I. INTRODUCTION
Bosentan (4-tert-butyl-N-[6-(2-hydroxyethoxy)-5-(2-methoxyphenoxy)-2,2’-bipyrimidin-4-yl]benzenesulfonamide, C27H29N5O6S; Figure 1) is a nonpeptide pyrimidine derivative approved by the FDA for the treatment of pulmonary arterial hypertension (PAH), when high blood pressure develops in the pulmonary arteries that carry oxygen-poor blood from the heart to the lungs (Enevoldsen et al., Reference Enevoldsen, Sahana, Wehland, Grimm, Infanger and Krüger2020). Bosentan is sold under the brand names Tracleer®, Safebo®, and others. It was the first endothelin-I receptor antagonist approved for the treatment of PAH, in 2001, with World Health Organization (WHO) functional classes III–IV (Mathier and Ishizawar, Reference Mathier and Ishizawar2010; Enevoldsen et al., Reference Enevoldsen, Sahana, Wehland, Grimm, Infanger and Krüger2020). The WHO divides pulmonary hypertension into five groups, I–V, based on its cause. Bosentan decreases both pulmonary vascular resistance and systemic vascular resistance, increasing cardiac output without increasing the heart rate. Therefore, it is an important therapeutic option.
Molecular diagram of bosentan.
A search in the Cambridge Structural Database (CSD), version 2024.1.0 (Groom et al., Reference Groom, Bruno, Lightfoot and Ward2016), led to five reports with crystal structures related to bosentan. Three of them were for the bosentan monohydrate phase, BSN·H2O (Refcodes NEQHEY, Kaur et al., Reference Kaur, Jasinski, Keeley, Yathirajan, Betz, Gerber and Butcher2013; NEQHEY01, Trzcińska et al., Reference Trzcińska, Łaszcz and Sidoryk2016; NEQHEY02, Łaszcz et al., Reference Łaszcz, Witkowska, Trzcińska, Kubiszewski and Kuziak2012). The structure determinations reported in those entries were carried out at 123 and 100 K. The two additional reports found were a cocrystal phase with succinic acid (Refcode DEGPEO, Barbas et al., Reference Barbas, Portell, Hunter, Prohens and Frontera2022) and a zinc complex (Refcode NAMGIU, Khandar et al., Reference Khandar, Mirzaei-Kalar, White, Hosseini-Yazdi, Kebriaeezadeh and Jouyban2017). Barbas and coworkers also prepared two new cocrystal phases with resorcinol and 4-hydroxybenzoic acid, respectively, reporting the X-ray powder diffraction patterns, the space group, and the unit cell parameters. The report found in the ICDD PDF-5+ Database (Kabekkodu et al., Reference Kabekkodu, Dosen and Blanton2024) contains the powder diffraction pattern of bosentan monohydrate (PDF 05-006-0054) calculated using the crystal structure data at 123 K reported for NEQHEY (Kaur et al., Reference Kaur, Jasinski, Keeley, Yathirajan, Betz, Gerber and Butcher2013).
After searching for “Bosentan monohydrate” on the Google Patents site (https://patents.google.com), many entries are encountered. Some of them refer to the preparation of BSN·H2O and various forms of sodium and potassium derivatives. Only in one of the patents (Sidoryk et al., Reference Sidoryk, Łuniewski, Puchalska, Łaszcz, Maruszak and Kościuch2014), the unit cell parameters of a BSN·H2O phase are reported. This phase corresponds to the phase reported in the CSD under the Refcodes NEQHEY01 (Trzcińska et al., Reference Trzcińska, Łaszcz and Sidoryk2016) and NEQHEY02 (Łaszcz et al., Reference Łaszcz, Witkowska, Trzcińska, Kubiszewski and Kuziak2012).
Several studies associated with the characterization of bosentan monohydrate have been published in the open literature. Some of them involve the preparation and characterization of microparticles (Lee et al., Reference Lee, Kang, Lee, Kim, Rhee, Kim, Park and Park2016), inclusion complexes (Jadhav and Pore, Reference Jadhav and Pore2017), and nanoparticles (Giménez et al., Reference Giménez, Sperandeo, Faudone, Noriega, Manucha and Kassuha2019; Mohapatra et al., Reference Mohapatra, Dash, Bhise, Kandalkar, Vasu, Pahuja and Bhajipale2022). There are also reports of one intermediate (Sagar et al., Reference Sagar, Yathirajan, Jasinski and Glidewell2016) and various derivatives of bosentan (Easwaramoorthi et al., Reference Easwaramoorthi, Rajendran, Rao, Arun, Balachandran, Perumal, Nobuhiko, Mahalingam, Duraipandiyan and Al-Dhabi2015). The amorphization of crystalline bosentan monohydrate has been studied by Minecka et al. (Reference Minecka, Chmiel, Jurkiewicz, Hachuła, Łunio, Żakowiecki, Hyla, Milanowski, Koperwas, Kamiński, Paluch and Kamińska2022). They prepared an amorphous phase by vitrification and cryomilling. The resulting phase was characterized by X-ray powder diffraction, thermogravimetry, differential scanning calorimetry, Fourier-transformed infrared spectroscopy, and broad-band dielectric spectroscopy (Minecka et al., Reference Minecka, Chmiel, Jurkiewicz, Hachuła, Łunio, Żakowiecki, Hyla, Milanowski, Koperwas, Kamiński, Paluch and Kamińska2022). The processes of dehydration and amorphization of this drug were also followed by diffraction, thermal, and spectroscopic techniques and by saturation solubility and dissolution studies (Krupa et al., Reference Krupa, Danède, Węgrzyn, Majda and Willart2022). The unindexed powder diffraction plot reported by these authors cannot be used to properly identify the monohydrate phase of bosentan in a phase ID process because the powder diffraction data are not included in this publication.
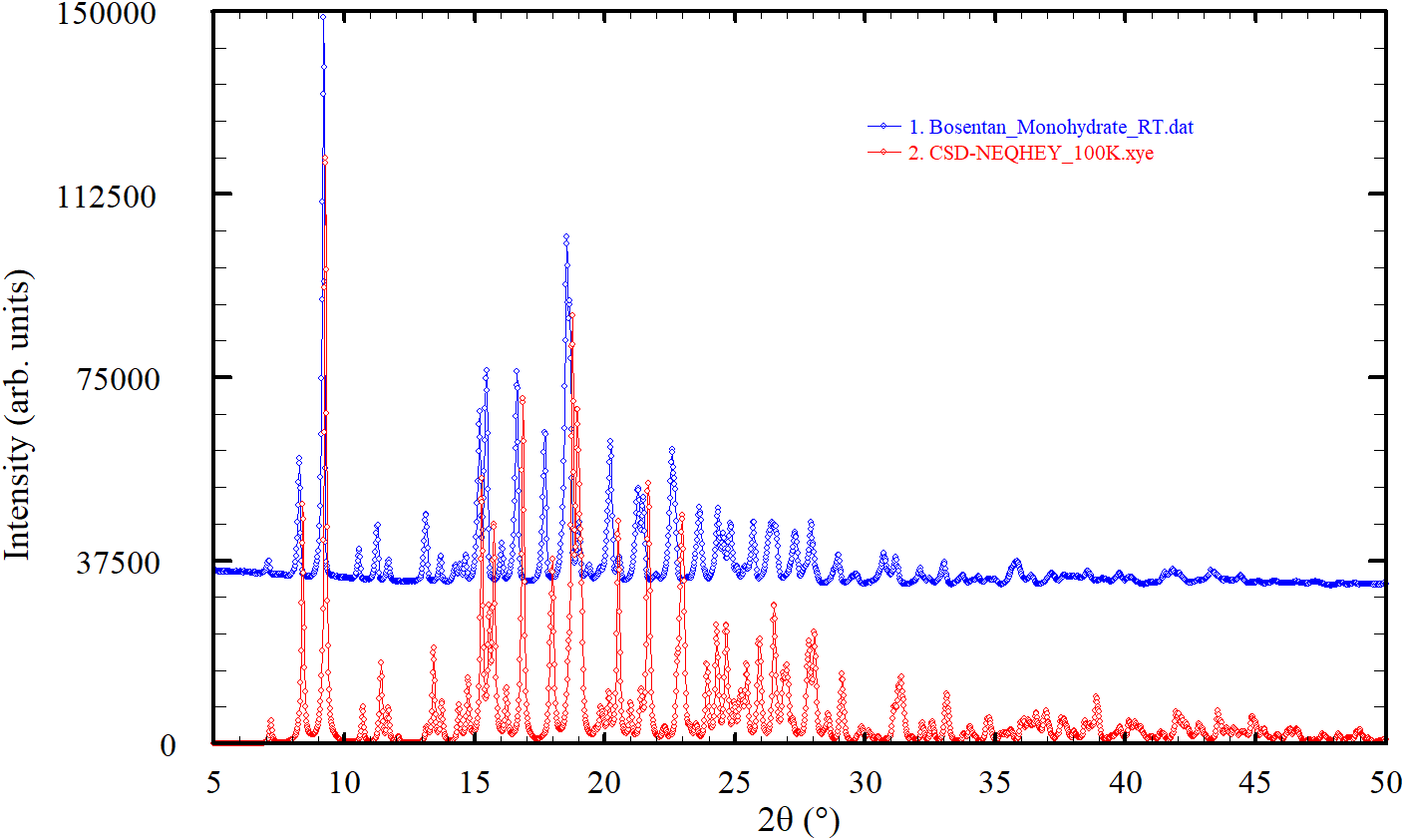
