Introduction
In nursing homes, infections are a major cause of resident morbidity and mortality. 1 Over half (57%) of nursing home residents are colonized with a multidrug-resistant organism (MDRO), including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), and extended-spectrum β-lactamase (ESBL)-producing organisms. Reference Mody, Foxman and Bradley2,Reference Rodríguez-Villodres, Martín-Gandul and Peñalva3 MDROs can spread when healthcare workers (HCWs) acquire contamination on their hands or clothing through physical contact with a resident who is colonized or infected and subsequently transmit it to other residents. Reference Cochard, Aubier, Quentin, Mee-Marquet and Centre4,Reference Lydecker, Osei and Pineles5 Many activities of daily living (ADLs) involve close, prolonged contact between HCWs, residents, and their immediate surroundings. Consequently, these particular activities pose a high risk of HCWs’ hands or clothing becoming contaminated with or transmitting MDROs, such as MRSA or ESBL-producing organisms. Reference Roghmann, Johnson and Sorkin6–Reference Blanco, Pineles and Lydecker9
Prior research has identified common challenges that contribute to the risk of transmission during high-contact activities, such as bathing or changing resident briefs. Reference Lydecker, Osei and Pineles5,Reference Mumma, Gannon, Regina, Luciano, Kellogg and Jacob10 These challenges arise from multilevel factors, including resident-related factors (e.g., immobility, incontinence, aggression) and organizational constraints (e.g., inadequate staffing, limited or poor-quality supplies). Such challenges can disrupt HCWs’ workflow or undermine their adherence to infection prevention and control (IPC) practices such as hand hygiene, use of personal protective equipment (PPE), and environmental surface cleaning or disinfection. However, understanding how these challenging workflows influence transmission during resident care remains limited, yet it is important for identifying barriers to existing IPC practices or where new interventions may be needed.
This study aimed to characterize certified nursing assistant (CNA) workflows and IPC practices during two high-contact resident care activities and examine how these behaviors may contribute to MDRO spread within nursing homes. CNAs were the focus of the present study because they provide the majority of direct resident care, including high-contact activities such as bathing and brief changes. Reference Nelson Chang, Leecaster and Fridkin11 To this end, we conducted high-fidelity simulations of resident care with CNAs using a bacteriophage as a surrogate marker for MDRO transmission, combined with behavioral analyses of simulation recordings.
