting traditions of the Near East. Where this approach diverges from the typical understanding of a chaîne opératoire is that as metallic iron was not the desired product of these operations, there was no intentionality of the practitioners in producing it. It must therefore be the result of other intended strategies which may form the link between extractive copper metallurgy and the transition to the use of iron. Thus not only the processes but also the intentions of each stage of production are considered. Yet to what extent are these practices archaeologically recoverable? To borrow the concept of a recipe, we can perhaps be more confident of reconstructing the ‘ingredients’, that is, a list of raw materials as well as the tools and facilities used, than the ‘rules’ or the specific actions taken and instances of problem-solving during each stage of production (Schiffer and Skibo Reference Schiffer and Skibo1987:597). Evidence for both categories is largely confined to physical remains, which presents another challenge, as the majority of smelting assemblages were deposited after the terminal stage of production. Formation processes, recovery of products and deliberate destruction or deposition often destroys the raw materials and disrupts the original contexts of smelting operations.
Technological contexts cannot be defined purely as physical or chemical processes; however, they represent the technical practices, pyrotechnologies, constructs and apparatus that together formed a recipe for the production of metal artefacts (O’Brien and Shennan Reference O’Brien and Shennan2010:8). Much of the evidence for extractive technologies in the archaeological record is fragmentary relying upon the rare survival of structures such as furnaces or non-perishable elements of some forms of bellows (Craddock Reference Craddock2000:158–160). Obtaining sufficient data to reconstruct these technological elements therefore requires close integration of site survey and analysis of artefacts with archaeomaterials techniques. Fortunately there has been a productive trend towards this level of integration over recent years which has contributed significantly towards a greater understanding of extractive metallurgy in the Late Bronze Age Near East (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020; Van Brempt and Kassianidou Reference Van Brempt and Kassianidou2016; Yagel et al. Reference Yagel, Ben-Yosef and Craddock2016; Yahalom-Mack et al. Reference Yahalom-Mack, Eliyahu-Behar and Martin2017). This section will begin by defining the technological contexts of copper production across the period in terms of the strategies employed as well as the technologies and raw materials that facilitated them.
By the middle of the 2nd millennium BCE, the art of extracting copper from native ores had reached a degree of sophistication across the Near East that is testament to the skill of the practitioners. Through the application of pyro-technology these traditions were able to exploit a wide variety of copper minerals by manipulating firing conditions, demonstrating an understanding of the behaviour of a multitude of materials at high temperatures within a strongly reducing atmosphere (Craddock Reference Craddock2000:164; Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020; Knapp and Kassianidou Reference Knapp and Kassianidou2008:144; Yahalom-Mack and Eliyahu-Behar Reference Yahalom-Mack and Eliyahu-Behar2015). In this section the technological contexts of extractive cuprous metallurgy that existed during this period are defined within the format of a chaîne opératoire. For ease of comparison, this format has been adopted throughout Sections 2 and 3 of this Element to represent a theoretical sequence of formation processes leading to the production of a copper artefact. This does not strictly reflect actual organisational or processual circumstances and simply provides a structure for reference. I begin with an overview of the ore minerals that were exploited, from the geographic distribution of deposits to ore chemistry and beneficiation, before concluding with a discussion of the extent to which exploitation of these ores may have introduced iron into the smelting process. From here the section progresses on to the types of furnaces, bellows and technical ceramics that had become established across the region. Estimated operating parameters for these pyro-technologies are presented based upon the available data for furnace construction, dimensions and artificial means of forced draught. The application of these methods is then considered in terms of the technical operations and physical and chemical conditions required for the extraction of copper from its ores. Lastly I assess the evidence for the casting of the copper metal or secondary production, primarily for the production of ingots. Of course the production of copper was not the sole objective of extractive metallurgical activity in this period which was employed to supply a breadth of products from ochres to lead, tin, speiss and precious metals (Pigott Reference Pigott, Man, Mining, Körlin, Steffens and Cierny2003:375; Roberts et al. Reference Roberts, Thornton and Pigott2009:1014–1017; Thornton et al. Reference Thornton, Rehren and Pigott2009:308; Veldhuijzen and Rehren Reference Veldhuijzen and Rehren2007:190). Many of these processes introduced large quantities of iron into pyro-technological processes raising the possibility that they played a role in the beginnings of iron metallurgy in the region. However, temperatures favourable for the extraction of iron (exceeding 1200°C) often risked volatilization of the desired product: for example, lead volatises at 1450°C, tin from 900°C and arsenic at 300-600°C (Hauptmann Reference Hauptmann2020:281,344). Furthermore the removal of undesirable components such as sulphur from these ores required oxidizing conditions that were typically insufficient to reduce iron from its ores or even relied upon its preferential oxidation (Dube Reference Dube2006:19; Lechtman And Klein Reference Lechtman and Klein1999; Pigott Reference Pigott, Man, Mining, Körlin, Steffens and Cierny2003:375). The role that non-cuprous products could have played in the beginnings of iron metallurgy is therefore unclear given that the conditions favoured for the extraction of iron are virtually prohibitive for these processes. For this reason this Element will exclusively focus on the methods for the extraction of copper that had become established by the mid 2nd millennium BCE in the Near East.
2.1 Start with the Ore
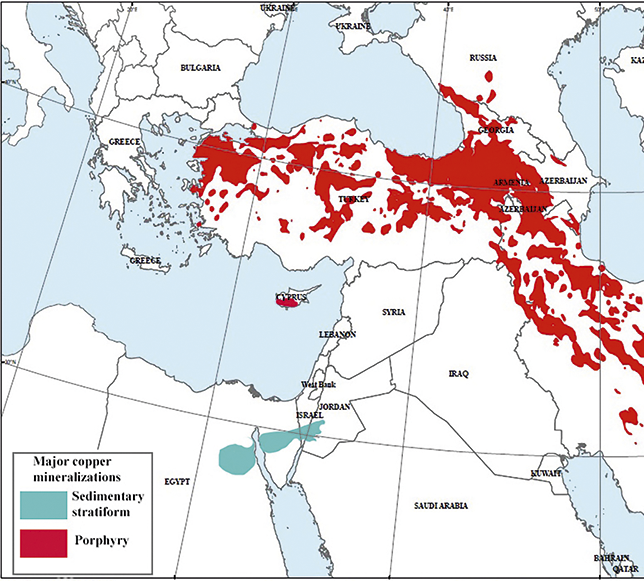
Copper is among the most abundant metals in the Earth’s crust, in weight around 300 billion metric tonnes of copper can be found in porphyry deposits globally (Kesler and Wilkinson Reference Kesler and Wilkinson2008). Even today, annual copper production worldwide amounts to just under 0.01% of this total, highlighting the relative scarcity of accessible deposits. Across the Near East, the majority of copper mineralizations originate from a ‘belt’ of porphyry mass sulphides extending east from the Aegean and into Iran at the intersection of the Euro-Anatolian and African-Arabian plates, termed the Tethys range (see Figure 1). Throughout prehistory, access to such deposits was dependent upon supergene processes of erosion and weathering causing exposure of these sulphides. Oxidation of surficial deposits of mass sulphides often result in a distinctive reddish ‘cap’ of copper and iron oxides termed a ‘gossan’, a notable example is the kokkinorotsos or ‘red-rock’ deposit in central Cyprus (Guilbert and Park Reference Guilbert and Park2007:391; Knapp et al. Reference Knapp, Kassianidou and Donnelly2001:208). The second major group of deposits begin in north-east Egypt and bisect the Sinai and southern Israel across a narrow range into Jordan. Here, the leaching of sulphide minerals through chloride ions in seawater or thermal springs formed copper carbonate hydroxides which are precipitated as crystal aggregates or banded crusts (Guilbert and Park Reference Guilbert and Park2007:24–125,774–781; Ilani et al. Reference Ilani, Flexer and Kronfeld1987:269). Typified by the Timna complex in Israel, hydrothermal solutions of copper sulphides were deposited in ‘karsts’ away from parent volcanogenic sulphides within sedimentary stratiform rocks (dolostones). The reaction of these solutions with proximal anions (i.e. silicates and carbonates from limestone) then precipitated the economically significant copper carbonate and silicate minerals (Dill Reference Dill2015). The chemistry of the ore contained throughout both types of deposit can vary considerably as a result of both hypogene and supergene processes as well as orebody displacement. Nevertheless these deposits can be characterised as belonging to two groups based on the dominant chemistry of the ores: the sulphides and the oxides. Technological adaptation to attain consistent yields from either category of ore was a determining factor in the development of extractive metallurgy across the region, with one consequence of exploiting iron-rich ores being the introduction of iron into the smelting process.
Map of Near East showing major copper mineralizations in porphyry or sedimentary stratiform rock formations. The red ‘belt’ or Tethys range extending from western Anatolia through to eastern Iran is visible, along with the smaller hydrothermal and volcanic mineralizations of northern Egypt and east across the Sinai into Jordan. Weathering of porphyry can produce copper carbonates and oxides; however, the far greater proportion of copper in these deposits is held in sulphides. Adapted from United States Government Geological survey.
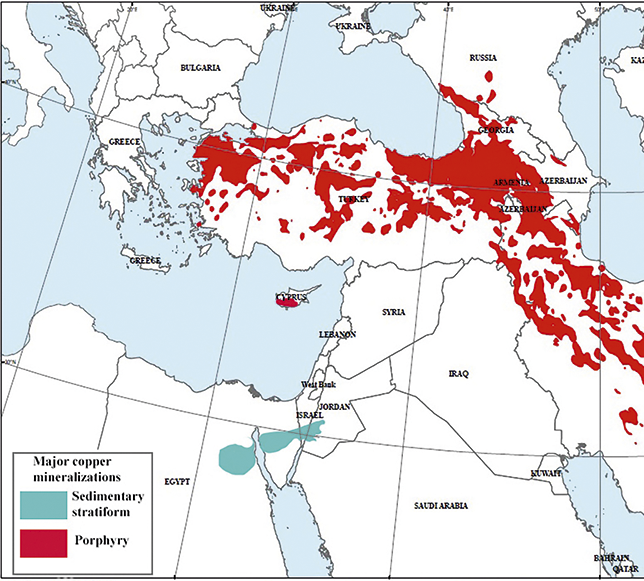
Of the sulphide group, major deposits exploited by the Late Bronze Age in the Near East largely fell within the Tethyan range from the Balkans and east across into Anatolia and northern Iran, including the southern occurrence of the Troodos ophiolite on Cyprus and extending north into the Madneuli formation in Bolnisi, Georgia (Kassianidou Reference Kassianidou2013a; Moritz and Baker Reference Moritz and Baker2019; Popkhadze et al. Reference Popkhadze, Moritz, Gialli and Jonsson2013). Copper-bearing minerals originating from these mass sulphide deposits occur as facies with other paragenetic minerals and include chalcopyrite (CuFeS2), covellite (CuS) and chalcocite (Cu2S) (Guilbert and Park Reference Guilbert and Park2007:212–220). Though early exploitation may have relied on the oxides found at the surface of these deposits, the use of sulphidic ores to the north of the region may have begun as early as the fifth millennium BCE indicated by high sulphur content in copper base artefacts of the Bulgaria hosted Varna and Gumelnitsa cultures as well as from central Anatolia at the 4th-millennium BCE site of Çamlıbel Tarlası (Lehner Reference Lehner2015:44; Ryndina et al. Reference Ryndina, Indenbaum and Kolosova1999). By the late 2nd millennium BCE their use had become fully established and formed the basis of some of the most intensive copper smelting activity in the Near East, from the isle of Cyprus to the south Caucasus (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020; Knapp and Kassianidou Reference Knapp and Kassianidou2008). An important factor in the exploitation of these ores was the introduction of considerable quantities of iron into pyro-metallurgical operations. This iron is contained both in ferrous copper minerals such as chalcopyrites and within extraneous pyrites, olivines and other iron-rich minerals sharing facies with the copper ores. As will be discussed later in the section, the iron could only be removed through beneficiation to a limited extent and conversely its presence may have been desirable due to its advantageous role in achieving superior yields of copper during the smelting process.
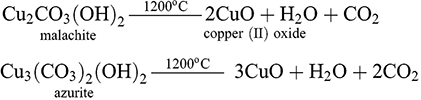
To the south of the region, the principal source of copper in prehistory was found in the abundant copper carbonates (e.g. malachite Cu2CO3(OH)2 and azurite Cu3(CO3)2(OH)2) contained within the karstic deposits extending from western Jordan and laterally across the Dead Sea rift valley into the Sinai and Egypt (Hauptmann Reference Hauptmann2007:68). The Bronze Age exploitation of these deposits is well documented with the discovery of the mining complex at Timna in southern Israel in the 1970s and subsequent projects revealing mining activity along the length of the Wadi Arabah valley into Jordan (Hauptmann Reference Hauptmann2007:68; Yagel et al. Reference Yagel, Ben-Yosef and Craddock2016). Mining was confined to the occurrences of paleokarsts or chambers containing copper carbonates located within the soft dolostones of local outcrops resulting in the excavation of distinctive narrow shafts. Copper production in proximity to these deposits has been dated from the 5th millennium BCE at Chalcolithic sites in Timna before reaching peak intensity towards the end of the 2nd millennium BCE with the introduction of new extractive technologies and organisational practices (Rothenberg and Merkel Reference Rothenberg and Merkel1998; Yagel et al. Reference Yagel, Ben-Yosef and Craddock2016). Through the same hydrothermal mechanisms responsible for the deposition of copper minerals, iron minerals originating from pyrites contained within parent mass sulphides frequently occur alongside copper carbonates in many of these paleokarsts. Iron oxides such as hematite (Fe2O3) and magnetite (Fe3O4) precipitated from solutions within karstic chambers before undergoing cementation with silicates to produce so-called ‘tile ores’ of a red hue and brickish consistency (Hauptmann Reference Hauptmann2007:68–70; Killick Reference Killick, Roberts and Thornton2014:21–22). The paragenesis of oxidic copper and iron minerals within these deposits meant their exploitation resulted in the smelting of particularly iron rich charge compositions. This can be seen in the comparison between the smelting complexes at Timna in Israel, and Wadi Faynan in Jordan. In the former locality greater quantities of slag (predominantly iron silicates) were produced during smelting operations than at Faynan, which corresponds with the higher average iron contents of the ores from deposits in proximity to Timna (Hauptmann Reference Hauptmann2007:78). Intentional addition of extraneous iron oxides may have been another contributing factor which led to rising iron contents in local charge compositions.