In the last few years, several compounds of pharmaceutical interest have been studied in our laboratory (Dugarte-Dugarte et al., Reference Dugarte-Dugarte, Toro, van de Streek, Henao, Fitch, Dejoie, Delgado and Díaz de Delgado2023; Toro et al., Reference Toro, Dugarte-Dugarte, van de Streek, Henao, Delgado and de Delgado2022; Dugarte-Dugarte et al., Reference Dugarte-Dugarte, Toro, van de Streek, Henao, de Delgado and Delgado2022; Dávila-Miliani et al., Reference Dávila-Miliani, Dugarte-Dugarte, Toro, Contreras, Camargo, Henao, Delgado and de Delgado2020). Because no powder diffraction pattern for bosentan monohydrate at room temperature has been included in the PDF-5+ database and the crystal structure of this material was determined at 100 and 123 K, for identification purposes at ambient conditions, it was decided to record and analyze the powder diffraction pattern at ambient temperature for inclusion in ICDD’s Powder Diffraction File™ (PDF®). Figure 2 illustrates the need to provide an experimental pattern at room temperature, useful for phase ID. As can be seen, the differences between the pattern recorded at room temperature and the pattern calculated from the single crystal data registered at 123 K make it difficult to properly identify bosentan monohydrate from powder data collected under normal laboratory conditions.
Comparison of the powder pattern recorded in the present study (room temperature) with the pattern calculated from single crystal data at 123 K (CSD Refcode NEQHEY, Kaur et al., Reference Kaur, Jasinski, Keeley, Yathirajan, Betz, Gerber and Butcher2013).
Given the importance that hydrogen bonding and other intermolecular interactions have on the use and performance of pharmaceutical compounds, an analysis of hydrogen bonds, C▬H⋯π, and π⋯π interactions, combined with Hirshfeld surface analysis, energy frameworks calculations, and hydrate analysis, is also presented.
II. EXPERIMENTAL METHODS
A small portion of the sample, provided by Tecnoquímicas (Cali, Colombia), was ground and mounted on a zero-background holder. The X-ray powder diffraction data were registered at room temperature with a BRUKER D8 ADVANCE diffractometer working in the Bragg-Brentano geometry. This instrument is equipped with a Cu Kα source, working at 40 kV and 30 mA, and a LynxEye detector. The pattern was recorded from 5.00 to 60.00° in steps of 0.01526° (2θ) at 1 s per step. The standard instrument settings (Ni filter of 0.02 mm, primary and secondary Soller slits of 2.5°, divergence slit of 0.2 mm, scatter screen height of 3 mm) were used.
III. CALCULATIONS AND COMPUTATIONAL STUDIES
The analysis of hydrogen bonds, C▬H⋯π, and π⋯π interactions was carried out with PLATON (Spek, Reference Spek2020) and Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020). Hirshfeld surface analysis, “fingerprint plots”, and energy frameworks calculations (Spackman and Jayatilaka, Reference Spackman and Jayatilaka2009) were performed with CrystalExplorer21 (Spackman et al., Reference Spackman, Turner, McKinnon, Wolff, Grimwood, Jayatilaka and Spackman2021). The energy frameworks were calculated using the molecular wavefunction at the B3LYP/6-31G(d,p) level, within a radius of 3.8 Å, and neighboring molecules within this shell were generated by symmetry operations. The space-filling environment and the interaction preferences of the water molecule in the structure were studied with the hydrate analyzer tool implemented in Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020).
IV. RESULTS AND DISCUSSION
A. Indexing and pattern fitting
The indexing of the recorded pattern carried out with DICVOL14 (Louër and Boultif, Reference Louër and Boultif2014), as implemented in the PreDICT graphical user interface (Blanton et al., Reference Blanton, Papoular and Louër2019), using the first 20 peaks of the pattern produced a monoclinic unit cell with unit cell parameters: a = 12.418(5) Å, b = 15.067(5) Å, c = 15.058(6) Å, β = 95.08(4)°, V = 2806.35 Å3. This cell accounted for the 77 diffraction maxima registered. The de Wolff (de Wolff, Reference de Wolff1968) and Smith–Snyder (Smith and Snyder, Reference Smith and Snyder1979) figures of merit obtained were M
20 = 27.7 and F
20 = 95.9 (0.0052, 40), respectively. It must be noted that the unit cell with the highest figures of merit, obtained with CONOGRAPH (Esmaeili et al., Reference Esmaeili, Kamiyama and Oishi-Tomiyasu2017) using the first 30 peaks, was similar to the cell obtained with DICVOL14. The figures of merit obtained with CONOGRAPH were M
30 = 21.162,
![]() $ {M}_{30}^{\mathrm{Wu}} $
= 20.279,
$ {M}_{30}^{\mathrm{Wu}} $
= 20.279,
![]() $ {M}_{30}^{\mathrm{Rev}} $
= 3.328,
$ {M}_{30}^{\mathrm{Rev}} $
= 3.328,
![]() $ {M}_{30}^{\mathrm{Sym}} $
= 70.424, NN = 78, and number of solutions = 4 (Oishi-Tomiyasu, Reference Oishi-Tomiyasu2013). The unit cell parameters obtained agree with the values reported by Kaur et al. (Reference Kaur, Jasinski, Keeley, Yathirajan, Betz, Gerber and Butcher2013) and by Sidoryk et al. (Reference Sidoryk, Łuniewski, Puchalska, Łaszcz, Maruszak and Kościuch2014), indicating that the material under study corresponds to the reported phase of bosentan monohydrate.
$ {M}_{30}^{\mathrm{Sym}} $
= 70.424, NN = 78, and number of solutions = 4 (Oishi-Tomiyasu, Reference Oishi-Tomiyasu2013). The unit cell parameters obtained agree with the values reported by Kaur et al. (Reference Kaur, Jasinski, Keeley, Yathirajan, Betz, Gerber and Butcher2013) and by Sidoryk et al. (Reference Sidoryk, Łuniewski, Puchalska, Łaszcz, Maruszak and Kościuch2014), indicating that the material under study corresponds to the reported phase of bosentan monohydrate.
The powder pattern recorded has been submitted to the ICDD through the GiA program to be incorporated into the Powder Diffraction File™ (PDF®). For this submission, integrated intensities were obtained by Le Bail refinement (Le Bail et al., Reference Le Bail, Duroy and Fourquet1988) using the FULLPROF software (Rodriguez-Carvajal, Reference Rodriguez-Carvajal1990). Weak reflections with intensities less than 0.5% of the maximum intensity were omitted. The refinement led to the following unit cell parameters: a = 12.4520(7) Å, b = 15.110(1) Å, c = 15.0849(9) Å, β = 95.119(5)°, V = 2827.0(3) Å3.
