1. Introduction
The stable isotope composition of terrestrial matter (environmental samples, animals, humans) at coastal sites can be distinctly affected by sea spray aerosols. Seawater and sea spray aerosols contain a variety of different cations and anions, such as Na+, K+, Mg2+, Ca2+, Sr2+, Cl–, HCO3 –, CO3 2–, and SO4 2– (e.g., Bates et al. Reference Bates, Quinn, Frossard, Russell, Hakala, Petäjä, Kulmala, Covert, Cappa and Li2012; Keene et al. Reference Keene, Maring, Maben, Kieber, Pszenny, Dahl, Izaguirre, Davis, Long and Zhou2007; Wright and Colling Reference Wright, Colling, Suckow, Weisbroth and Franklin1995b). Marine aerosols, entering the terrestrial environment via sea spray, can cause a shift in the isotope composition of biological tissues towards a seemingly marine isotope signature, masking the terrestrial isotopic fingerprint.
Besides a direct sea spray effect, caused by the uptake of isotopically enriched ions of marine origin (e.g., Clementz et al. Reference Clementz, Goswami, Gingerich and Koch2006; Clementz and Koch Reference Clementz and Koch2001; Hobson Reference Hobson1999; Koch Reference Koch2007; Mook Reference Mook1971; Nehlich Reference Nehlich2015), physiological and biochemical reactions in the plants can cause associated indirect sea spray effects due to discrimination caused by, e.g., salinity stress. These physiological reactions are visible in the stable isotope signature of plants, including δ13Ccellulose, δ18Ocellulose, δ18Osulfate, δ34Ssulfate, δ34Sorganic S, and δ34Stotal S (Ellsworth and Sternberg Reference Ellsworth and Sternberg2014; Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a, Reference Göhring, Hölzl, Mayr and Strauss2023b; Krouse Reference Krouse, Rundel, Ehleringer and Nagy1989; Liu et al. Reference Liu, Yang, Gong, Schäufele and Schnyder2017; O’Leary Reference OʼLeary1981; Sternberg et al. Reference Sternberg, Ish-Shalom-Gordon, Ross and O’Brien1991; Trust and Fry Reference Trust and Fry1992; Yakir et al. Reference Yakir, DeNiro and Ephrath1990). Physiological effects can partly overlay or even mask the actual sea spray effect, making it difficult to distinguish between isotopic shifts caused by the (direct) sea spray effect and those caused by associated physiological (indirect) effects (see below).
Atmospheric CO2 taken up by plants during photosynthesis is used for a variety of metabolic pathways, such as gluconeogenesis or the Krebs cycle (see, e.g., Poschenrieder et al. Reference Poschenrieder, Fernández, Rubio, Pérez, Terés and Barceló2018). However, plants can also take up CO2 (aq.) as well as dissolved CO3 2– or HCO3 –. CO2 and HCO3 – can be reversely interconverted (CO2 + H2O ↔ HCO3 – + H+) by carbonic anhydrases (see, e.g., Tiwari et al. Reference Tiwari, Kumar, Singh and Ansari2005). As has been demonstrated previously, HCO3 – cannot only be taken up via the roots of the plants but also via their leaves (Bedri et al. Reference Bedri, Wallace and Rhoads1960; Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a; Overstreet et al. Reference Overstreet, Ruben and Broyer1940; Poschenrieder et al. Reference Poschenrieder, Fernández, Rubio, Pérez, Terés and Barceló2018; Rasmussen et al. Reference Rasmussen, Kusliene, Jacobsen, Kuzyakov and Eriksen2013; Schäfer Reference Schäfer1988; Stringer and Kimmerer Reference Stringer and Kimmerer1993; Zamanian et al. Reference Zamanian, Pustovoytov and Kuzyakov2017). Marine-derived HCO3 –, which constitutes about 90% of inorganic carbon in the seawater (Poschenrieder et al. Reference Poschenrieder, Fernández, Rubio, Pérez, Terés and Barceló2018), could, thus, potentially enter plant tissues via sea spray. Under controlled greenhouse conditions, the sea spray effect at the Baltic coast caused an enrichment in 13Ccellulose by about 6‰ as quantified by Göhring et al. (Reference Göhring, Hölzl, Mayr and Strauss2023a). This shift is not only resulting from the uptake of HCO3 – of marine origin, enriched in 13C (Craig Reference Craig1953; Mook and Vogel Reference Mook and Vogel1968; Poschenrieder et al. Reference Poschenrieder, Fernández, Rubio, Pérez, Terés and Barceló2018; Saltzman and Thomas Reference Saltzman, Thomas, Gradstein, Ogg, Schmitz and Ogg2012), but also due to a physiological reactions in the plants (indirect “sea spray” effect). Salinity stress has a distinct effect on δ13Ccellulose due to stomatal closure. Reduced stomatal conductance leads to a decreased discrimination against 13C and, thus, results in less negative δ13C values (Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a, Reference Göhring, Hölzl, Mayr and Strauss2023b; McCarroll and Loader Reference McCarroll and Loader2004; Roden et al. Reference Roden, Bowling, McDowell, Bond and Ehleringer2005; Zhang et al. Reference Zhang, Hartmann, Gleixner, Thoma and Schwab2019). Similarly, high HCO3 – stress, caused by a very high concentration of HCO3 –, results in stomatal closure (see, e.g., Gao et al. Reference Gao, Sagi and Lips1998; Mukhtar et al. Reference Mukhtar, Shahid, Khan, Balal, Iqbal, Naz, Zubair and Ali2016; Poschenrieder et al. Reference Poschenrieder, Fernández, Rubio, Pérez, Terés and Barceló2018), also affecting δ13Ccellulose (Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a).
Radiocarbon dating of archaeological remains from coastal sites may need to be corrected for a marine reservoir age as the marine reservoir is depleted in 14C activity due to pre-aged water mixture. Marine organisms usually reflect both the actual 14C decay and the reservoir 14C activity due to isotope uptake via the food chain. The so-called reservoir 14C age R(t) describes the age offset between the marine reservoir and the atmosphere. For regional differences, caused by different oceanographic conditions, a so-called ΔR was defined, which accounts for the offset between regional and global ocean apparent 14C ages (Stuiver and Braziunas Reference Stuiver and Braziunas1993). For the Baltic Sea, for example, Fischer and Olsen (Reference Fischer and Olsen2021) determined a marine reservoir age of 273 ± 18 14C years (ΔR = –234 ± 61 years). This is younger than the (pre-bomb) global marine reservoir effect of about 585 14C years, with a regional ΔR of –140 years for the Western Baltic (marine reservoir age: 585 – 140 = 445 14C years; CALIB rev. 8, Stuiver and Reimer Reference Stuiver and Reimer1993; Heaton et al. Reference Heaton, Bard, Bronk Ramsey, Butzin, Hatté, Hughen, Köhler and Reimer2023, Reference Heaton, Köhler, Butzin, Bard, Reimer, Austin, Bronk Ramsey, Grootes, Hughen, Kromer, Reimer, Adkins, Burke, Cook, Olsen and Skinner2020).
By consuming marine food sources, e.g., fish, the marine isotope signal could be transferred to the terrestrial consumer, e.g., humans, via the food chain. However, the impact of uptake of dissolved inorganic carbon (DIC) of marine origin, i.e., H14CO3 –, on the radiocarbon signature of plants has so far not been investigated. Such a linkage could potentially affect the 14C signature of humans either through direct consumption of the plants or by consuming, e.g., the meat of herbivores which in turn are directly or indirectly influenced by the plant sources affected by sea spray. A marine impact by consuming plants influenced by sea spray would, however, not be visible in the δ13Ccollagen or δ15Ncollagen values of consumers, which reflect the protein sources in their diet (e.g., Johansen et al. Reference Johansen, Gulliksen and Nydal1986; Richards et al. Reference Richards, Fuller and Molleson2006; Richards and Hedges Reference Richards and Hedges1999; Schoeninger and DeNiro Reference Schoeninger and DeNiro1984; Schoeninger et al. Reference Schoeninger, DeNiro and Tauber1983). The latter would (correctly) indicate terrestrial protein sources even though an individual or its diet was affected by sea spray (see Göhring et al. Reference Göhring, Mauder, Vohberger, Nehlich, von Carnap-Bornheim, Hilberg, Kröger and Grupe2018, Reference Göhring, Mayr and Grupe2020).
This study complements an earlier published study focusing on the stable and radiogenic isotope composition (δ13C, δ18O, δ34S, 87Sr/86Sr) of plants grown in a greenhouse and treated with an artificial sea spray (Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a). Here, we examine the sea spray effect on the radiocarbon composition of these plants.
2. Material and methods
2.1 Greenhouse experiment
European beach grass (Ammophila arenaria, L.; Jelitto Staudensamen GmbH, Schwarmstedt, Germany) was grown in planters on a mixture of lawn sand and lawn soil (ratio 1:7; Floragard GmbH, Oldenburg, Germany), prepared for all treatment groups (see below), in the greenhouse of the Biocenter of the Ludwig Maximilian University Munich (Figure 1) in June (group 1) and July 2020 (group 2) and sampled end of December 2020. The plants grew under constant environmental conditions, at 20°C with a relative humidity of ca. 60%, and under long-day light conditions (0.9 klx).
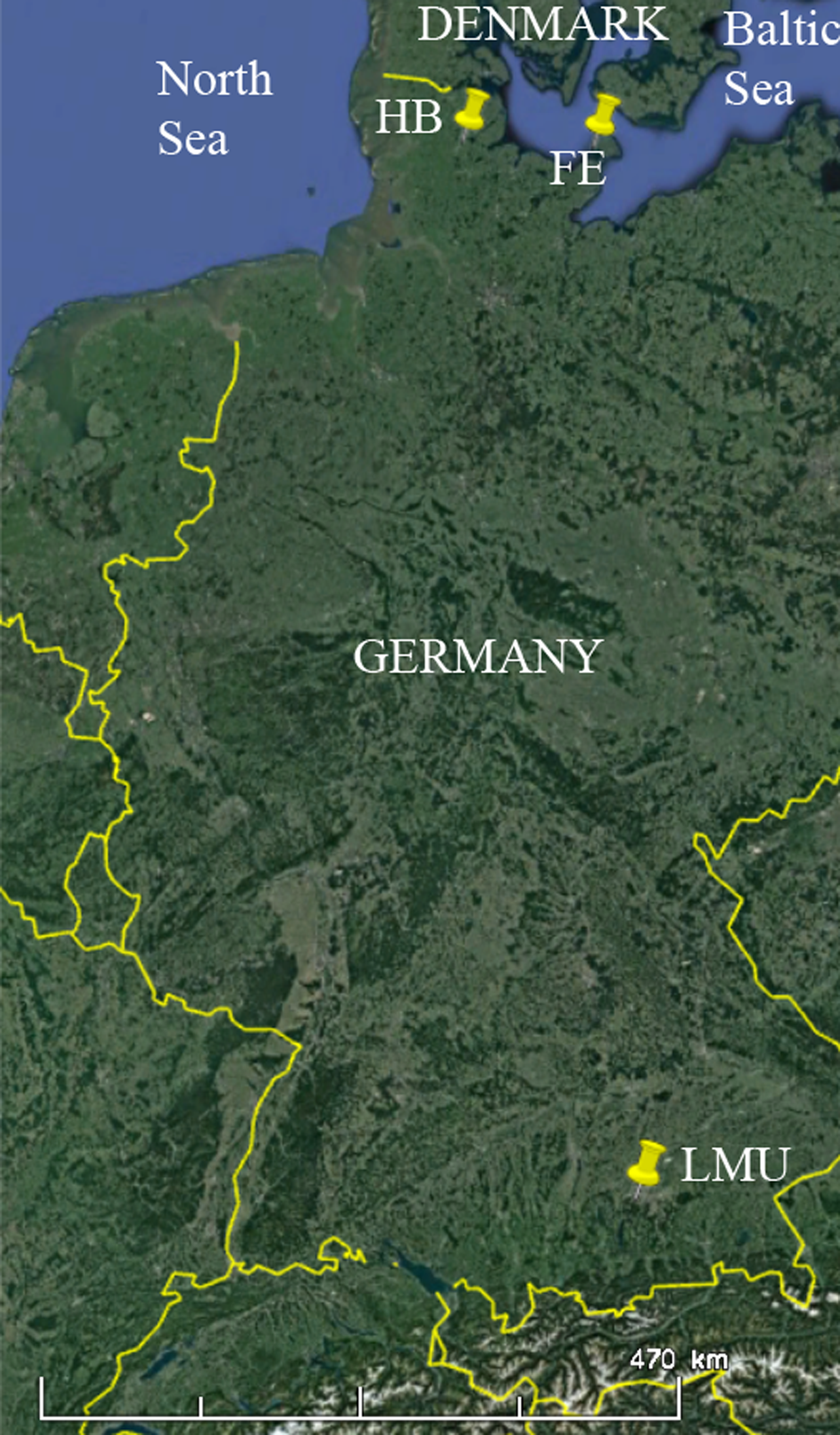
Map indicating the sampling locations for the spray water (HB = Haithabu (G-5), FE = Fehmarn (G-6)) as well as the greenhouse of the Biocenter of the LMU Munich. Google Earth Pro, Google 2022, http://www.earth.google.com, 12/14/2015, 51.327532 °N, 10.271728 °E, eye altitude 1808.12 km.