While ore geology may have determined the presence of iron to a greater or lesser extent during smelting, its impact upon the development of extractive iron metallurgy was limited until technological developments allowed for the maintenance of conditions sufficient to extract iron. Despite exploitation of the same deposits beginning from the 5th millennium BCE in the Near East, the production of ferrous slags during Chalcolithic smelting operations was infrequent (Bougarit Reference Bourgarit2007:5; Rothenberg and Merkel Reference Rothenberg and Merkel1998:2). Even for those traditions that began to exploit iron-rich sulphidic ores, such as the 4th-millennium BCE site of Shahr-I-Sokta in southern Iran, the production of ferrous slags remained limited (Hauptmann et al. Reference Hauptmann, Rehren and Schmitt-Strecker2003:204). This is in marked contrast to the 2nd millennium BCE which saw the deposition of vast quantities of iron rich slags at smelting sites across the region, demonstrating the impact that the introduction of new pyro-metallurgical technologies had upon these processes (Craddock and Meeks Reference Craddock and Meeks1987; Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020; Hauptmann Reference Hauptmann2007:68; Knapp and Kassianidou Reference Knapp and Kassianidou2008). Among the most significant developments was the innovation of the furnace, which was a major step towards achieving control over the reaction parameters during a smelting operation. In the following section the development of the furnace in the Near East will be explored along with other innovations that arose as a consequence of the new possibilities afforded by this technology.
2.2 Furnaces and Forced Draught
The furnace, which at its most basic level consisted of a lined chamber to contain the reactants and fuel during combustion, may have been in use in parts of the Near East from the 3rd millennium BCE (Ackerfield et al. Reference Ackerfeld, Abadi-Reiss and Yagel2020; Craddock Reference Craddock2000; Thorton Reference Thornton, Rehren and Pigott2009). Starting from shallow pits or bowls these structures had developed into thick-walled shafts of larger dimensions by the mid 2nd millennium BCE necessitating investment into forced draught to drive oxygen into these greater reaction volumes (Craddock Reference Craddock2000:161–162). Structures identified as furnaces dating to this period have been attested to at a number of metal production sites across the Near East in Israel (Berna et al. Reference Berna, Behar and Shahack-Gross2007), Georgia (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020), Cyprus (Karageorghis and Kassianidou Reference Karageorghis and Kassianidou1999:178–180), Egypt, Anatolia (Craddock Reference Craddock2000) and the Sinai (Yahalom-Mack and Eliyahu-Behar Reference Yahalom-Mack and Eliyahu-Behar2015). Generally these belong to two types dependent upon whether the majority of the shaft exists below ground level as a sub-structure or above ground level as a superstructure which hereinafter will be referred to as pit and shaft furnaces respectively. Of the pit furnaces, many examples have been excavated following recent fieldwork in Georgia at numerous late 2nd millennium BCE sites on the banks of rivers such as the Choloki, Ochkhomuri and Supsa-Gubazeuli in the Guria region (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020). These consist of pits dug into the substrate of around 1 metre in depth and diameter, floored with stones and lined in clay (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020:28). Provision of air into these furnaces may have required the use of nozzles, recovered in great numbers at the sites, to direct the blast of attached bellows down into the shaft from the surface though an alternate method requiring interlocking tubes that extended into the pits has been speculated (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020; Khakhutaishvili Reference Khakhutaĭshvili2009:63). The remains of shaft or superstructure furnaces, usually reduced to fragments of clay walls and bolstering materials, have been recovered from Late Bronze Age sites in Anatolia, Cyprus, the Levant and southern Israel (Abdel-Motelib et al. Reference Abdel-Motelib, Bode and Hartmann2012; Craddock Reference Craddock2000:164; Given and Knapp Reference Given and Knapp2003; Yahalom-Mack and Eliyahu-Behar Reference Yahalom-Mack and Eliyahu-Behar2015). These furnaces have an interior diameter between 30 and 50 cm with a tuyere or opening through which air would be supplied at the base of the shaft, angled into a shallow pit beneath the furnace walls (Craddock Reference Craddock2000:162–163; Hein et al. Reference Hein, Kilikoglou and Kassianidou2007:142–143). As extant remains of furnace walls are fragmented it is difficult to estimate the height of the shafts which may have been anywhere from 30 cm to a metre in height (Craddock Reference Craddock2000:162; Hein et al. Reference Hein, Kilikoglou and Kassianidou2007:143). The internal volume of the pit furnaces may have been greater (0.8m3) than those of shaft furnaces (0.3–0.5m3); however, the reaction volumes of both types may have been similar and are estimated at 300 litres, situated at the hottest part of the furnace during firing around a ‘cone’ of air entering from a tuyere or bellow nozzle (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020; Rehder Reference Rehder2000:75,80).
With an increase in furnace dimensions, the introduction of forced draught through the use of bellows allowed for the maintenance of the same or higher rates of reaction across a greater volume and mass of material (Craddock Reference Craddock2000:162; Killick Reference Killick, Roberts and Thornton2014:32). This required an input of energy that was an order of magnitude greater than for earlier practices, as it had to both facilitate the endothermic Boudouard reaction to maintain the partial pressures of carbon monoxide across a larger volume while also achieving temperatures that could render the greater mass of the slag liquid and mobile to prevent entrainment of the copper metal (Killick Reference Killick, Roberts and Thornton2014:34). The delivery of greater volumes of air to drive up the rate of the exothermic combustion of the carbon fuel could not be consistently achieved through natural means such as wind and the open furnace structures this method often necessitated reduced the insulation the furnace could provide (Craddock Reference Craddock2000:164; Rehder Reference Rehder2000). Only with the innovation of the bellows would the use of larger dimensions of furnaces have become feasible, with the two technologies likely converging at some point in the late 3rd to early 2nd millennium BCE in Eurasia (Rehder Reference Rehder2000). In the Near East, the use of bellows is in evidence from the early 2nd millennium BCE with the appearance of often vitrified ceramic tuyere fragments and nozzles and have been found in huge quantities in the most metallurgically active parts of the region such as Politiko Phorades on Cyprus, the Wadi Arabah complex in Israel and at the Supsa-Gubazeuli sites in the south Caucasus (Erb-Satullo, Gilmour and Khakhutaishvili Reference Erb-Satullo, Gilmour and Khakhutaishvili2014:8; Hauptmann Reference Hauptmann2020:303; Kassianidou Reference Kassianidou2011). More direct evidence has also been recovered in the form of the ‘pot bellow’, a ceramic basin which may have operated through the working of an animal hide ‘diaphragm’ attached to the opening or by a more complicated interior fan, a number of which have been found across the Near East from the mid 2nd millennium BCE (Davey Reference Davey1979; Kassianidou Reference Kassianidou2011:43). Based on reconstructions of Near Eastern examples such bellows were found by Humphris et al. to deliver around 3 litres of air per stroke estimated on the volume of the bellows at maximum extension of the hide (Reference Humphris, Charlton, Keen, Sauder and Alshishani2018:406–408). This volume, though not necessarily greater than could be provided by human lungs, was found to be sufficient to reduce iron ores into small blooms by Humphris et al. which may have been a result of more consistent delivery of air to maintain rates of reaction in the furnace (Reference Humphris, Charlton, Keen, Sauder and Alshishani2018:408; Rehder Reference Rehder1994:346).
2.3 Firing
Sections 2.1 and 2.2 established the reactants, namely the ore, to be smelted and the reactor, or furnaces, that the ores were smelted in to produce the copper metal. In this section the reaction mechanisms by which the two groups of ores identified in Section 2.1, oxides and sulphides, were exploited will be explored before moving on to the technical process employed in the smelting of the ores during the firing procedure. Following this, the evidence from Late Bronze Age smelting sites in the Near East that provides an indication of the reaction parameters achieved during firing will be assessed. As discussed in Section 2.1, the oxide group more specifically relates to the copper carbonate hydroxides (e.g. malachite Cu2CO3(OH)2, azurite Cu3(CO3)2(OH)2) which through supergene oxidation and hydrothermal activity could occur in karstic deposits in dolostones but also less abundantly within surficial proximity to mass sulphide deposits from weathering (Hauptmann Reference Hauptmann2020:24,39). The reduction of these minerals required two steps which were achieved simultaneously within the contexts of prehistoric smelting: (1) the thermal decomposition of the ore into copper oxides and (2) the reduction of these oxides in the presence of carbon monoxide to produce metallic copper (Hauptmann Reference Hauptmann2020:310–314):
 (1)
(1) (2)
(2)
Decomposition and reduction of these ores can take place at temperatures as low as 700°C (Bougarit et al. Reference Bourgarit2007:7; Craddock Reference Craddock2000:154). At these temperatures, the metallic copper would precipitate from the ore and remain in solid state as prills which would have to be collected and melted in a crucible to consolidate it into a greater mass. By the 2nd millennium BCE, developments in pyrotechnology with the introduction of forced draught and furnaces of greater dimensions allowed higher temperatures to be achieved that would render any copper in the furnace molten, allowing it to collect into one mass at the base of the furnace through density segregation which made retrieval more efficient (Bourgarit Reference Bourgarit2007:7; Craddock Reference Craddock1995:47; Hauptmann Reference Hauptmann2007:219).
The production of copper from sulphidic ores (principally chalcopyrite CuFeS2 but also chalcocite Cu2S or covellite CuS) necessitated removal of the sulphur (desulphurization) of an ore before its reduction into metallic copper (Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:2). In prehistoric smelting, this was primarily achieved through oxidation by heating the ore in the presence of oxygen to produce sulphur dioxide gas and copper oxides. Since the reduction of the copper oxides required a more strongly reducing environment with little oxygen availability these two processes could not be achieved simultaneously in the furnace; adding a degree of complexity to sulphide smelting not encountered in the smelting of oxidic ores (Craddock Reference Craddock1995:162). By the mid 2nd millennium BCE this complexity had resulted in the development of an array of production strategies for the exploitation of these ores, many of which still defy explanation (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020; Van Brempt and Kassianidou Reference Van Brempt and Kassianidou2016). Based on the evidence from these sites and from analogy with modern industrial processes, archaeometallurgists have hypothesised three potential methods for the extraction of copper from sulphidic ores in prehistory: the direct-to-copper smelt (Bougarit et al. Reference Bourgarit, Mille, Prange, Ambert and Hauptmann2003; Rostoker et al. Reference Rostoker, Pigott and Dvorak1989:74), dead roasting or ‘black copper’ smelting (Eibner Reference Eibner and Hänsel1986; Rostoker et al. Reference Rostoker, Pigott and Dvorak1989:80) and matte smelting (Craddock Reference Craddock1995:167; Rostoker Reference Rostoker1975; Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:127–129). The direct method required the (1) heating of the ore in an oxygen-rich environment to decompose the ore into a mixture of sulphides and oxides. Then in a more reducing environment with less oxygen availability, the sulphides reduce the copper oxide into metallic copper (2–3):
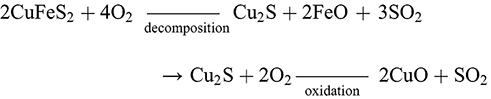 (1)
(1)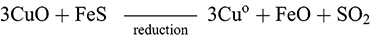
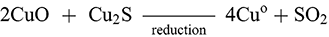 (3)
(3)This process may have begun to be exploited during the Chalcolithic with the co-smelting of oxide ores such as malachite with chalcocite and other sulphide ores, allowing the sulphide ores to reduce the copper oxides over a short firing (Bougarit Reference Bourgarit2007). This single firing method would have been inefficient without the introduction of more intentional control of the conditions starting from more oxidising conditions in the furnace and progressing towards reducing conditions as the ores convert from sulphides to oxides and then to metal, a technically demanding procedure illustrated by Chiarantini et al. in a study of a ninth-century BCE site in Populonia, Italy (Reference Chiarantini, Benvenuti and Costagliola2009). This practice does not appear to have been common among traditions of the 2nd millennium BCE in the Near East.
The ‘dead-roasting’ or ‘black copper’ process began with the roasting of the ore in an oxygen-rich environment for a long duration over which the sulphide ore would (1) decompose and the copper sulphides would gradually convert to (black) copper oxide with the production of sulphur dioxide gas (2):
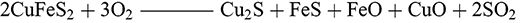 (1)
(1)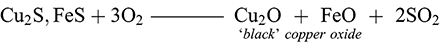 (2)
(2)This ‘black’ copper could then be collected and re-smelted in a simple reduction step within the furnace to produce copper metal:

Evidence for the employment of similar methods for the production of copper from sulphidic ores mostly exists in the form of roasting pits, found at numerous sites across the Near East from Cyprus to the South Caucasus, as well as the presence of black copper oxide in some production contexts (Fasnacht et al. Reference Fasnacht, Peege and Hedley2000; Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020; Knapp and Kassianidou Reference Knapp and Kassianidou2008). While this offers one explanation, historical accounts of dead-roasting processes stress that maintaining such a long duration of roasting was fuel intensive, bringing into question its adoption as a viable production strategy in the contexts of Late Bronze Age copper production (Percy Reference Percy1861:415).
The third method is defined by a multi-stage process in which an intermediary product called matte is made in an initial stage and then converted into copper metal in a final stage (Craddock Reference Craddock1995:165; Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011: 75, 127). Similarly to the dead-roasting process, the sulphide ore would be roasted to begin decomposition of the ore before being collected and smelted at temperatures up to 1200°C to form an intermediary product termed matte and a slag:
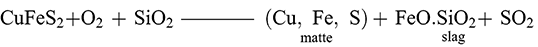 (1)
(1)
This matte, consisting mostly of copper sulphide and some iron sulphide (the iron is partially removed into the slag), could then be collected and (2) smelted in a ‘coppermaking’ stage until it was converted into copper metal via oxidation and reduction of the copper oxides:
 (2)
(2)
Many elements of the matte smelting process are shared with dead roasting, from the roasting stage to the production of a copper-rich sulphide intermediary, raising the possibility that the two processes could be virtually indistinguishable in the archaeological record. However, we can identify two main features of matte smelting that are distinct from dead roasting: the production of an iron-rich slag and two stages requiring temperatures up to and exceeding 1200°C. The first smelt requires these higher temperatures in order to render the available silica molten which upon contact causes the liquid oxy-sulphides from the ore to split into an iron-rich matte and a slag (Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:74–75). Matte smelting may have been beneficial as the production of a slag removed much of the iron from the ore before the coppermaking stage. Converting the matte could be achieved efficiently as the higher specific gravity of matte compared to siliceous slags allowed it to sit under the slag, allowing any metallic copper that precipitated from it to sink to the base of the furnace without risking entrainment in the slag (Hauptmann Reference Hauptmann2020:47; Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:84–88). Iron content in the matte also lowered its viscosity, aiding in the mobility of the copper during density segregation in the furnace (Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:82). For these reasons matte smelting can be characterised as introducing particularly high quantities of iron into the furnace during firing, perhaps in greater amounts than any other form of smelting employed during the Late Bronze Age. This raises the possibility that iron could become reduced more frequently during this process compared to other methods by sinking through the slag and being picked up by the mobile copper as it passed through the matte to the furnace base. Evidence for the matte process being employed in the Near East begins in the 4th millennium BCE at the sites of Shahr-i-Sohkta in Iran and in northern Turkey at Murgul in small scale operations (Hauptmann et al. Reference Hauptmann, Rehren and Schmitt-Strecker2003:202). By the 2nd millennium BCE this practice intensified, indicated by the large concentrations of slag heaps from successive smelting operations, large flat ‘plate’ slags of concave appearance and indicators of high temperature smelting such as vitrified furnace linings at smelting sites. These features have all been identified at sites in the South Caucasus and on Cyprus (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020; Van Brempt and Kassianidou Reference Van Brempt and Kassianidou2016).
