Introduction
Scientific advances to fight the world’s infectious diseases in recent decades have been nothing short of breathtaking. Cutting edge treatments attack complex viruses at multiple sites. Diagnoses that previously required advanced laboratories can be done with a swab from a local pharmacy. Instead of live virus and using chicken eggs, mRNA vaccines now instruct cells to make proteins that will trigger antibodies.
International law and global governance systems have sought, and often failed, to keep pace with science — to secure equity in global access and effective use of technologies to stop diseases, including those spreading as pandemics. One model from the AIDS response has enabled the global sharing of scientific know-how and production, at scale, to advance quality, affordable access. Born out of the tragedy of this century’s deadliest pandemic and secured through the work of a powerful transnational social movement, this model reflected a remarkable shift in international law and global governance. Yet this shift has remained incomplete and contested. In the last decade, the fracturing of the global health governance space for infectious disease has given rise to competing paradigms about how, and when, to use the power of law to secure timely and equitable access.
This article traces global health law in this area — from the emergence of the first truly global legal paradigm for equitable access through its confrontations, transfer to other diseases, and near-complete circumvention during COVID-19. Global law and governance institutions eventually saved millions of lives but failed during the most recent pandemics. The question for the field, then, is what can be rescued from the “AIDS model” and whether innovation can build a new paradigm to meet this moment of global health turmoil.
The “AIDS Model” of Ensuring Access to Medicines in a Pandemic
As Gostin writes in Global Health Law, “It is impossible to overstate the importance of new institutions in mobilizing resources and sharing science…”Reference Gostin1 in the global AIDS response — not just for HIV but for global health writ large. Among these were law and policy rules that structure access to antiretroviral medicines (ARVs).
Immediately after the development of effective ARVs to treat HIV, multilateralism and global governance failed. Deaths from AIDS plummeted in the United States and Europe — dropping 47% in the first year in the US.2 But in global South countries in Africa, Asia, and Latin America deaths from AIDS continued to increase because the medicines / ARVs saving lives were both too expensive and largely unavailable.3 An estimated 12 million people living with HIV died on the African continent alone, waiting for access in the decade after treatment became available.Reference Nkengasong4
Eventually a powerful movement of people living with HIV, activists, lawyers, researchers, scientists, clinicians, and allies in governments of the global South came together to force change.Reference Mbali5 Through lobbying, protest, strategic litigation, and negotiation, the international movement cast the issue of high prices and abuse of patent monopolies in ethical terms too, and mobilized the political and legal will to act.6
The model built by international cooperation that followed had 4 key law and governance elements: (1) use of law by national governments to compel sharing, (2) decentralized generic manufacturing, (3) mechanisms for voluntary sharing of patents and technology transfer, and (4) substantial international funding. These were synergistic and, in combination, created a remarkable new ecosystem: prices on medicines fell by more than 99 percent, factories in Southern countries produced for millions, and today 3/4 of all people living with HIV are accessing lifesaving and HIV-preventing ARVs.7
The first element reflected an important departure from maximalist intellectual property (IP) rules. The Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS), part of the treaties establishing the WTO in 1994, required many countries to begin offering patents on medicines for the first time. The agreement had largely been driven by the commercial interests of industrialized nations and their companies, with little consideration for public health.Reference Sell8 The result for medicines was the problematic international harmonization of patent norms, creating barriers against generic production of AIDS drugs.Reference Correa9 Reframing this as a rights violation, activists and governments of the global South successfully challenged the WTO and, in 2001, the WTO’s Doha Declaration pronounced TRIPS “can and should be interpreted and implemented in a manner supportive of WTO members’ right to protect public health and, in particular, to promote access to medicines for all.”10 The declaration established several “flexibilities” and removed the obligation for least-developed countries to comply with TRIPS rules on patents until 2016 (since continued).