Methods
Participants
Between August and November 2023, we conducted 10 standardized resident care simulations with CNAs who worked in long-term care or mixed-care units (e.g., residents receiving long-term or skilled nursing) in a nursing home. Participants had a median of 21 years (IQR = 17.2–28.3) of experience as a CNA and worked on either a long-term (50%) or mixed-care (50%) unit. Participants were compensated with a $25 gift card and a parking voucher for their time. All research activities were approved by Emory University’s Institutional Review Board (STUDY00004038).
High-fidelity simulation scenario
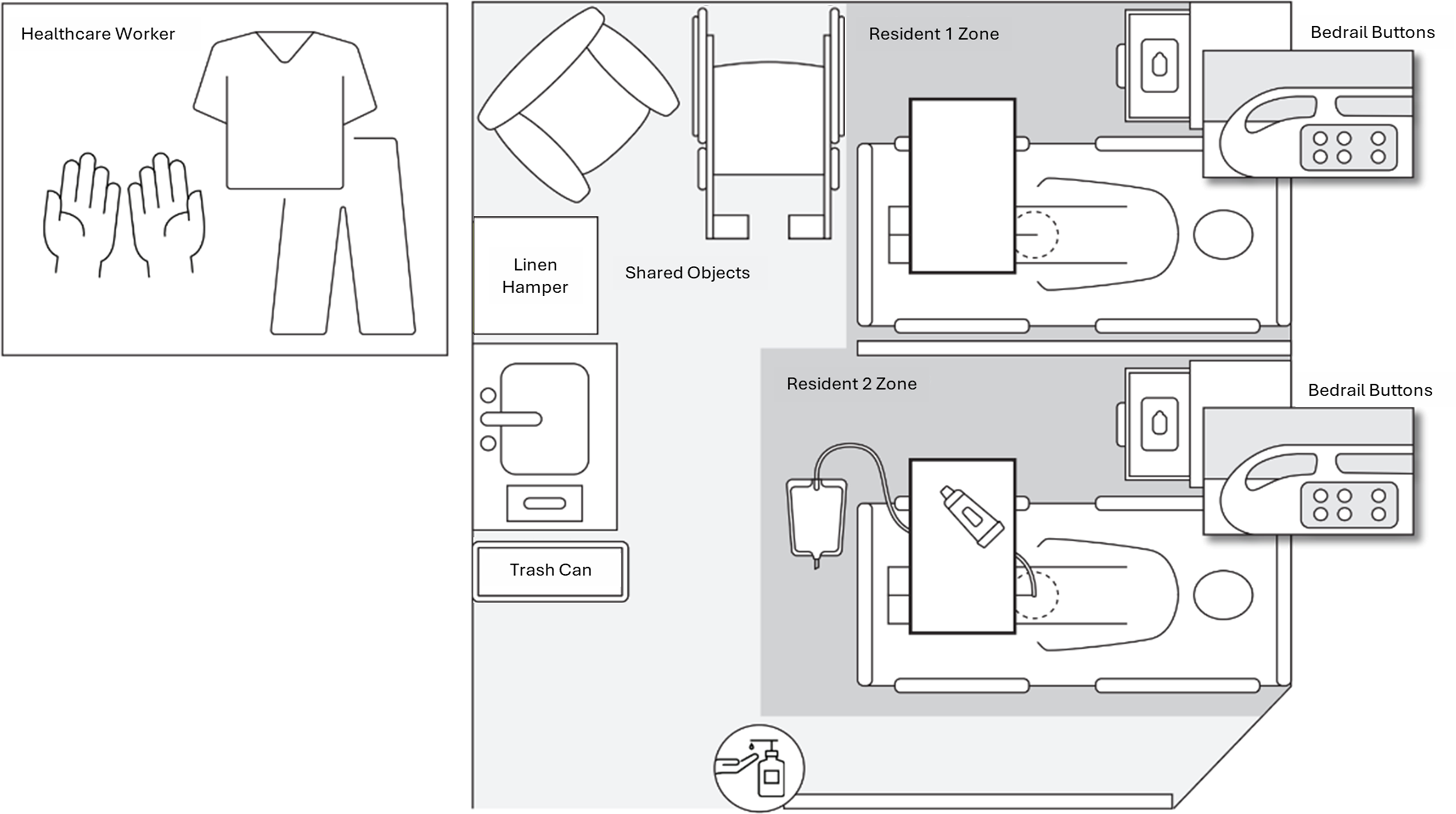
We developed a high-fidelity simulation scenario of high-contact resident care activities involving two adult male nursing home residents (Figure 1). The residents, represented by mannequins, shared a single room with a curtain dividing their immediate surroundings (“zones”), a common configuration in nursing homes. Resident 1 was incontinent of urine, had simulated skin breakdown in the groin area, and needed his brief changed and skin barrier cream applied to the skin breakdown. Resident 2 was incontinent of stool, had an indwelling urinary catheter, and needed a complete bed bath and linen change. Options for hand hygiene included alcohol-based hand rub dispensers located at the entryway of the resident’s room or a sink with hand soap in the middle of the shared room. Gloves were available on the sink in the resident’s room, while isolation gowns and containers of disinfecting wipes, which are typically stored outside of resident rooms, Reference Jacobs Slifka, Kabbani and Stone12 were available in a separate supply room. A trash can and a linen hamper were located next to the sink.
Layout of a simulated double occupancy room in a nursing home.

Based on prior qualitative research with CNAs, the simulation scenario also included common challenges to performing care activities that may contribute to transmission by promoting lapses in IPC practices or increasing the amount of physical contact between the CNA, residents, and their environment. Reference Mumma, Gannon, Regina, Luciano, Kellogg and Jacob10 These challenges in the simulation included limited availability of resident care supplies (e.g., skin barrier cream), environmental clutter (e.g., furniture, a wheelchair), having residents with fecal or urinary incontinence, and residents who require total care (i.e., are completely dependent on staff for ADLs).