Slags are a glassy silicate that mainly consist of structures of long polyions of acidic oxides, the principal of these being silicas which are found commonly in ancient metallurgical slags as olivines: a group of metallic (Mg, Fe, Ca etc.) silicates (Hauptmann Reference Hauptmann2020:246; Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:74). Acidic oxides have a high viscosity which is an undesirable trait during smelting, as this increases the amount of copper that becomes entrained in the slag and does not remove other unwanted acidic oxides (Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:74). The addition of basic oxides such as calcium and magnesium oxide disrupts these polyion structures and greatly reduces the viscosity of the slag and increases the solubility of acidic oxides. Similarly, when FeO enters the slag it dissociates into Fe2+, Fe3+ and O2- and these free oxygen ions react with bridging oxygen to depolymerise the slag network and lower the viscosity. The lower melting points of neutral oxides (FeO, Cu2O) in comparison to silica means their addition would further decrease the viscosity and melting point of the slag (Bougarit et al. Reference Bourgarit2007: 7–11; Kaiura et al. Reference Kaiura, Toguri and Marchant1977:157–158; Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:74–76). The lower melting point of the most iron-rich olivine member fayalite (Fe2+2SiO4) at 1200°C compared to silica at 1710°C meant that fayalitic slag was an ideal composition for prehistoric smelting as it could be rendered free running, allowing the copper to fall through the slag and collect at the furnace base (Bourgarit Reference Bourgarit2019:215; Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:74–75). In oxide smelting, the role of the slag was to collect extraneous material in the furnace preventing the entrainment of the copper metal and allowing it to collect at the base (Craddock Reference Craddock2000:154). The production of less viscous slags was therefore encouraged during firing with the addition of iron-bearing minerals in a practice referred to as slagging. The iron oxides contained in these minerals would react with the available silica from the copper ores and furnace lining to produce a highly mobile non-viscous slag (Craddock Reference Craddock2000:154; Park et al. Reference Park, Park and Sohn2011:694). If enough slag was produced to obstruct the airflow into the furnace, it could be evacuated from the interior by means of an opening in the furnace walls at the base in a practice termed tapping (Craddock Reference Craddock1995:31; Hauptmann Reference Hauptmann2020:233). Incontrovertible evidence for the addition of iron oxides has remained elusive though iron minerals have been discovered in stores at copper smelting sites in the Near East and within spoil heaps and slag scatters at Middle to Late Bronze Age sites in the Levant, Jordan and the Sinai (Hauptmann Reference Hauptmann2007:69). Before this period, the typically poor eutectic (low melting) compositions of smelting products suggests that there was no intentional addition of iron-rich minerals as a means to improve slag liquefaction (Hauptmann et al. Reference Hauptmann, Rehren and Schmitt-Strecker2003:204). Iron bearing minerals such as hematite (Fe2O3) or iron hydroxides are known to occur regularly alongside both sedimentary and metamorphic copper ore deposits leading to the characterisation of them as ‘self fluxing’ or ‘self slagging’ (Hauptmann Reference Hauptmann2020:54–56). At the exemplary sites at Timna, hematite and hydroxide (tile) ores were found sorted into piles within late 2nd to early 1st millennium BCE metallurgical installations at site 30 presumably with the intent of addition to the copper oxide charge (Hauptmann Reference Hauptmann2007:232). Setting aside the question of intentionality, the practice of slagging as evidenced by increasing quantities of iron rich slags at smelting sites dating from the 3rd to 2nd Millennia BCE likely originated within a transitional phase of extractive metallurgy in the Near East that accompanied pyro-technological innovations such as furnaces and bellows (Craddock Reference Craddock2000:157; Hauptmann Reference Hauptmann2020:203–204; Thorton et al. Reference Thornton, Rehren and Pigott2009). Following this phase full comprehension of the behaviour of slags led to the establishment of this practice by the Late Bronze Age across the region.
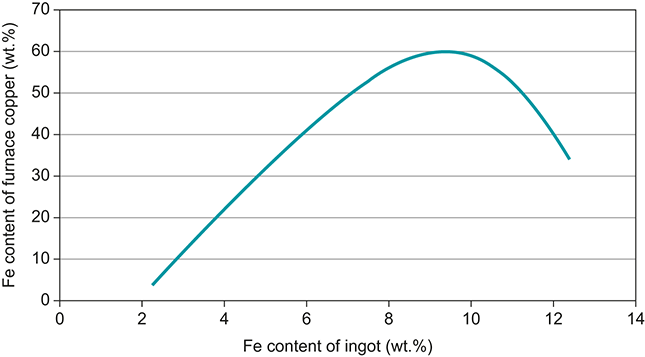
Slags can also provide an indication of the physical processes that occur during smelting (Hauptmann Reference Hauptmann2020:217). If the furnace reached temperatures sufficient to bring a large volume of slag to liquidus and render it free running this is reflected in the morphology of the slag as well as the phases visible within the matrix. The melting points of slags varies depending upon the presence of metallic oxides; however, many of the iron-rich (fayalitic) copper smelting slags recovered from Late Bronze Age sites in the Near East would have become molten around 1200°C (Schlesinger et al. Reference Schlesinger, King, Sole and Davenport2011:74). Tap slags recovered at smelting sites from this period suggest that furnaces may have regularly exceeded this temperature to allow the slag to remain free-running for some time after exiting the furnace and rapidly cooling (Erb-Satullo, Gilmour and Khakhutaishvili Reference Erb-Satullo, Gilmour and Khakhutaishvili2014:10; Hauptmann Reference Hauptmann2020:233; Veldhuijzen and Rehren Reference Veldhuijzen and Rehren2007:193–194). The glassy highly vitreous nature of some slags has also been linked to higher temperature processes, many of which have been found abundantly in the South Caucasus (Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020). Similarly, the vitrified remains of furnace linings and tuyere fragments which appear frequently at 2nd-millennium BCE sites across the Near East points to temperatures sufficient to partially melt silicates of around 1200-1400°C (Craddock Reference Craddock2000:158–162; Erb-Satullo, Gilmour and Khakhutaishvili Reference Erb-Satullo, Gilmour and Khakhutaishvili2014:6–7; Knapp and Kassianidou Reference Knapp and Kassianidou2008; Yahalom-Mack et al. Reference Yahalom-Mack, Eliyahu-Behar and Martin2017). Redox conditions during smelting can mainly be extrapolated from the ratio of oxides in the wüstite-magnetite system which is oxygen sensitive (Hauptmann Reference Hauptmann2020:268). In a more reducing environment wüstite (FeO) will be favoured, appearing more frequently in smelting slags whereas in less reducing conditions magnetite (Fe3O4) will form. The atmosphere within a furnace is not homogenous with certain regions becoming more or less strongly reducing based upon the direction of the draught and the temperature during smelting. Nevertheless the more common occurrence of either oxide can provide limited evidence for redox conditions during firing. In general, smelting operations in the Late Bronze Age Near East favoured less strongly reducing environments than those witnessed in the later true iron smelting operations of the Iron Age, with magnetite occurring more frequently in smelting slags (Craddock Reference Craddock2000; Erb-Satullo, Gilmour and Khakhutaishvili Reference Erb-Satullo, Gilmour and Khakhutaishvili2014:11; Van Brempt and Kassianidou Reference Van Brempt and Kassianidou2016; Yahalom-Mack et al. Reference Yahalom-Mack, Eliyahu-Behar and Martin2017). Nevertheless, conditions within the furnace could become substantially more reducing as evidenced by the occurrence of metallic iron prills in the smelting slags and also reflected indirectly through the rising iron contents of copper base alloys between the early to late 2nd millennium BCE, perhaps the result of iron-rich matte processes (Craddock and Meeks Reference Craddock and Meeks1987; Gilmour et al. Reference Gilmour, Cox, Erb-Satullo, Khakhutaishvili and Pollard2020:29).
2.4 Casting
Ingots provide an important source of quantitative data for copper base alloys and have been recovered in considerable quantities from Late Bronze Age contexts across the Near East, most spectacularly from the mass depositions within shipwrecks including the Cape Gelidonya and Uluburun wrecks in the eastern Mediterranean (Kassianidou Reference Kassianidou2013b:51–52; Pulak Reference Pulak and Cline1997). As the first stage in the secondary production of an artefact, retrieval of the copper from the furnace and its casting into ingots was regularly practised for ease of mobility and trade. This process would have been highly oxidizing with the exposure of the molten copper to the air within an open crucible, causing most of the waste material from the furnace still trapped in the copper (silica, iron, calcium, aluminium etc.) to precipitate on the surface of the molten copper as oxides known as dross. These less dense oxides could then be skimmed and removed from the surface of the molten copper (Hauptmann Reference Hauptmann2020:383). In the Late Bronze Age, this was likely the extent of ‘refinement’ of the copper as no evidence for investment into further refinement of copper alloys through the use of an oxygenated ‘blast’ of air has been found in the Near East (Hauptmann Reference Hauptmann2020:318). Further refinement was impractical and simply unnecessary, the trace amounts of remaining iron and other impurities being undetectable. Rising iron contents of copper base artefacts from the Middle to Late Bronze Age instead suggests that re-oxidation of the copper during secondary production remained consistent and was insufficient to remove the increasing quantities of iron entering the furnace copper (Cooke and Aschenbrenner Reference Cooke and Aschenbrenner1975; Craddock and Meeks Reference Craddock and Meeks1987). Ingot casting was just the first stage in the secondary production of an artefact and in theory the copper might be subjected to a number of other processes from alloying, re-casting and eventual recycling before deposition as an artefact. Though this may have provided more opportunity for the mechanical segregation of the iron and other impurities, if these quantities remained low enough to stay in solution their removal would be negligible. This section provided an overview of the technological contexts of extractive metallurgy in the Late Bronze Age Near East. In the following section these contexts will be explored in a series of experiments designed to reproduce the actual conditions of copper production in this period.
3 Iron in Practice
Based upon the technological contexts presented in the previous section, we can identify key developments within extractive traditions that may correlate with the increase of average iron contents in copper base alloys over the course of the 2nd millennium BCE in the Near East. The first of these is the exploitation of increasingly iron-rich ores, either as a result of natural composition or the addition of extraneous minerals, for the production of copper. With iron forming a greater proportion of the furnace charge, more iron was available in the system. Reaction volumes also increased as furnaces (reactors) of bigger dimensions were built and methods of forced draught were introduced to maintain or increase rates of reaction across these greater volumes. Raising the rate of combustion of the carbon fuel resulted both in higher temperatures and created a more intensely reducing atmosphere by favouring the Boudouard reaction with abundant carbon availability at temperatures around 1200°C. Yet without a complete production assemblage, it is impossible to assess what the actual consequences of these developments were in terms of the production of metallic iron. As discussed in Section 1.2, it is necessary to design an experiment around specific objectives or in other terms, the knowledge that the experiment is intended to generate. The primary objective of the experiments presented here was to reproduce a production assemblage for a typical Late Bronze Age copper smelt using the practices employed by traditions across the Near East. This requires the recreation of conditions subject to the same variables that affected the prehistoric process and so the application of actualistic methods is appropriate. The secondary objective was to reveal the extent to which the available iron in the furnace charge can become reduced during these smelting operations.
To this end a single variable that was subject to change in prehistoric smelting was identified: the iron content of the furnace charge, which acted as the controlled variable throughout the experiments. The experimental methodology chosen incorporated four smelting experiments and three casting operations. For each smelting experiment a twenty kilogram charge of ore was prepared and fired within a full scale reconstructed shaft furnace for an undefined period based upon the addition of the ore and fuel. The charges contained copper and iron oxides in varying proportions of a twenty kilogram total mass (see Table 1, above). The furnaces were coil-built, with clay tubes laid in successive concentric layers and kneaded and compacted around a foundation consisting of a shallow ‘bowl’ pit dug some 10 cm into the substrate with a diameter of 45 cm. This process was repeated until conical walls extended from the base to a height of 70–80 cm with a wall thickness of 15 cm. A circular passage of 4 cm in diameter was cut into the rear of the furnace (wall at top of incline facing away from front passage) about 8 cm up from the base of the walls to form a tuyere. The interior volume of the furnace can be estimated at 0.06m3 with a reaction volume in the lower half of the chamber surrounding the tuyere of around 0.03m3. A fire was set on the furnace floor and the front passage was sealed. An electric blower (motor-powered air pump), shown to deliver 0.3m3 of air per minute through testing with an anemometer, was set up to direct the blast through the tuyere at the rear of the furnace. In total each experimental smelt lasted for a duration of seven to nine hours over which time 60 to 75 kilograms of charcoal was added to the furnace, with similar rates of fuel consumption observed across all four smelts. Around two kilograms of the copper oxide and hematite/pellet ore charge was added to the furnace at roughly twenty minute intervals until the total twenty kilograms of charge had been added within the fourth hour of the smelt. After a further period of up to five hours the draught was stopped and the furnace allowed to cool before the contents were evacuated. Samples weighing two to three kilograms were then taken from the metal phases of three of the smelting assemblages and rendered molten at 1200°C before being cast into three ingots. The section begins with a comparison of the compositions of the experimental ores in comparison to those exploited during the Late Bronze Age in the Near East. The experimental firing of the ores is then assessed in 3.2 in terms of operating parameters and reactions based on observations of the smelting process and analysis of the smelting assemblages. In 3.3 the results of the metallographic analysis of the metal phases recovered from the experimental smelts are presented alongside compositional data alongside the data from a selection of Late Bronze Age furnace coppers for comparison. At the end of the Section in 3.4 the results of the ingot castings are discussed and compared to the compositions of Late Bronze Age copper base ingots recovered across the eastern Mediterranean.
