Introduction
Preterm infants, because of their immaturity, are prone to serious infections. Since premature infants spend long stays in hospitals and often require invasive procedures, they are at a particularly high risk of healthcare-associated infections (HAI).Reference Shane, Sanchez and Stoll1 Birth weight and gestational age as well as previous placement of a peripheral or central venous catheter (CVC) have been identified as independent risk factors for healthcare-associated sepsis (HAS).Reference Geffers, Gastmeier, Schwab, Groneberg, Ruden and Gastmeier2,Reference Schwab, Geffers, Barwolff, Ruden and Gastmeier3 The implementation of a local surveillance system whose reference information for benchmarking allowed timely adjustment of locally implemented infection prevention and control (IPC) measures led to a decrease in HAS incidence density rates of 24% (from 8.3 to 6.4) over a three-year period.Reference Schwab, Geffers, Barwolff, Ruden and Gastmeier3 In light of this, it is surprising that in many countries continuous nationwide surveillance of HAI among high-risk neonates is not routine practice and that knowledge of the contribution of long-term surveillance to the prevention of HAI in neonates is still limited. In Germany, though, participation in such a surveillance system has been mandatory for neonatal units seeking classification as high-level (level 1 or level 2) perinatal centers since 2006, and is therefore indirectly linked to financial reimbursement.4 This has encouraged the participation of neonatal units in a surveillance system. Because NEO-KISS has in essence been the only established surveillance system in Germany, data has been reported to it nationwide by almost all neonatology departments in Germany. The aim of this paper is to describe the development of HAI rates among high-risk neonates since nationwide implementation of NEO-KISS and to present risk factors in the development of HAI.
Methods
Data collection and study population
NEO-KISS is a prospective, patient-based surveillance system for HAI and device use in infants with a birth weight of less than 1500 g. The program was launched in Germany in 2000 by the National Reference Centre for Surveillance of Nosocomial Infections (NRZ). Its focus is HAS, healthcare-associated pneumonia (HAP), and necrotizing enterocolitis (NEC) using modified Centers for Disease Control and Prevention (CDC) definitions.Reference Gastmeier, Geffers, Schwab, Fitzner, Obladen and Ruden5–Reference Hentschel, de Veer, Gastmeier, Ruden and Obladen8 In this context, the term HAI is used throughout the manuscript as an umbrella term encompassing all conditions captured by the NEO-KISS surveillance system, even if NEC is not uniformly attributable to an infectious cause.Reference Gordon, Swanson, Attridge and Clark9 Upon enrollment in the program, neonatology departments indicate their level of care as defined by the German joint federal committee. This definition distinguishes four such levels, with level one corresponding to the highest and level four to the lowest. The first two levels include maintenance of a neonatal intensive care unit (NICU).10 Case definitions of HAI in NEO-KISS are described in detail in the current NEO-KISS protocols developed by the NRZ.Reference Gastmeier, Geffers, Schwab, Fitzner, Obladen and Ruden5,11 It specifies a HAI be recorded in the database if symptom-onset occurs more than 72 hours after birth/admission. It is considered central venous catheter-associated HAS (CVC-S) or invasive mechanical ventilation-associated HAP (VAP) if the device (CVC, including an umbilical catheter, or a tube) was in place for at least three consecutive calendar days (regardless of the time of placement) either on the day of the first onset of symptoms or on the day before onset of symptoms. Subtypes of HAS are (i) laboratory-confirmed bloodstream infection (BSI) with a blood (or cerebrospinal fluid) culture positive for a recognized pathogen other than coagulase-negative staphylococci (non-CoNS BSI), (ii) BSI with a blood culture positive for CoNS (CoNS BSI), and (iii) clinical HAS without cultured pathogens (clinical HAS). All three subtypes require the presence of at least two clinical symptoms (temperature >38 °C, <36.5 °C, or unstable; tachycardia or bradycardia; apnea; extended recapillarization time; metabolic acidosis; hyperglycemia; or other signs of sepsis) without signs of infection at another site. In addition, a recording of CoNS BSI requires at least one laboratory finding (increased values for C-reactive protein, for interleukin, or for immature and total neutrophil ratio; decreased values for platelets or white blood cells); a recording of clinical HAS requires at least five days of adequate, anti-infective therapy. Overall, the NEO-KISS surveillance system allows reporting of the following categories of HAS: Total HAS events with subtypes CoNS BSI, non-CoNS BSI and clinical HAS; total CVC-S events (as a subset of HAS events) with subtypes CVC-associated CoNS BSI (CVC-CoNS BSI), CVC-associated non-CoNS-BSI (CVC-non-CoNS BSI), and clinical CVC-S.
