I. INTRODUCTION
We know Isaac Newton today primarily as a mathematician and a physicist. But the co-creator of calculus (with G. W. Leibniz) and the discoverer of the law of universal gravitation also spent some 40 years studying “chymistry,” the early modern discipline that blended practices akin to modern chemical science with transmutational beliefs drawn from alchemy. In fact, Newton left a body of manuscripts consisting of about 1 million words on the subject of chymistry in his hand. Among these cryptic documents, one even finds two dated laboratory notebooks describing Newton’s laboratory experiments. In total, these notebooks consist of well over 200 folios (400 pages), filled with quantitative records expressed in the apothecaries’ weights that were customary in Newton’s time. One might think that such a record left by one of the greatest scientific minds of all time would have been thoroughly mined for its content by previous scholars and that modern researchers might even have tried to replicate Newton’s experiments in the laboratory. However, multiple factors have unfortunately conspired to prevent that from happening until the present. First, Newton’s chymical manuscripts were long kept under lock and key, not to be divulged to the public, by his heirs, the Earls of Portsmouth, until the late nineteenth century. At that point, a catalog was drawn up by a team or “syndicate,” and the manuscripts that were deemed to be of scientific value were given to the library of the University of Cambridge. But even then, the chymical notebooks proved to be impenetrable, as the brief and unsatisfactory notes that the chemist George D. Liveing, a member of the examining team, appended to them show. This leads to the second, and greater, problem faced by the interpreter of Newton’s notebooks. Newton did not use chymical terminology in a straightforward fashion, even by the standards of his contemporaries. For example, the term “vitriol” in Newton’s day usually meant a crystalline metallic sulfate, such as copper(II) sulfate pentahydrate. To Newton, it meant something else entirely, a synthetic crystalline product made by combining a metal or metal compound with a solution that he called “liquor of antimony” (a solution of Sb2S3, the mineral stibnite, and NH4Cl [sal ammoniac] in nitric acid) and then allowing the solution to crystallize (Newman, Reference Newman2019). This is only one of many examples where Newton employed his own idiosyncratic terminology in the materials described in his notebooks. A final barrier to the understanding of Newton’s chymistry lies in the institutional barriers typically separating scientific and historical research. Only in recent years has the emergence of Experimental History of Science as a distinct field encouraged historians and scientists to join efforts in deciphering pre-modern laboratory work (Fors et al., Reference Fors, Principe and Sibum2016).
Fortunately, a recently discovered document provides a new key to deciphering Newton’s Cambridge laboratory notebooks (Newman, Reference Newman2019). A letter found in the William Andrews Clark Library at UCLA contains a series of chymical instructions that Newton gave his good friend, the Genevan mathematician Nicolas Fatio de Duillier, in 1693. Comparison of these sequential directions with Newton’s laboratory notebooks has allowed one of us (W.R.N.) to show that Newton was giving Fatio simplified results of the projects found in Newton’s own experimental records. Because the descriptions in Fatio’s letter are written in plain, unencoded language (albeit in Latin), it has been possible for us to reconstruct the processes described in them by Newton and to analyze the products.
Fundamental to Newton’s chymical quest was his desire to render the then-known metals volatile. Thinking in terms of a hierarchical corpuscular theory akin to the modern idea of a complex atom composed of subatomic particles held together by forces, Newton believed that metals could in principle be broken down into more primitive components and then rebuilt however one wished. One marker of this decomposition to Newton was volatility, as a smaller corpuscle of a given substance might reasonably be thought to be more capable of undergoing sublimation than a larger corpuscle of the same material. Newton’s instructions to Fatio show that his process began with the dissolution of a copper compound in order to produce a vitriol that could later be sublimed with other materials. Newton did this by reacting a material that he calls viride aeris (probably verdigris) with his so-called liquor of antimony. He then allowed the decanted solution to crystallize slowly by evaporation at room temperature. The resulting product of our reproduction of these experiments revealed a mixture of different solid constituents that are all capable of being sublimed, vaporized, or decomposed (partly into gases) at temperatures below 400 °C. Most components of the crystallized solid mass were identified by powder X-ray diffraction (XRD). However, one of these phases, the main subject of this contribution, has not been identified heretofore and was investigated using single-crystal XRD methods.