| Charge (20 kg) | Fe (wt.%) | Cu (wt.%) | Fe (kg) | Cu (kg) | Ratio (Cu:Fe) |
|---|---|---|---|---|---|
| Timna (Ore only) | 7.29 | 23.7 | 1.45 | 4.74 | 3:1 |
| Timna Fluxed (3:1 ore to hematite) | 21.35 | 17.7 | 4.74 | 3.55 | 1:1 |
| CUFE 1 | 36.7 | 63.1 | 7.32 | 12.62 | 2:1 |
| CUFE 2 | 18 | 82 | 3.6 | 16.4 | 4:1 |
| CUFE 3 | 83.5 | 23.8 | 16.7 | 4.76 | 1:4 |
| CUFE 4 | 56 | 43.2 | 11.2 | 8.64 | 1:1 |
3.1 The Ores
In Section 2 the types of ore that were exploited in the Near East by the 2nd millennium BCE were discussed in relation to the distribution of geological deposits and their composition within two major groups of the sulphides and oxides. The ores selected for the four experiments were exclusively oxidic, which was prioritised over sulphides for the following reasons. Firstly, oxides are commercially available as chemicals that are close to their stoichiometric compositions. This allows for greater control over the proportions of copper and iron in the charge while also permitting greater accuracy in quantitative estimations. The smelting of oxides is also representative of coppermaking processes in the smelting of sulphides, whereby the remaining oxides from the desulphurization of the sulphide ores are reduced to produce copper metal. The final reason is that while the categorization of ores exploited during the 2nd millennium BCE in the Near East may form a basis for comparison, the chemical composition of these ores does not necessarily provide accurate quantitative data on the proportion and weight of iron to copper in the charge during prehistoric smelting. Compositional data on ores that can be considered authentic, or with a good chance of having been exploited during this period, is rare. As surficial deposits nearest to the oxidation zone, oxidic ores such as malachite and azurite can become rapidly depleted by ancient and modern mining activity leaving only the least economically viable ores by content of metal. The exceptional discovery of the New Kingdom metallurgical complex at Timna, situated within the Wadi Arabah in southern Israel, provided a rich source of non-depleted ores for chemical analysis which Tylecote et al. used to produce a compositional dataset of major and trace components (Reference Tylecote, Ghaznavi and Boydell1977; Hauptmann Reference Hauptmann2007:68). From this data, the quantitative composition of iron and copper in a hypothetical 20 kg charge of Timna sourced ores can be calculated and compared to the quantitative data for the 20 kg charges of ore fired during the series of experiments presented in this Element hereafter referred to as CUFE 1–4. The data for both is shown in Table 1, page 28.
The artificial charges (CUFE) were clearly richer in copper than the Timna ores, this is to be expected given the artificial compounds consisted of pure copper oxide (CuO), containing 79 wt.% copper in comparison to the Timna ore containing 23.7 wt.% or just over three times as much in the same weight of ore. The iron content of the artificial charges, contained within hematites and iron oxide pellet, was roughly equal to the Timna hematites used in the fluxed charge though made up a larger proportion of the total artificial charge than in the Timna charges. This disparity between the Timna charges and the artificial charges was partially offset by the smelting of smaller charges by weight in the experiments compared to speculative weights of an average charge during the smelting operations at Timna, raising the total metal contents in the furnace during the experiments to quantities approximating those added to the furnace over the course of a typical Timna smelt (Merkel Reference Merkel1990; Rothenberg Reference Rothenberg1999). In addition, the act of fluxing with the addition of hematite to the charge (row 2 of Table 1) is estimated by Merkel to have been around 1 parts hematite to 3 parts copper ore (Reference Merkel1990:112). This would have introduced up to three times more iron into the furnace during firing bringing the total iron content closer in line with the experimental charges. Given the disparity in metal content of the same quantity of Timna ore to the experimental charges the ratio of copper to iron can be used as an alternative basis for comparison. In Table 1 the experimental charge closest in proportion of copper to iron within the unfluxed Timna charge (3:1) is CUFE 1 (2:1). The closest to the fluxed Timna charge is CUFE 4, both having a roughly 1:1 proportion of copper to iron in the charge, whereas CUFE 2 and CUFE 3 represent copper rich and iron rich outliers respectively. The implications of the ratio of iron to copper in the Timna ore charges and the experimental charges in regards to the production of copper iron alloys will be addressed in 3.3.
3.2 In the Furnace
With considerable quantities of iron entering the ore charge for the experimental smelts, what was the outcome of firing such iron-rich compositions in a furnace? We can begin by defining the operating parameters of the furnaces by identifying key indicators that reflect the actual conditions in the furnace interior including temperature and rates of reduction as a function of the partial pressure of carbon monoxide. Through examination of the slags the conditions in the furnace interior can be revealed both in the morphology and appearance of the slags as well as from the composition. For all four experimental smelts the greater part of the smelting assemblage consisted of a vitreous dark grey slag containing numerous voids. The glassy appearance of the slag, which had consolidated into a distinct phase immediately above the metal in the furnace, suggests that interior temperatures had been sufficient to render much of the slag molten and mobile, allowing the copper to sink through to the furnace base. Vitrification of the furnace walls also confirmed that temperatures in excess of 1200°C had resulted in much of the silica in the clay lining becoming partially liquefied before entering the slag. Redox conditions within the furnace were reflected in the ratios of wüstite to magnetite present in the slag, which is determined by the partial pressures of oxygen and carbon monoxide. Across all of the experiments the slags contained both wüstite and magnetite in dendritic forms (see Figure 2) along with fayalite. The composition of the slags can be characterised as particularly iron-rich though the occurrence of magnetite suggests conditions were slightly less reducing than typical of prehistoric iron smelting.
Micrograph of a cross-section of slag from smelt CUFE 3. Dendritic wüstite (white) and laths of fayalite (light grey) in a glassy matrix (dark grey). Prills of copper (reddish-brown) and α-iron (white, top left) are visible.

Given the strongly reducing atmosphere and temperatures in excess of 1200°C it is perhaps unsurprising that the copper metal across all of the experiments had formed a consolidated phase with a convex base at the interface with the furnace floor, containing many voids and incorporating slag. From the larger metal phase rivulets of copper metal had percolated to the furnace floor forming ‘springy’ offshoots around the substrate. Each assemblage weighed around five kilograms and measured up to 30 cm in diameter and height. The metal phases of the assemblages can be seen to consist mainly of reddish-brown copper; however, for the more iron-rich smelts (CUFE 1, 3 and 4) inclusions of grey iron are visible ranging from a few millimetres up to >2 cm in size. The largest of these inclusions appeared to have formed in the hottest region of the furnace surrounding the tuyere, which was apparent in some of the assemblages as an opening retained in the material upon cooling (see Figure 3). Despite this the iron phases were largely dispersed throughout the copper metal and no degree of separation between the two metals was evident.
Cross-section of assemblage CUFE 4, which lay in the furnace inversely to this photograph. The tuyere is visible as the round opening in the assemblage, centre showing how the main metal phase developed just below the tuyere. Reddish-brown copper with large inclusions of light-grey iron is visible, encased within a glossy black slag.

In prehistoric iron smelting, the metallic iron reduced from ferrous oxides consolidates into a bloom which attaches itself to the furnace wall below the tuyere around the hottest and most reducing region in the furnace (Hauptmann Reference Hauptmann2020:365). Why then have the substantial quantities of iron that formed during the CUFE series of smelts entered into the copper rather than form into a bloom or become entrained in the slags? One possible mechanism for the iron to enter into the copper would be the dissolution of the iron into the copper during the firing with the solubility limit being dependent upon the temperatures achieved in the furnace. Craddock and Meeks suggest that there was ‘ample opportunity for the copper to pick up iron whilst the small droplets drained slowly through the iron-rich slag’ (Reference Craddock and Meeks1987:192). This may certainly account for some of the iron content of the copper; however, the slags themselves contained almost no metallic iron for the copper to ‘pick up’ and iron content of the copper was in some cases far in excess of the c.6 wt% of iron that is soluble in copper at firing temperatures (Turchanin Reference Turchanin, Agraval and Nikolaenko2003:315). Another explanation is that this phenomenon may arise from the close densities of the two metals (copper has a specific gravity of 8.95 and iron 7.85) which would limit the degree to which the two could separate in the furnace, causing them to form a contiguous metal phase beneath the slag (Bachmann Reference Bachmann2016:31). While this may have some bearing on the segregation of the two metal phases in the furnace, the iron would remain in solid state at firing temperatures whereas the copper would reach liquidus, making it substantially more mobile than the iron. A difference of 1 in the specific gravity of a phase is proficient for density segregation for slags and mattes, mattes and speiss or speiss and metal in the furnace (Hauptmann Reference Hauptmann2020:271). The denser and more highly mobile copper would surely be able to separate from the iron into a distinct phase during firing, leaving the less mobile iron to become entrained in the slag. Alternatively, both metals may be produced within the most reducing region of the furnace, causing the iron from the ore to be reduced and enter instantaneously into the surrounding copper phase as γ-iron at temperatures exceeding 910°C before changing to α-iron upon cooling (Cooke and Aschenbrenner Reference Cooke and Aschenbrenner1975:259). This region, located in proximity to the tuyere, contains the highest partial pressures of carbon monoxide due to the increased rates of reaction of carbon dioxide and carbon from the Boudouard interaction. This is perhaps demonstrated by the assemblage shown in Figure 3 where the largest inclusions of iron in the copper occur around the tuyere. Regardless of the mechanisms for the entry of the iron into the copper, substantial quantities of iron were able to enter the copper across all of the experiments. With the entry of so much iron into the copper, it is necessary to evaluate whether this phenomenon was the result of a freak event, mismanagement of the furnace during firing or if it was a regular and unavoidable occurrence during the co-smelting of iron-rich charges with copper ores. This first explanation appears unlikely given the consistency in firing strategies employed for all of the smelts which all produced copper containing iron. Mismanagement during firing is a possibility that was raised by Merkel (Reference Merkel1982) and Tylecote et al. (Reference Tylecote, Ghaznavi and Boydell1977) with the ‘overblowing’ of the furnace in the presence of excess carbon causing the more frequent reduction of iron from available oxides which enters into the copper (Craddock and Meeks Reference Craddock and Meeks1987:201). While this may apply to the experiments to a certain extent with the clear reduction of much of the iron oxide into iron, the equal ratio of magnetite to wüstite in the smelting slags indicates that the atmosphere was less strongly reducing than typically achieved during prehistoric iron smelting. In fact the slags were largely indistinguishable from prehistoric copper smelting slags in composition, with wüstite only occurring in proximity to the most iron-rich metal phases. Contrary to a random event or result of mismanagement during firing, iron contents of the copper appeared to correlate directly with the proportion of iron in the charge which will be demonstrated in the following section.
3.3 The Metal
Co-reduction of the copper and iron oxides during the experimental smelts resulted in the production of a large copper metal phase containing substantial quantities of metallic iron. This iron exists as abundant inclusions in its alpha allotrope (α-Fe) within the copper metal. Within the temperature range of prehistoric smelting, much of the iron upon reduction may have entered the copper as its beta (γ-Fe) allotrope before changing to α-Fe or ferrite upon cooling to room temperature. Across all of the smelts this iron was present as numerous oblong and spheroidal phases within the copper metal (see Figure 4), producing a bi-phasic alloy with various degrees of magnetism based upon iron content. In 3.2 based upon the dispersal of the iron in the metal phase in proximity to the most reducing region of the furnace, co-reduction of both metal oxides within this zone was speculated to be the mechanism for entry of the iron into the copper. Yet to what extent is this reflected in the metal?
Furnace copper from smelt CUFE 2. On the left the large iron phases (white) are visible as oblong and spheroidal inclusions in dense formations within the copper (red-brown). On the right the much smaller dendroidal inclusions of iron (white-grey) that has precipitated from solution are present.

Proceeding from 1200°C, only around 6 wt.% iron can be dissolved in the copper. From 1100°C this would reduce to 2.8 wt.% iron in solution, with the remainder precipitated as a solid phase in the ratio of 28.5:1 parts copper to γ-Fe. From 1094°C the isothermal (peritectic) reaction of the liquid copper alloy and precipitated iron results in a solid solution with a peritectic composition of 96.0 wt.% copper and 4.0 wt.% iron. Below this temperature the peritectic alloy is completely solid and the 4 wt.% of iron begins to re-precipitate from the alloy as it cools, entering in a finely divided state within the copper (Cooke and Aschenbrenner Reference Cooke and Aschenbrenner1975:259). By 835°C this precipitated γ-Fe transforms into ferromagnetic α-Fe in a fine dispersal of small dendroidal inclusions. Though this may account for some of the iron content of the metal assemblages (see Figure 4, right image) it is clearly insufficient to account for the much greater quantities of iron entering into the copper far in excess of the c.6 wt.% of precipitated iron from solution.
Analysis of the phase structure of the iron inclusions revealed infrequent instances of spherical ‘meshes’ of α-Fe contained within the copper which resemble the formations of iron in the blooms and smelting slags of prehistoric iron smelting (see Figure 5). This may further support the theory that the iron oxides were reduced along with the copper in the same region of the furnace, immediately becoming entrained in the larger copper metal phase as it formed. Dispersal of the iron once it had entered the copper may have been caused by the greater mobility and density of the molten copper which allowed the iron to consolidate into the main metal phase at the base of the furnace before it became trapped in the slags immediately above this phase.
Micrographs of metal samples from experiment CUFE-1, left and an iron bloomery smelting slag, right. In both examples the iron metal (white) has formed a ‘mesh’ within the copper (red-brown) or smelting slag (dark grey) suggesting that the iron oxides present were reduced into iron metal directly within the copper or slag phases.