Procedure
Before each simulation, four genetic variants of λ phage were applied to predetermined sites on residents’ bodies and environmental surfaces in the simulation space. Resident 1 had λ Temp applied to simulated skin breakdown in the groin area and the incontinence brief and λ Kan on the front bedrails and bedside table. Resident 2 had λ Vir in simulated stool that was in their brief, on their indwelling urinary catheter, on their right back bedrail, and on one side of the bedside table. λ Chl was applied to Resident 2’s front bedrails and the side of the bedside table that was not contaminated by λ Vir. Each λ variant was applied with a spray bottle containing 10 mL of a 108 plaque-forming units (PFU)/mL solution. Reference Burke, Berryhill and Garcia13
Upon arrival, participants were consented and completed a demographic questionnaire. Research staff then oriented the participant to the simulation space, supplies, and resident care activities. Participants then donned a pair of disposable scrubs and a head-mounted camera (GoPro®). The simulation began after the research staff gave a scripted handover for each resident to the participant, who could choose which task and resident to begin with. Participants were told that contamination was present in the simulation but were not aware of its locations.
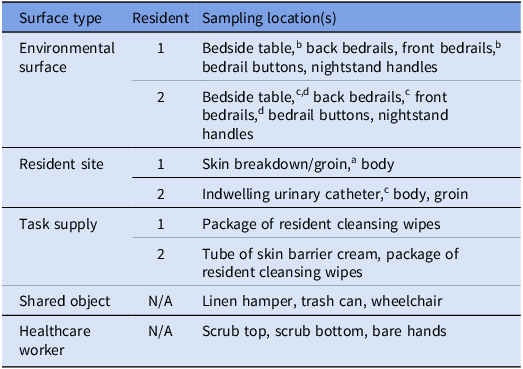
After each simulation, 24 predetermined sites were sampled for the presence of each λ variant (Table 1). These surfaces comprised environmental surfaces (n = 10 sampling sites), resident sites (n = 5), task supplies (n = 3), shared objects (n = 3), and surfaces on participants (n = 3). After sampling, surfaces were disinfected using 70% ethanol, DNA Away™, RNase Away™, and Lysol® wipes. The identity of λ variant(s) from positive surface samples was determined through direct polymerase chain reaction testing. A “transmission event” occurred when a λ variant was detected on a sampling surface on which that variant was not applied prior to the simulation (Table 1). Detailed methods for contamination, surface sampling, and decontamination procedures are described and validated elsewhere. Reference Burke, Berryhill and Garcia13
Sampling surfaces and sources of contamination in each simulation

N/A, not applicable.
a-dSampling locations inoculated with λTemp (a), λKan (b), λVir (c), or λChl (d) prior to the start of each simulation.
Behavioral coding
We developed a behavioral coding scheme to capture sequences of hand-to-surface contacts and performance of IPC practices in video recordings of simulations (see Supplemental Material). Contact with a surface was coded whenever a participant grabbed or held a new surface with either hand but was not coded when repeatedly touching the same surface or when transferring the same object between hands. If a participant grabbed or held multiple new surfaces simultaneously, contact with all surfaces was coded. For hand hygiene, we recorded whether participants used alcohol-based hand rub or soap and water. For glove use, we coded when both gloves were fully donned or doffed. For surface cleaning and disinfection, we coded each time an object or surface was wiped and whether participants used a resident cleansing wipe, a washcloth or towel, or a disinfecting wipe.
After developing the initial behavioral coding scheme, three researchers independently coded one pilot simulation video. The researchers then discussed disagreements, revised the coding scheme, and independently coded a video of a different pilot simulation. Interrater reliability was acceptable (Cohen’s κ = 0.68; 95% CI: 0.64–0.71), and each researcher subsequently coded 3–4 simulation videos. Behavioral coding was performed using The Observer XT software. Reference Zimmerman, Bolhuis, Willemsen, Meyer and Noldus14
Workflow analysis
To characterize CNA workflows in the simulations, we conducted a two-phase analysis of the behavioral coding data. In the first phase, we performed a lag sequential analysis (LSA) using The Observer XT software. Reference Zimmerman, Bolhuis, Willemsen, Meyer and Noldus14 LSA calculated the total frequency at which contact with each object (or performance of an IPC practice) immediately followed contact with every other object or IPC practice in each simulation. Before performing LSA, data were preprocessed by consolidating interactions with objects that had similar potential for hand-to-surface transmission. For example, both grabbing a bedside table and cleaning it with a resident cleansing wipe were counted as contact with the bedside table. In contrast, the use of disinfecting wipes on a surface was not counted as contact, given their ability to eliminate pathogens, including bacteriophage. Reference Burke, Berryhill and Garcia13
In the second phase of analysis, we examined the structure of CNA workflows using the Walktrap community detection algorithm Reference Pons and Latapy15 in the igraph package in R.16 The Walktrap algorithm identifies densely connected communities of nodes (i.e., objects or IPC practices) by simulating short, random walks on a network where the probability of transitioning between two nodes is proportional to the weight of the edge connecting them. Reference Pons and Latapy15 In terms of workflows, communities are groups of contiguous behaviors, which may correspond to distinct tasks or subtasks. To apply the Walktrap algorithm, we first converted the output of the LSA into an undirected, weighted graph by retaining edges between objects and IPC practices only if transitions occurred in both directions at least once. The weight of each edge was then calculated as the sum of transitions in both directions.