II. EXPERIMENTAL DETAILS
A. Material synthesis
Preparation of Newton’s vitriol proceeded by first preparing his liquor of antimony. In accordance with the practices recorded in Newton’s laboratory notebooks, 155 g reagent-grade NH4Cl was added to 450 ml 1.75 M HNO3 and dissolved at ~308 K. A total of 91 g technical-grade Sb2S3 (confirmed by powder XRD) was added, and after the ensuing boiling reaction ceased, the solution was left overnight and was then decanted and filtered (yielding Newton’s liquor of antimony). Next, 50 g reagent-grade Cu(CH3COO)2 (Aldrich, Newton’s verdigris) was added to the filtered solution, which was allowed to sit for 1 h. The solution was then decanted and filtered, leaving a green precipitate (sample NS#24d) as a cake on the filter paper. This green solution was then allowed to crystallize in open air in a crystallizing dish for several weeks, yielding a heterogeneous layer of blue-green crystals with dispersed clear centimeter-sized rosettes on the surface (Figure 1).
Photograph of sample NS#24, showing the rosettes (NS#24a) and the underlying blue-green crust (NS#24b). The diameter of the dish is 12 cm.

B. X-ray powder diffraction
The crystalline material resulting from these experiments was manually separated into the clear rosettes (our sample NS#24a) and the underlying blue-green crystals (sample NS#24b). Both components were gently crushed in an agate mortar and pestle with no liquid, as the products are soluble in many liquids, and the resulting powders were placed in 1-mm-deep cavities milled into Ti mounts. The green precipitate (sample NS#24d), which formed before the crystallization of these two samples, was also analyzed. Powder XRD data were measured on a Bruker D8 Advance diffractometer with Cu Kα radiation and a SolX energy-discriminating detector. The instrument was calibrated using NIST SRM 660a. Data were measured from 2° to 120° 2θ with a step size of 0.02° in continuous-scanning mode, counting for 30 s/step (25 min/° for #24a) or 17 s/step (14.2 min/° for #24b and #24d). Additional measurements were made under controlled relative humidity (RH) using an RH-controlled cell (InstruQuest Inc. V-Gen controller) on the diffractometer. Rietveld refinements used Bruker AXS Topas Version 6 (Bruker AXS, 2016) using a fundamental-parameters approach. The emission spectrum was modeled by a series of five Lorentzians (Berger, Reference Berger1986), and the background was modeled by a fourth-order Chebyshev polynomial. Lorentzian crystallite size and strain contributions were refined for the major phases. Refinements for NS#24a used a March function preferred orientation correction for NH4Cl, NH4NO3, and (NH4)2CuCl4(H2O)2, and a spherical harmonics preferred orientation correction was applied for (NH4)2Cu(SO4)2(H2O)6. No correction was applied for the remaining phases. Refinements for NS#24b used a March function preferred orientation correction for NH4Cl, the newly discovered phase, and (NH4)2CuCl4(H2O)2, and no correction was applied for the remaining phases.
C. Single-crystal X-ray diffraction
Numerous crystals were extracted from sample NS#24b and analyzed, but most were either unsuitable or were the materials identified by powder XRD (e.g., NH4Cl, CuCl4(NH4)2(H2O)2, and NH4NO3). A blue crystal obtained from sample NS#24b (~0.465 × 0.447 × 0.362 mm3) was placed onto the tip of a MiTeGen™ loop and mounted on a Bruker Venture D8 diffractometer equipped with a PhotonIII detector. Data were measured at 173(2) K using Mo Kα radiation (microfocus sealed tube, multilayer mirror monochromator) with an exposure time of 0.75 s and a detector distance of 4.00 cm. Data were collected with 1° ω and ϕ scans to a resolution of 0.75 Å. A total of 1,395 images were collected over a total measurement time of 0.29 h. The images were integrated with the Bruker SAINT software package (V8.41; Bruker AXS, 2024).
