Introduction
Schizophrenia (SZ) is a complex psychiatric disorder affecting ~24 million people or 1 in 300 individuals worldwide, according to the 2021 census (WHO, 2022). Antipsychotic drugs are prescribed to such patients alone or in combination (with other antipsychotic agents) to alleviate symptoms (Doane et al., Reference Doane, Sajatovic, Weiden, O’Sullivan, Maher, Bjorner, Sikora Kessler, Carpenter-Conlin, Bessonova and Velligan2020; Lähteenvuo & Tiihonen, Reference Lähteenvuo and Tiihonen2021). There is substantial evidence in the literature showing the immunomodulatory effect of antipsychotic drugs (Drzyzga, Obuchowicz, Marcinowska, & Herman, Reference Drzyzga, Obuchowicz, Marcinowska and Herman2006). For example, one study reported that in female SZ patients, increasing doses of antipsychotic drugs were associated with higher neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) (Frota et al., Reference Frota, de Oliveira, De Lima, Costa Filho, Menezes, Soares, Chaves Filho, Lós, Moreira, Viana, Campos, Vasconcelos, Seeman, Macêdo and Sanders2023). Chen and colleagues reported suppression of interferon-γ (IFNγ) production by clozapine in peripheral blood mononuclear cells (PBMCs) (Chen et al., Reference Chen, Tsai, Wang, Lin, Tsai, M-C and Tsai2012) and noted that both risperidone and clozapine inhibited the production of interleukin-6 (IL-6), IL-8, and IL-12, and increased IL-10 production in lipopolysaccharide (LPS)-stimulated macrophages (Chen et al., Reference Chen, Wu, Tsai, L-K and Tsai2013). Similarly, in polyinosinic:polycytidylic acid (PIC) and LPS-stimulated PBMC cultures, haloperidol, clozapine, risperidone, and quetiapine increased the production of anti-inflammatory cytokines (IL-4 and IL-10), while decreasing the production of pro-inflammatory cytokine (IFNγ) (Al-Amin, Uddin, & Reza, Reference Al-Amin, Uddin and Reza2013). In vitro studies also show decreased neutrophil survival rate post-clozapine treatment (48 h, 50 μM clozapine) (Goto et al., Reference Goto, Yoshimi, Nagai, Ukigai, Mouri, Ozaki and Noda2017). It is well documented that clozapine is associated with neutropenia in 3% of cases and agranulocytosis in 0.8% of cases (Matsui et al., Reference Matsui, Ishibashi, Kawano, Oshibuchi, Ishigooka, Nishimura and Inada2020; Mijovic & MacCabe, Reference Mijovic and MacCabe2020). Furthermore, a recent meta-analysis demonstrated that risperidone had clear anti-inflammatory effects, with a decrease in IL-6, IL-1β, and tumor necrosis factor-α (TNF-α) in the plasma/serum of patients, and no effect was observed in the case of clozapine (S. R. Patlola, Donohoe, & McKernan, Reference Patlola, Donohoe and McKernan2023a).
Studies have also shown the immunomodulatory effect of antipsychotic drugs on Toll-like receptors (TLRs), whose activity regulates the production of cytokines. One particular study on human postmortem brain determined that the patients on antipsychotic drugs showed higher protein expression of TLR4 and MyD88 compared to controls, and only for MyD88, messenger RNA (mRNA) expression was significantly higher in the antipsychotic drug group compared to the drug-naïve SZ group (García-Bueno et al., Reference García-Bueno, Gassó, MacDowell, Callado, Mas, Bernardo, Lafuente, Meana and Leza2016). Other studies have reported that the percentage of TLR4+ and TLR5+ monocyte cells in blood decreased in patients after risperidone or olanzapine (OLZ) treatment (8 weeks), whereas the percentage of TLR2+ monocytes increased (Kéri, Szabó, & Kelemen, Reference Kéri, Szabó and Kelemen2017). Balaji and colleagues reported no change in mRNA expression levels of TLR3 and 4 in PBMCs after 3 months of antipsychotic medication (Balaji et al., Reference Balaji, Subbanna, Shivakumar, Abdul, Venkatasubramanian and Debnath2020). At the cellular level, antipsychotic drugs seem to contribute toward the changes in immune cell counts, while at the molecular level, they seem to affect the TLR number, activity, and their downstream signaling products, such as cytokines.
