Overview of RWE in reimbursement
While randomized controlled trials (RCTs) are the gold standard for assessing efficacy and safety of treatments in regulatory decision-making, they are limited in their ability to provide evidence of an intervention’s value in a real-world context (Reference Monti, Grosso, Todoerti and Caporali1). Real-world evidence (RWE), generally defined as observational evidence derived outside RCTs, can fill data gaps and inform reimbursement decisions (2;Reference Tadrous, Ahuja, Ghosh and Kropp3). Canada aims to be one of the first jurisdictions to implement RWE to support RCTs in informing health technology assessment (HTA)/reimbursement (4). In 2023, the Canadian Drug Agency–L’Agence des médicaments du Canada (CDA-AMC) partnered with Health Canada and Institut national d’excellence en santé et services sociaux to create the Guidance for Reporting Real-World Evidence, which promotes RWE standardization by providing core conduct and reporting guidance for RWE studies being submitted for reimbursement (5).
However, uncertainty remains around when RWE generation should be prioritized and conducted for reimbursement. While many health technologies are coming to market, not all will benefit equally from RWE to optimize patient access. RWE studies are time-consuming and require considerable resources. Therefore, judicious RWE generation planning is essential during asset development to ensure that once approved, access is optimized to meet patient and health-system needs.
This article outlines our efforts to determine how and when RWE is used for reimbursement. We did this by identifying past CDA-AMC reimbursement reviews that included RWE to understand how RWE has been considered in past Canadian HTA. We also collected insights from key Canadian, regional, and global stakeholders, including pharmaceutical industry representatives, payers, and patient advocates, on when RWE generation should be prioritized for reimbursement. We integrated our findings to identify common themes around when it is impactful to generate RWE for reimbursement and barriers/facilitators to generating these data. Details on methodology are presented in the Supplementary Methods.
Use of RWE in Canadian reimbursement reviews
We reviewed 379 CDA-AMC reimbursement submissions from 2017 to 2022 and found that RWE was included in approximately one-third of submissions (n = 145). No clear trends were identified in the inclusion of RWE over the 6 years. The most common therapeutic areas for RWE inclusion were oncology (50 percent), genetic disorders (10 percent), and neurology (9 percent). Forty-three percent of reviews for rare diseases included RWE.
We conducted a detailed data extraction on a random sample of 76 reviews with RWE to assess the RWE’s characteristics, purpose, and impact (i.e., whether it was discussed in any level of detail in the report) and influence (i.e., whether it was mentioned in the final recommendation report, regardless of the decision). RWE was most often generated to fill gaps in economic model inputs (27 percent), comparative efficacy evidence (18 percent), efficacy and safety data (14 percent), and to assess the validity of outcome measures (8 percent).
We found large variability in the impact of RWE on decision-making based on a preliminary analysis of 11 reimbursement reviews from 2022. There was a lack of correlation between whether the CDA-AMC committee discussed the RWE beyond simply including the evidence in the report (e.g., highlighting limitations in methodology and raising concerns about generalizability to the Canadian population) and if the RWE was deemed influential on the final recommendation. While the lack of correlation prevents making robust conclusions on the role of RWE in influencing decisions, the inclusion of even low-quality RWE (e.g., small sample size and risk of bias or confounding due to the study design) suggests that these data may still influence decision-making if they represent the best available evidence.
Several analyses of HTA reimbursement reviews containing RWE have been published (Reference Lau and Dranitsaris6–Reference Jaksa, Louder and Maksymiuk10). However, compared to our analysis, most of these studies had a more focused scope (e.g., oncology drugs only, use of a specific database, and use of an external control arm) or aimed to answer a different research question.