Criteria for HAP and NEC are defined in the NEO-KISS protocols11 and described in Supplementary Table S1. Briefly, diagnosis of HAP requires one radiological finding, deterioration in saturation or gas exchange and at least four clinical criteria. NEC is recorded if at least one radiological sign and two clinical signs are present, or a histological report. Recording of more than one HAI of the same HAI-type per infant is possible if the new episode occurs after an interval of 14 days and if there was an interval that was symptom-free. All neonates admitted to a participating neonatology department with a birth weight below 1500 g are followed up in NEO-KISS until they reach a weight of 1800 g, with the exception of cases which drop out of surveillance because they have been discharged, transferred to another hospital/department, or have died. Key methodological specifications and definitions of NEO-KISS are summarized in Supplementary Table S1 and compared with current CDC/NHSN definitions.
Here, we present an analysis of all infants who were admitted between January 01, 2008, and December 31, 2022, enrolled in NEO-KISS, and subsequently followed up. We did not include data in our analysis from 2006 and 2007, that is two years after financial reimbursement was in place, in order to give new centers an opportunity to successfully navigate the implementation process involved. Primary outcome parameters are device utilization (device days per 100 patient days) and HAI (total number and incidence densities per either 1,000 patient days or 1,000 device days). Secondary outcome was mortality under surveillance (per 100 patient days).
