New Zealand has a strategy of eliminating SARS-CoV-2 that has resulted in a low incidence of coronavirus-19 disease (COVID-19). The first case was reported on 26 February 2020, and the country entered a strict nationwide lockdown one month later for 49 days [Reference Baker, Wilson and Anglemyer1]. Through rigorous border control and managed isolation and quarantine facilities for new arrivals, New Zealand has since remained largely COVID-19 free. Globally, serological surveillance has been utilised throughout the pandemic to define the cumulative incidence, including estimations of missed cases and/or asymptomatic infection. Due to lockdowns and movement restrictions, blood donors have been used as a sentinel population in many settings [Reference Uyoga2, Reference Gidding3]. The aim of this study was to describe the spread of SARS-CoV-2 in New Zealand via a blood donor serosurvey. Though the pandemic response has been highly effective, PCR testing was initially restricted due to limited diagnostic reagents [Reference Geoghegan4] and there have been occasional border incursions and small community outbreaks, including a cluster in August 2020 with no identified link to the border.
Samples were collected by the New Zealand Blood Service via nine static collection centres and 36 mobile collection services over a 4-week period (3 December 2020–6 January 2021) from individuals aged 16–88 years. Duplicates were removed, leaving 9806 samples for analysis. Compared with the 2018 New Zealand census, participants were more likely to be aged 40–59 years (43.3% vs. 25.9%) and of European ethnicity (77.8% vs. 61.0%) but had a similar proportion of females (49.1% vs. 50.7%) and were geographically spread with 16 of 20 district health board regions represented (Table 1 and Supplementary Appendix). This study was assessed by the Health and Disability Ethics Committee, and additional consent was not required (21/CEN/21).
Demographics of the blood donors, 2018 Census population and COVID-19 cases in New Zealand
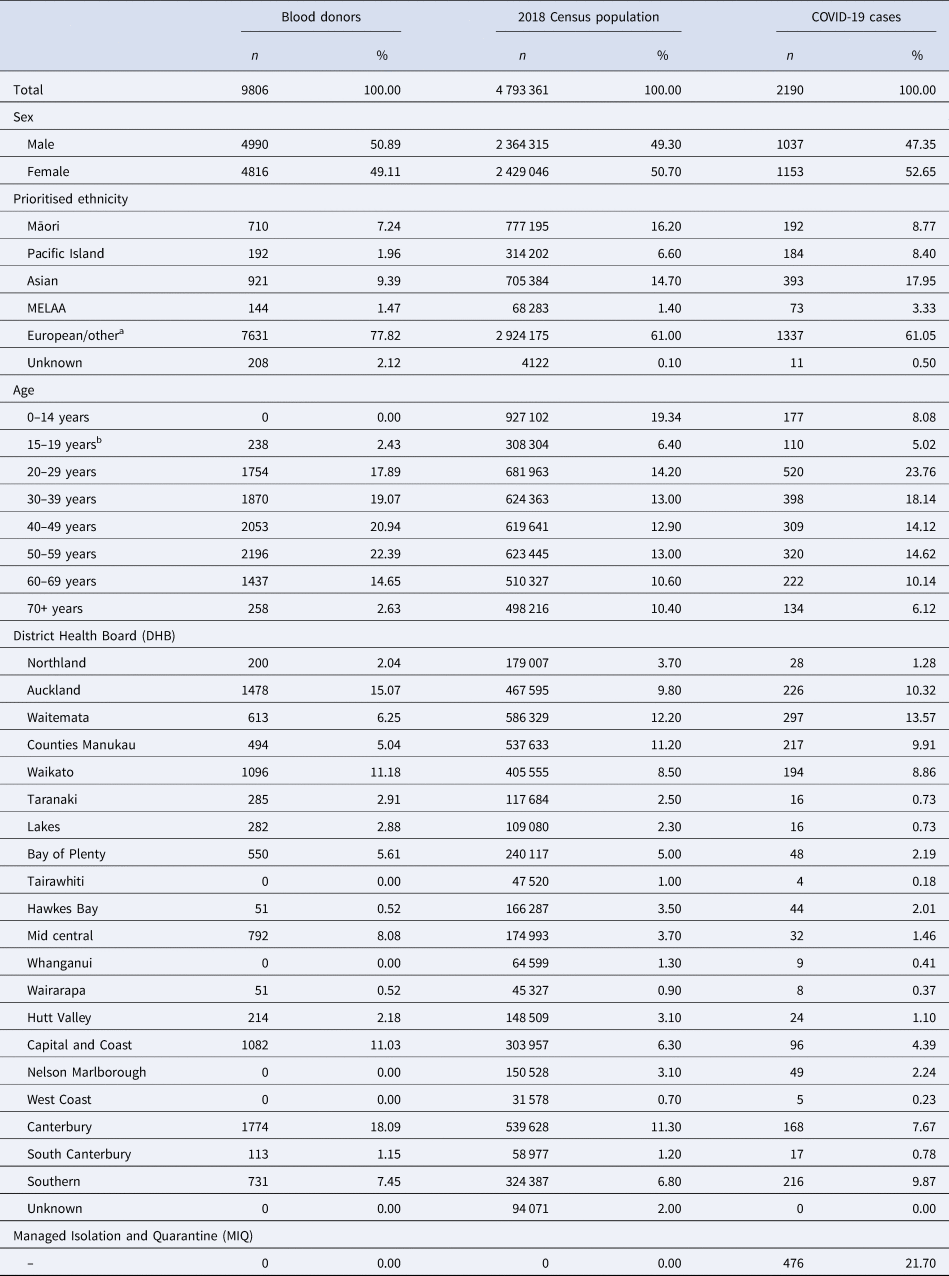
MELAA, Middle Eastern, Latin American and African.
