Introduction
Entomopathogenic nematodes (EPNs) are important biological control agents for pests in natural ecosystems. These nematodes, primarily represented by Steinernema and Heterorhabditis species, exhibit remarkable host-seeking capabilities, entering insect hosts through natural openings (mouth, anus) or integumentary wounds (Stuart et al. Reference Stuart, Barbercheck, Grewal, Taylor and Hoy2006). They not only effectively suppress pest populations and mitigate crop losses, but also substantially reduce dependence on chemical pesticides, thereby contributing to more sustainable agricultural systems (Koppenhöfer et al. Reference Koppenhöfer, Shapiro-Ilan and Hiltpold2020; Nurashikin-Khairuddin et al. Reference Nurashikin-Khairuddin, Abdul-Hamid, Mansor, Bharudin, Othman and Jalinas2022; van Zyl and Malan Reference van Zyl and Malan2014). The remarkable pathogenicity of EPNs stems from their symbiotic relationship with specific bacteria. Steinernema species exclusively harbor Xenorhabdus bacteria, while Heterorhabditis species carry Photorhabdus bacteria as their symbiotic partners. During host infection, these muturalistic bacteria are released into the insect hemocoel, rapidly inducing host mortality through acute septicemia (San-Blas Reference San-Blas2013; Stuart et al. Reference Stuart, Barbercheck, Grewal, Taylor and Hoy2006).
The EPNs exhibit a near-global distribution, having been reported from all continents except Antarctica. Extensive field surveys have demonstrated their widespread occurrence across diverse ecosystems, particularly in temperate and tropical regions (Bhat et al. Reference Bhat, Chaubey and Askary2020). The past decade has witnessed significant taxonomic progress in the genus Steinernema, with the descriptions of numerous novel species significantly expanding both our understanding of the group’s biodiversity and the available options for biological pest control applications (Lacey and Georgis Reference Lacey and Georgis2012).
The described biodiversity of EPNs currently included 113 validated species in the genus Steinernema and 21 species in its sister genus Heterorhabditis (Půža et al. Reference Půža, Nermuť, Konopická and Habuštová2024). The classification of these nematode species employs an integrative taxonomic approach incorporating diagnostic morphological characteristics, species-specific ecological habits, and their relationship with symbiotic bacteria. Molecular phylogenetic analysis based on the sequences of the internal transcribed spacer (ITS) region of ribosomal RNA (rRNA) genes has resolved the species of the genus Steinernema into 12 well-defined clades, comprising nine multispecies clades and three monotypic clades (Spiridonov and Subbotin Reference Spiridonov and Subbotin2016).
From 2020 to 2024, we conducted a comprehensive survey investigating the occurrence and distribution of EPNs in the Tarim Basin of Xinjiang, China. The survey yielded more than 20 EPN isolates, including three Steinernema isolates that were initially identified as conspecificity based on morphological characters, and belonged to the ‘kushidai-clade’ based on molecular characters (Spiridonov and Subbotin Reference Spiridonov and Subbotin2016). The ‘kushidai-clade’ currently comprises four described species including S. akhursti Qiu et al. Reference Qiu, Hu, Zhou, Mei, Nguyen and Pang2005, S. anatnagense Bhat et al. Reference Bhat, Machado, Abolafia, Askary, Půža, Ruiz-Cuenca, Rana, Sayed and Al-Shuraym2023, S. kushidai Mamiya, Reference Mamiya1988), and S. populi Tian et al. Reference Tian, Zhu, Li, Zhang, Půža, Shapiro-Ilan, Zhao, Liu, Zhou, Ding, Wang, Ma, Zhu, Li and Li2022. Following detailed morphological examinations and phylogenetic analyses, three isolates were confirmed as identical and nominated as Steinernema tarimense n. sp. For the taxonomic characterization of this new species, we selected isolate Z32 as the type material, which serves as the basis for both morphological description and biological characterization.
Materials and methods
Nematode isolation and cultivation
During the survey conducted in April 2024, three Steinernema isolates (R31, R39, and Z32) were collected from soil of the Populus euphratica forest in Yuli County at Tarim Basin of Xinjiang, China. The soil samples were collected from a square of 10 m × 10 m area and taken in a depth of 10–30 cm using a 5-point cross-sampling method. Approximately 500 g homogenized soil from all five sampling points constituted one soil sample. All soil samples were sealed in plastic bags and transported to the laboratory. Seven mature larvae of Galleria mellonella were subsequently placed into each bag and mixed thoroughly with the soil. The bags were then kept in the dark at 25°C. After 2–3 days later, the dead G. mellonella larvae were collected from soil samples, and the third-stage infective juveniles (IJs) of EPN were recovered from each larva by the White trap method (White Reference White1927). The emerged IJs were collected and cleaned 3–4 times using sterilized water. The IJs suspensions were adjusted to a concentration of approximately 1,000 IJs/ml using a 1% formalin solution, and stored at 10°C in petri dishes (diam. 9 cm). During the preservation period, the IJs were recovered with G. mellonella larvae every 3–4 months.
Morphological observations
Different life stages of the isolate Z32 were obtained from the infected G. mellonella larvae, which were exposed to 1400 IJs/insects in a 9-cm-diameter Petri dish lined with two moistened filter papers and kept in the dark at 25°C. The first- and second-generation adult nematodes were obtained by dissecting infected G. mellonella cadavers in Ringer’s solution after 2 and 5 days infection, respectively, under an Olympus SZ61 microscope (Tokyo, Japan). The IJs were collected from the cadavers after 6–8 days infection.
For light microscopy (LM) observation, the above adults and juveniles were killed in 60°C water, fixed in triethanolamine formalin (TAF), and dehydrated using the slow evaporation method (Nguyen Reference Nguyen, Nguyen and Hunt2007a). Dehydration was performed by placing the fixed nematodes in an embryo dish with a 5% glycerin solution and allowing them to slowly evaporate for at least a week. After processed in pure glycerin, the nematodes were subsequently mounted in anhydrous glycerin on permanent slides. The morphological observations, morphometrics, and photographics were taken under an Olympus CX41 phase contrast microscope. The drawings of morphological characters were performed using a drawing tube connected to an Olympus BHA light microscope, and in conjunction with the Adobe Illustrator CS5.
For scanning electron microscopy (SEM) observation, the first-generation adult males and IJs were fixed in 4% formalin buffered with 0.1 M sodium cacodylate (pH 7.2) for 24 h at 8°C. They were subsequently fixed with a 2% osmium tetroxide solution for 12 h at 25°C, then dehydrated through a graded series of ethanol, critical point dried using liquid carbon dioxide, mounted on SEM stubs, and coated with gold (Nguyen Reference Nguyen, Nguyen and Hunt2007a). Specimens were then photographed using a Regulus 8100 microscope (Hitachi, Tokyo, Japan).
DNA extraction
The DNA of approximate 1,000 IJs from each of three isolates was extracted according to the methods described in Nguyen (Reference Nguyen, Nguyen and Hunt2007a). The IJs were collected into a 1.5 ml centrifuge tube and surface-sterilized with 0.1% NaOCl for 30 min. After being centrifuged at 1,000 r/min for 5 min, the nematode precipitate was washed 3 to 5 times with sterile water. After adding proteinase K (final concentration 60 μg/ml), the tube was put in liquid nitrogen for 3 to 5 min and subsequently incubated at 65°C for 5 min. Afterward, the tube was quickly put in liquid nitrogen again for another 3 to 5 min and incubated at 65°C for 5 min. After these steps were repeated 2 to 3 times, the tube was incubated at 95°C for 10 min to degenerate proteinase. Finally, the tube was centrifuged at 12000 r/min for 2 min, and the supernatant was collected as the template for PCR amplification or stored at -20°C.
PCR amplification and sequencing
The near full-length fragment of ITS regions of rRNA genes was amplified from the DNA template with the forward primer 18S (5’-TTG ATT ACG TCC CTG CCC TTT-3’) and the reverse primer 26S (5’-TTT CAC TCG CCG TTA CTA AGG-3’) (Vrain et al. Reference Vrain, Wakarchuk, Levesque and Hamilton1992). The fragment of D2-D3 regions of 28S rRNA gene was amplified with the forward primer D2F (5’-CCT TAG TAA CGG CGA GTG AAA-3’) and the reverse primer 536 (5’-CAG CTA TCC TGA GGA AAC-3’) (Nguyen, Reference Nguyen, Nguyen and Hunt2007a). The fragment of 12S mitochondrial gene (mt12S) was amplified with the forward primer 505F (5′-GTT CCA GAA TAA TCG GCT AGA C-3′) and the reverse primer 506R (5′-TCT ACT TTA CTA CAA CTT ACT CCC CC-3′) (Nadler et al. Reference Nadler, Bolotin and Stock2006).
The PCR reaction was carried out in a total volume of 25 μl containing 2.5 μl of DNA template, 2.5 μl of 10× PCR buffer, 1 μl of a dNTP mixture (10 mM each), 1 μl of each primer (10 mM), 1.5 μl of MgCl2 (50 mM), and 0.25 μl of Taq DNA polymerase (5 U μl), and adding distilled water to the full volume. The PCR programs for amplifying ITS and 28S fragments were followed by the description in Nguyen (Reference Nguyen, Nguyen and Hunt2007a) as: an initial step of 94°C for 7 min; 35 cycles of 94°C for 60 s, 50°C for 60 s, and 72°C for 60 s; a final step of 72°C for 10 min. The PCR program for mt12S fragment was as follows: an initial step of 94°C for 3 min; 30 cycles of 94°C for 30 s, 50°C for 30 s, 72°C for 45 s; and a final step of 72°C for 15 min. PCR products were separated on 1% agarose gels and visualised by staining with ethidium bromide. PCR products of sufficiently high quality were purified for cloning and sequencing by the Sangon Bioengineering Co., Ltd. (Shanghai, China).