Ethical approval and individual informed consent are not required and institutional review boards were not consulted, firstly because surveillance of HAI and the associated data collection by hospitals is legally mandated by the German Protection Against Infection Act (Infektionsschutzgesetz §23)12; and secondly because our analysis and publication of aggregated information for the purpose of improving infection prevention and control is one of our responsibilities, as defined by Robert Koch Institute, in our role as the NRZ for the surveillance of nosocomial infections in Germany on behalf of the Federal Ministry of Health.13
Statistical analysis
Multivariable regression models were used to assess the effect of the development of HAI over time. Three five-year periods of admission (2008–2012, 2013–2017, and 2018–2022) were set up to correspond with the reference period intervals. Cox-proportional hazard regression models with cluster effects were applied which adequately considered the time to event and clusters in a department to calculate hazard ratios (HR) for the first HAS, HAP and NEC of each case.
Each type of HAI (HAS, HAP, NEC) and each subtype of HAS (non-CoNS BSI, CoNS BSI, clinical HAS), including CVC-S and VAP, were analyzed separately. We calculated crude and adjusted hazard ratios with 95% confidence intervals for the period of admission. Separate models were calculated for each type of HAI using variable selection stepwise backward, starting with the full adjusted model. P < .05 was considered statistically significant. Data was analyzed using SAS version 9.4 and IBM SPSS Statistics version 29. Figures were created in Microsoft Excel 2019.
Results
Overall, 251 neonatology departments participated in NEO-KISS during the period January 2008 to December 2022. Of these, the majority were certified as perinatal centers level 1 (n = 163, 64.9%) or level 2 (n = 67, 26.7%). In the period between 2008 and 2022, the median length of NEO-KISS participation among departments was 15 years (interquartile range (IQR): 11–15). The median number of patients per department was 414 (IQR: 117–695), with a median of 14,244 patient days (IQR: 2,708–25,214) observed (Supplementary Table S2).