Immune modulation by antipsychotic drugs has been associated with changes in cognitive function. For example, Kéri et al. (Reference Kéri, Szabó and Kelemen2017) reported that higher percentages of TLR4+ and TLR5+ monocyte levels were found to be associated with decreased cognitive performance, which was not observed post-antipsychotic drug administration (Kéri et al., Reference Kéri, Szabó and Kelemen2017). Frota et al. reported that higher antipsychotic drug doses were associated with performance decline in working memory, processing speed (PS), and executive function, possibly due to changes in the immune system (NLR and PLR ratios) (Frota et al., Reference Frota, de Oliveira, De Lima, Costa Filho, Menezes, Soares, Chaves Filho, Lós, Moreira, Viana, Campos, Vasconcelos, Seeman, Macêdo and Sanders2023). Furthermore, a recent article discusses the cognitive benefits of reducing the antipsychotic drug dose. They also discuss the effects of antipsychotic drugs on neuroinflammation, further modulating neurotransmitter pathways and cognition (Allott et al., Reference Allott, Chopra, Rogers, Dauvermann and Clark2024). Consequently, based on current research, antipsychotic drugs might have an immunomodulatory effect and negatively affect cognitive performance. However, it is unclear if immune alteration mediates the cognitive changes due to antipsychotic drugs. Moreover, our recent work showed that TLR2 and TLR4 activity negatively impact cognition via cytokines (Patlola et al., Reference Patlola, Holleran, Dauvermann, Rokita, Laighneach, Hallahan, McManus, Kenyon, McDonald, Morris, Kelly, Donohoe and McKernan2025). Hence, we are interested in understanding if the changes in the immune system and cognition are affected by antipsychotic drugs.
Another crucial factor that needs to be taken into account is the anticholinergic burden (ACB). Most drugs used to treat psychiatric disorders, including antipsychotic drugs, mood stabilizers, and antidepressants, tend to have anticholinergic side effects (Lieberman 3rd, Reference Lieberman2004) and can cause cognitive impairment. Past studies have demonstrated that ACB has a significant impact on the functional capacity and cognition of individuals with SZ (Joshi et al., Reference Joshi, Thomas, Braff, Green, Gur, Gur, Nuechterlein, Stone, Greenwood, Lazzeroni, MacDonald, Molina, Nungaray, Radant, Silverman, Sprock, Sugar, Tsuang, Tsuang and Light2021; O’Reilly et al., Reference O’Reilly, O’Connell, Donohoe, Coyle, O’Sullivan, Azvee, Maddock, Sharma, Sadi, McMahon and Kennedy2016). This effect was shown to be more pronounced in verbal memory, mediating the worsening of functioning in first-episode psychosis cohort (Ballesteros et al., Reference Ballesteros, Sánchez Torres, López-Ilundáin, Mezquida, Lobo, González-Pinto, Pina-Camacho, Corripio, Vieta, de la Serna, Mané, Bioque, Moreno-Izco, Espliego, Lorente-Omeñaca, Amoretti, Bernardo and Cuesta2021). Therefore, we incorporated both antipsychotic drug dose and ACB in our analysis.
In this study, we hypothesized that antipsychotic drugs have an immunomodulatory effect on cytokine levels and TLR activity. We also aim to investigate whether antipsychotic medication affects cognition and whether the antipsychotic drug dose moderates the relationship between immune biomarkers and cognition.
Methods and materials
Participants
From a total of 300 participants recruited as part of the ‘Immune Response & Social Cognition in Schizophrenia’ research project, 280 (N = 79 patients and 201 healthy controls [HCs]) had the necessary data available for this study. This is a cross-sectional and observational study design. The patient group consisted of clinically stable patients with either a diagnosis of SZ or schizoaffective disorder. Diagnosis was confirmed using the Structured Clinical Interview for Diagnostic Statistical Manual-IV, and patients were recruited from local outpatient clinics and mental health services. The Positive and Negative Syndrome Scale (PANSS) was used to measure the symptoms and severity of SZ, with the Hamilton Depression and Rating Scale (HAM-D) utilized to measure depressive symptoms. Both the PANSS and HAM-D (17-item) have high reliability and validity metrics (Kay, Fiszbein, & Opler, Reference Kay, Fiszbein and Opler1987; Maier & Philipp, Reference Maier and Philipp1985). Healthy participants were recruited from the general population. Participants were excluded based on the following criteria: (1) a history of acquired brain injury causing loss of consciousness of >1 min; (2) substance abuse in the last 6 months, and (3) intellectual disability. All participants were aged between 18 and 65 years. Additional information on the sample, including full inclusion and exclusion criteria, has been detailed in the Supplementary Methods. All individuals gave informed written consent before the study, and assessments were conducted in accordance with the relevant ethics committee approval.
Blood collection and plasma isolation
Venous blood was collected from HCs and SZ patients in 10 mL EDTA tubes (Catalog# BD367873) for plasma and 2.5 mL PAXgene® Blood tubes for RNA (Qiagen, Catalog# 762165) at ~9.00 AM (for every participant). Approximately 3–4 mL of blood (EDTA tubes) was centrifuged at 1,200 g for 10 min at room temperature, and the resultant supernatant (plasma) was then aliquoted and stored at −80 °C for further analysis. The remaining blood was used for stimulation studies carried out immediately after collection. Additional information is available in the Supplementary Methods.