In order to relate the charge compositions of the CUFE series of experimental smelts to the iron contents of the metal assemblages, p-XRF sampling was conducted using the cross-sections prepared for the optical microscopic analysis. This ruled out non-representative sampling from surficial enrichment or contamination by extraneous materials. An average iron and copper content (wt.%) was calculated from five samples taken from each cross-section and are displayed in Table 2. For the third experiment CUFE 3, two compositions are given to represent the most iron-dense formations and the least within the cross-section. With the iron contents of the experimental assemblages established, this forms a basis for comparison to furnace coppers of the Late Bronze Age Near East. As a raw material that in prehistory was subject to secondary production, raw or furnace copper is rarely encountered in the archaeological record. Fortunately a ‘spill’ of raw copper, interpreted as a store of raw copper before secondary production, was found near a structure at Site 3 in the Timna New Kingdom metallurgical complex (Yagel et al. Reference Yagel, Ben-Yosef and Craddock2016:43). This copper cannot be directly related to a single smelting operation and may have been waste material from particularly iron-rich smelts that accumulated over time to form a single large deposit. As such this copper may not be representative of the average composition of furnace coppers produced during these smelting operations. By comparing the iron contents of this copper to the copper produced by Tylecote et al. (Reference Tylecote, Ghaznavi and Boydell1977) using the Timna ores, the extent to which these compositions are indicative of an average copper smelt at Timna can be assessed. The copper and iron compositions for the Timna furnace copper, the experimental copper produced by Tylecote et al. (Reference Tylecote, Ghaznavi and Boydell1977) and the metal assemblages from the experiments presented in this Element are displayed in Table 2.
| Furnace Copper | Cu (wt.%) | Fe (wt.%) |
|---|---|---|
| CUFE 1 | 87.2 | 12.3 |
| CUFE 2 | 96.2 | 3.59 |
| CUFE 3 (Cu-rich) | 59.2 | 39.7 |
| CUFE 3 (Fe-rich) | 18.9 | 78.4 |
| CUFE 4 | 65.1 | 34 |
| Experiment 7 no flux | 97.9 | 2.1 |
| Experiment 7 w. Fe flux | 76.1 | 23.9 |
| Experiment 10 no flux | 99.6 | 0.4 |
| Experiment 10 w. Fe flux | 76 | 24 |
| Experiment 13 no flux | 98.8 | 1.2 |
| Experiment 13 w. flux | 71.1 | 28.9 |
| Timna 346 | 98 | 1.85 |
| Timna 329 | 90 | 4.1 |
| Timna 345 | 85 | 7.15 |
| Timna 347 | 87.5 | 5.5 |
| Timna 348 | 90 | 0.33 |
| Timna 349 | 94 | 0.51 |
| Timna 350 | 89 | 2.9 |
| Timna 351 | 83.5 | 9.72 |
| Timna 353 | 92 | 2.52 |
| Timna 354 | 90 | 4.84 |
| Timna 355 | 89 | 0.72 |
| Timna 331 | 96 | 2.7 |
Beginning with the experiments of Tylecote et al. the smelting of an unfluxed (no addition of hematite) Timna ore containing 7.29 wt.% of iron resulted in a copper-iron alloy containing 0.4–2.0 wt.% iron (Reference Tylecote, Ghaznavi and Boydell1977). This shares the smallest disparity with experiment CUFE 2 which produced an alloy containing 3.59 wt.% iron from a charge with an iron content of 18.2 wt.%. This might be expected given the artificial charge contained just over twice as much iron as the Timna ore. When fluxed to create a charge containing equal parts copper and iron, the experiments by Tylecote et al. produced an alloy containing around 25 wt.% iron. This correlates closest to experiment CUFE 4 which produced an alloy containing 34 wt.% iron from an ore ratio in the charge of roughly equal parts, resulting in an alloy containing 2:1 copper to iron in comparison to the 3:1 copper to iron ratio in the fluxed Timna alloys. Taking the results of smelts CUFE 2 and CUFE 4 as the range of expected compositions for the smelting of a Timna ore, we can then draw comparison to the furnace coppers recovered at Site 3 at Timna. In Table 2 the compositions of the twelve samples of furnace copper range from an iron content of 0.33 to 9.72 wt.% iron with an average contents of 3.57 wt.%. These alloys are thus very similar in composition to the alloy produced for experiment CUFE 2, likely being produced from a charge containing around four parts copper to one part iron oxides. The most iron-rich furnace copper, Timna 351 containing 9.72 wt.% iron, is closest in composition to the alloy produced from experiment CUFE 1, possibly being produced from a charge containing as much as two parts copper to one part iron. In tangent with the experimental alloys, from the compositions of the furnace coppers of Site 3 at Timna we can define the average smelt at the complex as the firing of a charge containing up to 20–30% of iron oxides in a ratio of around one parts iron oxide to three parts copper oxide. Firing conditions were sufficiently reducing to render much of these oxides into iron metal with a conversion rate of up to 30% of the iron oxides in the charge, resulting in a copper-iron biphasic alloy with an average iron content of 3 wt.% with iron-rich outliers containing as much as 10 to 30 wt.%. If iron was able to enter the copper in such quantities during the firing of iron-rich ores, to what extent is this reflected in the compositions of copper base artefacts of the Late Bronze Age? This question will be addressed in the final part of the section.
3.4 Casting
As we’ve seen in the previous sections, the entry of iron into the copper during the firing of iron-rich charges resulted in the production of copper with an iron content that could in rare cases exceed 10 wt.%. This raises the question of why iron is not evident in greater quantities in contemporary copper base alloys, a discrepancy noted by Craddock and Meeks: ‘the question should be why do most copper alloy artefacts from the Bronze Age on contain so little iron?’ (Reference Craddock and Meeks1987:192). Discussed in Section 2.4, despite an increase in the iron content of copper base artefacts from the Middle to Late Bronze Age in the Near East, the average iron content remained only around a few weight per cent. This indicates that the iron content could be reduced by as much as 10 weight percent during the casting of furnace coppers into ingots. To evaluate whether this was practicable within the scope of a single casting procedure, analysis of the metal produced during an experimental crucible melting and casting of the CUFE furnace copper was carried out using p-XRF sampling and the results are shown in Table 3.
| Ingot Section | Cu (wt.%) | Fe (wt.%) |
|---|---|---|
| CUIN 1 (CUFE 4) | 87.4 | 12.4 |
| CUIN 2 (CUFE 2) | 97.5 | 2.25 |
| CUIN 3 (CUFE 3) | 90.9 | 8.73 |
For CUIN 1, the iron content of the furnace copper from CUFE 4 was reduced by 22 wt.%, remaining the most iron-rich in the ingot series. CUIN 2 saw a reduction of 1.34 wt.% iron, the lowest loss of iron during the casting as expected given the furnace copper from CUFE 2 contained the least iron. CUIN 3 was subject to by far the greatest loss of iron of 70–30 wt.% (sample dependent) showing an even more drastic reduction than in CUIN 1 despite the furnace copper containing up to 35 wt.% more iron. The variation between iron contents in the ingots reveals two major implications for the casting of copper iron biphasic alloys. In the most iron-rich alloys the larger phases of iron face greater exposure to the atmosphere with the copper being insufficient to prevent contact with oxygen. Rapid oxidation of the iron disrupted the casting procedure of CUIN 3 with the production of excess dross as black oxides. This produced a copper containing less iron than CUIN 1, where the greater proportion of copper in the metal prevented oxidation of the smaller iron phases (see Figure 6). Secondly, below 5 wt.% of iron in the copper, removal of the iron becomes exponentially more difficult within the contexts of a single casting operation (see Figure 7), allowing a few percent of iron to persist in many copper base artefacts even after multiple castings (Craddock and Meeks Reference Craddock and Meeks1987:188). Further removal of the iron (refining) would require increased oxygen potentials from a blast of oxygen-enriched air: a feat for which there remains no evidence among prehistoric practices (Hauptmann Reference Hauptmann2020:317).
Macrograph of ingot section CUIN 1, x1.5 objective. Iron (light grey) in copper (reddish-brown).

Graph showing relationship of iron content of experimental furnace coppers (CUFE) to ingots (CUIN).
The discovery of copper-base ingots dating to the 2nd millennium BCE across the Near East at production sites and from their deposition during transit provides a far more extensive record of copper compositions before further alloying than the rare occurrences of raw or furnace coppers. With the removal of over three quarters of the iron content of the experimental furnace copper during casting (see Table 3), the compositions of these prehistoric ingots can be re-evaluated with regards to iron content. Copper and iron compositions for a selection of ingots from southern Israel, Jordan and the Sinai (based on chemical provenance) are listed in Table 4.
| Ingot description/code | Speculative origins | Cu (wt.%) | Fe (wt.%) |
|---|---|---|---|
| METB31 (Timna, Site 2) | Timna, 2nd Mil. BCE | 87.8 | 2.49 |
| METB32 (Timna, Site 2) | Timna, 2nd Mil. BCE | 90.1 | 1.86 |
| METB33 (Timna, Beer Ora) | Timna, 2nd Mil. BCE | 86.5 | 1.83 |
| METB34 (Sinai, Bir Nasib) | Bir Nasib, Sinai, 2nd Mil. BCE | 56 | 25 |
| METB35 (Deir Alla, Jordan) | Timna, 2nd Mil. BCE | 91.9 | 1.48 |
| METB52 (Hazor, Israel) | Faynan, Jordan 2nd Mil. BCE | 92 | 0.41 |
| METB67 (Hazor, Israel) | Timna | 86.8 | 0.14 |
Iron content within ingots may not necessarily originate from metallic iron phases, with much of the iron occurring either as iron sulphides or oxides in trapped matte or slag inclusions. This limits the use of quantitative analyses for iron content to determine if metallic iron was extracted during the original smelting operation that produced the copper in an artefact, though microstructural analysis of the ingots in Table 4 revealed that much of the iron was indeed present as metal phases within the copper matrix (Merkel and Barrett Reference Merkel and Barrett2000; Roman 1990; Yahalom-Mack et al. Reference Yahalom-Mack, Galili and Segal2014:167). This allows for closer comparison of these ingots with the compositions of the experimental ingots presented here. Starting with the most iron-rich ingot, METB34 contains two times the iron content of the most iron-rich experimental ingot CUIN 1. Yahalom-Mack et al. characterise this ingot as a primary product of an unsuccessful smelt, which given the similarity of the iron contents to the most iron-rich experimental furnace coppers (CUFE 3 and CUFE 4, see Table 2) appears reasonable (Reference Yahalom-Mack, Galili and Segal2014:172). It is also possible this ‘ingot’ was cast directly from the furnace via tapping into a pit or crucible with no secondary casting of the metal, limiting the exposure of the iron to oxidation. The charge composition of the smelt that produced ingot METB34 must have been extremely iron-rich likely containing iron and copper in equal parts. METB31 and the ingots containing >1 wt.% iron correlate closest to the composition of the experimental ingot CUIN 2. These ingots may have derived from furnace copper with 3 to 4 wt.% iron content, smelted from a charge containing four parts copper to one part iron oxides, which could not be lowered below 2.5 wt.% within a single casting. The remaining ingots containing <2 wt.% iron may have derived from less iron-rich copper while a prolonged casting process may have oxidised more of the iron which was more efficiently segregated into and removed in the dross. From this basis we can begin to re-evaluate the evidence from copper base alloys of the late 2nd millennium BCE to reveal the consequences of the smelting of increasingly iron-rich charges in the more reducing conditions of the Late Bronze Age furnace. In many cases, iron must have comprised a minimum of a fifth of the total metal content of the charge, often forming a bi-phasic metal alloy with the copper with a composition between 3 and just over 10 wt.% iron. The implications of this are explored in the following section.
4 Iron in Perspective
In section three the consequences of the developments in extractive metallurgy that were established by the 2nd millennium BCE across the Near East were demonstrated with the experimental production of copper-iron biphasic alloys. The experimental casting of these alloys revealed that this phenomenon may have been far more frequent than anticipated from the compositions of Late Bronze Age copper base artefacts based on the rapid oxidation and removal of this iron within a single casting procedure. Given the regular occurrence of metallic iron in copper metal produced via these extractive methods, what were the implications of this for metal production in the region? At this stage, the production of iron-rich copper was an undesirable outcome of the drive to improve copper yields through the consolidation of the metal into a single distinct phase in the furnace while limiting entrainment of the copper in the slag or matte. These alloys were disadvantageous for casting as the mass could reduce significantly with the loss of iron through oxidation, a process that could be so volatile that the forming oxides would disrupt the process. The working of copper-iron biphasic alloys is plausible as both metals largely retain their properties which can result in a greater tensile strength and yield strength than pure copper (Yamashita et al. Reference Yamashita, Koga and Kawasaki2021). This presents an intriguing scenario for an experimentation with these alloys; however, their use in the production of artefacts is not known from the 2nd millennium BCE, occurring only in the rare instances of the ‘bun’ shaped ingots (see Table 4, METB34) found sporadically across the eastern Mediterranean. What role such a problematic material could play in the later establishment of iron metallurgy across the region is therefore ambiguous. This leaves two routes whereby this phenomenon could have developed into the later bloomery methods of iron extraction, firstly via the direct production of iron through anomalous events during smelting previously termed the ‘adventitious’ theory, or through a process of observation and innovation within existing production strategies to gradually eliminate copper from the charge with the goal of producing iron. The adventitious route will be assessed in 4.1 in comparison to the results of the experiments detailed in Section 3.
Though we can identify the potential of the extractive traditions of the Late Bronze Age Near East to produce iron, this does not account for how this anomaly might have instigated a process leading to the establishment of new practices to exclusively extract iron at a scale comparable to the contemporary production of copper. A first step to addressing the ‘gulf’ between an anomaly and an invention is to conceptualise this as a process incorporating two stages which can be defined as ‘observation’ and the ‘mode of response’ (Arthur Reference Arthur2007:276; Roberts and Radivojevic Reference Roberts and Radivojević2015:301). As an inherently cognitive event, demonstrating acts of observation of phenomena among past societies may appear implausible. Yet archaeologists are in a position to assess the potential for observation to occur through revealing the existence and extent of anomalies within the formation processes associated with a technology (Roberts and Radivojevic Reference Roberts and Radivojević2015). In Section 4.2 I determine the most probable scenario for the discovery of iron in copper-iron biphasic alloys and assess the likelihood and frequency of its observation among the extractive traditions of the Near East over the course of the 2nd millennium BCE.
The second stage represents the exploitation of an anomaly achieved through the manipulation of formation processes to reach the desired outcome (Shortland Reference Shortland, Bourriau and Phillips2004:2–3). Within the contexts of a singular event, evidence for this is unlikely to be visible in the archaeological record (Schiffer Reference Schiffer2010:240). However, this can be viewed as the starting point that initiates a longer process of innovation based upon trial and error, with modified practices yielding new observations which then proliferate novel strategies and investment. This extended process is far more likely to survive in the archaeological record (Schiffer Reference Schiffer2010:240; Walsh et al. Reference Walsh, Riede and O’Neill2019). In 4.3 I outline the innovations required for the extractive traditions of the Late Bronze Age Near East to extract iron at a scale comparable to the established bloomery methods employed by the 1st millennium BCE in the Near East. From this basis we can determine the minimum requirements for those traditions to produce practicable quantities of iron and begin to document the kinds of changes in production strategies that would permit the transition to the wholesale production of iron.