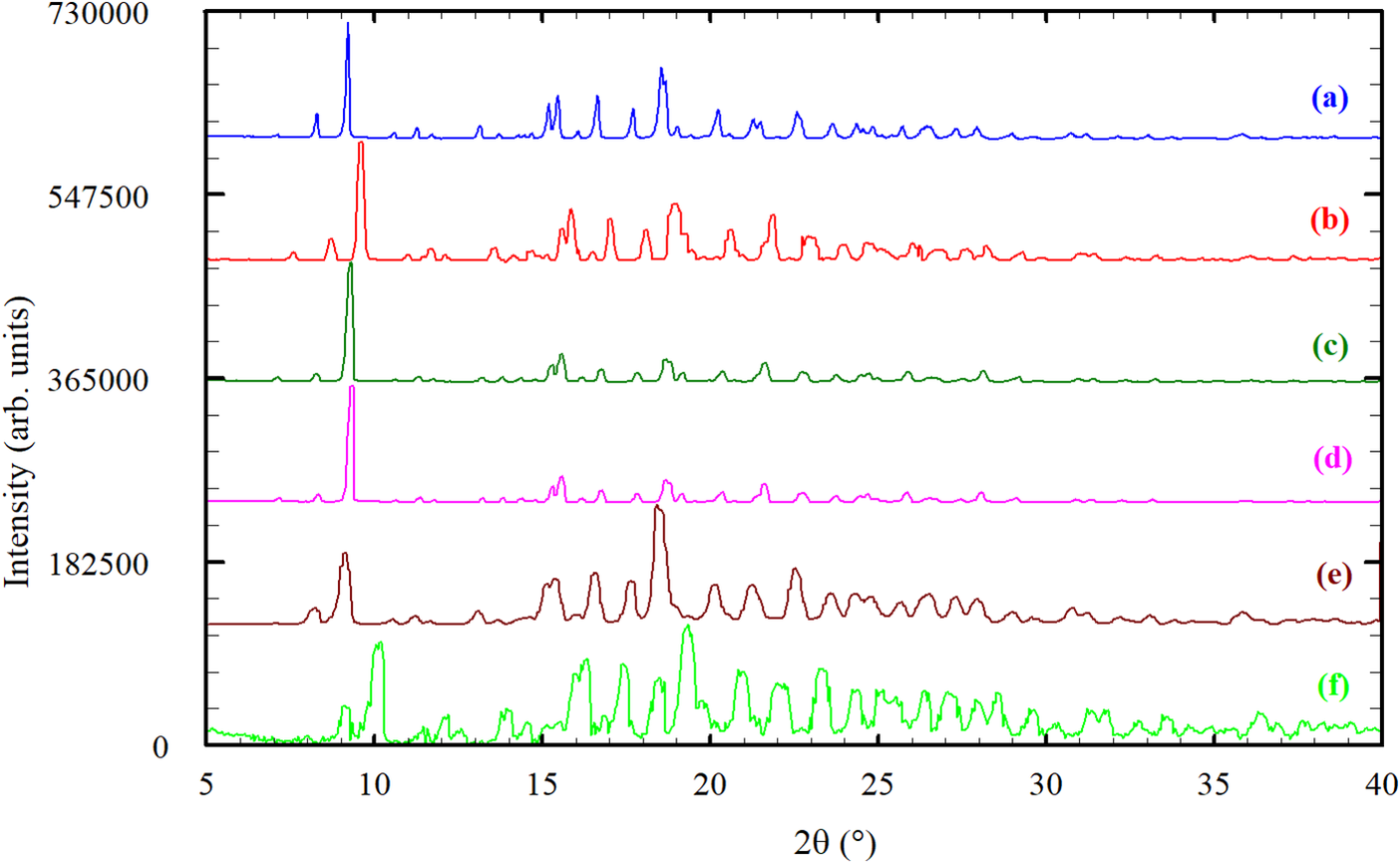
The powder patterns reported in the patents (Mathad et al., Reference Mathad, Niphade, Jagtap and Gaikwad2011; Andretto et al., Reference Andretto, Colli, Cotarca, Maragni, Melloni, Melotto, Michieletto, Verzini and Volpicelli2011; Cotarca et al., Reference Cotarca, Verzini, Melotto, Michieletto, Melloni, Maragni, Volpicelli, Andretto and Colli2011; Sidoryk et al., Reference Sidoryk, Łuniewski, Puchalska, Łaszcz, Maruszak and Kościuch2014; Teksi̇n and Yilmaz Usta, Reference Teksi̇n and Usta2020), digitized using the online JADE® Pattern Digitizer (ICDD, 2022), and the pattern recorded in the present work are shown in Figure 3. All the patterns are similar, indicating that they correspond to the same monohydrated phase.
Comparison of the powder pattern recorded in the present study (a) for Bosentan monohydrate (BSN·H2O) with the reported powder patterns from the patents: (b) Mathad et al., Reference Mathad, Niphade, Jagtap and Gaikwad2011; (c) Andretto et al., Reference Andretto, Colli, Cotarca, Maragni, Melloni, Melotto, Michieletto, Verzini and Volpicelli2011; (d) Cotarca et al., Reference Cotarca, Verzini, Melotto, Michieletto, Melloni, Maragni, Volpicelli, Andretto and Colli2011; (e) Sidoryk et al., Reference Sidoryk, Łuniewski, Puchalska, Łaszcz, Maruszak and Kościuch2014; (f) Teksi̇n and Yilmaz Usta, Reference Teksi̇n and Usta2020.
A Pawley fit (Pawley, Reference Pawley1981) of the recorded pattern was carried out by modeling the background, sample displacement errors, absorption, cell parameters, and peak shape parameters (including anisotropic broadening) using TOPAS-Academic (Coelho, Reference Coelho2018). The background was modeled by a 15-term Chebyshev polynomial. The intermediate Gaussian–Lorentzian function was used with a correction for axial divergence as proposed by the program. The Pawley refinement produced a good fitting of all the diffraction maxima recorded with residuals R p = 0.0173, R wp = 0.0237, and GoF = 1.932, once again confirming that the unit cell was correct and the single-phase nature of the material under investigation. The analysis of the reflection conditions implemented in the CONOGRAPH software suggested P21/c as the possible space group. This space group was also suggested by the Bayesian extinction symbol algorithm in DASH 4.0.0 (Markvardsen et al., Reference Markvardsen, David, Johnson and Shankland2001) and by DAJUST (Vallcorba et al., Reference Vallcorba, Rius, Frontera, Peral and Miravitlles2012).
An initial molecular model from the CIF file containing the structural data obtained at 100 K, reported in CSD entry NEQHEY01 (Trzcińska et al., Reference Trzcińska, Łaszcz and Sidoryk2016) was used for Rietveld refinement carried out with TOPAS-Academic (Coelho, Reference Coelho2018). The refinement included an overall scale parameter, background, sample displacement correction, peak shapes (including anisotropic broadening), unit cell parameters, absorption correction, atomic coordinates, four B iso parameters, and a March–Dollase parameter. The bond distances and angles were restrained based on the values suggested by Mogul Check Geometry (Bruno et al., Reference Bruno, Cole, Kessler, Luo, Motherwell, Purkis, Smith, Taylor, Cooper, Harris and Orpen2004). Four planar restraints for the molecule with a standard deviation of 0.01 Å were also established. The isotropic atomic displacement parameters for the hydrogen atoms were 1.2 times the parameter of the C, N, or O atom to which they are attached.