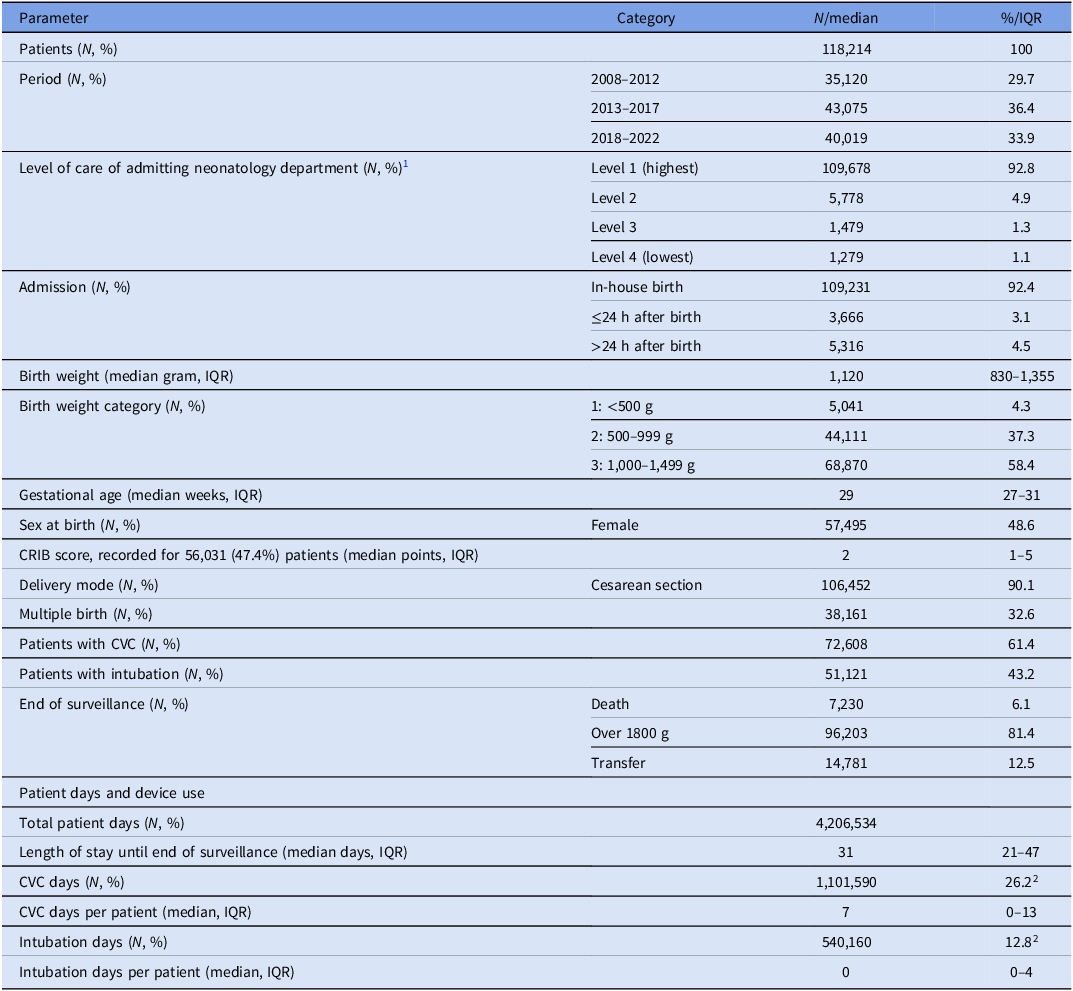
A total of 118,214 infants were admitted during the three surveillance periods, comprising 4,206,534 patient days. Almost all infants were admitted to a perinatal center level 1 (92.8%) or level 2 (4.9%). The majority of births were in hospital (92.4%) and by Cesarean section (90.1%). The median birth weight was 1120g (IQR: 830–1355g), with 5,041 (4.3%) neonates weighing less than 500g. The median gestational age was 29 weeks (IQR: 27–31). The distribution of gestational age and birth weight per five-year period is shown in Figure 1A–D.
Distribution of gestational age (A: frequency, B: percentage) and birth weight (C: frequency, D: percentage) of neonates during the three surveillance periods, 2008–2012 versus 2013–2017 versus 2018–2022.

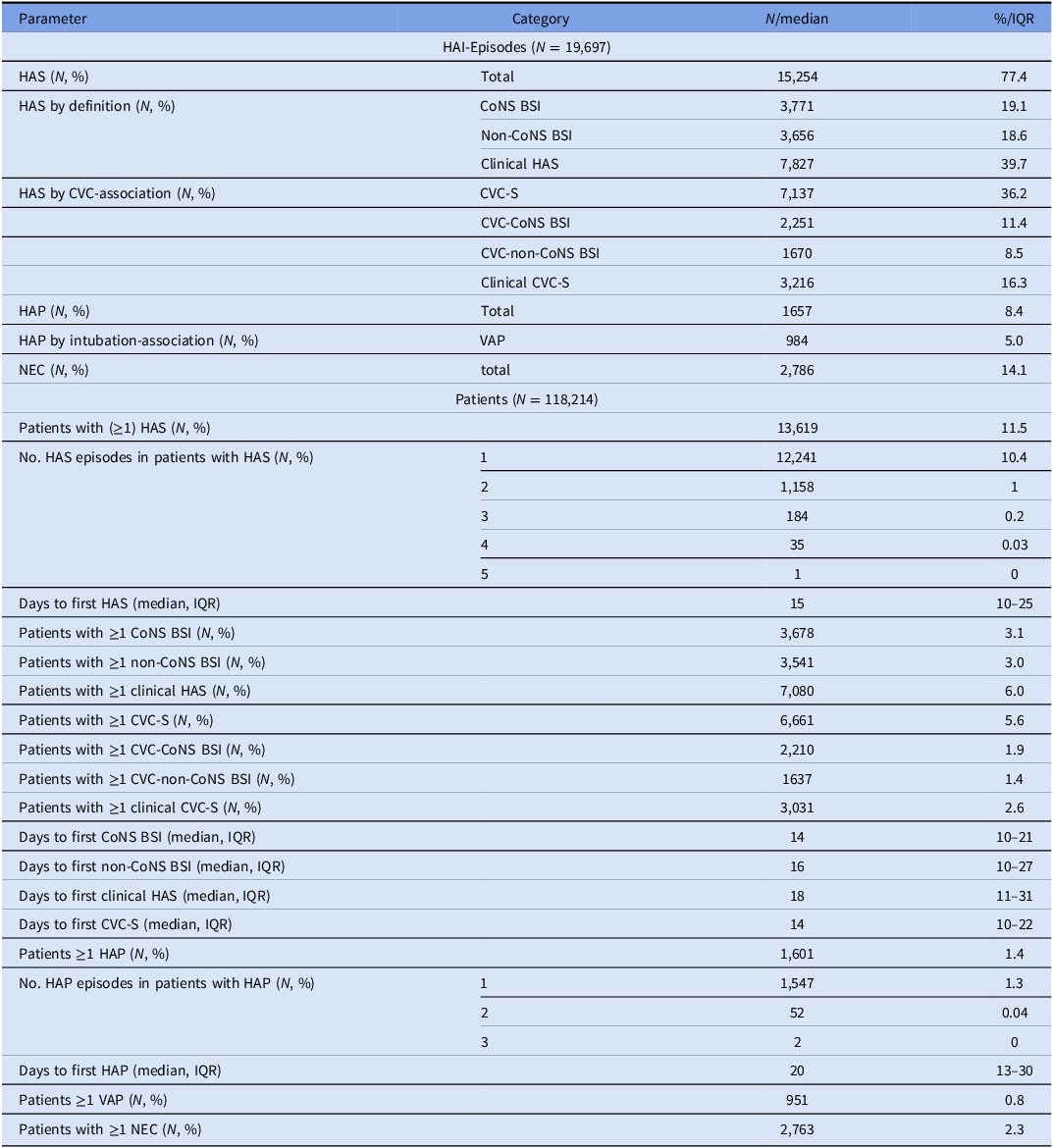
A total of 61.4% of the infants had at least one CVC day and 43.2% received at least one day of invasive mechanical ventilation. Regarding device utilization rates, infants were equipped with a CVC on an average of 26.2% of patient days and were intubated on an average of 12.8% of patient days. Overall, 96,203 (81.4%) infants were followed up until they reached the surveillance end point of 1800 g body weight, 14,781 (12.5%) were transferred or discharged, and 7,230 (6.1%) died while under surveillance. The median length of surveillance was 31 days (IQR 21–47). Table 1 presents an overview of the respective demographic data. In total, 15,254 HAS, 1,657 HAP and 2,786 NEC were counted in 13 619 (11.5%), 1601 (1.4%), and 2,763 (2.4%) cases, respectively (Table 2).
Descriptive data for 118,214 neonates analyzed in the study, NEO-KISS 2008–2022, Germany

CRIB, Clinical risk index for babies; CVC, Central venous catheter; IQR, Interquartile range.