Shortly after the Doha Declaration, certain Southern countries led a surge in compulsory licensing, albeit with time-consuming difficulties, and opposition.Reference ’t Hoen11
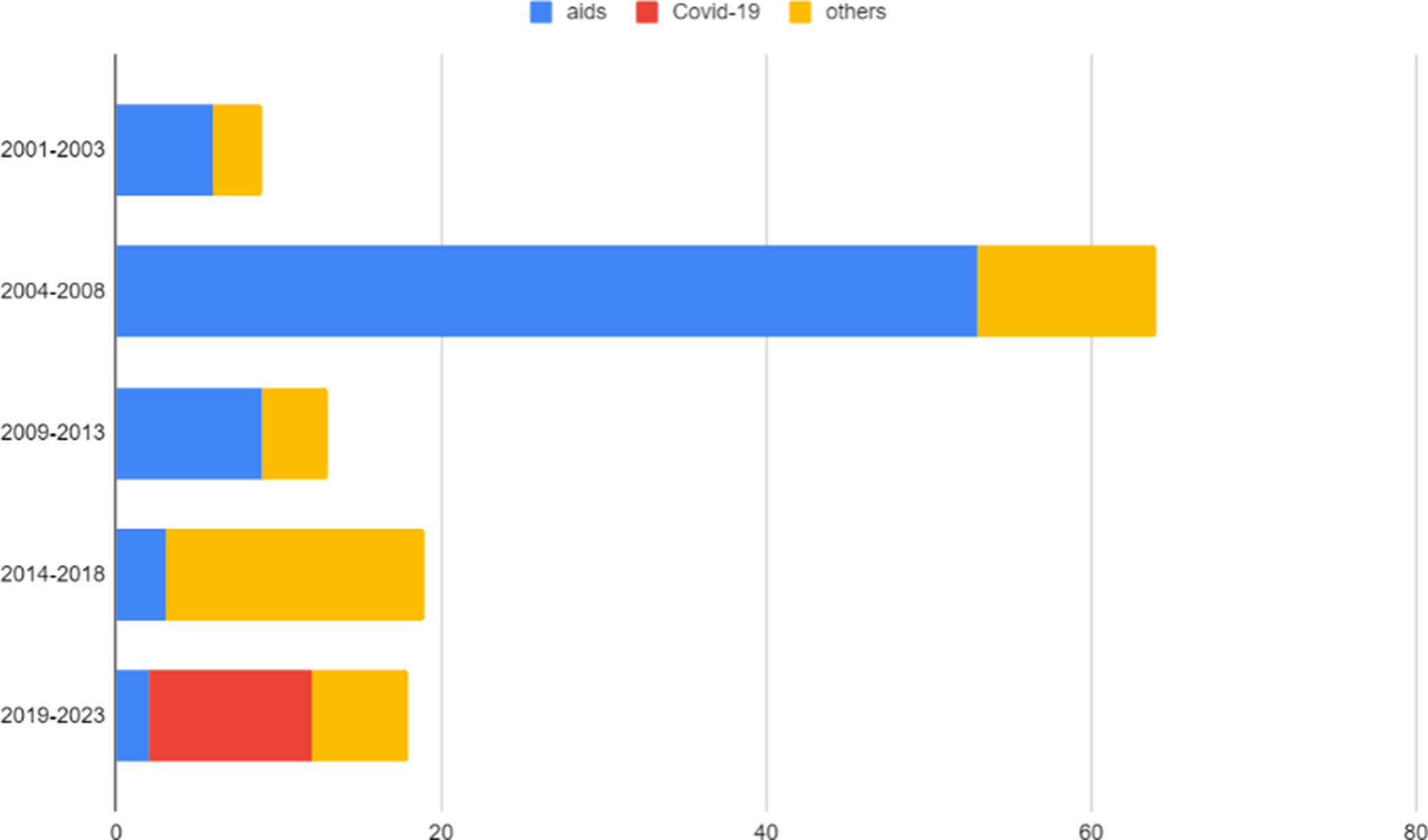
Figures 1 and 2 show the number of compulsory license requests since the Doha Declaration was adopted. From 2004–2008, there were 64 compulsory license requests mostly involving AIDS medicines (53), with the vast majority led by governments (44). Almost all took place in the South. More than 80 percent of these requests concluded in the granting of the request and others leveraged drug price reductions.