Immune assays for protein quantification
Enzyme-linked immunosorbent assay (ELISA) was used to quantify cytokine and C-reactive protein (CRP) levels in blood plasma and from stimulated whole blood cultures. For stimulations, TLR ligands were used, such as TLR2 – heat-killed Listeria monocytogenes (1010 cells of HKLM) (Invivogen, Catalog# tlrl-hklm), TLR3 – 10 μg/mL PIC (Invivogen, Catalog# tlrl-pic), and TLR4–1 μg/mL LPS (Invivogen, Catalog# tlrl-eklps). Blood cultures were treated with the ligands for 24 h, and the supernatant was harvested. This was stored at −80 °C for later analysis. Cytokines, such as IL-6, IL-8, IL-10, and TNF-α, were assessed using the supernatant by DuoSet ELISA kits (Bio-Techne Ltd.; R&D Systems) and plasma cytokines by Quantikine High Sensitivity ELISA kits (and Quantikine kit for CRP) from Bio-Techne Ltd. The procedures and technical information for both assays are detailed in the Supplementary Methods.
Cognitive assessment
The participant’s cognitive performance was recorded using Wechsler scales and Cambridge Neuropsychological Test Automated Battery (CANTAB) tests. The tasks measured and utilized in this study include digit symbol coding (scores 0–133), logical memory-recall (scores 0–75; 25/iteration), full-scale intelligence quotient, letter-number sequence (21 items; scores 0–21), paired associates learning (total errors scores from 6 shapes adjusted), and reading in the mind of the eyes task (18 items; 0–18). Furthermore, these cognition tasks were assigned to specific domains (Supplementary Methods and Supplementary Table S1).
OLZ dose equivalents calculation
The dose from each antipsychotic drug was converted to the equivalent of OLZ based on the defined daily dose method from the World Health Organization, calculated by Leucht, Samara, Heres, & Davis (Reference Leucht, Samara, Heres and Davis2016). In the case of polypharmacy (multiple antipsychotic drugs prescribed in combination) or parenteral administrations, drug doses per day were calculated, which were then converted to OLZ equivalents. These antipsychotic drug daily doses were added to get the total OLZ equivalent dose/day/individual.
ACB scores calculation
ACB scores were calculated based on a scale from 0 to 3, with 0 being no anticholinergic effect, a score of 1 indicating a possible anticholinergic effect, and scores of 2 and 3 indicating a definite anticholinergic effect (Fox et al., Reference Fox, Richardson, Maidment, Savva, Matthews, Smithard, Coulton, Katona, Boustani and Brayne2011). Using an online tool (King Rebecca, Reference King Rebecca2024), ACB scores were calculated for all drugs, including antipsychotic drugs, and psychiatric and nonpsychiatric drugs. Individual drug ACB scores were summed for every participant to get a cumulative ACB score, which is used in the analysis.
Statistical analysis
The data were tested for normality using the Shapiro–Wilk test and homogeneity of variance using Fisher’s test. Plasma cytokines, TLR activity, and cognition were all analyzed using Mann–Whitney unpaired t-test using GraphPad Prism V.10.3.1. Bonferroni correction was applied to these tests to factor for multiple testing. Multiple linear regressions, principal component analysis (PCA), and moderation analysis were conducted using IBM SPSS V.29 (IBMCorp, 2023). Bootstrapping was performed in these analyses to address the disproportionate sample variances between controls and patients, along with increasing the confidence of our results. Sample size variations in linear regression data reflect the missing data either in the dependent or independent variables, or both.
Model summaries, sample sizes, and analysis of variance were used to check the quality of multiple linear regressions. For multiple linear regressions, the Kaiser–Meyer–Olkin measure of sampling adequacy, Bartlett’s test of sphericity, and scree plots were used as quality control.
We used moderation analysis in our study, and this investigates if the relationship between two variables is strengthened or weakened depending on the level of the third variable knowns as a moderator. Moderation analysis was conducted using PROCESS V.4.2 in SPSS. ‘Model 1’ (Stride, Gardner, Catley, & Thomas, Reference Stride, Gardner, Catley and Thomas2015) was used in our study, and its quality was determined using model summaries. TLR2 and 4 activities are composite variables based on the PCA of IL-6, IL-8, IL-10, and TNF-α levels from blood stimulations (data presented in Patlola et al. [Reference Patlola, Holleran, Dauvermann, Rokita, Laighneach, Hallahan, McManus, Kenyon, McDonald, Morris, Kelly, Donohoe and McKernan2025]). The detailed list of quality checks and model summaries is available in the Supplementary Documents.