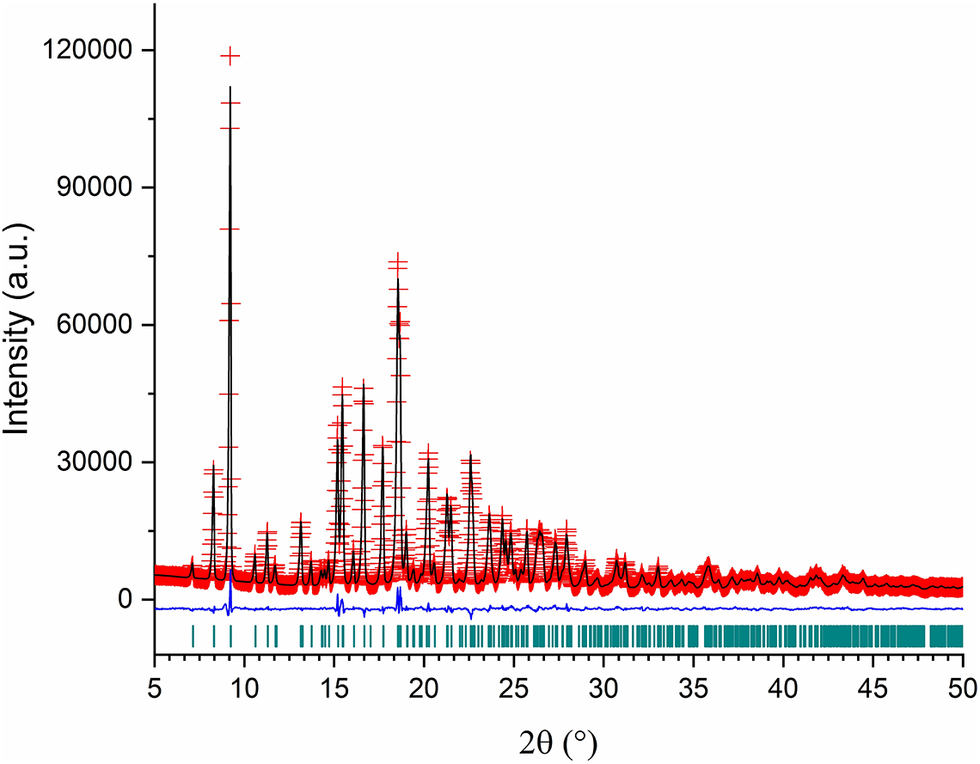
The refinement performed with TOPAS-Academic (Coelho, Reference Coelho2018) was very stable. Figure 4 shows the final Rietveld refinement plot. The refinement included 277 parameters, 2012 data points (502 reflections), 197 restraints, and 4 constraints. The final whole pattern fitting converged with good figures of merit: R e = 0.0150, R p = 0.0287, R wp = 0.0355, and GoF = 2.371. The March–Dollase preferred orientation parameter in the (1 1 0) plane was 0.923(4).
Rietveld refinement plot for Bosentan·H2O.
B. A closer look at the molecular structure, hydrogen bonding, C▬H⋯π, and π⋯π interactions
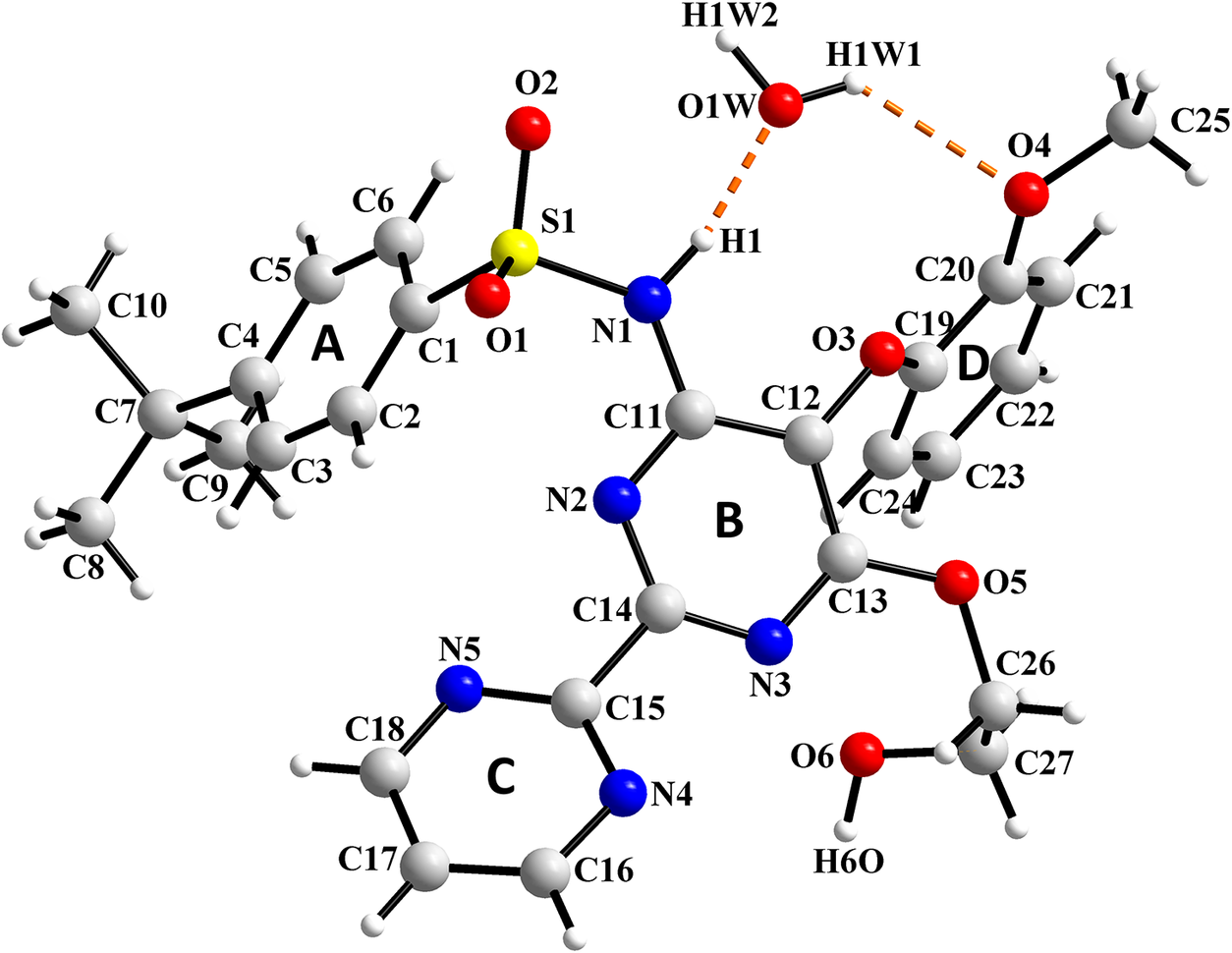
The molecular structure of BSN·H2O with the labeling scheme for atoms and rings is presented in Figure 5 drawn with Diamond 5.0 (Putz and Brandenburg, Reference Putz and Brandenburg2023). Bond distances and angles and torsion angles are collected in Supplementary Tables S1–S3. Their values are in agreement with the distances and angles previously reported. The comparison of the refined structure with the CSD entries NEQHEY and NEQHEY01, using the Packing Similarity feature implemented in Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020) led to RMSCD values of 0.0790 and 0.090 Å, respectively (0.0728 and 0.0806, respectively, if using the Molecular Similarity feature). A superposition of the molecules from this study with those reported in the CSD is presented in Figure 6.
Molecular structure of Bosentan·H2O with the labeling scheme for atoms and rings.
Superposition of the molecule of Bosentan obtained in this work (cyan) with the molecule from CSD entries NEQHEY (red) and NEQHEY01 (blue).
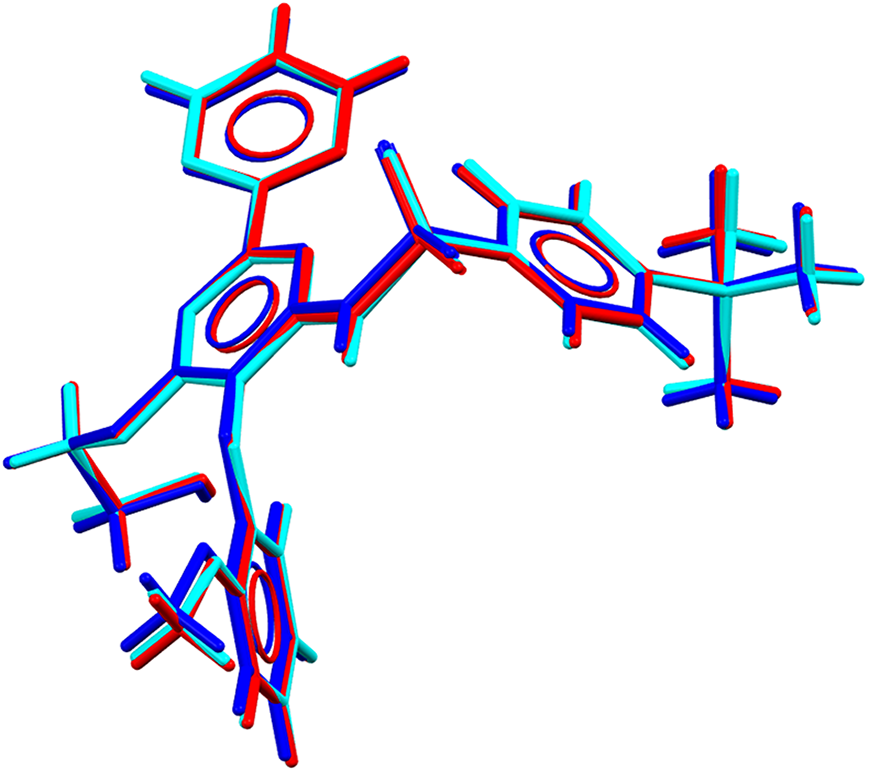
The molecule of bosentan adopts a bent conformation. Important torsion angles (Supplementary Table S3) are N1–C11–C12–O3 [−4.1(9)°], C1–S1–N1–C11 [−74.2(6)°], and C12–O3–C19–C20 [−176.5(6)°]. The planes of the pyrimidine rings, represented by B and C in Figure 5, make an angle of 28.7(3)°. Ring A is twisted by 69.8(3)° and 42.9(3)° with respect to rings B and C, respectively. Ring D makes angles of 68.0(3)° and 83.4(3)° with rings B and C, respectively.
