Introduction
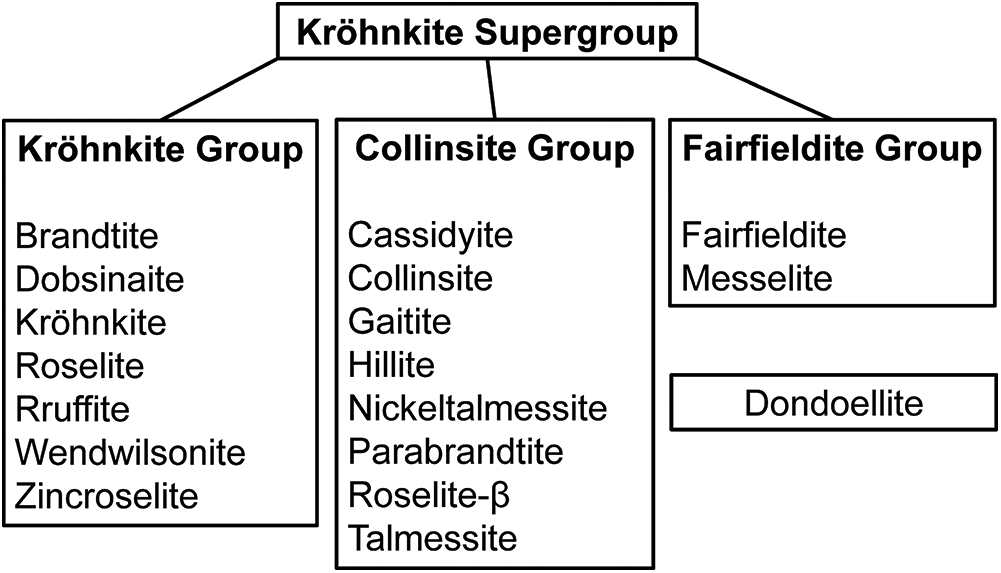
A mineral supergroup consists of two or more groups that have essentially the same structure and are composed of chemically similar elements (Mills et al., Reference Mills, Hatert, Nickel and Ferraris2009). The minerals of the kröhnkite, talmessite and fairfieldite groups have the general formula
where X = Na or Ca; M2+ = Mg, Fe, Mn, Co, Ni, Zn or Cu; and T = S, P or As. There are currently 17 IMA-approved minerals in these three groups (Table 1) plus a single mineral, dondoellite, with a structure different from but related to the structures of the minerals of these three groups. The Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (IMA–CNMNC) has approved the creation of the kröhnkite supergroup (Bosi et al., Reference Bosi, Hatert, Pasero and Mills2024). The details of the minerals in the supergroup and their division into three groups are shown in Fig. 1. The currently named kröhnkite, talmessite and fairfieldite groups are retained, and belong to the kröhnkite supergroup. Dondoellite is an isolated structure within the kröhnkite supergroup but cannot form a group until an isotypic mineral is approved whereupon a new group can be proposed.
The classification of the minerals of the kröhnkite supergroup.