The New Zealand blood service donations were collected between the 3rd of December 2020 and the 6th of January 2021. Of the 9806 individuals, 9771 are blood donors and 35 are living tissue and stem cell donors. All donors must be free of illness and weigh >50 kg. Notable travel and a history SARS-CoV-2 infection (or contact with a positive case) are recorded prior to collection. The health board region provided for the donors is based on donation location. Demographics for COVID-19 cases were obtained from the New Zealand Ministry of Health and include probable and confirmed infections up to and including the 6th of January 2021. The most recent New Zealand census took place in March 2018. Priority ethnicity is reported as defined by the New Zealand Department of Statistics.
a ‘Other’ comprises of n = 28 (0.20%) of the blood donor population, and n = 51 447 (1.10%) of the New Zealand census population.
b All blood donors are at least 16 years of age.
Antibodies to the Spike (S) protein and receptor-binding domain (RBD) persist for many months after infection, compared with antibodies to the nucleocapsid (N) protein [Reference Whitcombe5, Reference Carvalho, Krammer and Iwasaki6], providing a rationale for the use of S protein-based assays in serosurveys. The overall serological testing algorithm was optimised for specificity given the low number of reported COVID-19 cases in New Zealand (2190 as of 6 January 2021) and the associated period prevalence of 0.04%, which limits the positive predictive value of tests with reduced specificity [Reference Bond7]. Samples were first screened with a widely used and well-validated two-step ELISA that comprises a single point dilution assay against the RBD followed by titration against trimeric S protein (Supplementary Appendix) [Reference Amanat8, Reference McGregor9]. Samples above the cut-off were tested on two further immunoassays – the EuroImmun SARS-CoV-2 IgG ELISA (EuroImmun AG, Lübeck, Germany) and the cPass surrogate Viral Neutralisation Test (sVNT) (GenScript, New Jersey, USA) and the values deemed seropositive if above the cut-off on both commercial assays. Sensitivity and specificity for these assays were determined by Receiver Operator Characteristic (ROC) curves based on previous analyses (413 pre-pandemic negatives, 99 PCR confirmed cases) (Supplementary Appendix) [Reference McGregor9, Reference Craigie10].