Compulsory Licenses by Diseases (2001-2023)
Source: own elaboration based on data from the Medicines Law and Policy TRIPS Flexibilities Database 12
Compulsory licensing by requestor (2001-2023)

Brazil, Thailand, and many other governments used the threat and at times issuance of compulsory licenses to build ARV programs by producing or procuring from generic makers.Reference ’t Hoen13 In South Africa AIDS advocates won a victory over drug companies to remove price and patent monopolies over first line ARV production in the Hazel Tau anti-trust case, and that settlement resulted in a long-running precedent that HIV drug makers should license for generic production where affordability is a barrier.Reference Beresford14
These actions opened the door to the second key element of the “AIDS model” — a robust global ecosystem of ARV producers in India, South Africa, Uganda, Brazil, Thailand, and beyond. That includes government producers like Thailand’s GPO, parastatals like Brazil’s Fiocruz, and many private producers, like India’s Cipla, which produced some of the first generic ARVs and showed low-cost, high-quality production was possible.Reference Menghaney15 This ecosystem was nurtured by substantial public support, including strategic regulatory shaping and capital for private sector producers.Reference Vella16
These elements, alongside movement political mobilization, built pressure for a third element, as companies provided voluntarily licenses and technology transfer — such that originators control the high-income markets, but Southern producers supply the South. Initially this came in the form of bilateral licenses by ARV originators to a handful of companies, largely in India. Later the Medicines Patent Pool (MPP) was created with support from UNITAID to negotiate global licenses for HIV medicines. Today it has agreements with nearly all the ARV patent-holding companies, though middle-income countries (as defined by the World Bank) are often left out and must rely on other recourses or compulsory measures.
The final piece of the puzzle has been robust international solidarity through funding of access to ARVs through major institutions like the multilateral Global Fund to Fight AIDS, TB, and Malaria; UNITAID; and the U.S. bilateral PEPFAR program. These institutions are at the center of about $8.1 billion in international funding that supports over 30 million on treatment.17
Acting in unison, these four elements built a model for greater access to ARVs. For example, a decade after the start of this model, originator companies were still offering first-line ARVs at prices of $1,033 per year to low- and lower-middle income countries (LMICs). At that price, it would cost $30 billion per year today just to buy ARVs in LMICs (without staff or programming for treatment, prevention, and care) — 1.5 times total AIDS financing,18 all to a handful of US/European companies. Instead, today’s leading HIV treatment (TLD) is available worldwide and costs less than $45/year in LMICs.19 Indeed, the “success” can be seen in the breach — as in Colombia, which was excluded from the TLD licenses and asked to pay several thousand dollars per person, per year, by the originator.20 The country attempted to negotiate a more affordable price, but eventually issued a compulsory license to secure generic access.
Using The “AIDS Model” Beyond AIDS
Beyond AIDS, the MPP expanded its mandate in 2018 to include small molecule drugs across disease areasReference Burrone21 and again in 2021 to include biotherapeutics.Reference Morin22 It has often struggled to achieve the kind of leverage it has on HIV, however. Some drugs for Hepatitis C have been licensed through the MPP, and others through bilateral licensing.23 On TB, neither MPP nor bilateral licenses have been easy — with, recently, a major pressure campaign using pre-grant opposition mechanisms (India, Brazil) and Competition Law (South Africa) initiated by activists globally, to push Johnson & Johnson to agree not to enforce its evergreening patents on bedaquiline in 134 LMICs and to substantially drop its price.Reference Tu24
This weak leverage is directly linked to governments’ reduced use of the legal authority that pushed companies into voluntary action. Efforts to increase compulsory licenses beyond AIDS have been met with resistance from industry and high-income country governments. Graph 1 shows that although civil society and generic companies have sought compulsory licensing roughly at the same rate as during the AIDS peak, governments have recently been far less interested in using this tool. While this does not capture the instances where government officials have invoked compulsory licensing as leverage in negotiations, one can expect that formal proceedings correlate.