III. RESULTS AND DISCUSSION
A. X-ray powder diffraction results
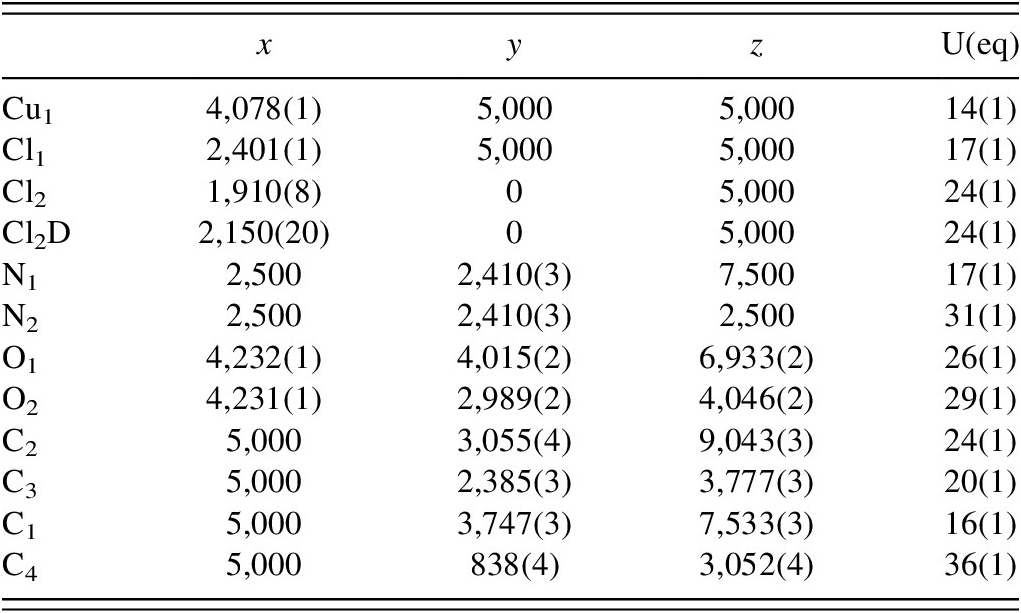
XRD data for powders of the centimeter-sized rosettes (sample NS#24a) were measured under room conditions (291.75 K and 35% RH) and also under controlled-RH conditions at 0% and 40% RH at 292.7 K, and both measurements yielded identical diffraction data, with the exception of several very weak low-angle peaks. Qualitative analysis of the data supported by successful Rietveld refinement showed the presence of (NH4)2Cu(SO4)2(H2O)6 (ICSD 2019135) (62(1)%), NH4NO3 (ICSD 28069) (15.8(9)%), NH4Cl (8.3(3)%), (NH4)2CuCl4(H2O)2 (ICSD 420586) (4.8(3)%), and (NH4)[Cu(NH3)2Cl3]⋅2H2O (ICSD 120) (5.3(3)%). Traces (1.0(1)%) of the new (NH4)2[Cu2Cl2(C2H3O2)4]·2NH4Cl phase characterized in sample NS#24b and of CuSO4·5H2O (ICSD 196306) (1.2(2)%) were identified in NS#24a. These phases accounted for all but several weak peaks, which include peaks at 10.72°, 17.42°, 18.65°, and 27.34° 2θ; these peaks did not fit anything in the PDF-5 database (Kabekkodu et al., Reference Kabekkodu, Dosen and Blanton2024). A portion (low-intensity, high-angle data are not shown) of the plot of the data showing the fit with these phases is shown in Figure 2 (R wp = 8.58, Rp = 5.27, GOF = 4.01). The largest difference peaks are from (NH4)2Cu(SO4)2·6H2O, and it appears that these differences may be at least partly attributable to inadequacies in the structure model for this phase.
Plot of powder X-ray diffraction data for sample NS#24a (blue), with the results of Rietveld refinement (red). The difference curve is in gray. The tick marks at the bottom of the plot reflect the positions of possible reflections for the corresponding color-keyed phase.