Phylogenetic analysis
The newly obtained sequences were analyzed using BLAST to identify closely related species and compare them with existing sequences in the GenBank database. The sequences of relevant Steinernema species were downloaded from the GenBank. Multiple sequence alignments were generated for these sequences using the default Clustal X 1.8 configuration in software MEGA 7 and were subsequently optimized manually in BioEdit (Hall Reference Hall1999). Sequences similarities were calculated by Pairwise distances using the MegAlign Pro 17. The sequence datasets were analyzed with the Bayesian inference (BI) on CIPRES Science Gateway v.3.3 (Miller et al. Reference Miller, Pfeiffer and Schwartz2010) using MrBayes 3.2.3 (Ronquist et al. Reference Ronquist, Teslenko, van der Mark, Ayres, Darling, Höhna, Larget, Liu, Suchard and Huelsenbeck2012). The best-fitting models were identified based on the Akaike information criterion using the ModelFinder (Kalyaanamoorthy et al. Reference Kalyaanamoorthy, Minh, Wong, von Haeseler and Jermiin2017). The BI analysis of ITS sequences was performed under the GTR + F + G4 model, while the 28S and mt12S sequences were analyzed using the GTR + F + I + G4 model, and the tree topology was confirmed using IQ-TREE (Nguyen et al. Reference Nguyen, Schmidt, von Haeseler and Minh2015). Four chains were run for 1 × 107 generations, after which 25% of the sampled trees were discarded as burn-in. The Markov Chain Monte Carlo Algorithms (Larget and Simon Reference Larget and Simon1999) was employed within a Bayesian framework to estimate the posterior probabilities (PP) of phylogenetic trees and to generate a 50% majority-rule consensus tree. Trees were visualized and modified by using the FigTree v. 1.4.3 (Rambaut Reference Rambaut2016) and the Adobe Illustrator CS5.
Results
Steinernema tarimense n. sp.
Description
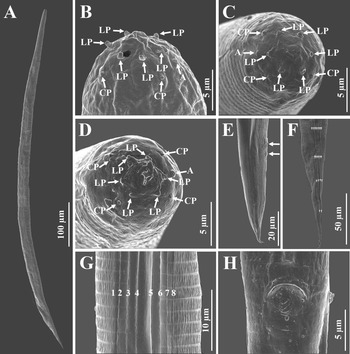
First-generation male
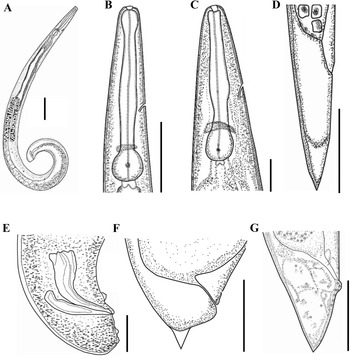
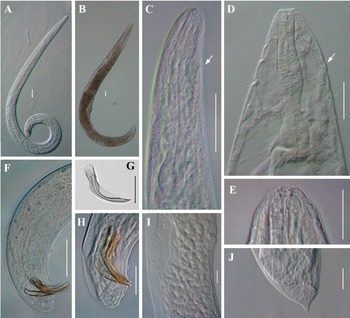
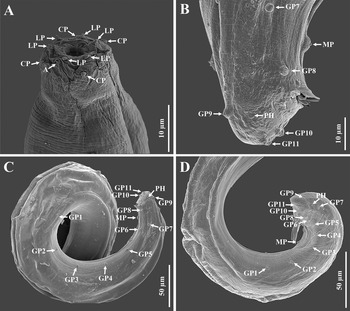
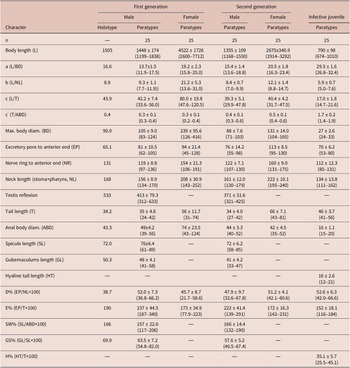
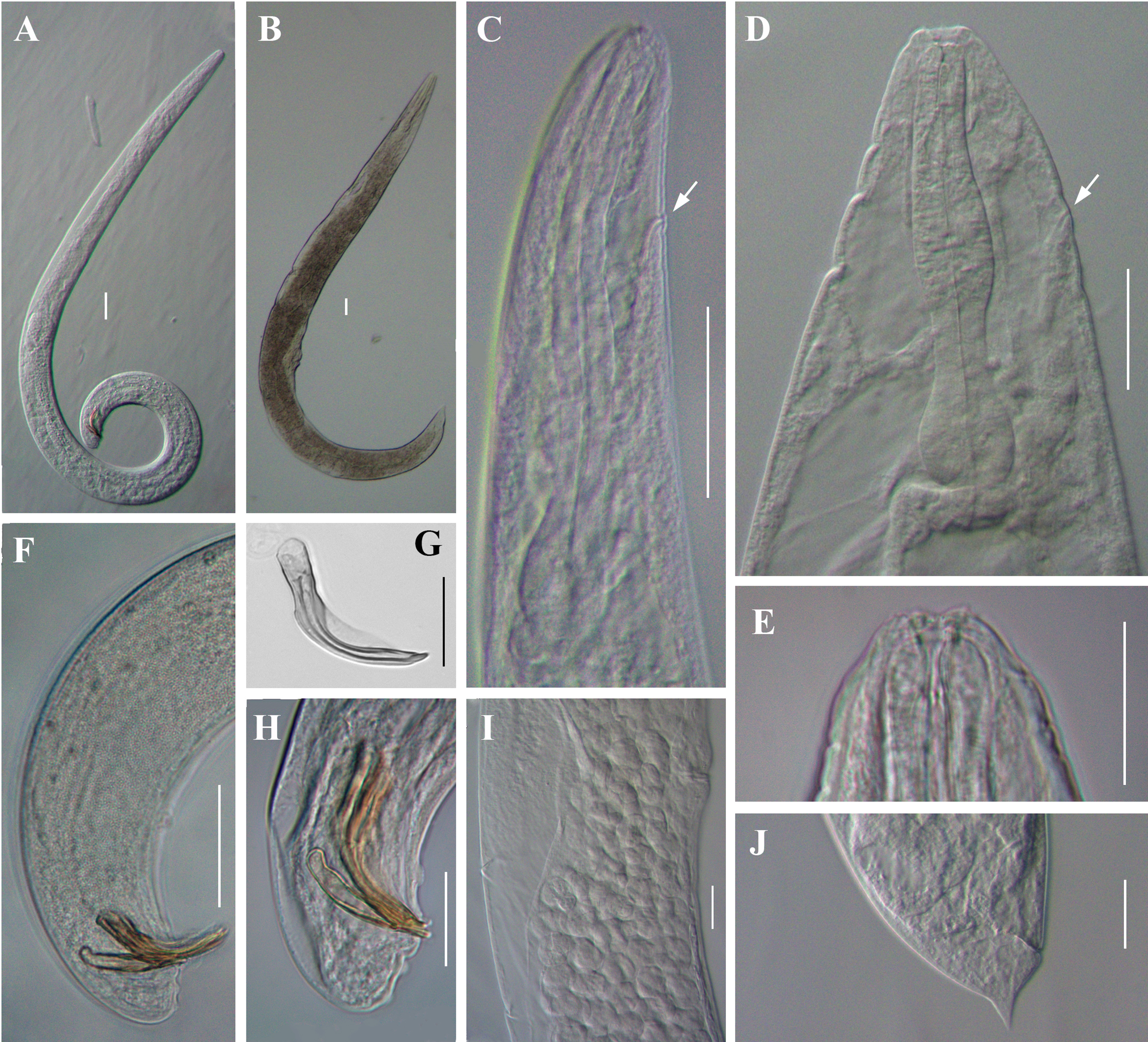
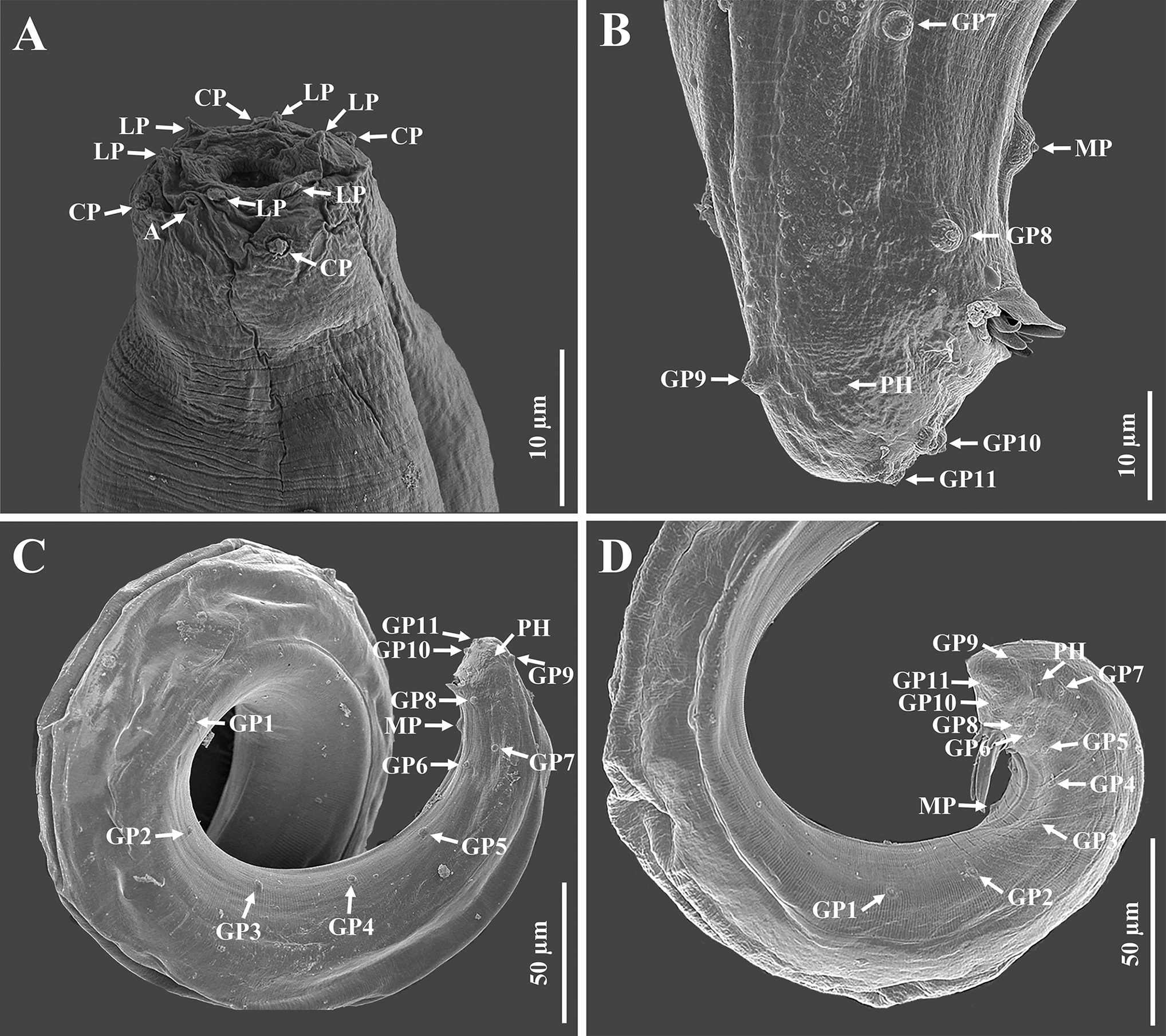
Body slender, ventrally curved posteriorly, strong J-shaped when heat-killed (Figure 1A,Figure 2A). Cuticle annuli appearing slightly visible under SEM (Figure 3A). Lateral fields scarcely marked. Lip region round, continuous with body. Six lips amalgamated, with one acute labial papilla and one low and larger cephalic papilla each, except lateral lips. Amphidial apertures small, located at lateral lips posterior to lateral labial papillae (Figure 3A). Stoma shallow, funnel-shaped, short and wide, with inconspicuous sclerotized walls. Deirids inconspicuous. Pharynx muscular with a cylindrical procorpus, a slightly swollen and non-valvate metacorpus, narrower isthmus and basal bulb spheroid with reduced valves. Nerve ring usually located at anterior part of the basal bulb. Secretory-excretory pore located at level of metacorpus. Cardia prominent, conoid. Intestine tubular without differentiations. Reproductive system monorchic, ventrally reflexed (Figure 1B,Figure 2C). Spicules paired, symmetrical, slightly ventrally curved with manubrium elongated rhomboidal (Figure 1E,Figure 2F), calamus narrower and lamina moderately curved (Figure 2G), slightly arched at anterior part, bearing two longitudinal