Stakeholder insights on the prioritization of RWE for reimbursement
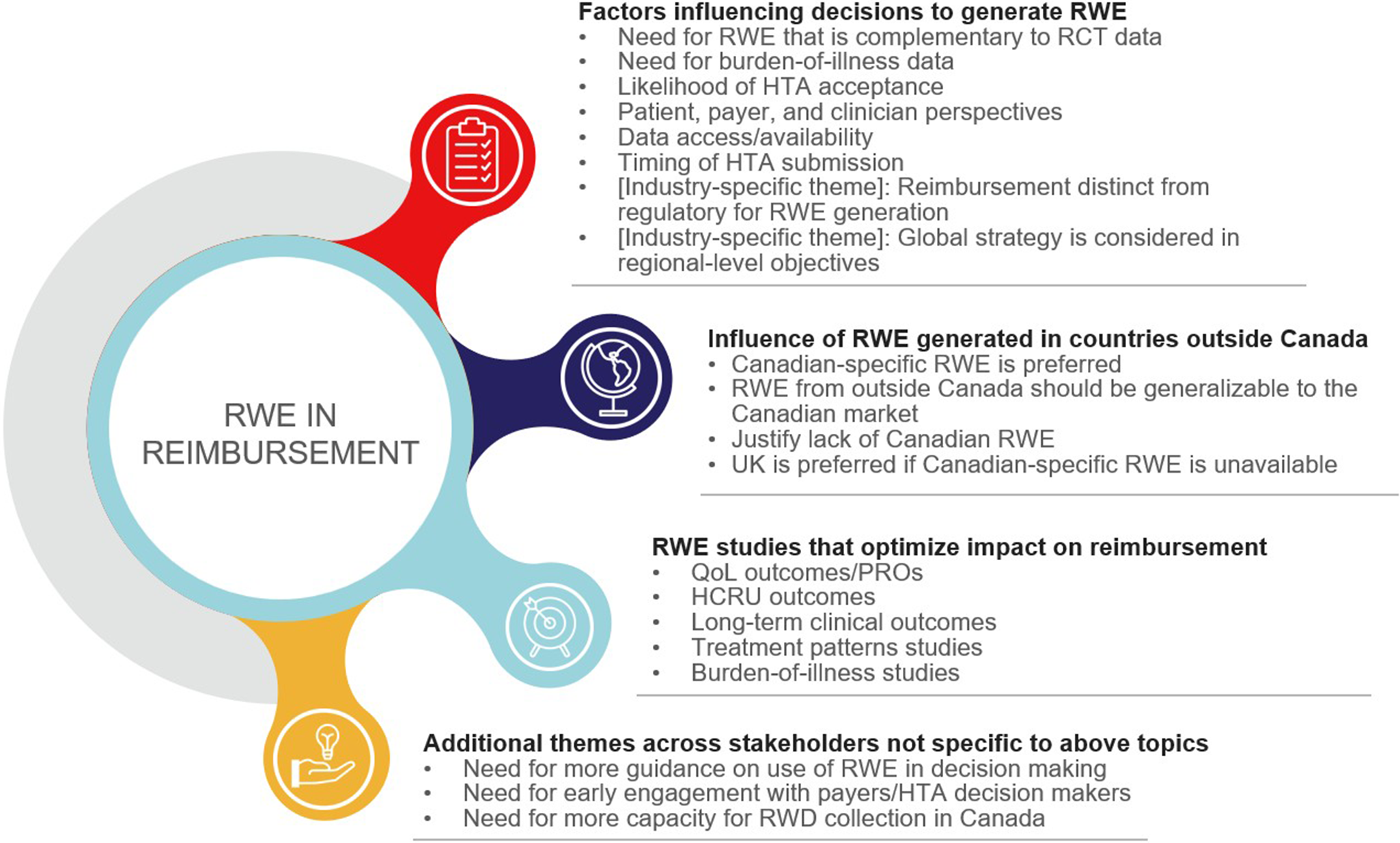
We next sought to draw upon the expertise of different stakeholder groups to identify key learnings and current practices around generating and prioritizing RWE studies for reimbursement in Canada and globally. We conducted 1-1 interviews with a total of 13 stakeholders; 3 prespecified topics were discussed: (a) what factors influence decisions to conduct RWE studies, (b) when RWE generated in other countries influences this decision, and (c) when RWE can optimize reimbursement. Based on a thematic analysis of the interviews, the stakeholders agreed on several themes related to each prespecified topic, as well as additional themes that emerged during the interviews (Figure 1; Table 1).