1As defined by the German joint federal committee.10
2Percent of patient days.
Numbers and percentages of different healthcare-associated infections and affected patients from all 118,214 neonates analyzed in the study, NEO-KISS 2008–2022, Germany
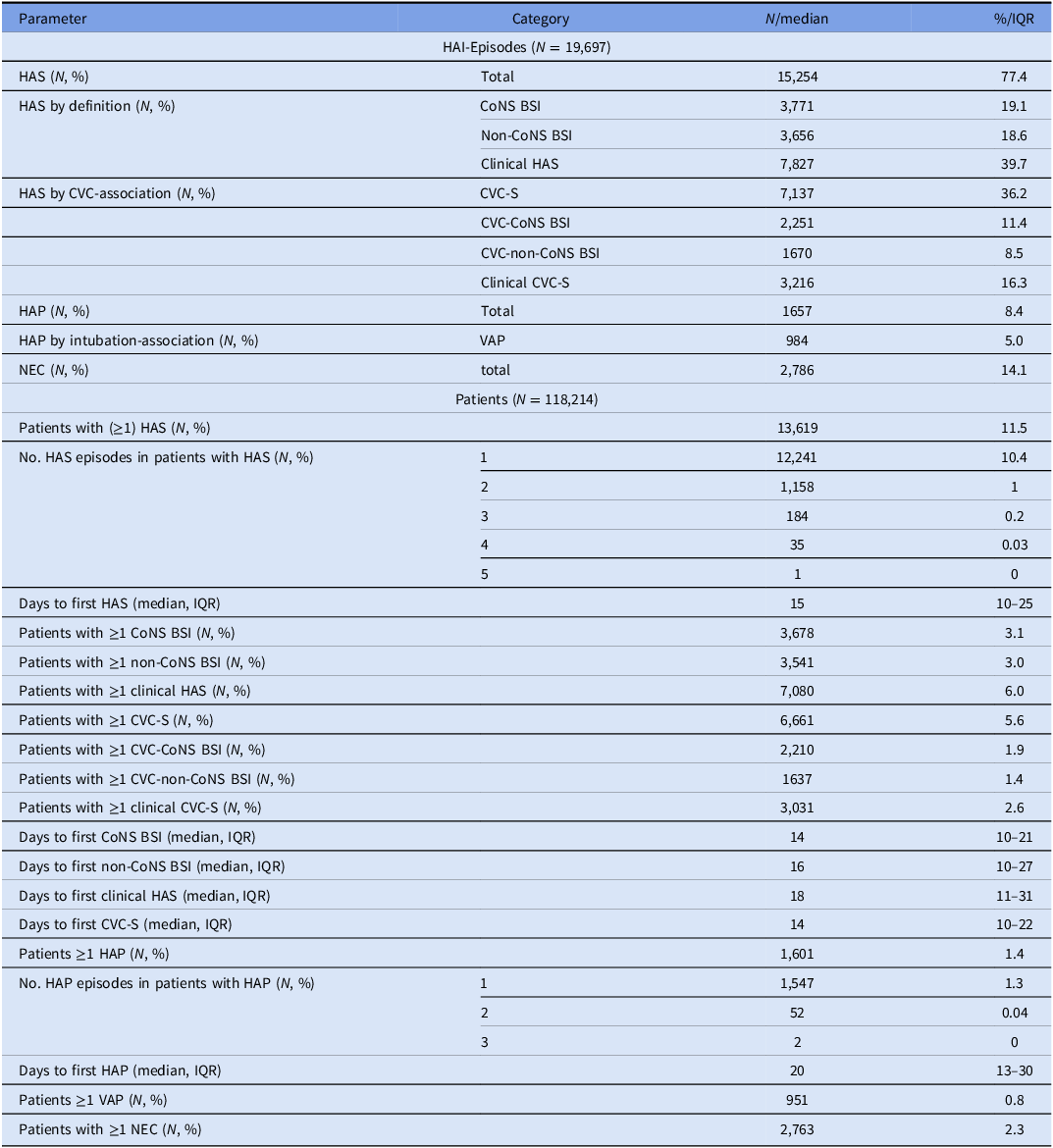
CVC-S, central venous catheter-associated sepsis; CoNS, coagulase-negative staphylococci; HAS, healthcare-associated sepsis; HAP, healthcare-associated pneumonia; IQR, interquartile range; NEC, necrotizing enterocolitis; VAP, invasive mechanical ventilation-associated pneumonia.
Healthcare-associated bloodstream infections
With respect to the different categories of HAS, the total number of 15,254 episodes consist of 3,771 CoNS BSI (24.7%) recorded from 3,678 patients, 3,656 non-CoNS BSI (24.0%) from 3,541 patients, and 7,827 clinical HAS (51.3%) from 7,080 patients. More than one HAS was detected in the course of the admission in 1,378 of all affected patients (10.1%). In addition, 7,137 CVC-S episodes (46.8%) were observed in 6,661 infants. Of these, CVC-CoNS BSI accounted for 2,251 episodes (31.5%), which corresponds to 68% more than the number of CoNS BSI in non-CVC-S (n = 1,520 / 8,117, 18.7%) (Supplementary Figure S1). Additional descriptive results for the entire observation period are listed in Table 2; results of the individual surveillance periods 2008–2012, 2013–2017, and 2018–2022 are shown in Supplementary Table S3.
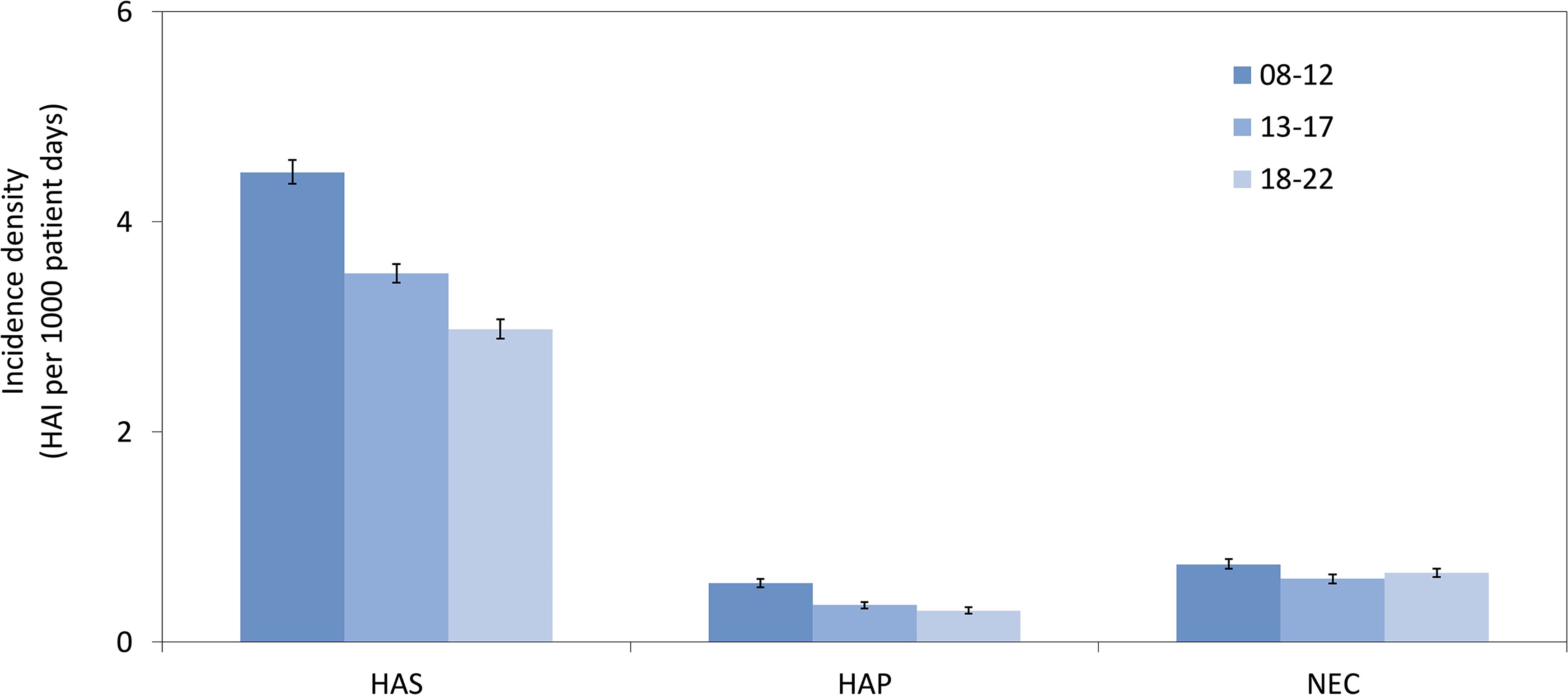
The overall HAS incidence density was 3.63 (per 1,000 patient days), which represents a 33.3% decrease over each of the five-year surveillance periods from 4.47 in 2008–2012 to 2.98 in 2018–2022 (Figure 2).
Incidence densities of healthcare-associated sepsis (HAS), healthcare-associated pneumonia (HAP) and necrotizing enterocolitis (NEC) in neonates during the three surveillance periods, 2008–2012 versus 2013–2017 versus 2018–2022. HAI: healthcare-associated infection.