ribs, and ending in a bluntly pointed terminus, with scarcely developed velum not reaching spicule tip, without rostrum or retinaculum (Figure 2H). Gubernaculum fusiform, with curved and enlarged tip, cuneus pointed, corpus closed posteriorly, about 70% of the length of spicules (Figure 2H). Tail conoid rounded without a fine mucron (Figure 2F, H). Bursa absent. There are 23 genital papillae (GP) (11 pairs and one single) arranged as follows: five pairs subventral precloacal, one pair lateral precloacal, one single mid-ventral papilla precloacal, two pairs sub-ventral ad-cloacal, one pair subdorsal post-cloacal, and two pairs of subventral terminal papillae. Phasmids terminal, located between GP9 and GP10 pairs (Figure 3B-D).
Line drawings of Steinernema tarimense n. sp. A: Entire body of first-generation male; B: Stoma and pharynx region of first-generation male; C: Stoma and pharynx region of first-generation female; D: Posterior end of third-stage infective juvenile; E: Posterior end of second-generation male; F: Posterior end of first-generation female with tail mucron; G: Posterior end of second-generation female. Scale bars: A = 100 μm; B, C, E-G = 50 μm; D = 25 μm.

Light microscope micrographs of first-generation adults of Steinernema tarimensis n. sp. A, B: Entire body of male and female, respectively; C, D: Stoma and pharynx region of male and female, respectively; E: Lip region and stoma of female; F, H: Posterior end of male; G: Male spicule; I: Vulval region of female; J: Posterior end of female. White arrow pointing the excretory pore. Scale bars: A–D, F, I, J = 50 μm; E, G, H = 25 μm.
Scanning electron microscope micrographs of the first-generation male of Steinernema tarimense n. sp. A: Lip region shown in both dorsal and lateral views; B: Tail region; C, D: Posterior part of body. (Abbreviations: A = amphid; CP = cephalic papilla; LP = labial papilla; MP = mid-ventral papilla; PH = phasmid; GP + number = genital papillae)
Second-generation male
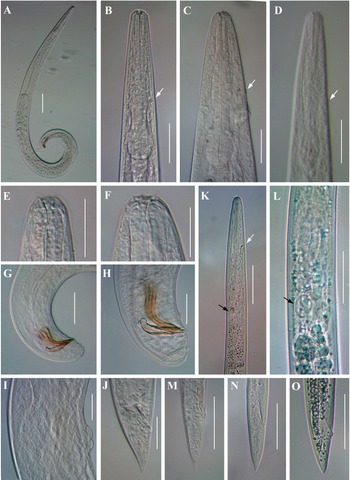
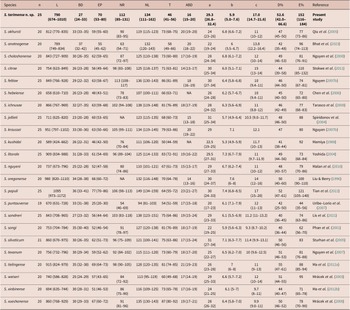
General morphology similar to that of first-generation males (Figure 4B, E), but smaller in size and slenderer (Figure 4A). Tail mucron absent. Spicules ventrally curved, with manubrium rounded, calamus slightly narrower than manubrium (Figure 4G), and lamina ventrally curved at anterior part, lanceolate posterior part with finely rounded tip, reduced ventral velum, and two longitudinal lateral ribs. Gubernaculum slenderer than that of first-generation male, with manubrium ventrad bent, corpus robust, and narrow and slender terminus (Figure 4H). Genital papillae and phasmids with arrangement similar to that in first-generation male.
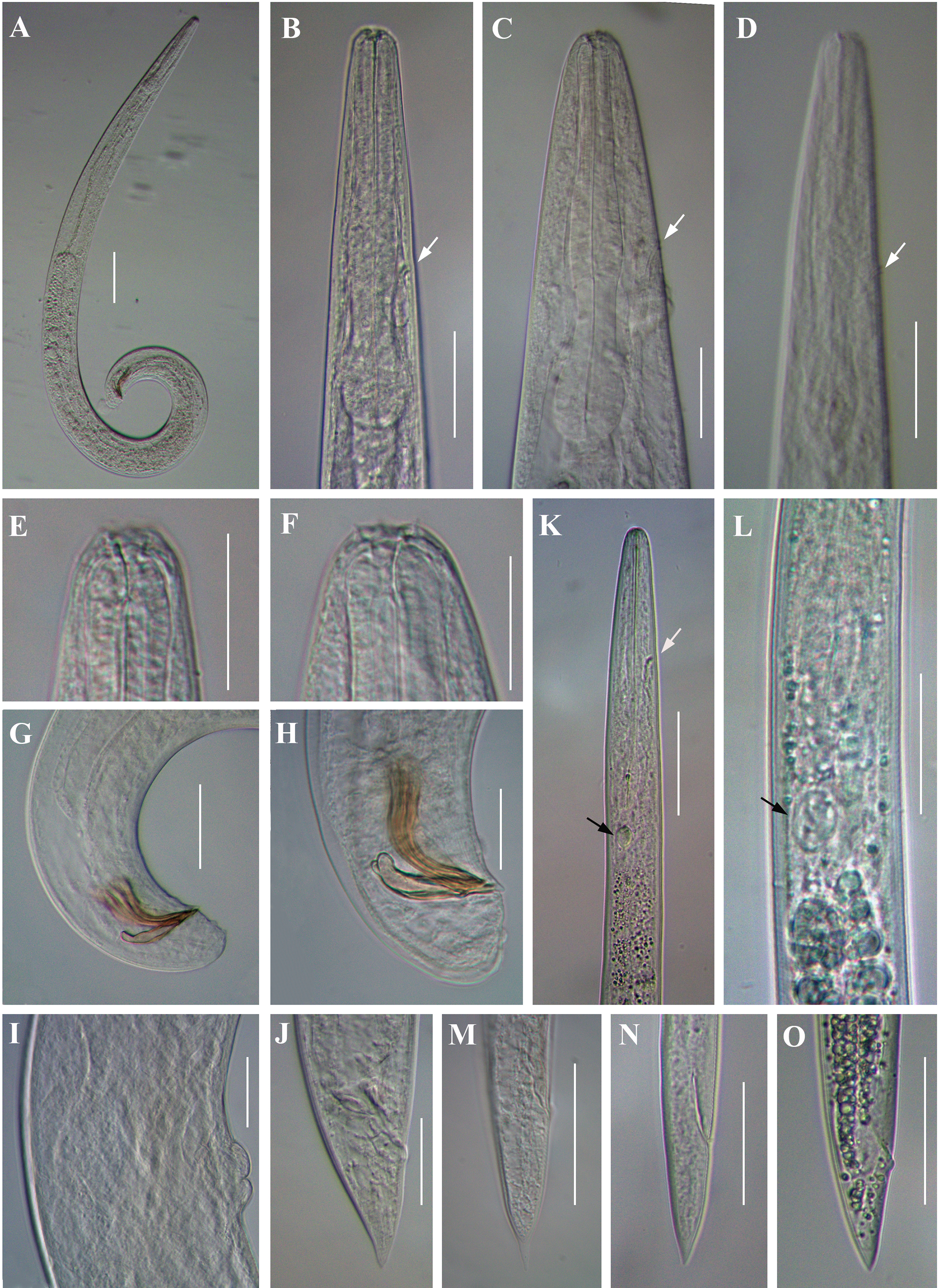
Light microscope micrographs of second-generation adults and infective juvenile (IJ) of Steinernema tarimense n. sp. A: Entire body of male; B–D: Stoma and pharynx region of male female and IJ, respectively; E, F: Stoma and pharynx region of male and female, respectively; G, H: Posterior end of male; I: Vulval region of female; J: Posterior end of female; K: Anterior body of IJ; L: Pharynx and intestine junction region of IJ; M, N: Posterior end of fixed IJ; O: Posterior end of fresh IJ. White arrow pointing the excretory pore, black arrow pointing the bacteria sac. Scale bars: A = 100 μm; B–D, G-K, M–O = 50 μm; E, F, L = 25 μm.