Plants were watered with tap water (G-GW) about three days a week (ca. 150 mL/week). Irrigation water (tap water) was filled into a jerrycan, stored in the greenhouse chamber. Munich tap water mainly originates from the Mangfall as well as Loisachtal Valley. Artificial sea spray treatment (ca. 2.5 mL/week) started two weeks after sowing. At this time, germlings reached a height of about 1.5 to 2.0 cm. The used amount of spray water was just enough to result in a sufficient moistening of the leaves, thus simulating the effect of sea spray aerosol droplets on the leaves.
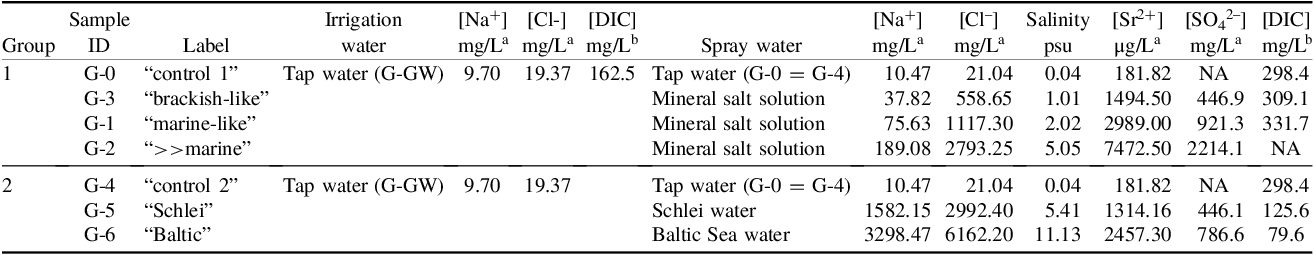
Seven treatments have to be distinguished according to the experimental setting in the greenhouse (see Table 1). Plants of group 1 (G-0, G-1, G-2, G-3) were sprayed with mineral salt solution, prepared by dissolving mineral salt (Preis Aquaristik KG, Bayerfeld, Germany) of varying ion/mineral concentrations, containing only traces of NaCl (see Table 1), in tap water. Tap water (and corresponding mineral salt solution) was restocked in the spray bottles of the treatment groups (G-1 to G-3) and control groups (G-0/G-4) on a regular basis. These treatments aim to allow quantification of the impact of different ions (e.g., bicarbonate) taken up by the plants without any associated salinity impact (in contrast to group 2). As a control, plants of G-0 (“control 1”) were sprayed with tap water only. The mineral salt solutions for G-1 to G-3 were prepared by dissolving the mineral salt in Munich tap water (identical to control G-0). The artificial “marine-like” spray water for treatment G-1 was prepared with an ion concentration (e.g., HCO3 –, SO4 2-, Sr2+) comparable to seawater, but containing only a low amount of NaCl (2 g mineral salt dissolved in 500 mL tap water; see Table 1) compared to about 10556 mg/L Na+ and about 18980 mg/L Cl– in seawater (Wright and Colling Reference Wright, Colling, Suckow, Weisbroth and Franklin1995a). This treatment should resemble a marine spray water without salinity, i.e., NaCl, impact. The salinity level of this treatment, based on chlorinity (S = 1.80655*chlorinity [g/L]; see Wright and Colling (Reference Wright, Colling, Suckow, Weisbroth and Franklin1995a)), was about 2 psu (practical salinity unit; 1 psu ≙ 1 g/L). For comparison, the salinity of the open ocean varies between about 33 and 37 psu, while brackish water of epeiric seas like the Baltic Sea has a salinity of less than 25 psu (Wright and Colling Reference Wright, Colling, Suckow, Weisbroth and Franklin1995a). Similarly, the artificial “brackish-like” spray water of the treatment G-3 was prepared with a brackish-like ion solution, again with a low NaCl concentration (1 g mineral salt dissolved in 500 mL tap water; see Table 1) and a respective salinity of about 1 psu. For the plants of treatment G-2 (“>>marine”) we prepared a spray water containing an ion concentration 2.5 times higher than found in natural seawater (5 g mineral salt dissolved in 500 mL tap water; see Table 1). The Na+ concentration (189 mg/L) and the Cl– concentration (2793 mg/L) are beyond seawater levels (see above). Accordingly, salinity levels were still low with about 5 psu.
Overview of the plant, irrigation water, and spray water samples for the different treatments in group 1 and 2, respectively, including [Na+] and [Cl−] in irrigation water as well as [Na+], [Cl−], salinity (psu), [Sr2+], [SO4 2-], and [DIC], measured as mg CO2, in the spray water samples (see text for details). NA = not available. nd = not determinable ([SO4 2-] below detection level)
a Data from Göhring et al. (Reference Göhring, Hölzl, Mayr and Strauss2023a).
b This study.
In contrast, plants of group 2 were sprayed with salty water, containing NaCl (see Table 1), from (i) the Schlei inlet (treatment G-5; “Schlei”), sampled at the Viking Haithabu site (54.491528°N, 9.5693333°E), and (ii) with water from the Baltic Sea (treatment G-6; “Baltic”), collected at the western coast of Fehmarn (Flügger Strand; 54.452361°N, 11.004722°E) in July 2020. The spray water samples for G-5 and G-6 were stored in jerrycans (at 4°C). Similar to the control G-0, the control G-4 (“control 2”) was sprayed with tap water only. This spray water was identical to the water used for G-0. Spray water of the treatments G-5 and G-6 was naturally salty and, thus, contained elevated amounts of NaCl. The salinity for the spray water of G-5 and G-6 was about 5.4 psu and 11.1 psu, respectively (see Table 1; Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a).
The control plants G-0 (group 1) and G-4 (group 2), growing on the same soil as the plants of the other treatment groups, sprayed with tap water also used to prepare the mineral salt solutions for group 1 treatments, and irrigated with the same tap water (G-GW) as all other plants, provide the isotopic signature expected for plants unaffected by sea spray. This enables a comparison with the plants of the different treatment groups growing under artificial sea spray. In addition, the controls also reflect all potential changes over the growth period (although limited in a constant greenhouse environment), including potential changes in the atmospheric CO2 and chemical or isotopic composition of tap water, as well as the background soil signal. Moreover, while we cannot fully exclude fossil fuel effects on the greenhouse plants (see above), any such effects would also influence the isotopic signature of the control plants growing under the same atmosphere as all other plants. Hence, we assume that a deviation in the isotopic signature of the treated plants from that of the control plants is the result of the artificial sea spray.
Plants were treated with the artificial sea spray five days a week (Monday to Friday) by spraying the leaves of the plants with about 0.5 mL of spray water per planter per day. This treatment was conducted from June 2020 (group 1) or July 2020 (group 2) until the end of December 2020. For measurements, one grass blade, reflecting the whole growth period, of each treatment group was sampled (length G-0: ca. 47 cm, G-1: ca. 48 cm, G-2: ca. 42 cm, G-3: ca. 50 cm, G-4: ca. 40 cm, G-5: ca. 41 cm, G-6: ca. 45 cm). The samples were processed for further analyses as described in section 2.2.
2.2 Laboratory analyses
This study focuses on radiocarbon measurements. The relevant parts of the laboratory analyses are presented below. The protocols for stable isotope analysis or elemental analysis of sampled plant leaves and spray as well as irrigation water samples are published elsewhere (see Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a). The radiocarbon analyses were conducted at the Leibniz Laboratory for Radiometric Dating and Stable Isotope Research in Kiel.
The sampled plant leaves were washed twice in an ultrasonic bath (Bachofer) using distilled water for 15 min each to remove surficial salts and subsequently dried in a drying oven (SALVIS, KCTS 11). Plant sample material selected for radiocarbon analysis was inspected for macroscopic contamination under the microscope. Plant material was successively treated with 1% HCl, 1% NaOH (60°C), and 1% HCl. The resulting alkali residue was combusted following Nadeau et al. (Reference Nadeau, Grootes, Schleicher, Hasselberg, Rieck and Bitterling1998).
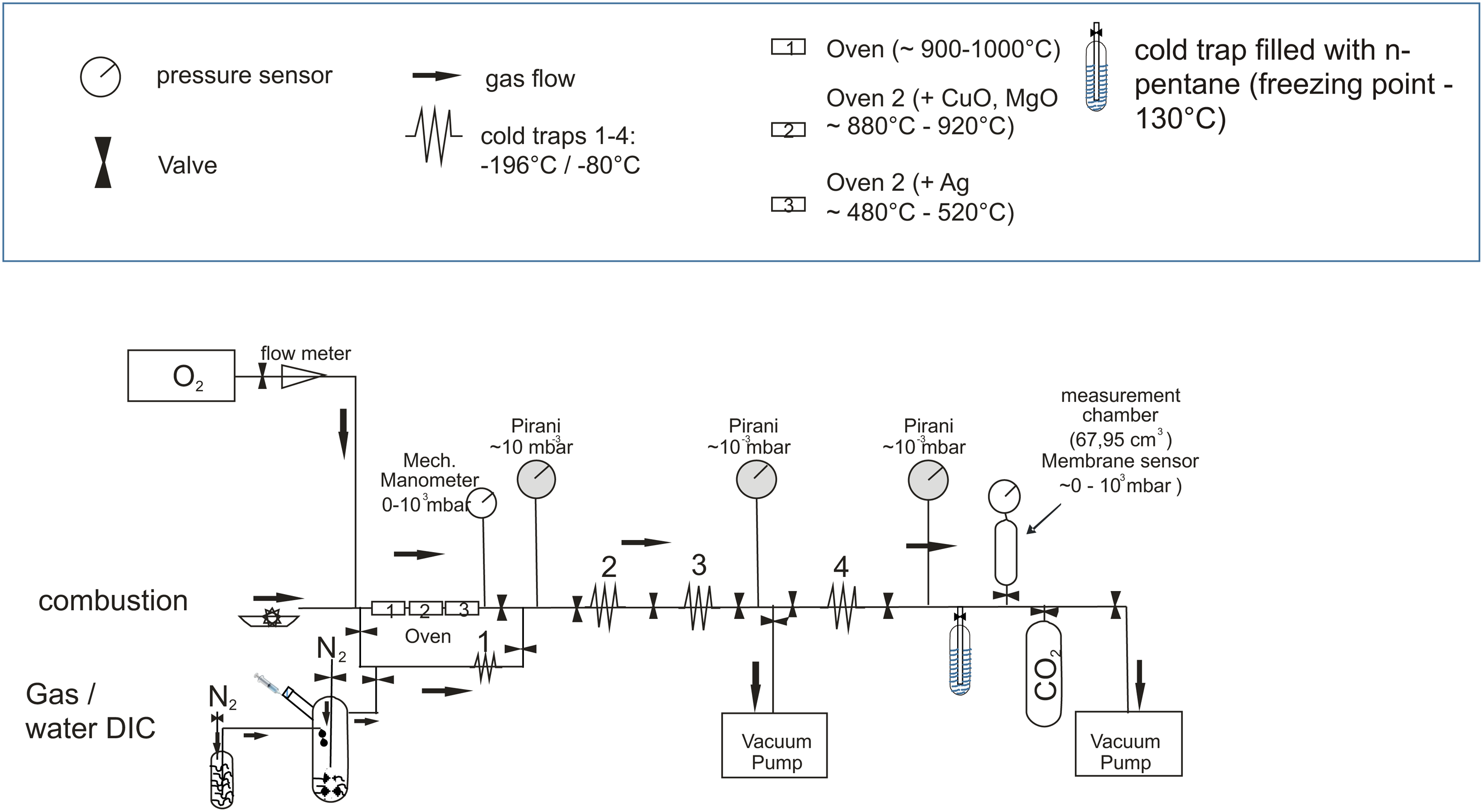
For the water samples used during the growth experiments (i.e., irrigation and spray water), a high-vacuum Multi-Purposed-CO2-Extraction Line (MPEL; see Figure 2) was used. The CO2 extraction line was conditioned by evacuation below 1 mbar, flushed three times with ultra-pure N2, and a final N2 fill, pressured between 400–600 mbar. After water sample transfer under an ultra-pure N2 gas stream, 5 mL degassed 100% H3PO4 was added to acidify the sample. The dissolved inorganic carbon (DIC) was stripped under a N2 gas stream (50 mL/min for about 30–40 min) as CO2. CO2 was subsequently cryogenically collected and purified with liquid nitrogen (–196°C) and a dry-ice/alcohol slurry (–80°C) under vacuum (< 0.001 mbar). The amount of CO2 was estimated by measuring the pressure of the gas in a defined volume (in mg CO2/L).