The degree of decline varied among the different subtypes of HAS. The incidence densities of clinical HAS, CoNS BSI, and non-CoNS BSI decreased from 2.42 to 1.41 (–41.7%), from 1.04 to 0.73 (–29.8%) and from 1.01 to 0.84 (–16.8%), respectively, (Supplementary Table S3). It is noteworthy that from 2013–2017 to 2018–2022 non-CoNS BSI rates increased by 9% from 0.77 to 0.84 with an associated higher percentage of non-CoNS BSI (relative to all HAS) compared to the earlier periods (Figure 3).
Proportions of different types of healthcare-associated sepsis (HAS) in neonates during the three surveillance periods, 2008–2012 versus 2013–2017 versus 2018–2022. CoNS: coagulase-negative staphylococci.

Although CVC days per patient did not decline during the observation period (Supplementary Table S3, Supplementary Figure S2), the CVC-S rate decreased by 28.7% from 7.74 to 5.52. In terms of birth weight and gestational age of infants, incidence densities of all types of HAS decreased significantly as birth weight and gestational age increased (Supplementary Table S5).
Healthcare-associated pneumonia and necrotizing enterocolitis
Regarding HAP, 984 of 1,657 episodes (59.4%) were assessed as VAP (Table 2) a trend that decreased slightly over the surveillance periods (Supplementary Table S3). The overall incidence density was 0.39, which declined by 46.7% from 0.56 during 2008–2012 to 0.3 in 2018–2022 (Figure 2, Supplementary Table S4). Rates of VAP fell from 2.22 to 1.44 (–35.1%) (Supplementary Figure S2), while the intubation rates decreased by 24.5% from 14.7 to 11.1 (Supplementary Table S3). In contrast, NEC incidence densities did not show a clear trend over time (Figure 2). However, both HAP and NEC rates decreased as birth weight and gestational age increased (Supplementary Table S5).