Of the 9806 samples, 18 were positive for both Spike IgG (EuroImmun) and antibodies that block the RBD-hACE-2 interaction (sVNT), with the values highly correlated (Pearson r 0.7993, P < 0.0001) (Fig. 1). Further analysis of the 18 seropositive samples with a multiplex bead-based assay that detects antibody isotype reactivity to RBD, S and N proteins [Reference Whitcombe5] revealed a pattern consistent with infections that occurred weeks or months prior; a dominance of RBD and S protein IgG with few samples positive for N protein IgG, nor IgA or IgM against any of the three antigens (Fig. 1). Within these 18 seropositive samples, six were retrospectively matched to donors with previously confirmed SARS-CoV-2 infections. That all confirmed cases were detected supports the rationale of the testing algorithm applied. A further four seropositive samples were from donors with 2020 travel history in settings with a high risk of SARS-CoV-2 exposure (UK and Europe), suggesting likely infection outside New Zealand. The remaining eight seropositive samples were from seven different district health regions, giving a crude seroprevalence estimate of 0.082% (95% confidence intervals (CI) 0.035–0.16%). Applying the Rogan–Gladen estimate with the Lang–Reiczigel CI method to account for test sensitivity and specificity resulted in a true seroprevalence estimate of 0.103% (95% CI 0.09–0.12%) (Supplementary Appendix). This corresponds to an infection to case ratio of 2.3, based on notified cases on 6 January 2021, suggesting some undiagnosed infections have occurred. However, this ratio needs to be interpreted with caution for two reasons. First, the limitations of the sampling population including the absence of those aged <16 years of age, and a lower proportion of those of Māori and Pacific ethnicity compared with the 2018 census population. Second, the extremely small number of seropositive donors makes extrapolation unreliable and this also precludes any subgroup analysis.
Antibody characteristics of the seropositive donors (n = 18). (a) Seropositivity was confirmed by EuroImmun S1 IgG (top) and the surrogate Viral Neutralisation Test (sVNT, bottom). Six donors had PCR-confirmed SARS-CoV-2 infection (dark grey), four had relevant travel history (dark turquoise) and eight were identified in this study (orange). The manufacturer cut-offs are shown (black dotted line). (b) Pearson correlation of sVNT and the Euroimmun IgG ELISA (n = 18). (c) Rose plot showing the percentage of seropositive donors over baseline for IgG, IgA and IgM antibodies against the RBD, Spike (S) and nucleocapsid (N) proteins determined using a multi-plex Luminex bead assay.

The very low seroprevalence of SARS-CoV-2 infection in New Zealand implies that undetected community transmission has been limited. This seroprevalence is broadly similar to a recent study conducted in the low prevalence city of Sydney in Australia [Reference Gidding3], and markedly lower than estimates of >10% from serosurveys in Europe and North America where the pandemic has been poorly controlled (https://serotracker.com). This study provides robust, serological evidence of New Zealand's successful elimination strategy ahead of vaccine roll-out and highlights the value of a nationwide blood donor service to monitor viral spread during the pandemic.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0950268821001643
Data
Data, in addition to those available in the Supplementary information, are available from the authors on request.
Acknowledgements
This work was funded by the School of Medicine Foundation (University of Auckland), and the COVID-19 Innovation Acceleration Fund (Ministry of Business, Innovation and Employment). The 2018 Census data used in this study were supplied by Statistics New Zealand (Stats NZ) and accessed via its Integrated Data Infrastructure (IDI).
Statistics New Zealand Disclaimer
Access to the data used in this study was provided by Stats NZ under conditions designed to give effect to the security and confidentiality provisions of the Statistics Act 1975. The results presented in this study are the work of the author, not Stats NZ or individual data suppliers.