Illustration of the Multi-Purposed-CO2-Extraction Line (MPEL). © C. M. Hüls.
After graphitization of the resulting CO2 (Nadeau et al. Reference Nadeau, Schleicher, Grootes, Erlenkeuser, Gottdang, Mous, Sarnthein and Willkomm1997) from either combustion or DIC extraction, AMS measurements were performed with a HVE 3MV Tandetron 4130 at the Leibniz Laboratory in Kiel. The simultaneously measured isotope ratios 14C/12C and 13C/12C of the sample were compared with a CO2 measurement standard (Oxalic Acid II) and successively corrected for effects caused by contamination with external carbon during sample processing, determined by a double-blind test. The resulting 14C content of the sample is corrected for isotope fractionation and is given as F14C related to the hypothetic atmospheric value in AD 1950 according to
with the normalized specific activity of the sample (ASN) and the normalized specific activity of Oxalic Acid II (AON), whereby the specific activity of the standard is normalized to δ13C = –25‰ (Reimer et al. Reference Reimer, Brown and Reimer2004). The conventional radiocarbon age (14C age) can be calculated according to
The proportion of fossil carbon (fossil 14C (%)) in the plants can be calculated by
with F14Cplant and F14Catm being the apparent 14C concentration in the plant samples and the contemporary atmosphere, respectively. Atmospheric 14C during the experiment’s growth period (group 1 [G-0 to G-3]: June–December 2020, group 2 [G-4 to G-6]: July–December 2020) can be calculated to about F14Catm = 1.0048 (Δ14Catm = –3.6560‰) for group 1 and about F14Catm = 1.0044 (Δ14Catm = –4.0200‰) for group 2, using the Hohenpeissenberg (HPB; 47.8011°N, 11.0246°E) atmospheric 14CO2 dataset (Kubistin et al. Reference Kubistin, Plaß-Dülmer, Arnold, Kneuer, Lindauer, Müller-Williams and Schumacher2024). The HPB atmosphere data used here are only an approximation of the atmosphere present in the greenhouse (distance between HPB and LMU Biocenter: ca. 50 km). The real atmospheric 14C could be different from the HPB data, also due to potential urban fossil influences. Due to controlled air flows (air filters) in the climate chamber and the suburban location of the greenhouse, this potential impact is most likely very small, even though it cannot be fully excluded. The percentage of dissolved inorganic carbon from water taken up by the plants (DIC 14C [%]) is based on the relative difference between the F14Catm value (see above) and the F14C value in the plant (F14Cplant) as well as the water (F14Cwater (modeled)):
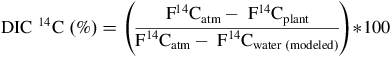 $${\rm{DIC}}\;_{}^{14}{\rm{C}}\;\left( \% \right) = \left( {{{{{\rm{F}}^{14}}{{\rm{C}}_{{\rm{atm}}}} - \;{{\rm{F}}^{14}}{{\rm{C}}_{{\rm{plant}}}}} \over {{{\rm{F}}^{14}}{{\rm{C}}_{{\rm{atm}}}} - \;{{\rm{F}}^{14}}{{\rm{C}}_{{\rm{water}}\;\left( {{\rm{modeled}}} \right)}}}}} \right){\rm{*}}100$$
$${\rm{DIC}}\;_{}^{14}{\rm{C}}\;\left( \% \right) = \left( {{{{{\rm{F}}^{14}}{{\rm{C}}_{{\rm{atm}}}} - \;{{\rm{F}}^{14}}{{\rm{C}}_{{\rm{plant}}}}} \over {{{\rm{F}}^{14}}{{\rm{C}}_{{\rm{atm}}}} - \;{{\rm{F}}^{14}}{{\rm{C}}_{{\rm{water}}\;\left( {{\rm{modeled}}} \right)}}}}} \right){\rm{*}}100$$
F14Cwater (modeled) is calculated according to Eq. (5). The treated plants had two different water sources, namely spray water (ca. 1.64 vol%) and irrigation water (ca. 98.36 vol%; see section 2.1) with measured F14Cspray water and F14Cirrigation water concentration. DIC potentially taken up by the plants via their roots (mainly) originates from irrigation water. All treatment groups were irrigated with the same irrigation water (G-GW) as well as with the same amount of irrigation water. Accordingly, isotope differences between treatment groups are not caused by irrigation water. Nevertheless, irrigation water is a relevant source of DIC for all plants and has to be considered:
The DIC concentration of the modeled water is a mixture of the concentration found in the spray and irrigation water, calculated as
Potential differences in the DIC concentration of the different water sources (spray water, irrigation water) are reflected by F14Cwater (modeled) due to the existing relationship between the radiocarbon concentration and the DIC concentration in water (see, e.g., Wang et al. Reference Wang, Lin, Burr, Wang and Lin2022).
Plants discriminate against 13C or 14C in their carbon source (e.g., atmospheric CO2). Discrimination against 13C (Δ13C) or 14C (ΔΔ14C) can be calculated based on the following formula:
 $$ \Delta = {\rm{\;}}{{{{{{\rm{\delta }}_{{\rm{source}}}}} \over {1000}}{\rm{\;}} - {\rm{\;}}{{{{\rm{\delta }}_{{\rm{plant}}}}} \over {1000}}} \over {1 + {\rm{\;}}{{{{\rm{\delta }}_{{\rm{plant}}}}} \over {1000}}}}\ {\rm{*}}\ 1000\ \left( {{\rm{after \ Farquhar \ et \ al}}.{\rm{ }}\ 1989} \right) $$
$$ \Delta = {\rm{\;}}{{{{{{\rm{\delta }}_{{\rm{source}}}}} \over {1000}}{\rm{\;}} - {\rm{\;}}{{{{\rm{\delta }}_{{\rm{plant}}}}} \over {1000}}} \over {1 + {\rm{\;}}{{{{\rm{\delta }}_{{\rm{plant}}}}} \over {1000}}}}\ {\rm{*}}\ 1000\ \left( {{\rm{after \ Farquhar \ et \ al}}.{\rm{ }}\ 1989} \right) $$
In contrast to δ13C and Δ14C, Δ13C and ΔΔ14C allow investigation into biological processes (Farquhar and Lloyd Reference Farquhar and Lloyd1993). For discrimination against 13C and 14C of atmospheric CO2, δsource was set to δ13Catm = −8.5‰ (Cernusak and Ubierna Reference Cernusak and Ubierna2022) and Δ14Catm = –3.6560‰ (group 1) or Δ14Catm = –4.0200‰ (group 2; see above), respectively. For δplant, the measured IRMS δ13Ccellulose values (see also Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a) were used and the age-corrected Δ14Cplant values were calculated according to
with
![]() ${\lambda _{\rm{c}}} = {{\ln \left( 2 \right)} \over {5730\;{\rm{a}}}}$
and x = 2020 (year of growth/sampling). The discrimination of plants against 13C and 14C of DIC in spray water is based on the δ13CDIC and Δ14CDIC values measured for the spray water samples and can be calculated according to Eq. (6) (with source = DIC).
${\lambda _{\rm{c}}} = {{\ln \left( 2 \right)} \over {5730\;{\rm{a}}}}$
and x = 2020 (year of growth/sampling). The discrimination of plants against 13C and 14C of DIC in spray water is based on the δ13CDIC and Δ14CDIC values measured for the spray water samples and can be calculated according to Eq. (6) (with source = DIC).
The control plants (G-0/G-4) grew in the same chamber and, thus, under the same atmosphere, as the different treatment groups. They were also irrigated with the same tap water as all other groups. Accordingly, deviations in the isotopic composition of the plants of the different treatment groups from the isotopic signature observed for the corresponding control groups is an indicator for sea spray related effects.
3. Results
Our main objective was to study possible effects in the 14C composition of plants which were sprayed during growth with water containing different amounts of minerals, including DIC, and salt (NaCl), and thereby mimicking effects of sea spray.
The radiocarbon composition of the water used for the experiments is presented in Table 2. All plants were irrigated with tap water G-GW with a radiocarbon concentration of F14C 0.9337 ± 0.0031, indicating the admixture of a few percent of fossil carbon within the aquifer. The tap spray water used for the control samples G-0 (group 1) and G-4 (group 2), was identical. This water contained a radiocarbon concentration of F14C 0.9191 ± 0.0029 and thus contained a bit more fossil carbon than the irrigation water. The mineral salt mixed to the spray water solutions of G-3 (“brackish-like”) and G-1 (“marine-like”) apparently contained a higher amount of fossil carbon compared to the control samples since the resulting spray water show a F14C of about 0.9006 ± 0.0030 and 0.8707 ± 0.0030, respectively. Unfortunately, the radiocarbon measurements of the spray water of G-2 (“>>marine”) failed due to a broken sample container. The salty spray water of G-5 (“Schlei”) and G-6 (“Baltic”) were more enriched in 14C (G-5: 0.9437 ± 0.0030; G-6: 0.9638 ± 0.0031) compared to the spray water of G-0/G-4, G-3, and G-1.
14C age, F14C (± standard deviation σ), and Δ14C (see Eq. 7) for plant leaves, spray water, and irrigation water samples from the greenhouse as well as modeled F14C and corresponding Δ14C (see Eq. 7) in water (F14Cmodeled; see Eq. 5), calculated percentage of fossil 14C in plants (fossil 14C (%); see Eq. 3), and calculated percentage of 14C in plants originating from dissolved inorganic carbon (DIC) in (spray or irrigation) water (DIC 14C (%); see Eq. 4). NA = not available
The irrigation water G-GW and the spray water of the control treatments G-0 (“control 1”) and G-4 (“control 2”) contained 162.5 mg/L and 298.4 mg/L dissolved inorganic carbon (DIC), measured as mg CO2, respectively. Spray water used for the treatments G-3 and G-1 contained a slightly higher DIC content (309.1 mg/L and 331.7 mg/L, respectively). The natural salty water from the Schlei inlet (G-5) and the Baltic Sea (G-6) contained markedly lower DIC concentrations of 125.6 mg/L and 79.6 mg/L, respectively (see Figure S1 and Table 1).
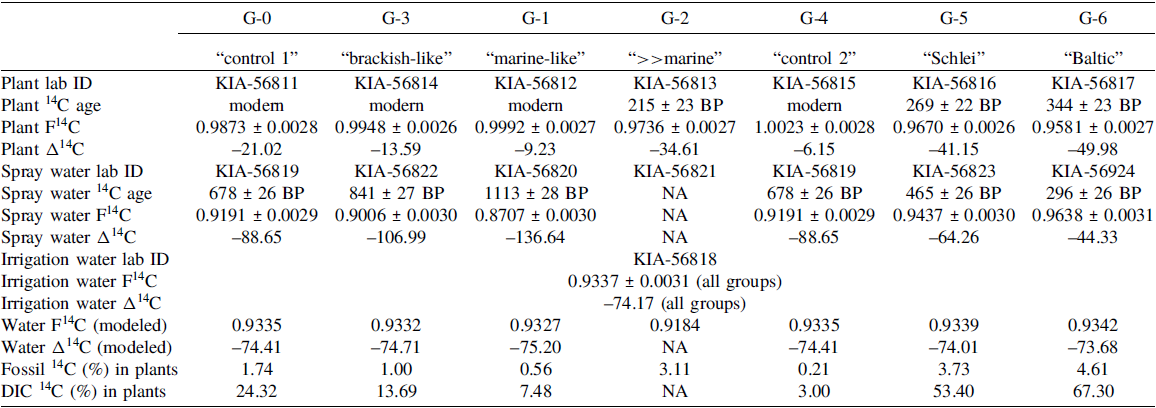
The results for the radiocarbon analyses on the plant samples are presented in Table 2 and visualized in Figure 3A. Estimated radiocarbon concentration for plants of the treatments G-0 (“control 1”), G-3 (“brackish-like”), and G-1 (“marine-like”) give 14C signatures which are depleted with respect to atmospheric 14C. The plant sample of treatment G-4 (“control 2”) exhibit, as expected, a radiocarbon concentration close to average atmospheric 14C over the growth period. The experimental conditions of treatments G-0 and G-4 differ in the growth period of both treatments (G-0: June–December; G-4: July–December), while the composition of used irrigation and spray water are the same. The depleted radiocarbon content of G-0 plant tissue with a magnitude comparable to treatments G-1 and G-3 seem to indicate a larger influence of the fossil carbon containing irrigation and spray waters, as compared with treatment G-4 and need further verification.