Results of regression analysis
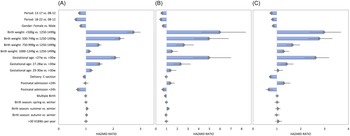
Multivariate regression analysis revealed significant risk reduction for (the first) HAS during the second (2013–2017) and third (2018–2022) period of admission compared to 2008–2012 with hazard ratios (HR) of 0.76 (95% confidence interval (CI): 0.73–0.79) and 0.63 (95% CI: 0.61–0.66) (Supplementary Table S6). A reduced risk was also observed for females (HR 0.81, 95% CI 0.79–0.84), Cesarean births (HR 0.93, 95% CI 0.88–0.98), and admission > 24 h after birth (HR 0.7, 95% CI: 0.63–0.78). An increased risk was found for decreasing birth weight (HR 1000–1249 g vs. 1250–1499 g: 1.12, 95% CI: 1.05–1.2; HR < 500 g vs. 1250–1499 g: 2.74, 95% CI: 2.5–3) and younger gestational age (HR 29–30 weeks vs. >30 weeks: 1.18, 95% CI: 1.1–1.27; HR <27 weeks vs. >30 weeks: 2.07, 95% CI 1.91–2.25) (Figure 4A).
Results of multivariable cox-proportional hazard regression analysis for the occurrence of healthcare-associated sepsis (A), healthcare-associated pneumonia (B) and necrotizing enterocolitis (C) in neonates. VLBW: very low birth weight (<1500 g) infants.

While CoNS BSI, clinical HAS, and CVC-S yielded similar results, the risk of non-CoNS BSI increased again slightly from 2013–2017 to 2018–2022 (Supplementary Table S7).
Admission during a later surveillance period also reduced the risk of HAP. However, no decrease was observed for NEC. HAP was less frequent in cases of multiple birth (HR 0.89, 95% CI: 0.81–0.98) and NEC occurred less often after Cesarean sections (HR 0.73, 95% CI 0.66–0.82) (Figure 4B–C).
Discussion
With almost 120,000 preterm infants and over four million patient days, this paper describes one of the largest cohorts of high-risk neonates ever prospectively monitored for surveillance purposes. When compared with the total number of live births in Germany between 2008–2022 (n = 132,138, including two years with estimated numbers) with a birth weight less than 1500 g,14,15 the reported data encompasses more than 80% of all infants born in Germany during this time period with a birth weight below 1500 g. Moreover, the median length of participation in NEO-KISS of 15 years per department indicates that most departments participated continuously. Therefore, the sample can be considered a comprehensive representation of nationwide data and demonstrates the feasibility of continuous HAI surveillance in neonates in a high-income EU country.
The observed incidence of HAS of 11.5% (13,619/118,214) for the years 2008–2022 was lower than the sepsis incidence of 17.7% identified using ICD-10 codes among infants with a birth weight of <1500 g, as previously reported in a population-wide analysis from Germany.Reference Born, Dame and Matthaus-Kramer16 However, in contrast to NEO-KISS, the authors of that study did not exclude cases of early-onset sepsis.
A strength of NEO-KISS is that not only are laboratory-confirmed cases of HAS registered, but blood culture-negative, clinically diagnosed HAS are as well. Although the clinical decisions associated with this type of HAS tend to be more subjective—and therefore a recording of clinical HAS as well—more than 50% of all observed HAS occurred with negative cultures. Since a percentage of 90% of blood culture-negative cases were found in other studies for late-onset sepsis,Reference Hornik, Fort and Clark17 monitoring for clinical sepsis is an essential part of the surveillance of high-risk neonates.
The observation of a higher percentage of CoNS BSI and a smaller percentage of clinical HAS in CVC-S compared to non-CVC-S is in line with other studies that detected CoNS in 75% of central line-associated BSI vs. 15–68% in other BSI.Reference Jansen, van der Hoeven and van den Akker18 Of note, in the study by Jansen et al., a HAS was defined by at least one positive blood culture and could not be diagnosed based on clinical judgment alone.
The overall decrease in the HAI incidence density (Figure 2) is well aligned with the global decrease in sepsis incidence across all patient groups since 1990.Reference Rudd, Johnson and Agesa19 This may in general reflect improved medical care. Given the fact that CoNS BSI are more likely to be associated with contamination,Reference Souvenir, Anderson and Palpant20 the greater reduction in CoNS BSI incidence density compared with non-CoNS BSI incidence density indicates that IPC measures are among the improvements that have been made, including improved CVC management, early enteral and human milk feeding, use of closed systems for blood sampling, strict adherence to sterile techniques, and, in some settings, the use of probiotics.Reference Denkel, Schwab, Garten, Geffers, Gastmeier and Piening21 Interestingly, no relevant reduction in HAI rates were observed in other longitudinal cohorts that included preterm infants.Reference Jansen, van der Hoeven and van den Akker18,Reference Ceparano, Sciurti and Isonne22 However, these cohorts were not placed under surveillance or did not originate from a single center.