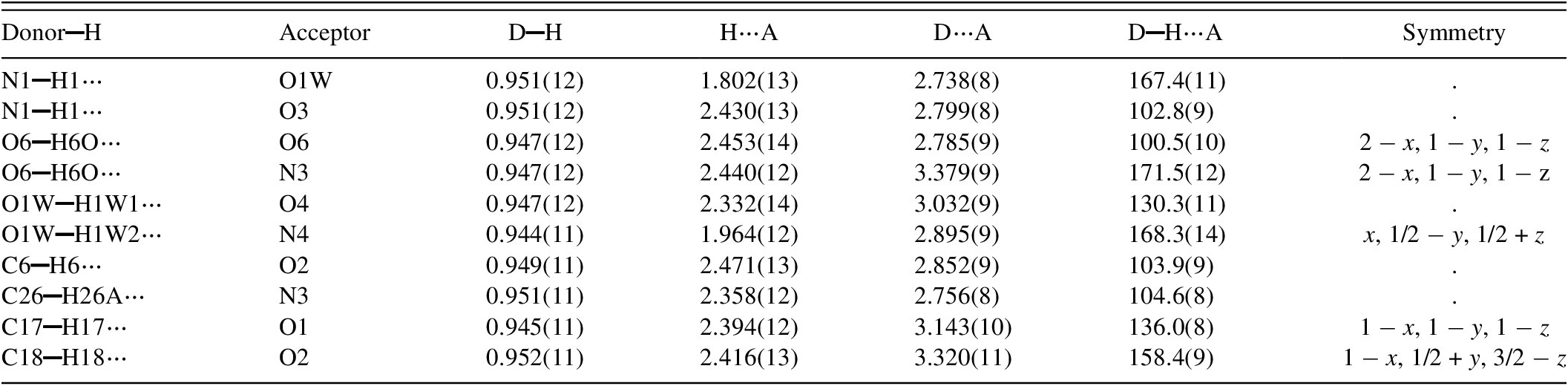
The bent conformation of bosentan provides the appropriate geometry for several intramolecular interactions. The analysis, carried out with PLATON (Spek, Reference Spek2020), indicates that there are five intramolecular N▬H⋯O, O▬H⋯O, C▬H⋯O, and C▬H⋯N hydrogen bonds involving the water molecule and the bosentan moiety. In addition, five intermolecular hydrogen bonds occur involving O▬H⋯O, O▬H⋯N, and C▬H⋯O interactions between molecules related by the c-glide perpendicular to [010]. Some of the hydrogen bonds reported herein are similar to those reported by Kaur et al. (Reference Kaur, Jasinski, Keeley, Yathirajan, Betz, Gerber and Butcher2013) for NEQHEY in their single crystal study at 123 K. Table I contains the geometrical parameters for the hydrogen bonds depicted in Figure 7a,b.
Hydrogen bond geometry (Å, °) for Bosentan·H2O.
(a) Partial view of the chains of hydrogen bonded molecules related by a c-glide along the c-axis; (b) chains connected via hydrogen bonding; color scheme: (i) x, y, z in light grey; (ii) −x, ½ + y, ½ − z in green; (iii) −x, −y, −z in gold; (iv) x, ½ − y, ½ + z in magenta; (c) sequence of molecules connected by one C▬H⋯π and the π⋯π interactions.
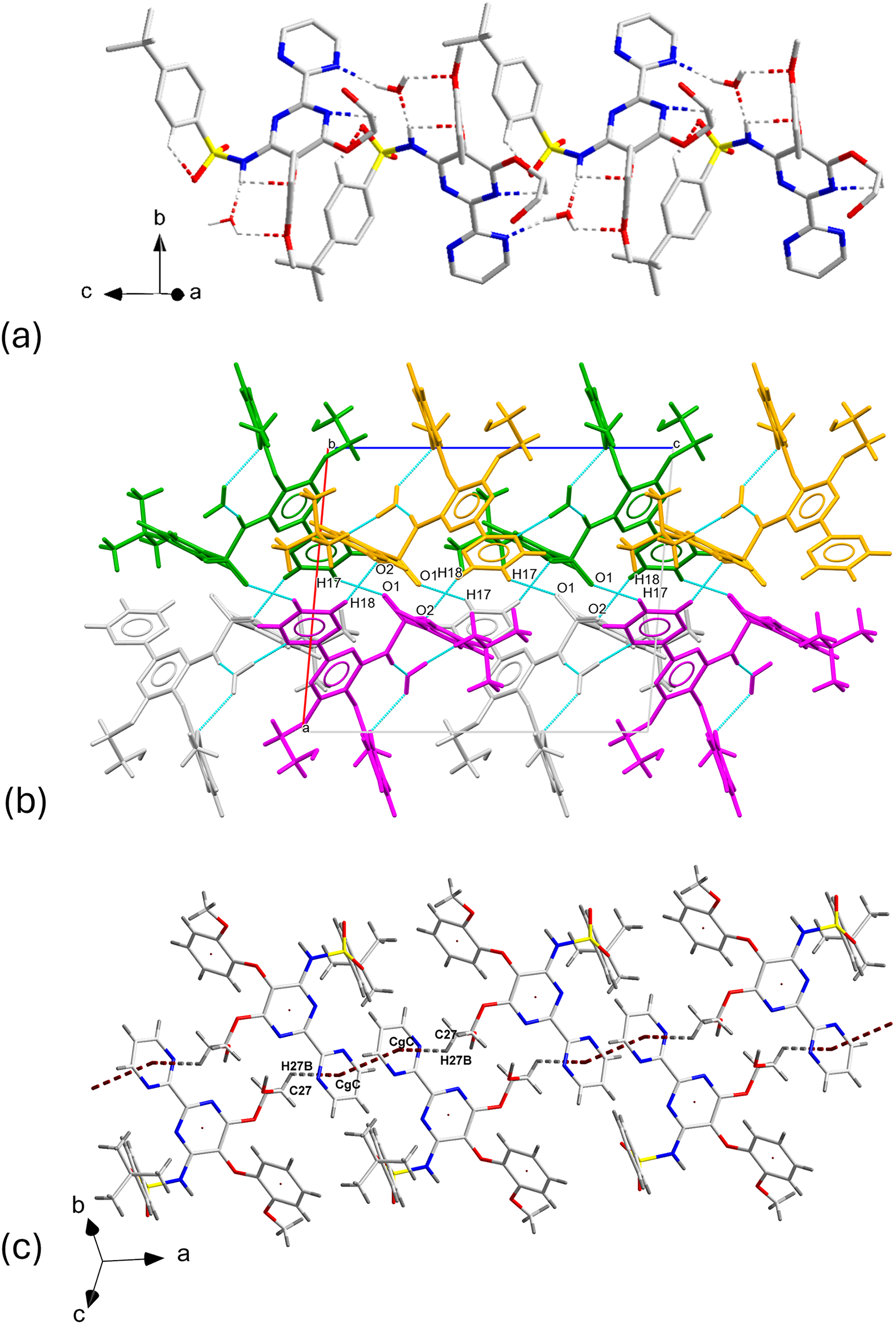
In addition to extensive hydrogen bonding, C▬H⋯π, and π⋯π interactions play a role in stabilizing the structure. The geometric parameters of these interactions are presented in Table II. The strongest π⋯π contact (shown in Figure 7c) occurs between the centroids of rings C of two bosentan molecules related by inversion. The centroid-to-centroid (CgC ▬CgC ) distance is 3.563(4) Å. At the same time, the C27–H27B end of two neighboring molecules participate in C▬H⋯π interactions with the two CgC in contact by π⋯π interactions, at distances of 2.995(10) Å.