Key themes from stakeholder insights by topic. Abbreviations: HCRU, healthcare resource utilization; HTA, health technology assessment; PRO, patient-reported outcome; QoL, quality of life; RCT, randomized controlled trial; RWD, real-world data; RWE, real-world evidence.

Key themes from stakeholder insights by topic, with theme descriptions and representative stakeholder quotes

Abbreviations: CDA-AMC, Canadian Drug Agency–L’Agence des médicaments du Canada; HCRU, healthcare resource utilization; HTA, health technology assessment; PRO, patient-reported outcome; QoL, quality of life; RCT, randomized controlled trial; RWD, real-world data; RWE, real-world evidence; UK = United Kingdom.
All stakeholders were aligned on the value RWE can provide as complementary evidence to support RCT data, to fill evidence gaps left by RCTs, or to validate RCT results in larger and/or different populations; the theme “need for RWE that is complementary to RCT data” had high concordance (defined as ≥8 stakeholders) in our analysis.
There was general agreement among all stakeholders that certain types of RWE studies are usually more important for reimbursement purposes, specifically those characterizing patient-reported outcomes, current treatment patterns, healthcare resource utilization, and long-term clinical outcomes (Figure 1). RWE can be used to reduce uncertainty around these topics by establishing existing disease burden and addressing evidence gaps from RCTs.
For reimbursement in Canada, stakeholders agreed that Canadian data is preferred, but they believe data availability limits the feasibility of generating such evidence (the theme “data access/availability” had high concordance). This belief echoes an analysis of stakeholder perspectives in Canada, which identified inadequacies in real-world data (RWD) infrastructure and access as the greatest barrier to using RWE for decision-making for cancer drugs (Reference Clausen, Mighton and Kiflen11). Canadian stakeholders in our analysis stated that when not possible, justification should be provided for the use of data from outside of Canada, including generalizability to the Canadian population (5). From our assessment of CDA-AMC reimbursement submissions, manufacturers rely heavily on RWD from other countries, with more than half of reviews using RWE from other regions (n = 24 out of 43 reviews that reported the region[s] for all included RWE studies). Other regions frequently represented included the United States, United Kingdom, and France, which aligned with Canadian stakeholders’ insights, indicating the importance of similarities in healthcare systems. Canadian patient advocate stakeholders noted that for rare diseases, consolidating data from other markets, and particularly the United States, may be a reasonable approach to obtaining RWD rather than focusing only on Canadian data. Stakeholders with global experience also cited a preference for local data, followed by data that are generalizable to the region of interest.
Industry stakeholders considered the likely acceptability by HTA bodies and payers when making decisions about RWE generation and noted this may vary by region. Trends in RWE acceptance in Europe were explored in a recent survey, which found that European Network for Health Technology Assessment member HTA organizations were more likely to accept RWE for orphan drugs and when there is a companion diagnostic, and were least likely to accept non-local RWE, although the impact on decision-making was not assessed (Reference Hogervorst, Pontén, Vreman, Mantel-Teeuwisse and Goettsch12). As we found in our analysis, some stakeholders highlighted that early engagement with HTA bodies and payers can identify data gaps that RWE can fill and opportunities to optimize RWE study design. Payer stakeholders emphasized that RWE should be robust and generated in studies conducted with high methodological rigor.
Incorporation of the patient perspective in RWE was deemed important by Canadian patient advocate stakeholders as they believe these data provide valuable insight into the patient and caregiver experience – insight that until recently had not been routinely considered in reimbursement decision-making. However, they emphasized that low patient involvement is a challenge and suggested providing patient education around the benefit of RWE study participation and involving patients in early RWE generation planning to encourage engagement. Involving patients in initiatives to support RWE incorporation in cancer drug HTA in Canada was previously recommended by stakeholders in a 2020 study (Reference Clausen, Mighton and Kiflen11).