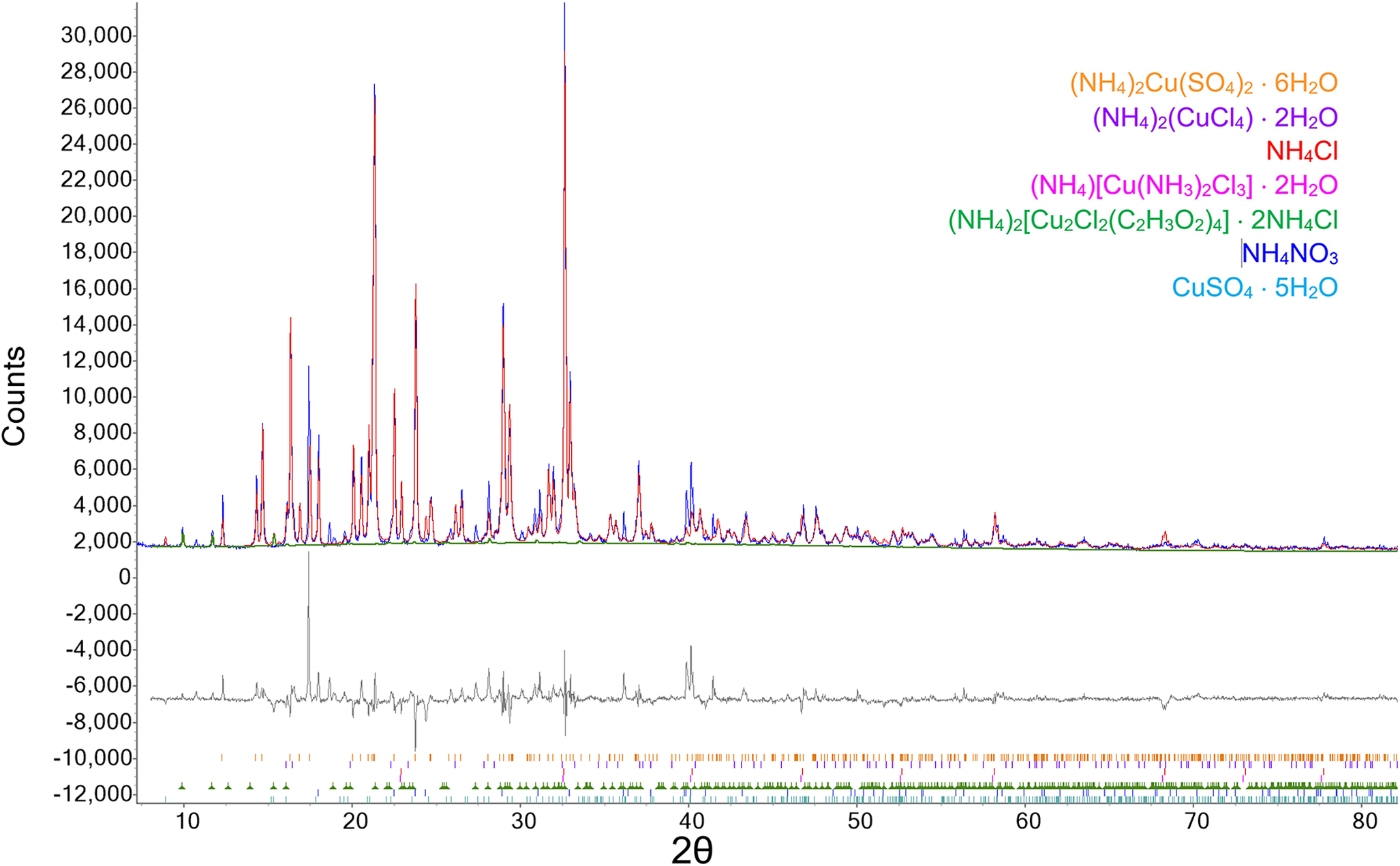
Several chunks of the massive, crystalline dark green material covering the bottom of the dish were extracted (our sample NS#24b), attempting to avoid the overlying rosettes. The material was ground as above, yielding a dry powder that was not hygroscopic or sticky. Data were measured at 294.5 K and 21% RH as above. Qualitative analysis of the data and subsequent Rietveld refinement revealed the presence of NH4Cl (ICSD 22141), (NH4)2CuCl4(H2O)2 (ICSD 420586), (NH4)[Cu(NH3)2Cl3]⋅2H2O (ICSD 120), and NH4NO3 (ICSD 28069), but there was a significant other phase, along with a weak, broad peak at ~7.2° 2θ. We have observed this broad peak for other products from similar experiments, and the peak does not appear in patterns of other materials using the same mount. Although there is a slight beam overlap onto the Ti sample holder at 7.2° 2θ with our 1° divergence slit, the holder is pure Ti metal and shows no unusual low-angle or background features. Measurement of the inverted holder gives the expected diffraction pattern from crystalline Ti. A search using the 2024 ICDD PDF-5 database (Kabekkodu et al., Reference Kabekkodu, Dosen and Blanton2024) resulted in no matches for the additional crystalline phase. A search of the Inorganic Crystal Structure Database (ICSD release 5.3.0; Zagorac et al., Reference Zagorac, Müller, Ruehl, Zagorac and Rehme2019) likewise produced no satisfactory matches to the unmatched peaks, within the constraints of our known chemistry. Single-crystal analysis (see below) of a crystal extracted from NS#24b gave a formula of (NH4)2[Cu2Cl2(C2H3O2)4]·2NH4Cl, and the inclusion of this phase in the Rietveld refinement for NS#24b gave an excellent fit with only 1 or 2 weak unmatched peaks remaining. The weak, broad 7.2° 2θ band was independently fit with a Pearson VII profile. The final refined composition was 5.4(2)% (NH4)2[Cu2Cl2(C2H3O2)4]·2NH4Cl, 60.7(5)% NH4Cl, 21.6(4)% (NH4)2CuCl4 (H2O)2, 8.4(3)% (NH4)[Cu(NH3)2Cl3]⋅2H2O (ICSD 120), and 3.9(2)% NH4NO3 (ICSD 28069). Atomic positions were not refined for any of the component phases, and only unit-cell parameters were allowed to vary. Refined unit-cell parameters for the new acetate phase were a = 14.547(6) Å, b = 8.851(1) Å, and c = 9.118(2) Å. A plot of the data showing the fit using these phases is shown in Figure 3 (R wp = 7.74, Rp = 5.30, GOF = 2.20). All of the largest difference peaks are from NH4Cl.