(A) F14C plotted against δ13C for beach grass leaves (α-cellulose; washed), spray water (dissolved inorganic carbon (DIC)), and irrigation water (G-GW; DIC) for group 1 (mineral salt solution; control G-0 (“control 1”), G-3 (“brackish-like”), G-1 (“marine-like”), G-2 (“≫marine”)) and group 2 (control G-4 (“control 2”), Schlei water next to Haithabu (G-5, “Schlei”) and Baltic Sea water next to Fehmarn (G-6, “Baltic”)), respectively. (B) δ13C data for α-cellulose and bulk in beach grass leaves (washed) as well as for dissolved inorganic carbon (DIC) in spray water and irrigation water for group 1 and group 2 (see above; modified after Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a). * = δ13CDIC values were measured via AMS (could contain fractionation effects from ionization) and are given only for indicating the magnitude in 13C composition (see indicated ranges (grey) in (A)); see Tables 2 and S1.
In contrast to the above mentioned treatments, plants of treatments G-2 (“>>marine”), G-5 (“Schlei”), and G-6 (“Baltic”) give distinctly more depleted 14C concentrations of 0.9736 ± 0.0027, 0.9670 ± 0.0026, and 0.9581 ± 0.0027, respectively (see Figure 3A and Table 2).
The plant tissue of treatments G-5 and G-6 exhibit radiocarbon concentrations close to the corresponding spray water samples in the range expected for modern Baltic Sea water (i.e., F14C 0.967 ± 0.002; Fischer and Olsen Reference Fischer and Olsen2021) in case of G-6 (“Baltic”) and rather close to the (pre-bomb) Western Baltic seawater value (ca. F14C 0.946; CALIB rev. 8, Stuiver and Reimer Reference Stuiver and Reimer1993; Heaton et al. Reference Heaton, Köhler, Butzin, Bard, Reimer, Austin, Bronk Ramsey, Grootes, Hughen, Kromer, Reimer, Adkins, Burke, Cook, Olsen and Skinner2020) in case of G-5 (“Schlei”; see Figure 3A, Table 2). While we cannot say anything about the isotopic signature of the spray water used for treatment G-2 (see above), the measured 14C concentration of the corresponding plant sample is comparable to the concentration measured in plant tissues of treatments G-5 and G-6, and again close to the range for Baltic Sea water, although these plants were not sprayed with natural brackish water (see section 2.1).
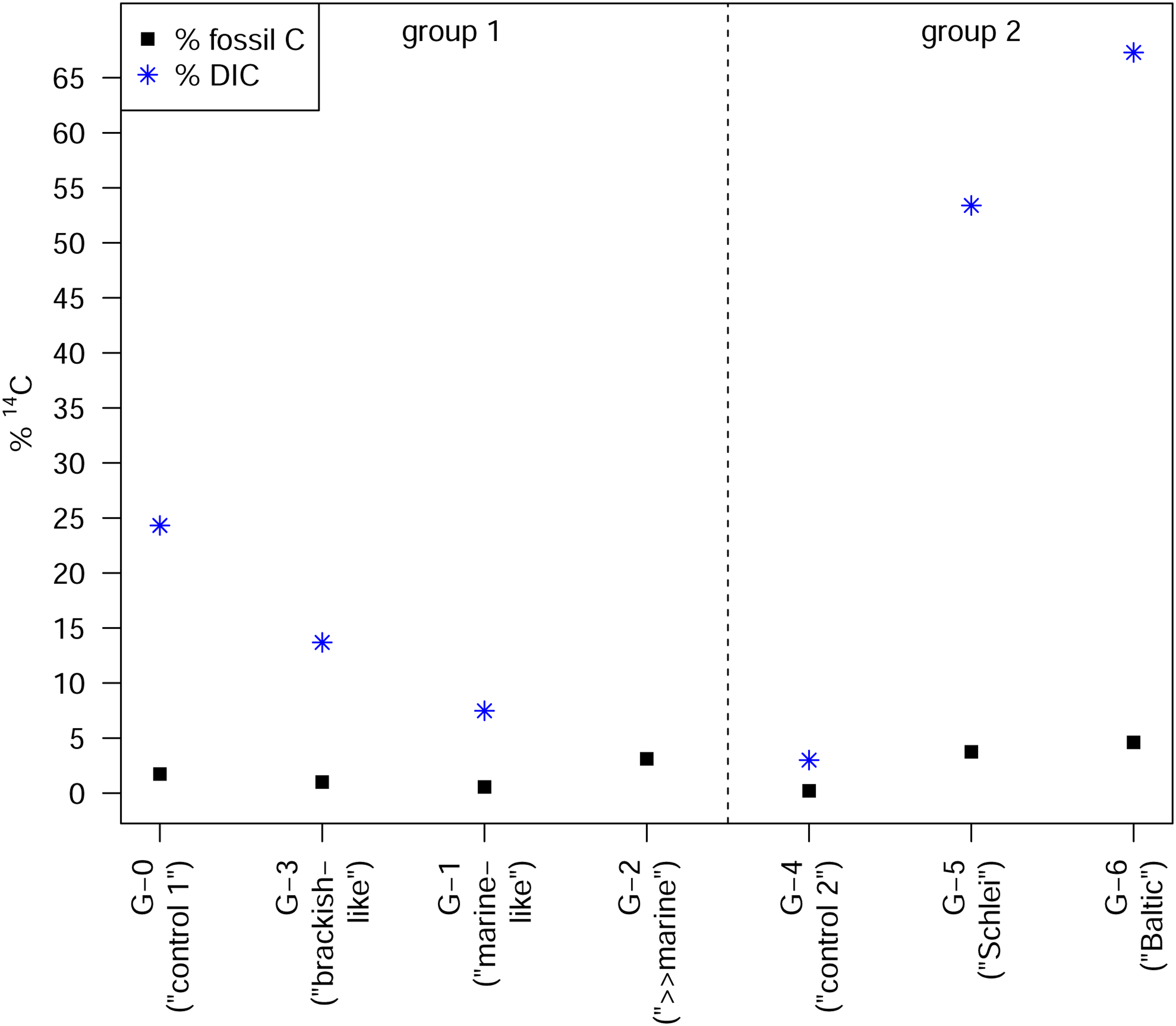
Assuming atmospheric CO2 was the major carbon source for the plants, we calculated the discrimination of the plants of the different treatment groups against atmospheric 13C (Δ13C) and 14C (ΔΔ14C) (see Eq. 6), respectively (see Table 3). The discrimination against atmospheric 13C as well as 14C decreases in plants of group 1 (non-salty) treatments G-3 (“brackish-like”) and G-1 (“marine-like”) compared to the corresponding control plants of G-0 (see Figure 4, Table 3). With an increasing mineral concentration in the spray water (G-0 < G-3 < G-1; see Table 1), the discrimination in 14C becomes smaller as compared to the discrimination against 13C (Figures S2 and S3) resulting in a lowering of the calculated ΔΔ14C/Δ13C ratio (Table 3). Plants of treatment G-2 (“>>marine”) show a discrimination against 13C of comparable magnitude as seen for G-1 and G-3. However, with respect to 14C the discrimination is about 2-times higher than the discrimination against 13C, comparable to group 2 treatments (Figures S2–S4, Table 3). For the group 2 plants of treatments G-4 (“control 2”), G-5 (“Schlei”), and G-6 (“Baltic”) the discrimination against atmospheric 13C decreases with increasing salinity (psu; see Table 1). In contrast, discrimination against 14C strongly increases in G-5 and G-6 plants compared to that found for the control treatment G-4 or the group 1 treatments. The isotopic discrimination against atmospheric 13C is, however, of comparable magnitude as seen in group 1 treatments of G-3 and G-1 (Figures S2A and S3A). Assuming a major carbon uptake via the atmosphere and a fossil 14C concentration of 0 F14C, the proportion of atmospheric fossil carbon in the plants can be calculated (see Eq. 3). The estimated fossil carbon contribution is low, is up to about 3% (G-2) in group 1 and up to about 5% (G-6) in group 2 (Figure 5, Table 2).
Discrimination against 13C (Δ13C) and 14C (ΔΔ14C) as well as calculated relative ratio of discrimination against 13C and 14C (ΔΔ14C/Δ13C) assuming atmospheric CO2 or DIC in spray water or modeled water as the only carbon source for the investigated plants. NA = not available
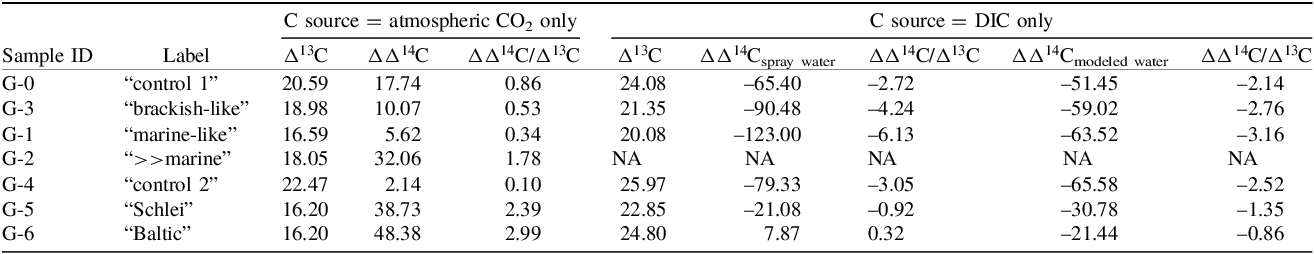
Discrimination against 13C (Δ13C) versus discrimination against 14C (ΔΔ14C) in atmospheric CO2 (filled symbols) for group 1 (mineral salt solution; control G-0 (“control 1”), G-3 (“brackish-like”), G-1 (“marine-like”), G-2 (“≫marine”)) and group 2 (control G-4 (“control 2”), Schlei water next to Haithabu (G-5, “Schlei”) and Baltic Sea water next to Fehmarn (G-6, “Baltic”)), respectively (see Table 3).