Our thematic analysis highlighted that the majority of stakeholders recognized that the value of RWE lies in its ability to complement RCT data, and that RWE is typically not sufficient to support reimbursement on its own. Furthermore, most stakeholders discussed the need to consider feasibility, as well as the perspectives of different stakeholders to optimize RWE for reimbursement.
Limitations
A key limitation of the analysis of CDA-AMC reimbursement reviews was variability in how RWE was described in the CDA-AMC reports, which may have led to undercounting the reviews with RWE if not clearly documented. Detailed analysis was conducted only on a small random sample of CDA-AMC reviews, and the resulting data may not be representative of larger datasets. In addition, our study only used RWE information included in the CDA-AMC reports and did not assess the cited RWE studies in depth.
The stakeholder insights research was a small sample. Additionally, due to fewer patient advocates and payer stakeholders, results may be biased toward the industry perspective. Our study was designed to elicit preliminary insights from a sample of stakeholders. As such, the insights presented are high-level and may not be representative of all stakeholders of a given type or region.
Gaps and potential solutions
Key gaps identified in our analysis include a lack of guidance on the practical implications of RWE in reimbursement decision-making, as well as challenges in RWD availability, access, and collection in Canada. The recent guidance published by CDA-AMC encouraging robust design of RWE studies, and the general preference of HTA agencies for high-quality data, may contribute to greater acceptability of RWE in Canada; however, these guidelines – as well as recent global RWE guidelines – are heavily focused on methodology (5). Developing a methodologically robust, practical framework that could be tailored to different regions, helps stakeholders prioritize when to generate RWE for reimbursement, and incorporates stakeholder perspectives would provide value. Given that HTA bodies are increasingly including non-payer stakeholders in decision-making, earlier involvement of such stakeholders in RWE planning could better inform manufacturers and facilitate the inclusion of valuable perspectives. Additionally, greater engagement in scientific advice programs offered by HTA agencies could increase the impact of RWE (13–16).
Although Canadian-specific data would be preferred for generalizability of RWE, access to and collection of Canadian data vary across provincial/territorial jurisdictions, making it difficult for manufacturers to use for RWE. This suggests a need for cross-country collaboration among data managers to build and maintain pan-Canadian RWD sources (Reference Facey, Rannanheimo, Batchelor, Borchardt and de Cock17). Several recent initiatives have launched within Canada (such as the Pan-Canadian Real-World Health Data Network) (Reference Smith, McGrail and Schull18–Reference Dai, de Oliveira and Blommaert20), all of which focus on consolidating available real-world datasets in a central database for easy accessibility. Such offerings could increase RWE generation and have valuable downstream uses in reimbursement decision-making. Improvements in RWE generation capabilities would improve data quality and, thus, enable RWE to be more influential in HTA decisions.
Key takeaways
Taken together, our analysis of CDA-AMC reimbursement reviews indicates that while RWE was included in some submissions, presumably representing the best available evidence in those cases, it was not commonly used in Canadian HTA during the period of analysis. Only approximately one-third of submissions included in our analysis included RWE, indicating there may be an opportunity for greater utilization of RWE in reimbursement decision-making in Canada.
Our research highlights important areas to consider when prioritizing RWE studies for reimbursement. RWE should primarily be used as complementary evidence to support, validate, or fill gaps left by RCTs. Current guidance provided globally by HTA bodies and regulatory agencies focuses on RWE methodology, but there is a need for further practical guidance, such as a robust decision framework to support optimized RWE generation for reimbursement.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0266462325103401.
Acknowledgments
The authors would like to thank Christie Blaskovich, PharmD, for contributing to the analysis of the CDA-AMC reimbursement reviews.
Funding statement
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
All authors are employed by PPD Evidera Health Economics & Market Access, Thermo Fisher Scientific – a healthcare research firm that provides consulting and other research services to pharmaceutical, device, government, and non-government organizations. In this salaried position, each author works with a variety of companies and is explicitly precluded from accepting any payment or honoraria directly from them for services rendered.