Results
Sociodemographic data
The detailed sociodemographic data are presented in Table 1. Patients (SZ) in our study were older (p < 0.0001) and spent a similar time in education (p = 0.43) compared to HCs. Individuals with SZ had a higher body mass index (BMI) (p < 0.0001) and showed a higher incidence of depression (p < 0.0001). Patients also showed significantly lower cognitive performance (p < 0.05) than controls in full-scale IQ, digit symbol coding, logical memory, letter-number sequence, paired associates learning, and reading of mind in the eyes tasks. The results shown here belong to subset of full cohort (Patlola et al., Reference Patlola, Holleran, Dauvermann, Rokita, Laighneach, Hallahan, McManus, Kenyon, McDonald, Morris, Kelly, Donohoe and McKernan2025). The subset was filtered to remove patients not taking any medication or presented incomplete information on medication. Participants were also excluded if they had high CRP levels (>10 mg/L), as it could be due to an underlying health condition that was not known to the participant at that time or was not reported.
Sociodemographic and cognition data

Note: P-values in this table are based on a non-parametric (Mann–Whitney test) T-test between HC and SZ. BMI, body mass index; PANSS, Positive and Negative Symptom Scale; HAM-D 17, Hamilton Depression Rating Scale 17 items; antipsychotic drug, antipsychotic drug; OLZ, olanzapine; IQ, intelligence quotient; DSC, digit symbol coding; LM, logical memory; PAL, paired associates learning; LNS, letter number sequencing; RME, reading the mind in the eyes. N, sample size; Std. Dev., standard deviation; Bold, significant.
TLR activity and plasma cytokines
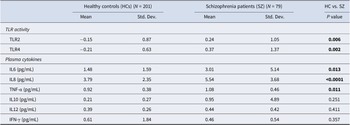
TLR activity is a composite score measure derived from a PCA based on the levels of stimulated cytokine release (IL-6, IL-8, IL-10, and TNF-α) following TLR activation for 24 h using agonists specific for each TLR, for example, TLR2 – HKLM (1010 cells), TLR3 – Poly I:C (10 μg/mL), and TLR4 – LPS (1 μg/mL). Participants in the SZ cohort displayed higher mean TLR2 and four activities (p < 0.01) compared to HCs (Table 2). Patients showed significant elevation in circulating cytokines (excluding IL-10, IL-12, and IFN-γ; p > 0.05) compared to HCs. We have reported this for the full cohort, but as this is a subset, we are reporting the new values (Patlola et al., Reference Patlola, Holleran, Dauvermann, Rokita, Laighneach, Hallahan, McManus, Kenyon, McDonald, Morris, Kelly, Donohoe and McKernan2025). Only TLR2, TLR4, and IL-8 survived the Bonferroni multiple-test correction.
TLR2 and 4 activity and cytokine data

Note: P values in this table are based on a non-parametric (Mann–Whitney test) t-test between HCs and patients. TLR, Toll-like receptor; IL, interleukin; TNF, tumor necrosis factor; IFN, interferon; pg/ml, picograms per milli-liter; N, sample size; Std. Dev., standard deviation; Bold, significant.
Antipsychotic drugs/OLZ equivalent doses and other medications
After calculating the OLZ equivalents, the participants were categorized based on the number of antipsychotic drugs they were taking at the time of study (Supplementary Table S2). Fifty-four patients are taking one antipsychotic drug, and the rest are on multiple antipsychotic drugs (22 SZ – 2 antipsychotic drugs; 3 SZ – 3 antipsychotic drugs). Patients with no antipsychotic drugs or incomplete information were excluded from further analysis. Other medications acting on the central nervous system (CNS) and non-CNS drugs were categorized separately. Only 25 HCs were on some form of medication or supplements. The medication details for both patients and controls are illustrated in Figure 1.
Breakdown of all the medication taken by patients. (a) Doughnut chart; N = 79 and controls. (b) Pie chart; N = 25. The number below the drug(s) indicates the number of participants taking that medication at the time of the study. CNS, central nervous system; ACE, acetylcholinesterase.

Antipsychotic drug dose does not predict the decline in cognition
Multiple linear regression was performed on the whole population using antipsychotic drug dose as the independent variable and cognition as the dependent variable, controlling for age, sex, BMI, full-scale IQ, and duration of illness (Table 3). This analysis showed that antipsychotic drug dose does not have any significant association with cognition in our cohort. An increase in duration of illness, age, and BMI (in some cases) significantly predicted a decline in cognition (p < 0.05) when the remaining variables were kept constant. Additionally, a greater IQ was associated with increased cognitive performance when the rest of the variables were kept constant. Model summary and specifics are detailed in Supplementary Doc 1.