Results
Workflow analysis
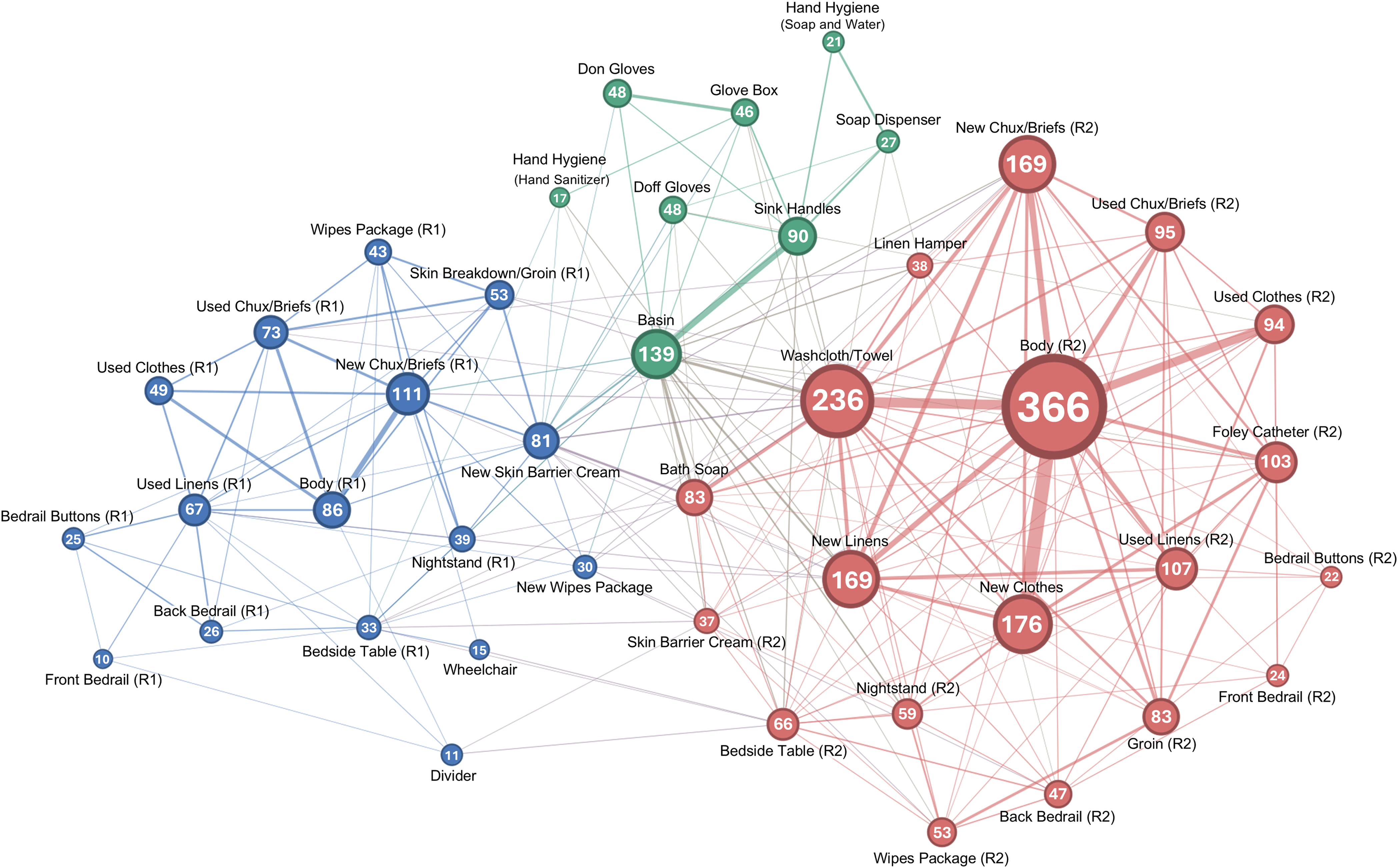
Simulations lasted a median of 38.5 minutes (IQR: 32.5–46). Across the 10 simulations, there were 3,083 hand-to-surface contacts (median = 282.5 contacts per simulation, IQR: 228.75–378). Most contacts (78%, 2,398/3,083) occurred within resident zones (i.e., a resident and their immediate surroundings; Figure 1), with contact within Resident 2’s zone alone accounting for 58% (1,783/3,083) of all contacts. Resident 2, who required a complete bed bath and linen change, was also the single most frequently contacted object (Median = 33.5 contacts per simulation, IQR: 24.25–47.5).