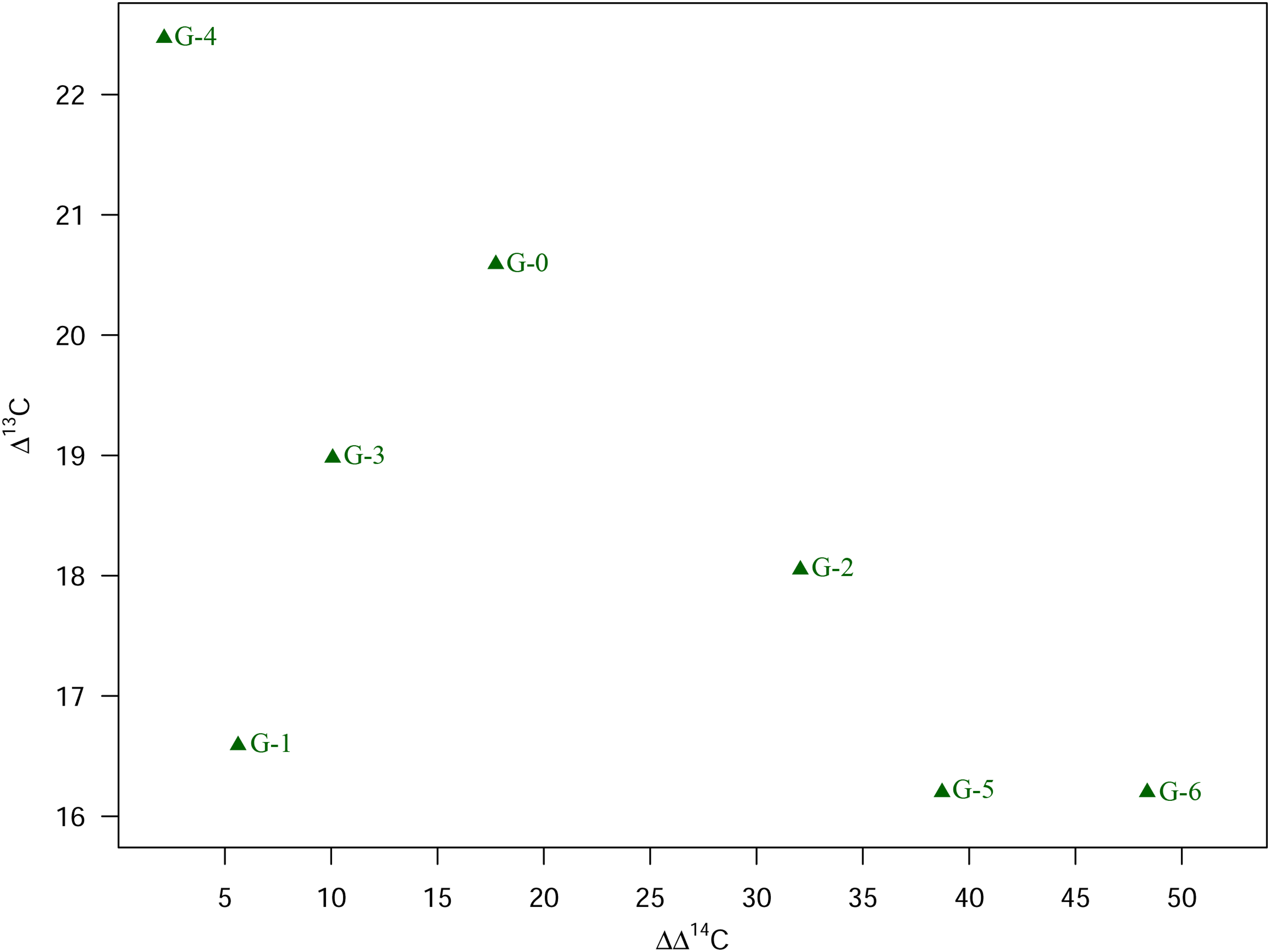
Calculated proportion of fossil (atmospheric) 14C as well as proportion of 14C from water (DIC) taken up by the greenhouse plants of group 1 (mineral salt solution; control G-0 (“control 1”), G-3 (“brackish-like”), G-1 (“marine-like”), G-2 (“≫marine”)) and group 2 (control G-4 (“control 2”), Schlei water next to Haithabu (G-5, “Schlei”) and Baltic Sea water next to Fehmarn (G-6, “Baltic”)), respectively; see also Table 2.
The above discussed carbon isotope discrimination factors were calculated relative to atmospheric CO2 as the sole carbon source for the plants and, thus, ignoring carbon derived from water, i.e. DIC. A comparable estimation against water DI13C isotopic composition, i.e. Δ13CDIC, is hampered by the fact that water δ13CDIC was only measured by AMS and not by conventional IRMS, thus also contain effects caused by the ionization process. Uncertainties in the analysis as well as accuracies are worse and measurements might deviate from conventional IRMS analyses by about 1.5–2‰. Nevertheless, a brief comparison of the discrimination effects relative to spray water DIC is given (see Table 3).
If the carbon uptake in the plant tissue during the growth experiment did not only occur via atmospheric CO2 but also (via the water path (irrigation and spray water), the proportion of 14C from DIC can be calculated. According to Eq. (4), plants of G-5 (“Schlei”) and G-6 (“Baltic”) received about 53% and 67% of the carbon from DIC. The percentage of 14C originating from DIC is lower for plants of group 1 with values between 7% (G-1) and 24% (G-0) (Figure 5). Unfortunately, no water DIC proportion could be calculated for G-2 (for reasons given above). As visible from Figure S4, the percentage of 14C taken up from DIC by group 2 plants G-5 and G-6 increases with increasing salinity levels, compared to their control group G-4. We observe an opposite such trend for plants of group 1, with (slightly) increasing DIC, [Na+], and [Cl–] levels (G-0 < G-3 < G-1; see Figures S2, S4–S6). Figure 3B illustrates that the δ13Ccellulose values of the plants are not only influenced by the δ13CDIC value of the irrigation water (–3.6‰; Figure 3B, Table S1) or of atmospheric CO2 (ca. –8.5‰; Cernusak and Ubierna Reference Cernusak and Ubierna2022). With δ13CDIC values of about –1.5‰ (G-5, “Schlei”) and 0.4‰ (G-6, “Baltic”) the spray water samples are as expected for Baltic Sea water (brackish water) and seawater (δ13CDIC ≈ 1.5‰ ± 0.8‰) in general (Hoefs Reference Hoefs2021; Kroopnick et al. Reference Kroopnick, Weiss and Craig1972; Kroopnick Reference Kroopnick1985).
The sea spray effect on the stable and radiogenic isotopic signature of the sprayed plants can also be visualized for δ34S and 87Sr/86Sr (Figures S7B and S8B, Table S1): Plants of treatments G-5 and G-6 are shifted towards seawater values with δ34S ≈ 20–21‰ and 87Sr/86Sr ≈ 0.7092, respectively (Andersson et al. Reference Andersson, Wasserburg and Ingri1992; Rees et al. Reference Rees, Jenkins and Monster1978; Tostevin et al. Reference Tostevin, Turchyn, Farquhar, Johnston, Eldridge, Bishop and McIlvin2014). While plants of G-3 are enriched in 34S compared to control G-0 and while plants of G-1 are enriched in 34S compared to G-3, depletion in 34S is observable for G-2 (Figure S7B, Table S1). With respect to 87Sr/86Sr, plants of group 1 are shifted towards lower values, i.e. towards the isotope ratio of their corresponding spray water, with increasing Sr concentration. G-2 plants, sprayed with water exhibiting the highest Sr concentration within group 1, are shifted the most (Figure S8B, Table S1; see also Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a).
We, thus, have to distinguish two different models of behavior in both the stable isotope fingerprints as well as the radiocarbon signature (F14C, Δ14C) as a result of the artificial sea spray treatment: a shift towards a seemingly marine stable isotope as well as radiocarbon signature (G-5, G-6) in the (salty) group 2 and a shift towards a seemingly marine radiocarbon signature, but divergent stable isotope data (G-2; (mineral salt solution) group 1), resulting from different mechanisms (see section 4.1).
4. Discussion
4.1 Sea spray effect, salinity stress, HCO3– stress, and 14C analysis of plant samples
Our study demonstrates the impact of sea spray on radiocarbon analyses of terrestrial plants. In principle, two types of spray water have been used, i.e. (i) mineral salt solution, originating from tap water, containing increasing ion concentrations (e.g., DIC, Na+/Cl–), and slightly higher radiocarbon concentrations (group 1; G-1 to G-3; control G-0), and (ii) salty water (higher NaCl concentration), originating from marine sources (group 2; G-5 and G-6; control G-4).
With increasing ion concentration within the (non-salty) spray water of group 1, the radiocarbon content in the water decreases (G-0: 0.9191 ± 0.0029, G-3: 0.9006 ± 0.0030; G-1: 0.8707 ± 0.0029; see Figure 3A), probably the result of differences in the DIC content (see also Figure S1). This is in accordance with the findings of Wang et al. (Reference Wang, Lin, Burr, Wang and Lin2022) who found a linear relationship between 14C concentration and 1/[DIC]. For spray waters of group 2 with larger NaCl concentrations we observe higher 14C concentrations compared to the tap water (see Table 2).
Soil respiration is known to be a potential source of atmospheric CO2, originating from soil carbon. Soil respiration is mainly driven by three biological processes, namely microbial respiration, root respiration, and faunal respiration and soil manuring has a high impact on soil respiration. While microorganisms might have been naturally present in the soil used for the experiment, no larger organisms (e.g. earthworm) were present in the soil. Thus, we can exclude faunal respiration. The soil was not manured during the experiments. Thus, manuring effects on the soil can also be excluded. Sandy soils, as used for this experiment (see section 2.1), typically have a low SOM (soil organic matter) and SOC (soil organic carbon) content and low available water capacity. This limits soil respiration and mineralization of nitrogen. Therefore, sandy soils show a very low soil respiration activity. In ecosystems where the soil CO2 production is very low due to low biological activity, CO2 consumptive processes (dissolution in water, biological activities, chemical reactions) in the soil are higher than CO2 production processes (e.g. Sánchez-Cañete et al. Reference Sánchez-Cañete, Barron-Gafford and Chorover2018). The impact of SOC and DIC as source of atmospheric CO2 via respiration processes can be neglected in our study. In addition, any minor impact would have affected the atmosphere of all treatment groups, growing under the same atmosphere in the climate chamber, including the control groups. The radiocarbon content of the plants is known due to the experimental design and should reflect the atmospheric 14C concentration during the growth period, i.e. they should exhibit a modern 14C signature (group 1, June–December 2020: F14Catm = 1.0048, group 2, July–December 2020: F14Catm = 1.0044). The atmospheric 14C concentration should only be understood as an approximation for the climate chamber’s atmosphere, reflecting the HPB atmosphere during the growth period. F14C values close to the HPB atmospheric F14C values are observed for treatment G-4 (“control 2”; 1.0023 ± 0.0028). In contrast, plants of the control treatment G-0 (0.9873 ± 0.0028) exhibit an unexpected low 14C concentration, compared to the contemporary atmospheric concentration. Differences between control plants of G-0 and G-4 are also observable to some extent with respect to δ13Ccellulose (G-0: –28.01‰, G-4: –29.80‰), δ34Stotal S (G-0: 5.50‰, G-4: 5.96‰), and 87Sr/86Sr (G-0: 0.709340, G-4: 0.708845) (see Table S1). The corresponding δ18Ocellulose values, however, do not differ (G-0: 30.98‰, G-4: 31.01‰; see Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a for details).
The control groups grew in the same greenhouse chamber, thus under the same atmosphere, and were treated identically to each other (sprayed with identical spray water, irrigated with identical tap water). The group 1 experiment started in June 2020, while the group 2 experiment started in July 2020. These differences (also with respect to atmosphere) should be minor; however, a potential effect of the different starting point (and run time) cannot fully be excluded. Another difference potentially affecting the radiocarbon concentration in the G-0 and G-4 plants could result from the fact that two different seed batches, delivered in two different packages, were used when growing the plants of group 1 and 2, respectively. In addition, a new bag of lawn soil and lawn sand has to be used to set up the soil for group 2. We cannot fully exclude differences in potentially different stocks of seeds or soil/sand. Preliminary isotope analyses on seed, plant, and soil samples (δ13Ccellulose, δ18Ocellulose, δ34Stotal S, 87Sr/86Sr), however, point to inter-individual differences rather than batch differences (see Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a).
Accordingly, differences in the control plants of G-0 and G-4 with respect to F14C as well as stable isotope values might partly be due to differences within the growth period, with a shorter growth period (G-0: June–December; G-4: July–December), with some additional influences of potential inter- (or also intra-) plant variation (see Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a for intra-leaf variation in δ13Ccellulose and δ18Ocellulose). Further analyses, including multiple isotopic measurements of samples of the same treatment groups, are required to investigate the observed differences.
The treatments G-1 (“marine-like”) and G-3 (“brackish-like”) also exhibit radiocarbon concentrations slightly lower than the (expected) atmospheric value, indicating some impact of DIC via spray or irrigation water, or both.