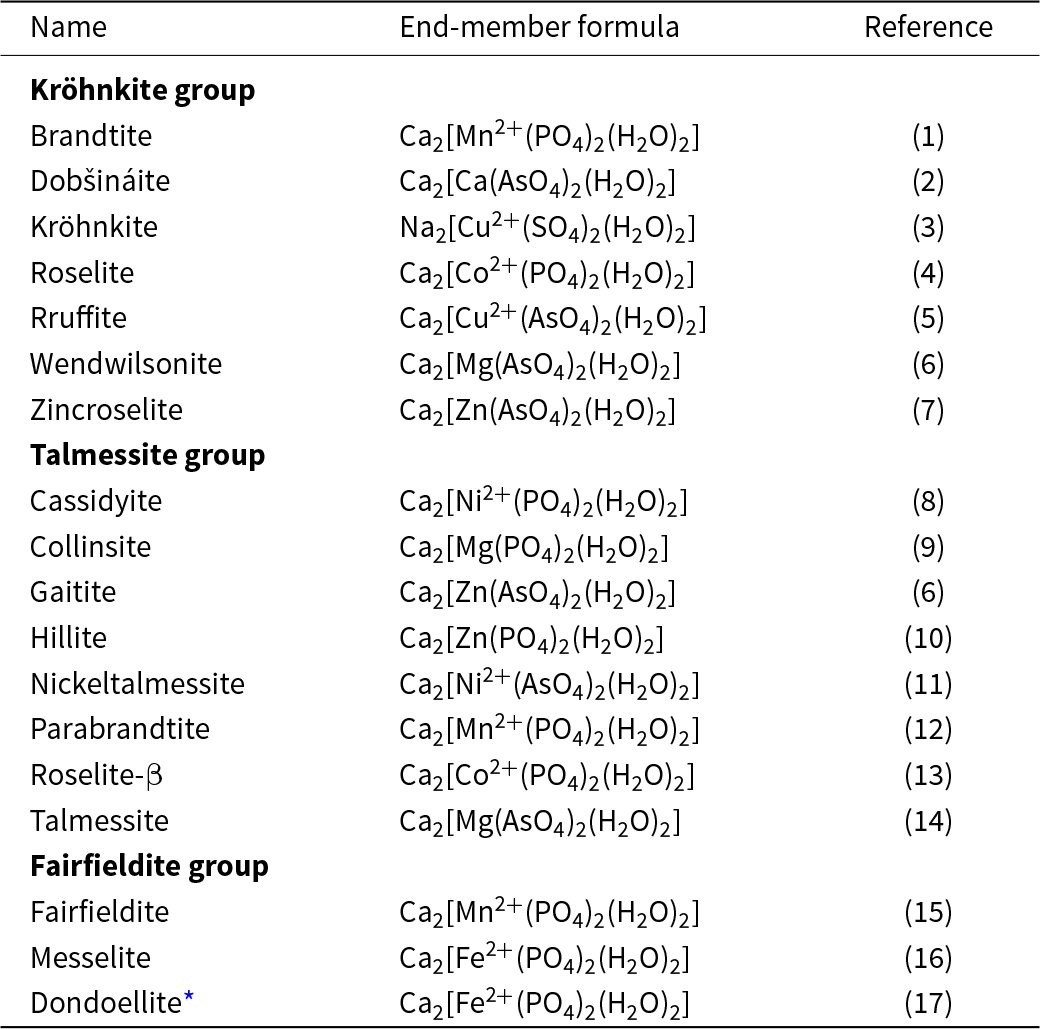
Minerals of the kröhnkite supergroup

* Dondoellite falls within the kröhnkite supergroup but not within the kröhnkite, talmessite and fairfieldite groups; it does not (as yet) have any isostructural minerals and hence cannot form a group, but it is likely that this will happen in due course.
References: (1) Dahlman (Reference Dahlman1951); (2) Sejkora et al. (Reference Sejkora, Števko, Škoda, Víšková, Toman, Hreus, Plášil and Dolníček2021); (3) Hawthorne and Ferguson (Reference Hawthorne and Ferguson1975); (4) Hawthorne and Ferguson (Reference Hawthorne and Ferguson1977); (5) Yang et al. (Reference Yang, Jenkins, Downs and Evans2011); (6) Kolitsch and Fleck (Reference Kolitsch and Fleck2006); (7) Keller et al. (Reference Keller, Lissner and Schleid2004); (8) White et al. (Reference White, Henderson and Mason1967); (9) Brotherton et al. (Reference Brotherton, Maslen, Pryce and White1974); (10) Yakubovich et al. (Reference Yakubovich, Massa, Liferovich, Gavrilenko, Bogdanova and Tuisku2003); (11) Chukanov et al. (Reference Chukanov, Mukhanova, Möckel, Belakovsky and Levitskaya2020); (12) Dunn et al. (Reference Dunn, Sturman and Nelen1987); (13) Frondel (Reference Frondel1955); (14) Catti et al. (Reference Catti, Ferraris and Ivaldi1977); (15) Fanfani et al. (Reference Fanfani, Nunzi and Zanazzi1970); (16) Fleck and Kolitsch (Reference Fleck and Kolitsch2003); (17) Yang et al. (Reference Yang, Gibbs, Mcglasson, Jenkins and Downs2022).