Most CNAs (80%, 8/10) began the simulation with Resident 1, who required a brief change and application of skin barrier cream to skin breakdown on his groin. CNAs’ workflows were otherwise highly modular, forming three distinct communities: one community encompassed all objects in Resident 1’s zone (blue; Figure 2), another all objects in Resident 2’s zone (red), and a third (green) comprising IPC practices (e.g., hand hygiene, glove use) and objects needed to perform them (e.g., sink, glove box). Communities also included new supplies from the supply room or shared objects that were closely associated with a resident and their activities, such as the linen hamperin Resident 2’s community. The separation of IPC practices indicates that these activities were performed outside of, rather than during episodes of resident care, while the separation between resident communities reflects how CNAs tended to complete all care for one resident before beginning care for another. Within resident communities, CNAs frequently transitioned between touching the resident, their immediate surroundings, and task supplies.
Weighted, undirected graph of transition frequencies between objects or IPC practices (nodes). Node values and edge thickness represent the total frequency of transitions to and bidirectional transitions between nodes, respectively. The trashcan was excluded due to no bidirectional transitions. Abbreviations: R1, resident 1; R2, resident 2.

Transmission events
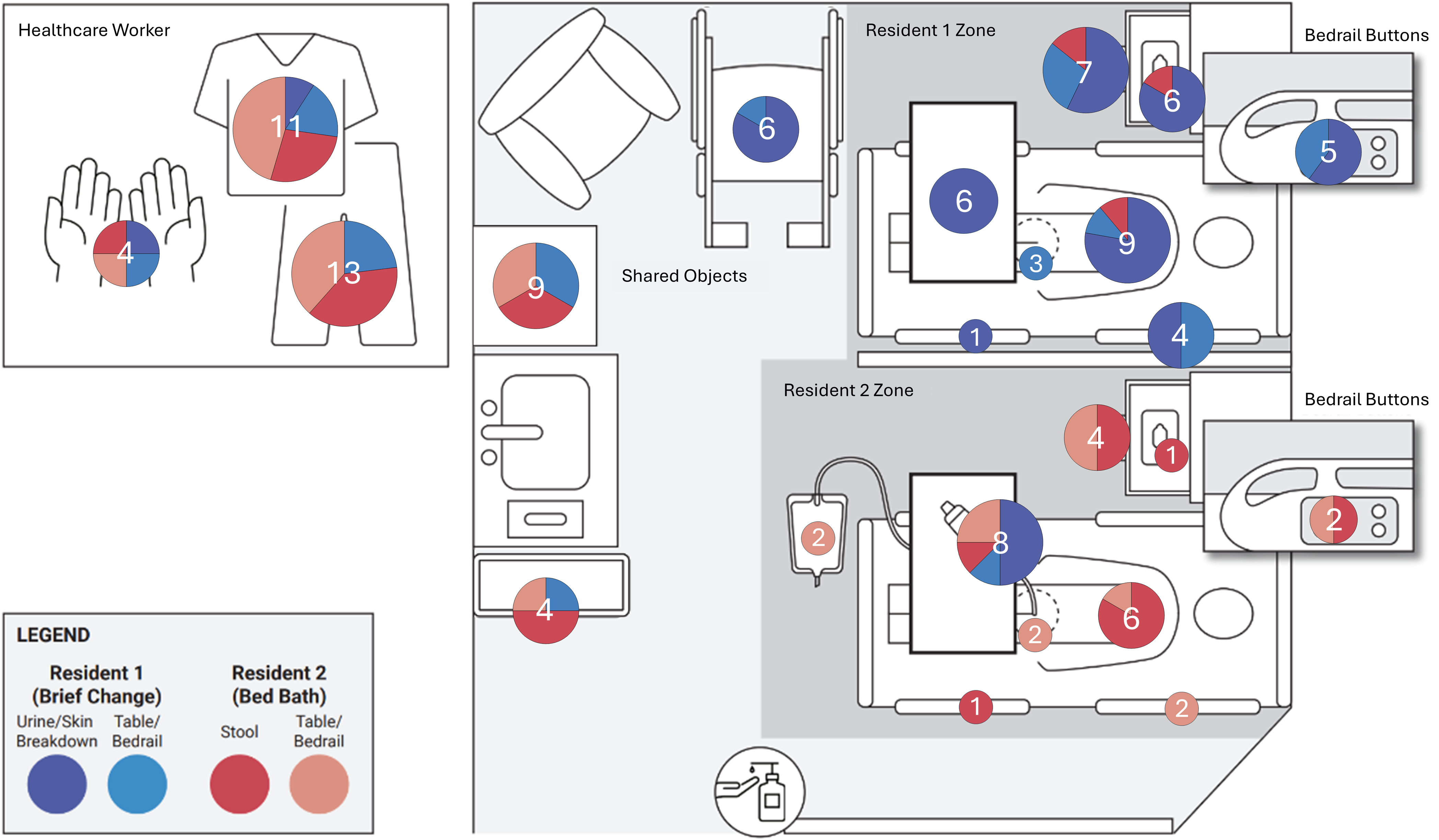
Across the 10 simulations, there were 116 transmission events (median = 11.5 events per simulation, IQR: 7.25–15.75). Most events (60%, 69/116) occurred within resident zones (Figure 3) and overwhelmingly involved transmission of a λ variant originating from that zone (88%, 61/69). Transmission between resident zones was less common (12%, 8/69), occurring primarily in Resident 2’s zone and involving transmission from Resident 1’s skin breakdown to Resident 2’s skin barrier cream, which was a limited supply in the simulation. Reference Mumma, Gannon, Regina, Luciano, Kellogg and Jacob10
Pie charts show the total number of transmission events on a sampling surface (in white) and the proportion of transmission events on sampling surfaces from each λ variant.