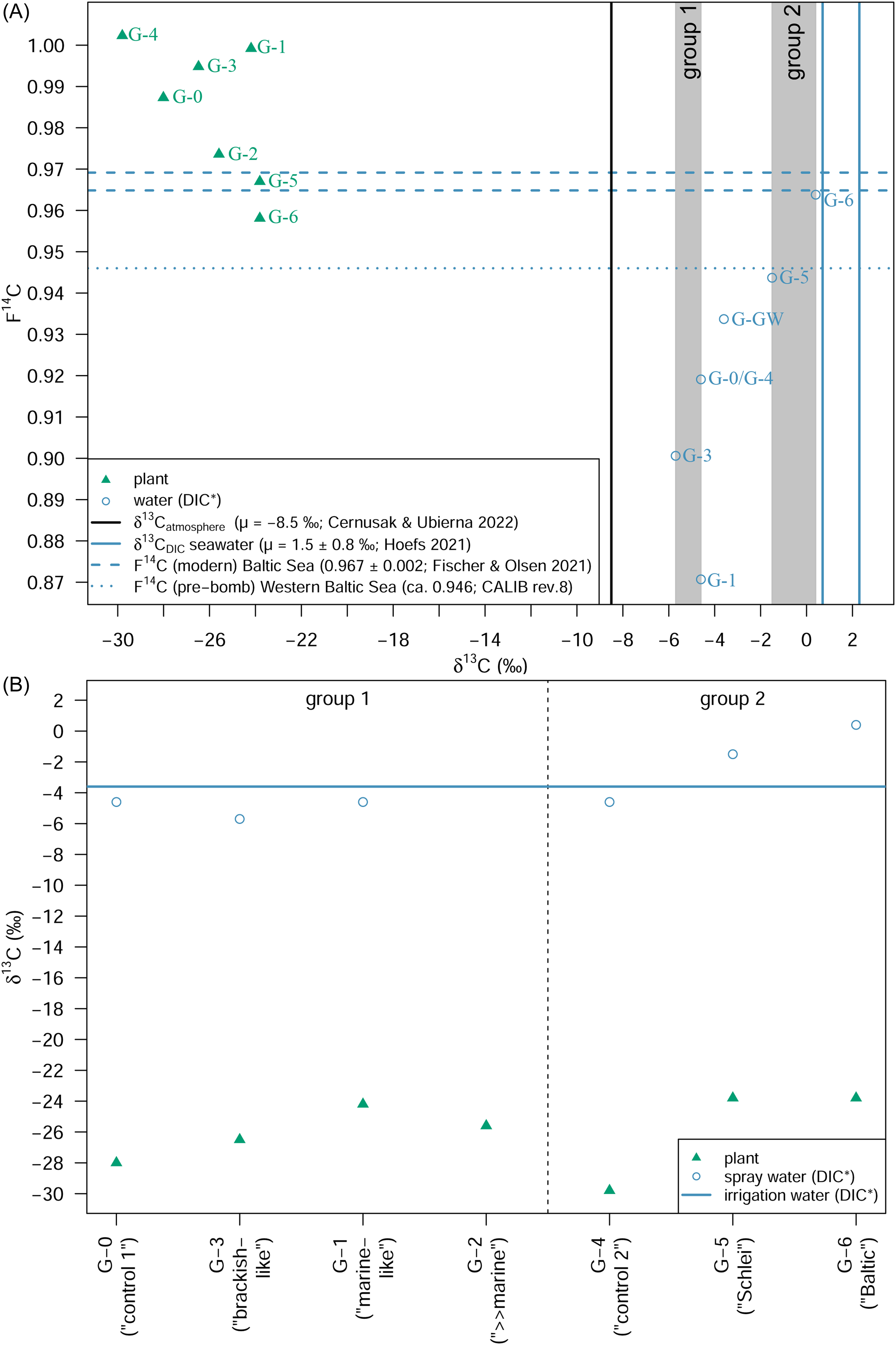
Figure 6 shows that the δ13C and Δ14C signatures of the investigated plant treatment groups are influenced in different ways. We can differentiate between several influencing factors that affect the δ13C or the Δ14C signature of the plants. The processes involved are explained in the following:
δ13C values for plants, atmospheric CO2 and spray water (DIC) as well as irrigation water (DIC; G-GW) plotted against the corresponding age-corrected Δ14C values for the different treatment groups (see Tables 2 and S1): group 1 (mineral salt solution; control G-0 (“control 1”), G-3 (“brackish-like”), G-1 (“marine-like”), G-2 (“≫marine”)) and group 2 (control G-4 (“control 2”), Schlei water next to Haithabu (G-5, “Schlei”) and Baltic Sea water next to Fehmarn (G-6, “Baltic”)). The arrows indicate major effects on the isotope values (see text). The bars on the edges serve for an illustration of the range of the δ13C and Δ14C values in plant (green), spray water (blue), atmosphere (black). [color online].
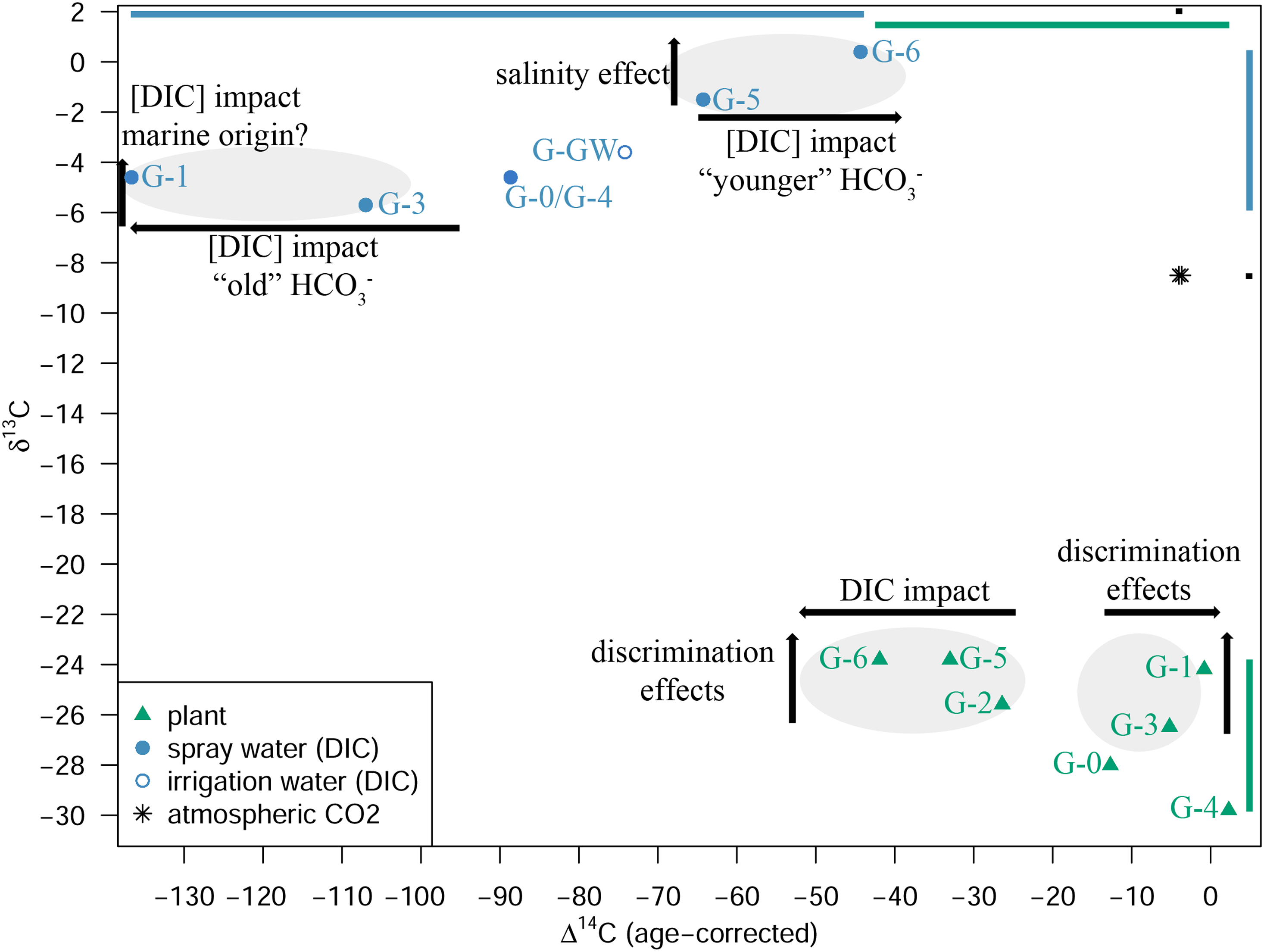
Figures S5 and S6 illustrate decreased discrimination against both 13C and 14C with increasing levels of, e.g., Na+ or Cl-, when considering plants of the G-0 (“control 1”), G-3 (“brackish-like”), and G-1 (“marine-like”) treatment groups. The DIC concentration (i.e., HCO3 –) apparently has a similar effect on the isotopic composition of plants of treatment groups G-3 and G-1 (Figures S1–S4, see also Table 3). As shown by Tavakkoli et al. (2011; Reference Tavakkoli, Rengasamy and McDonald2010) stomatal conductance decreases with increasing levels of both Na+ and Cl–. While the Na+ levels are comparably low in the irrigation water (G-GW; 9.70 mg/L) as well as the spray water of G-3 (37.82 mg/L) and G-1 (75.63 mg/L), respectively, both spray water sources contain already rather high levels of Cl− (G-3: 558.65 mg/L; G-1: 1117.30 mg/L). Far lower Cl− levels can already have a distinct impact on stomatal conductance, CO2 assimilation rate, or on the ratio of intracellular versus atmospheric CO2 concentration (ci/ca) (Tavakkoli et al. 2011; Reference Tavakkoli, Rengasamy and McDonald2010). The DIC concentration in spray water used for treatments G-0/G-4, G-3, and G-1 varies between 298.4 mg/L (G-0/G-4) and 331.7 mg/L (G-1) which is distinctly more than the amount measured in the natural salty waters of the Schlei inlet (G-5, 125.6 mg/L) and the Baltic Sea (G-6, 79.6 mg/L; see Table 1). A higher amount of DIC available to the plants should elevate partial intercellular CO2 pressure (pi) via carbonic anhydrase reactions (HCO3 – → CO2), resulting in an increased discrimination against 13C as well as 14C. Accordingly, for group 1 treatments, increasing Cl– as well as elevated DIC concentrations could cause a reduced stomatal conductance, resulting in decreased isotopic discrimination, as documented in Table 3 and Figure 6 (see also Figures S2 and S6).
Plants of treatments G-5 (“Schlei”) and G-6 (“Baltic”) give 14C concentrations of F14C 0.9670 (± 0.0026) and F14C 0.9581 (± 0.0027), respectively. In addition, also plants of treatment G-2 (“>>marine”) show a low 14C concentration (0.9736 ± 0.0027; Figure 3A, Table 2). Plants of G-5 (“Schlei”) and G-6 (“Baltic”) grew under salinity stress as spray water of both treatment groups contained quite high Na+ and Cl– levels (see Table 1). These concentrations are markedly higher than that found for the other spray water of group 1 as well as higher than those in tap water used as spray water source or for irrigating the plants of all treatment groups.
On a physiological level, salinity causes reduced stomatal conductance in plants (Brugnoli and Lauteri Reference Brugnoli and Lauteri1991; Poschenrieder et al. Reference Poschenrieder, Fernández, Rubio, Pérez, Terés and Barceló2018; Seemann and Critchley Reference Seemann and Critchley1985), which in turn has an effect on e.g., δ13Ccellulose in plants, expressed by enrichment in 13C (Figure 3B, Table 3) (Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a; Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023b; Roden et al. Reference Roden, Bowling, McDowell, Bond and Ehleringer2005; Zhang et al. Reference Zhang, Hartmann, Gleixner, Thoma and Schwab2019). Low stomatal conductance causes a decrease in the intercellular partial pressure of CO2 (pi) and, thus, a reduced intercellular CO2 concentration (ci) (Seemann and Critchley Reference Seemann and Critchley1985; Winter Reference Winter1981). Under salt stress, the ratio of intercellular and atmospheric CO2 concentration (ci/ca) is reduced (Farquhar et al. Reference Farquhar, OʼLeary and Berry1982). Accordingly, discrimination against 13C decreases with increasing salinity levels as seen in Δ13C of treatment groups G-4 and G-5/G-6 (see Figures 6 and S3A, Table 3). We would expect similar and even more pronounced effects for ΔΔ14C, but instead plants of treatments G-5 and G-6 show a ΔΔ14C of about 38‰ and 48‰, respectively, distinctly larger than in plants of control G-4 (6.6‰) or group 1 treatments G-1 and G-3 (Figure 6, Table 3). Apparently, some carbon, depleted in 14C relative to atmospheric carbon, still enter the plant as H14CO3 – via the roots. The proportion of 14C originating from DIC within both spray water and irrigation water (%DIC) in the plants increases in treatment G-5 (ca. 53%) and G-6 (ca. 67%) compared to the corresponding control plants of G-4 (“control 2”; ca. 3%; see Figures 5 and S4, Table 2). Although atmospheric CO2 uptake is reduced due to stomatal closure, the high amount of available DIC (i.e., HCO3 –) in the brackish water (G-5, G-6) – compared to the amount of DIC available to the control plants G-4 – results in an increased discrimination against 14C (see also Figure 6), which is even more pronounced than the reverse effect of reduced stomatal conductance. Interestingly, we do not see this effect in the corresponding Δ13C values. The reduced stomatal conductance due to salinity stress seems to dominate the discrimination against 13C, masking the opposite effect of the DIC taken up by the plants.