Multiple linear regression of cognition, TLR activity, and cytokines

Note: Sample size variations in the data reflect the missing data either in the dependent or independent variables or both. FSIQ, full-scale intelligence quotient; PS, processing speed; VLM, verbal learning and memory; VisLM, visual learning and memory; WM, working memory; SC, social cognition; TLR, Toll-like receptors; IL, interleukin; BMI, body mass index; p, p-value; Bold, significant relationships.
Antipsychotic drug dose does not predict changes in TLR activity or cytokines
Multiple linear regression was performed in the whole population using TLR activity and IL-8 (only cytokine to survive Bonferroni correction) (Table 3) as dependent variables and antipsychotic drug dose as an independent variable, factoring in age, sex, duration of illness, and BMI. We found that antipsychotic drug dose could not predict changes in TLR2/TLR4 activity and IL-8 levels in the participants. However, an increase in IQ was associated with a decrease in plasma IL-8 levels. Model summary and specifics are detailed in Supplementary Doc 1.
Antipsychotic drug doses moderate the relationship between TLR4 activity and cognition
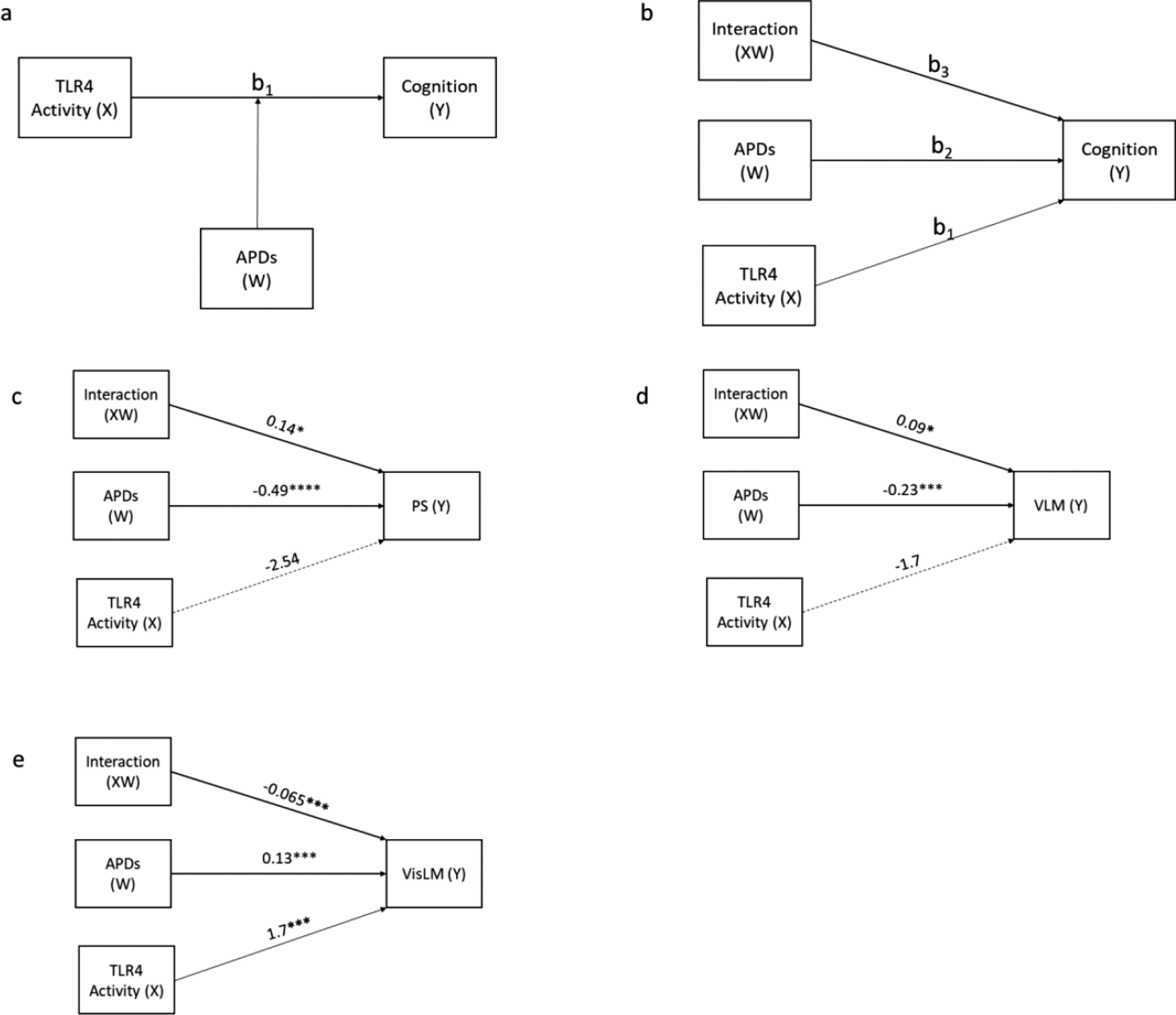
We had previously investigated and established the mediating effects of cytokines between TLR activity and cognition in the same cohort (Patlola et al., Reference Patlola, Holleran, Dauvermann, Rokita, Laighneach, Hallahan, McManus, Kenyon, McDonald, Morris, Kelly, Donohoe and McKernan2025). Therefore, in the current study, we investigated whether the antipsychotic drug dose moderates (Figure 2) the relationship between immune response (inflammation) and cognition. Therefore, we investigated the moderating effect of antipsychotic drug doses on the relationship between TLR activity and cognition. The results indicate that there is no moderating effect of antipsychotic drug dose on the relationship between TLR2 activity and any of the six cognition domains. The detailed moderation analysis data and model statistics are present in Supplementary Doc 2.