Geometry of C▬H···π, and π···π interactions in Bosentan·H2O.
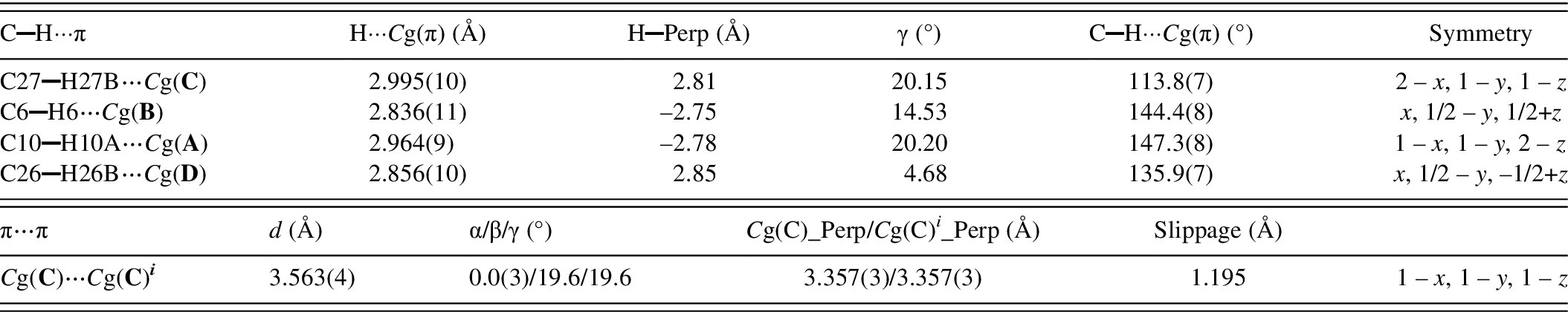
Cg(π) denotes the centroids Cg(A), Cg(B), Cg(C), and Cg(D) of rings A, B, C, and D (π-rings) respectively, as depicted in Figure 5. The geometry of the contacts is defined in PLATON [Spek, Reference Spek2020] by the following parameters: H—Perp = perpendicular distance of H to the plane of rings A, B, C, or D (Figure 5); γ = angle between the Cg(π)—H or the Cg(C)→Cg(C) i vector and the normal to plane C; d = Cg(C)··· Cg(C) i distance; α = dihedral angle between Planes C and C i ; β = angle between the Cg(C)→Cg(C) i vector and the normal to plane C; Cg(C) Perp = perpendicular distance of Cg(C) on ring C i ; Cg(C) i _Perp = Perpendicular distance of Cg(C) i on ring C; Slippage = distance between Cg(C) and the perpendicular projection of Cg(C) i on ring C. The symmetry operation 1-x, 1-y, 1-z is denoted by the superscript i .
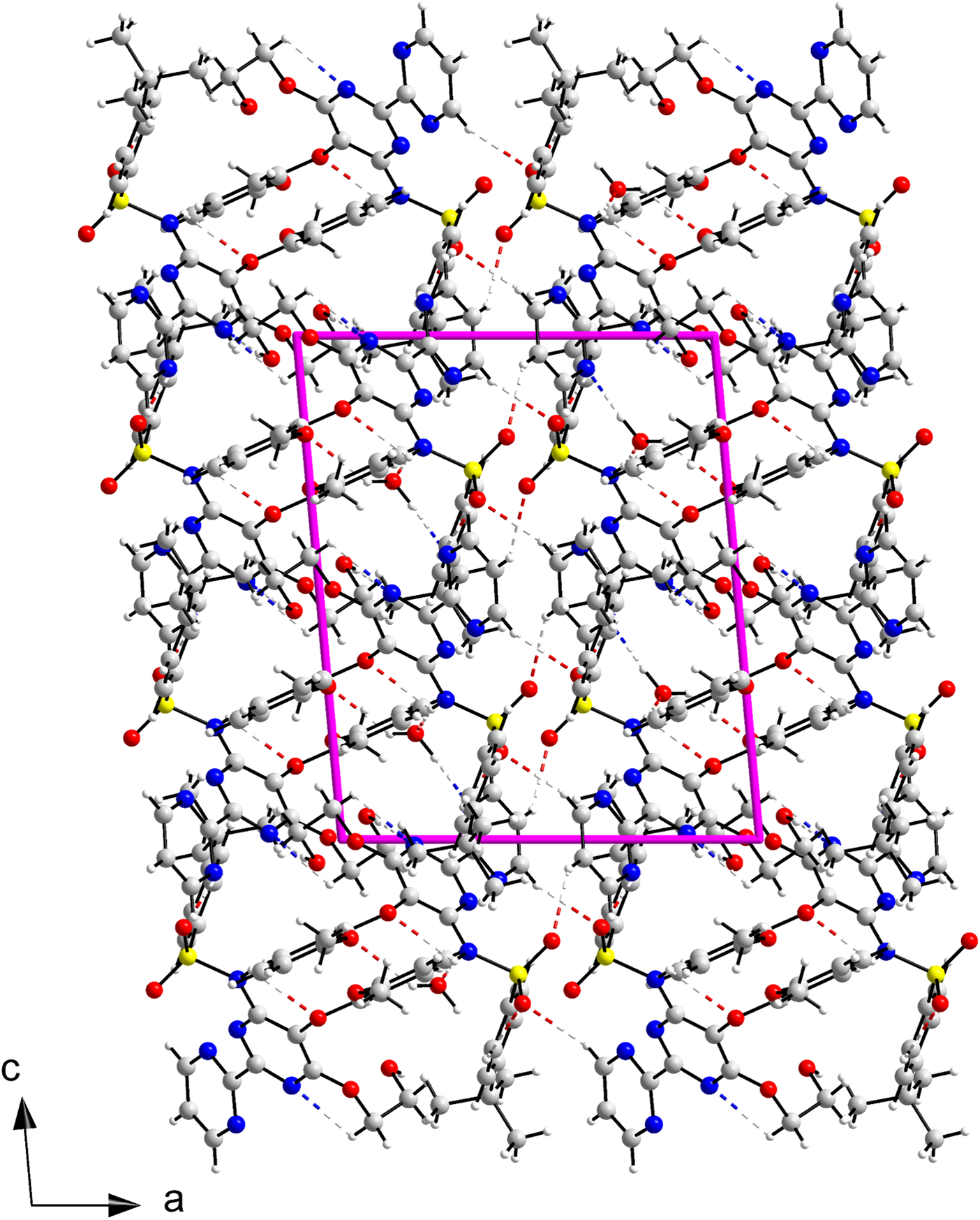
A view of the structure of BSN·H2O is shown in Figure 8. The structure can be described in terms of columns of bosentan and water molecules that run along the c-axis and pack along the a-axis joined by hydrogen bonds where only bosentan molecules participate.
Crystal structure of Bosentan·H2O viewed down the b-axis.