First-generation female
Body C-shaped when heat-relaxed and fixed (Figure 2B). Cuticle with poorly visible annuli. Lateral fields not observed. Deirids inconspicuous, difficult to observe even under SEM. Labial region rounded, continuous with the adjacent part of body (Figure 1C,Figure 2E). Stoma and pharynx region similar to males (Figure 1C). Nerve ring surrounding isthmus, located just anterior to basal bulb. Excretory pore located at level of metacorpus (Figure 2D). Cardia prominent. Reproductive system didelphic, amphidelphic. Ovaries reflexed in dorsal position; oviducts well developed with glandular spermatheca, and uteri tubular with numerous uterine eggs; vagina short, with muscular walls; vulval protruding, in the form of transverse slit located slightly post-equatorial with lips slightly protruding, asymmetrical, without small epiptygmata (Figure 2I). Rectum 0.23 to 0.46 times the body diam., with three rectal glands. Tail conoid, shorter than anal body diam., terminus bearing a minute mucron (Figure 1F, Figure 2J). Phasmids located at anterior part of tail, at 40% to 48% of tail length.
Second-generation female
Similar to first-generation female but smaller. Tail conoid and straight, longer than that of first-generation female (Figure 1G), tapering to a blunt end, lacking mucron (Figure 4J). Excretory pore located at level of metacorpus, similar as that in the first-generation female (Figure 4C). Phasmid located at posterior part of tail, approximately at 60% of tail length
Third-stage infective juvenile
Body slender, straight or slightly curved when heat-killed, tapering gradually from the base of pharynx to the anterior end and from anus to the distal end. Cuticle striated (Figure 5A). appearing well-developed annuli (Figure 5C). Lateral fields begin as a single ridge close to anterior end, increasing to eight ridges unequally spaced, posteriorly gradually reduced to six (anus level) and two (phasmid level) (Figure 5F, G). Lip region truncate, smooth or annulated, continuous or slightly offset from the adjacent part of body, with six lips and prominent six labial and four cephalic papillae (Figure 5B, D). Amphidial apertures rounded and pore-like. Stoma reduced almost closed, with small cheilostom and elongate gymno-stegostom. Pharynx reduced with narrow corpus, lightly swollen metacorpus, slightly narrower isthmus, and elongated pyriform basal bulb with reduced valves. Nerve ring surrounding isthmus. Excretory pore located at level of metacorpus. Hemizonid present, located at anterior of pharynx base. Cardia conoid and small. Deirids inconspicuous (Figure 4D, K). Intestine lumen narrow, bearing a bacterial sac at its anterior part (Figure 4L). Rectum long, almost straight (Figure 1D). Anus distinct (Figure 4M-O, Figure 5H). Genital primordium obscure. Tail conoid, tapering gradually with pointed terminus; hyaline part occupying ca. 35.1% of tail length. Phasmids prominent, located at 65% to 75% of tail length (Figure 5E).
Scanning electron microscope micrographs of infective juveniles of Steinernema tarimense n. sp. A: Entire body; B–D: Lip region; E: Tail in lateral view, showing anus and caudal pailla by arrow, respectively; F: Lateral field at tail region, incisures indicated by arrows; G: Lateral field at mid-body, incisures indicated by number 1–8; H: Anus in ventral view. (Abbreviations: A = amphid; CP = cephalic papilla; LP = labial papilla)

Diagnoses and relationships
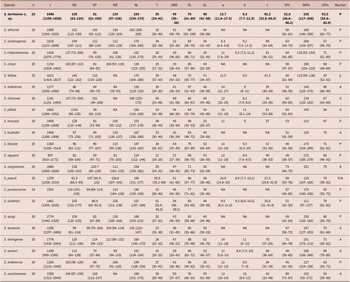
Steinernema tarimense n. sp. adults have short stoma, pharynx robust with rounded basal bulb; males monorchid with ventrally curved spicules, gubernaculum fusiform in the first and second generations, tail conoid slightly ventrally curved, with blunt terminus; females didelphic-amphidelphic with shorter conoid tail bearing a fine mucron in the first generation (31–74 μm, c = 47.6–120.5, c´ = 0.2–0.4, mucron = 3.7–5.0 μm) and longer conoid tail lacking mucron in the second generation (43–81 μm, c = 31.7–47.5, c´ = 0.4–0.6); and IJs with short body (674–1010 μm), poorly developed pharynx (111–162 μm), H% (25.5–45.1), D% (42.0–66.6), and E% (116–184), lateral fields with eight longitudinal ridges, and tail elongated conoid (41–56 μm, c = 14.7–21.6, c´ = 1.4–1.9) (Table 1).
Morphometrics of Steinernema tarimensis n. sp. All measurements are in μm and in the form: mean ± s.d. (range)
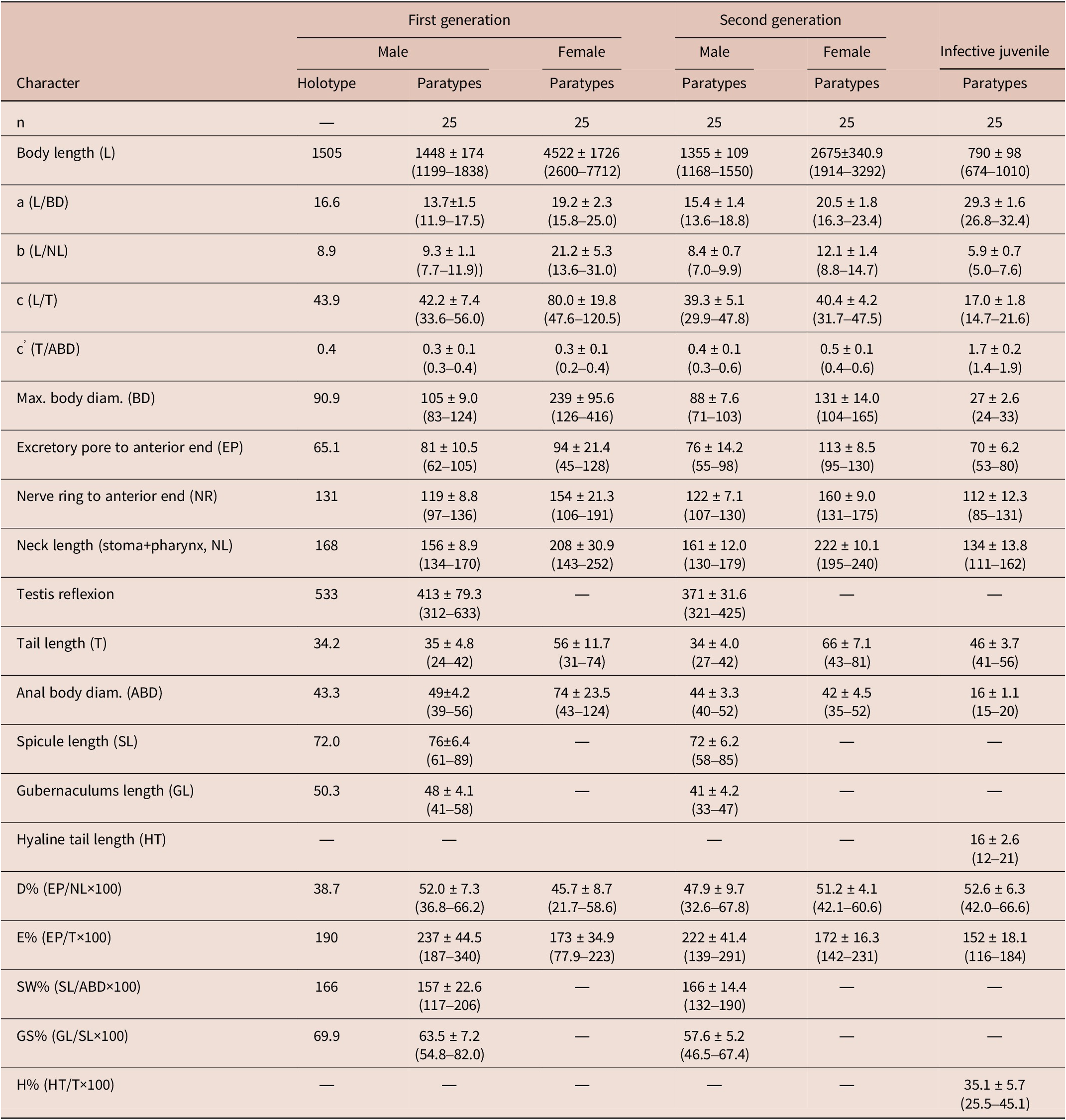
Steinernema tarimense n. sp. belongs to the ‘kushidai-clade’ as defined by Spiridonov and Subbotin (Reference Spiridonov and Subbotin2016). Phylogenetic analysis revealed that S. tarimense n. sp. is sistered to other members of the ‘kushidai-clade’, including S. akhursti, S. anatnagense, S. kushidai, and S. populi. These species are characterized by the infective juvenile (IJ) body lengths ranging from 700 to 1,000 μm (Bhat et al. Reference Bhat, Machado, Abolafia, Askary, Půža, Ruiz-Cuenca, Rana, Sayed and Al-Shuraym2023).