Clearly, our plant growth experiment under two different spray water regimes (group 1 vs. group 2) seem to indicate two different reactions of the plant metabolism. Plants of group 1 treatments, sprayed with increasing amounts of mineral (including DIC), enriched non-salty tap water, indicate that plants react with increasing stomatal closure, limiting thereby access of atmospheric CO2. As a result, discrimination against the heavier carbon isotopes decreases. At certain DIC concentrations and with additionally increasing salinity levels (group 2), stomatal closure must be so tight that the only remaining carbon source for plant tissue formation via photosynthesis could be the DIC originating from the irrigation water or the mixture of irrigation and spray water, as indicated by the apparent large proportion of carbon in plant tissues of group 2 treatments G-5 and G-6 originating from DIC (Figure S4, Table 2). This is also reflected by the large discrimination of the plants of G-5 and G-6 against atmospheric 14C, but comparably low discrimination against water DI14C (Figure 4, Table 3). To some extent, treatment G-2, sprayed with (non-salty) water containing very high mineral salt concentration (including DIC) seem to indicate a similar process as seen in salt-water sprayed treatments (G-5 and G-6).
G-2 plants (“>>marine”) exhibit a 14C concentration which is distinctly too depleted, namely 0.9736 ± 0.0027 (Table 2). As the corresponding spray water contained only traces of NaCl (see Table 1), this shift cannot be explained by salinity stress. The plants of G-2 faced several types of stress, including HCO3 – stress, relevant for the present study:
Stomatal closure is not only caused by salinity but also by high CO2 or HCO3 – concentrations. The critical concentration level is probably species-specific (see, e.g., Engineer et al. Reference Engineer, Hashimoto-Sugimoto, Negi, Israelsson-Nordström, Azoulay-Shemer, Rappel, Iba and Schroeder2016; Kolla et al. Reference Kolla, Vavasseur and Raghavendra2007; Misra et al. Reference Misra, De Armas, Tong and Chen2015; Mrinalini et al. Reference Mrinalini, Latha, Raghavendra and Das1982; Tian et al. Reference Tian, Hou, Ren, Pan, Jia, Zhang, Bai, Zhang, Zhu and He2015). Under high HCO3 – concentration, the excess HCO3 – can also be excreted via the roots (Poschenrieder et al. Reference Poschenrieder, Fernández, Rubio, Pérez, Terés and Barceló2018).
G-2 plants discriminate about twice as much against atmospheric 14C (ca. 32‰) than against atmospheric 13C (ca. 18.05‰), as would be expected for, e.g., chemical processes (O’Leary Reference OʼLeary1981; Stern and Vogel Reference Stern and Vogel1971). Although the Na+ and, in particular, Cl– concentration of the spray water of G-2 plants was higher than that of the G-1 plants, there is no further decrease in discrimination compared to G-1 plants (see Table 2). Instead, high HCO3 – concentration in the spray water results in increased intercellular partial pressure of CO2, due to the conversion of HCO3 – to intercellular CO2 by carbonic anhydrases, and, thus, an increased pi/pa ratio. The resulting high pi is reflected by an increased discrimination against both 13C and 14C (see Figure 6).
The proportion of 14C in plants originating from DIC is remarkably high (see Table 2), especially for the plants of the treatment groups G-5 (Schlei water; ca. 53%) and G-6 (Baltic Sea water; ca. 67%). The observed DIC effect and the underlying mechanisms seem to be similar to the freshwater reservoir effect observable in, e.g., aquatic plants (e.g., Ascough et al. Reference Ascough, Cook, Church, Dugmore, McGovern, Dunbar, Einarsson, Friðriksson and Gestsdóttir2007; Geyh et al. Reference Geyh, Schotterer and Grosjean1998; Philippsen Reference Philippsen2013). Freshwater bodies can show a (variable) depletion in 14C, resulting from the input of groundwater containing DIC originating from the dissolution of calcareous bedrock, old soil carbonates, and geothermal processes (e.g., Ascough et al. Reference Ascough, Cook, Church, Dugmore, McGovern, Dunbar, Einarsson, Friðriksson and Gestsdóttir2007; Geyh et al. Reference Geyh, Schotterer and Grosjean1998; Philippsen Reference Philippsen2013). The 14C-depleted DIC, incorporated by the plants, has a distinct impact on the radiocarbon concentration of the plants and also enters the food chain, thus affecting the radiocarbon concentration of consumers of freshwater food. However, DI14C can also affect the radiocarbon concentration of terrestrial plants.
As demonstrated by 14C tracer studies, the uptake of H14CO3 – into plant tissues depends on various factors, including its concentration (e.g., Pelkonen et al Reference Pelkonen, Vapaavuori and Vuorinen1985; Vapaavuori and Pelkonen Reference Vapaavuori and Pelkonen1985; Viktor and Cramer Reference Viktor and Cramer2005), the overall nutrient supply (e.g., Werth and Kuzyakov Reference Werth and Kuzyakov2005), and physiological factors such as water stress (e.g., Bota et al Reference Bota, Stasyk, Flexas and Medrano2004). Distinct species-differences (e.g., Pelkonen et al Reference Pelkonen, Vapaavuori and Vuorinen1985) as well as differences between bicarbonate uptake in different plant organs or compounds are observable (e.g., Bialczyk and Lechowski Reference Bialczyk and Lechowski1992; Domanski et al Reference Domanski, Kuzyakov, Siniakina and Stahr2001; Pelkonen et al. Reference Pelkonen, Vapaavuori and Vuorinen1985; Vapaavuori and Pelkonen Reference Vapaavuori and Pelkonen1985; Werth and Kuzyakov Reference Werth and Kuzyakov2005; Zamanian et al. Reference Zamanian, Pustovoytov and Kuzyakov2017). Accordingly, the published proportion of 14C derived from carbonates and incorporated in plant tissues varies.
Schäfer (Reference Schäfer1988) observed that after root labeling with NaH14CO3 almost 50% of the 14C was incorporated into the sugar and starch fraction of spring wheat (Ador sp.). Acid-stable plant products (= excluding inorganic carbon) contained up to 79% of 14C taken up from the medium labeled with NaH14CO3 in willow (Salix aquatic gigantean) and up to 63% in sunflower (Helianthus annuus) plants (Pelkonen et al. Reference Pelkonen, Vapaavuori and Vuorinen1985; Vapaavouri and Pelkonen Reference Vapaavuori and Pelkonen1985). The proportional contribution of root-derived 14C to leaf total photosynthesis in paper mulberry (Broussonetia papyrifera) was about 28%, measured 10 and 20 days after treatment. In contrast, for white mulberry (Morus alba) after 10 days about 8% DIC input was measured, while it was 0% after 20 days due to low root-zone bicarbonate concentration (Wu and Rao Reference Wu, Rao, Wu and Rao2023; Wu and Xing Reference Wu and Xing2012).
Zamanian et al. (Reference Zamanian, Pustovoytov and Kuzyakov2017) investigated root- and shoot-labeled field gromwell (Buglossoides arvensis), growing on sand and loess soil, respectively. Fruits of plants growing on calcareous soil exhibit apparent 14C ages which are overestimated by several hundred years. The proportion of 14C taken up from soil pore water containing dissolved carbonates and stored in fruit carbonates was estimated to about 6% (Zamanian et al. Reference Zamanian, Pustovoytov and Kuzyakov2017), thus, much lower than for our plants growing under salinity stress (ca. 53% (G-5) and 67% (G-6); Table 2). The physiological stress conditions of the G-5 and G-6 plants most likely enhanced the uptake of DIC (see above), thus resulting in the observed high DI14C proportions. This example actually demonstrates the large effect of the uptake of even a small proportion of H14CO3 – on the 14C concentration in plants (Zamanian et al. Reference Zamanian, Pustovoytov and Kuzyakov2017). The available proportion of DI14C in the spray water of both G-5 (Schlei water; 125.6 mg/L) and G-6 (Baltic Sea water; 79.6 mg/L; see Table 1) was higher than that in the calcareous soil in the study by Zamanian et al. (Reference Zamanian, Pustovoytov and Kuzyakov2017; ≤ 13 mg/L). Seawater usually contains HCO3 – levels of about 140 mg/L (Wright and Colling Reference Wright, Colling, Suckow, Weisbroth and Franklin1995b). The HCO3 – concentration in the spray water of G-2 was even higher (ca. 191 mg/L HCO3 – in mineral salt (Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a), equivalent to about 436 mg/L CO2 after dissolving the mineral salt in tap water G-0).
The resulting shift in the 14C concentration due to the admixture of assumed 14C free fossil carbon in G-6 (measured F14C 0.9581) vs. the contemporary atmospheric carbon concentration (F14C: 1.0044) with a calculated fossil carbon contribution of about 4.6% (see Figure 5 and Table 2) due to salinity or HCO3 – stress is similar to the shift observed by Zamanian et al. (Reference Zamanian, Pustovoytov and Kuzyakov2017) (ca. 500 14C years, equivalent to about 6% in F14C), resulting from the uptake of soil carbonate derivatives. It is conceivable that the actual shift, caused by either a direct or an indirect sea spray effect, could be even higher than observed in this pilot study. However, both the observed results and the potential variation in the DI14C contribution of coastal plants needs to be further verified in the future. Besides the sea spray effect, our findings also suggest that changes in the isotopic composition or ion concentration of groundwater, respectively pore water can affect the radiocarbon data of the investigated plants. A changed proportion of 14C from DIC taken up by the plants causes shifts in the radiocarbon dating. This can, e.g., also be of interest for regions which are (regularly) flooded, not only by seawater but also by terrestrial aquatic sources with a depleted 14C signature. In addition, physiological reactions in plants, besides salinity and HCO3 – stress, as a result from environmental conditions (e.g., temperature, solar radiation, precipitation, aridity/humidity, drought stress) may affect radiocarbon dating as well. This is especially true for those environmental factors influencing the stomatal conductance as well as the photosynthesis rate, also affecting the δ13C signature of plants (see section 1).
It is important to differentiate between an “indirect” sea spray effect which is caused by associated physiological reactions in the plants as a result from, e.g., salinity stress and a “direct” sea spray effect, caused by the uptake of ions of marine origin (aerosols). The indirect and direct sea spray effect can be distinguished with respect to the stable isotope fingerprints of the treated plants of group 1 and 2. A direct sea spray impact on the radiocarbon signature is visible by shifted radiocarbon concentrations towards values expected for seawater or similar to the marine spray water (see G-5 and G-6). Apparently, the plants of G-5 and G-6 take up a calculated proportion of (old) fossil 14C of about 3.7% and 4.6%, respectively, incorporated in the plants (Figure 5, Table 2). Comparison of the fossil 14C contribution in control plants G-4 (0.2%) with that in G-5 and G-6 plants suggests that the fossil 14C signal detected in the plant tissue of G-5 and G-6 originates from the spray water, whereas the signal in the corresponding control groups can be understood as atmospheric (or irrigation water) background noise with only low anthropogenic impact, relative to the HPB atmospheric data. Our values are distinctly lower than those observed for industrial (urban) regions with values of even 10% (e.g., Varga et al. Reference Varga, Barnucz, Major, Lisztes-Szabó, Jull, László, Pénzes and Molnár2019). The fossil 14C signal in seawater, resulting in the marine reservoir effect (see section 1), also slightly affects the plants of treatment group 2, which were sprayed with brackish water from the Baltic Sea and the Schlei inlet, which is fed by Baltic Sea water, thus contributing to the overall fossil 14C contribution in the plants. We can see smaller such values for plants of treatment group 1, nevertheless, a distinct uptake of fossil 14C cannot be demonstrated with the current experimental settings (see Figure 4, Table 3). Using (much) older tap water for both irrigation and spraying (mineral salt solutions) than the used Munich tap water could result in clearer results with respect to fossil 14C, thus, for differentiating between the indirect and direct sea spray effect on radiocarbon analyses. As outlined above, the Δ14C values of the plants of the treatment groups G-5 and G-6 are mainly affected by the uptake of 14C from DIC (and respective DIC/HCO3 – concentration levels; see also Figures S2B and S4). The applied spray water results in reduced stomatal conductance, which in turn causes a decrease in carbon isotopic discrimination against both 13C and 14C, also documented by decreased ΔΔ14C/Δ13C ratios (Table 3). At the present stage of research, it is not possible to clearly distinguish between the influence of DIC in irrigation water and in spray water (Schlei water, Baltic Sea water). However, it seems likely that the higher HCO3 – levels as well as the lower 14C concentrations in the spray water have a higher impact than tap water used for irrigating the plants. Nevertheless, further experiments are required to verify the impact of both spray and irrigation water.