C. Hirshfeld surface analysis, energy frameworks calculations, and hydrate analysis
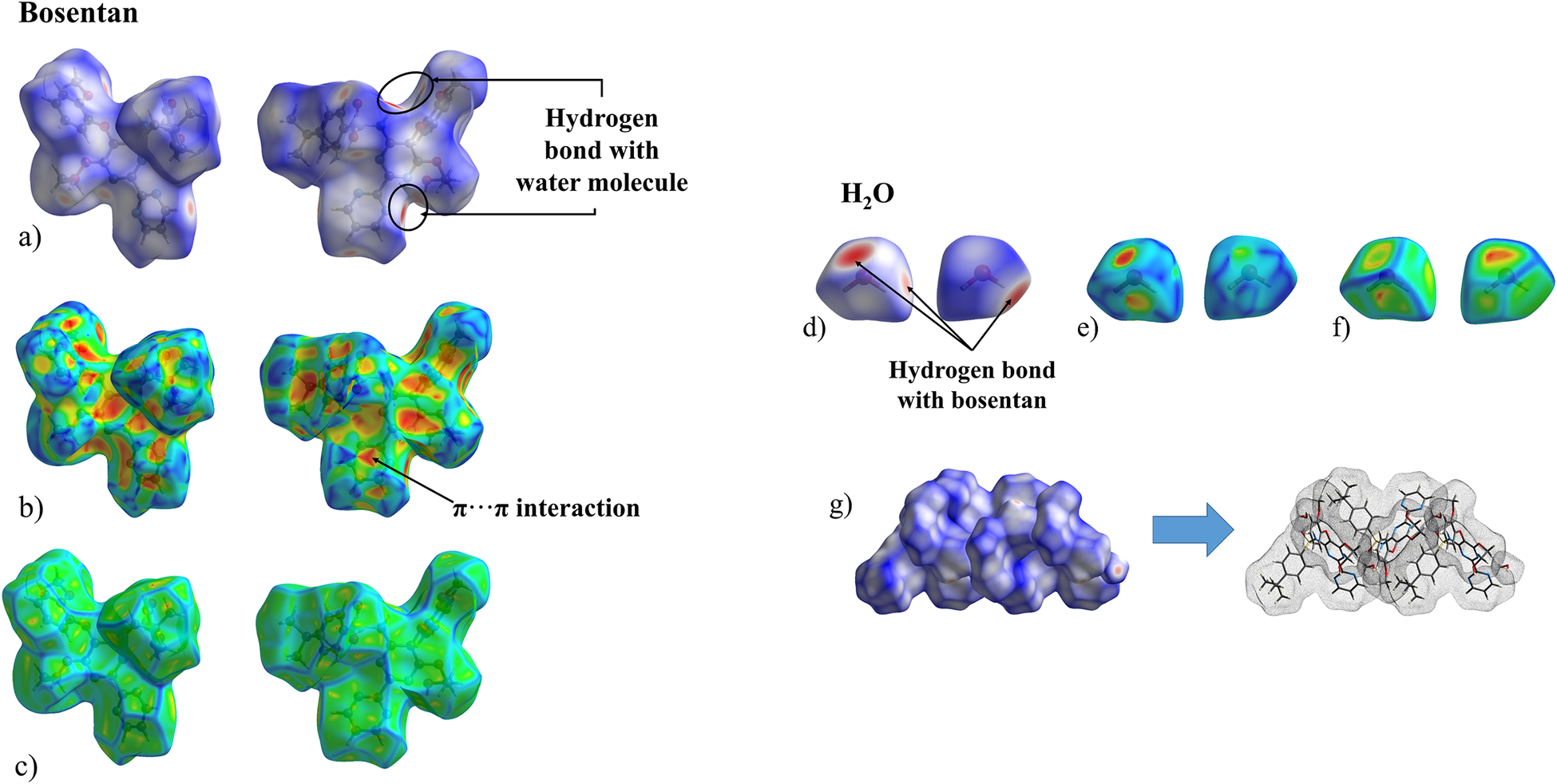
The Hirshfeld surface (HS), calculated with CrystalExplorer21.5 (Spackman et al., Reference Spackman, Turner, McKinnon, Wolff, Grimwood, Jayatilaka and Spackman2021), provides useful tools for the analysis and representation of intermolecular contacts. Figure 9 depicts the HS mapped onto several functions for the BSN and the water moieties: d norm (a,d), shape index (b,e), and curvedness (c,f). In Figure 9a,d, the red spots represent the intermolecular contacts shorter than the sum of van der Waals radii, while the longest contacts are represented in blue. The indentations where the red spots occur indicate the points of contact of the water molecule. The HS analysis reveals that the most important intermolecular interactions correspond, as expected, to the hydrogen bonds between the BSN and the water molecules. In the shape index function mapped on the HS (shown in Figure 9b,e) the blue bumps and the red-orange dents indicate where interactions occur. The bosentan and water molecules are arranged along the b-axis next to each other, in an alternating fashion, with the BSN shape index dents being complementary to the water shape index bumps (Figure 9b,e). The interactions between the C aromatic rings appear as triangular spots on the map of the shape index and correspond to the π⋯π interactions described in the previous section, summarized in Table II. The curvedness function for the BSN molecule (Figure 9c,f) displays flat areas, the most relevant of them located on the C ring where the π⋯π interactions take place. The curvedness function mapped for the water molecule (Figure 9f) is characterized by flat faces where the bosentan molecule is “anchored” through the hydrogen bonds. These contacts contribute to the crystal packing of the material in layers of alternating BSN and water molecules (Figure 9g).
Hirshfeld surface mapped onto d norm, shape index, and curvedness for the Bosentan molecule (a, b, c, respectively) and the H2O molecule (d, e, f, respectively) in Bosentan·H2O; g depicts the packing of Bosentan and water molecules.
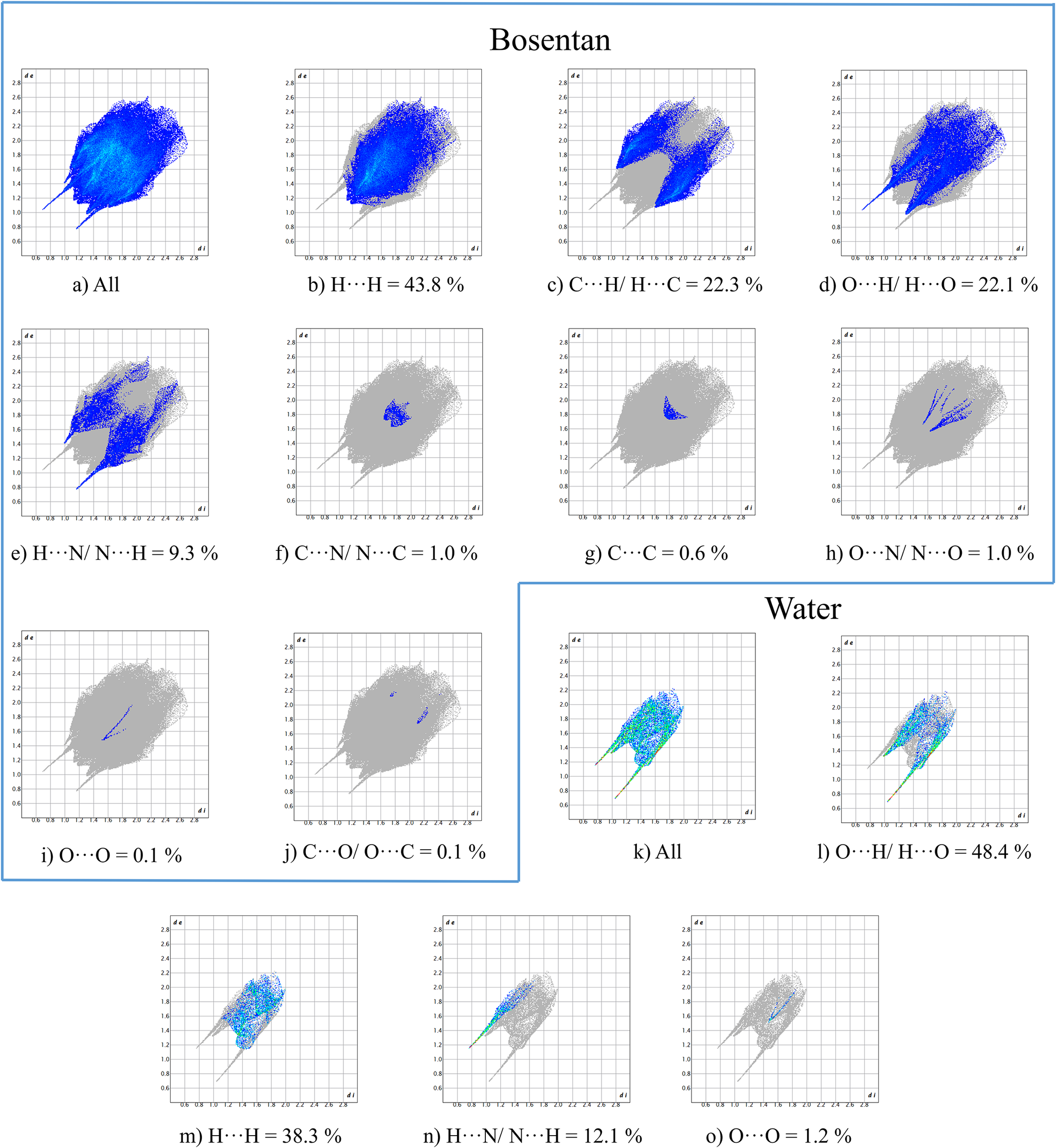
The “fingerprint plots” (Figure 10) representing distances of intermolecular interactions (Spackman and Jayatilaka, Reference Spackman and Jayatilaka2009) provide additional insight into the contributions of such interactions. Figure 10a (for bosentan) and Figure 10k (for water) represent all interactions in which these two moieties are involved. In the subsequent diagrams the contributions of each contact are plotted. They indicate that, in bosentan, the most important contacts are H⋯H, C⋯H/H⋯C, and O⋯H/H⋯O, contributing 43.8, 22.3, and 22.1%, respectively. H⋯N/N⋯H contacts represent a 9.3% while other interactions contribute less than 1%. For the water molecule, the O⋯H/H⋯O contact represents 48.4%, the H⋯H a 38.3%, and the H⋯N/N⋯H a 12.1%.