Structure
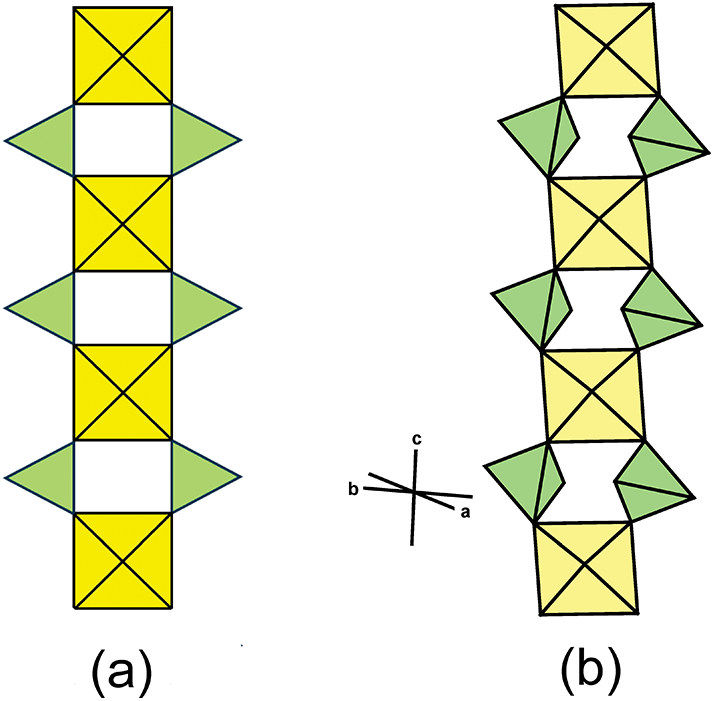
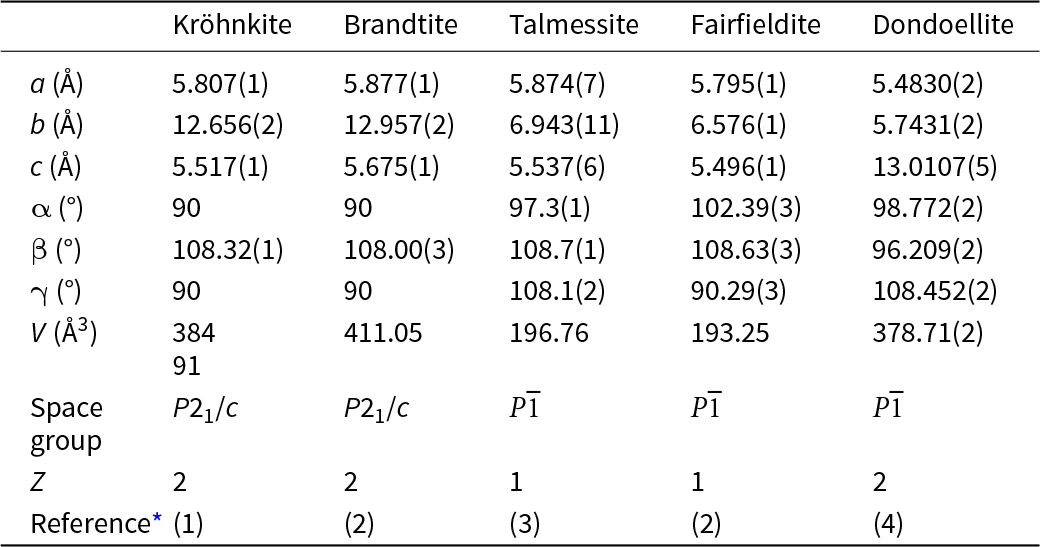
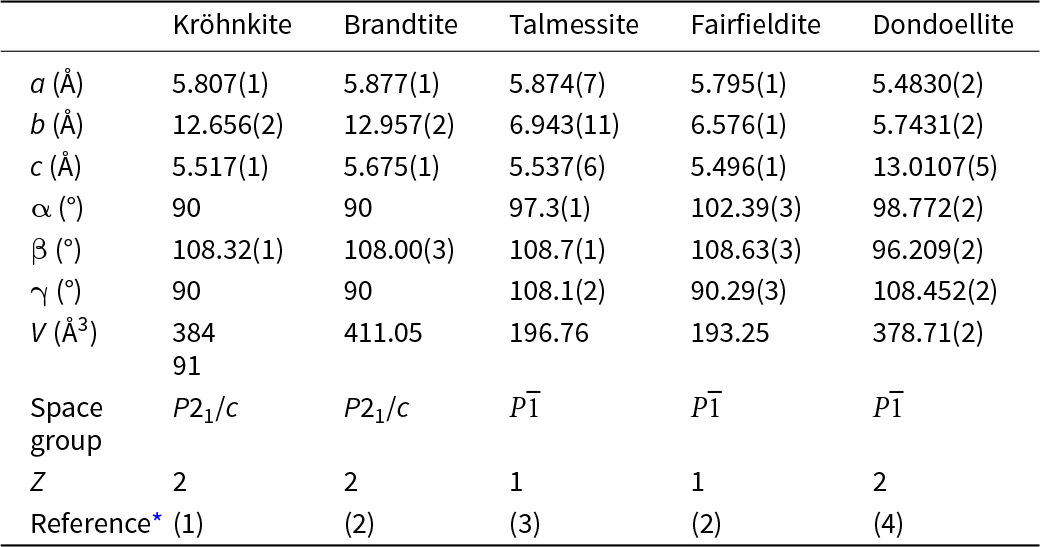
All minerals in the kröhnkite supergroup are either monoclinic or triclinic. The cell dimensions and associated space-group symmetries of the minerals in these three groups and dondoellite are distinct (Table 2), but the structures of these minerals are strongly related as they have topologically identical structural units (Lima-de-Faria et al., Reference Lima-de-Faria, Hellner, Liebau, Makovicky and Parthé1990) that define the c-dimension of each group (Fig. 2). The structural unit is an [M(TO4)2Φ2] chain (Hawthorne, Reference Hawthorne1985) as shown idealised in Fig. 2a and as in the structure of brandtite in Fig. 2b. As noted by Hawthorne (Reference Hawthorne1983), the linkage between polyhedra in Fig. 2 maximises the accord with the valence-sum rule (Brown, Reference Brown2016; Hawthorne, Reference Hawthorne2012, Reference Hawthorne2015) for linkage of isolated octahedra and isolated tetrahedra into a chain without octahedron–octahedron or tetrahedron–tetrahedron linkages.
The [M(TO4)2Φ2] chain (Φ = unspecified anion) that forms the structural unit in the minerals of the kröhnkite supergroup; (a) the generalized chain (modified from Hawthorne, Reference Hawthorne1985); and (b) the [Mn2+(PO4)2(H2O)2] chain in brandtite. Drawn using ATOMS V6.4 (Dowty, Reference Dowty2016).

Crystallographic properties of kröhnkite, brandtite, collinsite, fairfieldite and dondoellite
* References: (1) Hawthorne and Ferguson (Reference Hawthorne and Ferguson1975); (2) Herwig and Hawthorne (Reference Herwig and Hawthorne2006); (3) Catti et al. (Reference Catti, Ferraris and Ivaldi1977); (4) Yang et al. (Reference Yang, Gibbs, Mcglasson, Jenkins and Downs2022).