Plot of powder X-ray diffraction data for sample NS#24b (blue), with the results of Rietveld refinement (red) and difference curve (gray). The tick marks at the bottom of the plot reflect the positions of possible reflections for the corresponding color-keyed phase.

It is noteworthy that we detected no significant crystalline Sb-containing compound in either of these samples, although the component in NS#24b contributing to the broad peak at ~7° 2θ may contain Sb. Chemical analysis of sample NS#24d, the precipitate (~50% amorphous with (NH4)2CuCl4 (H2O)2 and NH4Cl), revealed significant Cu (18.65%) and Sb (19.89%), and it appears that the amorphous component in NS#24d is an Sb oxide, based on the absence of Cl and any crystalline Sb-containing phase.
B. Single-crystal X-ray diffraction results
Integration of the single-crystal XRD data using an orthorhombic unit cell yielded a total of 38,684 reflections to a maximum θ angle of 28.29° (0.75 Å resolution), of which 1,484 were independent (average redundancy 26.067, completeness = 99.2%, R int = 7.44%, R sig = 3.12%) and 1,376 (92.72%) were greater than 2σ(F 2). Final unit-cell parameters of a = 14.4535(4) Å, b = 8.7728(3) Å, c = 9.1061(3) Å, and volume = 1,154.63(6) Å3 are based on the refinement of the XYZ centroids of 9,928 reflections above 20 σ(I) with 5.287° < 2θ < 56.56°. Differences between these values and the Rietveld-refined values are consistent with the lower temperature of measurement for the single-crystal data (173 vs. 293.9 K). Data were corrected for absorption effects using the Multi-Scan method (SADABS; Krause et al., Reference Krause, Herbst-Irmer, Sheldrick and Stalke2015). The ratio of minimum to maximum apparent transmission was 0.661. The calculated minimum and maximum transmission coefficients (based on crystal size) are 0.4090 and 0.4840.