4.1 Direct to Iron?
Theories placing the origins of extractive iron metallurgy within the metallurgical practices of earlier traditions have circulated since the early twentieth century (Rickard Reference Rickard1939). Resulting narratives asserted that these traditions which had developed over the course of the Bronze Age in Eurasia were uniquely situated to exploit associated technologies and practices for the production of iron (Pickles and Peltenburg Reference Pickles and Peltenburg1998:79–80). Nowhere else it seemed were the necessary parameters for the reduction of iron from ferrous minerals achieved across the breadth of human endeavour (Killick Reference Killick, Roberts and Thornton2014:32–35 Pleiner Reference Pleiner2000:212–213; Wertime and Muhly Reference Wertime and Muhly1980:6). Often termed the ‘adventitious’ discovery of iron, from the outset these theories first hinged upon analogy with historic industries. After observing the production of a large sintered mass of iron nicknamed a ‘bear’ or ‘salamander’ during lead smelting in Iran in an expedition in the 1960s, Theodore Wertime and colleagues raised the possibility that a similar event may have been responsible for the development of iron metallurgy among copper smelting traditions (Arab and Rehren Reference Arab, Rehren, Stöllner, Slotta and Vatandoust2004). This prompted numerous theories to propose that iron may have been produced as a ‘by-product’ during the smelting of copper (Craddock and Meeks Reference Craddock and Meeks1987; Gale et al. Reference Gale, Bachmann, Rothenberg, Stos-Gale and Tylecote1990; Tylecote and Boydell Reference Tylecote, Ghaznavi and Boydell1977; Wertime and Muhly Reference Wertime and Muhly1980:16), copper slags (Tholander Reference Tholander1971), lead (Healy Reference Healy1982; Pickles Reference Pickles1988:10) or copper matte production (Shell Reference Shell1997). Following the surge of interest in the copper production complex uncovered at Timna in Israel headed by Beno Rothenburg, there was renewed interest in the by-product theory (Gale et al. Reference Gale, Bachmann, Rothenberg, Stos-Gale and Tylecote1990). In a series of experiments smelting local iron-rich ores, Tylecote et al. (Reference Tylecote, Ghaznavi and Boydell1977) and Merkel (Reference Merkel1983) were able to produce copper-iron biphasic alloys. Gale et al. expanded upon this work to conclude that a series of iron objects enriched with copper found in a New Kingdom Temple of Hathor at Timna may have derived from a similar context, stating: ‘Iron artefacts dating between 1318 and 1156 BCE, found both at Site 2 and at the site of the Hathor Temple contain noticeable amounts of copper. There is a strong indication that the iron used to make these artefacts was produced locally as an adventitious by-product of copper smelting’ (1990:189). The possibilities raised by the discoveries at Timna led to the re-evaluation of early iron at other sites across the Near East from Cyprus (Kassianidou Reference Kassianidou1994) to Mesopotamia (Moorey Reference Moorey1999:279) and the Levant (Waldbaum Reference Waldbaum and Pigott1999:39). Yet this hypothetical provenance would come to be dismantled by Merkel and Barrett who re-analysed the Hathor artefacts using EPMA and discovered the copper became rapidly depleted towards the core of the objects (2000). The authors were forced to conclude that the high copper contents of the Timna artefacts likely resulted from surface enrichment, detected during the initial XRF analysis, originating from corroded copper artefacts that lay in close proximity. In the absence of direct evidence for the production of iron artefacts from metal derived from copper smelting, many seminal works on the origins of iron metallurgy expressed doubt over the feasibility of the ‘adventitious’ route. In ‘The Coming of the Age of Iron’ (Wertime and Muhly Reference Wertime and Muhly1980), ‘Early Metal Mining and Production’ (Craddock Reference Craddock1995) and ‘Iron in Archaeology’ (Pleiner Reference Pleiner2000) the authors all claimed that deriving workable iron from a copper smelt was implausible. These arguments cited the abundant impurities that iron produced in this context would contain, which would necessitate removal via refinement, a process deemed too technically demanding for Late Bronze traditions to achieve (Craddock Reference Craddock1995:255; Wertime and Muhly Reference Wertime and Muhly1980:14–15). This has led to the general recognition that while the extractive technologies fostered within preceding copper smelting practices remain the best candidate for the conception of extractive iron metallurgy, the process by which this might have occurred is obscure, with the ‘adventitious’ theory being irreconcilable with the impracticability of iron derived from copper smelting barring the hypothetical consolidation of a purer ‘bloom’ or ‘bear’ of iron under certain conditions (Killick Reference Killick, Roberts and Thornton2014:33; Pigott Reference Pigott1981:82–84). The application of theories such as Shortland’s (Reference Shortland, Bourriau and Phillips2004) ‘hopeful monsters’ concept might provide new relevance for the adventitious route to iron especially as a model for how extreme technological outliers arising from atypical conditions may have a delayed influence upon technological change as an early source of analogy for later observation and experimentation during the inventive process.
Based on the experiments detailed in Section 3, these arguments can be re-assessed through comparison to the series of full scale and duration smelts undertaken by the author. With the entry of the majority of the metallic iron into the main metal phase in the furnace, there appears to be no opportunity for the iron to separate into an isolated mass that would resemble the ‘bear’ phenomenon witnessed in historical processes. As the main metal phase lay at the base of the furnace, density segregation even over an extended period of time would not facilitate separation of the two metals. With the formation of both metals immediately around the tuyere, any metallic iron formed from the reduction of proximal oxides became entrained in the phases of metallic copper, preventing its consolidation into a bloom and attachment to the furnace wall below the tuyere. Deriving sufficiently pure iron metal from this context for working into an artefact appears totally unfeasible with the exception of the most iron-rich experimental smelt, CUFE-3. In this experiment an alloy containing an average 60 wt.% Fe was produced with some of the phases of iron consisting of spheroidal inclusions large enough to be mechanically separated from the main metal phase. With copper being considerably less soluble in iron than vice versa, this iron may have contained only a few percent of copper. This remaining copper could be removed with the addition of carbon, which greatly lowers the solubility of copper in iron as demonstrated by Tylecote et al. (Reference Tylecote, Ghaznavi and Boydell1977:331), who successfully isolated pure iron from a copper-iron biphasic alloy through heating with charcoal (Busolic et al. Reference Busolic, Parada and Parra2011:33). However, producing a metal phase containing so much iron must have been atypical for these processes. Intentionally smelting copper from a charge composition containing four parts iron to one part copper is an extremely unlikely scenario and alternately if the objective was to produce iron there would be little reason to retain any copper minerals in the charge. The ‘adventitious’ mode of discovery can therefore be ruled out as there was no direct method for deriving metallic iron from the contexts of a Late Bronze Age copper smelt. Clearly, attaining iron from such contexts would have required considerable investment into refinement of the products, processes that were not facilitated by pre-existing practices for the production of copper.
4.2 Discovering Iron
The point at which an anomaly becomes actionable and is able to transform the existing structure must be determined by the capacity of the individual to perceive it. In humans, the ability to identify differences is largely dependent on physiological constraints which may differ slightly between individuals based on the sensory stimulus (i.e. eyesight, hearing), yet research indicates is mostly constant across a population (Eerkens and Lipo Reference Eerkens and Lipo2005:320–321). The other factor which can be described as the ‘logical structure of individual cognition’ relates to the selection of the most ‘useful’ parts of the stimulus data, as the total amount of data available from our sensory organs is too much to process in a given moment (Eerkens and Lipo Reference Eerkens and Lipo2005:321; van Doorn et al. Reference Van Doorn, Van de Grind and Koenderink2020:3–7). This results in a ‘perception bias’ which may stem from cognitive functions such as individual/automatic learning, for instance through the extrapolation of analogous information to interpret a novel context (deBeaune Reference de Beaune, Coolidge and Wynn2009:6) or from social/cultural learning, which may be facilitated through imitation of others or through social direction (Boyd and Richerson Reference Boyd and Richerson2005:19–35). This also may be subject to individual variation; however, we can expect considerable homogeneity within the same cultural and social contexts where the lived experience of individuals will be similar and concepts, ideas and technical skills are freely transmitted. If we accept that human perception remains as a constant in a given population, we can estimate the likelihood of an anomaly being perceived based upon the size or intensity of that phenomenon relative to the existing structure (Eerkens and Lipo Reference Eerkens and Lipo2005:321). The greater the anomaly the more disruptive it will be and the greater the chance that it falls within the threshold values of perception for a population. With the application of analytical methods from archaeomaterials, the direct comparison of products to formation processes is possible, allowing archaeologists to reveal the magnitude and extent of such anomalies (Roberts and Radivojevic Reference Roberts and Radivojević2015:301). The reproduction of production assemblages during the experiments demonstrates the strengths of a combined approach, presenting a rare opportunity to assess the point at which the incidental production of iron may have fallen within perceptible parameters of the copper smelting traditions of the Late Bronze Age Near East. As the earliest point of contact and engagement with the material, retrieval of the main metal phase from the furnace once the smelting process had completed must have been the first opportunity for the practitioner to directly observe phases of metallic iron in the metal. The extent that these phases of iron metal would be visible to the naked eye is determined by numerous factors ranging from their size, quantity and distribution within the copper metal. From observation of the experimental assemblages it is clear that these factors correlate directly to the proportion of iron in the metal. Upon retrieval of the metal assemblage from the furnace, surface oxidation and accruing slag also obscures the visibility of any iron phases present. This could have affected visibility variably; however, further processing of the assemblage would have provided ample opportunity to observe the numerous exposed surfaces of the metal phase encountered on the experimental assemblages. Of the 6 wt.% of iron that can be dissolved in copper at 1200°C, discussed in Section 3.3, we can infer that almost none of the iron precipitated from solution would be visible in the copper given its dispersal as minute dendrites of iron in the copper (see Figure 8, left image). Copper produced from a charge composition of four parts copper to one part iron is an unlikely scenario to lead to the discovery of iron though infrequent larger phases of iron may have been present.
Macrographs (x1.5 objective) of cross-sections from samples taken from experimental assemblage CUFE-2 measuring 25 cm in length, left and CUFE-1 measuring 35 mm, right.

Beginning around >10 wt.% iron content in the copper, the perceptibility of iron in the copper increases as the dimensions of the phases of α-iron begin to exceed c.1 cm diameter from 30 wt.%. The minimum range of detectability can be fixed between 10 and 30 wt.% iron with the widely dispersed and frequent inclusions becoming clearly visible as a distinct grey metal. To achieve the production of copper with an iron content that fell within this range the charge composition would need to contain anywhere from two parts copper to one parts iron up to an equal or even greater part of iron. Once the iron content exceeded 40–50 wt.% the iron phases are readily detectable with the largest inclusions possibly being mechanically extricable from the mass. The production of copper with such a high iron content would require the charge to contain at least twice as much iron as copper, surely an exceptional circumstance if the objective was to produce copper. From the limited evidence available for furnace copper, producing copper with an iron content greater than 10 wt.% may have become a semi-regular occurrence in parts of the region including the Timna and Wadi Faynan complexes by the mid to late 2nd millennium BCE (Tylecote and Boydell Reference Tylecote, Ghaznavi and Boydell1977; Yagel et al. Reference Yagel, Ben-Yosef and Craddock2016:46; Yahalom-Mack et al. Reference Yahalom-Mack, Galili and Segal2014:167). Furnace copper with an iron contents within this range likely saw a reduction in iron contents to within a few per cent during casting suggesting that it was a far more frequent occurrence than anticipated even from the limited examples of furnace copper available for analysis. Rare instances of extremely iron-rich ingots such as the Bir Nasib ingot (see Table 4) containing 24 wt.% iron further reinforces this hypothesis as a product of these processes, despite representing an outlier.
Having established the perceptibility of the anomalous production of iron at the scale of the individual, it is possible to place this event in the wider contexts of the copper producing traditions of the Near East over the course of the 2nd millennium BCE. Estimates for the total yields in metal produced at a site, complex or over space and time can be calculated by dividing the predicted weight or volume of slag produced during a single smelting operation by the sum total contained within any given deposit. From this figure, the number of smelting operations represented by the deposit can be multiplied by the expected yield in metal from a single smelt to provide a rough estimate of the total yield over the lifetime of the deposit. Here I consider the typesites of Timna in Southern Israel and Wadi Nasib in the Sinai. Beginning with an expedition to the site in 1959 by Beno Rothenburg, the exploitation of the karstic deposits of copper oxides at Timna has long been recognised as one of the most significant examples of copper production during this period throughout the Near East (Rothenburg Reference Rothenberg1999). Multiple phases of occupation and activity at the site, comprising numerous ‘camps’ dedicated to the production of copper, proved challenging when constructing site chronologies (Yagel et al. Reference Yagel, Ben-Yosef and Craddock2016). Nevertheless three main phases of activity at the site have been identified beginning with a sparse Chalcolithic presence during the 4th millennium BCE before becoming most active during the Ramesside dynasties towards the end of the New Kingdom period (1550–1070 BCE) under Egyptian influence (Yagel et al. Reference Yagel, Ben-Yosef and Craddock2016). Revised dates attained for smelting sites in the area revealed that the majority of these belonging to the late 2nd millennium BCE lay in the north of the Arabah valley and consisted of small and undefended clusters of structures with more dispersed slag scatters and deposits than those encountered during the Iron Age phases (Yagel et al. Reference Yagel, Ben-Yosef and Craddock2016:46–47). In all, five sites are believed to have been active during this period: sites 2, 3, 13, 14 and 15. At a single site, it is estimated that around 120 tonnes of copper was produced within a 150-year period and that total production across the entire Timna complex during the Ramesside period may have reached up to 1000 tonnes of metal (Hikade Reference Hikade1998:48). When applied to the five sites identified by Yagel et al. (Reference Yagel, Ben-Yosef and Craddock2016) this provides a total yield of 600 tonnes over a 150-year period. If the iron content of the copper produced during these operations fell within the ranges of the two closest experimental alloys CUFE-1 and CUFE-2, to the compositions obtained for furnace coppers at Site 13, this would give an average contents of 8 wt.% Fe. This provides a total estimate of around 48 tonnes of iron produced over a 150-year period or around 350 kilograms produced each year. Though the intensity of production at the Timna complex fluctuated with intermittent phases of abandonment and re-occupation, even over a relatively short period substantial quantities of iron could have been produced, rendering its observation among local traditions highly probable. At Wadi Nasib in the western Sinai, smelting activity took place at camps occupied from the reign of Amenemhet III (1842–1794 BCE) up to the New Kingdom period exploiting the local carbonate ore deposits. In total around 5000 tonnes of copper may have been produced during the 2nd millennium BCE based on estimates drawn from the slag heaps in the locality (Hikade Reference Hikade1998:46). Using the Bir Nasib ingot (METB34, see Table 4) as an extreme example, over a 200-year period the total yield of 1500 tonnes of metal produced with the same composition would contain a staggering 300 tonnes of iron. If the copper produced at the site contained a more modest average of 12 wt.% Fe then around a single tonne of iron may have been produced annually over the same period. These estimates provide some insight into the scale of the extraction of iron over successive smelting operations at these sites. Once an anomaly had become so prevalent it would certainly fall within the thresholds of human perception, actively requiring management. As demonstrated in Figure 9, the casting of copper containing over 30 wt.% iron is subject to the violent re-oxidation of this iron which greatly hinders casting attempts. Managing this problem to prevent flawed castings required an understanding of iron as a distinct component that could enter the copper during smelting and result in this undesirable occurrence.