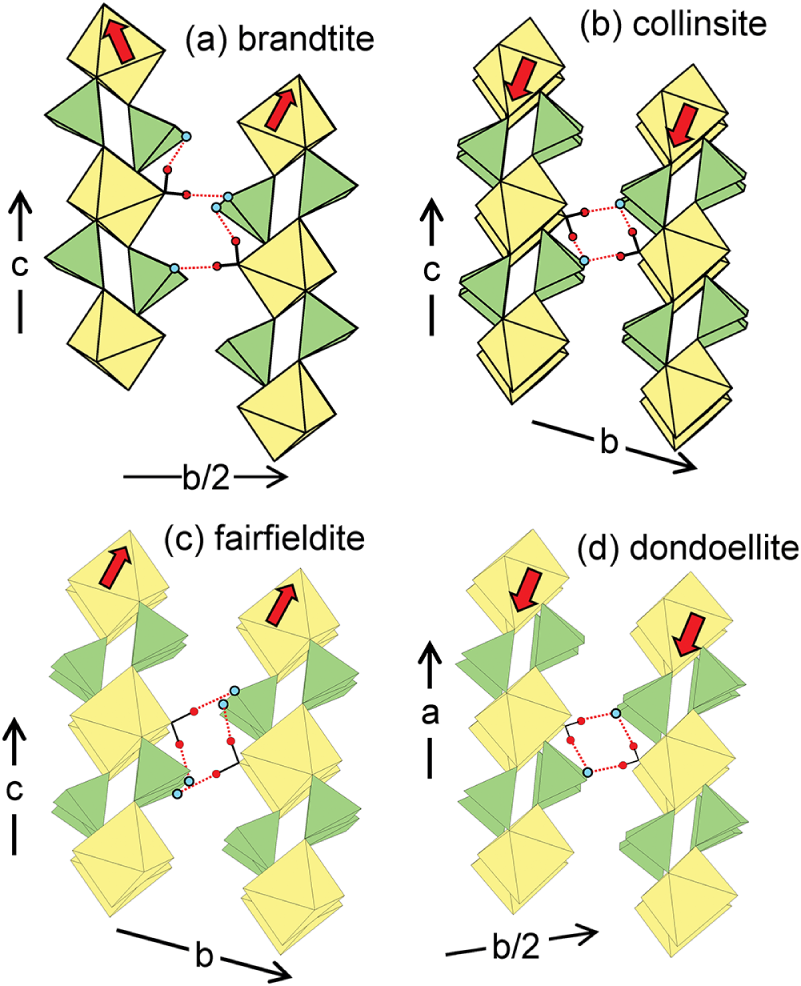
Differences in structure between the different groups involve the disposition of adjacent [M2+(TO4)2(H2O)2] chains and the details of the hydrogen bonds that link adjacent [M2+(TO4)2(H2O)2] chains. As indicated by the red arrows in Fig. 3, in the structures of the kröhnkite-group minerals (Fig. 3a), the octahedra of the structural units point alternately northeast and northwest in adjacent chains; in the structures of the talmessite-group minerals (Fig. 3b), the octahedra of the structural units point southwest in adjacent chains; in the structures of the fairfieldite-group minerals (Fig. 3c), the octahedra of the structural units point northeast in adjacent chains; and in the structure of dondoellite (Fig. 3d), the octahedra of the structural units point southwest in adjacent chains.
The crystal structures of (a) brandtite; (b) collinsite; (c) fairfieldite; and (d) dondoellite, projected onto (100). M octahedra are yellow, T tetrahedra are green, and H are small red circles; solid black lines: Odonor–H bonds; red dotted lines: hydrogen bonds; small blue circles: Oacceptor anions. The red arrows show the relative attitude of octahedra in adjacent chains. Drawn using ATOMS V6.4 (Dowty, Reference Dowty2016).

In the structures of the minerals of all groups, one H atom of the (H2O) group hydrogen-bonds to a (TO4) oxygen of the same chain and the other H atom of the (H2O) group hydrogen-bonds to a (TO4) oxygen of an adjacent chain. As indicated in Fig. 3a, in the kröhnkite-group structures, only two chains are involved in this mutual linkage of opposing (H2O) groups. In the collinsite-group structures, the fairfieldite-group structures, and in dondoellite, four chains are involved in this mutual linkage of opposing (H2O) groups (Fig. 3b,c,d).