C. Structure solution and refinement
Space group Pmna was determined based on intensity statistics and systematic absences. The structure was solved and refined using the SHELX suite of programs (Sheldrick, Reference Sheldrick2015a, Reference Sheldrick2015b). All non-hydrogen atoms were refined with anisotropic displacement parameters, whereas the hydrogen atoms were placed in ideal positions and refined as riding atoms with relative isotropic displacement parameters. One of the (NH4)+ ions, the Cl− ions, and the methyl groups are disordered and were refined using geometrical and displacement restraints and constraints. Half of the formula unit is crystallographically unique, that is, all ions are situated in special positions, including inversion centers, two-fold axes, and a mirror. Both NH4 molecules are located on crystallographic twofold axes, and displacement of N2 suggests disorder near the twofold. However, such a refinement failed to converge. The hydrogen atoms of N1 follow twofold symmetry, whereas those of N2 (as observed by electron density in their bonds) do not, and two distinct orientations were refined; constraints were applied to obtain and then maintain a chemically reasonable conformation. Displacement of Cl− at a single site indicated disorder, with both Cl sites remaining on a twofold axis. The site occupancy was freely refined to the reported values. The methyl groups are located on a mirror but do not feature mirror symmetry and therefore must be disordered over the symmetry element, in our case a twofold (thus 50:50). Hydrogen atoms were refined as riding on the parent atom, allowing torsion while maintaining a chemically sensible geometry at 50% occupancy; the equivalent positions provide the second orientation of those hydrogen atoms.
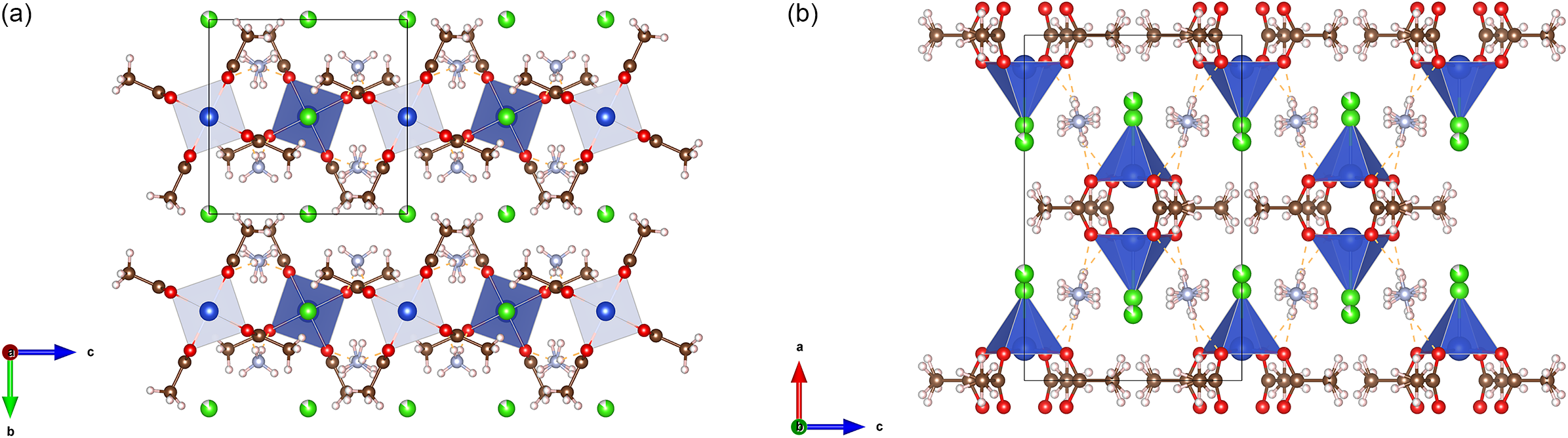
The final anisotropic full-matrix least-squares refinement on F 2 with 72 variables converged at R1 = 3.49% for the observed data and wR2 = 9.29% for all data. The goodness of fit was 1.244. The largest peak in the final difference electron density synthesis was 0.484 e−/Å3 and the largest hole was −0.880 e−/Å3 with an RMS deviation of 0.118 e−/Å3. On the basis of the final model, the calculated density was 1.660 g/cm3 and F(000) = 588 e−. Hydrogen bonding was observed between the ammonium ion and carboxylate groups and the free chlorine anion. Table I provides atomic coordinates for (NH4)2[Cu2Cl2(C2H3O2)4]·2NH4Cl. A diagram of the structure is shown in Figure 4, and packing is shown in Figure 5a,b.