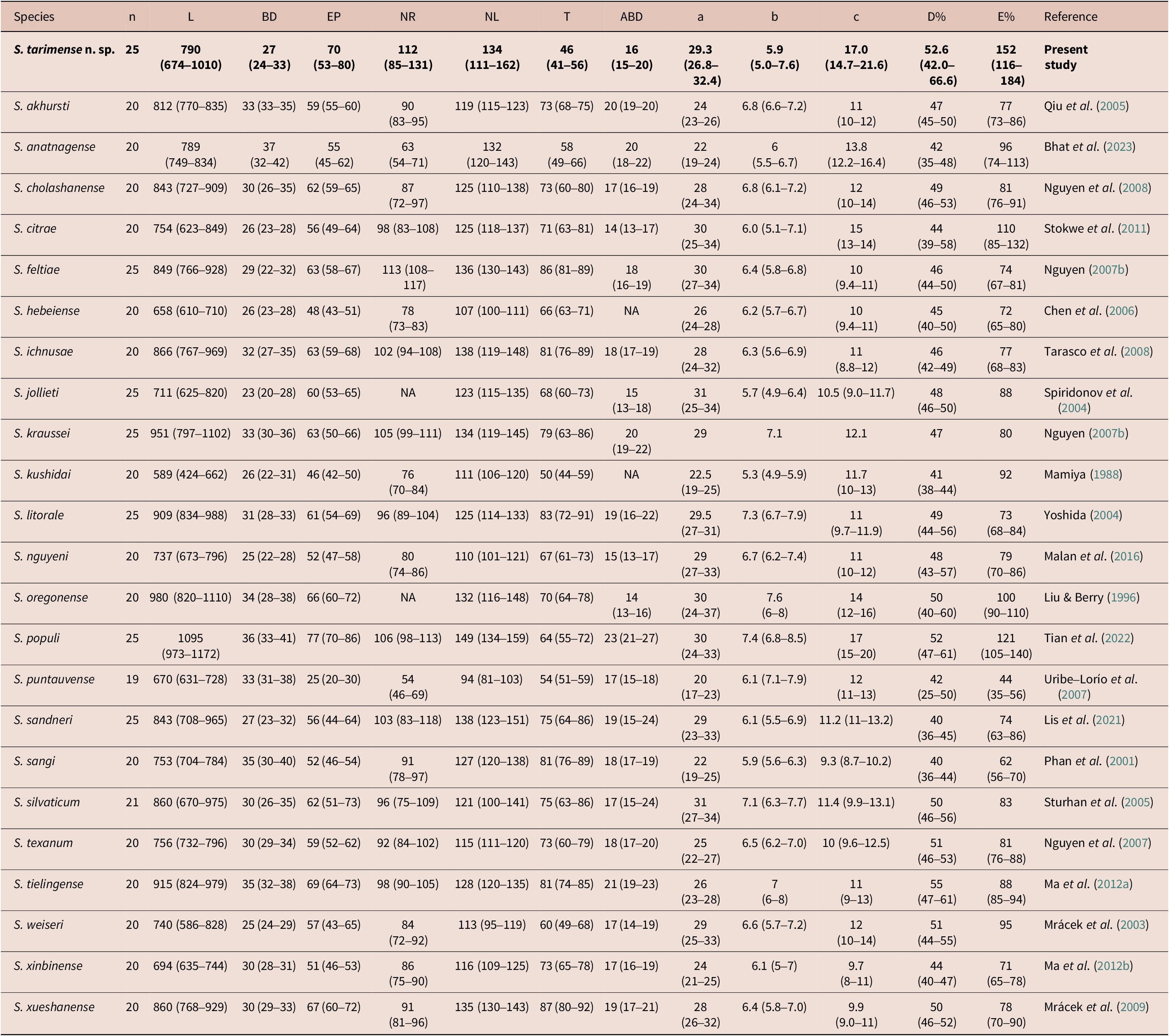
The IJs of S. tarimense n. sp. differ from S. akhursti in body diameter (24–33 μm vs 33–35 μm), tail length (41–56 μm vs 68-75 μm), a ratio (26.8–32.4 vs 23–26), c ratio (14.7–21.6 vs 10–12), D% value (42.0–66.6 vs 45–50), and E% value (116–184 vs 73–86); from S. anatnagense in body diameter (24–33 μm vs 32–42 μm), the distances from anterior end to excretory pore (53–80 μm vs 45–62 μm), to nerve ring (85–131 μm vs 54–71 μm), tail length (41–56 μm vs 49–66 μm), a ratio (26.8–32.4 vs 19–24), c ratio (14.7–21.6 vs 12.2–16.4), D% value (42.0–66.6 vs 35–48), and E% value (116–184 vs 74–113); from S. kushidai in body length (674–1010 μm vs 424–662 μm), the distances from anterior end to excretory pore (53–80 μm vs 42–50 μm) and to nerve ring (85–131 μm vs 70–84 μm), ratio a (26.8–32 vs 19–25), ratio c (14.7–21.6 vs 10–13), D% value (42.0–66.6 vs 38–44), and E% value (116–184 vs 92); and from S. populi in body diameter (24–33 μm vs 33–41 μm), tail length (41–56 μm vs 55–72 μm), and anal body width (15–20 μm vs 21–27 μm).
The first-generation males of S. tarimense n. sp. differ from S. akhursti in distances from anterior end to nerve ring (97–136 μm vs 120–163 μm), and to end of pharynx (134–170 μm vs 168–205 μm), spicule length (61–89 μm vs 85–100 μm), and gubernaculum length (41–58 μm vs 58–68 μm); from S. anatnagense in body diameter (83–124 μm vs 167–211 μm), anal body width (39–56 μm vs 25–36 μm), ratio a (11.9–17.5 vs 6.4–9.8), spicule length (61–89 μm vs 56–70 μm), gubernaculum length (41–58 μm vs 31–43 μm); from S. kushidai in spicule length (61–89 μm vs 48–72 μm); and from S. populi in body diameter (83–124 μm vs 66.3–95 μm), tail length (24–42 μm vs 39.2–68 μm), ratio c (33.6–56.0 vs 19.8–32.9), and D% value (36.8–66.2 vs 59–78).
Moreover, the comparison of IJs for species in the ‘feltiae-kushidai-clade’, which comprises over 20 Steinernema species (Table 2) revealed that S. tarimense n. sp. can be distinguished from all these species, especially S. cholashanense Nguyen, Puza & Mracek, 2008, S. feltiae (Filipjev, 1934) Wouts, Mracek, Gerdin & Bedding, Reference Wouts, Mracek, Gerdin and Bedding1982, and S. oregonense Liu & Berry, Reference Liu and Berry1996 by the smaller body length, longer distance of excretory pore to anterior end, shorter tail length, as well as the lower ratio b value, higher values in ratio c, D%, and E%. The comparisons of the first-generation males (Table 3) also revealed that the new species differs from S. cholashanense, S. feltiae, and S. oregonense by the longer spicules, lower D% and higher SW% values.