There is no apparent sorting of structure type by cation radii and there are several compositions that occur as polymorphs that belong to different groups: brandtite–parabrandtite–fairfieldite; roselite–roselite-β; wendwilsonite–talmessite; zincroselite–gaitite; and messelite–dondoellite.
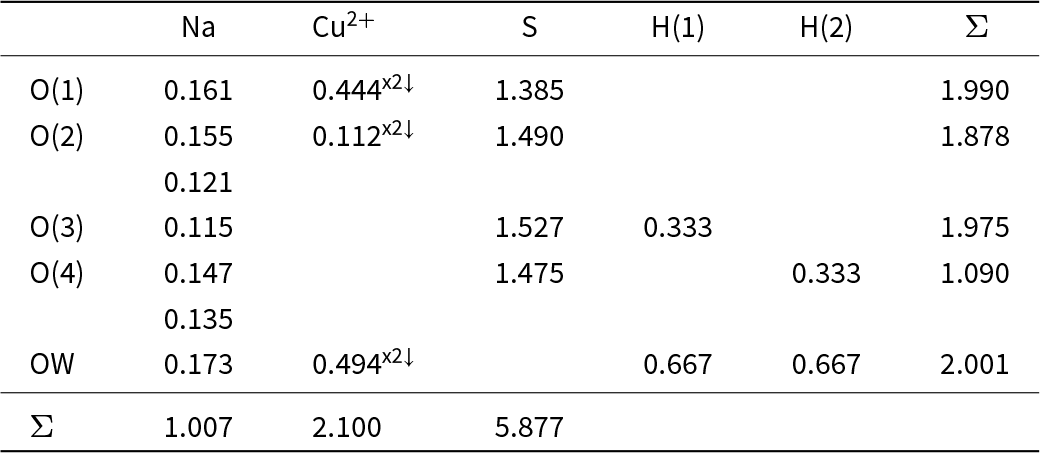
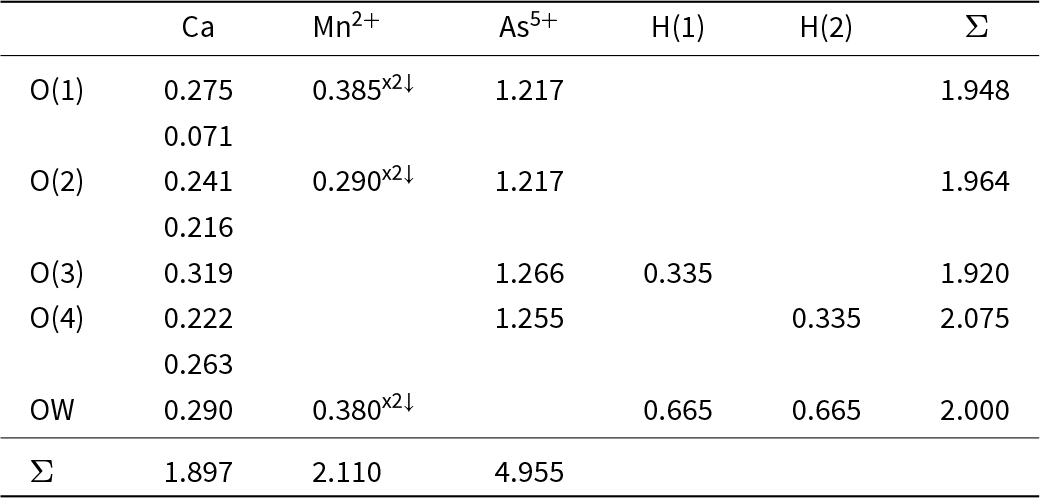
The bond-valence tables for kröhnkite and brandtite are shown in Tables 3 and 4, respectively. The bond topology of the two structures is the same and hence they have a similar pattern of bond valences. The coordination numbers of the anions, omitting the hydrogen bonds, is the same: O(1) = [3], O(2) = [4], O(3) = [2], O(4) = [3], OW = [2], and an unusual feature of each structure is the fact that the OW (H2O) group has significant incident bond-valence: 0.667 and 0.660 valence units (vu) and hence the associated hydrogen bonds are stronger than usual for bonded (H2O) groups in minerals. These strong hydrogen bonds compensate for the variation in coordination number of the anions and ensure that the valence-matching principle is satisfied (Tables 3 and 4). The incident bond-valence at the OW (H2O) group is the same in both kröhnkite and brandtite (∼0.67 vu) and yet the oxidation states of the coordinating cations are 1+ and 2+ in kröhnkite and 2+ and 2+ in brandtite; how can the same incident bond-valence be maintained in these two minerals? The key to this issue is the fact that the divalent cation in kröhnkite is Cu2+. Octahedrally coordinated Cu2+ is Jahn-Teller active (Burns and Hawthorne, Reference Burns and Hawthorne1996) and typically shows four short meridional bonds and two long apical bonds (Eby and