Photograph of ingot CUIN-3. Rapid oxidation of the iron in the CUFE-3 furnace copper disrupted the casting process despite resulting in copper containing just a few percent of iron.

4.3 Setting the Stage
Given the frequency and scale of the anomalous production of iron among the extractive traditions of the Near East as demonstrated in the previous sections, there is sufficient basis that we can be confident that those traditions had become aware of this phenomenon to a varying extent. Moving from this awareness to a ‘solution’ or means of exploiting this development was described by Usher as the setting of the stage or a ‘[m]ediated contingency of a systematic effort to find the solution by trial and error’ (Arthur Reference Arthur2007:275). Before attempting to model this process, it is necessary to define what solutions were required to permit the exclusive extraction and production of iron. For this we can rely upon the practices of later traditions, after the problems inherent to extractive iron metallurgy had been resolved and this technology had became established across Eurasia. As methods for the production of iron remained broadly consistent from the beginning of the 1st millennium BCE up until around 1000 CE there is a wealth of evidence available both in the archaeological and experimental literature that allows us to reconstruct this process far more confidently than for copper production before this period (Crew Reference Crew, Dungworth and Doonan2013; Killick Reference Killick, Roberts and Thornton2014; Pleiner Reference Pleiner2000). Beginning with ores, total elimination of any copper-bearing minerals in the charge is required to prevent the entrainment of iron phases in the copper. Even as little as c.10 wt.% copper in the metal phase is sufficient to disrupt the production of a ‘bloom’ as demonstrated in experiment CUFE-3. Achieving the total elimination of copper from a charge may have been more problematic than expected for the extractive traditions of the Late Bronze Age Near East. Many of the ore deposits exploited by these traditions contained copper and sulphide minerals originating from volcanogenic mass sulphides, discussed in Section 2.1. Thus iron-bearing minerals in situ to these deposits, ranging from the mixed karstic deposits of the Wadi Arabah to the Gossans of the Cyprus ophiolites and Central Anatolia, could without beneficiation contain considerable quantities of copper (Hauptmann Reference Hauptmann2007:43; Knapp and Kassianidou Reference Kassianidou, Knapp, 67Blake and Knapp2005).
The basic elements of the shaft furnace as employed during the Iron Age were largely established during the 2nd millennium BCE. Differences in materials, construction and dimensions did not affect the function of the furnace with reaction volumes likely remaining fairly consistent within the scope of a single smelting operation, containing the maximum quantity of ore that could be heated and reduced into metal before being retrieved and forged into a bloom (Craddock Reference Craddock2000:162). Very little innovation was required for the furnace of the Late Bronze Age to be repurposed to produce iron exclusively as a result. Methods for forced draught, having developed in tandem with the furnace, were likely sufficient to achieve the temperatures and reducing atmosphere across the reaction volume of the furnace to reduce iron-bearing minerals into iron metal. The bloomery process of the Iron Age does not appear to have represented a revolution in technology then, rather it was a transformation of technical practices. The first major difference in firing strategy was the longer duration of the firing required during bloomery smelting to consolidate the iron metal into a bloom, which remained in a less mobile semi-solid state at the temperatures achieved in the furnace. This was not necessarily more fuel intensive than copper smelting which is less fuel efficient than iron smelting over the same duration. Typically for the production of a 4 kg bloom Crew (Reference Crew, Dungworth and Doonan2013) noted a firing period of around nine to ten hours was required, which compared to the firing time necessary to reduce and consolidate copper into a metal phase may have been up to 40% longer (Crew Reference Crew, Dungworth and Doonan2013; Pleiner Reference Pleiner2000:135). Operation of the bellows or means of forced draught may have required alternate strategies to increase the partial pressures of carbon monoxide in the furnace required to reduce the iron from its oxides. The dangers of ‘overblowing’ the furnace, noted by experimental practitioners such as Crew (Reference Crew, Dungworth and Doonan2013) can result in lowering the pressure of carbon monoxide leading to excessive losses of iron into the slag as oxides. If too little oxygen is delivered into the furnace this will lower the rate of combustion and will not favour the boudouard reaction, also causing the partial pressure of carbon monoxide to fall below ideal parameters for reduction. The extent that these factors would require new technical strategies to avoid is difficult to ascertain, often amounting to a considerable challenge in experimental attempts when compared to the experimental smelting of copper (Crew Reference Crew, Dungworth and Doonan2013).
Once the bloom had formed within the furnace, its retrieval required a radically different procedure from copper smelting. Over the course of firing, the bloom becomes attached to the rear wall of the furnace directly below the tuyere due to the limited mobility of the forming iron phases (Pleiner Reference Pleiner2000:136). In the vicinity of the tuyere where the ambient temperature can reach up to 1600°C these phases can be rendered molten and gradually move towards the furnace base before consolidating with proximal iron phases and turning solid once temperatures drop below the tuyere. The most efficient means of removal is to breach the furnace while the iron contents remain at welding temperatures (1400°C) and detach the bloom from the furnace interior, aided by the mobility of the slag at heat. The bloom can then be forged with percussive strikes to weld the iron at the phase boundaries into a solid mass and expel accruing slag (Hauptmann Reference Hauptmann2020:366). This is perhaps the most technically demanding aspect of the bloomery system, with the easiest viable alternative being the recovery of the bloom once the furnace has cooled, before heating it once more and forge welding it. This method risked excessive losses of the iron due to oxidation in the furnace and entry into the slag over the period of cooling, with successive heating losing a greater mass of the iron to re-oxidation (Crew Reference Crew, Dungworth and Doonan2013:36). From the comparison with bloomery smelting methods, it is clear that two developments in the extractive metallurgical traditions of the Late Bronze Age Near East were required to permit the production of iron at commensurate scales with copper: the elimination of copper-bearing minerals from ore charges and new technical strategies for the retrieval of the iron. Returning to theory, the next section hypothesises the cognitive process that may have led to these new production strategies being adopted by the extractive traditions of the late 2nd to early 1st millennium BCE Near East.
5 Iron in Theory
From the conclusions drawn in the previous section it appears that the copper-producing traditions of the Near East must have encountered and had the opportunity to observe smelted iron in phases with furnace and cast copper metal with semi-regularity over the course of the 2nd millennium BCE. Though a direct means to derive workable iron from a copper smelting context is less probable than an indirect process of elimination of copper from the ore charge, neither hypothesis is viable without first reconstructing how observation of this anomaly and the mode of response could instigate the invention of extractive iron metallurgy via the bloomery method. This process can be defined as the succession of interactions between people and the given anomaly that gave rise to a new technology. In Section 1.3, the last step in Schiffer’s scheme to reveal invention in the human past was to ‘formulate research questions in behavioural-not archaeological-terms’ or to relate technological change to the activities that took place in the life history of an artefact (Reference Schiffer2010:236). Here the application of theoretical models may be instrumental in allowing us to reconstruct these activities to find the most plausible scenario for these inventive processes. Over the past few decades, ongoing attempts to explain the diversification of human culture have led to the development of numerous theories that have contributed towards the construction of theoretical approaches towards explaining technological change (Erb-Satullo Reference Erb-Satullo2020:38–40). In this section I apply the three major theoretical approaches of behavioural, evolutionary and cognitive archaeology to attempt to explain a hypothetical process that may have led to invention of the bloomery method for the production of iron among the copper producing traditions of the Late Bronze Age Near East.
5.1 Iron by Analogy
If the production of iron only required a modification of existing extractive practices but not unprecedented technologies, the mechanism behind the development of methods for the extraction of iron may lie within a mental process described as ‘analogical reasoning’ in cognitive psychology, outlined in Section 1.3. This process can be expanded into three steps which de Beaune defines as:
1. A ‘source’ situation stemming either from shared knowledge passed on generation to generation or from the subject’s individual experience is stored in long term memory. 2. Transfer of knowledge is used to solve a technical problem, either through insight or through the scanning of possible solutions in the memory. This translates into an activation of previous knowledge, or transitory representations and presupposes a mental projection of the result of the action. 3. The new solution is stored in the memory to serve later as a ‘source’ situation. (Reference de Beaune2004:151)
As the new solution becomes the source situation this results in the acceptance of the novelty by the group and its transmission assuming favourable technical, social and psychological contexts. If we apply these steps to the contexts of copper smelting among the extractive traditions of the Near East, upon encountering the anomalous production of iron and its entry into the main copper phase, the source situation that the individual could draw upon would be the existing extractive methods for the production of copper which largely originated from shared knowledge transmitted within those traditions (O’Brien and Bentley Reference O’Brien and Bentley2011:317). In the second step, the solution to the ‘technical problem’ of deriving workable iron from the firing could be found in the similar yet non-identical source situation of the extraction of copper, and transferred to the new situation so that iron-bearing minerals would be chosen to make up a greater or total part of the furnace charge with the new goal of obtaining iron. Direct observation of this phenomenon, with the entry of increasing quantities of iron metal into the copper as iron-bearing minerals formed a greater proportion of the charge, would reinforce this analogical deduction through positive confirmation until becoming fully recognised. Once this transferral was complete, the solution of firing iron-bearing minerals exclusively would become the new source situation, facilitating its adoption and transmission within extractive traditions. This process may have been gradual, relying on successive experiences that formed as new contingencies arose which added new components to the source situation. Crucially, as the decision to adopt the solution as a new source situation from which to act relies upon favourable technical, social or psychological contexts it remains conditional upon existential factors (de Beaune Reference de Beaune2004:151). The presence and detection of an anomaly will in itself not necessarily lead to an instance of analogical reasoning and the achievement of a new technology if acting upon it does not result in a desirable outcome for the individual or society at large.
5.2 Iron by Selection
The model of analogical reasoning undoubtedly addresses one of the core cognitive mechanisms that facilitates the modification of existing practices to achieve new outcomes but for those innovations that lay outside of the shared knowledge and experience of a tradition it may not be sufficient to explain the broader series of interactions that lead to their development. The retrieval of an iron bloom during firing and the percussive welding of the iron grains into a billet, described in Section 4.3, had no close analogy within the shared corpus of experience of the extractive traditions of the 2nd millennium BCE in the Near East. This could only have been achieved after an extended series of complex interactions, alluded to by de Beaune as the storage of experience for analogical reference upon encountering new problems, which may benefit from a model that addresses the broader contexts of the developmental process (Reference de Beaune2004:150). The behavioural approach exemplified by Schiffer (Reference Schiffer1996, Reference Schiffer2004, Reference Schiffer2005; Reference Schiffer2010; Schiffer and Skibo Reference Schiffer and Skibo1987) and colleagues maintains that invention operates upon the selection of variants possessing favourable ‘performance characteristics’. These characteristics relate to the behaviours of a material or activity that allow it to fulfil a function to a greater or lesser extent (Schiffer and Skibo Reference Schiffer and Skibo1997). Thus under the behavioural model, the technical choice to eliminate copper-bearing minerals from the charge and also to retrieve the iron bloom at heat and forge weld it would produce a material with superior formal properties, resulting in a favourable performance characteristic for these activities (Schiffer and Skibo Reference Schiffer and Skibo1997:31). Whether or not a performance characteristic is positive is determined by the selective contexts which may be immediate, that is, any activity directly related to the life history of an artefact, or extended as any activity that is coupled to the immediate selective context (O’Brien and Bentley Reference O’Brien and Shennan2010:312). Thus the drive to reproduce those actions with the most desirable performance characteristics under these selective contexts acts as a selection pressure, reducing variants until the new variant activity is replicated and adopted as a new technology (Schiffer Reference Schiffer2010:238). In this way, the variant activities of exclusively firing iron-bearing minerals and the retrieval of furnace contents at heat may have become selected for their positive performance characteristics in obtaining workable iron before being fully integrated as persistent technical components into the extractive metallurgical traditions of the Near East.
Yet the anomalous production of iron cannot have possessed a favourable performance characteristic, while the objective of these traditions remained the production of copper. This discrepancy is mirrored in the analogical reasoning model of de Beaune, which requires a mental projection of the desired outcome before the transferral of advantageous knowledge from the long term memory (de Beaune Reference de Beaune2004:151). Conversely, it was actually a negative performance characteristic arising from the introduction of furnaces, forced draught and the addition of iron-bearing minerals to the charge, the consequences of which included the disruption and complication of casting procedures, excess weight of copper base alloys and susceptibility to corrosion of the iron phases contained within. The reason these practices persisted regardless is explained by Schiffer, as ‘[s]election pressures in the immediate selective context lead to artifacts that embody design compromises of many kinds, as in trade-offs between performance characteristics pertaining to manufacture, use, and maintenance processes … or even between activities within a given process. Compromises are necessitated because, ordinarily, no single design can maximize an artifact’s entire set of activity-specific performance characteristics’ (Reference Schiffer1996:654). Consequently even though these practices favoured the extraction of iron, only once the objective became the production of iron instead of copper could those traditions begin to select for those performance characteristics among their behaviours that favoured this new goal. This adoption of a new objective or in other words a change in the selective contexts is referred to by Schiffer (Reference Schiffer1996) as stimulated variation (O’Brien and Bentley Reference O’Brien and Bentley2011:312–313). This places the onus on identifying what caused the selective contexts to change and whether this change occurred within the immediate or extended selective contexts or through interactions between the two.
A variant with ideal performance characteristics becoming selected and adopted just once is extremely improbable, with its implementation likely leading to emergent performance problems that in turn stimulate the creation of more variants and re-selection for favourable attributes to overcome these problems (O’Brien and Bentley Reference O’Brien and Bentley2011:314). This may lead to a succession of inventions, selection of the best variants among these and then adoptions that Schiffer (Reference Schiffer2005) describes as cascades. Recognition of performance problems is not restricted to the immediate or extended selective contexts of an artefact; indeed, O’Brien and Bentley point out that it often may come from the user: ‘consumers are the selective context’ (Reference O’Brien and Bentley2011:313). In the case of the development of new practices for the manufacture of iron implements, as iron came to be adopted as a material to replace copper base alloys it would increasingly become selected for properties that facilitated this function which were directly impacted through the extractive methods used in its production. The percussive expulsion of slag and consolidation of the iron grains in the bloom through welding, described in Section 4.3, represents one such practice that could have developed through a cascade as the poor performance characteristics of iron that was not treated in this way became recognised by smiths. Inventors may adopt numerous different variants to solve performance problems as they arise, potentially resulting in technological diversity at every stage in the life history of an artefact (O’Brien and Bentley Reference O’Brien and Bentley2011:314). Any technology which is preceded by a diverse array of variants is therefore likely to have developed from a cascade process.