Moderation ‘Model 1’ illustrates (a) the general model and (b) the statistical model. The moderating effect of antipsychotic drug dose (APD) on the relationship between TLR4 activity and (c) processing speed (PS), (d) verbal learning and memory, and (e) visual learning and memory. TLR, Toll-like receptor; antipsychotic drug, antipsychotic drug dose; X, independent variable; Y, dependent variable; W, – moderator; path b1, direct effect of TLR4 activity on cognition scores; path b2, direct effect of antipsychotic drug dose on activity on cognition scores; path b3, interaction term, effect of TLR4 activity on cognition when antipsychotic drug dose is factored. P > 0.05, not significant; *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001.

However, in the case of TLR4 activity, we observed three models that showed significant associations. The results indicate that an increase in the antipsychotic drug dose strengthens the relationship between TLR4 activity and PS (B = 0.14; p < 0.05) (Figure 2c) and TLR4 activity and verbal learning and memory (VLM) (B = 0.09; p < 0.05) (Figure 2d). However, post hoc analysis shows that the conditional effects (Supplementary Figures S1 and S2) at low, moderate, and high doses are not significant, indicating that although the moderation effect is statistically significant, the effect of TLR4 activity on either PS or VLM is weak to negligible. Interestingly, we found that the antipsychotic drug dose significantly weakens the relationship between TLR4 activity and visual learning and memory (VisLM) (Figure 2e). Further post hoc analysis (conditional effects) (Supplementary Figure S3) revealed that, at low/no doses of antipsychotic drugs, an increase in TLR4 activity leads to increased scores (PAL error scores) in the VisLM domain, indicating greater cognitive deficits. The strength of this relationship decreases with an increase in dose, and finally, at high doses, the moderating effect becomes negligible.
Bonferroni multiple test correction p-value is set at p < 0.0083 for these analyses (0.02/6). TLR4/VisLM is the only model that survived the correction. All of the abovementioned models with TLR2, TLR4 activity, and cognition are not significant when covaried for age, sex, BMI, and duration of illness.
Discussion
The effect of antipsychotic drugs on cognition has been studied for more than two decades (Baldez et al., Reference Baldez, Biazus, Rabelo-da-Ponte, Nogaro, Martins, Kunz and Czepielewski2021; Keefe, Silva, Perkins, & Lieberman, Reference Keefe, Silva, Perkins and Lieberman1999). Evidence suggests that antipsychotic drugs provide symptom alleviation to some extent and improve cognitive deficits (Haddad et al., Reference Haddad, Salameh, Sacre, Clément and Calvet2023; Hou et al., Reference Hou, Xie, Yuan, Cheng, Han, Yang, Yu and Shi2020) in specific domains (case-dependent). A recent network meta-analysis indicates that individual antipsychotic drugs may not have a better outcome compared to the placebo group, but in combination with other antipsychotics show a better outcome on cognition (Feber et al., Reference Feber, Peter, Chiocchia, Schneider-Thoma, Siafis, Bighelli, Hansen, Lin, Prates-Baldez, Salanti, Keefe, Engel and Leucht2024). However, the exact mechanism by which they impact cognition is not clearly understood. In previous meta-analyses, we showed the suppressive effect of antipsychotic drugs on inflammatory cytokines (S. R. Patlola et al., Reference Patlola, Donohoe and McKernan2023a) and also the detrimental effects of elevated cytokines on cognition (Patlola, Donohoe, & McKernan, Reference Patlola, Donohoe and McKernan2023b). We have also recently determined that TLR activity may influence cognitive deficits in patients (Patlola et al., Reference Patlola, Holleran, Dauvermann, Rokita, Laighneach, Hallahan, McManus, Kenyon, McDonald, Morris, Kelly, Donohoe and McKernan2025). We, therefore, investigated the effect of the antipsychotic drug dose on cognition and the immune system. To the best of our knowledge, this is the first study investigating whether antipsychotic drugs moderate the relationship between TLR activity and cognition.