Hawthorne, Reference Eby and Hawthorne1993; Gagné and Hawthorne, Reference Gagné and F.C2020). This is the case in kröhnkite. As the OW (H2O) group in kröhnkite is a meridional anion, it has a very strong bond incident from Cu2+ which compensates for the weaker Na–OW bond in kröhnkite (Table 3) compared to the stronger Ca–OW bond in brandtite (Table 4). This accounts for the absence of mineral compositions with the general formula Na2[6]M2+(SO4)2(H2O)2 other than kröhnkite with M2+ = Cu2+.
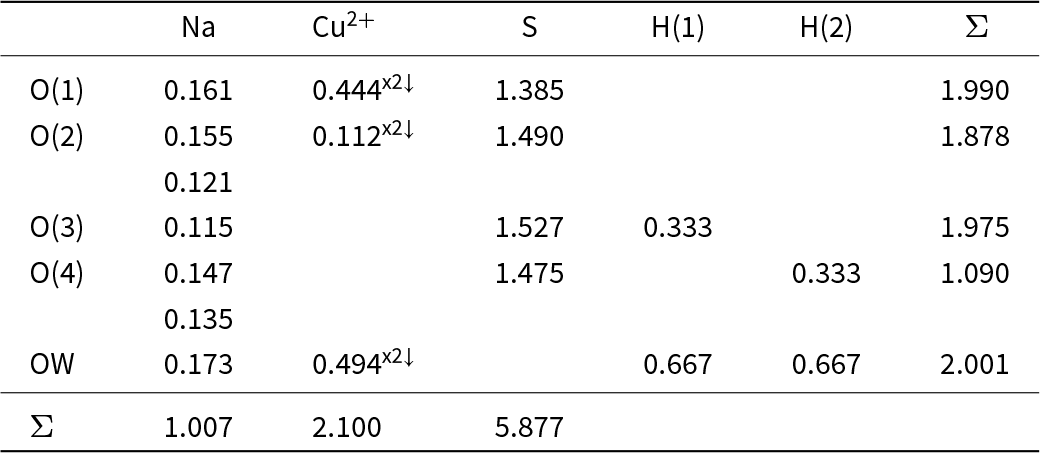
* Bond-valence curves from Gagné and Hawthorne (Reference Gagné and F.C2015);
** O(1) and O(3) have been interchanged to make the site labels consistent in kröhnkite and brandtite.
Bond-valence (vu) table * for brandtite

* Bond-valence curves from Gagné and Hawthorne (Reference Gagné and F.C2015).
Controls on chemical composition in kröhnkite-supergroup structures
The general formula for the kröhnkite-supergroup minerals is X2M2+(TO4)2(H2O)2. What ions are possible in this topological structure-type at the various sites in the structure? Inspection of Table 1 shows that X = Na+ or Ca2+; M2+ = Mg, Fe, Mn, Co, Ni, Zn or Cu; and T = S6+, P5+ and As5+. What other ions can feasibly enter a crystal structure with the kröhnkite bond topology? In regard to this question, there has been considerable work on synthetic analogues of these minerals (e.g. Fleck et al., Reference Fleck, Kolitsch and Hertweck2002; Fleck and Kolitsch, Reference Fleck and Kolitsch2003; Kolitsch and Fleck, Reference Kolitsch and Fleck2005, Reference Kolitsch and Fleck2006; Wildner and Stoilova, Reference Wildner and Stoilova2003) as they show potential for industrial applications.