Comparison of morphometrics of infective juveniles of Steinernema tarimense n. sp. with other members of ‘feltiae-kushidai-clade’. Measurements are in μm except n, ratio and percentage. Data for new species is in bold
Comparison of morphometrics of the first-generation males of Steinernema tarimense n. sp. with other members of ‘feltiae-kushidai-clade’. Measurements are in μm except n, ratio and percentage. Data for new species is in bold
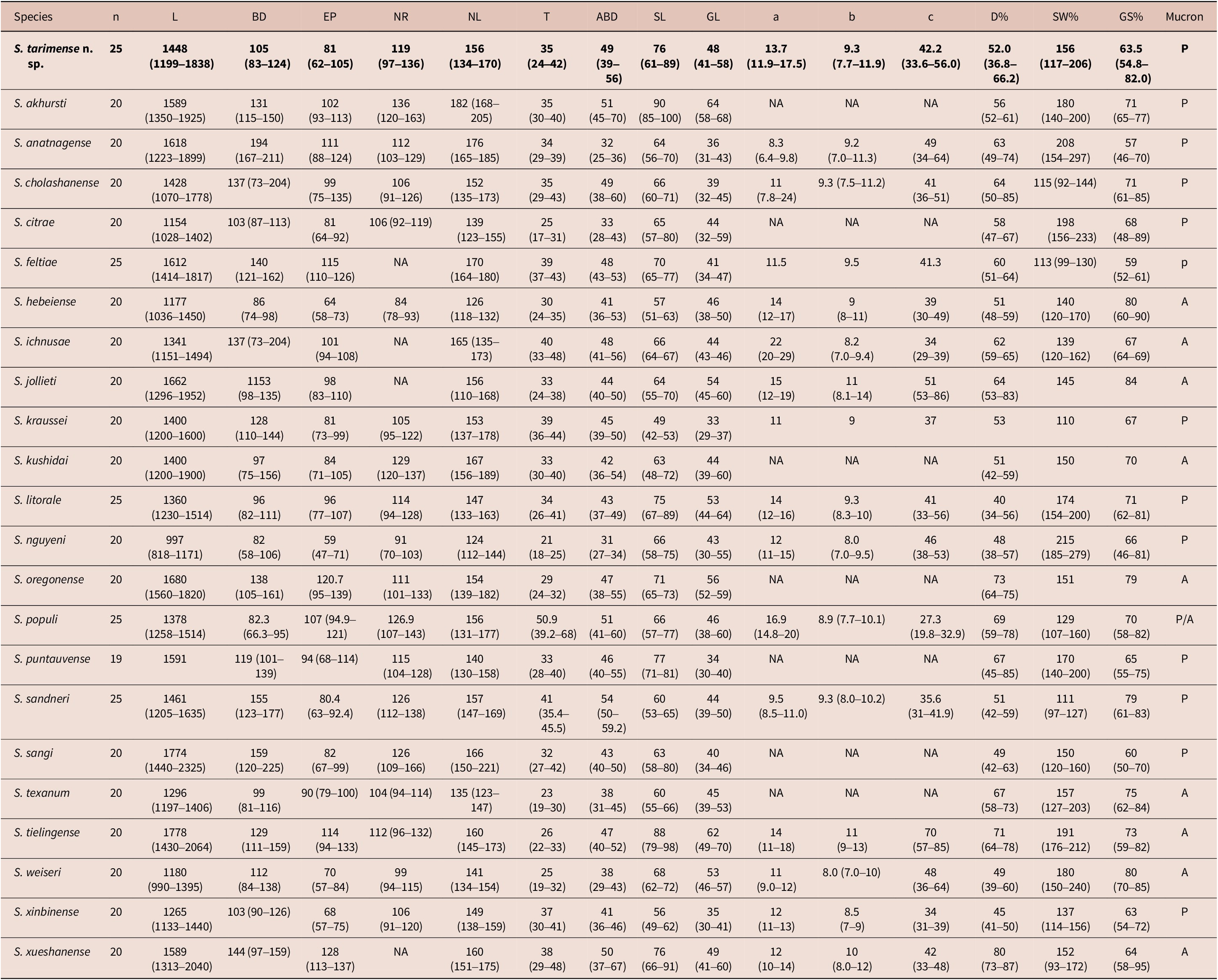
Note: NA = Not available; P = Present; A = Absent.
Life cycle
Steinernema tarimense n. sp. demonstrated effective infestation and development in G. mellonella larvae (Figure 6), exhibiting significantly faster development rates compared to several congeneric species. At 25°C, S. tarimense n. sp. completed its life cycle more rapidly than S. cholashanense, S. feltiae, S. litorale, and S. populi. When infected with 100 IJs of S. tarimense n. sp., G. mellonella larvae succumbed within 2–3 days, compared to 5–6 days for the other species examined. The first- and second-generation adults of S. tarimense n. sp. appeared in host cadavers within 2–3 days and 5–6 days post-infection, respectively, while the comparative species required 8–9 days and 11–12 days for the same developmental stages. Notably, the IJs of S. tarimense n. sp. emerged from cadavers with 5–6 days after initial infection, whereas the other species showed emergence times of 8–12 days.
Infection of Steinernema species to Galleria mellonella larvae. A: Cadavers of G. mellonella larvae infested with S. populi and S. tarimense n. sp. isolate Z32, respectively; B: Third-stage infective juveniles of S. tarimense n. sp. migrated out from cadavers.

Type host and locality
The type hosts are unknown as the nematodes of this genus can be hosted by different insect species (Fallet et al. Reference Fallet, Gianni, de Machado, Bruno, Bernal, Karangwa, Kajuga, Waweru, Bazagwira and Degen2022; Kajuga et al. Reference Kajuga, Hategekimana, Yan, Waweru, Li, Li, Yin, Cao, Karanja and Umulisa2018; Yan et al. Reference Yan, Waweru, Qiu, Hategekimana, Kajuga, Li, Edgington, Umulisa, Han and Toepfer2016). Specimens of S. tarimense n. sp. were isolated from mixed soil samples collected in a Populus euphratica forest located along the Tarim River (Yuli County, Xinjiang, China) using the Galleria mellonella baiting technique (Bedding and Akhurst Reference Bedding and Akhurst1975; White Reference White1927). The geographic coordinates of the sampling site for isolate R31 is 41°3’44.3592”N and 86°7’2.0279”E, for isolate R39 is 41°4’20.37”N and 86°7’9.6312”E, and for isolate Z32 is 41°4’25.0824”N and 86°7’3.3708”E.
Type material
Isolate Z32 was designated as the type material for Steinernema tarimense n. sp. Three slides of each stage, including first-generation adults (males and females), second-generation adults (males and females), and IJs, were deposited in the US Department of Agriculture Nematode Collection (USDA NC), Beltsville, Maryland, USA. Holotype: one male of F1, USDANC accession numbers is T-811t. Paratype: males of F1, with USDANC numbers from T-8097p to T-8099p; males of F2, with numbers from T-8103p to T-8105p; females of F1, with numbers from T-8106p to T-8108p; females of F2, with numbers from T-8109p to T-8110p; IJs, with numbers from T-8100p to T-8013p.
Many males and females of the first generation and several IJs were deposited in the National Parasitic Resources Center (NPRC-2019-194-30) of China, with accession No. CSTR:15507.06. YZ1CR1001KpLC.
Numerous males and females of the first generation, along with several IJs, were deposited in the Institute of Microbiology, Xinjiang Academy of Agricultural Sciences, China.
Etymology
The species epithet refers to the region where the species was recovered (Tarim Basin in Xinjiang, China).
Nematode molecular characterization
The ITS sequences of Steinernema tarimense n. sp. isolates R31, R39, and Z32 were identical in length of 1019 bp, with GenBank accession number PQ590687, PQ590686, and PQ590685, respectively (Table 4). The sequence similarity of three isolates is 100%. Comparative sequence analysis revealed that the ITS region of S. tarimense n. sp. exhibited 76–124 nucleotide differences relative to related species, corresponding to sequence similarity values ranging from 84.50% to 92.09% (Supplementary table 1). The sequence similarity of the new species to S. anantnagense (OQ407501) is 92.09%; to S. akhursti (DQ375757), 91.40%; to S. kushidai (AB243440), 90.17%; to S. cholashanense (EF431959), 84.99%; and to S. populi (MZ367622), 84.50%.