We first investigated whether antipsychotic drug dose predicted cognitive function. Interestingly, we found no association between antipsychotic drug doses and cognition. Our results are in contrast to studies in the literature showing improved cognition in animals (MacDowell et al., Reference MacDowell, Munarriz-Cuezva, Caso, Madrigal, Zabala, Meana, García-Bueno and Leza2017; Neill et al., Reference Neill, Grayson, Kiss, Gyertyán, Ferguson and Adham2016) and humans (Noh et al., Reference Noh, Na, Park, Kim, Evins and Roh2020) post-antipsychotic drug administration. Moreover, a past meta-analysis of 35 studies (SZ patients) (Nielsen et al., Reference Nielsen, Levander, Kjaersdam Telléus, Jensen, Østergaard Christensen and Leucht2015) indicated that clozapine and OLZ use resulted in a decline in verbal working memory in individuals with SZ. The study also showed that clozapine and OLZ improved verbal fluency. Furthermore, it was observed that first-generation antipsychotics and clozapine may have detrimental effects on cognition (Feber et al., Reference Feber, Peter, Chiocchia, Schneider-Thoma, Siafis, Bighelli, Hansen, Lin, Prates-Baldez, Salanti, Keefe, Engel and Leucht2024). In our cohort, 41.8% and 22.8% of patients are on clozapine and OLZ, respectively; nearly one-third of the patients (31.7%) are on more than a single antipsychotic drug. This makes it difficult to assess the effect of each drug independently in our cohort. A newer study reports a decline in cognitive function in patients with SZ, considering the cumulative lifetime antipsychotic drug use by each patient (Husa et al., Reference Husa, Moilanen, Murray, Marttila, Haapea, Rannikko, Barnett, Jones, Isohanni, Remes, Koponen, Miettunen and Jääskeläinen2017). Furthermore, studies suggest that reducing the antipsychotic drug dose improves cognition in SZ (Kawai et al., Reference Kawai, Yamakawa, Baba, Nemoto, Tachikawa, Hori, Asada and Iidaka2006; Singh et al., Reference Singh, Kumar, Pathak, Jacob, Venkatasubramanian, Varambally and Rao2022).
Next, we investigated whether antipsychotic drugs affect inflammatory cytokines and TLR activity. In our previous meta-analysis, we reported that antipsychotic drugs, specifically risperidone administration, reduced plasma cytokine levels (S. R. Patlola et al., Reference Patlola, Donohoe and McKernan2023a). However, we did not find such evidence in plasma cytokines in this current study. In our study, there were only 12.7% patients on risperidone. Unlike all the studies included in the meta-analysis, our study design did not investigate the effects of antipsychotic drugs before and after administration; instead, we investigated whether the OLZ equivalent doses of antipsychotic drugs could help predict the circulating cytokine levels. This could be the reason for such contrasting findings.
Additionally, we report that antipsychotic drug dose could not predict TLR activity. Both TLR2 and TLR4 receptors play a vital role in the innate immune system. They belong to a family of pattern recognition receptors and can recognize molecular patterns, including those present in LPSs from Gram-negative bacteria and some polymers from Gram-positive bacteria, as well as damage-associated molecular patterns, such as hyaluronan, fibronectin, and other endogenous ligands released during cell damage or stress, further promoting cytokine production (Molteni, Gemma, & Rossetti, Reference Molteni, Gemma and Rossetti2016). Moreover, in animals, stimulation of TLR4 has been associated with cognitive decline (Li et al., Reference Li, Chen, Gou, Li, Tong, Zhou, Xie, Yu, Feng, Li, Chen, Tian, Tan, Wang, Pan, Li, Luo, Zhang, Huang and Tan2022), and knocking out this receptor improved specific cognitive domains (Connolly, Potter, Sexton, & Kohman, Reference Connolly, Potter, Sexton and Kohman2021; Fei et al., Reference Fei, Y-n, Lv, Ding, Wei, Wu, He, Fei and Fei2022). Hence, we were interested in investigating whether increased TLR activity negatively impacted cognition (which we have previously shown as a mediation analysis with both TLR2 and TLR4 – (Patlola et al., Reference Patlola, Holleran, Dauvermann, Rokita, Laighneach, Hallahan, McManus, Kenyon, McDonald, Morris, Kelly, Donohoe and McKernan2025) and whether the antipsychotic drug dose moderated this relationship.