To approach this question, we use binary representation (Schindler and Hawthorne, Reference Schindler and Hawthorne2001; Hawthorne and Schindler, Reference Hawthorne and Schindler2008) whereby a structure is divided into a structural unit and an interstitial complex. The ions that form the interstitial complex of a mineral and the structural unit must satisfy the principle of correspondence of Lewis acidity–basicity (Hawthorne, Reference Hawthorne2012, Reference Hawthorne2015; Hawthorne and Schindler, Reference Hawthorne and Schindler2008), a mean-field version of the valence-matching principle (Brown, Reference Brown, O’Keeffe and Navrotsky1981; Hawthorne, Reference Hawthorne1994) whereby stable structures will form when the Lewis acid strength of the cation closely matches the Lewis base strength of the anion. Thus an ion may enter a structure with the kröhnkite bond topology if the resultant structure accords with the principle of correspondence of Lewis acidity–basicity.
The structural unit with the kröhnkite bond topology may be written as follows:
The number of bonds in the structural unit = 1 × [6] + 2 × [4] + 4 × [2] = 22. Note that although two of the hydrogen bonds exit the structural unit, they bond to anions in adjacent structural units and the two exident hydrogen-bonds are balanced by the two incident hydrogen-bonds from adjacent structural units. The number of bonds needed for [4]-coordination of all simple anions and [3]-coordination of (H2O) = 8 × [4] × 2 × [3] = 38. The number of additional bonds to the structural unit to achieve this coordination = 38 – 22 = 16 and hence the structural unit needs 16 bonds from the interstitial complex.
The charge on the structural unit [[6]M2+([4]Tn+O4)2([2]H2O)2] = 2+ + 2 × n + + 4 × 1+ – 2– × 10 = 6 + 2n – 20 = 2n – 14. The Lewis basicity of the structural unit = charge/bonds = (2n – 14)/14 vu.
For n = 5+ (P5+, As5+), the Lewis basicity of the structural unit = 4/16 = 0.250 vu. The Lewis basicity of Ca2+ = 0.264 vu (Gagné and Hawthorne, Reference Gagné and F.C2017) is the closest match with the Lewis basicity of the structural unit containing pentavalent cations. The Lewis basicity of Pb2+ = 0.266 vu but no Pb-bearing species occur; this may be a steric effect due to the large size of [8]Pb2+ (2.697 Å) compared to that of [8]Ca2+ (2.498 Å) (values from Hawthorne and Gagné, Reference Hawthorne and O.C2024).
For n = 6+ (S6+), the Lewis basicity of the structural unit = 2/16 = 0.125 vu. The Lewis basicity of Na+ is 0.159 vu but the coordination of Na+ in kröhnkite is [7] (Table 3) which reduces the effective Lewis acidity to 1/7 = 0.143 vu. The Lewis basicity of K+ is 0.108 vu (Gagné and Hawthorne, Reference Gagné and F.C2017). K-bearing kröhnkite-supergroup minerals have not been found but K-bearing synthetics have been synthesised with a view to their use in alkali-metal batteries (e.g. Barpanda et al., Reference Barpanda, Oyama, Ling and Yamada2014; Marinova et al., Reference Marinova, Kostov, Nikolova, Kukeva, Zhecheva, Sendova-Vasileva and Stoyanova2015; Watcharatharapong et al., Reference Watcharatharapong, Thienprasert, Barpanda, Ahuja and Chakraborty2017) and various possible optical applications (e.g. de Oliveira Neto et al., Reference de Oliveira Neto, Lang, Rodrigues, Gutiérrez, Murillo, de Sousa, Filho and dos Santos2022).
Acknowledgements
I thank Aaron Lussier and two anonymous reviewers for their comments on this paper.
Financial statement
This work was supported by a Discovery Grant from the Natural Sciences and Engineering Research Council of Canada.
Competing interests
The author declares none.