5.3 Iron by Error
Selection also plays a prominent role in evolutionary schemes of technological change with the adoption and transmission of certain ‘traits’ or cultural information as part of the dual inheritance model (O’Brien and Shennan Reference O’Brien and Shennan2010:9). Yet evolutionary archaeologists emphasise that selection is not the cause of variation, which can only be generated through individual experimentation or errors in the transmission of information which can be likened to mutation and transcription errors in the genes of organisms respectively (O’Brien and Bentley Reference O’Brien and Bentley2011:318). In Section 1.3, I suggested that the evolutionary model that best represents the development of a technology from an anomaly is that of guided variation. This model removes social learning as a factor in the generation of variation, reducing the impact that errors in transmission may have and instead attributing variation largely to individual experimentation (O’Brien and Shennan Reference O’Brien and Shennan2010:10). The individual can then compare the results of this trial and error to the environmental data obtained to judge whether an alteration in practices yields an adaptive advantage before deciding to adopt the new behaviour or retain the behaviours learnt from unbiased transmission, perhaps within a tradition (O’Brien and Bentley Reference O’Brien and Bentley2011:317). Guided variation may proceed from an individual experimenting with behaviours learnt and replicated from a tradition or cultural group, with this experimentation being stimulated by observation of an anomalous outcome produced through replication of these learnt behaviours (O’Brien and Bentley Reference O’Brien and Bentley2011:317). From the conclusions drawn in Section 4 it is clear that a process of guided variation could certainly apply to the development of methods for the production of iron among the extractive traditions of the Late Bronze Age Near East. Section 4.2 demonstrated that there was ample opportunity for individuals among those traditions to observe iron in phases within the copper, which then could have stimulated individual experimentation. Copy errors in replicating existing practices also may have resulted in the infrequent production of copper alloys containing over 30 wt.% iron, contributing greatly to its perceptibility. Experimentation, possibly facilitated through analogical reasoning and selection of advantageous behaviours discussed earlier in the section, could then have led to the modification of these practices with the elimination of copper bearing minerals and retrieval and forging of an iron bloom during firing.
At first glance, a major shortfall of the application of this model is that the transition to the use of iron in the Near East appears to be far more rapid than could be explained through the typically gradual process of guided variation (O’Brien and Bentley Reference O’Brien and Bentley2011:317). This process remains slow as it is dependent upon individual efforts to generate variation through trial and error but also may be constrained by individual learning biases (O’Brien and Bentley Reference O’Brien and Bentley2011:317). Where specific selection forces are absent, populations will tend to select variants that favour these biases especially if guided variation is weak or individuals are presented with little opportunity or motivation to experiment and transmit new variants (O’Brien and Bentley Reference O’Brien and Bentley2011:317). These biases are defined by O’Brien and Shennan as ‘[t]he predictions of a cost-benefit model in which individual fitness is the index of success’ and so leave ‘[l]ittle opportunity for the evolution of behaviours that benefit the group as a whole’ (Reference O’Brien and Shennan2010:10). The reticence of extractive traditions in the Near East to select variants in favour of the production of iron, while the continued production of copper was of greater cost-benefit to the individual, might explain why iron wasn’t adopted sooner even when its anomalous production became increasingly perceptible. Another outcome of this may have been the divergence and creation of multiple innovations that were ‘locally optimal’ (O’Brien and Shennan Reference O’Brien and Shennan2010:11). A possible example of this may be the Late Cypriot (ca.1200 BCE) bronze riveted iron daggers found on the isle of Cyprus, where localised iron production may have supplied an emerging market of the novel and prestige objects in the sub-region (Kassianidou Reference Kassianidou1994). This begs the question, how can a process of guided variation in a multi-modal adaptive landscape, where multiple variants may be viable, produce the cohesive package of innovations (recipe) that accompanied the rapid adoption of iron in the Near East during the late 2nd millennium BCE?
In answer, Mesoudi and O’Brien suggest that individuals would, through guided variation within the multi-modal adaptive landscape, converge upon multiple innovations that represented locally optimal ‘peaks’ (O’Brien and Shennan Reference O’Brien and Shennan2010:11). Then via indirect bias, the individual or tradition may select from among these innovations the globally optimal peak in imitation of the most successful variant in their environment. Thus the optimal innovations that originated from guided variation may be transmitted through indirect bias resulting in ‘behaviourally homogenous local populations’ which permitted the much more rapid evolution and adoption of group-beneficial behaviours (O’Brien and Shennan Reference O’Brien and Shennan2010:10). In the contexts of the extractive traditions of the Late Bronze Age Near East, this model would predict multiple local innovations across the region over a long time period that resulted in the limited and localised production of iron. This pattern would continue until those cultures began to display an indirect bias towards a particular production strategy, perhaps favouring iron that is more amenable to working as it began to displace copper base alloys for use in implements. This bias would result in the selection of globally optimal innovations such as the exclusion of copper-bearing minerals from the charge and forging of the bloom at heat. Through this process of selection and adoption, a recipe for the bloomery method of iron extraction may have developed which was quickly adopted and contributed towards locally homogenous production strategies becoming established by the 1st millennium BCE.
6 Conclusions
Arguments over diffusion and independent invention ignore the complex metallurgic crafts leading to iron.
Over the past half century since Wertime made his titular statement, invention among past societies has increasingly come to be viewed as the product of human engagement with the material world. It is through these interactions that knowledge is generated but crucially also transmitted between individuals and within traditions, for which there was no means of learning other than direct participation and personal instruction. At the beginning of this Element I argued that developments within the extractive copper traditions of the 2nd millennium BCE in the Near East remain the most likely explanation behind the advent of iron in the region by the turn of the millennium. These traditions presented the ideal conditions for the process of invention to occur, moving from observation of the new with the anomalous production of iron, to a response drawing upon a shared corpus of experience and ending with the adoption of a complete recipe for extractive iron metallurgy. I advocated for an approach with three objectives: firstly to establish the technological contexts of those extractive traditions and then to employ analogy to reveal the potential of those contexts to initiate a process of invention before relating the findings back to theory to uncover the cognitive and social mechanisms behind this process. Having achieved these objectives, what insights can we extrapolate from the results?
This Element first sought to establish the technological contexts of extractive metallurgy across the region. Section 2 charted major developments in the 2nd millennium BCE with the employ of furnaces of greater dimensions than in the previous millennium and the related introduction of forced draught. These technologies accompanied new production strategies that encouraged the smelting of increasingly iron-rich charges arising from exploitation of mixed sulphide ores or the addition of iron oxides to promote slag viscosity. In Section 3, the outcome of these developments was demonstrated with the experimental production of copper-iron biphasic alloys under actualistic conditions from charges of variable iron content. These results suggest that all of the factors identified in Section 2 would have contributed towards the more frequent co-extraction of iron alongside copper during firing, largely confirming the academic consensus that these developments present the best hypothetical explanation for the apparent increase in iron content of copper base alloys over the course of the Middle to Late Bronze Age in the Near East. What was not anticipated was just how much iron could enter into the copper during these practices and how widespread this occurrence might have been, resulting from the loss of much of this iron over even a single casting procedure to consistently low quantities in the copper.
If this anomalous co-production of iron was indeed the route to cultures of the Near East becoming iron using, Section 4 presented two developmental trajectories through which this phenomenon could have instigated the advent of iron over the course of the 2nd millennium BCE. The first and most widely speculated hypothesis of a direct to iron process is not corroborated by the experiments undertaken in this Element. At no point during the experimental firing of the mixed charges was iron isolated from the main metal (copper) phase in the furnace in sufficient quantities to permit the limited manufacture of goods. As a consequence, no opportunity was presented for detached grains of iron to become welded into larger masses or for these grains to be collected and forge welded into small ‘blooms’: a conjectured alternative for the deriving of metallic iron from the contexts of a copper smelt (Killick Reference Killick, Roberts and Thornton2014:33; Pigott Reference Pigott1981:82–84). More significant is that the direct to iron route fails to address how this infrequent occurrence could have led to the development of new practices for the production and retrieval of iron among the extractive traditions of the Near East. This does not preclude the role that the anomalous production of iron could have played in this process, however, as this phenomenon would have become increasingly perceptible as iron-bearing minerals formed an ever-greater proportion of the charge. This opens the way for a second trajectory whereby the extractive traditions of the Near East modified existing practices to exploit this newly emergent anomaly.
The conceptual ‘leap’ between the observation of iron as phases within copper and the response, which must ultimately have resulted in the new practices of exclusively firing iron-bearing minerals and the post-firing retrieval and forging of blooms, can only be understood as a function of human cognition. Transferral of the known domain of the extraction of copper from its ores to the new domain of iron production may have facilitated the early experimentation that later gave rise to novel technical strategies. Emergent practices would have become subject to a selective environment whereby their capacity to fulfil a desired outcome, the production of iron, led to a transmission bias in favour of those deemed most optimal. Viewed in isolation this theoretical process of selection may appear unilinear and deterministic, a criticism that has often been levelled at evolutionary and behavioural schemes of technological change, yet that would be to ignore numerous intervening factors which at any time may have altered the eventual outcome (Erb- Satullo Reference Erb-Satullo2020:40; Wylie Reference Wylie, Skibo, Walker and Nielsen1995:206). From a behavioural perspective, a change in selective contexts was necessary before selection among existing variant practices could have created new innovations via stimulated variation (Schiffer Reference Schiffer1996). In the case of developing new strategies for the extraction of iron, we can conceptualise this as the shift in priorities for local traditions from the production of copper, for which the reduction of excessive amounts of iron was deleterious, to the exclusive production of iron. This change in selective contexts may have been triggered by cultural and social factors rather than the functional fitness of the new technology. Even at the cognitive level the committal of only those situations that prove to be advantageous into the long term memory as ‘source situations’ for future analogical transfer are largely dependent on the social environment of the individual. Transmission of a selected variant that favoured this new objective was also far from inevitable as the introduction of copy errors and bias relating to methods of learning may have intervened in the selection process with varying results.
This is perhaps exemplified by the discrepancy between the typically slow and localised forces of guided variation, whereby individuals transmit knowledge gained through individual learning leading to the alteration of activities over the course of time, with the seemingly rapid spread of extractive iron metallurgy in the region at the end of the 2nd millennium BCE. The rapid adoption of new practices for the extraction of iron may be a prime example of selection operating unevenly across the region upon a diverse range of strategies as a consequence of existential factors. Within this multi-modal adaptive landscape, for which we can identify outliers such as the Late Cypriot trade in bronze riveted iron daggers as an example of a local adaptation (Sherratt Reference Sherratt and Karageorghis1994), the appearance of a homogenous package of strategies for the production of iron may have displaced local strategies as producers strove to imitate the best actor. This may have been triggered by a shift in priorities as producers sought to fulfil a new demand for iron, leading to a cascade in innovations until a superior strategy was established. As discussed in Section 5, the driving force behind this reorientation of priorities may have come from changes in the selective contexts, many of which have been addressed over the past century of scholarship. These range from tin shortages to fuel economy, the material advantage of iron and the disruption of sociopolitical structures in the region (Erb-Satullo Reference Erb-Satullo2020:559–560). More significant still may be the development of increasingly sophisticated techniques for the secondary production of iron. As smiths became increasingly skilled with the shaping and forge welding of iron, the need for greater quantities of iron and the appreciation for metal that was of a superior quality would have become ever more apparent. This demand began with consumers who desired iron goods that could displace those of copper, which in turn caused secondary producers to seek a superior product from primary producers. Revealing the factors that lay behind these changes in selective contexts among local extractive traditions towards the end of the 2nd millennium BCE will be crucial to future attempts at charting the advent of iron in the Near East. The promising trend among recent studies of integration of site excavation with archaeomaterials and experimental archaeology as a component of research methodologies may be instrumental in achieving this objective.
To return to the concessions made by Basalla (Reference Basalla1988) at the beginning of the Element, we can certainly conclude that iron was not necessarily invented for the manufacture of those implements which for the majority of the 2nd millennium BCE had been facilitated by copper base alloys in the Near East. Second, it is beyond doubt that modern civilization is an iron-centric civilization that has carried ferrous metallurgy to a high state of development. Third and foremost, however, iron is not uniquely suitable or necessary for all roles and to all people, at all times. When searching for those deposits we might expect to contain evidence for the invention of extractive iron metallurgy we should be mindful of these truths or risk conflating the consequences of its adoption with the circumstances of its invention. It may be likely that smelted iron was discovered in a limited and local capacity and put to some small use long before making the transformative impact it would come to have upon the cultures that came to adopt it wholesale. Nor did its earliest appearances necessarily lead to the mature technologies associated with the major expansion in its use during the 1st millennium BCE, with chance discoveries, transient experiments or even a short-lived opportunistic trade in iron potentially becoming abandoned as interest fluctuated. In this the transition to the use of iron can be seen mirrored in modern technologies, with the telephone being adopted by only 42% of United States households in the early 1900s since its invention decades previously. By contrast, in 1990 the mobile phone achieved the same penetration after just five years in circulation (Mercer Reference Mercer2006:XX, 105–108). This stands as an example of how a technology may persist in a limited use by a few, until new innovations unlock its potential in fulfilling a wider demand to the benefit of society at large.
A. Mark Pollard
University of Oxford
A. Mark Pollard is Emeritus Professor of Archaeological Science at the University of Oxford. His publications include Beyond Provenance: New Approaches to Interpreting the Chemistry of Archaeological Copper Alloys (University of Leuven Press, 2018), Archaeological Chemistry (Royal Society of Chemistry, 2017), and Handbook of Archaeological Sciences (Wiley, 2001).
Chris Gosden
University of Oxford
Chris Gosden is Professor of European Archaeology at the University of Oxford, and his publications include Celtic Art in Europe: Making Connections (2014), A Technology of Enchantment? Exploring Celtic Art 400 BC – AD 100 (2012) and Archaeology and Colonialism (2004).
Editorial Board
Ann Brysbaert, Leiden University
Shadreck Chirikure, University of Oxford
Rosemary Joyce, University of California, Berkeley
Mei Jianjun, University of Cambridge
Joanna Sofaer, University of Southampton
About the Series
Examining technology on a worldwide basis from the earliest human use of tools to the early modern period, this series focuses on new archaeological findings, and integrates these with historical textual sources where they exist. It considers how things were done, why they were done that way, and how an understanding of the world was generated through making and using materials.