Surfaces on CNAs, such as their scrubs and bare hands, accounted for 24% (28/116) of transmission events and had the highest contamination rate of all types of sampling surfaces (median = 2 events per simulation, IQR: 2–3.75). Most transmission to CNAs was to their scrubs (86%, 24/28), which originated mostly from Resident 2 (71%, 20/28).
Lastly, transmission to shared objects, such as the linen hamper and trash can, accounted for 16% (19/116) of events. Transmission to these objects originated from Residents 1 and 2 to similar degrees (53% [10/19] versus 47% [9/19], respectively) except for the wheelchair, which only had contamination from Resident 1.
Infection prevention and control practices
CNAs performed hand hygiene a median of 3.5 times per simulation (IQR: 2–5.75), using soap and water and alcohol-based hand rub in 55% (21/38) and 45% (17/38) of instances, respectively. Hand hygiene was most often performed at the start of the simulation (29%, 11/38), after completing care for one resident but before beginning care for the next (29%, 11/38), after completing care for both residents (26%, 10/38), and least often, during resident care itself (16%, 6/38).
Regarding PPE use, CNAs donned and doffed gloves a median of 5 times per simulation (IQR: 3–6), performing hand hygiene either before donning or after doffing gloves in 42% (20/48) of instances, both before donning and after doffing (8%, 4/48), and never (50%, 24/48). No participant wore an isolation gown despite them being available in the supply room.
Lastly, CNAs attempted to clean or disinfect environmental surfaces a median of 4 times per simulation (IQR: 2.25–5), using non-disinfecting wipes in 82% (39/48) of instances, which included resident cleansing wipes or “improvised” wipes made from a paper towel and hand sanitizer, washcloths in 10% (5/48) of instances, and disinfecting wipes in 8% (4/48) of instances.
Discussion
This study used high-fidelity simulations and surrogate markers to examine how contamination spreads during high-contact resident care activities in nursing homes. Contamination of HCWs, residents, their immediate environments, and shared objects was common. Behavioral analyses indicated that transmission is shaped not only by adherence to IPC practices but also by the organization and flow of resident care activities.