Molecular information for nematode species used in phylogenetic analyses. Data of the new species are shown in bold
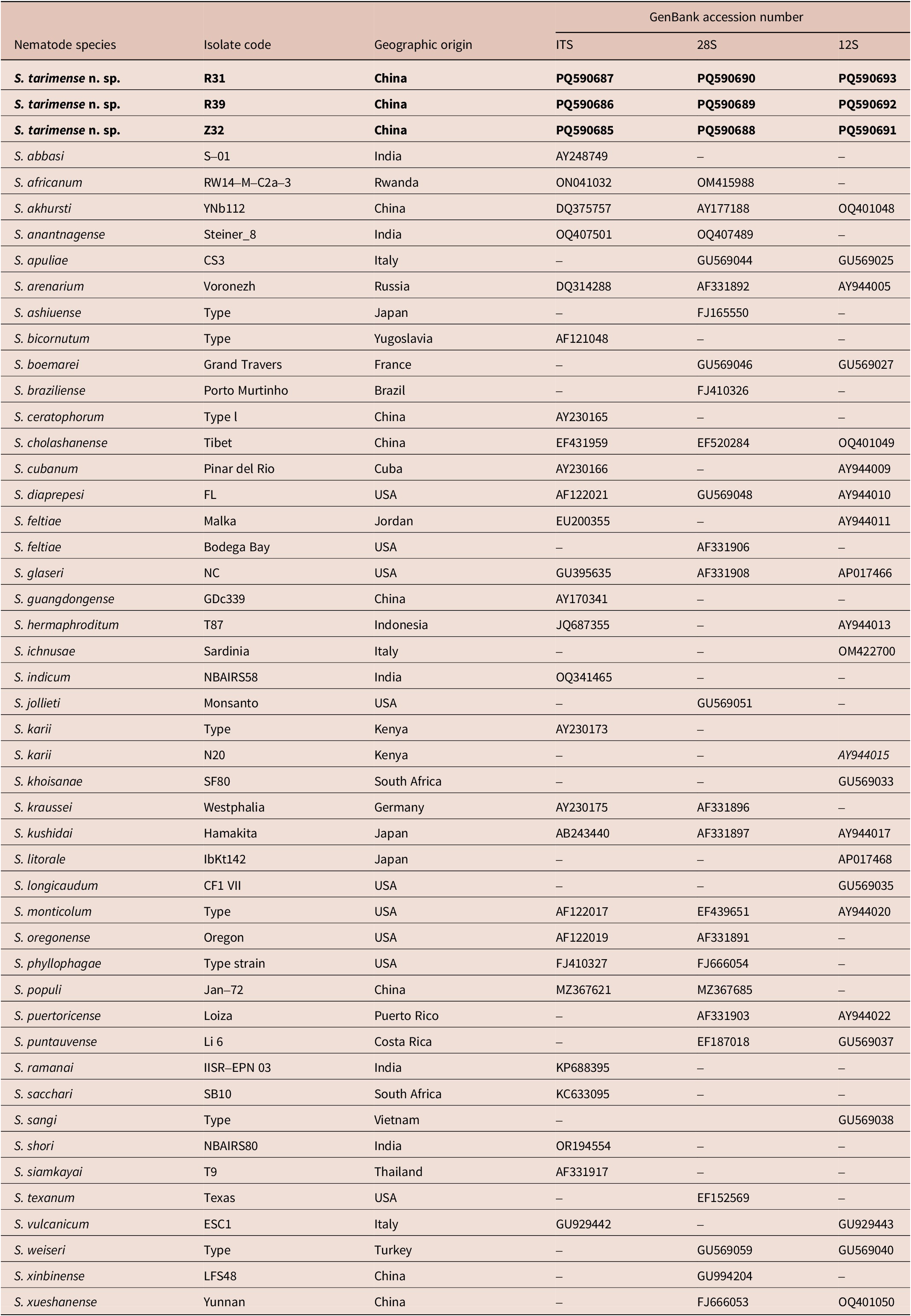
Note: “–” indicates data unavailable.
The 28S sequences of isolates R31, R39, and Z32 were identical in length of 908 bp, with GenBank No. PQ590690, PQ590689, and PQ590688, respectively (Table 4). Three sequences of the new species showed 100% similarity, while exhibiting 8–20 nucleotide differences from related species, corresponding to sequence similarities of 97.74–98.70% (Supplementary table 2). The sequence similarity of the new species to S. akhursti (DQ375757) is 98.47%; to S. kushidai (AB243440), 97.91%; and to S. populi (MZ367622), 97.74%.
The sequences of mt12S gene of isolates R31, R39, and Z32 were identical in length of 546 bp, with GenBank No. PQ590693, PQ590692, and PQ590691, respectively (Table 4). The three sequences of the new species were identical to each other but exhibited significant divergence from related species, with 33–46 nucleotide differences corresponding to sequence similarities of 90.02–91.49% (Supplementary table 3). The sequence similarity of the new species to S. akhursti (OQ401048) is 91.49%; to S. vulcanicum (GU929443), 91.27%; and to S. kushidai (AY944017), 90.02%.
Although S. tarimense n. sp. shared relatively low similarity in the ITS, 28S, and mt12S sequences with all other known Steinernema species, the comparative sequences analysis revealed that the new species is most similar to S. akhursti, S. anantnagense, and S. kushidai, supporting its designation as a novel taxonomic entity within the ‘kushidai-clade’.
Nematode phylogenetic relationships
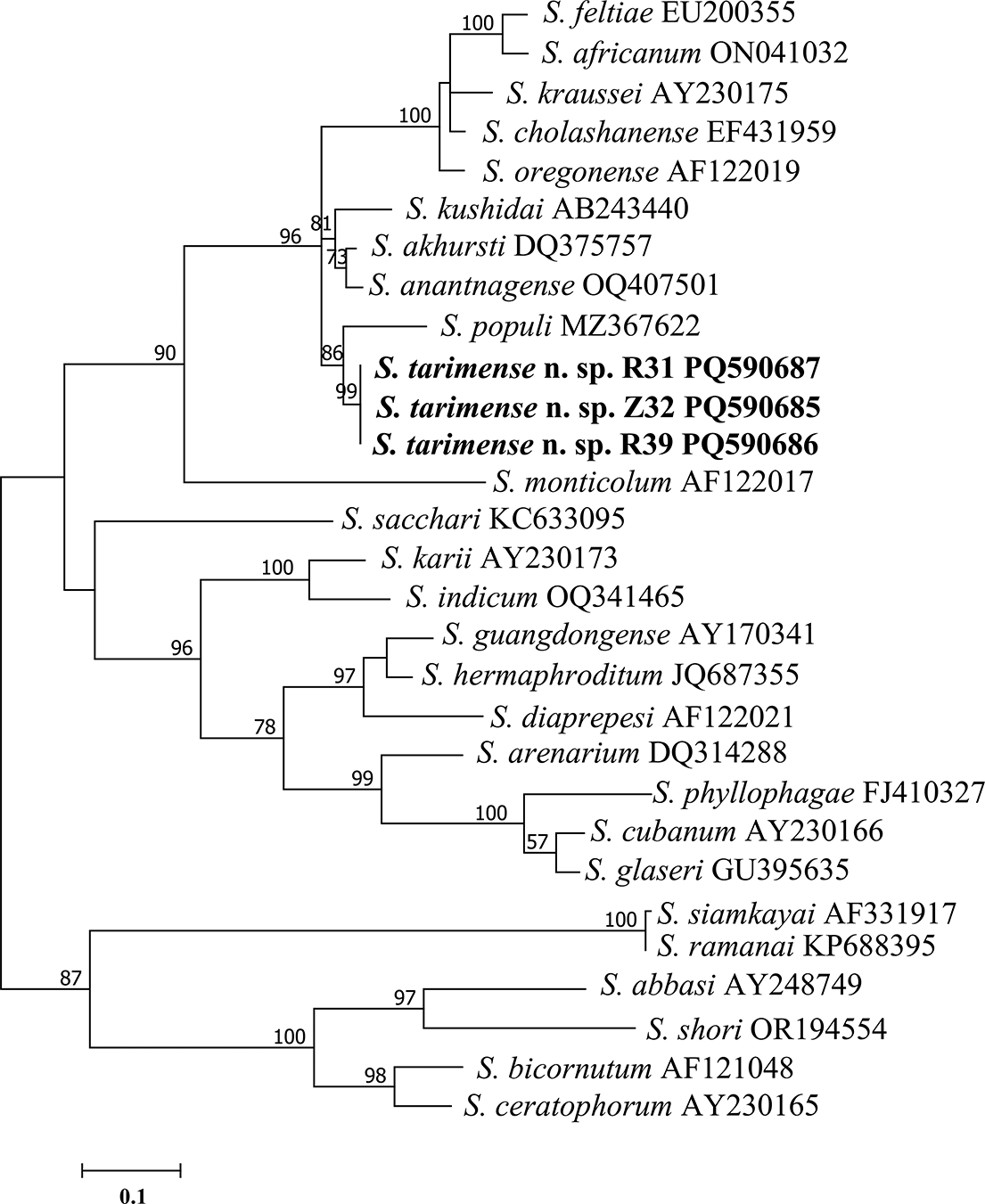
For the ITS sequences, the Bayesian inference (BI) analysis revealed the alignment comprising 1335 nucleotide sites, including 364 constant, 735 parsimony-informative, and 248 singleton sites. The phylogenetic relationships among 27 Steinernema species are shown in Figure 7. Three S. tarimense n. sp. isolates grouped with S. populi in one branch, which further grouped with the branch consisting of S. akhursti, S. anantnagense and S. kushidai and the branch of ‘feltiae-clade’ species including S. africanum, S. cholashanense, S. feltiae, S. kraussei, and S. oregonense, into a higher statistically supported ‘feltiae-kushidai-clade’ (PP = 96).
Bayesian 50% majority-rule consensus tree was inferred from the ITS rRNA sequences of Steinernema tarimense n. sp., utilizing the GTR + F + G4 model. Bayesian posterior probabilities (PPs) exceeding 50% are indicated for relevant clades. The scale bar represents the number of nucleotide substitutions per site. The new species is indicated in bold.