Atomic coordinates (× 104) and equivalent isotropic displacement parameters (Å2 × 103) for (NH4)2[Cu2Cl2(C2H3O2)4]·2NH4Cl

U(eq) is defined as one-third of the trace of the orthogonalized Uij tensor.
Molecular structure of (NH4)2[Cu2Cl2(C2H3O2)4]·2NH4Cl. Thermal ellipsoids are pictured at the 50% probability level, and H atoms are shown as spheres of arbitrary radius. Cu atoms are light blue, Cl green, O red, N blue, C gray, and H white. Hydrogen bonding is displayed with dashed lines. Disorder has been omitted for clarity.

Packing plots with Cu environments shown as polyhedra: (a) viewed down the a-axis and (b) viewed down the b-axis. Disorder of the methyl groups and NH4+ is illustrated, and disorder of the Cl− atoms is omitted for clarity.
IV. SUMMARY
The results of these replication experiments have considerable implications for our understanding of Isaac Newton’s work in chymistry. A few years after the bulk of Newton’s alchemical papers were sold by the celebrated auction house Sotheby’s in 1936, the economist John Maynard Keynes, who had purchased a large number of the manuscripts, wrote a seminal article in which he expressed his opinion about Newton’s chymical endeavors. Keynes famously stated that Newton was not “the first of the age of reason,” but “the last of the magicians.” Moreover, Keynes asserted in the same article that Newton’s chymical manuscripts were “wholly magical and wholly devoid of scientific value” (Keynes, Reference Keynes1947). A similar sentiment has been echoed by many subsequent historians (Monod, Reference Monod2013; Strathern, Reference Strathern2019). However, these comments about Newton’s “magical” experiments raise a serious question. How is it that one of the greatest experimental minds in history could have persisted in his chymical laboratory work for some three decades without experiencing any scientific success at all? Is it really the case that his chymistry was “wholly devoid of scientific value“? Before answering this question, we should bear in mind that it was the same Newton whose work on prisms overthrew some two millennia of optical theory when he demonstrated that white light consists of a mixture of unaltered spectral colors. Is it really likely that such a gifted and fastidious experimenter would continue for nearly 30 years in an entirely unfruitful project that offered no prospect of success? Our results suggest otherwise and provide a starkly countervailing image of Newton’s laboratory work. He was in fact doing innovative and serious chemistry even by modern standards, a fact that his production of a compound unreplicated until the present, (NH4)2[Cu2Cl2(C2H3O2)4]·2NH4Cl, makes undeniable. Our replications of a number of his other experiments have similarly yielded new, previously unidentified compounds. His synthesis methods provided pathways that, even today, are not often pursued (e.g., sublimation). Newton must have realized that his volatile metallic compounds provided a path to future research that his contemporaries had not yet realized.
DEPOSITED DATA
Crystallographic data for (NH4)2[Cu2Cl2(C2H3O2)4]·2NH4Cl have been deposited with the Cambridge Crystallographic Data Center (CCDC) (Deposition No. 2400719) and can be obtained free of charge from the CCDC via www.ccdc.cam.ac.uk.
ACKNOWLEDGEMENTS
W.R.N. thanks Dr. Cate Reck for the use of her laboratory for synthesis experiments.
CONFLICTS OF INTERESTS
The authors declare no competing interests.
FUNDING STATEMENT
Support for the acquisition of the Bruker Venture D8 diffractometer through the Major Scientific Research Equipment Fund from the Office of the President of Indiana University and the Office of the Vice President for Research is gratefully acknowledged. W.R.N. acknowledges support from NSF grant SES-2240879.