Battles Over The Proper Use Of Law In COVID-19
The “AIDS model” was available to the world when the COVID-19 pandemic hit and scientific breakthroughs brought effective mRNA vaccines within a year. In many ways, the model was ready-made for tackling the challenge with high-quality decentralized production, with networked multilateral organizations ready to support technology transfer and IP sharing and a set of public and private sector producers in LMICs already making pandemic products. Before any vaccine was available, Costa Rica’s president proposed an agreement among WHO member states to share technology to help stop the pandemic.Reference Quesada25 Several countries amended their patent laws to facilitate the use of compulsory licenses for COVID-19 technologies. South Africa and India proposed a temporary waiver at the WTO to remove any IP barriers.Reference Waiver26 The WHO put together the COVID-19 Technology Access Pool (C-TAP), an initiative modeled upon the MPP, but with an expanded mandate.27
But instead of mobilizing the full, synergistic model of government legal action, decentralized manufacturing, voluntary licensing, and financing during COVID-19, global health institutions focused mainly on the last — financing and on donations.Reference Kavanagh and Singh28 The few compulsory measures were largely in high-income countries for development, as when the US granted Moderna unauthorized use of patented vaccine technology.29 Voluntary licenses were also scarce, more in treatment than vaccines.Reference Sharma30 Meanwhile high-income countries opposed a WTO waiver31 and successfully delayed and limited its scope such that it had little effect.32 No major vaccine developer agreed to put its technology into C-TAP. While companies like Moderna showed they could rapidly transfer know-how to new factories producing vaccines within months (e.g. in Switzerland), and an mRNA Hub showed they could produce mRNA vaccines in South Africa, originator companies were neither compelled nor incentivized to share technology with global South producers, to serve global South populations, as is standard for HIV.Reference Baker, Hassan and Gonzales33 Indeed, some doses made in Africa were shipped to Europe and North America while Africans waited.Reference Robbins and Mueller34 High-income countries and companies successfully opposed deployment of the full “AIDS model,” resulting in preventable illness and death.Reference Adikari and Wijesinghe35
Global equity efforts focused primarily on financing purchase of vaccines from originator companies. The COVAX facility, managed by Gavi, launched 9 months before any vaccines were approved, aiming to halt the pandemic’s acute phase with “rapid, fair and equitable access” to vaccines.Reference Berkley36 To start, COVAX would secure 2 billion doses in 2021 — to cover 20% of participating countries’ populations.Reference Usher37 It used advanced purchase agreements to incentivize development, negotiations with companies making vaccines, and pooled demand-signaling as a market-based incentive for producers.38 But, despite having been heavily funded by governments, companies maintained monopoly control over production of each vaccine. It was originator companies who decided whether to sell doses to COVAX and/or to LMICs, in what quantity, and on what timeline.Reference Hassan39 Neither companies nor states were compelled to prioritize COVAX orders. Middle-income countries like South Africa were pushed to sign highly one-sided secret contracts with unfair terms and no guarantee of delivery dates or quantities to obtain vaccines for their populations.40
Ultimately, with only a few producers and a resultant self-created scarcity of vaccine doses, vaccine nationalism fatally undermined this effort, much as high prices undermined early global HIV treatment efforts. High income countries mobilized political and economic power to secure agreements for enough doses to vaccinate their populations many times over. COVAX could not compete, reaching less than half its goal.41
A year after the first vaccines were registered, 9 billion doses had been administered, but just 1% of them were delivered in low-income countries.42 By the end of 2021, more booster shots had been administered in HICs than first shots in LMICs,43 while just 1 in 4 African health workers had received a full course of vaccine.44 Global vaccination did not come close to equity.
The Future of Global Health Law in Infectious Disease Medicines
Today the global governance of access to medicines for infectious diseases and pandemics is deeply unsettled. Two potential paths present themselves: Despite its challenges and failures, some policy makers still favor expanding the COVAX-type model and making it permanent, albeit with a more rapid funding mechanism and the hope of avoiding the “entirely unexpected” behaviors by states and companies.Reference Berkley45 This is particularly favored by industry and high-income countries. But it is not clear why this model will function better in a future pandemic than it did during COVID-19.
In negotiations on a Pandemic Treaty, many developing countries and civil society groups pushed to bring in more elements of the AIDS model — commitments to share technology promptly, waive IP obligations to support government action, build decentralized factories, and finance pandemic response. If these make it into the final pandemic agreement, they could shift the focus toward rapid knowledge sharing and worldwide production in a pandemic instead of relying on high-income country producers to behave equitably.46
But even if they make it into a future treaty, and even apart from questions of compliance and enforceability, this will be insufficient to secure equity. Indeed, in the recent Mpox outbreak in Africa we see a repeat of lack of access to vaccines.Reference Furlong and Kresge47 WTO’s role in governing IP on medicines needs to be rethought. In the face of a crisis of unfair trade in lifesaving vaccines, a multi-year debate on allowing national governments to act that ends in gridlock is intolerable.Reference Fischer48 And there are big questions for infectious and non-communicable diseases outside of pandemics. Prices for NCD drugs are sky-high. Increasingly even high-income countries seek to use certain elements of what worked for AIDS to reign them in — the Biden administration’s move to use “march-in” rights to access patented, federally-funded products is an example.49 These might portend a global shift away from North vs. South politics on access and instead to a greater use of law to address inequality. If accelerated, it might portend a rebalancing toward public health.