Therefore, we performed a moderation analysis, and we observed that antipsychotic drug dose negatively affected cognition. Most importantly, we did not observe any moderation effect of the antipsychotic drug dose, except for the case of TLR4/VisLM (Figure 2e), which indicates that the negative effect of TLR4 activity on cognition is dependent on the dosage levels of antipsychotic drugs. Similar findings were reported in the past in an outpatient SZ group, but with IL-6 instead of TLR4 activity (Ribeiro-Santos et al., Reference Ribeiro-Santos, de Campos-Carli, Ferretjans, Teixeira-Carvalho, Martins-Filho, Teixeira and Salgado2020). They observed that the association between higher serum IL-6 and cognitive decline depended on the antipsychotic drug dose. In the current study, post hoc analysis indicated that at low or no antipsychotic doses, an increase in TLR4 activity was associated with a decline in VisLM (Supplementary Figure S3); at moderate doses, this association weakened but remained significant. However, at higher doses, this relationship weakened further and was not significant. In the future, this trend is worth investigating with larger longitudinal cohorts.
While the findings reported are novel, this study also has limitations. First, this is a cross-sectional study; therefore, we could not capture the effects of the antipsychotic drugs over time, which would have been valuable to understanding the effect of antipsychotic drugs on the immune system and cognition as the illness progressed. Moreover, the sample size variation between patients (N = 79) and controls (N = 201) is considerable. Therefore, results need to be carefully interpreted. We incorporated bootstrapping into the analysis to increase the confidence in the results. Second, as the patient population was on a variety of antipsychotic drugs, such as clozapine, OLZ, aripiprazole, and other antipsychotic drugs, investigating individual antipsychotic drug effects would not be possible due to the low sample size. In addition to antipsychotic drugs, some patients were also on other CNS drugs, such as antidepressants and mood stabilizers that could impact the immune system and cognition. This factor needs to be acknowledged because ACB has been demonstrated to impact cognition; therefore, a separate moderated-moderation analysis using ACB scores was performed (Supplementary Doc 3). The results indicate that only at high doses of antipsychotic drugs, an increase in TLR4 activity led to improvement in the VLM domain. This was applicable for low, moderate, and high ACB scores. However, the limitation of this is that ACB scores are qualitative and do not account for the dose of the drug. Therefore, a better score or measure needs to be created to account for this.
Third, nearly 32% of our patient cohort (25) was on more than one antipsychotic drug. This is important because despite being all second-generation drugs, they have different receptor affinities and side effects. Feber and colleagues have also suggested that the varying effects of antipsychotic drugs on cognition are due to their different receptor-binding profiles. Their findings also suggest that antipsychotic drugs’ effects on cognition are drug-specific (Feber et al., Reference Feber, Peter, Chiocchia, Schneider-Thoma, Siafis, Bighelli, Hansen, Lin, Prates-Baldez, Salanti, Keefe, Engel and Leucht2024). Variations in pharmacodynamics are difficult to integrate into the analysis. However, it was partly managed by converting them all into OLZ equivalents. Hence, despite the small sample sizes (within-group), we tested for differences between individuals taking one (54) and more than one antipsychotic drugs (25). The results showed that patients on more than one antipsychotic drug presented higher BMI, plasma CRP levels, more significant depressive symptoms, and poorer PS (Supplementary Doc 2) compared to patients on a single antipsychotic drug. However, it is but a trend as they do not survive Bonferroni multiple test correction. This could perhaps be because people on higher dosages or a greater number of antipsychotic drugs to begin with had a more severe illness with greater levels of symptomatology that had been unresponsive to treatment (Gallego et al., Reference Gallego, Nielsen, De Hert, Kane and Correll2012).
Lastly, we did not include the participants’ lifetime cannabis or tobacco exposure in our analysis. Evidence indicates that they both have a modulating effect on TLR activity (Cui Sun, Otálora-Alcaraz, Prenderville, & Downer, Reference Cui Sun, Otálora-Alcaraz, Prenderville and Downer2024; Semlali, Witoled, Alanazi, & Rouabhia, Reference Semlali, Witoled, Alanazi and Rouabhia2012). In our cohort, based on a self-reporting questionnaire, 80.4% of controls and 87.6% of patients never consumed cannabis in their lifetime or in the last 12 months. Similarly, 74.9% of controls and 60.5% of patients never consumed tobacco in their lifetime. The data we have are qualitative in nature, and the reliability of the quantity of cannabis and tobacco consumed from the questionnaire could be questionable, as the forms of consumption are different and measuring the exact dose for every consumption is not possible.
In conclusion, there was no evidence in this participant cohort that antipsychotic drugs moderated the impact of the immune system on cognition. However, we did find that the antipsychotic drug dose is significantly associated with cognitive decline and elevated TLR4 activity.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0033291726103596.
Data availability statement
The data used in this study are available upon reasonable request.
Acknowledgments
The authors sincerely thank all participants who took part in the study. The authors would also like to thank past and present iRELATE team members and Dr. Daniel Kerr for their assistance with the project.
Funding statement
This work was funded through the Hardiman Scholarship, University of Galway (to SRP), and a grant from the European Research Council (ERC-2015-STG-677467) to GD.
Competing interests
The authors declare none.