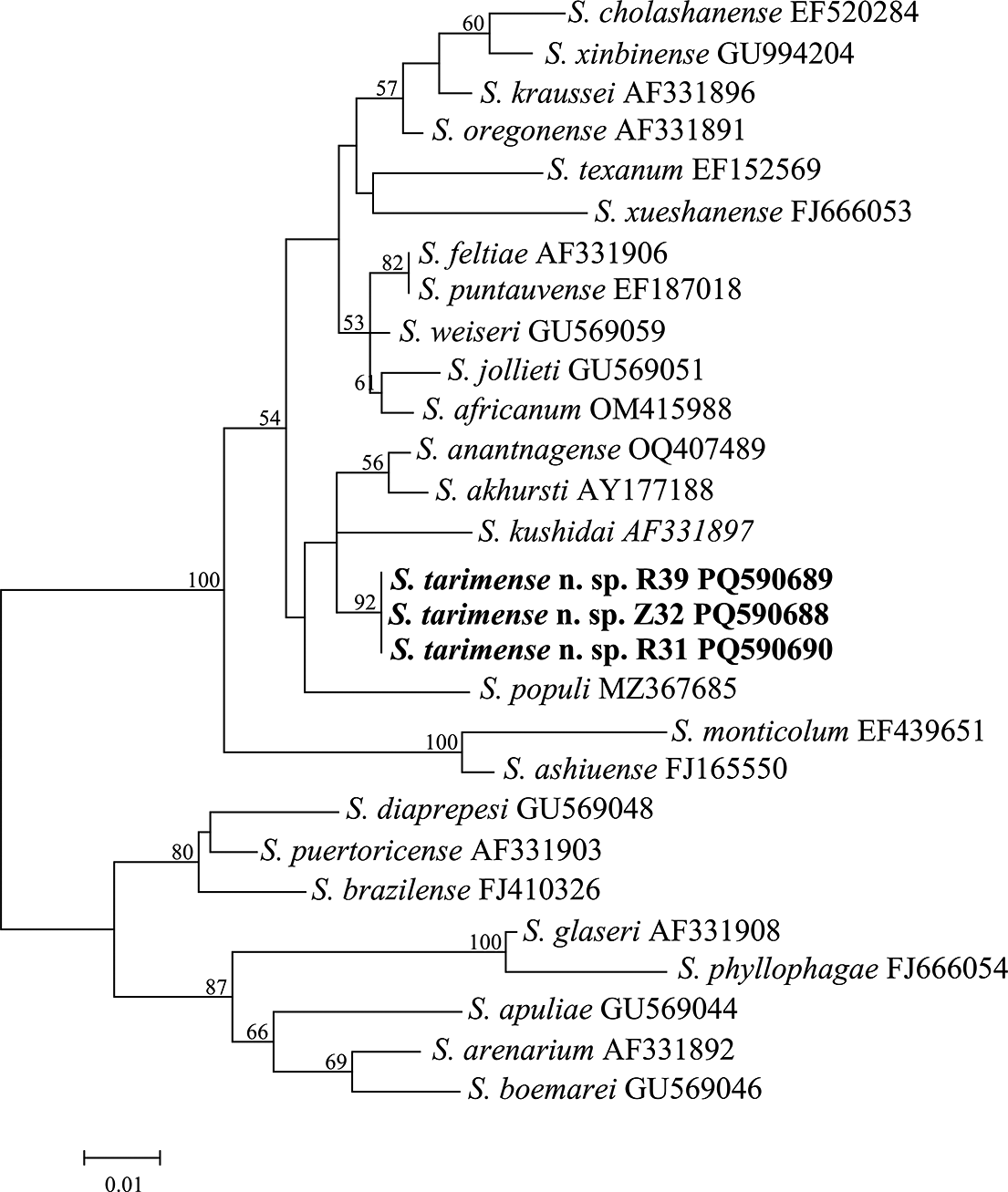
For the 28S sequences, the BI analysis revealed the alignment comprising 1030 nucleotide sites, including 583 constant, 267 parsimony-informative, and 180 singleton sites. The phylogenetic relationships among 26 Steinernema species are illustrated in Figure 8. Three S. tarimense n. sp. isolates grouped with the ‘kushidai-clade’ members including S. akhursti, S. anantnagense, S. kushidai, and S. populi in a lower supported clade (PP < 50). This clade is further grouped with the members of the ‘feltiae-clade’ and ‘monticolum-clade’ into a superclade with a highest support (PP = 100).
Bayesian 50% majority-rule consensus tree was inferred from the D2D3 regions of 28S rRNA of Steinernema tarimense n. sp., utilizing the GTR + F + I + G4 model. Bayesian posterior probabilities (PPs) exceeding 50% are indicated for the relevant clades. The scale bar represents the number of nucleotide substitutions per site. The new species is indicated in bold.
For the mt12S sequences, the BI analysis revealed the alignment encompassing 558 nucleotide sites, including 278 constant, 211 parsimony-informative, and 69 singleton sites. The phylogenetic relationships among 24 Steinernema species are illustrated in Figure 9. Three S. tarimense n. sp. isolates also grouped with the ‘kushidai-clade’ members S. akhursti and S. kushidai in a lower supported clade (PP < 50). All phylogenetic analyses based on ITS, 28S, and mt12S sequences demonstrated that three isolates of S. tarimense n. sp. are conspecific and closed to members in the ‘kushidai-clade’.
Bayesian 50% majority-rule consensus tree was inferred for the mt12S gene of Steinernema tarimense n. sp. using the GTR + F + I + G4 model. Bayesian posterior probabilities (PPs) exceeding 50% are provided for the relevant clades. The scale bar represents the number of nucleotide substitutions per site. The new species is indicated in bold.

Discussion
Entomopathogenic nematodes (EPNs) have obtained increasing attention as effective biopesticides for insect pest control. While the diversity of EPNs in China remains incompletely characterized, at least eighteen steinernematids have been originally described from various regions, including Hebei, Jilin, and Yunnan provinces (Chen et al. Reference Chen, Li, Yan, Spiridonov and Moens2006; Ma et al. 2012 a, b; Qiu et al. Reference Qiu, Hu, Zhou, Mei, Nguyen and Pang2005; Tian et al. Reference Tian, Zhu, Li, Zhang, Půža, Shapiro-Ilan, Zhao, Liu, Zhou, Ding, Wang, Ma, Zhu, Li and Li2022). A survey conducted in Xinjiang during 2020 to 2024 has recovered more than 20 EPN isolates, among which three morphologically and genetically identical isolates were identified as a new species – Steinernema tarimense n. sp. This species was collected from Populus euphratica riparian forests in the Tarim Basin, though its natural insect host remains undetermined. Notably, Xinjiang covers approximately 1.6 million km2 (one-sixth of China’s total territory), making this discovery particularly significant. As the first EPN species described from this vast region, S. tarimense n. sp. represents an important addition to our understanding of EPN biodiversity and biogeography in China.
Although S. tarimense n. sp. exhibited relatively low similarity in ITS, 28S, and mt12S sequences compared to other known Steinernema species (Table 4), phylogenetic analyses consistently placed it within an evolutionary clade containing the ‘kushidai-clade’ members (S. akhursti, S. anantnagense, S. kushidai, and S. populi). These taxa collectively formed a well-supported ‘feltiae-kushidai-clade’ with members of the ‘feltiae-clade’. Morphological diagnosis clearly distinguished S. tarimense n. sp. from its closest phylogenetic relatives in the ‘kushidai-clade’. Comparative morphometrics of both infective juveniles (Table 2) and first-generation males (Table 3) further confirmed its distinctiveness from other members of the ‘feltiae-kushidai-clade’. The combined evidence from comprehensive morphological characterization, detailed morphometric analyses, and robust phylogenetic reconstruction unequivocally supports the recognition of S. tarimense as a novel species within the genus Steinernema.
Steinernema tarimense n. sp. presented several distinctive biological characters compared to congeners. Notably, G. mellonella larvae infested with S. tarimense n. sp. displayed light black or nearly colorless cadavers, contrasting sharply with the reddish coloration induced by S. populi (Figure 6A). The new species demonstrates accelerated developmental rates. Infective juveniles (IJs) and other developmental stages emerged in water within 5–6 days post-infestation (Figure 6B), compared to 10–15 days for S. populi (Tian et al. Reference Tian, Zhu, Li, Zhang, Půža, Shapiro-Ilan, Zhao, Liu, Zhou, Ding, Wang, Ma, Zhu, Li and Li2022). First-generation adults appeared in cadavers within 2–3 days, followed by second-generation adults at 5–6 days, whereas S. akhursti required 3–4 days and 7–8 days, respectively (Qiu et al. Reference Qiu, Hu, Zhou, Mei, Nguyen and Pang2005). The unique biological traits observed in the new species likely represent evolutionary adaptations responsed to the extreme environment of its native habitat – the Populus euphratica forests along the Tarim River. This region is characterized by aridity, low precipitation, nutrient-poor soils, and limited insect host availability. To survive under these environmental constraints, S. tarimense n. sp. appears to have evolved a suite of adaptive traits through natural selections: 1) rapid development by optimizing reproduction within narrow temporal windows; 2) high biomass conversion efficiency by utilizing all host tissues except the cuticle; and 3) ecological opportunism by maximizing population persistence despite resource limitations. These traits collectively enhanced the fitness of S. tarimense n. sp. in an ecosystem where parasitic opportunities were ephemeral and unpredictable.
Conclusions
Integrated morphological and molecular analyses confirmed Steinernema tarimense n. sp. as a new taxon within the genus. The species demonstrates remarkable thermobiological efficiency, with its infective juveniles completing development at 25°C significantly faster than related species (S. cholashanense, S. feltiae, S. litorale, and S. populi). As the first EPN species described from Xinjiang region, this discovery expands the known biogeographic distribution of Steinernema in Asia, represents a valuable indigenous resource for biological control programs in arid ecosystems, and provides novel genetic material for studying thermal adaptation in EPNs.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0022149X25100448.
Acknowledgements
This research was supported by the National Natural Science Foundation of China (Grant No. 32160377), the Public Welfare Project of Xinjiang Uygur Autonomous Region (No. KY2024024), and the Project of Fund for Stable Support to Agricultural Sci-Tech Renovation (xjnkywdzc-2023004-3). We thank the Team of Beneficial Crop Microorganisms at the Institute of Microbiology, Xinjiang Academy of Agricultural Sciences, for their assistance in collecting, isolating, and preserving soil samples. Additionally, special gratitude goes to Dr. Xue Qing from the Department of Plant Pathology of Nanjing Agricultural University for his helpful supports.
Competing interests
None.
Ethical standard
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional guides on the care and use of laboratory animals.