It became apparent that the tap water used for spraying the control groups (G-0/G-4) or used for the preparation of the mineral salt solution for group 1 treatment, taken in the laboratory, was different from tap water used for irrigating all plants (G-GW), taken in the climate chamber of the greenhouse. This is not only the case with respect to the stable isotope data (δ13CDIC: G-0/G-4: –4.6‰, G-GW: –3.6‰; 87Sr/86Sr: G-0/G-4: 0.708335, G-GW: 0.708280; Table S1) and the (trace) elemental composition (e.g., [Ca]: G-0/G-4: 99.51 mg/L, G-GW: 50.49 mg/L; [Sr]: G-0/G-4: 181.82 µg/L, G-GW: 157.78 µg/L; see also Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a), but also with respect to radiocarbon dating (G-0/G-4: 678 ± 26 BP, G-GW: 551 ± 27 BP) or F14C (G-0/G-4: 0.9191 ± 0.0029, G-GW: 0.9337 ± 0.0031). Laboratory and greenhouse were located in two adjacent buildings of the LMU Biocenter, obviously using different water pipes, i.e., galvanized pipes in the laboratory building (containing traces of Zn and Pb). The measured calcium as well as DIC concentration for G-0/G-4 ([Ca]: 99.51 mg/L, [DIC]: 298.4 mg/L) and G-GW water ([Ca]: 50.49 mg/L, [DIC]: 162.5 mg/L; see Table 1 and Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a) also points to differences in the overall calcium carbonate concentration in the two tap water sources. Furthermore, the residence time of the water in the pipe of the greenhouse chamber was most likely different from that in the pipe of the laboratory, located in different buildings and at different floors. It also has to be emphasized that the presented isotopic composition of the irrigation water samples and the tap water used to prepare the mineral solutions of group 1 and for spraying the control groups could have (slightly) varied over the growth experiment. However, all variations would have equally influenced all plants, including the control groups.
4.2 Dealing with the marine reservoir effect and the sea spray effect in 14C data
The magnitude of the salinity-induced (indirect) sea spray effect, visible in plants grown under salinity stress (G-5 (“Schlei”): 269 ± 22 BP; G-6 (“Baltic”): 344 ± 23 BP; see section 4.1), is comparable to the (modern) marine reservoir age of the Baltic Sea (273 ± 18 14C years) as determined by Fischer and Olsen (Reference Fischer and Olsen2021) or the (pre-bomb) seawater reservoir age in the Western Baltic Sea of about 445 14C years (Stuiver and Reimer Reference Stuiver and Reimer1993; Heaton et al. 2023; Reference Heaton, Köhler, Butzin, Bard, Reimer, Austin, Bronk Ramsey, Grootes, Hughen, Kromer, Reimer, Adkins, Burke, Cook, Olsen and Skinner2020). Since our analyses are based on a very small dataset, these findings are not yet valid. Nevertheless, our data imply that the sea spray effect on 14C might behave in a similar way as a reservoir effect, with certain dependence on physiological reactions in the plants (see section 4.1).
Consumption of marine food sources by humans is known to affect the radiocarbon dating of these humans (marine reservoir effect; see section 1). Humans - or omnivores in general - living in coastal regions or obtaining their food from such regions can show a 14C age intermediate between that of marine mammals (e.g., seal) and terrestrial herbivorous mammals (e.g., deer), depending on their dietary composition (see, e.g., Yoneda et al. Reference Yoneda, Tanaka, Shibata, Morita, Uzawa, Hirota and Uchida2002).
A marine impact on 14C is commonly controlled by investigating the δ13Ccollagen and δ15Ncollagen values which, beyond others, can give information on the consumption of marine protein sources (see section 1). Based on the isotopic signature of these two isotopic systems, terrestrial herbivores are supposed to be unaffected by the marine reservoir effect and radiocarbon data are corrected against the calculated marine reservoir age based on a marine and terrestrial end member (see, e.g., Ascough et al. Reference Ascough, Cook and Dugmore2005; Cook et al. Reference Cook, Ascough, Bonsall, Hamilton, Russell, Sayle, Scott and Bownes2015; Dury et al. Reference Dury, Eriksson, Fjellström, Wallerström and Lidén2018; Sayle et al. Reference Sayle, Cook, Ascough, Gestsdóttir, Hamilton and McGovern2014). However, even if herbivorous mammals have not consumed any marine food, there can be a marine bias due to the consumption of plants influenced by the sea spray effect. So far, the marine impact on terrestrial samples caused by sea spray aerosols has not been addressed.
The sea spray signal enters the isotopic fingerprint along the terrestrial food chain. Accordingly, this impact must not be ignored in both stable isotope as well as radiocarbon analyses. Neither δ13Ccollagen nor δ15Ncollagen are capable of giving hints on a potential sea spray impact as no marine but terrestrial protein (e.g., terrestrial plants) is consumed in such a case (Göhring et al. Reference Göhring, Mauder, Vohberger, Nehlich, von Carnap-Bornheim, Hilberg, Kröger and Grupe2018; Göhring et al. Reference Göhring, Mayr and Grupe2020). Accordingly, investigating only these two isotopic systems would be insufficient at coastal sites. For identifying a sea spray effect in archaeological skeletal remains, the investigation of additional isotopic systems is, thus, required. The sea spray signal in, e.g., α-cellulose carbon (δ13Ccellulose) is, moreover, also transferred to the terrestrial consumer via the diet, resulting in a seemingly marine isotope signal in the δ13Ccarbonate values of terrestrial animals as well as humans (Göhring et al. Reference Göhring, Mauder, Vohberger, Nehlich, von Carnap-Bornheim, Hilberg, Kröger and Grupe2018, Reference Göhring, Mayr and Grupe2020). This was further evidenced in recent studies (Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023a, Reference Göhring, Hölzl, Mayr and Strauss2023b). Accordingly, it is mandatory to control for the sea spray by measuring (several) other isotopic systems potentially affected by sea spray. By investigating both terrestrial herbivores and marine piscivorous mammals it is possible to identify a potential sea spray impact and correct for the sea spray also in human consumers (Göhring et al. Reference Göhring, Mauder, Vohberger, Nehlich, von Carnap-Bornheim, Hilberg, Kröger and Grupe2018, Reference Göhring, Mayr and Grupe2020).
4.3 Potential implications for bioarchaeological studies
While our results have to be understood as preliminary data and require additional detailed investigations based on a larger dataset, the observed indirect sea spray effect on radiocarbon data of (modern) terrestrial plants, visible in F14C as well as in the apparent 14C age, is of interest for archaeological studies in coastal regions. With respect to stable isotopes, the sea spray effect identified in the greenhouse experiments was also validated in environmental samples collected next to the Baltic coast (Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023b). Thus, the greenhouse 14C data might be an indicator for the sea spray impact at coastal sites, as well.
Based on our preliminary results, we expect a reservoir age of about 250–290 years (see G-5, “Schlei”) for radiocarbon dating of plant samples from (or nearby) the archaeological site of Haithabu. In the case of archaeological material found in Haithabu (cal AD 804–1066) and its successor town Schleswig (Rathausmarkt site cal AD 1070–1210, St. Clements graveyard cal AD 1250–1350), located at opposing sides of the Schlei inlet close to the Baltic Sea (see, e.g., Grupe et al. Reference Grupe, von Carnap-Bornheim and Becker2013; Hilberg Reference Hilberg and Brink2008; Jahnke Reference Jahnke2006; Jankuhn Reference Jankuhn1986; Müller Reference Müller2016; Schlesinger Reference Schlesinger1972), a sea spray-induced shift by about 200 or 300 years can actually be problematic.
For illustration, let us have a look at the shipwreck “Wrack 4” found in the harbor of Haithabu. The wood was 14C-dated to 1024 ± 25 BP, calibrated to 987–1017 AD. Accordingly, this ship would be allocated to the Viking Age period. In contrast, dendrochronology determined that the investigated tree was logged around or after 1184 and, thus, during medieval times (Kalmring Reference Kalmring2010; Nakoinz Reference Nakoinz2005; von Carnap-Bornheim et al. Reference von Carnap-Bornheim, Hartz, Kühn and Nakoinz2002; von Carnap-Bornheim et al. Reference von Carnap-Bornheim, Kühn and Nakoinz2003). “Wrack 4” would be associated to Schleswig based on dendrochronology, while radiocarbon analysis points to an association with Haithabu. The difference between calibrated 14C dating and dendrochronology is 167 to 197 years. The 14C-dated ship planks (Nakoinz Reference Nakoinz2005) were made of oak wood (Crumlin-Pedersen Reference Crumlin-Pedersen1969). Oaks grew locally in the region (see Behre Reference Behre1983). Thus, the oakwood likely originated from the close vicinity of Haithabu/Schleswig. Accordingly, we suppose that the difference between radiocarbon and dendrochronological age could have been caused by the sea spray effect. For comparison, the artificial sea spray with Schlei water (G-5, expected Haithabu signal) resulted in a divergence of 269 ± 22 years (247–291 years). This shift, based on greenhouse data, however, clearly has to be evaluated based on modern local samples.
5. Conclusion
European beach grass (Ammophila arenaria, L.), grown in planters in a greenhouse between June and December 2020, have been irrigated with tap water and sprayed with mineral salt solution, with low NaCl concentration but partly high concentration in, e.g., HCO3 –, as well as natural salty, i.e. NaCl enriched, water. By analyzing the carbon isotope inventory of irrigation and spray water as well as treated plants this study aimed to investigate possible reservoir age effects in grown plant tissues since all waters used were depleted in radiocarbon concentration in comparison to contemporary atmospheric CO2.
Based on the presented preliminary results we observed a potential sea spray impact on the radiocarbon composition of terrestrial plants treated with an artificial sea spray, which is resulting from the uptake of 14C-depleted DIC as well as from physiological reactions in the plants and accompanied discrimination effects. We emphasize, however, that further analyses are required in order to verify a potential sea spray-induced reservoir effect as indicated by our study.
Measured carbon isotope composition in the plant tissue indicates two distinctively different effects. Group 1 plants grown under the influence of spray water enriched in minerals, including DIC, are depleted in 14C. Overall, the discrimination against atmospheric 14C relative to the discrimination against atmospheric 13C indicates a reduction in the stomatal conductance, probably caused by increasing DIC as well as Na+/Cl– contents in the spray water. In difference from the group 1 experiments, group 2 plants were sprayed with naturally salty (NaCl) water with lower DIC content compared to the group 1 experiment. While discrimination against 13C was of comparable magnitude as seen for G-1 (“marine-like”), the discrimination against 14C increased markedly. Salinity stress also induces a reduced stomatal conductance, similar to the effect seen in group 1, but so intense that apparently DIC in the irrigation water as well as admixed spray water delivered the necessary carbon for plant growth, causing an apparent aging in plant tissue.
As a consequence, we want to emphasize that plants or terrestrial herbivores from coastal sites should not be used as a control for a 100% terrestrial 14C signal without checking stable or radiogenic isotopic systems besides δ13Ccollagen and δ15Ncollagen (e.g., δ13Ccarbonate, δ18Ocarbonate, δ18Ophosphate, δ34Scollagen, 87Sr/86Sr). In addition, we want to point out that the sea spray signal in stable isotopic systems can be detected in samples located several kilometers, potentially even about several hundred kilometers distant from the shoreline (e.g., Alonzi et al. Reference Alonzi, Pacheco-Forés, Gordon, Kuijt and Knudson2020; Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023b; Kochergina et al. Reference Kochergina, Novak, Erban and Stepanova2021; Nehlich Reference Nehlich2015; Snoeck et al. Reference Snoeck, Ryan, Pouncett, Pellegrini, Claeys, Wainwright, Mattielli, Lee-Thorp and Schulting2020). Regional and seasonal differences could also have a distinct effect on the local sea spray signal (Göhring et al. Reference Göhring, Hölzl, Mayr and Strauss2023b).
Based on our presented preliminary results it is important to consider the sea spray effect when performing radiocarbon analyses on terrestrial individuals (plants, animals, humans) from coastal sites. They might be prone to a sea spray reservoir effect. Further (greenhouse) experiments for the quantification of the indirect as well as the direct sea spray effect in plants and animals are required in order to further reveal the reservoir effect on terrestrial individuals, including humans, caused by sea spray and associated physiological or metabolic reactions as well as to correct for the sea spray impact on radiocarbon analyses.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/RDC.2025.1
Acknowledgments
This research project was supported by LMUexcellent as part of LMU Munich’s funding as University of Excellence within the framework of the German Excellence Strategy.
We thank the team of the greenhouse of the Biocenter of the LMU Munich, i.e., Dr. Anja Schneider and Mr. Ariel Wenzel, for the permission to carry out the study in one of the climate chambers.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.