Fingerprint plots for the Bosentan molecule (a–j) and for the water molecule (k–o) with percent contributions from specific pairs of interatomic interactions.
The structure of bosentan monohydrate is densely packed. The volume of the HS for the bosentan molecule is 664.70 Å3 and 24.27 Å3 for the water molecule, corresponding to a total volume of 688.97 Å3, which represents 98.26% of the volume of the unit cell.
The analysis of the space occupied by the water molecules and their interactions with the closest neighbors was carried out using the hydrate analyzer tool incorporated in Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020), and the graphical representation is shown in Figure 11. The water molecules occupy 59.51 Å3, representing 2.1% of the volume of the unit cell. Each water molecule is hydrogen bonded to two bosentan molecules and each bosentan binds to two water molecules. They arrange in double layers parallel to the bc-plane and stack along the a-axis connected by hydrogen bonds between bosentan moieties (Figure 11a). The interaction maps for the water probe are shown in Figure 11b. The greatest number of interactions are observed in the contours with the most intense color levels. The most important interactions of the water molecule are with the O3 and O4 oxygen atoms from ring D (magenta outline), with the hydroxyl group (orange circle) and with N3 from ring B (green ellipse). As can be seen, in the packing, the water molecules interact with almost the entire bosentan molecule.
(a) View down the b-axis of the volume occupied by the water molecules calculated using the hydrate analyzer tool in Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020) and (b) interaction map for the water molecules in Bosentan·H2O.

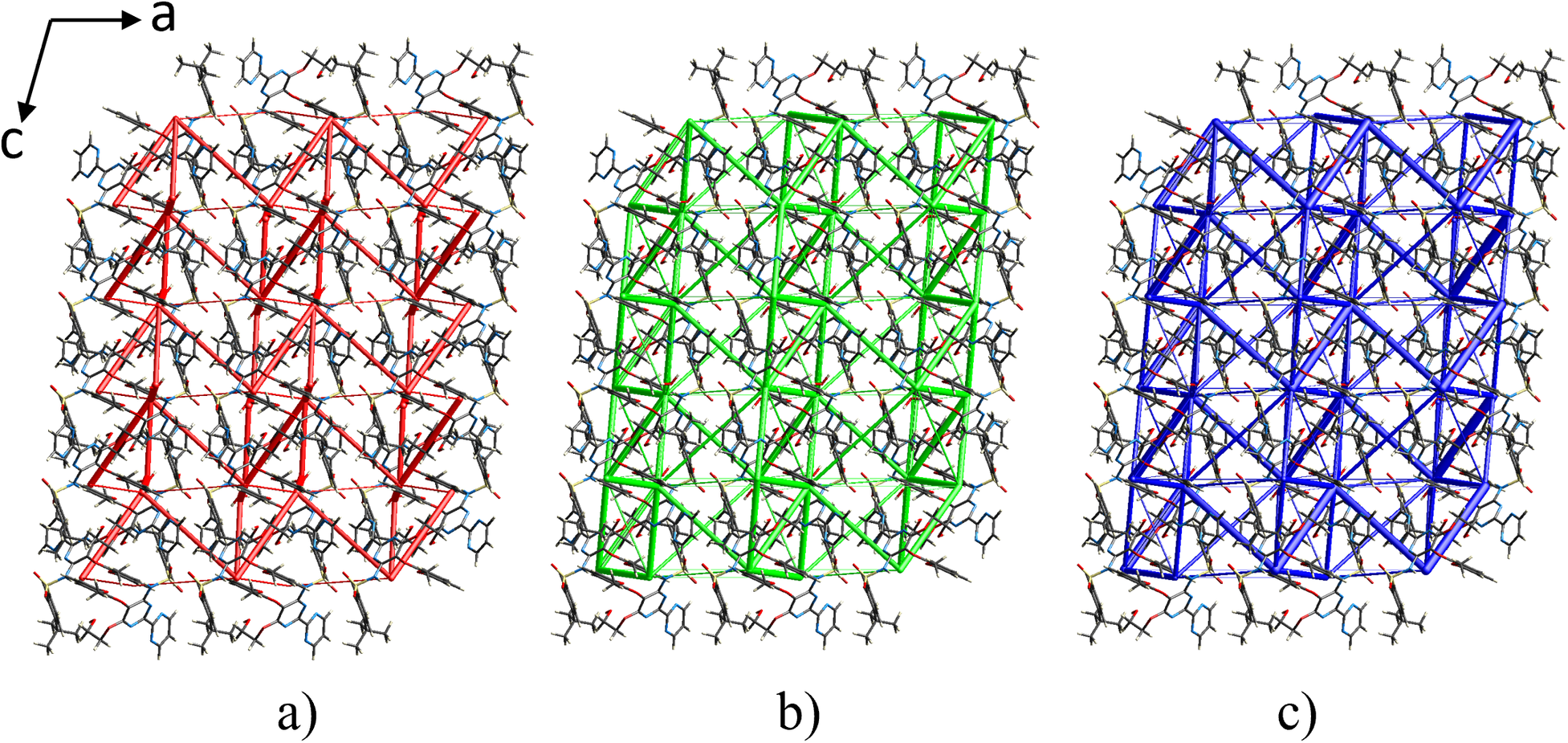
The interaction energies between the central molecule and neighboring molecules within a radius of 3.8 Å are shown in Supplementary Table S4. The highest stabilization interaction energies are observed between BSN molecules related by inversion (E tot = −84.1 kJ/mol and −64.1 kJ/mol), followed by interactions of neighboring molecules with symmetry operation −x, y + 1/2, −z + 1/2 (E tot = −51.5 kJ/mol) and the units related by x, −y + 1/2, z + 1/2 (E tot = −39.7 kJ/mol). The most relevant BSN interactions with the water molecule have energy values of −45.0 kJ/mol and −32.3 kJ/mol, corresponding to the hydrogen bonds O1W⋯H1 and N4⋯H1W2. The topology of the energy frameworks (Figure 12) shows that the −84.1 kJ/mol interaction extends along the c-axis forming chains that are connected parallel to the a-axis by contacts with energies of −64.1 kJ/mol and −33.0 J/mol while the interactions between BSN and water extend in a zigzag fashion along the c-axis.
Energy frameworks for Bosentan·H2O calculated with CrystalExplorer (Spackman et al., Reference Spackman, Turner, McKinnon, Wolff, Grimwood, Jayatilaka and Spackman2021) viewed down the b-axis. (a) electrostatic, E ele, red; (b) dispersive, E disp, green; (c) total energy, E tot, blue. The cylinder radii were scaled to 75 arbitrary units and the energy cutoff is 5 kJ mol−1.
V. DEPOSITED DATA
Crystallographic information framework (CIF) files containing the results of the Rietveld refinement and a CIF with the data submitted for the GiA program were deposited with the ICDD (Editorial Number G000465). The data can be requested at pdj@icdd.com. The crystal structure data were also deposited with the Cambridge Crystallographic Data Centre (CCDC 2372331).
Supplementary material
To view supplementary material for this article, please visit http://doi.org/10.1017/S0885715625000065.
Acknowledgments
The authors thank the support of Vicerrectoría de Investigación y Extensión of Universidad Industrial de Santander (UIS), Colombia. Access to the Cambridge Structural Database for Universidad de Los Andes (Venezuela) was possible through the Frank H. Allen International Research & Education Programme (FAIRE) from the Cambridge Crystallographic Data Centre.
Conflicts of interest
The authors declare no competing interests.
