This highlights the importance of a surveillance network like NEO-KISS, which allows for periodic feedback and benchmarking. We can therefore assume that engaging with the surveillance data, together with other IPC measures such as prevention bundles and better training,Reference Neill, Haithcock and Smith23,Reference Schelonka, Scruggs, Nichols, Dimmitt and Carlo24 has contributed to a reduction in the risk of CVC-S. A similar observation was made in a single center study from Nova Scotia that analyzed data from 16 years of surveillance of pediatric patients. A remarkable 84% reduction in central line-associated BSI rates was observed and associated with a simultaneous hand hygiene campaign.Reference Carter, Langley, Kuhle and Kirkland25
Comparable results were found for HAP. In relative terms, its decline over the years has been even greater than that of HAS (47% vs. 33%). Moreover, the rate of VAP decreased by 35%. In contrast to CVC, there was a remarkable drop in the intubation rate. This is most likely related to an increased use of noninvasive techniques like continuous positive airway pressure via supraglottic tube, binasal prongs, and especially high-flow nasal cannula intended to prevent morbidities related to invasive ventilation such as VAP in neonates.Reference Ramaswamy, Devi and Kumar26 Furthermore, the reduction of HAP rates can probably be attributed to an increasing effort to implement bundles to prevent HAP.Reference Bondarev, Ryan and Mukherjee27
NEC incidence densities fluctuated over the surveillance periods and did not show a clear trend. This might reflect the complex, multifactor etiology of this partially endogenous syndrome.Reference Denning, Bhatia, Kane, Patel and Denning28 Therefore, the link to causative health care processes and the effect of IPC interventions is difficult to assess for NEC. However, we consider surveillance of NEC a beneficial tool for identifying clusters nonetheless.Reference Magnusson, Ahle, Swolin-Eide, Elfvin and Andersson29
A later period of admission was independently associated with lower risk of HAS and HAP in the multivariate regression analysis. Moreover, a lower gestational week and birth weight were confirmed as risk factors for HAI, which is in line with the results from large cohort studies.Reference Fanaroff, Korones and Wright30,Reference Klinger, Bromiker and Zaslavsky-Paltiel31
The substantial reduction in the risk of NEC after a Cesarean section is also noteworthy. Although similar results have been reported in the past, there have also been reports showing no difference or even a higher risk following a Cesarean section.Reference Son, Grobman and Miller32 This indicates the need for further investigation of this question.
The following limitations need to be considered.
First, manual surveillance always depends on the expertise and accuracy of the person responsible for carrying out the surveillance. Therefore, the NRZ organizes yearly events to instruct local IPC staff in the validated clinical criteria in the NEO-KISS protocol. Participating hospitals are required to guarantee that the recording of data is performed only by IPC staff that have had this training. Second, in 2017, during the observation period, two relevant changes to the NEO-KISS protocol were introduced and implemented. These were i) a cutoff period of 14 days, in reference to which a second infection in the same organ or with the same bacterial species cannot be diagnosed until at least 14 days after the first infection, and ii) by definition, “device-association” applies only if the device has been in place for at least three days either on the day of the first onset of symptoms or on the day before onset of symptoms. Third, device days could not be incorporated into the analysis, as the available data do not distinguish between a device preceding infection and a device occurring as a consequence of the infection. Fourth, since blood culture sampling frequency is not recorded in NEO-KISS, the changes in HAS rates could ultimately have been the result of changes in blood culture sampling policies. Fifth, comparability of NEO-KISS with surveillance systems from other countries is limited, but our results strongly support the goal of initiatives that aim at introducing internationally harmonized surveillance systems for nosocomial infections in high-risk neonates similar to the NeoIPC Surveillance.33
Here, for the first time, data is presented from a nationwide surveillance system for HAI in high-risk neonates which has been maintained continuously in Germany for more than two decades. Evaluation based on surveillance periods shows a continuous reduction in incidence densities of HAS and HAP over time. In conclusion, we interpret this decrease as a sign of improved treatment, which includes improved IPC measures and the ongoing surveillance of HAI. The feasibility and success of a nationwide surveillance system for preterm infants should encourage wider adoption of methods like NEO-KISS.
Supplementary material
Supplementary material for this article can be found at https://doi.org/10.1017/ice.2026.10407.
Data availability
Not applicable, because all data were collected as part of the surveillance of (HAI) conducted in accordance with the German Protection Against Infection Act.
Acknowledgements
We thank all of the staff and clinicians of the neonatal departments who participated in the study for their support and participation. We thank Gerald Brennan for proofreading as a native speaker.
Financial support
The NEO-KISS receives financial support from the German Ministry of Health.
Competing interests
All authors report no conflicts of interest relevant to this article.
Ethical standard
Ethics approval and informed consent were not needed for this retrospective analysis of data routinely collected as part of the surveillance of (HAI) conducted in accordance with the German Protection Against Infection Act.
Use of artificial intelligence tools
OpenAI’s ChatGPT V4 was employed as a drafting tool to assist with refining the language, coherence, and structure of this manuscript. All scientific content, analyses, and conclusions were exclusively the responsibility of the authors.