Most transmission events involved the movement of phage within a resident’s zone, which reflected how CNAs frequently transitioned between touching a resident, their immediate surroundings, and care supplies during care activities. IPC practices, such as changing gloves, hand hygiene, and surface cleaning or disinfection occurred infrequently, leaving few barriers to interrupt transmission within resident zones. When surface cleaning or disinfection did occur, it was rarely performed with a disinfecting product; in nursing homes, cleaning products are stored outside resident rooms to prevent misuse by cognitively impaired residents, a practice that also limits staff access and use during care. Reference Jacobs Slifka, Kabbani and Stone12 By increasing environmental bioburden, transmission within resident zones creates opportunities for re-contaminating a resident (e.g., colonizing a new body site) or transmission outside a resident’s zone.17–19
Transmission outside of resident zones primarily involved surfaces that could facilitate spread between residents. Shared objects, such as trashcans, linen hampers, and wheelchairs, can indirectly transmit contamination through repeated handling by HCWs and use across multiple residents. Wheelchairs, in particular, can be an overlooked vector for transmission due to their frequent use and inconsistent disinfection between residents.20,21 In addition to shared objects, CNAs’ scrubs were frequently contaminated, especially during bed bathing of Resident 2. This finding aligns with prior evidence identifying HCW clothing as a vector for MDRO transmission.22–24 Behavioral analyses suggested two contributing factors: a high amount of physical contact during bed bathing and the absence of gown use, despite their availability. These findings reinforce the importance of targeted gown and glove use during high-contact care activities, as recommended in the Centers for Disease Control and Prevention’s concept of Enhanced Barrier Precautions (EBP).5,25 Additionally, other strategies to reduce contact during bed bathing should be considered. For example, alternatives to bathing, such as using no-rinse bath wipes, may decrease the intensity and duration of physical contact during bed bathing.26
Transmission between resident zones was rare and appeared to be related to the larger structure of workflows. CNAs typically completed all care activities for one resident before moving to the next, which limited opportunities for direct transmission between residents. When transmission did occur, it was mainly from Resident 1 to Resident 2 and was associated with having limited resident care supplies. Reference Mumma, Gannon, Regina, Luciano, Kellogg and Jacob10 Most CNAs began the simulation with Resident 1, who needed a brief change and skin barrier cream applied to the skin breakdown on his groin. Although cream was stocked in the supply room, it was only immediately available on resident 2’s bedside table. Retrieving the supply during Resident 1’s care may have disrupted the CNAs’ workflow and contributed to lapses in IPC practices at this critical moment. Reference Mumma, Gannon, Regina, Luciano, Kellogg and Jacob10
This study has several limitations. First, the small number of participants could not capture the full variability of workflows and adherence to IPC practices in nursing homes. Second, we assessed positivity rather than density of recovered phage from surfaces within a resident’s room as our focus was on identifying potential transmission pathways rather than quantifying the risk associated with these pathways. Third, participants were aware that they were being recorded during their simulations, which may have influenced their behavior. Lastly, we conducted the study before the 2024 Center for Medicare and Medicaid Services mandate for EBP in nursing homes, and outcomes, such as frequent scrub contamination, may have differed had EBP been followed in the simulations.
Overall, our findings suggest a close relationship between HCW workflows and transmission during high-contact resident care activities. Targeted interventions include providing adequate and accessible supplies, Reference Mumma, Gannon, Regina, Luciano, Kellogg and Jacob10 altering tasks to reduce HCW contamination (e.g., alternative approaches to bed bathing), improving hand hygiene and PPE use at the bedside (e.g., EBP), and environmental surface disinfection of shared objects and high-touch surfaces. High-fidelity simulation with surrogate markers, combined with behavioral analysis, is useful for identifying transmission pathways during resident care activities, and future research should use this method to evaluate potential interventions.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/ice.2026.10431.
Acknowledgements
We thank Kara Jacobs Slifka and Kiara McNamara for feedback on the design of simulations, Patricia Comer for helping with participant recruitment, and Myrlene (Mimi) Jeremie and Brandon Berryhill for helping conduct simulations. The authors used ChatGPT (OpenAI, San Francisco, CA, USA) to assist with minor language editing for clarity, conciseness, and grammar. All intellectual content, data interpretation, and conclusions were generated and verified by the authors.
Financial support
Research reported in this publication was supported through the Emory Prevention Epicenter Program (PEACH) which is funded by a cooperative agreement with the Centers for Disease Control and Prevention (CDC) [U54CK000601]. This content is solely the responsibility of the authors and does not necessarily represent the official views of the CDC.
Competing interests
All authors report no conflicts of interest relevant to this article.